1. Introduction
Urinary tract infections (UTIs) are the most prevalent and enduring infections observed after kidney transplantation [
1,
2,
3]. Researches indicates that the incidence of UTIs in kidney transplant recipients varies between 12% and 75%, contingent upon the specific population and geographic area examined [
4,
5,
6,
7]. The occurrence of a UTI in kidney transplant patients can have far-reaching consequences, affecting both the survival of the graft and the overall quality of life for the patient. In many cases, UTIs can lead to hospitalizations, prolonged recovery periods, and an increased risk of further complications, including morbidity and mortality[
8,
9]. The bacterial strains most commonly implicated in post-transplant urinary tract infections include Escherichia coli, which accounts for 30-80% of cases. Other notable pathogens include Klebsiella pneumoniae (12%), Enterococcus species (5%), as well as less common strains such as Pseudomonas aeruginosa and Proteus [
5,
6,
7]. These infections can vary from uncomplicated to severe, multi-drug-resistant infections that complicate treatment outcomes. Several patient-specific and transplant-related factors contribute to an increased risk of developing a UTI after transplantation. Factors include advanced age, as older patients exhibit greater susceptibility, and gender, with females at a higher risk compared to males. Diabetes mellitus, getting a kidney from a cadaveric donor, continues use of urinary catheters, and acute rejection episodes are additional significant risk factors [
1,
4,
6,
10]. All these characteristics can affect the frequency and severity of UTI episodes, complicating prevention and management in this demographic. This study attempts to enhance understanding of recurring and multi-drug resistance urinary tract infections in kidney transplant patients using a retrospective analysis of patient data collected two years post-transplant among two hospitals. The main objective is to evaluate the existing therapeutic options implemented in Saudi Arabia to manage these medical conditions. The study seeks to identify the principal risk variables that enhance the possibility of recurring UTIs, possibly influencing future prevention and treatment strategies for this patient group in Saudi Arabia.
2. Result
The medical records of 491 renal transplant recipients were reviewed, and 75 patients (15.3%) met the inclusion criteria for recurrent UTIs. The mean age of the patients was 48.2 ± 15.8 years, with 43 (57.5%) being women. The average BMI was 24.9 ± 5.9. A significant majority (85.3%) of the patients received living donor transplants
Table 1.
Key risk factors associated with recurrent UTIs included age over 45 (Mean = 48.2), DM (OR = 1.833, P = 0.039), the presence of multi-drug resistant organisms (MDRO) (OR = 3.14, 95% CI = 1.19-8.33, P = 0.02), ureteric stents (OR = 4.07, 95% CI = 1.05-15.67, P = 0.04), and complicated UTIs (OR = 4.60, 95% CI = 1.57-13.50, P = 0.005). Interestingly, patients with catheters were significantly less likely to experience recurrent UTIs compared to those without (OR = 0.19, 95% CI = 0.07-0.54, P = 0.002), in addition to female compared to male (OR = 0.382, 95% CI = 0.148-0.99, P = 0.048)
Table 2.
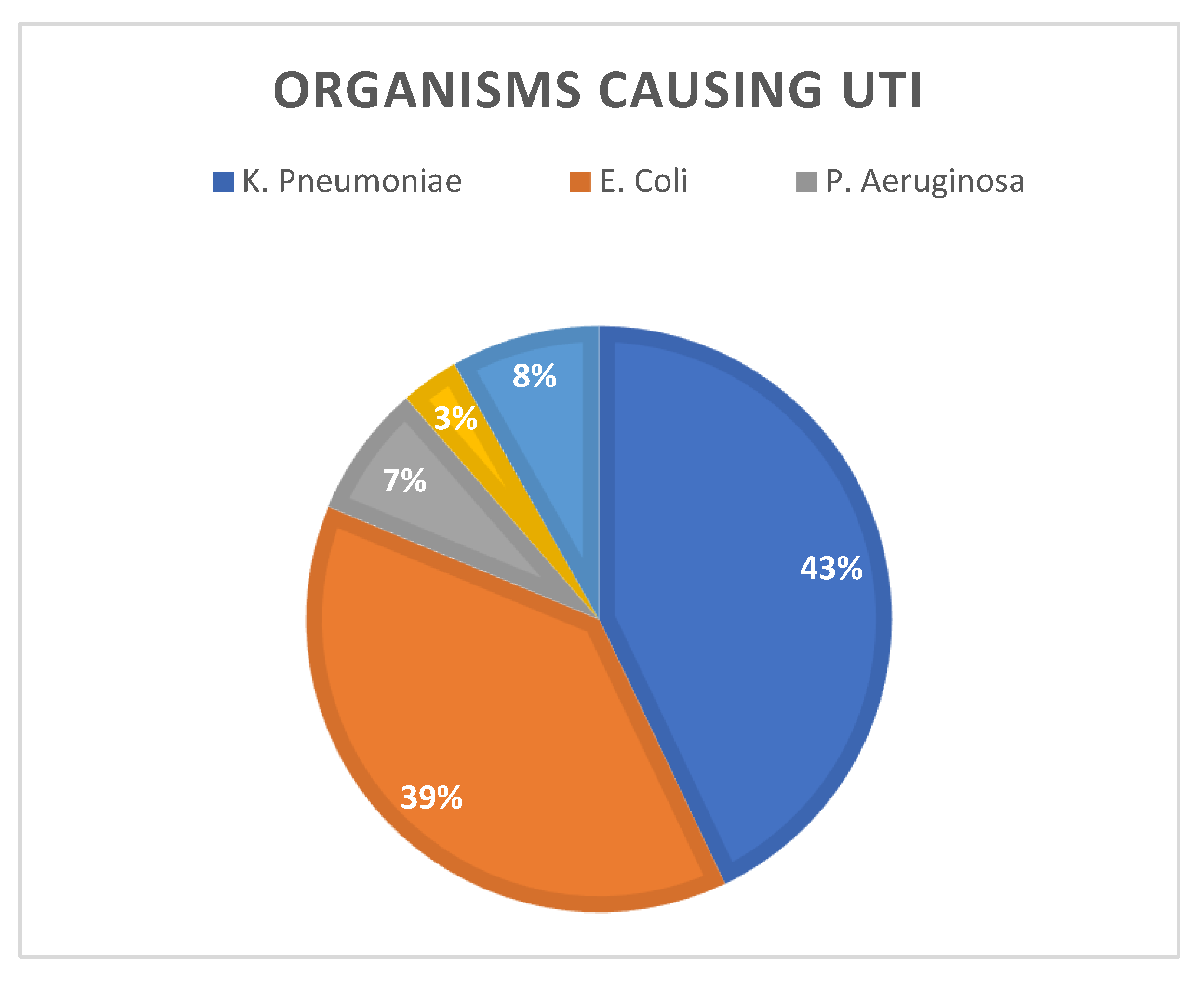
Logistic regression showed no significant association between baseline serum creatinine (OR = 1.006, P = 0.136) and white blood cell count (OR = 0.963, P = 0.472) with the recurrence of UTIs post-transplant. Similarly, no significant differences were found in serum creatinine levels or WBC counts between the first and second UTI episodes (P > 0.05). Additionally, the duration of prophylaxis (OR = 1.10, 95% CI = 0.97-1.22, P = 0.67) and treatment duration after the first (OR = 0.99, 95% CI = 0.86-1.14, P = 0.90) and second UTI episodes (OR = 1.06, 95% CI = 0.95-1.18, P = 0.29) did not increase the risk of recurrent UTIs. Out of 219 UTI episodes, Klebsiella pneumoniae was the most commonly identified organism (n = 94; 42.9%), followed by Escherichia coli (n = 84; 38.4%), Pseudomonas aeruginosa (n = 16; 7.3%), Proteus mirabilis (n = 7; 3.2%), and others (n = 18; 8.2%)
Figure 1.
The most frequently used antibiotics were Carbapenems (n = 95; 40.8%), Cephalosporins (n = 67; 28.2%), Penicillin (n = 28; 12%), and Fluoroquinolones (n = 27; 11.6%). The Wilcoxon signed-rank test revealed a significant rise in serum creatinine levels in post-transplant patients with recurrent UTIs compared to baseline levels (Z = -4.552, P < 0.001)
Table 3.
Serum creatinine increased in 58 patients, remained the same in three, and decreased in 14. The median post-transplant creatinine level was 97 µmol/L (IQR = 77), compared to a baseline median of 85 µmol/L (IQR = 48) (Z = -4.552, P < 0.001).
3. Discussion
Our results correspond with other research regarding risk factors for recurrent urinary tract infections [
1,
4,
6,
7,
11]. For instance, when we compared our findings with a meta-analysis encompassing around 72,600 individuals, both investigations recognized age exceeding 45 as a notable risk factor, with a mean age of 48 years in our investigation. Both studies also highlighted the use of ureteric stents as a major risk factor, with an odds ratio (OR) of 4.065 (95% CI: 1.054-15.670) in our study and OR = 1.54 (95% CI: 1.16–2.06) in the meta-analysis [
4]. In cases of UTIs caused by multi-drug-resistant organisms (MDROs), our findings were consistent with those of a prior study that showed this risk factor nearly tripled the chances of recurrence, with an OR of 2.75 (95% CI: 1.97-3.83). Our study produced similar results, with an OR of 3.143 (95% CI: 1.185-8.334) [
12]. Patients with complicated UTIs in our study had a significantly higher risk of recurrent UTIs (OR = 4.600, 95% CI: 1.567 - 13.502) and DM (OR = 1.833, P = 0.039), marking them as the primary risk factors identified. Although urinary catheters are widely recognized as a risk factor for urinary tract infections (UTIs), our analysis revealed that their presence correlated with a reduced incidence of recurrent UTIs (OR = 0.19, 95% CI = 0.07-0.54, P = 0.002), potentially attributable to the transient duration of catheter usage and its impact on UTIs Risks and Benefits of Early Catheter Removal After Renal [
13,
14,
15]. Certain studies indicate that early removal might reduce the occurrence of urinary tract infections. Further factors, including smoking, anatomical anomalies, and infections like CMV and BK virus, did not demonstrate significant correlations with recurrent UTIs. (
Table 2). All kidney transplant recipients in our study were given induction immunosuppressants, specifically thymoglobulin (ATG) (49.3%) or basiliximab (50.7%). Maintenance therapy included steroids, tacrolimus, and mycophenolate mofetil (MMF). Only 3 out of 75 patients (4%) experienced episodes of rejection. While most studies define E. coli as the predominant cause of UTIs post-kidney transplantation, our research identified Klebsiella pneumoniae as the most prevalent pathogen, accounting for 42.9%
of the cases. The primary pathogen in each UTI episode was followed by E. coli at 38.4%, Pseudomonas aeruginosa at 7.3%, Proteus mirabilis at 3.2%, and other organisms representing 8.2% of the total UTI episodes..Numerous organizations, including the Infectious Disease Society of America (IDSA), the American Society of Transplantation Infectious Diseases Community of Practice, and the European Association of Urology Guidelines on Urological Infections, offer recommendations regarding the classifications of urinary tract infections (UTIs) and suitable antibiotic regimens for each [
5,
16,
17]. In our study, we found that carbapenems (40%) were the most commonly used antibiotics to treat recurrent UTIs, followed by cephalosporins (28.8%), with an average treatment duration of around 9 days
Table 4.
The choice of antibiotic and treatment length varies based on factors such as the patient’s condition, the type of organism, whether the infection is complicated or uncomplicated, and the administration route [
18,
19,
20,
21,
22]. While some studies explore available antibiotic options to prevent recurrent infections [
23,
24,
25], all kidney transplant recipients in our study received UTI prophylaxis with daily trimethoprim-sulfamethoxazole for at least six months post-transplant, following KDIGO guidelines for the care of KTRs [
26]. Our findings indicate that carbapenems are statistically significant and effective in the treatment of recurrent complicated urinary tract infections (P < 0.001). Cephalosporins and penicillin were particularly beneficial in treating recurring uncomplicated urinary tract infections. (P < 0.001)
Table 5. [
27,
28,
29].
Numerous research have investigated the influence of UTIs on graft function, yielding varied outcomes. Certain study The Impact of Urinary Tract Infections [
30,
31] indicates no correlation between urinary tract infections (UTIs) and poor transplant function or survival, but other studies recognize UTIs as a notable risk factor for unfavorable graft outcomes Effects of recurrent urinary tract infections on graft and patient outcomes after kidney transplantation[
8,
9]. Our investigation revealed that blood creatinine levels were elevated subsequent to urinary tract infection episodes, especially following the initial infection. A Wilcoxon signed-rank test revealed a statistically significant rise in serum creatinine levels in KTRs following UTI incidents compared to baseline levels (Z = -4.552, p < 0.001), with serum creatinine increasing in 58 patients (77.3%). This study possesses multiple characteristics, notably its implementation at two major transplant hospitals in Saudi Arabia, which enabled access to an extensive dataset and supported the examination of certain risk factors over an extended duration. The findings correspond with existing research, and the thorough analysis of antibiotic utilization, pathogen prevalence, and their impact on serum creatinine offers significant insights into post-transplant management. However, there are some limitations. The retrospective design of the study may introduce bias resulting from differences in hospital systems, employment, and standards over time. The limited sample size and concentration on a single region may restrict the applicability of the findings to wider populations.
4. Materials and Methods
This cross-sectional study examined the medical records of kidney transplant patients at King Faisal Specialist Hospital & Research Center in Jeddah, along with data from King Fahad Hospital in Madinah, Saudi Arabia, from March to May 2022. The study was approved by the Institutional Review Boards of both hospitals (IRB 2022-22 for Jeddah and IRB 22-073 for Madinah). The inclusion criteria focused on adult patients who had received a kidney transplant and developed recurrent UTIs within two years post-transplant. Exclusion criteria: Patients who experienced only one or no UTI episodes post-transplant, those with incomplete data or lost to follow-up, and patients under 18 years of age. The study collected variables such as patient demographics, pre-treatment risk factors, transplantation-related factors, baseline lab values, immunosuppressant medications, and UTI episodes, along with their management. Definitions for classifications used in this study were based on previous research. Recurrent UTI: Defined as two or more UTI episodes in the past six months, three or more symptomatic UTIs in the past 12 months, or more than one UTI episode within two years of kidney transplantation. Uncomplicated UTI: Positive urine culture accompanied by urinary symptoms such as dysuria, urgency, frequency, or suprapubic pain. Complicated UTI: Systemic symptoms requiring hospital admission for intravenous antibiotic therapy. Clinical presentation: Symptoms such as fever, chills, dysuria, cloudy urine, flank pain, or tenderness in the grafted kidney. Urinalysis: Considered positive if leukocyte esterase is ≥ 250 and/or nitrite is positive. Risk factors recorded in the study included diabetes mellitus, smoking, structural factors (e.g., native kidney disease with urological malformations, renal cysts, calculi, benign prostate hyperplasia), neurogenic bladder, and operation-related factors (e.g., urinary catheter, ureteric stent, vesicoureteral reflux, and urinary fistula), as well as infections with CMV, BK virus, and UTI caused by E. coli.
We used statistical analyses to explore the relationship between various risk factors and recurrent UTIs in kidney transplant recipients. Descriptive statistics were applied to summarize the demographic and clinical characteristics of the participants. Categorical variables were reported as frequencies and percentages, while continuous variables were presented as means and standard deviations. To assess the association between risk factors and recurrent UTIs, we performed logistic regression analysis. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated to estimate the strength of these relationships, with a p-value of less than 0.05 indicating statistical significance. For comparing paired data, we employed non-parametric tests like the Wilcoxon signed-rank test. All analyses were conducted using SPSS version 25, with a significance threshold set at p < 0.05 for all statistical tests.
5. Conclusions
Klebsiella pneumoniae emerged as the most frequent pathogen in recurrent UTIs. Severe cases were typically treated with carbapenems, while simpler infections were managed with cephalosporins or penicillin. Despite the recurrence of these infections, preventive treatment was uncommon. However, recurrent UTIs did lead to increased serum creatinine levels, resulting in more hospitalizations and higher healthcare costs. There’s a clear need for improved prevention strategies to address this issue.
Author Contributions
All authors played a key role in multiple stages of this research, including the initial concept, study design, data collection, and subsequent analysis and interpretation. They were also actively involved in drafting and critically revising the manuscript to ensure its intellectual rigor. Additionally, each author approved the final version of the manuscript for submission, participated in the decision to select the journal, and accepted full responsibility for the accuracy and integrity of the research.
Funding
This study was carried out without any external financial assistance or funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board. Institutional Review Board (IRB) approval was obtained from both King Faisal Specialist Hospital & Research Center, Jeddah (IRB 2022-22), and King Fahad Hospital, Madinah (IRB 22-073),.
Informed Consent Statement
the need for informed consent was waived by the IRB, given that the study posed minimal risk to participants.
Data Availability Statement
Upon a justified request, the corresponding author can share the data.
Conflicts of Interest
The authors confirm that there are no conflicts of interest that could have influenced the findings of this study.
Abbreviations
The following abbreviations are used in this manuscript:
| Abbreviations |
| UTI |
Urinary Tract Infection |
| rUTI |
Recurrent Urinary Tract Infection |
| CMV |
Cytomegalovirus |
| BK |
BK virus (polyomavirus) |
| MDRO |
Multi-Drug Resistant Organism |
| DM |
Diabetes Meletus |
| KTR |
Kidney Transplant Recipient |
References
- Alangaden GJ, Thyagarajan R, Gruber SA, et al. Infectious complications after kidney transplantation: current epidemiology and associated risk factors. Clin Transplant 2006;20:401–9. [CrossRef]
- Alangaden, G. Urinary tract infections in renal transplant recipients. Curr Infect Dis Rep 2007;9:475–9. [CrossRef]
- Veroux M, Giuffrida G, Corona D, et al. Infective complications in renal allograft recipients: epidemiology and outcome. Transplant Proc 2008;40:1873–6. [CrossRef]
- Hosseinpour M, Pezeshgi A, Mahdiabadi MZ, et al. Prevalence and risk factors of urinary tract infection in kidney recipients: a meta-analysis study. BMC Nephrol 2023;24:284. [CrossRef]
- Goldman JD, Julian K. Urinary tract infections in solid organ transplant recipients: Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant 2019;33. [CrossRef]
- Wu X, Dong Y, Liu Y, et al. The prevalence and predictive factors of urinary tract infection in patients undergoing renal transplantation: A meta-analysis. Am J Infect Control 2016;44. [CrossRef]
- Behzad D, Hakimeh A, Hossein R, et al. A middle east systematic review and meta-analysis of bacterial urinary tract infection among renal transplant recipients; Causative microorganisms. Microb Pathog 2020;148. [CrossRef]
- Nascimento EHG, Nascimento E, Lasmar MF, et al. Effects of Bacterial Urinary Tract Infection on Clinical Outcome and Survival of Kidney Transplant Patients. Transplant Proc 2022;54:1262–9. [CrossRef]
- Britt NS, Hagopian JC, Brennan DC, et al. Effects of recurrent urinary tract infections on graft and patient outcomes after kidney transplantation. Nephrology Dialysis Transplantation 2017;32:1758–66. [CrossRef]
- Fiorentino M, Pesce F, Schena A, et al. Updates on urinary tract infections in kidney transplantation. J Nephrol 2019;32. [CrossRef]
- Velioglu A, Guneri G, Arikan H, et al. Incidence and risk factors for urinary tract infections in the first year after renal transplantation. PLoS One 2021;16. [CrossRef]
- Alevizakos M, Nasioudis D, Mylonakis E. Urinary tract infections caused by ESBL -producing Enterobacteriaceae in renal transplant recipients: A systematic review and meta-analysis. Transplant Infectious Disease 2017;19. [CrossRef]
- Rabkin DG, Stifelman MD, Birkhoff J, et al. Early catheter removal decreases incidence of urinary tract infections in renal transplant recipients. Transplant Proc 1998;30:4314–6. [CrossRef]
- Guler S, Cimen S, Hurton S, et al. Risks and Benefits of Early Catheter Removal After Renal Transplantation. Transplant Proc 2015;47:2855–9. [CrossRef]
- Werneburg, GT. Catheter-Associated Urinary Tract Infections: Current Challenges and Future Prospects. Res Rep Urol 2022;Volume 14:109–33. [CrossRef]
- Gupta K, Hooton TM, Naber KG, et al. International Clinical Practice Guidelines for the Treatment of Acute Uncomplicated Cystitis and Pyelonephritis in Women: A 2010 Update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clinical Infectious Diseases 2011;52:e103–20. [CrossRef]
- Kranz J, Bartoletti R, Bruyère F, et al. European Association of Urology Guidelines on Urological Infections: Summary of the 2024 Guidelines. Eur Urol 2024;86:27–41. [CrossRef]
- Barber AE, Norton JP, Spivak AM, et al. Urinary Tract Infections: Current and Emerging Management Strategies. Clinical Infectious Diseases 2013;57:719–24. [CrossRef]
- Pitout JDD. Infections with Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae. Drugs 2010;70:313–33. [CrossRef]
- El-Gamal MI, Brahim I, Hisham N, et al. Recent updates of carbapenem antibiotics. Eur J Med Chem 2017;131:185–95. [CrossRef]
- Tan X, Pan Q, Mo C, et al. Carbapenems vs alternative antibiotics for the treatment of complicated urinary tract infection. Medicine 2020;99:e18769. [CrossRef]
- Antimicrobial therapy of complicated urinary. 2012.
- Wagenlehner FME, Vahlensieck W, Bauer HW, et al. Prevention of recurrent urinary tract infections. Minerva Urol Nefrol 2013;65.
- Dason S, Dason JT, Kapoor A. Guidelines for the diagnosis and management of recurrent urinary tract infection in women. Canadian Urological Association Journal 2011:316–22. [CrossRef]
- Al-Badr A, Al-Shaikh G. Recurrent Urinary Tract Infections Management in Women : A Review = الإلتهابات المتكررة للمسالك البولية عند النساء و طرق علاجها : مراجعة. Sultan Qaboos Univ Med J 2013;13:359–67. [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) Transplant Work Group. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplant 2009;9 Suppl 3:S1-155. [CrossRef]
- Mehnert-Kay, SA. Diagnosis and management of uncomplicated urinary tract infections. Am Fam Physician 2005;72:451–6.
- Bono MJ, Leslie SW, Reygaert WC. Uncomplicated Urinary Tract Infections. 2024.
- Hooton, TM. Uncomplicated Urinary Tract Infection. New England Journal of Medicine 2012;366:1028–37. [CrossRef]
- Halskov ACL, Dagnæs-Hansen J, Stroomberg H V., et al. Incidence of and Risk Factors for Recurrent Urinary Tract Infections in Renal Transplant Recipients. Eur Urol Open Sci 2023;52:115–22. [CrossRef]
- Al Tamimi AR, Alotaibi WS, Aljohani RM, et al. The Impact of Urinary Tract Infections in Kidney Transplant Recipients: A Six-Year Single-Center Experience. Cureus 2023. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).