Submitted:
05 December 2025
Posted:
08 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
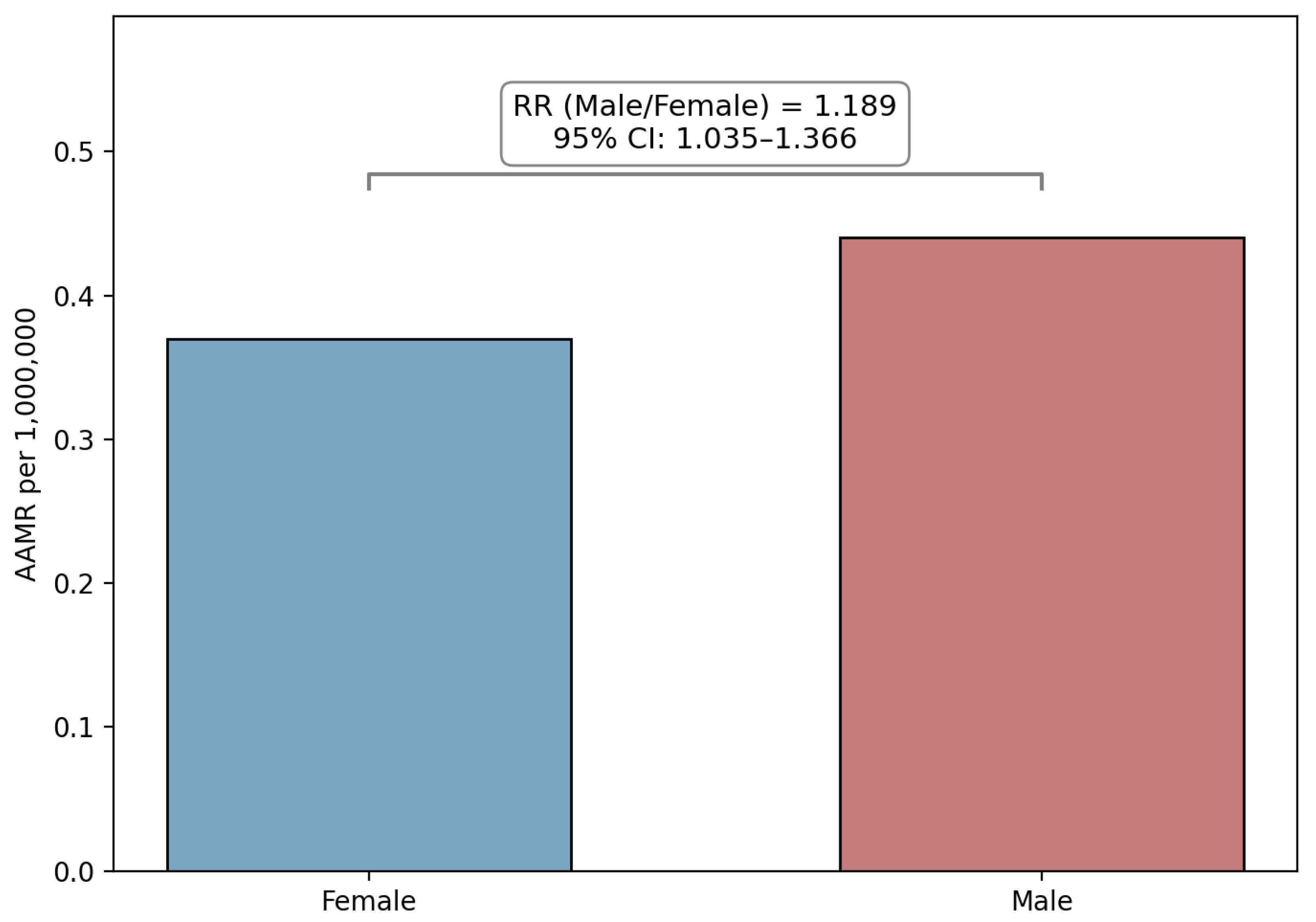
3.1. Overall Mortality and Sex-Based Disparities
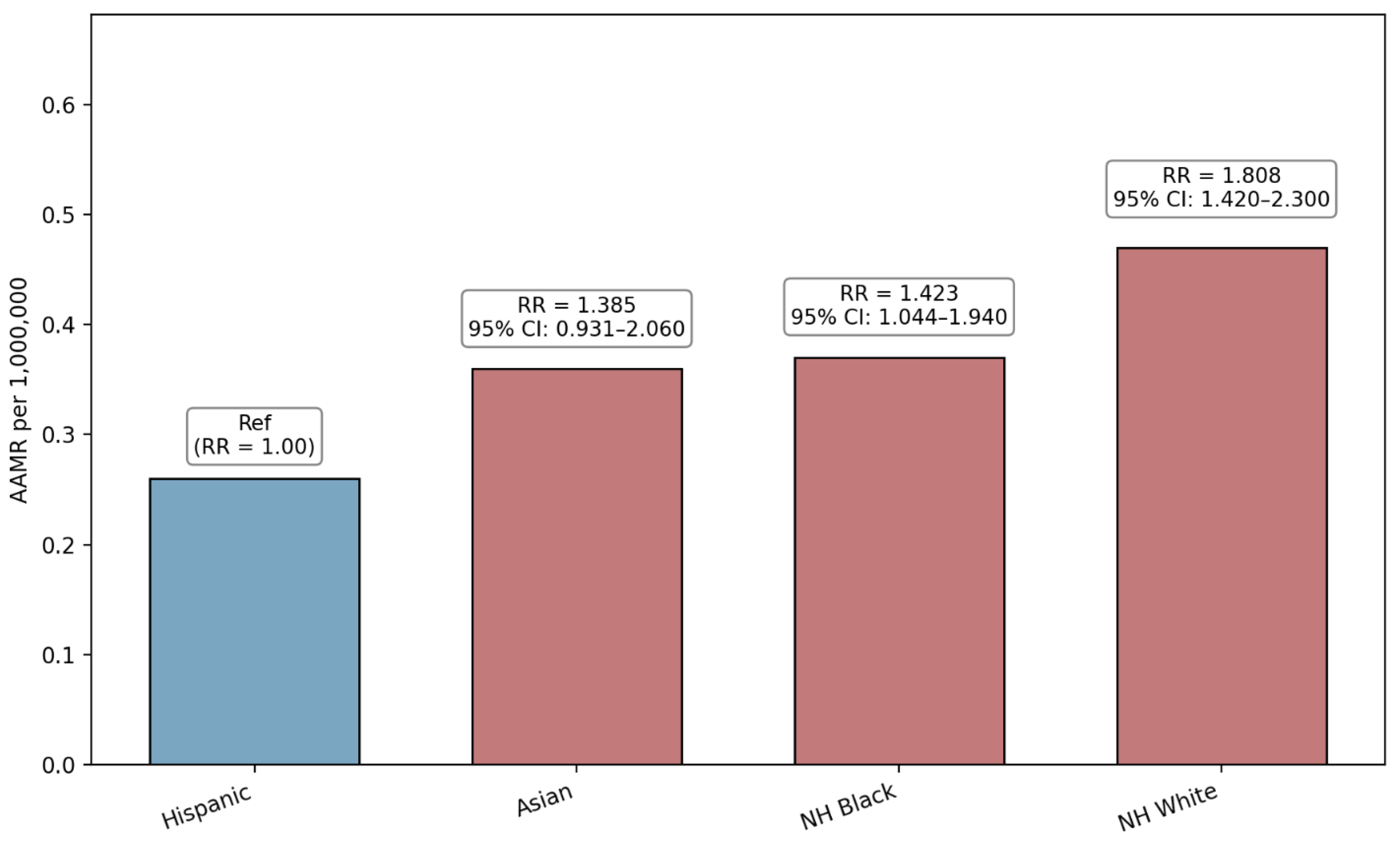
3.2. Race/Ethnicity-Based Disparities
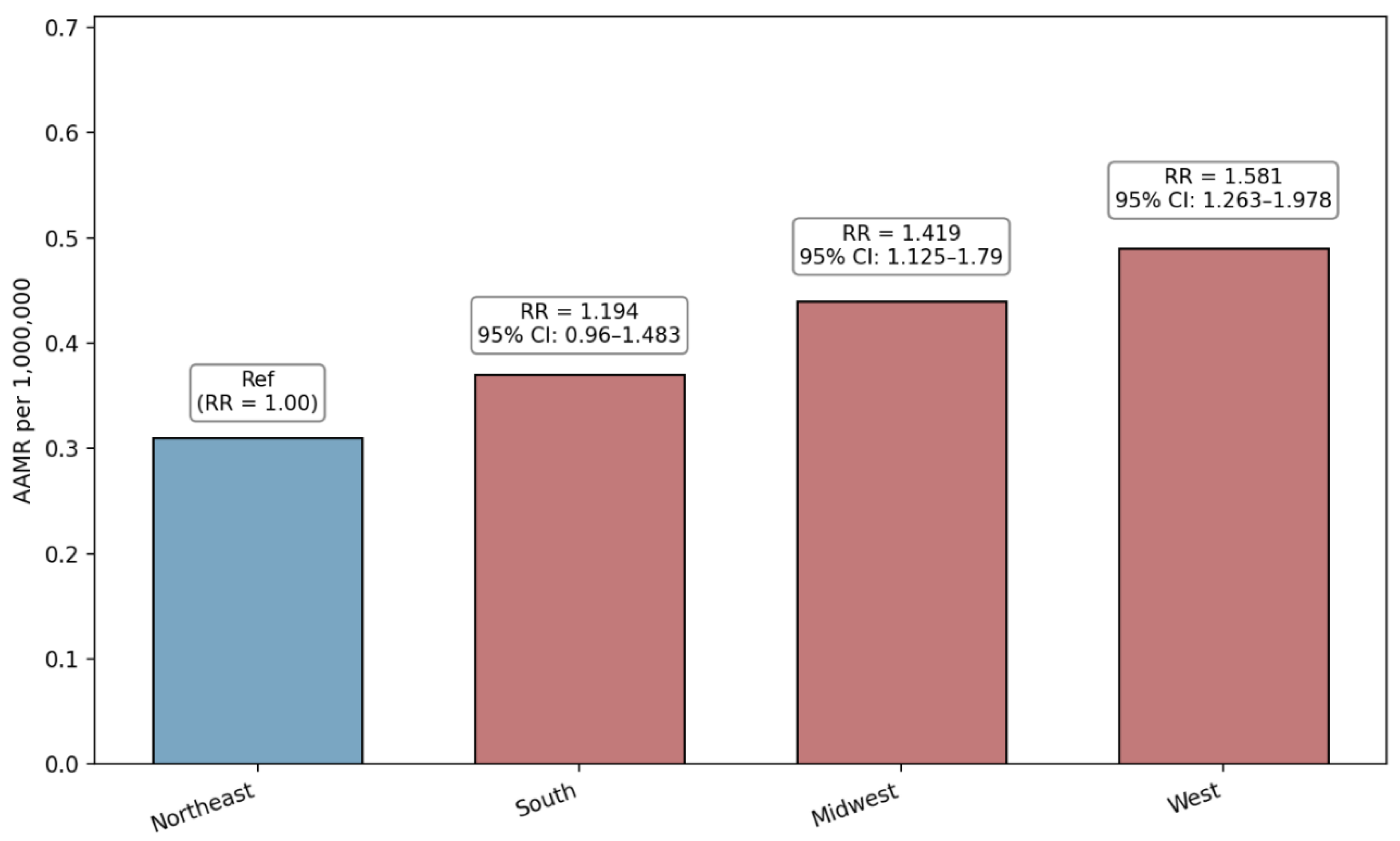
3.3. Region-Based Disparities
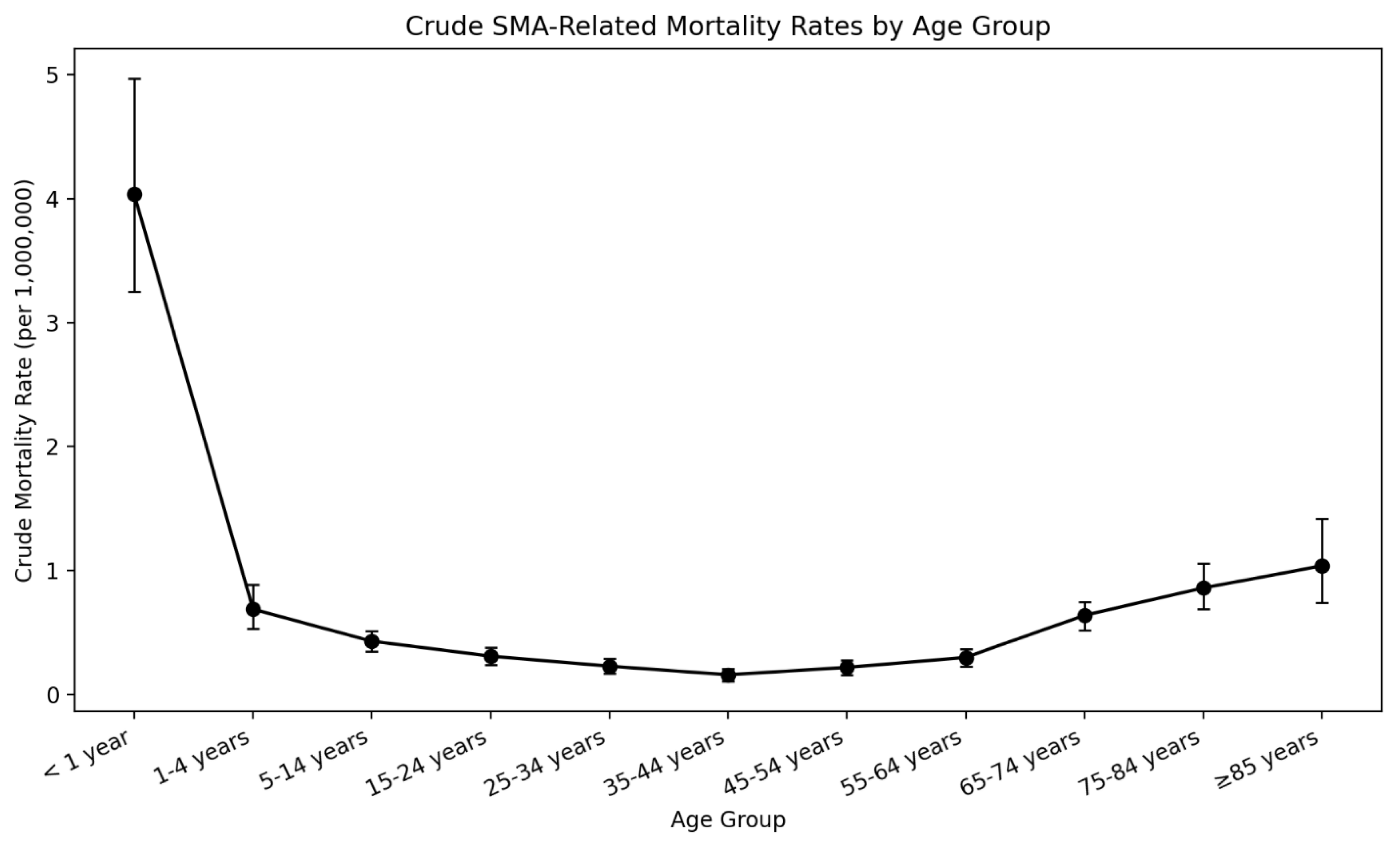
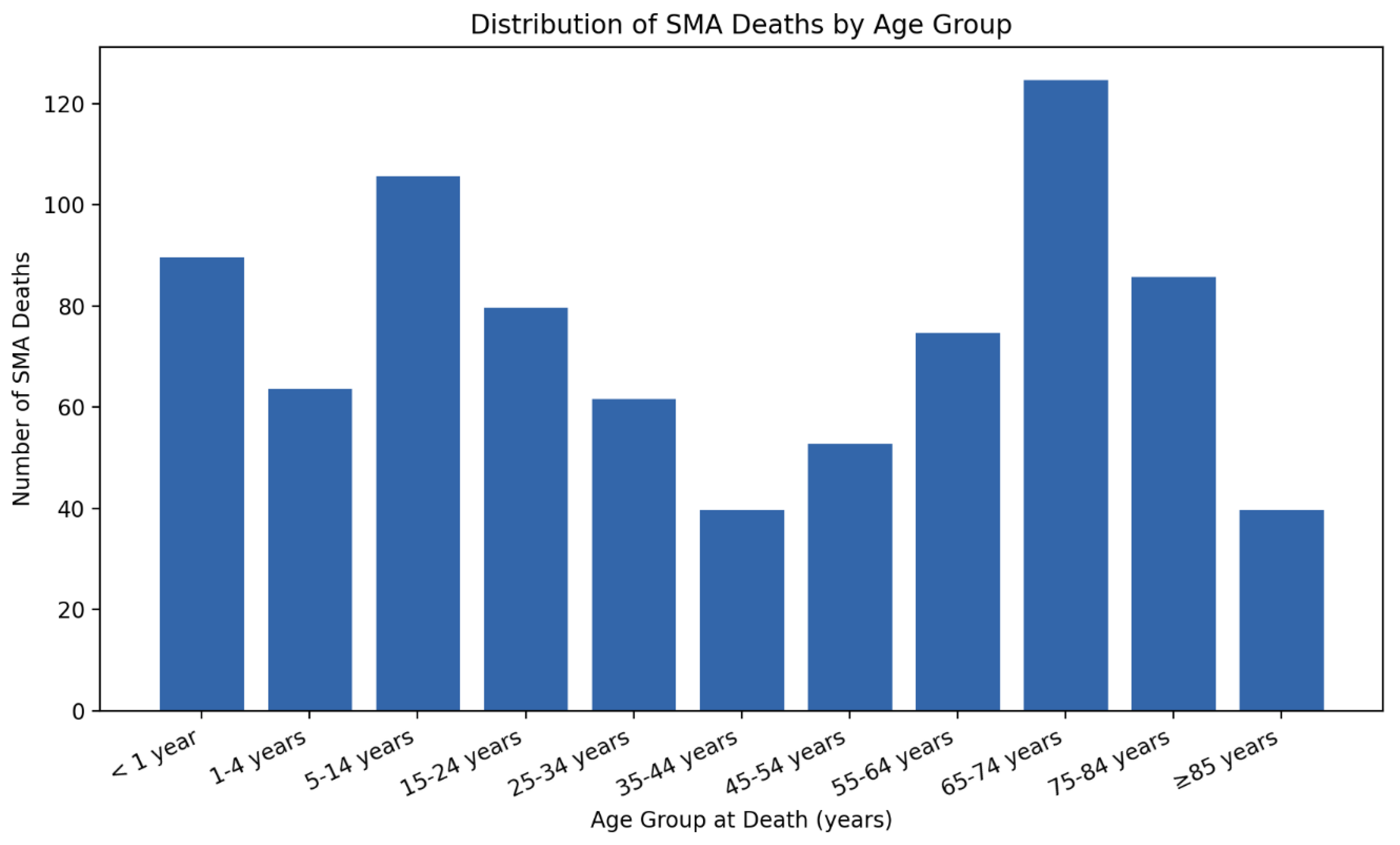
3.4. Age-Group Based Comparison
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mercuri, E.; Pera, M.C.; Scoto, M.; Finkel, R.; Muntoni, F. Spinal muscular atrophy — insights and challenges in the treatment era. Nat. Rev. Neurol. 2020, 16, 706–715. [Google Scholar] [CrossRef]
- Angilletta, I.; Ferrante, R.; Giansante, R.; Lombardi, L.; Babore, A.; Dell’elice, A.; Alessandrelli, E.; Notarangelo, S.; Ranaudo, M.; Palmarini, C.; et al. Spinal Muscular Atrophy: An Evolving Scenario through New Perspectives in Diagnosis and Advances in Therapies. Int. J. Mol. Sci. 2023, 24, 14873. [Google Scholar] [CrossRef]
- Lally, C.; Jones, C.; Farwell, W.; Reyna, S.P.; Cook, S.F.; Flanders, W.D. Indirect estimation of the prevalence of spinal muscular atrophy Type I, II, and III in the United States. Orphanet J. Rare Dis. 2017, 12, 175. [Google Scholar] [CrossRef]
- Global, regional, and national burden of motor neuron diseases 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 1083–1097. [CrossRef]
- Nishio, H.; Niba, E.T.E.; Saito, T.; Okamoto, K.; Takeshima, Y.; Awano, H. Spinal Muscular Atrophy: The Past, Present, and Future of Diagnosis and Treatment. Int. J. Mol. Sci. 2023, 24, 11939. [Google Scholar] [CrossRef]
- Li, Y.; Zeng, H.; Wei, Y.; Ma, X.; He, Z.-Y. An Overview of the Therapeutic Strategies for the Treatment of Spinal Muscular Atrophy. Hum. Gene Ther. 2023, 34, 180–191. [Google Scholar] [CrossRef] [PubMed]
- Viscidi, E.; Juneja, M.; Wang, J.; Wang, N.; Li, L.; Farwell, W.; Bhan, I.; Makepeace, C.; Laird, K.; Kupelian, V.; et al. Comparative All-Cause Mortality Among a Large Population of Patients with Spinal Muscular Atrophy Versus Matched Controls. Neurol. Ther. 11, 449–457. [CrossRef] [PubMed]
- Kochanek, K.D.; Murphy, S.L.; Xu, J.; Arias, E. Deaths: Final Data for 2020. Natl Vital Stat Rep. 2023, 72, 1–92. [Google Scholar]
- Ahmad, F.B.; Cisewski, J.A.; Xu, J.; Anderson, R.N. Provisional Mortality Data — United States, 2022. Mmwr-Morbidity Mortal. Wkly. Rep. 2023, 72, 488–492. [Google Scholar] [CrossRef]
- World Health Organization. ICD-10: international statistical classification of diseases and related health problems: tenth revision, 2nd ed.; World Health Organization, 2004. [Google Scholar]
- Ar Rochmah, M; Shima, A; Harahap, NIF; et al. Gender Effects on the Clinical Phenotype in Japanese Patients with Spinal Muscular Atrophy. Kobe J Med Sci. Published. 2017, 63, E41–E44. [Google Scholar] [PubMed]
- Zerres, K.; Rudnik-Schöneborn, S. Natural History in Proximal Spinal Muscular Atrophy. Clinical analysis of 445 patients and suggestions for a modification of existing classifications. Arch. Neurol. 1995, 52, 518–523. [Google Scholar] [CrossRef]
- Lipnick, S.L.; Agniel, D.M.; Aggarwal, R.; Makhortova, N.R.; Finlayson, S.G.; Brocato, A.; Palmer, N.; Darras, B.T.; Kohane, I.; Rubin, L.L. Systemic nature of spinal muscular atrophy revealed by studying insurance claims. PLOS ONE 2019, 14, e0213680. [Google Scholar] [CrossRef]
- Hendrickson, B.C.; Donohoe, C.; Akmaev, V.R.; A Sugarman, E.; Labrousse, P.; Boguslavskiy, L.; Flynn, K.; Rohlfs, E.M.; Walker, A.; Allitto, B. Differences in SMN1 allele frequencies among ethnic groups within North America. J. Med Genet. 2009, 46, 641–644. [Google Scholar] [CrossRef]
- A Sugarman, E.; Nagan, N.; Zhu, H.; Akmaev, V.R.; Zhou, Z.; Rohlfs, E.M.; Flynn, K.; Hendrickson, B.C.; Scholl, T.; Sirko-Osadsa, D.A.; et al. Pan-ethnic carrier screening and prenatal diagnosis for spinal muscular atrophy: clinical laboratory analysis of. Eur. J. Hum. Genet. 2012, 20, 27–32. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, W.K.; Hamilton, D.; Kuhle, S. SMA carrier testing: a meta-analysis of differences in test performance by ethnic group. Prenat. Diagn 2014, 34, 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- Mahungu, A.C.; Monnakgotla, N.; Nel, M.; Heckmann, J.M. A review of the genetic spectrum of hereditary spastic paraplegias, inherited neuropathies and spinal muscular atrophies in Africans. Orphanet J. Rare Dis. 2022, 17, 133. [Google Scholar] [CrossRef] [PubMed]
- Verhaart, I.E.C.; Robertson, A.; Wilson, I.J.; Aartsma-Rus, A.; Cameron, S.; Jones, C.C.; Cook, S.F.; Lochmüller, H. Prevalence, incidence and carrier frequency of 5q–linked spinal muscular atrophy – a literature review. Orphanet J. Rare Dis. 2017, 12, 124. [Google Scholar] [CrossRef]
- Carson, V.J.; Puffenberger, E.G.; E Bowser, L.; Brigatti, K.W.; Young, M.; Korulczyk, D.; Rodrigues, A.S.; Loeven, K.K.; A Strauss, K. Spinal muscular atrophy within Amish and Mennonite populations: Ancestral haplotypes and natural history. PLOS ONE 2018, 13, e0202104. [Google Scholar] [CrossRef]
- Ballreich, J.; Ezebilo, I.; Khalifa, B.A.; Choe, J.; Anderson, G. Coverage of genetic therapies for spinal muscular atrophy across fee-for-service Medicaid programs. J. Manag. Care Spéc. Pharm. 2022, 28, 39–47. [Google Scholar] [CrossRef]
- Lee, B.H.; Deng, S.; Chiriboga, C.A.; Kay, D.M.; Irumudomon, O.; Laureta, E.; Delfiner, L.; Treidler, S.O.; Anziska, Y.; Sakonju, A.; et al. Newborn Screening for Spinal Muscular Atrophy in New York State. Neurology 2022, 99, e1527–e1537. [Google Scholar] [CrossRef]
- Baker, M.; Griggs, R.; Byrne, B.; Connolly, A.M.; Finkel, R.; Grajkowska, L.; Haidet-Phillips, A.; Hagerty, L.; Ostrander, R.; Orlando, L.; et al. Maximizing the Benefit of Life-Saving Treatments for Pompe Disease, Spinal Muscular Atrophy, and Duchenne Muscular Dystrophy Through Newborn Screening: Essential Steps. JAMA Neurol. 2019, 76, 978–983. [Google Scholar] [CrossRef] [PubMed]
- Leon-Astudillo, C.; Byrne, B.J.; Salloum, R.G. Addressing the implementation gap in advanced therapeutics for spinal muscular atrophy in the era of newborn screening programs. Front. Neurol. 2022, 13, 1064194. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Deaths | AAMR per 1,000,000 | RR (95% CI) | p-value |
| Total | 821 | 0.4 | NA | NA |
| Biological Sex | ||||
| Female (ref) | 376 | 0.33 | 1 | NA |
| Male | 445 | 0.44 | 1.189 (1.035 to 1.366) | 0.014 |
| Race/Ethnicity | ||||
| Hispanic (Ref) | 99 | 0.26 | 1 | NA |
| NH White | 574 | 0.47 | 1.808 (1.420 to 2.300) | <0.0001 |
| NH Black | 86 | 0.37 | 1.4230 (1.0438 to 1.9400) | 0.0257 |
| Asian | 36 | 0.36 | 1.3846 (0.9306 to 2.0600) | 0.1084 |
| Census Region | ||||
| Northeast (Ref) | 113 | 0.31 | 1 | NA |
| South | 289 | 0.37 | 1.1935 (0.9604 to 1.4831) | 0.1104 |
| Midwest | 191 | 0.44 | 1.4193 (1.1254 to 1.7900) | 0.003 |
| West | 228 | 0.49 | 1.5806 (1.2629 to 1.9783) | <0.0001 |
| Age-Groups | Deaths | CMR per 1,000,000 | RR (95% CI) | p-value |
| < 1 year | 90 | 4.04 | 25.25 (18.30 to 34.83) | <0.000001 |
| 1-4 years | 64 | 0.69 | 4.31 (3.03 to 6.14) | <0.000001 |
| 5-14 years | 106 | 0.43 | 2.69 (1.98 to 3.65) | <0.000001 |
| 15-24 years | 80 | 0.31 | 1.94 (1.42 to 2.64) | <0.000001 |
| 25-34 years | 62 | 0.23 | 1.44 (1.01 to 2.05) | 0.045 |
| 35-44 years (Ref) | 40 | 0.16 | 1 | NA |
| 45-54 years | 53 | 0.22 | 1.38 (0.96 to 1.98) | 0.085 |
| 55-64 years | 75 | 0.3 | 1.88 (1.37 to 2.57) | <0.0001 |
| 65-74 years | 125 | 0.64 | 4 (2.94 to 5.43) | <0.000001 |
| 75-84 years | 86 | 0.86 | 5.38 (3.90 to 7.40) | <0.000001 |
| 85+ years | 40 | 1.04 | 6.50 (4.41 to 9.59) | <0.000001 |
| Total | 821 | 0.41 | NA | NA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




