Submitted:
01 October 2025
Posted:
02 October 2025
You are already at the latest version
Abstract
Background: Acute kidney injury (AKI) is a frequent and serious complication among individuals with type 2 diabetes, contributing to long-term renal decline and heightened cardiovascular risk. Glucagon-like peptide-1 receptor agonists (GLP-1 RAs), such as semaglutide, are widely prescribed and often regarded as kidney protective, yet post-marketing experience has linked them to AKI in some cases. Tirzepatide, a newer dual GIP/GLP-1 agonist, offers striking metabolic benefits, but its short-term renal safety in routine practice has not been fully characterized. Methods: We performed a disproportionality analysis of the U.S. FDA Adverse Event Reporting System (FAERS) from January 2022 to September 2025. Reports listing tirzepatide or semaglutide as the primary suspect drug were included, and AKI was defined using standardized MedDRA terms. Reporting odds ratios (RORs) and proportional reporting ratios (PRRs) were calculated to assess whether AKI was reported more frequently than expected. Results: A total of 92,807 adverse event reports for tirzepatide and 41,065 for semaglutide were identified. AKI was reported in 432 tirzepatide cases (0.47%) and 440 semaglutide cases (1.07%). Compared with semaglutide, tirzepatide was associated with a significantly lower frequency of AKI reporting (ROR 0.44, 95% CI 0.38–0.50; PRR 0.44). Sex distribution differed between agents, with a higher proportion of females among semaglutide cases. Year on year reporting increased for both drugs, consistent with broader clinical uptake. Serious outcomes, including death, hospitalization, and life threatening events, were more frequently documented with semaglutide. Conclusions: In this real-world pharmacovigilance analysis, semaglutide but not tirzepatide demonstrated a disproportionality signal for AKI. While spontaneous reporting cannot establish causality, these findings suggest a possible renal safety distinction between the two agents These findings support close renal monitoring, particularly when initiating semaglutide in higher-risk patients, and highlight the need for confirmation in well-designed epidemiologic studies.
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lee, J.; Liu, J.-J.; Liu, S.; Liu, A.; Zheng, H.; Chan, C.; Shao, Y.M.; Gurung, R.L.; Ang, K.; Lim, S.C. Acute kidney injury predicts the risk of adverse cardio renal events and all cause death in southeast Asian people with type 2 diabetes. Scientific Reports 2024, 14, 27027. [Google Scholar] [CrossRef] [PubMed]
- Caruso, I.; Giorgino, F. Renal effects of GLP-1 receptor agonists and tirzepatide in individuals with type 2 diabetes: seeds of a promising future. Endocrine 2024, 84, 822–835. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Sun, C. Can glucagon-like peptide-1 receptor agonists cause acute kidney injury? An analytical study based on post-marketing approval pharmacovigilance data. Front Endocrinol (Lausanne) 2022, 13, 1032199. [Google Scholar] [CrossRef] [PubMed]
- Begum, F.; Chang, K.; Kapoor, K.; Vij, R.; Phadke, G.; Hiser, W.M.; Wanchoo, R.; Sharma, P.; Sutaria, N.; Jhaveri, K.D. Semaglutide-associated kidney injury. Clin Kidney J 2024, 17, sfae250. [Google Scholar] [CrossRef] [PubMed]
- Komala, M.G.; Renthawa, J. A Case of Dulaglutide-Induced Acute Interstitial Nephritis After Many Years of Treatment With an Alternate GLP-1 Receptor Agonist. Clin Diabetes 2022, 40, 508–510. [Google Scholar] [CrossRef]
- Leehey, D.J.; Rahman, M.A.; Borys, E.; Picken, M.M.; Clise, C.E. Acute Kidney Injury Associated With Semaglutide. Kidney Med 2021, 3, 282–285. [Google Scholar] [CrossRef]
- Borkum, M.; Lau, W.; Blanco, P.; Farah, M. Semaglutide-Associated Acute Interstitial Nephritis: A Case Report. Kidney Medicine 2022, 4. [Google Scholar] [CrossRef]
- Sorli, C.; Harashima, S.I.; Tsoukas, G.M.; Unger, J.; Karsbøl, J.D.; Hansen, T.; Bain, S.C. Efficacy and safety of once-weekly semaglutide monotherapy versus placebo in patients with type 2 diabetes (SUSTAIN 1): a double-blind, randomised, placebo-controlled, parallel-group, multinational, multicentre phase 3a trial. Lancet Diabetes Endocrinol 2017, 5, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Aroda, V.R.; Erhan, U.; Jelnes, P.; Meier, J.J.; Abildlund, M.T.; Pratley, R.; Vilsbøll, T.; Husain, M. Safety and tolerability of semaglutide across the SUSTAIN and PIONEER phase IIIa clinical trial programmes. Diabetes Obes Metab 2023, 25, 1385–1397. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.L.; Sattar, N.; Pavo, I.; Haupt, A.; Duffin, K.L.; Yang, Z.; Wiese, R.J.; Tuttle, K.R.; Cherney, D.Z.I. Effects of tirzepatide versus insulin glargine on kidney outcomes in type 2 diabetes in the SURPASS-4 trial: post-hoc analysis of an open-label, randomised, phase 3 trial. Lancet Diabetes Endocrinol 2022, 10, 774–785. [Google Scholar] [CrossRef]
- Li, J.; Xie, J.; Han, Y.; Zhang, W.; Wang, Y.; Jiang, Z. A real-world disproportionality analysis of tirzepatide-related adverse events based on the FDA Adverse Event Reporting System (FAERS) database. Endocr J 2025, 72, 273–283. [Google Scholar] [CrossRef]
- Sakaeda, T.; Tamon, A.; Kadoyama, K.; Okuno, Y. Data mining of the public version of the FDA Adverse Event Reporting System. Int J Med Sci 2013, 10, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.J.; Waller, P.C.; Davis, S. Use of proportional reporting ratios (PRRs) for signal generation from spontaneous adverse drug reaction reports. Pharmacoepidemiol Drug Saf 2001, 10, 483–486. [Google Scholar] [CrossRef] [PubMed]
- Hartnell, N.R.; Wilson, J.P. Replication of the Weber effect using postmarketing adverse event reports voluntarily submitted to the United States Food and Drug Administration. Pharmacotherapy 2004, 24, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Feier, C.V.I.; Vonica, R.C.; Faur, A.M.; Streinu, D.R.; Muntean, C. Assessment of Thyroid Carcinogenic Risk and Safety Profile of GLP1-RA Semaglutide (Ozempic) Therapy for Diabetes Mellitus and Obesity: A Systematic Literature Review. International Journal of Molecular Sciences 2024, 25, 4346. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.F.E.; Hansen, T.; Idorn, T.; Leiter, L.A.; Marso, S.P.; Rossing, P.; Seufert, J.; Tadayon, S.; Vilsbøll, T. Effects of once-weekly subcutaneous semaglutide on kidney function and safety in patients with type 2 diabetes: a post-hoc analysis of the SUSTAIN 1-7 randomised controlled trials. Lancet Diabetes Endocrinol 2020, 8, 880–893. [Google Scholar] [CrossRef] [PubMed]
- Oesterle, T.S.; Ho, M.-F. Glucagon-like Peptide-1 Receptor Agonists: A New Frontier in Treating Alcohol Use Disorder. Brain Sciences 2025, 15, 702. [Google Scholar] [CrossRef] [PubMed]
- Rosenstock, J.; Fonseca, V.A.; Gross, J.L.; Ratner, R.E.; Ahrén, B.; Chow, F.C.; Yang, F.; Miller, D.; Johnson, S.L.; Stewart, M.W.; et al. Advancing basal insulin replacement in type 2 diabetes inadequately controlled with insulin glargine plus oral agents: a comparison of adding albiglutide, a weekly GLP-1 receptor agonist, versus thrice-daily prandial insulin lispro. Diabetes Care 2014, 37, 2317–2325. [Google Scholar] [CrossRef] [PubMed]
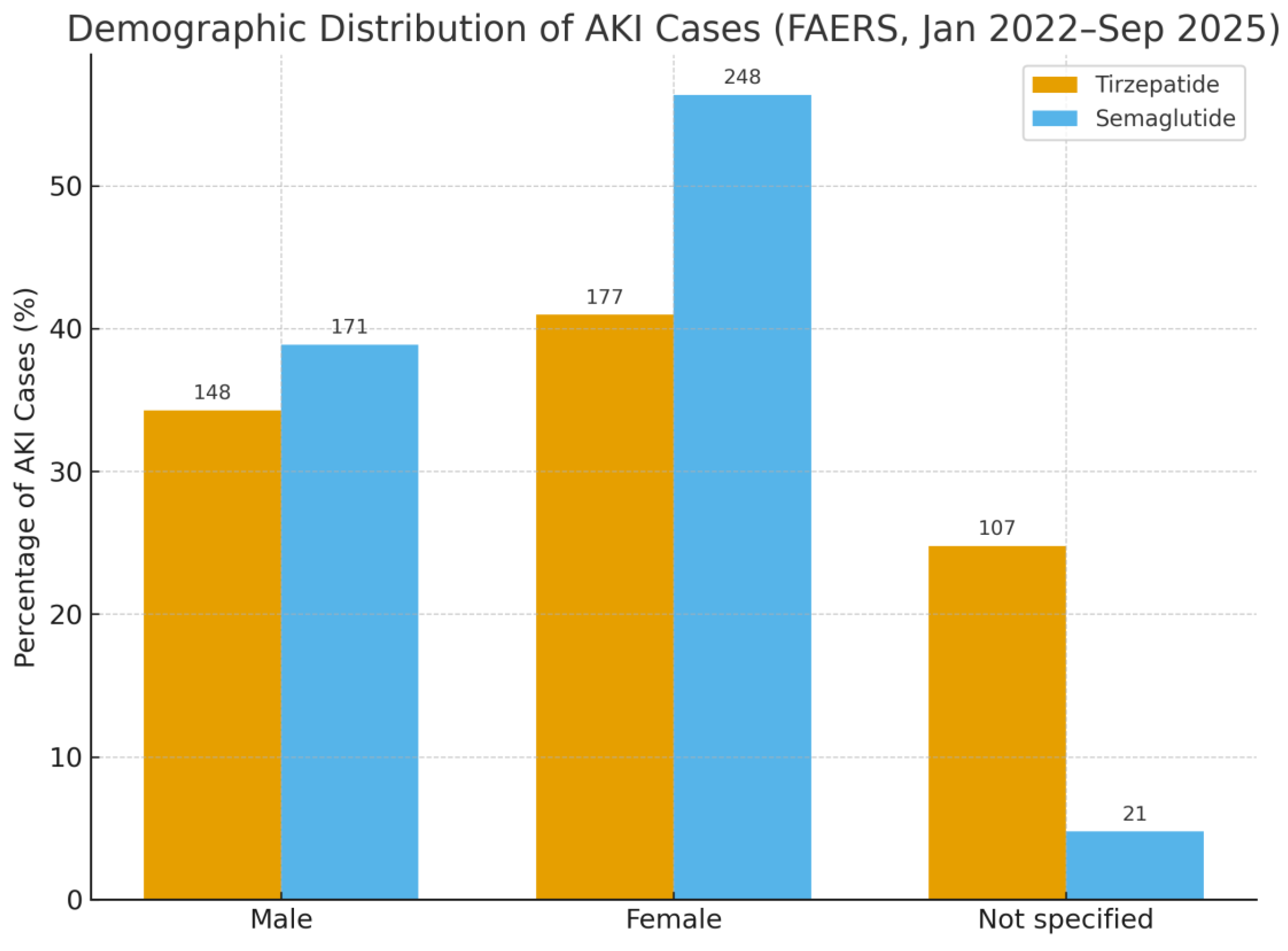
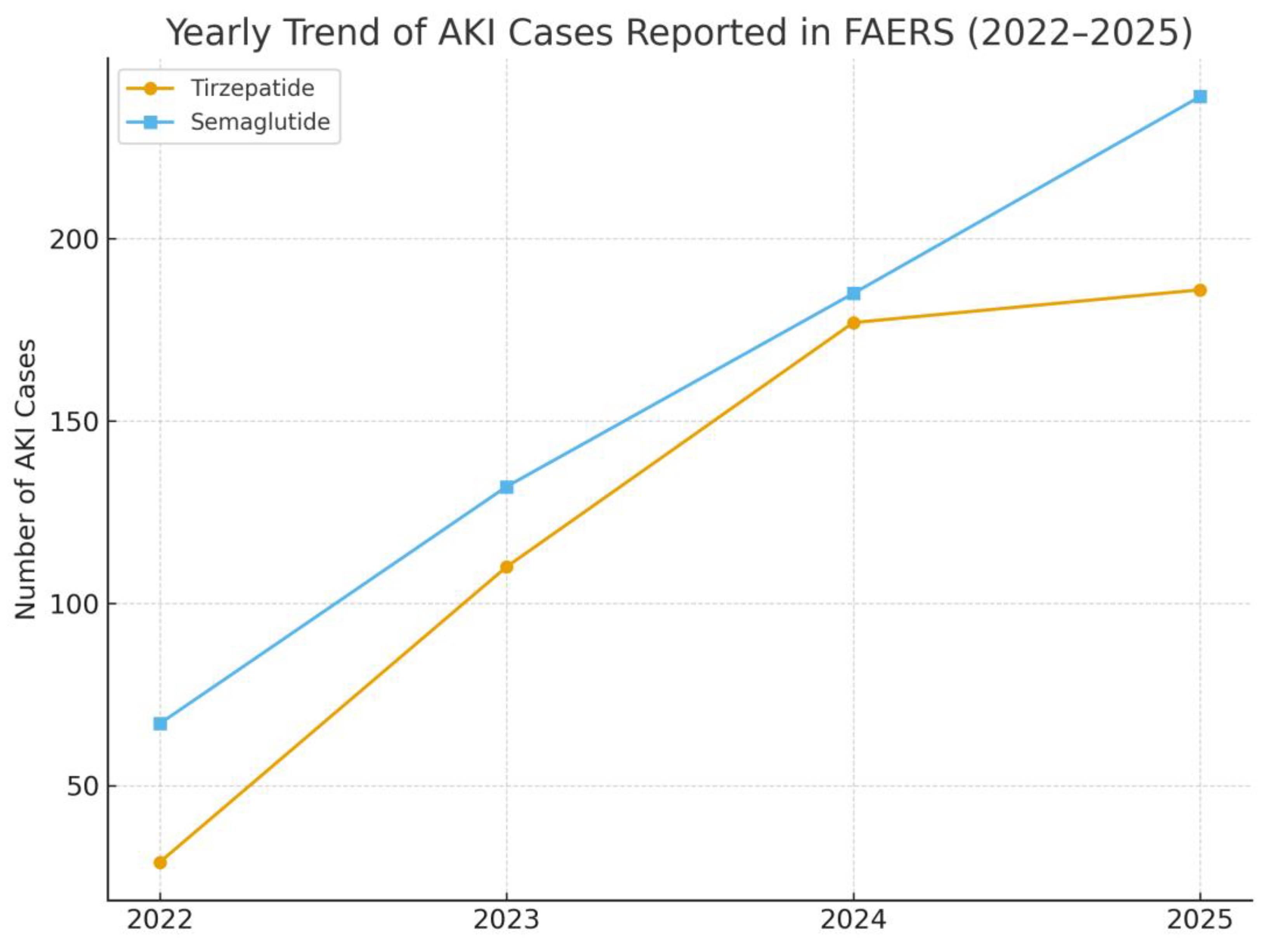
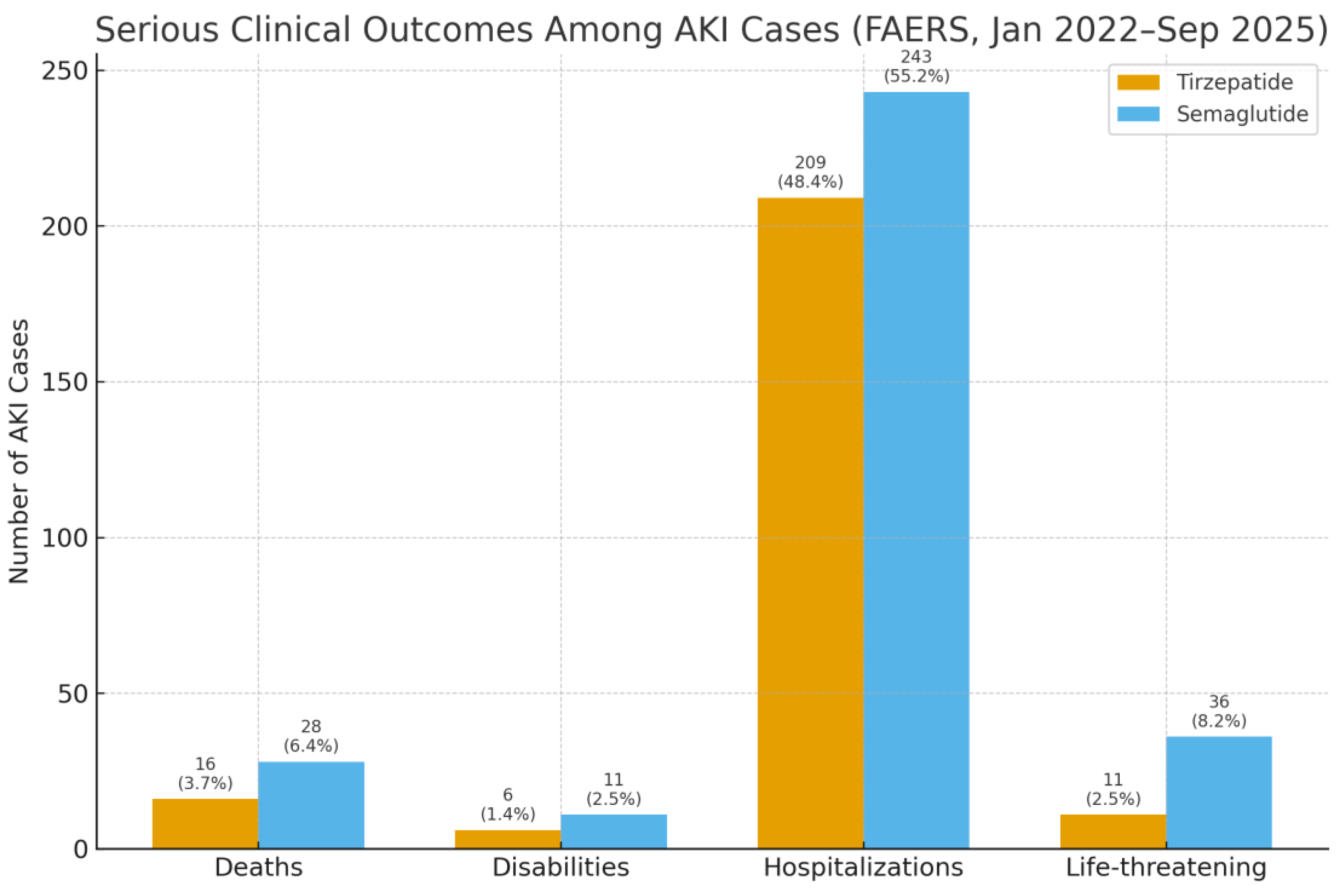
| Drug | Total AEs | AKI cases | AKI rate (%) | ROR (95% CI) | PRR |
|---|---|---|---|---|---|
| Tirzepatide | 92,807 | 432 | 0.47 | 0.44 (0.38–0.50) | 0.44 |
| Semaglutide | 41,065 | 440 | 1.07 | Reference | Ref |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




