Submitted:
09 September 2025
Posted:
10 September 2025
You are already at the latest version
Abstract
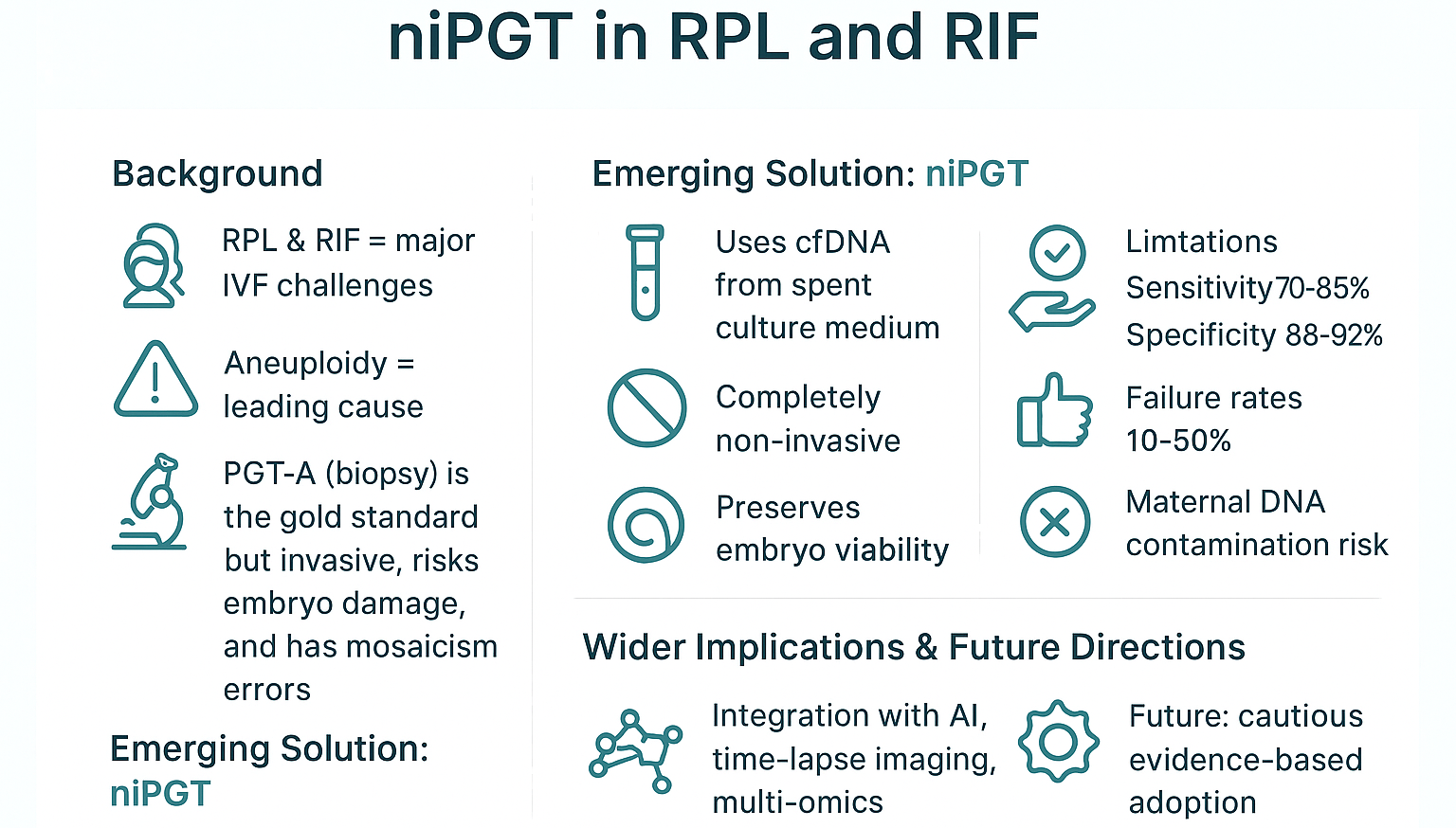
Background: Recurrent pregnancy loss (RPL) and recurrent implantation failure (RIF) are significant challenges in reproductive medicine. For both, embryonic aneuploidy is the leading etiological factor. Preimplantation genetic testing for aneuploidy (PGT-A) via trophectoderm biopsy is the current standard for embryo selection. However, it is limited by its invasiveness, potential for embryo damage, and diagnostic errors due to mosaicism. Rationale/Objectives: This review critically evaluates the emerging role of noninvasive PGT (niPGT). NiPGT analyzes cell-free DNA from spent blastocyst culture media, thus is a potential alternative for managing RPL and RIF. Hence, the primary objective is to determine whether current evidence supports niPGT as a reliable replacement for conventional biopsy-based PGT-A in these high-risk populations. Outcomes: The analysis reveals that niPGT offers significant theoretical advantages. These include complete non-invasiveness, enhanced embryo preservation, and high patient acceptability. However, its clinical application is hampered by substantial limitations. Key amongst them is the inconsistent and often suboptimal diagnostic accuracy (sensitivity 70-85%, specificity 88-92%) compared to biopsy. Other significant factors include the high rates of amplification failure (10-50%), vulnerability to maternal DNA contamination, as well as low DNA yield. Crucially, there is a definitive lack of robust, prospective randomized controlled trial (RCT) data demonstrating improved live birth rates or reduced miscarriage rates specifically in RPL and RIF cohorts. As, niPGT is not yet ready to be a standalone clinical adoption in RPL and RIF cases. However, it may serve as a valuable adjunct for rescue scenarios following biopsy failure or for ethical reasons. Wider Implications: The integration of niPGT with artificial intelligence, time-lapse imaging, and multi-omics profiling underlies a promising future. However, its transition from a predominantly research tool to a clinical standard necessitates various critical undertakings. These include rigorous multicenter RCTs, standardizing international protocol, and tailoring validation for the RPL and RIF subgroups. This review highlights the need for cautious optimism, positing that evidence-based integration, rather than premature adoption, is essential to realizing niPGT’s full potential without compromising patient care in these complex fertility scenarios.
Keywords:
Noninvasive Preimplantation Genetic Testing in Recurrent Pregnancy Loss and Implantation Failure: Breakthrough or Overpromise?
Introduction
1. Understanding RPL and RIF: Clinical Definitions and Burden
1.1. Definitions and Incidence
1.2. Common Etiologies
1.3. The Challenge of Idiopathic Cases
1.4. Psychological and Financial Toll
2. The Role of Embryonic Aneuploidy in RPL and RIF
2.1. Understanding Aneuploidy: Definition and Types
2.2. Aneuploidy as a Primary Cause of Implantation Failure and Miscarriage
2.3. Impact of Maternal Age and Diminished Ovarian Reserve (DOR)
2.4. Current Embryo Selection Strategies (Comparative Table)
2.5. Controversies
2.6. Evidence (Create an Image)
2.7. Summary
3. Invasive PGT-A: Current Standard and Its Limitations
3.1. Background – Purpose and Types (Images)
3.2. Common Techniques: Trophectoderm Biopsy and Blastocyst Stage Analysis (Images)
3.3. Benefits: Improved Selection and Reduced Miscarriage Rates
3.4. Limitations
3.5. Summary
4. Noninvasive PGT (niPGT): Science and Methodologies (Image)
4.1. Sources of Cell-Free DNA: Blastocoel Fluid vs. Spent Culture Medium (Differences Table)
4.2. Extraction, Amplification, and Sequencing Methods
4.3. Current Techniques
4.4. Challenges
4.5. Summary
5. Clinical Evidence: Can niPGT Replace Biopsy?
5.1. Key Studies and Meta-Analyses
5.2. Sensitivity and Specificity Compared to PGT-A
5.3. Real-World Success Rates in RPL/RIF Subpopulations
5.4. Cases Where niPGT Changed Clinical Management
5.5. Summary
6. Application in RPL/RIF: Hope or Hype?
6.1. Benefits: The Foundations of Hope
6.2. Drawbacks: The Reality Check
6.3. How IVF Centers Are Currently Using or Trialing niPGT
6.4. Integration with Time-Lapse, Morphology, and AI
6.5. Conclusion: Cautious Optimism Amid Significant Hurdles
7. Future Perspectives and Clinical Recommendations
7.1. Need for Prospective, Multicenter RCTs
7.2. Combining niPGT with Epigenetic and Metabolomic Profiling
7.3. Standardization of Protocols and Reporting
7.4. Tailored Approaches for Patient Subgroups
8. Conclusion: A Roadmap for Responsible Innovation
8.1. Reiterating the Promise and the Prerequisites
8.2. Confronting the Current Reality: Barriers to Clinical Adoption
8.3. Imperative Call for Cautious Integration and Further Research
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data availability statement
Conflicts of Interest
Abbreviations
| Abbreviation | Full Form |
| ADO | Allele Dropout |
| AI | Artificial Intelligence |
| AMA | Advanced Maternal Age |
| AMH | Anti-Müllerian Hormone |
| ANXA5 | Annexin A5 |
| APS | Antiphospholipid Syndrome |
| ART | Assisted Reproductive Technology |
| BF | Blastocoel Fluid |
| cfDNA | Cell-Free DNA |
| CLBR | Cumulative Live Birth Rate |
| DOR | Diminished Ovarian Reserve |
| EQC | External Quality Controls |
| ESHRE | European Society of Human Reproduction and Embryology |
| HLA | Human Leukocyte Antigen |
| ICM | Inner Cell Mass |
| IR | Implantation Rate |
| ISO | International Organization for Standardization |
| LBR | Live Birth Rate |
| LP-WGS | Low-Pass Whole Genome Sequencing |
| MDA | Multiple Displacement Amplification |
| NGS | Next-Generation Sequencing |
| niPGT | Noninvasive Preimplantation Genetic Testing |
| niPGT-A | Noninvasive Preimplantation Genetic Testing for Aneuploidy |
| niPGT-M | Noninvasive Preimplantation Genetic Testing for Monogenic disorders |
| niPGT-SR | Noninvasive Preimplantation Genetic Testing for Structural Rearrangements |
| NK cells (uNKs) | Natural Killer cells (Uterine Natural Killer cells) |
| NR | No Result |
| PCOS | Polycystic Ovary Syndrome |
| PGT | Preimplantation Genetic Testing |
| PGT-A | Preimplantation Genetic Testing for Aneuploidy |
| PGT-M | Preimplantation Genetic Testing for Monogenic disorders |
| PGT-SR | Preimplantation Genetic Testing for Structural Rearrangements |
| PPV/NPV | Positive Predictive Value / Negative Predictive Value |
| qPCR | Quantitative Polymerase Chain Reaction |
| RCT | Randomized Controlled Trial |
| RIF | Recurrent Implantation Failure |
| RPL | Recurrent Pregnancy Loss |
| SCM | Spent Culture Medium |
| SET | Single Embryo Transfer |
| STARD-PGT | Standards for Reporting Diagnostic Accuracy Studies - PGT |
| STR | Short Tandem Repeat |
| TE | Trophectoderm |
| TLI | Time-Lapse Imaging |
| Treg | Regulatory T cell |
| TSH | Thyroid-Stimulating Hormone |
| WGA | Whole Genome Amplification |
| WGS | Whole Genome Sequencing |
References
- Asghari, K.M.; Novinbahador, T.; Mehdizadeh, A.; Zolfaghari, M.; Yousefi, M. Revolutionized attitude toward recurrent pregnancy loss and recurrent implantation failure based on precision regenerative medicine. Heliyon 2024, 10. [Google Scholar]
- Cao, C.; Bai, S.; Zhang, J.; Sun, X.; Meng, A.; Chen, H. Understanding recurrent pregnancy loss: recent advances on its etiology, clinical diagnosis, and management. Medical Review 2023, 2, 570–589. [Google Scholar] [CrossRef] [PubMed]
- Barad, D.H.; Albertini, D.F.; Molinari, E.; Gleicher, N. IVF outcomes of embryos with abnormal PGT-A biopsy previously refused transfer: a prospective cohort study. Human Reproduction 2022, 37, 1194–1206. [Google Scholar] [CrossRef]
- Busnelli, A.; Somigliana, E.; Cirillo, F.; Baggiani, A.; Levi-Setti, P.E. Efficacy of therapies and interventions for repeated embryo implantation failure: a systematic review and meta-analysis. Scientific Reports 2021, 11, 1747. [Google Scholar] [CrossRef]
- Bakalova, D.N.; Navarro-Sánchez, L.; Rubio, C. (2025). Non-Invasive Preimplantation Genetic Testing.
- Capalbo, A.; Wells, D. The evolution of preimplantation genetic testing: where is the limit? Reproductive BioMedicine Online 2025, 50, 104845. [Google Scholar] [CrossRef]
- Cavalcante, M.B.; Sarno, M.; Barini, R. Immune biomarkers in cases of recurrent pregnancy loss and recurrent implantation failure. Minerva obstetrics and gynecology 2025, 77, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Hu, Z.; Lian, Y.; Han, Y.; Zhou, X.; Li, Y.; Xiang, L.; Jiang, W.; Li, M.; Zeng, P.; et al. The diagnostic accuracy of preimplantation genetic testing (PGT) in assessing the genetic status of embryos: a systematic review and meta-analysis. Reproductive Biology and Endocrinology 2025, 23, 39. [Google Scholar] [CrossRef]
- Chen, S.; Wang, L.; Hu, Y.; Yao, Y.; Gao, F.; Chang, C.; Zhang, L.; Huang, H.; Lu, D.; Xu, C. Noninvasive preimplantation genetic testing for aneuploidy using blastocyst spent culture medium may serve as a backup of trophectoderm biopsy in conventional preimplantation genetic testing. BMC Medical Genomics 2025, 18, 34. [Google Scholar] [CrossRef]
- Cheng, H.Y.H.; Chow, J.F.; Lam, K.K.; Lai, S.F.; Yeung, W.S.B.; Ng, E.H. Randomised double-blind controlled trial of non-invasive preimplantation genetic testing for aneuploidy in in vitro fertilisation: a protocol paper. BMJ open 2023, 13, e072557. [Google Scholar] [CrossRef]
- Chow, J.F.; Lam, K.K.; Cheng, H.H.; Lai, S.F.; Yeung, W.S.; Ng, E.H. Optimizing non-invasive preimplantation genetic testing: investigating culture conditions, sample collection, and IVF treatment for improved non-invasive PGT-A results. Journal of Assisted Reproduction and Genetics 2024, 41, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Nagori, C.; Panchal, S. (2022). Practical Guide to Recurrent Pregnancy Loss. Jaypee Brothers Medical Publishers.
- Ma, J.; Gao, W.; Li, D. Recurrent implantation failure: A comprehensive summary from etiology to treatment. Frontiers in Endocrinology 2023, 13, 1061766. [Google Scholar] [CrossRef]
- Sheikhansari, G.; Pourmoghadam, Z.; Danaii, S.; Mehdizadeh, A.; Yousefi, M. Etiology and management of recurrent implantation failure: a focus on intra-uterine PBMC-therapy for RIF. Journal of reproductive immunology 2020, 139, 103121. [Google Scholar] [CrossRef]
- Wang, C.; Lu, Y.; Ou, M.; Qian, L.; Zhang, Y.; Yang, Y.; Luo, L.; Wang, Q. Risk factors for recurrent implantation failure as defined by the European Society for Human Reproduction and Embryology. Human Reproduction 2025, deaf042. [Google Scholar] [CrossRef]
- Gonçalves, B.D.O. (2022). Aetiopathogenesis of Recurrent implantation failure after assisted reproductive techniques (Master's thesis).
- Linehan, L.; Hennessy, M.; O'Donoghue, K. Infertility and subsequent recurrent miscarriage: current state of the literature and future considerations for practice and research. HRB Open Research 2021, 4, 100. [Google Scholar] [CrossRef]
- Cuadrado-Torroglosa, I.; García-Velasco, J.A.; Alecsandru, D. Maternal--Fetal Compatibility in Recurrent Pregnancy Loss. Journal of Clinical Medicine 2024, 13, 2379. [Google Scholar] [CrossRef]
- Yang, X.; Tian, Y.; Zheng, L.; Luu, T.; Kwak-Kim, J. The update immune-regulatory role of pro-and anti-inflammatory cytokines in recurrent pregnancy losses. International journal of molecular sciences 2022, 24, 132. [Google Scholar] [CrossRef] [PubMed]
- Garmendia, J.V.; De Sanctis, C.V.; Hajdúch, M.; De Sanctis, J.B. Microbiota and recurrent pregnancy loss (RPL); more than a simple connection. Microorganisms 2024, 12, 1641. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; He, Z.; Ma, R.; Lin, N.; Li, L.; Li, Y.; Li, Y.; Ke, Y.; Meng, X.; Wu, Z. Narrative review of multifaceted approaches to managing recurrent implantation failure: insights and innovations. Clinical and Experimental Obstetrics Gynecology 2024, 51, 87. [Google Scholar] [CrossRef]
- Ebrahimi, F.; Omidvar-Mehrabadi, A.; Shahbazi, M.; Mohammadnia-Afrouzi, M. Innate and adaptive immune dysregulation in women with recurrent implantation failure. Journal of Reproductive Immunology 2024, 164, 104262. [Google Scholar] [CrossRef] [PubMed]
- Lash, G.E.; Raghupathy, R. Immune involvement in recurrent pregnancy loss. Frontiers in Immunology 2023, 14, 1330701. [Google Scholar] [CrossRef]
- Sfakianoudis, K.; Rapani, A.; Grigoriadis, S.; Pantou, A.; Maziotis, E.; Kokkini, G.; Tsirligkani, C.; Bolaris, S.; Nikolettos, K.; Chronopoulou, M.; et al. The role of uterine natural killer cells on recurrent miscarriage and recurrent implantation failure: from pathophysiology to treatment. Biomedicines 2021, 9, 1425. [Google Scholar] [CrossRef] [PubMed]
- Garmendia, J.V.; De Sanctis, C.V.; Hajdúch, M.; De Sanctis, J.B. Exploring the Immunological Aspects and Treatments of Recurrent Pregnancy Loss and Recurrent Implantation Failure. International Journal of Molecular Sciences 2025, 26, 1295. [Google Scholar] [CrossRef]
- Mukherjee, N.; Sharma, R.; Modi, D. Immune alterations in recurrent implantation failure. American Journal of Reproductive Immunology 2023, 89, e13563. [Google Scholar] [CrossRef]
- George, J.S.; Mortimer, R.; Anchan, R.M. (2022). The role of reproductive immunology in recurrent pregnancy loss and repeated implantation failure. In Immunology of Recurrent Pregnancy Loss and Implantation Failure (pp. 223-240). Academic Press.
- Nakagawa, K.; Kuroda, K.; Sugiyama, R. Helper T cell pathology and repeated implantation failures. Immunology of Recurrent Pregnancy Loss and Implantation Failure 2022, 273–285. [Google Scholar]
- Uța, C.; Tîrziu, A.; Zimbru, E.L.; Zimbru, R.I.; Georgescu, M.; Haidar, L.; Panaitescu, C. Alloimmune Causes of Recurrent Pregnancy Loss: Cellular Mechanisms and Overview of Therapeutic Approaches. Medicina 2024, 60, 1896. [Google Scholar] [CrossRef]
- Montazeri, F.; Tajamolian, M.; Hosseini, E.S.; Hoseini, S.M. Immunologic Factors and Genomic Considerations in Recurrent Pregnancy Loss: A Review. International Journal of Medical Laboratory 2023. [Google Scholar] [CrossRef]
- Davalieva, K.; Kocarev, D.; Plaseska-Karanfilska, D. Decoding recurrent pregnancy loss: insights from comparative proteomics studies. Biology of Reproduction 2025, 112, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Liaqat Ali Khan, N.; Nafee, T.; Shao, T.; Hart, A.R.; Elliott, S.; Ola, B.; Heath, P.R.; Fazeli, A. Dysregulation in multiple transcriptomic endometrial pathways is associated with recurrent implantation failure and recurrent early pregnancy loss. International Journal of Molecular Sciences 2022, 23, 16051. [Google Scholar] [CrossRef]
- Nørgaard-Pedersen, C. (2023). Recurrent Pregnancy Loss: immunological risk factors and immunomodulatory treatments.
- Zeng, Y.; Tu, W. Predicting Pregnancy Outcomes in the Recurrent Reproductive Failure. Immune Regulations in Reproductive Organs and Organ Transplant 2022, 18, 64216768. [Google Scholar]
- Sato, T.; Sugiura-Ogasawara, M.; Ozawa, F.; Yamamoto, T.; Kato, T.; Kurahashi, H.; Kuroda, T.; Aoyama, N.; Kato, K.; Kobayashi, R.; Fukuda, A.; Utsunomiya, T.; Kuwahara, A.; Saito, H.; Takeshita, T.; Irahara, M. Preimplantation genetic testing for aneuploidy: a comparison of live birth rates in patients with recurrent pregnancy loss due to embryonic aneuploidy or recurrent implantation failure. Human reproduction (Oxford, England) 2019, 34, 2340–2348. [Google Scholar] [CrossRef]
- Wang, X.; Chen, B.; Fang, L.; Wang, J.; Xu, A.; Xu, W.; Tong, X. Aneuploidy rates and clinical pregnancy outcomes after preimplantation genetic testing for aneuploidy using the progestin-primed ovarian stimulation protocol or the gonadotropin-releasing hormone antagonist protocol. Journal of Gynecology Obstetrics and Human Reproduction 2025, 54, 102883. [Google Scholar] [CrossRef] [PubMed]
- Lei, C.X.; Ye, J.F.; Sui, Y.L.; Zhang, Y.P.; Sun, X.X. Retrospective cohort study of preimplantation genetic testing for aneuploidy with comprehensive chromosome screening versus nonpreimplantation genetic testing in normal karyotype, secondary infertility patients with recurrent pregnancy loss. Reproductive and Developmental Medicine 2019, 3, 205–212. [Google Scholar] [CrossRef]
- Kato, K.; Kuroda, T.; Yamadera-Egawa, R.; Ezoe, K.; Aoyama, N.; Usami, A.; Miki, T.; Yamamoto, T.; Takeshita, T. Preimplantation genetic testing for aneuploidy for recurrent pregnancy loss and recurrent implantation failure in minimal ovarian stimulation cycle for women aged 35--42 years: live birth rate, developmental follow-up of children, and embryo ranking. Reproductive Sciences 2023, 30, 974–983. [Google Scholar] [CrossRef] [PubMed]
- Du, R.Q.; Zhao, D.D.; Kang, K.; Wang, F.; Xu, R.X.; Chi, C.L.; Kong, L.Y.; Liang, B. A review of pre-implantation genetic testing technologies and applications. Reproductive and Developmental Medicine 2023, 7, 20–31. [Google Scholar] [CrossRef]
- Wang, S.; Liu, L.; Ma, M.; Wang, H.; Han, Y.; Guo, X.; Yeung, W.S.; Cheng, Y.; Zhang, H.; Dong, F.; et al. Preimplantation genetic testing for aneuploidy helps to achieve a live birth with fewer transfer cycles for the blastocyst FET patients with unexplained recurrent implantation failure. Archives of Gynecology and Obstetrics 2023, 308, 599–610. [Google Scholar] [CrossRef]
- Du, Y.; Guan, Y.; Li, N.; Shi, C.; Zhang, Y.; Ren, B.; Liu, J.; Lou, H. Is it necessary for young patients with recurrent implantation failure to undergo preimplantation genetic testing for aneuploidy? Frontiers in Endocrinology 2023, 14, 1020055. [Google Scholar] [CrossRef]
- Guan, H.; He, Y.; Lu, Y.; Huang, J.; Wang, Y.; Zhu, Q.; Qi, J.; Lin, W.; Lindheim, S.R.; Wei, Z.; et al. Effect of Preimplantation Genetic Testing for Aneuploidy on Live Birth Rate in Young Women With Recurrent Implantation Failure: A Secondary Analysis of a Multicentre Randomised Trial. BJOG: An International Journal of Obstetrics Gynaecology 2024. [Google Scholar] [CrossRef]
- Kim, J.W.; Lee, S.Y.; Hur, C.Y.; Lim, J.H. P-507 Prediction of clinical efficacy of PGT-A based on maternal age and embryo quality in patients with recurrent implantation failure (RIF) or recurrent pregnancy loss (RPL). Human Reproduction 2024, 39(Supplement_1), deae108-847. [Google Scholar] [CrossRef]
- Kim, J.W.; Lee, S.Y.; Hur, C.Y.; Lim, J.H.; Park, C.K. Clinical outcomes of preimplantation genetic testing for aneuploidy in high-risk patients: a retrospective cohort study. Clinical and Experimental Reproductive Medicine 2023, 51, 75. [Google Scholar] [CrossRef]
- Kornfield, M.S.; Parker, P.; Rubin, E.; Garg, B.; O'Leary, T.; Amato, P.; Lee, D.; Wu, D.; Krieg, S. Patients with Recurrent Pregnancy Loss Have Similar Embryonic Preimplantation Genetic Testing Aneuploidy Rates and In Vitro Fertilization Outcomes to Infertility Patients. F&S Reports 2022, 3, 342–348. [Google Scholar] [CrossRef]
- Liang, Z.; Wen, Q.; Li, J.; Zeng, D.; Huang, P. A systematic review and meta-analysis: clinical outcomes of recurrent pregnancy failure resulting from preimplantation genetic testing for aneuploidy. Frontiers in Endocrinology 2023, 14, 1178294. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lan, X.; Lu, J.; Zhang, Q.; Zhou, T.; Ni, T.; Yan, J. Preimplantation genetic testing for aneuploidy could not improve cumulative live birth rate among 705 couples with unexplained recurrent implantation failure. The Application of Clinical Genetics 2024, 1–13. [Google Scholar] [CrossRef]
- Mei, Y.; Lin, Y.; Chen, Y.; Zheng, J.; Ke, X.; Liang, X.; Wang, F. Preimplantation genetic testing for aneuploidy optimizes reproductive outcomes in recurrent reproductive failure: a systematic review. Frontiers in Medicine 2024, 11, 1233962. [Google Scholar] [CrossRef] [PubMed]
- Mantravadi, K.; Mathew, S.; Soorve, S.; Rao, D.G.; Karunakaran, S. Does pre-implantation genetic testing for aneuploidy optimise the reproductive outcomes in women with idiopathic recurrent pregnancy loss? Fertility and Sterility 2020, 114, e437. [Google Scholar] [CrossRef]
- Sarkar, P.; Jindal, S.; New, E.P.; Sprague, R.G.; Tanner, J.; Imudia, A.N. The role of preimplantation genetic testing for aneuploidy in a good prognosis IVF population across different age groups. Systems Biology in Reproductive Medicine 2021, 67, 366–373. [Google Scholar] [CrossRef]
- Orvieto, R. Does preimplantation genetic testing for aneuploidy really improve IVF outcomes in advanced maternal age patients without compromising cumulative live-birth rate? Journal of Assisted Reproduction and Genetics 2020, 37, 159–159. [Google Scholar] [CrossRef]
- Kasaven, L.S.; Marcus, D.; Theodorou, E.; Jones, B.P.; Saso, S.; Naja, R.; Serhal, P.; Ben-Nagi, J. Systematic review and meta-analysis: does pre-implantation genetic testing for aneuploidy at the blastocyst stage improve live birth rate? Journal of Assisted Reproduction and Genetics 2023, 40, 2297–2316. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Bogale, B.; Tang, Y.; Lu, S.; Xie, X.S.; Racowsky, C. Noninvasive preimplantation genetic testing for aneuploidy in spent medium may be more reliable than trophectoderm biopsy. Proceedings of the National Academy of Sciences 2019, 116, 14105–14112. [Google Scholar] [CrossRef]
- Giuliano, R.; Maione, A.; Vallefuoco, A.; Sorrentino, U.; Zuccarello, D. Preimplantation genetic testing for genetic diseases: limits and review of current literature. Genes 2023, 14, 2095. [Google Scholar] [CrossRef]
- Kakourou, G.; Sofocleous, C.; Mamas, T.; Vrettou, C.; Traeger-Synodinos, J. The current clinical applications of preimplantation genetic testing (PGT): acknowledging the limitations of biology and technology. Expert Review of Molecular Diagnostics 2024, 24, 767–775. [Google Scholar] [CrossRef]
- Morales, C. Current applications and controversies in preimplantation genetic testing for aneuploidies (PGT-A) in in vitro fertilization. Reproductive Sciences 2024, 31, 66–80. [Google Scholar] [CrossRef]
- Viville, S.; Aboulghar, M. PGT-A: what's it for, what's wrong? Journal of Assisted Reproduction and Genetics 2025, 42, 63–69. [Google Scholar] [CrossRef]
- Phillips, K.R.; Kuzma-Hunt, A.G.; Neal, M.S.; Lisle, C.; Sribalachandran, H.; Carter, R.F.; Amin, S.; Karnis, M.F.; Faghih, M. Temporal Evaluation of a Minimally Invasive Method of Preimplantation Genetic Testing for Aneuploidy (mi-PGT-A) in Human Embryos. Reproductive Medicine 2024, 5. [Google Scholar] [CrossRef]
- He, F.; Yao, Y.X.; Wang, J.; Zhao, D.M.; Wan, A.Q.; Ren, J.; Lei, X. Non-invasive chromosome screening for embryo preimplantation using cell-free DNA. Reproductive and Developmental Medicine 2022, 6, 113–120. [Google Scholar] [CrossRef]
- Del Collado, M.; Andrade, G.M.; Gonçalves, N.J.N.; Fortini, S.; Perecin, F.; Carriero, M.M. The embryo non-invasive pre-implantation diagnosis era: how far are we? Animal Reproduction 2023, 20, e20230069. [Google Scholar] [CrossRef]
- Shitara, A.; Takahashi, K.; Goto, M.; Takahashi, H.; Iwasawa, T.; Onodera, Y.; Makino, K.; Miura, H.; Shirasawa, H.; Sato, W.; et al. Cell-free DNA in spent culture medium effectively reflects the chromosomal status of embryos following culturing beyond implantation compared to trophectoderm biopsy. PLoS One 2021, 16, e0246438. [Google Scholar] [CrossRef]
- Sialakouma, A.; Karakasiliotis, I.; Ntala, V.; Nikolettos, N.; Asimakopoulos, B. Embryonic cell-free DNA in spent culture medium: a non-invasive tool for aneuploidy screening of the corresponding embryos. in vivo 2021, 35, 3449–3457. [Google Scholar] [CrossRef]
- Xie, P.; Zhang, S.; Gu, Y.; Jiang, B.; Hu, L.; Tan, Y.Q.; Yao, Y.; Tang, Y.; Wan, A.; Cai, S.; et al. Non-invasive preimplantation genetic testing for conventional IVF blastocysts. Journal of Translational Medicine 2022, 20, 396. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, B.R. Great Challenges Remain for niPGT-A Reliability. Revista Brasileira de Ginecologia e Obstetrícia/RBGO Gynecology and Obstetrics 2022, 44, 721–722. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Li, Y.; Chen, K.; Li, M.; Tian, M.; Xiang, L.; Wu, X.; Zeng, P.; Li, M.; Shao, J.; et al. The comparison of two whole-genome amplification approaches for noninvasive preimplantation genetic testing (ni-PGT) and the application scenario of ni-PGT during the fresh cycle. The Journal of Molecular Diagnostics 2023, 25, 945–956. [Google Scholar] [CrossRef] [PubMed]
- Karami, N.; Iravani, F.; Bavarsad, S.B.; Asadollahi, S.; Hoseini, S.M.; Montazeri, F.; Kalantar, S.M. Comparing the advantages, disadvantages and diagnostic power of different non-invasive pre-implantation genetic testing: A literature review. International Journal of Reproductive BioMedicine 2024, 22, 177. [Google Scholar] [CrossRef]
- Sousa, L.N.; Monteiro, P.B. Non-invasive preimplantation genetic testing: a literature review. JBRA Assisted Reproduction 2022, 26, 554. [Google Scholar] [CrossRef]
- Rubio, C.; Navarro-Sánchez, L.; García-Pascual, C.M. (2022). Embryo Cell-Free DNA in the Culture Medium and Its Potential for Non-Invasive Aneuploidy Testing. In Handbook of Genetic Diagnostic Technologies in Reproductive Medicin (pp. 160-172). CRC Press.
- Vendrell, X.; Escribà, M.J. Non-invasive PGT. Medicina Reproductiva y Embriología Clínica 2021, 8, 100101. [Google Scholar] [CrossRef]
- Sakkas, D.; Navarro-Sánchez, L.; Ardestani, G.; Barroso, G.; Bisioli, C.; Boynukalin, K.; Cimadomo, D.; Frantz, N.; Kopcow, L.; Andrade, G.M.; et al. The impact of implementing a non-invasive preimplantation genetic testing for aneuploidies (niPGT-A) embryo culture protocol on embryo viability and clinical outcomes. Human Reproduction 2024, 39, 1952–1959. [Google Scholar] [CrossRef]
- Volovsky, M.; Scott Jr, R.T.; Seli, E. Non-invasive preimplantation genetic testing for aneuploidy: is the promise real? Human Reproduction 2024, 39, 1899–1908. [Google Scholar] [CrossRef]
- Rogers, A.; Menezes, M.; Kane, S.C.; Zander-Fox, D.; Hardy, T. Preimplantation genetic testing for monogenic conditions: is cell-free DNA testing the next step? Molecular diagnosis therapy 2021, 25, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Ai, Z.; Zhu, X.; Che, Z.; Pratikshya, A.; Tang, S.; Zhang, Q. Analysis of predictors of clinical pregnancy and live birth in patients with RIF treated with IVF-ET technology: A cohort study based on a propensity score approach. Frontiers in Medicine 2024, 11, 1348733. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; et al. Cell-free DNA in spent embryo culture medium reflects aneuploidy and mosaicism. Nature Communications 2016, 7, 11165. [Google Scholar]
- Rubio, C.; et al. Use of noninvasive preimplantation genetic testing (niPGT) to select euploid embryos in IVF: A prospective multicenter study. Fertility and Sterility 2020, 114, 538. [Google Scholar]
- Huang, L.; et al. Noninvasive preimplantation genetic testing for aneuploidy: Validation of a targeted next-generation sequencing-based method. Human Reproduction 2019, 34, 281. [Google Scholar]
- Hanson, B.M.; Tao, X.; Hong, K.H.; Comito, C.E.; Pangasnan, R.; Seli, E.; Jalas, C.; Scott, R.T., Jr. Noninvasive preimplantation genetic testing for aneuploidy exhibits high rates of deoxyribonucleic acid amplification failure and poor correlation with results obtained using trophectoderm biopsy. Fertility and Sterility 2021, 115, 1461–1470. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Luo, X.; Wu, R.; Qiu, L.; Lin, S.; Huang, X.; Wu, J. Evaluation of non-invasive gene detection in preimplantation embryos: a systematic review and meta-analysis. Journal of Assisted Reproduction and Genetics 2023, 40, 1243–1253. [Google Scholar] [CrossRef]
- Li Piani, L.; Petrone, P.; Brutto, M.; De Vos, A.; Van Der Kelen, A.; Vaiarelli, A.; Rienzi, L.; Conforti, A.; Cimadomo, D.; Verpoest, W. A systematic review and meta-analysis of double trophectoderm biopsy and/or cryopreservation in PGT: balancing the need for a diagnosis against the risk of harm. Human Reproduction Update 2025, 31, 102–115. [Google Scholar] [CrossRef]
- Li, X.; Hao, Y.; Chen, D.; Ji, D.; Zhu, W.; Zhu, X.; Wei, Z.; Cao, Y.; Zhang, Z.; Zhou, P. Non-invasive preimplantation genetic testing for putative mosaic blastocysts: a pilot study. Human Reproduction 2021, 36, 2020–2034. [Google Scholar] [CrossRef]
- Rubio, C.; Racowsky, C.; Barad, D.H.; Scott, R.T.; Simon, C. Noninvasive preimplantation genetic testing for aneuploidy in spent culture medium as a substitute for trophectoderm biopsy. Fertility and Sterility 2021, 115, 841–849. [Google Scholar] [CrossRef]
- Leaver, M.; Wells, D. Non-invasive preimplantation genetic testing (niPGT): the next revolution in reproductive genetics? Human reproduction update 2020, 26, 16–42. [Google Scholar] [CrossRef] [PubMed]
- Tiegs, A.W.; et al. Assessment of noninvasive preimplantation genetic testing: what are we missing? Current Opinion in Obstetrics and Gynecology 2021, 33, 138–145. [Google Scholar]
- Girardi, G.; Bremer, A.A. Advancing research on recurrent pregnancy loss: Overcoming obstacles and opportunities for translation. American Journal of Reproductive Immunology 2022, 87, e13508. [Google Scholar] [CrossRef]
- Oh, H.S.; Jang, J.M.; Yoon, H.J.; Choo, C.W.; Lim, K.S.; Lim, J.H.; Cheon, Y.P. The kinetics of nucleolar precursor bodies clustering at the pronuclei interface: Positive correlations with the morphokinetic characteristics of cleaving embryos and euploidy in preimplantation genetic testing programs. Korean Journal of Fertility and Sterility 2024. [CrossRef]
- Stankewicz, T. (2021). Optimizing IVF by controlling for both embryonic aneuploidy and endometrial receptivity using genetic testing. University of Kent (United Kingdom).
- Ying, L.I. (2022). Characterizing the Spectrum of Genome-wide Chromosomal Abnormalities in Early Pregnancy Loss by Lowpass Genome Sequencing (Doctoral dissertation, The Chinese University of Hong Kong (Hong Kong)).
- Peng, L.; Yang, W.; Deng, X.; Bao, S. Research progress on ANXA5 in recurrent pregnancy loss. Journal of Reproductive Immunology 2022, 153, 103679. [Google Scholar] [CrossRef] [PubMed]
- Petch, S.; Crosby, D. Updates in preimplantation genetic testing (PGT). Best Practice Research Clinical Obstetrics Gynaecology 2024, 102526. [Google Scholar] [CrossRef]
- Rubio, C.; Simón, C. Noninvasive preimplantation genetic testing for aneuploidy: Is the glass half-empty or half-full? Fertility and Sterility 2021, 115, 1426–1427. [Google Scholar] [CrossRef]
- Kakourou, G.; Mamas, T.; Vrettou, C.; Traeger-Synodinos, J. An update on non-invasive approaches for genetic testing of the preimplantation embryo. Current Genomics 2022, 23, 337–352. [Google Scholar] [CrossRef] [PubMed]
- Hoseini, S.M.; Montazeri, F.; Kalantar, S.M. Comparing the advantages, disadvantages and diagnostic power of different non-invasive pre-implantation genetic testing: A literature.
- Li, J.; Liu, Y.; Qian, Y.; Zhang, D. Noninvasive preimplantation genetic testing in assisted reproductive technology: current state and future perspectives. Journal of Genetics and Genomics 2020, 47, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; Wang, Y.; He, L.; Zheng, J.; Lin, Y.; Wang, F. Performance of preimplantation genetic testing for aneuploidy for patients with unexplained recurrent pregnancy loss and repeated implantation failure. Heliyon 2024, 10. [Google Scholar] [CrossRef] [PubMed]
| Domain | Standardization requirements |
|---|---|
| Culture conditions | Media type/volume, change intervals, incubation duration [2,56] |
| Sample handling | SCM collection timing, storage, contamination controls [56] |
| Analytical methods | WGA kits, sequencing depth, bioinformatics [2,56] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




