Submitted:
17 December 2025
Posted:
17 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
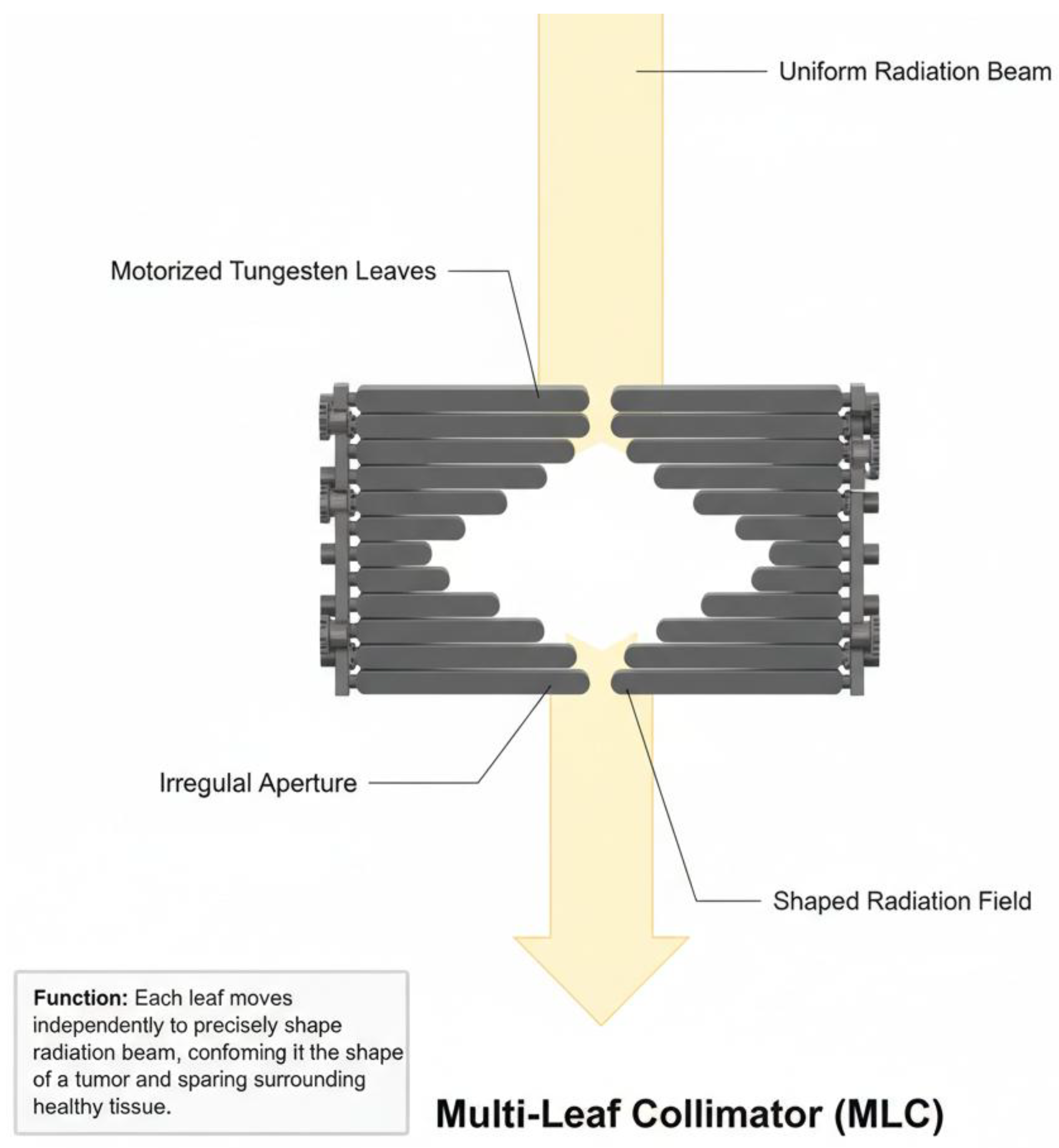
2. Materials and Methods
A. Materials
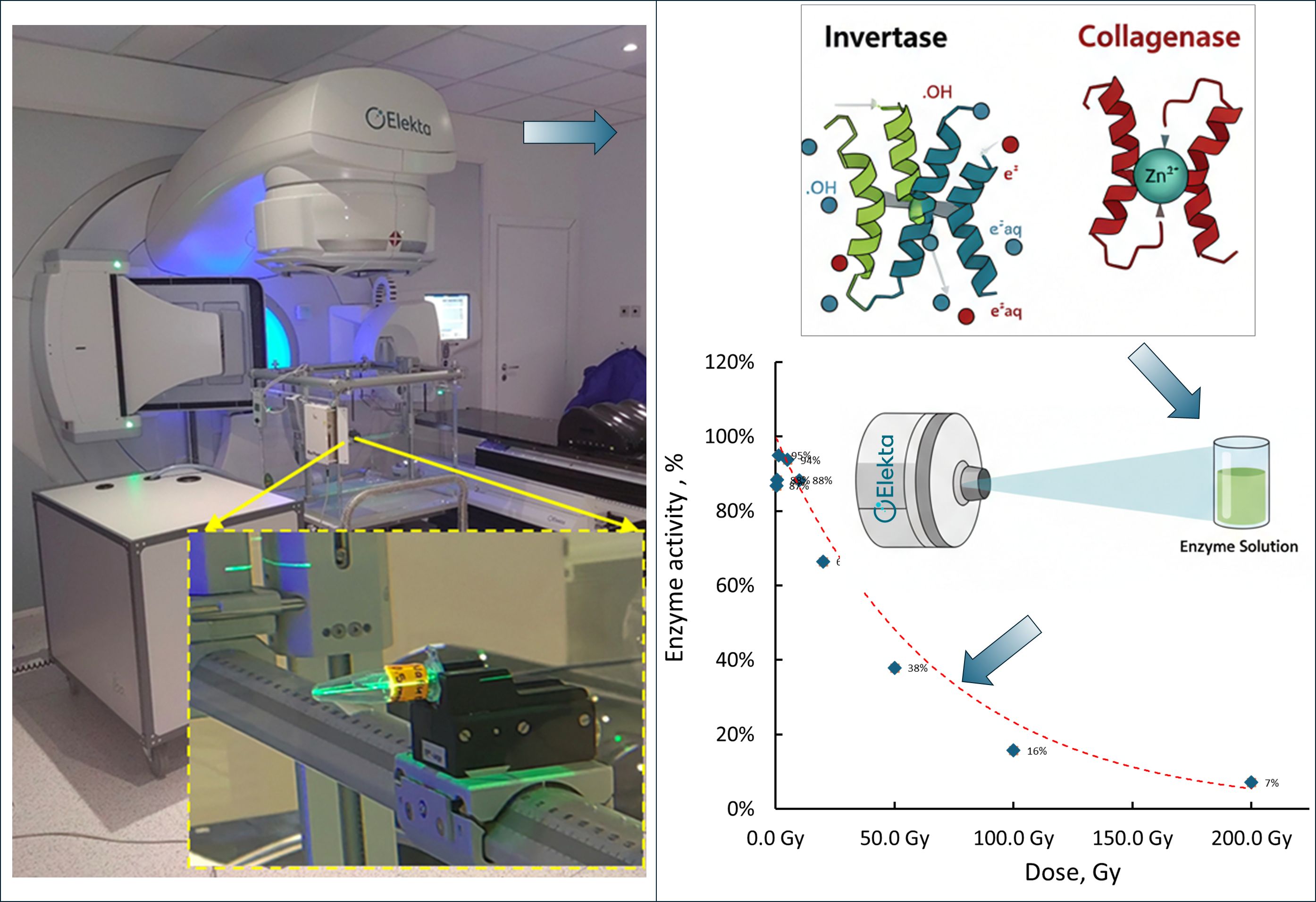
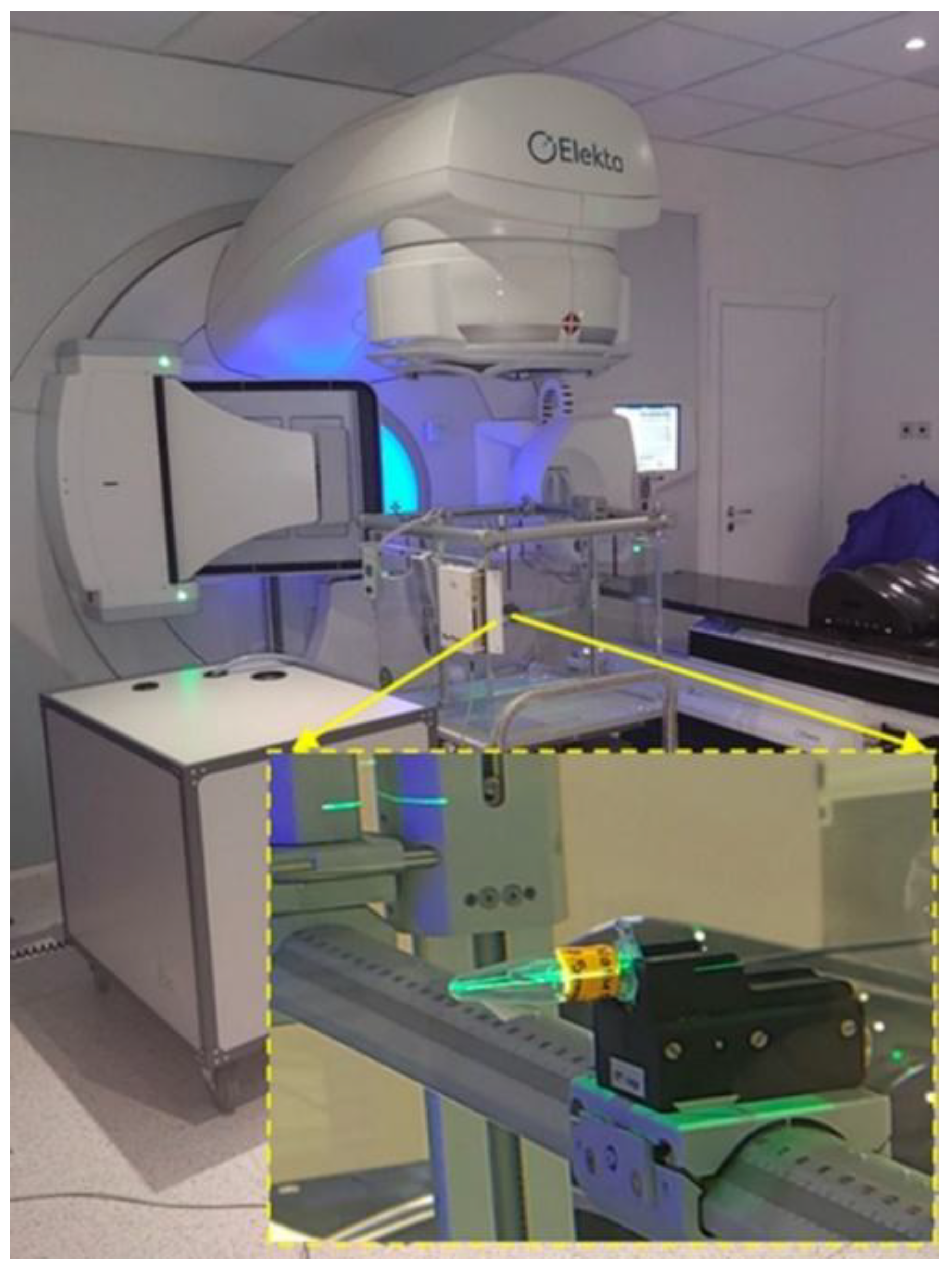
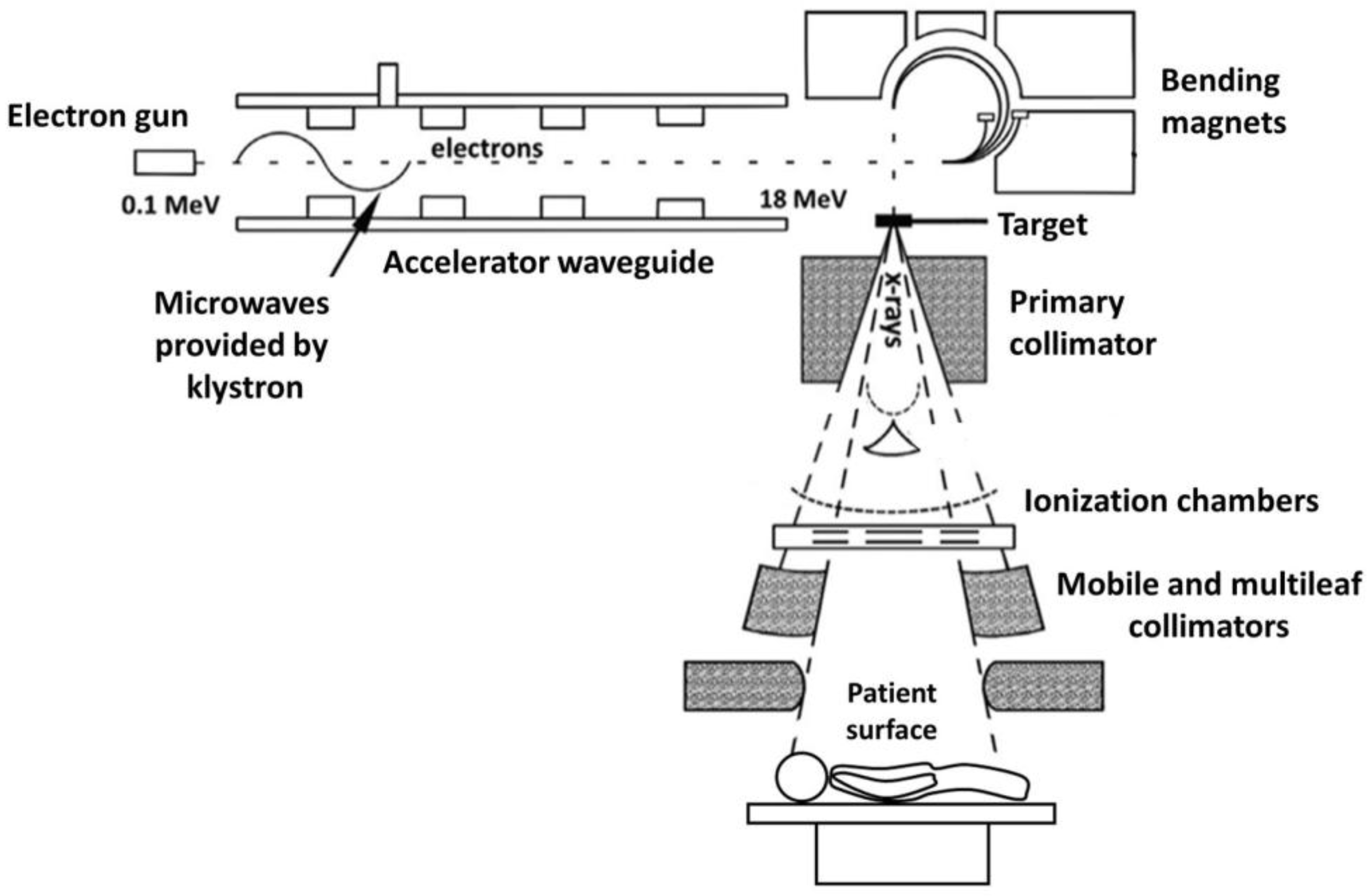
B. Irradiation at the LINAC suite
C. Enzyme Activity Assays
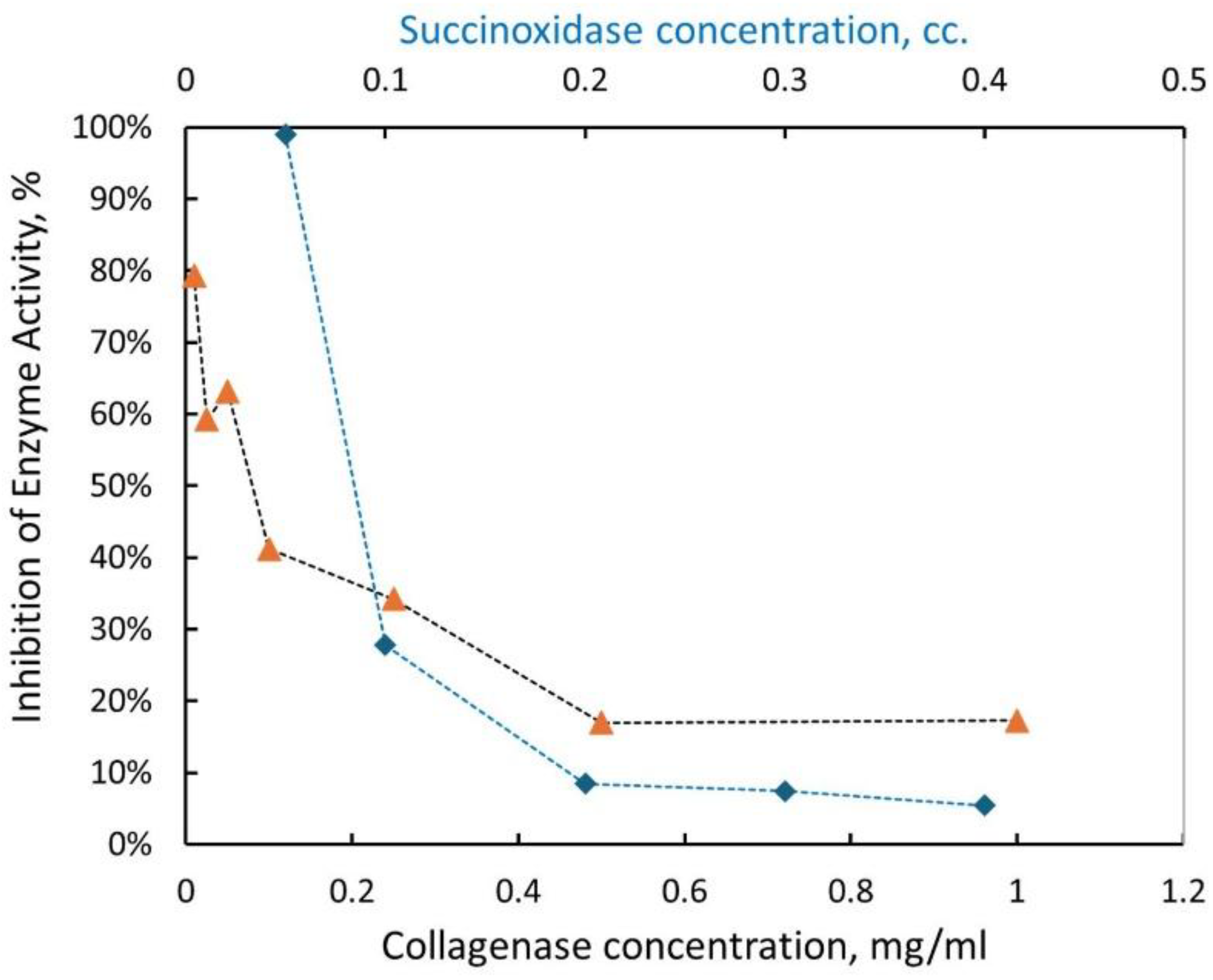
- Fixed Dose Experiment: All collagenase solutions (1.00, 0.50, 0.25, 0.10, 0.05, 0.025, and 0.01 mg/mL) were uniformly irradiated in the LINAC suite with a dose of 50 Gy to assess concentration-specific responses.
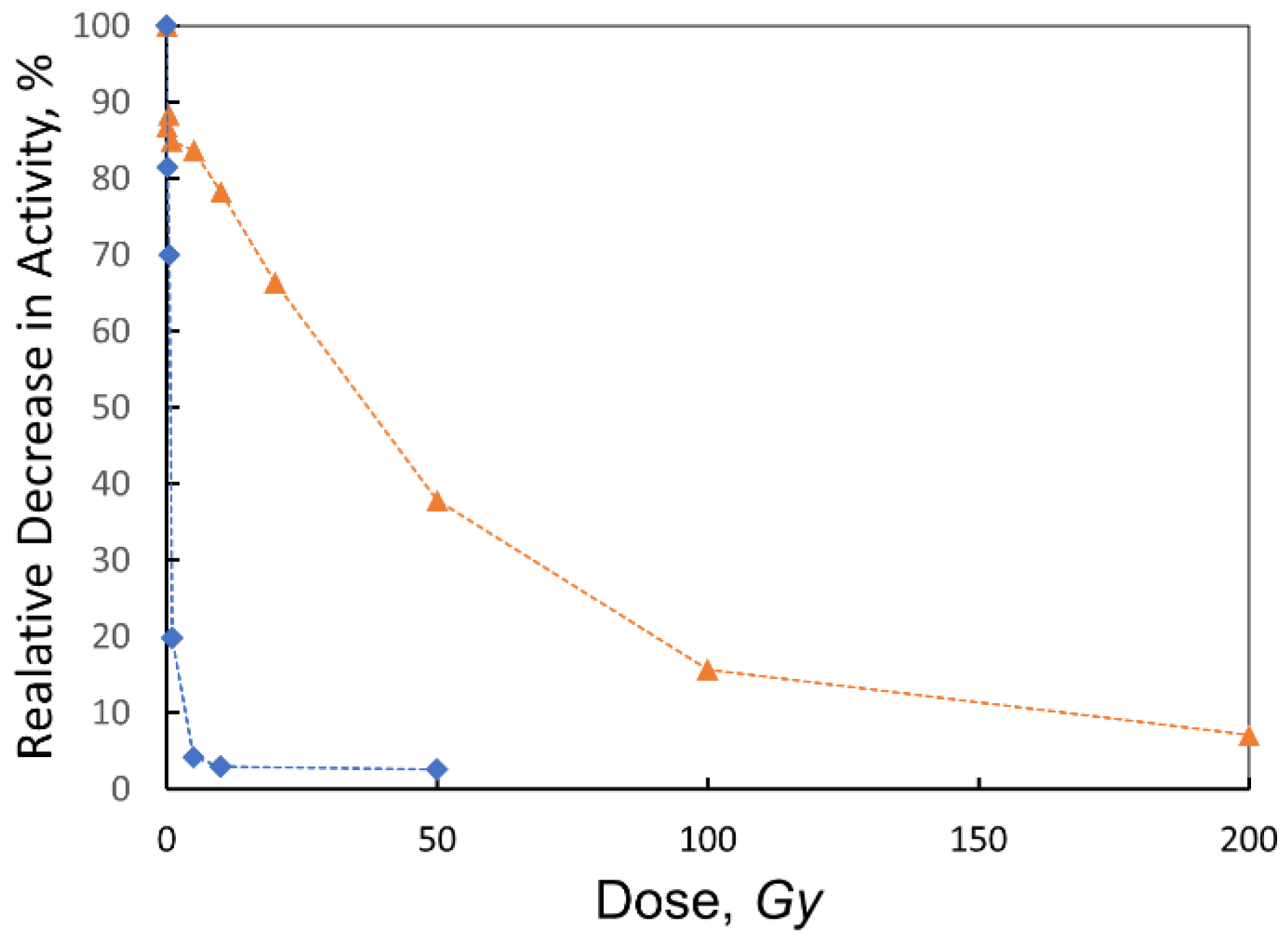
- Variable Dose Experiment: The 0.01 mg/mL solution, identified as the most responsive, was aliquoted into 1.5 mL vials and subjected to irradiation at doses of 0.1, 0.5, 1, 5, 10, 20, 50, 100, and 200 Gy to comprehensively evaluate dose-dependent activity changes.
3. Results
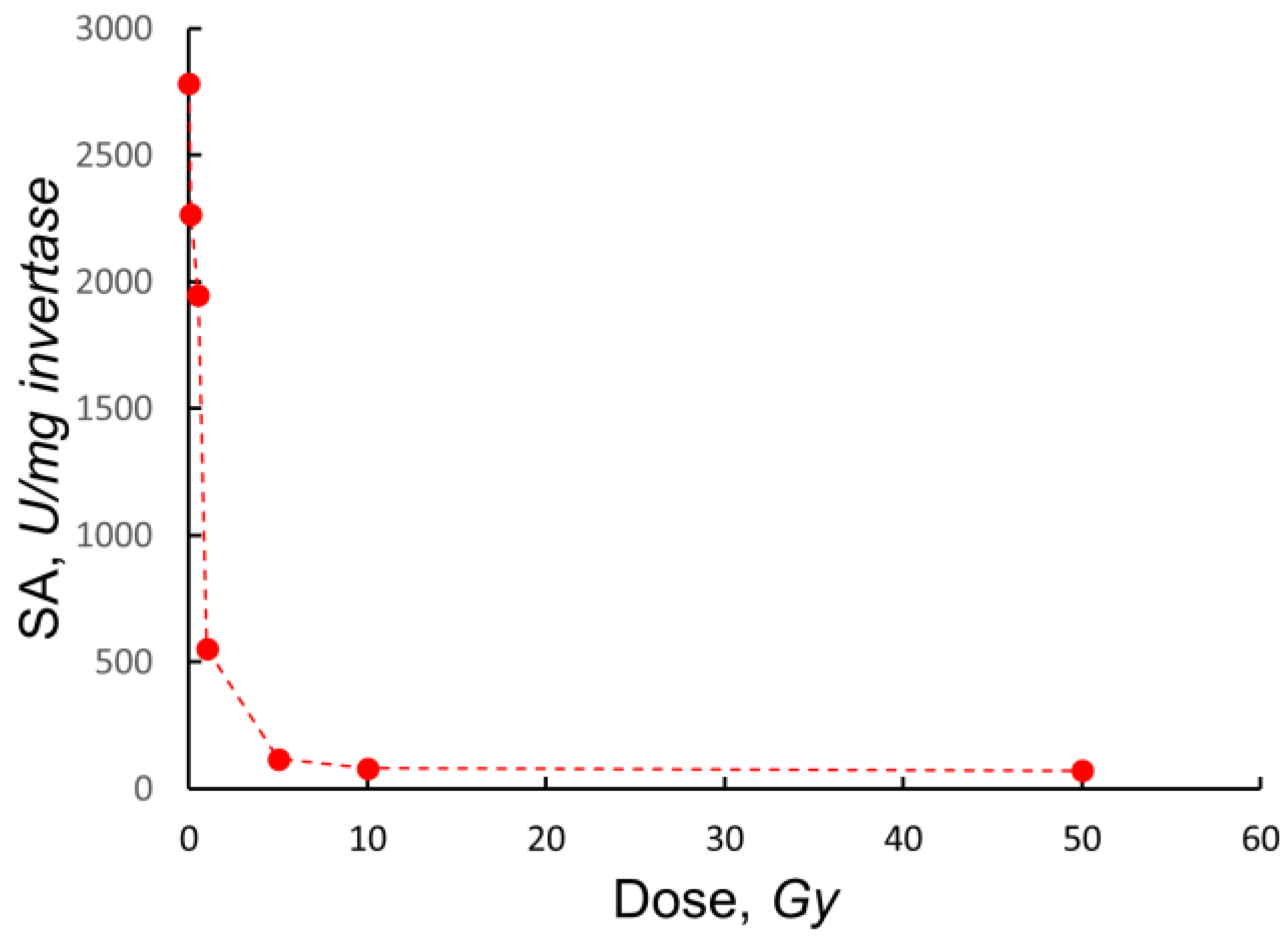
3.1. Effect of Ionizing Radiation on Invertase Activity
3.2. Collagenase and Its Treatment with Ionizing Radiation
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LINAC | Medical linear accelerator |
References
- Arena, V. Ionizing Radiation and Life: An Introduction to Radiation Biology and Biological Radiotracer Methods; Mosby: Saint Louis, 1971; ISBN 978-0-8016-0278-8. [Google Scholar]
- Belli, M.; Indovina, L. The Response of Living Organisms to Low Radiation Environment and Its Implications in Radiation Protection. Front. Public Health 2020, 8, 601711. [Google Scholar] [CrossRef]
- Chang, D.S.; Lasley, F.D.; Das, I.J.; Mendonca, M.S.; Dynlacht, J.R. Basic Radiotherapy Physics and Biology; Springer International Publishing: Cham, 2021; ISBN 978-3-030-61898-8. [Google Scholar]
- Ciernik, I.F.; Dizendorf, E.; Baumert, B.G.; Reiner, B.; Burger, C.; Davis, J.B.; Lütolf, U.M.; Steinert, H.C.; Von Schulthess, G.K. Radiation Treatment Planning with an Integrated Positron Emission and Computer Tomography (PET/CT): A Feasibility Study. International Journal of Radiation Oncology*Biology*Physics 2003, 57, 853–863. [Google Scholar] [CrossRef]
- Namiki, Y.; Fuchigami, T.; Tada, N.; Kawamura, R.; Matsunuma, S.; Kitamoto, Y.; Nakagawa, M. Nanomedicine for Cancer: Lipid-Based Nanostructures for Drug Delivery and Monitoring. Acc. Chem. Res. 2011, 44, 1080–1093. [Google Scholar] [CrossRef]
- Wain, J. Ionising Radiation Protection: A Primer. In Synthesis Lectures on Engineering, Science, and Technology; Springer Nature Switzerland: Cham, 2025; ISBN 978-3-031-65524-1. [Google Scholar]
- Leslie, S.W.; Hevly, B. Steeple Building at Stanford: Electrical Engineering, Physics, and Microwave Research. Proc. IEEE 1985, 73, 1169–1180. [Google Scholar] [CrossRef]
- Houée-Levin, C.; Sicard-Roselli, C. Radiation Chemistry of Proteins. In Studies in Physical and Theoretical Chemistry; Elsevier, 2001; Vol. 87, pp. 553–584. ISBN 978-0-444-82902-3. [Google Scholar]
- Dale, W.M. The Effect of X-Rays on Enzymes. Biochemical Journal 1940, 34, 1367–1373. [Google Scholar] [CrossRef]
- Sanner, T.; Pihl, A. Kinetics of Enzyme Inactivation in Solution by Ionizing Radiation. European Journal of Biochemistry 1967, 1, 33–35. [Google Scholar] [CrossRef]
- Kempner, E.S. Damage to Proteins Due to the Direct Action of Ionizing Radiation. Quart. Rev. Biophys. 1993, 26, 27–48. [Google Scholar] [CrossRef]
- Venkateswara Rao, M.; C. K., S.; Rawson, A.; D. V., C.; N., V. Modifying the Plant Proteins Techno-Functionalities by Novel Physical Processing Technologies: A Review. Critical Reviews in Food Science and Nutrition 2023, 63, 4070–4091. [Google Scholar] [CrossRef]
- Draganić, I.G.; Draganić, Z.D. The Radiation Chemistry of Water; Physical chemistry, a series of monographs; Academic Press: New York, 1971; ISBN 978-0-323-15878-7. [Google Scholar]
- Draganić, I.G. Radiolysis of Water: A Look at Its Origin and Occurrence in the Nature. Radiation Physics and Chemistry 2005, 72, 181–186. [Google Scholar] [CrossRef]
- Orlova, M.A. Radiation-Induced Inactivation of Proteolytic Enzymes. Russ. Chem. Rev. 1993, 62, 493–508. [Google Scholar] [CrossRef]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Ind J Clin Biochem 2015, 30, 11–26. [Google Scholar] [CrossRef]
- Gribkov, V.A.; Orlova, M.A. On Various Possibilities in Pulsed Radiation Biochemistry and Chemistry. Radiat Environ Biophys 2004, 43, 303–309. [Google Scholar] [CrossRef]
- Orlova, M.A.; Kost, O.A.; Gribkov, V.A.; Gazaryan, I.G.; Dubrovsky, A.V.; Egorov, V.A.; Troshina, N.N. Enzyme Activation and Inactivation Induced by Low Doses of Irradiation. ABAB 2000, 88, 243–256. [Google Scholar] [CrossRef]
- Saha, A. Photo-Induced Inactivation of Dihydroorotate Dehydrogenase in Dilute Aqueous Solution. International Journal of Radiation Biology 1997, 72, 55–61. [Google Scholar] [CrossRef]
- Wu, S.; Zhou, X.; Jin, Z.; Cheng, H. Collagenases and Their Inhibitors: A Review. Collagen & Leather 2023, 5, 19. [Google Scholar] [CrossRef]
- Sainz-Polo, M.A.; Ramírez-Escudero, M.; Lafraya, A.; González, B.; Marín-Navarro, J.; Polaina, J.; Sanz-Aparicio, J. Three-Dimensional Structure of Saccharomyces Invertase. Journal of Biological Chemistry 2013, 288, 9755–9766. [Google Scholar] [CrossRef]
- Drózdz, M.; Piwowarczyk, B.; Olczyk, K. Effects of ionizing radiation on the content of total collagen and its fractions and the activity of collagenolytic enzymes in rat tissues. Med Pr 1981, 32, 317–322. [Google Scholar] [PubMed]
- Nagrani, S.; Bisby, R.H. The Radiation-Induced Inactivation of External Yeast Invertase in Dilute Aqueous Solution. International Journal of Radiation Biology 1989, 55, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Marinov, P.; Petrov, I.; Stoilov, K.; Lazhovski, T.; Temnishki, P.; Petrova, S.; Balashev, K. Effect of Ionizing Radiation on the Activity of the Enzyme Invertase Studied by Polarimetry. C. R. Acad. Bulg. Sci. 2024, 77. [Google Scholar] [CrossRef]
- Ahnesjö, A.; Aspradakis, M.M. Dose Calculations for External Photon Beams in Radiotherapy. Phys. Med. Biol. 1999, 44, R99–R155. [Google Scholar] [CrossRef]
- Bortfeld, T. IMRT: A Review and Preview. Phys. Med. Biol. 2006, 51, R363–R379. [Google Scholar] [CrossRef]
- Sokač, T.; Šalić, A.; Dukarić, A.-M.; Tišma, M.; Planinić, M.; Zelić, B.; Božinović, M. Standardization of 3,5-Dinitrosalicylic Acid (DNS) Assay for Measuring Xylanase Activity: Detecting and Solving Problems. Croat. j. food sci. technol. (Online) 2023, 15, 151–162. [Google Scholar] [CrossRef]
- Chavira, R.; Burnett, T.J.; Hageman, J.H. Assaying Proteinases with Azocoll. Analytical Biochemistry 1984, 136, 446–450. [Google Scholar] [CrossRef]
- Neumann, N.P.; Lampen, J.O. Purification and Properties of Yeast Invertase*. Biochemistry 1967, 6, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Sirisatesuwon, C.; Ninchan, B.; Sriroth, K. Effects of Inhibitors on Kinetic Properties of Invertase from Saccharomyces Cerevisiae. Sugar Tech 2020, 22, 274–283. [Google Scholar] [CrossRef]
- Scott, C.M. Some Quantitative Aspects of the Biological Actions of X and γ-Rays. 1937. [Google Scholar]
- Barron, E.S.G.; Dickman, S.; Muntz, J.A.; Singer, T.P. STUDIES ON THE MECHANISM OF ACTION OF IONIZING RADIATIONS. Journal of General Physiology 1949, 32, 537–552. [Google Scholar] [CrossRef]
- Sanner, T.; Pihl, A. Kinetics of Enzyme Inactivation by Ionizing Radiation. Radiation Research 1963, 19, 12. [Google Scholar] [CrossRef]
- Patel, N.Y.; Baria, D.M.; Pardhi, D.S.; Yagnik, S.M.; Panchal, R.R.; Rajput, K.N.; Raval, V.H. Chapter 16 - Microbial Enzymes in Pharmaceutical Industry. In Biotechnology of Microbial Enzymes (Second Edition); Brahmachari, G., Ed.; Academic Press, 2023; pp. 375–403. ISBN 978-0-443-19059-9. [Google Scholar]
- Orlova, M.A. Radiation-Induced Inactivation of Proteolytic Enzymes. Russ. Chem. Rev. 1993, 62, 493–508. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



