Submitted:
29 January 2026
Posted:
30 January 2026
You are already at the latest version
Abstract
Keywords:
1. Background
2. Patients and Methods
2.1. Study Design
2.2. Ethical Consideration
2.3. Patient Selection Criteria
2.3.1. Patients Inclusion criteria
- 18 years old or older Patient diagnosed with refractory DME, RVO edema, or active n-AMD by clinical evidence, optical coherence tomography OCT and angiography (OCTA).
- Primary refractory patients show persistent subretinal or intraretinal fluid with no improvement BCVA nor a decrease in central retinal thickness after a minimum of 3 loading doses of Bevacizumab and Aflibercept (CRT>300 µm with BCVA either unchanged or worse compared with baseline).
- Ability to attend follow-up appointments and comply with treatment protocol.
2.3.2. Patient Exclusion Criteria
- Inconsistent treatment history: missed a dose of anti-VEGF doses, or failure to complete the three loading doses of Faricimab.
- Ocular comorbidities that could confound outcomes, such as: Visually significant cataract as Grade 2+ or more, corneal opacity, uncontrolled glaucoma (IOP > 25 mmHg on medication) with an increase in cup/disc ratio, Other macular pathologies (e.g., macular hole, epiretinal membrane ERM).
- Severe baseline vision loss (BCVA < 6/60).
- 4. Secondary non-responders: patients who initially showed improvement in BCVA and CRT after anti-VEGF therapy, but subsequently experienced deterioration despite continued treatment with bevacizumab and aflibercept.
2.4. Methods
2.5. Statistical Analysis
3. Results
3.1. Demographic Data and Medical Background of Patients
3.2. Characteristic of the Eyes to be Treated
3.3. Ocular Diagnosis
3.4. Demographic Data and Medical Background of Patients with DME
3.5. Clinical Parameters for Patients with DME
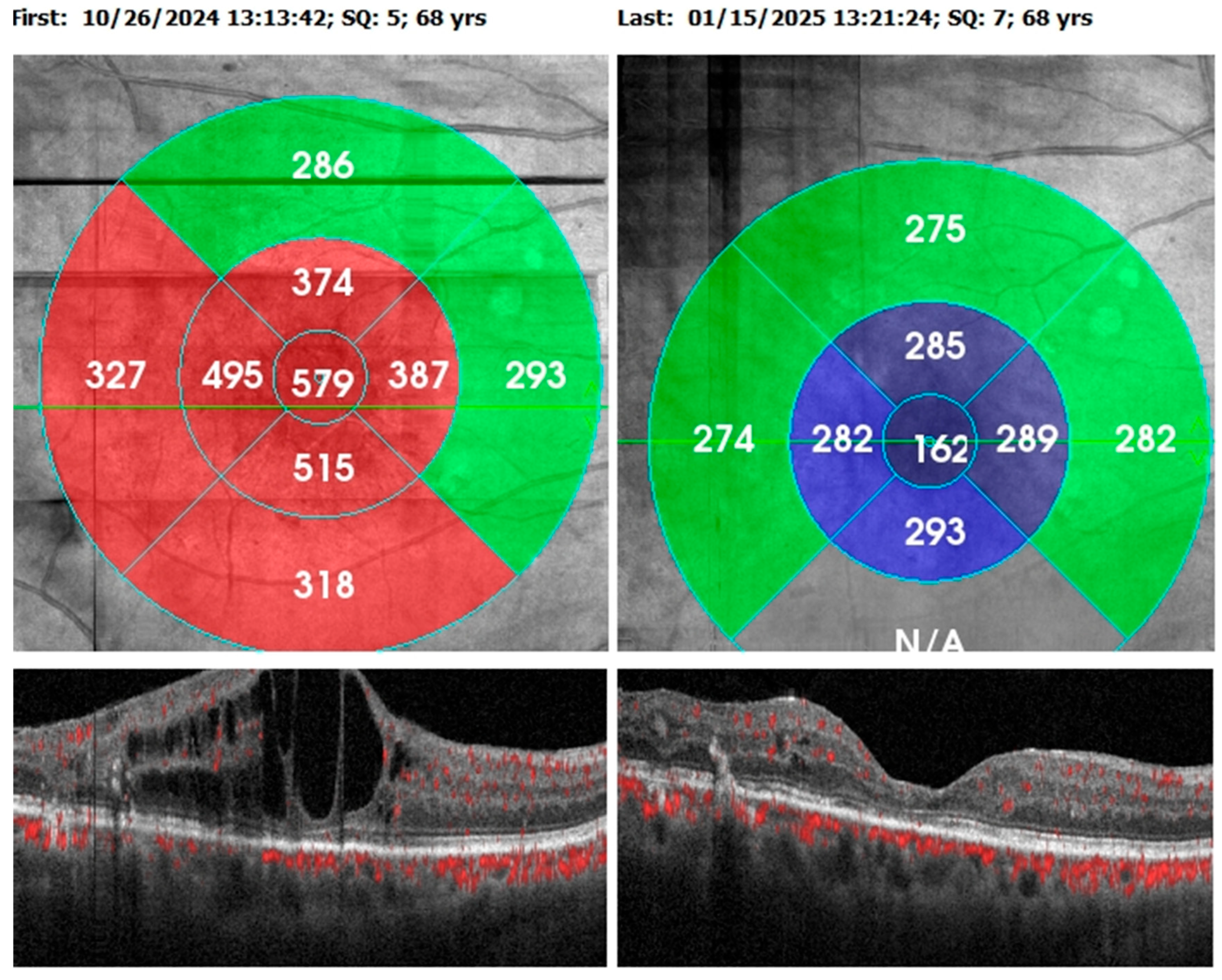
| Parameters | Pretreatment | Posttreatment | P* value | ||
| Mean | SD | Mean | SD | ||
| Visual Acuity LogMAR | 0.60 | 0.24 | 0.44 | 0.24 | <0.001 |
| Intraocular Pressure | 15.64 | 2.20 | 15.57 | 2.54 | 0.673 |
| Central Retinal Thickness | 464.74 | 112.99 | 288.5 | 85.04 | <0.001 |
| Parameters | No.46 | 100% | No.46 | 100% | P^ value |
| Subretinal Fluid | 15 | 32.60% | 2 | 4.34% | <0.001 |
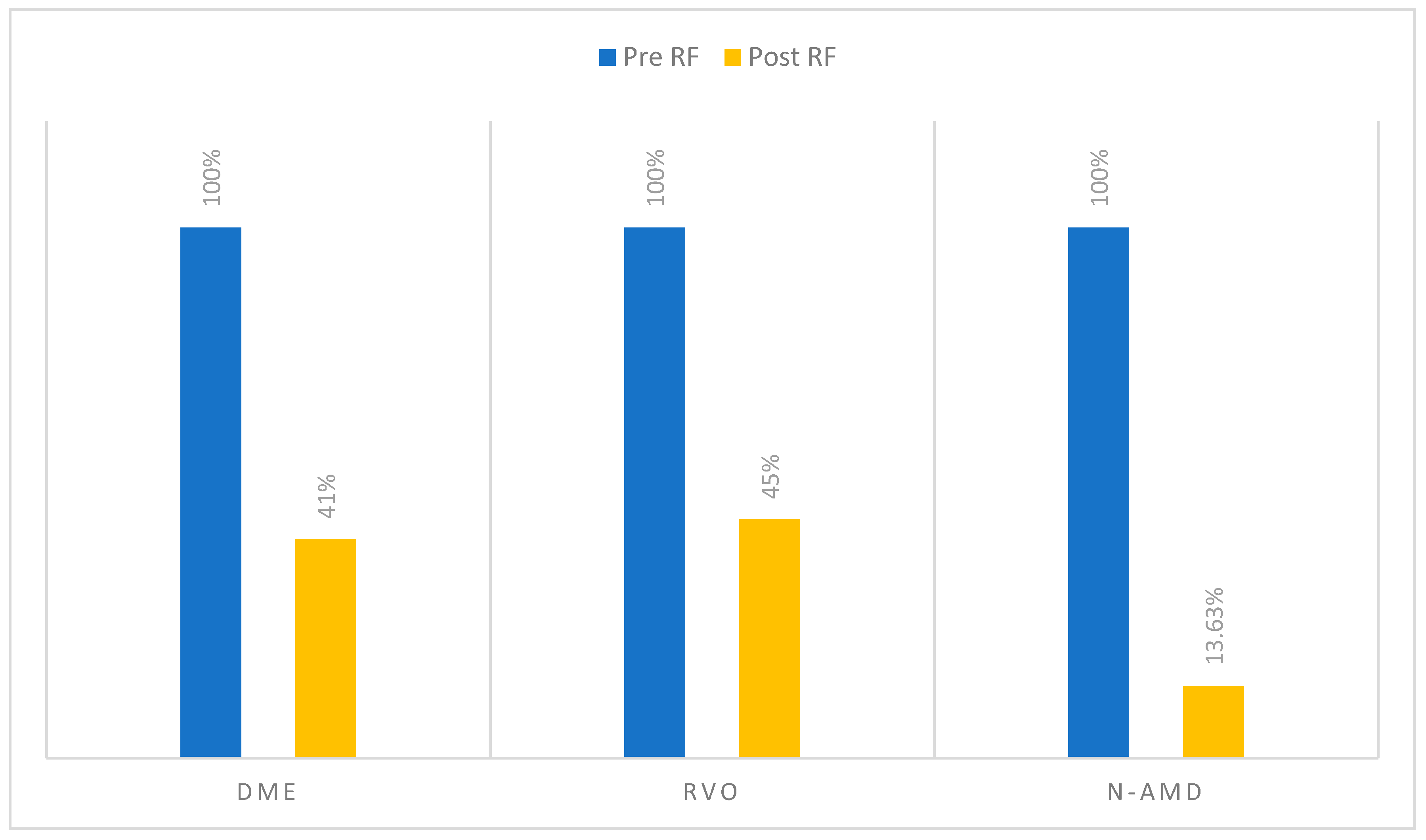
| Intraretinal Fluid | 46 | 100% | 19 | 41.30% | <0.001 |
3.6. Demographic Data and Medical Background of Patients with RVO
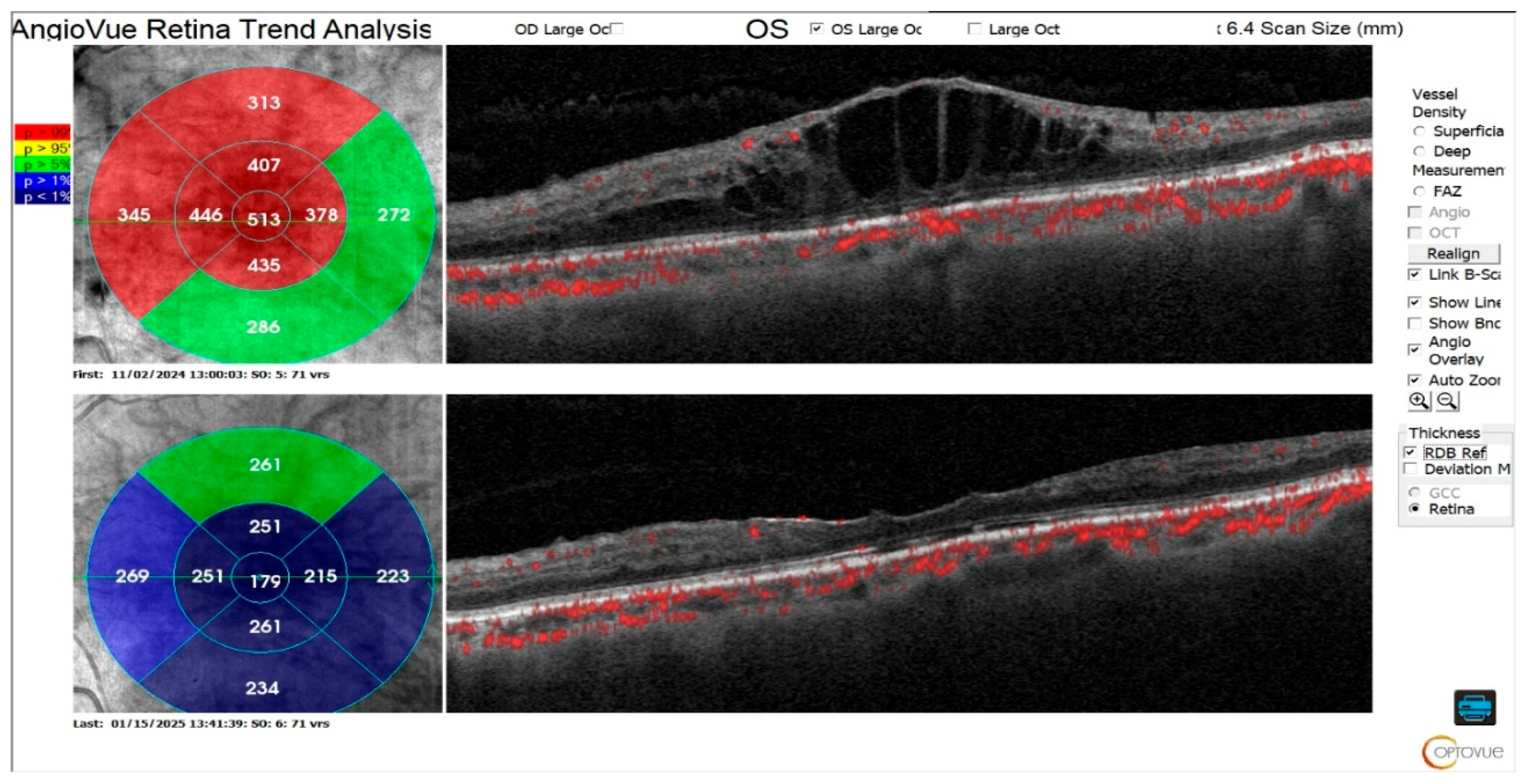
3.7. Clinical Parameters for Patients with RVO
| Parameters | Pretreatment | Posttreatment | P* value | ||
| Mean | SD | Mean | SD | ||
| Visual Acuity LogMAR | 0.71 | 0.25 | 0.48 | 0.27 | <0.001 |
| Intraocular Pressure | 17.65 | 2.13 | 17.5 | 1.88 | 0.408 |
| Central Retinal Thickness | 534.3 | 144.79 | 324.45 | 88.30 | <0.001 |
| Parameters | No.20 | 100% | No.20 | 100% | P^ value |
| Subretinal Fluid | 10 | 50% | 2 | 10% | 0.015 |
| Intraretinal Fluid | 20 | 100% | 9 | 45% | 0.002 |
3.8. Demographic Data and Medical Background of Patients with n-AMD
3.9. Clinical Parameters for Patients with n-AMD
| Parameters | Pretreatment | Posttreatment | P* value | ||
| Mean | SD | Mean | SD | ||
| Visual Acuity LogMAR | 0.63 | 0.18 | 0.39 | 0.28 | <0.001 |
| Intraocular Pressure | 14.64 | 2.44 | 14.91 | 2.83 | 0.216 |
| Central Retinal Thickness | 411.23 | 78.99 | 268.73 | 58.13 | <0.001 |
| Pigmented epithelial detachment | 176.91 | 225.71 | 68.91 | 100.94 | 0.007 |
| Parameters | No.22 | 100% | No.22 | 100% | P^ value |
| Subretinal Fluid | 22 | 100% | 3 | 13.63% | <0.001 |
| Intraretinal Fluid | 6 | 27.27% | 0 | 0% | <0.05 |
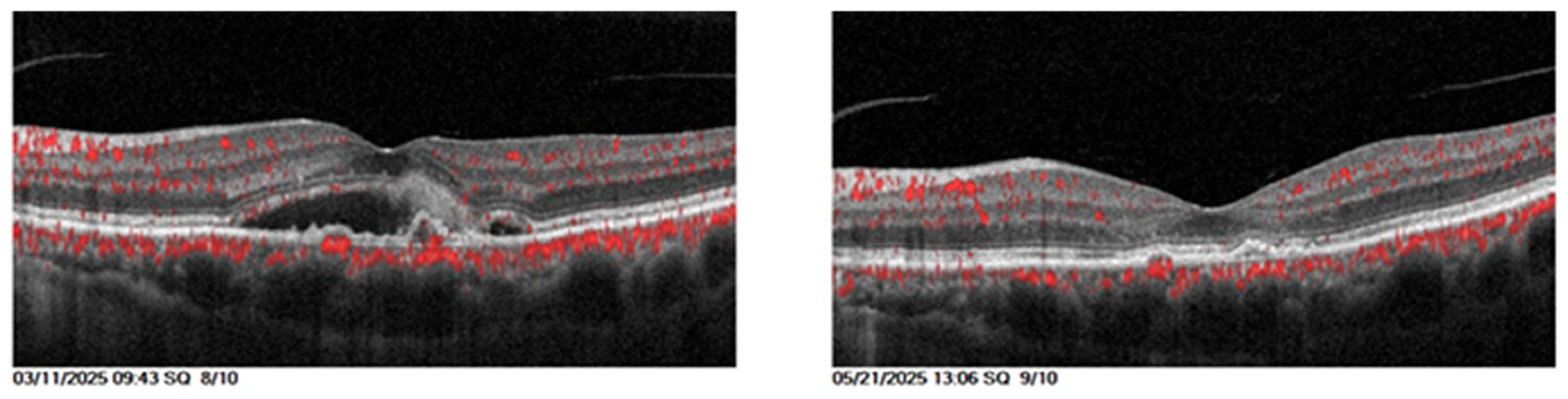
3.10. Retinal fluid
4. Discussion
4.1. Patient Demographics and Background
4.2. Best Corrected Visual Acuity
4.3. Anatomical Outcomes
5. Conclusions
Funding
Ethical Approval
Consent of Publication
Availability of Data and Materials
Acknowledgments
Conflicts of Interest
Abbreviations
| Ang2 | angiopoietin |
| BCVA | best corrected visual acuity |
| CRT | central retinal thickness |
| DME | diabetic macular edema |
| DM | diabetes mellitus |
| EDTR | Early treatment diabetic retinopathy study |
| HT | hypertension |
| IOP | intraocular pressure |
| IRF | intraretinal fluid |
| n-AMD | neovascular age related macular degeneration |
| OCTA | optical coherence tomography angiography |
| PED | pigmented epithelial detachment |
| PMH | past medical history |
| RVO | retinal vein occlusion edema |
| SD | standard deviation |
| SD-OCT | spectral domain optical coherence tomography |
| SRF | subretinal fluid |
| VA | visual acuity |
| VEGF | vascular endothelial growth factor |
References
- Panos, G.D., et al., Faricimab: transforming the future of macular diseases treatment-a comprehensive review of clinical studies. Drug design, development and therapy, 2023: p. 2861-2873. [CrossRef]
- Weis, S.M. and D.A. Cheresh, Pathophysiological consequences of VEGF-induced vascular permeability. Nature, 2005. 437(7058): p. 497-504. [CrossRef]
- Shen, J., et al., Targeting VE-PTP activates TIE2 and stabilizes the ocular vasculature. The Journal of clinical investigation, 2014. 124(10): p. 4564-4576. [CrossRef]
- Hussain, R.M., et al., Tie-2/Angiopoietin pathway modulation as a therapeutic strategy for retinal disease. Expert opinion on investigational drugs, 2019. 28(10): p. 861-869. [CrossRef]
- Agarwal, R., S.K. Gupta, and S. Agrawal, Efficacy and safety analysis of intravitreal bio-similar products of bevacizumab in patients with macular edema because of retinal diseases. Indian Journal of Ophthalmology, 2023. 71(5): p. 2066-2070. [CrossRef]
- Fogli, S., et al., Clinical pharmacology of intravitreal anti-VEGF drugs. Eye, 2018. 32(6): p. 1010-1020. [CrossRef]
- Platania, C.B., et al., Molecular features of interaction between VEGFA and anti-angiogenic drugs used in retinal diseases: a computational approach. Frontiers in pharmacology, 2015. 6: p. 248. [CrossRef]
- Yerramothu, P., New Therapies of Neovascular AMD—Beyond Anti-VEGFs. Vision, 2018. 2(3): p. 31. [CrossRef]
- Bressler, N.M., et al., Biosimilars of anti-vascular endothelial growth factor for ophthalmic diseases: A review. Survey of ophthalmology, 2024. 69(4): p. 521-538. [CrossRef]
- Nair, A.A., A.P. Finn, and P. Sternberg Jr, Spotlight on faricimab in the treatment of wet age-related macular degeneration: design, development and place in therapy. Drug Design, Development and Therapy, 2022: p. 3395-3400. [CrossRef]
- Thapa, R., et al., Prevalence, pattern and risk factors of retinal diseases among an elderly population in Nepal: the bhaktapur retina study. Clinical Ophthalmology, 2020: p. 2109-2118. [CrossRef]
- Shafiee, A., et al., Racial and Gender Disparities in Clinical Trial Representation for Age-Related Macular Degeneration Treatments: A Scoping Review. Journal of Clinical & Translational Ophthalmology, 2025. 3(3): p. 16. [CrossRef]
- Owusu-Afriyie, B., et al., Prevalence of risk factors of retinal diseases among patients in Madang Province, Papua New Guinea. International Journal of Clinical Practice, 2022. 2022(1): p. 6120908. [CrossRef]
- Rush, R.B. and S.W. Rush, Faricimab for treatment-resistant diabetic macular edema. Clinical Ophthalmology (Auckland, NZ), 2022. 16: p. 2797. [CrossRef]
- Machida, A., et al., Factors Associated with Success of Switching to Faricimab for Neovascular Age-Related Macular Degeneration Refractory to Intravitreal Aflibercept. Life, 2024. 14(4): p. 476. [CrossRef]
- Nichani, P.A., et al., Efficacy and safety of intravitreal faricimab in neovascular age-related macular degeneration, diabetic macular edema, and retinal vein occlusion: A meta-analysis. Ophthalmologica, 2024. 247(5-6): p. 355-372. [CrossRef]
- Rush, R.B., One year results of faricimab for aflibercept-resistant diabetic macular edema. Clinical Ophthalmology, 2023: p. 2397-2403. [CrossRef]
- Deiters, V., et al., Real-World Data on Morphological and Functional Responses After Switching to Faricimab in Recalcitrant, Chronic Diabetic Macular Edema. Ophthalmology and Therapy, 2025: p. 1-9. [CrossRef]
- Hattenbach, L.-O., et al., BALATON and COMINO: phase III randomized clinical trials of faricimab for retinal vein occlusion: study design and rationale. Ophthalmology Science, 2023. 3(3): p. 100302.
- Hafner, M., et al., Switching to Faricimab in Therapy-Resistant Macular Edema Due to Retinal Vein Occlusion: Initial Real-World Efficacy Outcomes. Journal of Clinical Medicine, 2025. 14(7): p. 2454. [CrossRef]
- Löw, K., et al., Real-Life Treatment Intervals and Morphological Outcomes Following the Switch to Faricimab Therapy in Neovascular Age-Related Macular Degeneration. Journal of Personalized Medicine, 2025. 15(5): p. 189. [CrossRef]
- Rush, R.B., One-year outcomes of faricimab treatment for aflibercept-resistant neovascular age-related macular degeneration. Clinical Ophthalmology, 2023: p. 2201-2208. [CrossRef]
- Raimondi, R., et al., Outcomes of treatment-resistant neovascular age-related macular degeneration switched from aflibercept to faricimab. Ophthalmology Retina, 2024. 8(6): p. 537-544. [CrossRef]
- Schneider, M., et al., Short-term outcomes of treatment switch to faricimab in patients with aflibercept-resistant neovascular age-related macular degeneration. Graefe’s Archive for Clinical and Experimental Ophthalmology, 2024. 262(7): p. 2153-2162. [CrossRef]
- Santos, A.R., et al., Measurements of retinal fluid by optical coherence tomography leakage in diabetic macular edema: a biomarker of visual acuity response to treatment. Retina, 2019. 39(1): p. 52-60.
- Lim, J.I., et al., Anatomic Control with Faricimab versus Aflibercept in the YOSEMITE/RHINE Trials in Diabetic Macular Edema. Ophthalmology Retina, 2025. [CrossRef]
- Sim, S.Y., et al., Real-world 1-year outcomes of treatment-intensive neovascular age-related macular degeneration switched to faricimab. Ophthalmology Retina, 2025. 9(1): p. 22-30. [CrossRef]
- Szigiato, A., et al., Short-term outcomes of faricimab in patients with neovascular age-related macular degeneration on prior anti-VEGF therapy. Ophthalmology Retina, 2024. 8(1): p. 10-17. [CrossRef]
- Pandit, S.A., et al., Clinical outcomes of faricimab in patients with previously treated neovascular age-related macular degeneration. Ophthalmology Retina, 2024. 8(4): p. 360-366. [CrossRef]
- Yen, W.-T., et al., Efficacy and safety of intravitreal faricimab for neovascular age-related macular degeneration: a systematic review and meta-analysis. Scientific Reports, 2024. 14(1): p. 2485. [CrossRef]
- Hoguet, A., et al., The effect of anti-vascular endothelial growth factor agents on intraocular pressure and glaucoma: a report by the American Academy of Ophthalmology. Ophthalmology, 2019. 126(4): p. 611-622.
- Callizo, J., et al., Real-world data: ranibizumab treatment for retinal vein occlusion in the OCEAN study. Clinical Ophthalmology, 2019: p. 2167-2179. [CrossRef]
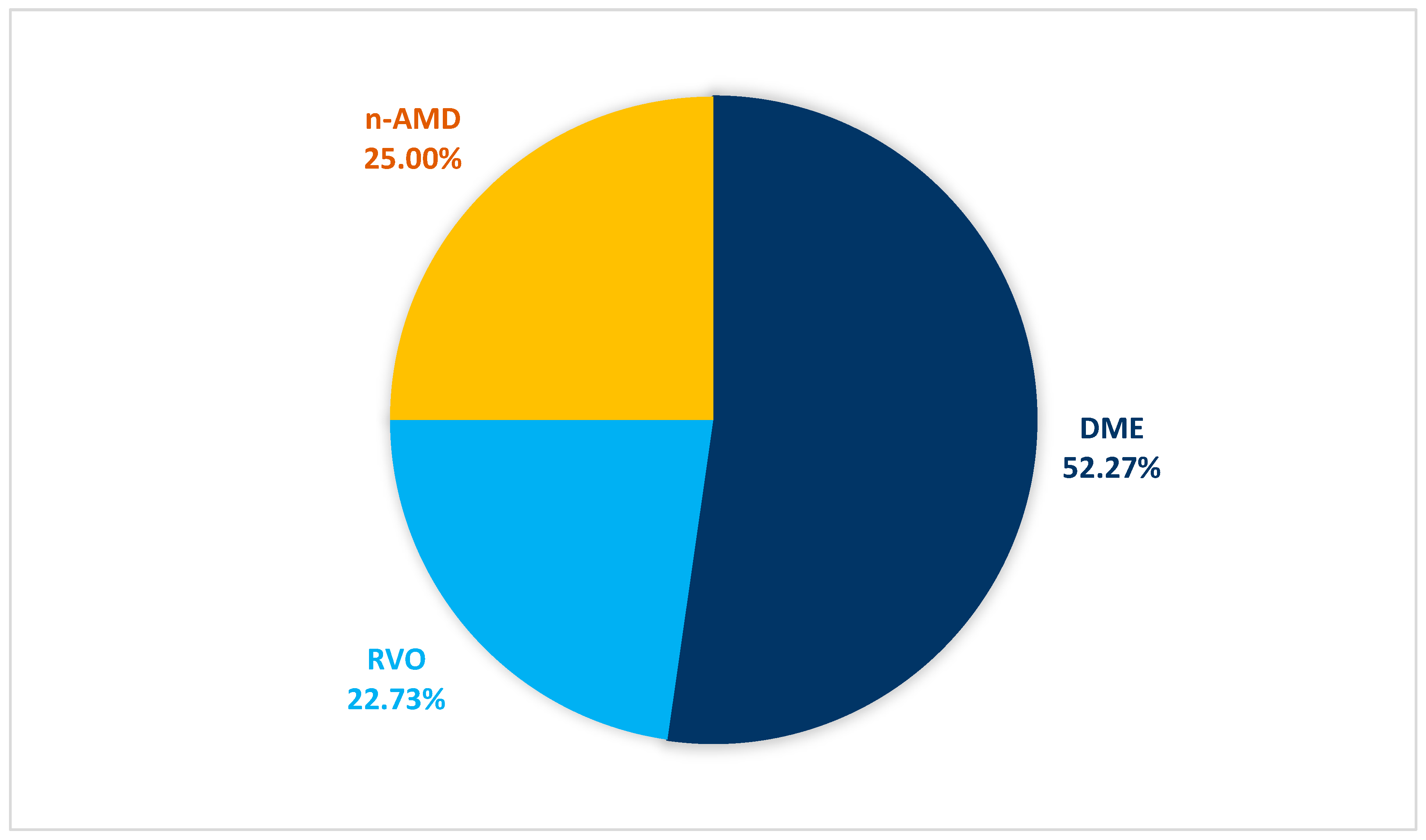
| Variables | No. (76) | 100% | |
|---|---|---|---|
| Mean age/years (mean±SD*) | 66.14±8.678 | ||
| Age ≥ 65 | 43 | 56.57% | |
| Age < 65 | 33 | 43.42% | |
| Gender | Male | 37 | 48.68% |
| Female | 39 | 51.32% | |
| Variables | No. (76) | 100% |
|---|---|---|
| PMH | 62 | 81.58% |
| HT | 30 | 39.47% |
| DM | 47 | 61.84% |
| Variables | No. (88) | 100% | |
|---|---|---|---|
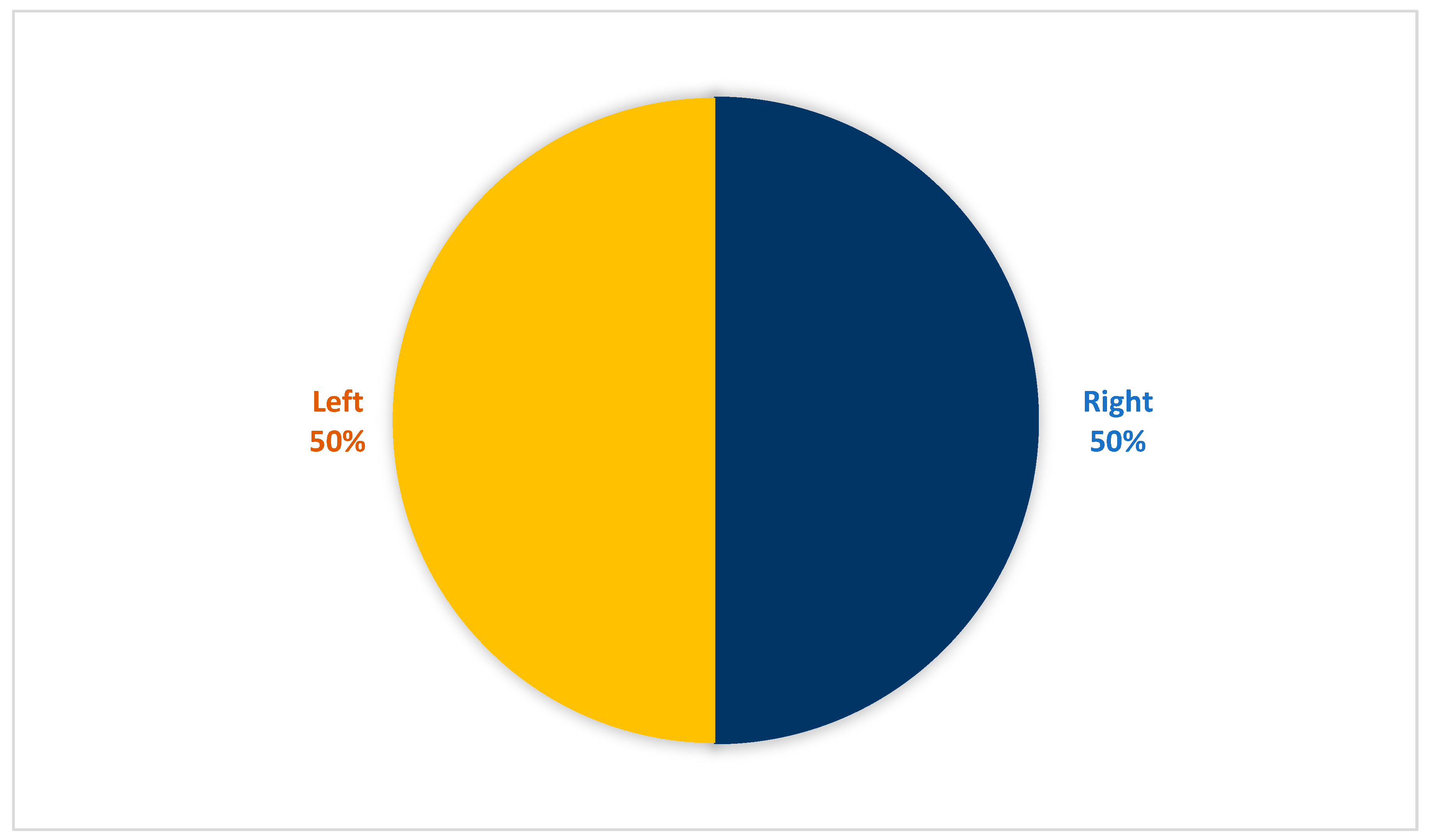
| Laterality | Right | 44 | 50% |
| Left | 44 | 50% | |
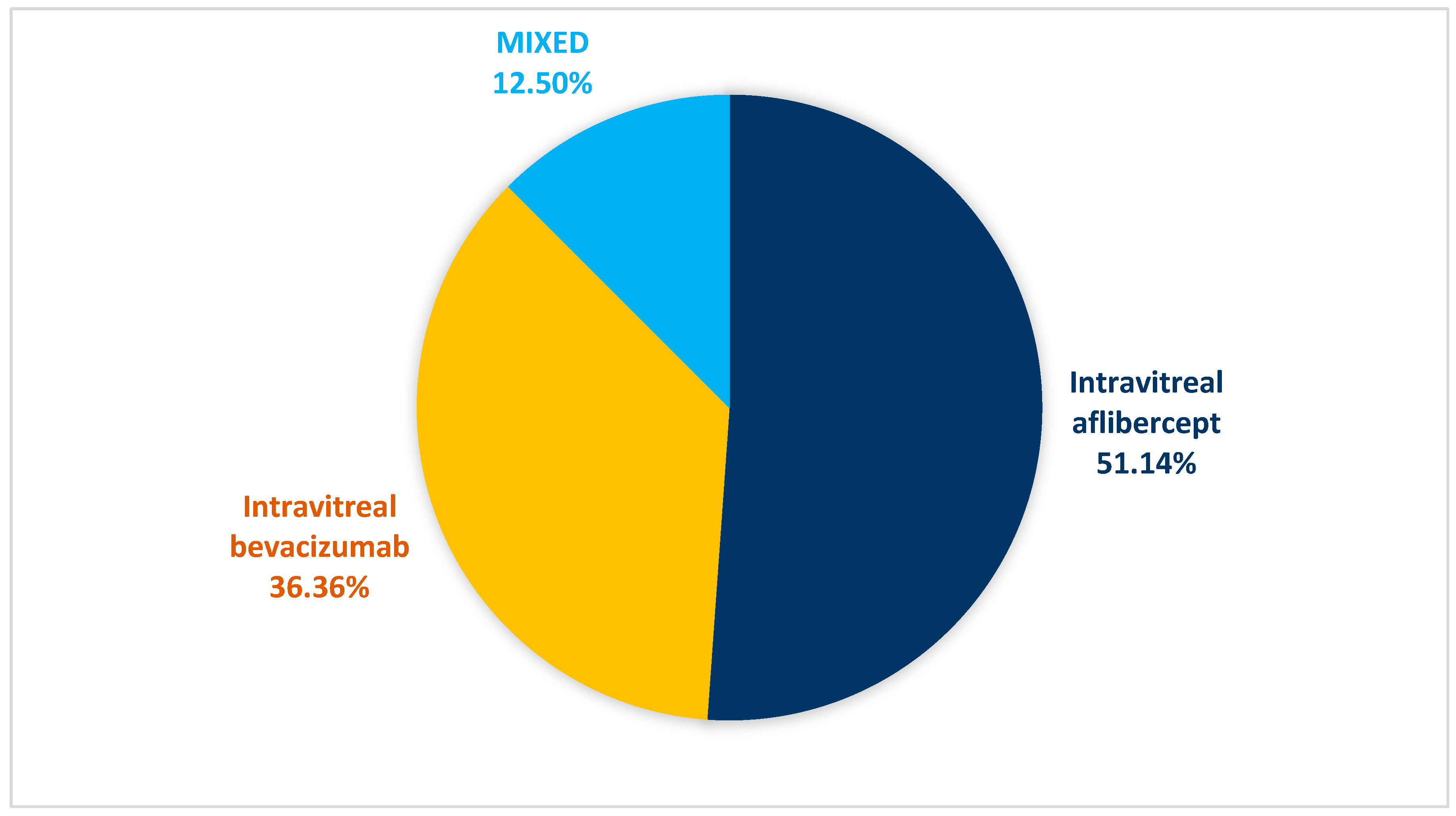
| Intravitreal aflibercept | 45 | 51.14% | |
| Intravitreal bevacizumab | 32 | 36.36% | |
| Intravitreal aflibercept + Intravitreal bevacizumab | 11 | 12.50% | |
| Dose (mean±SD*) | 7 | 1.77 | |
| Variables | No. (38) | 100% |
|---|---|---|
| Mean age/years (mean±SD*) | 64.8±7.9 | |
| Age ≥ 65 | 17 | 44.7% |
| Age < 65 | 21 | 55.3% |
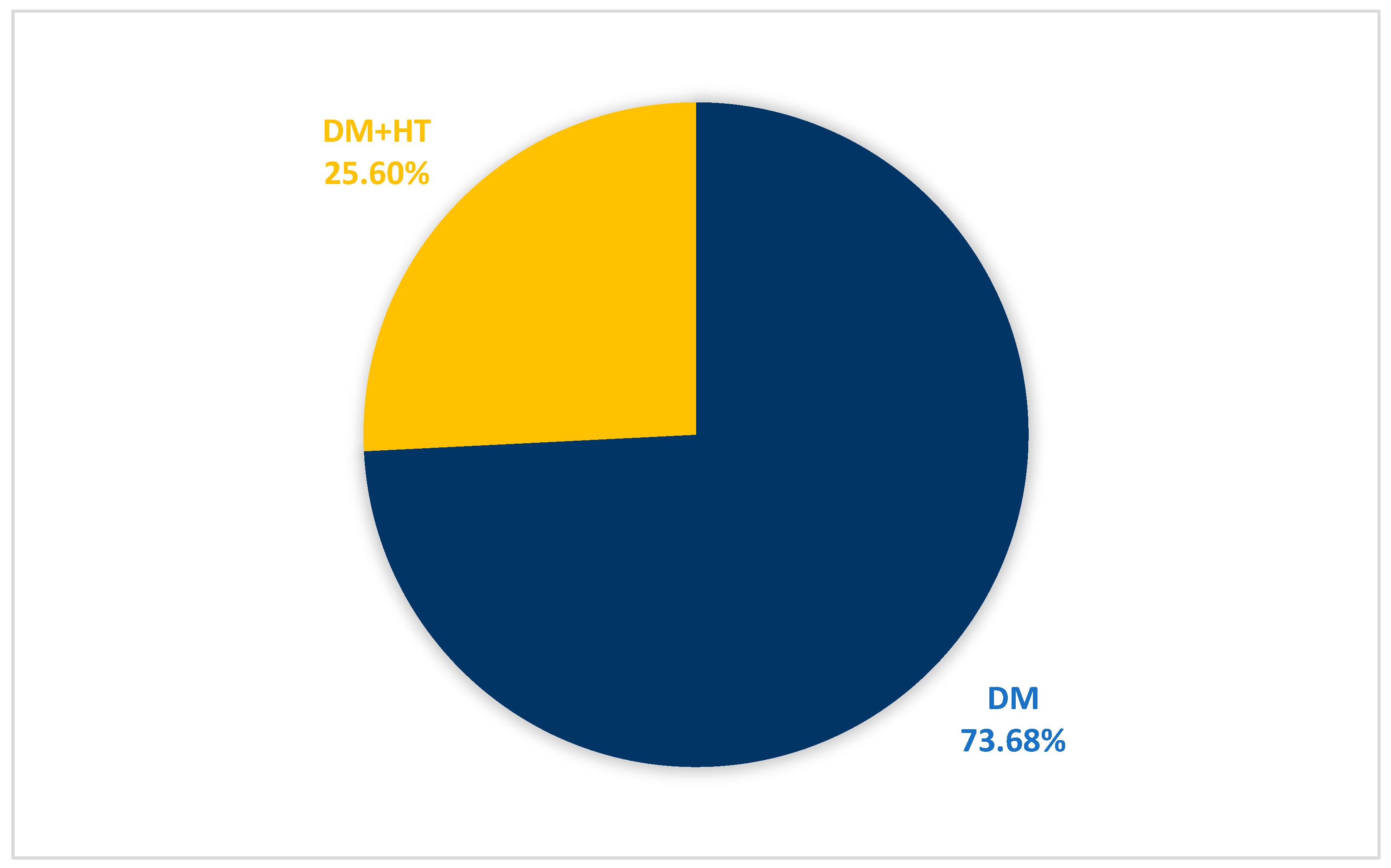
| PMH | 38 | 100% |
| DM | 38 | 100% |
| HT | 10 | 25.6% |
| Variables | No. (19) | 100% |
|---|---|---|
| Mean age/years (mean±SD*) | 63.6±8.6 | |
| Age ≥ 65 | 8 | 42.1% |
| Age < 65 | 11 | 57.9% |
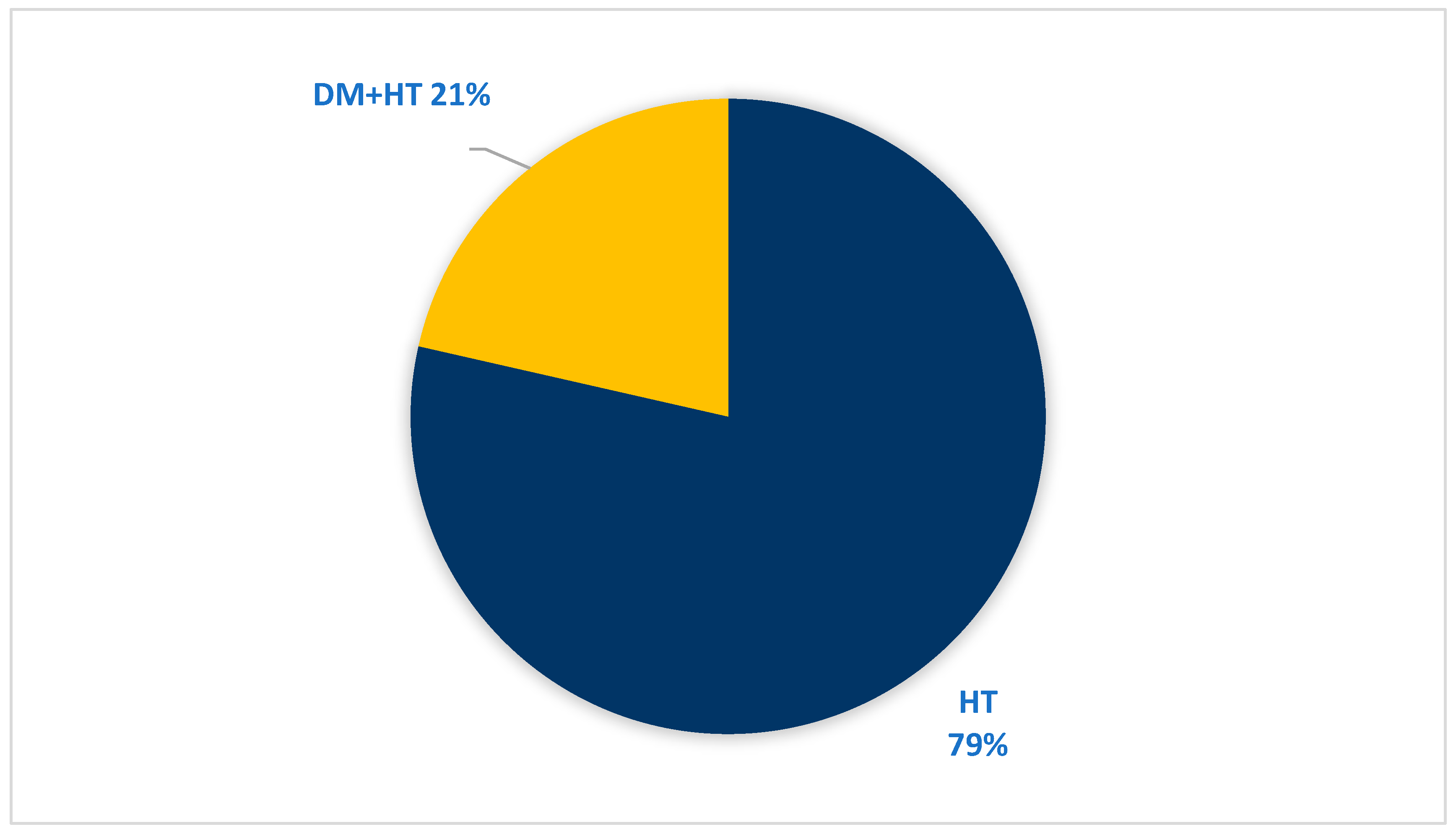
| PMH | 14 | 73.7% |
| HT | 14 | 73.7% |
| DM | 3 | 15.9% |
| Variables | No. (19) | 100% |
|---|---|---|
| Mean age/years (mean±SD*) | 74.1±5.4 | |
| Age ≥ 65 | 18 | 94.7% |
| Age < 65 | 1 | 5.3% |
| PMH | 10 | 52.6% |
| HT | 7 | 36.8% |
| DM | 6 | 31.6% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




