Submitted:
20 August 2025
Posted:
22 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
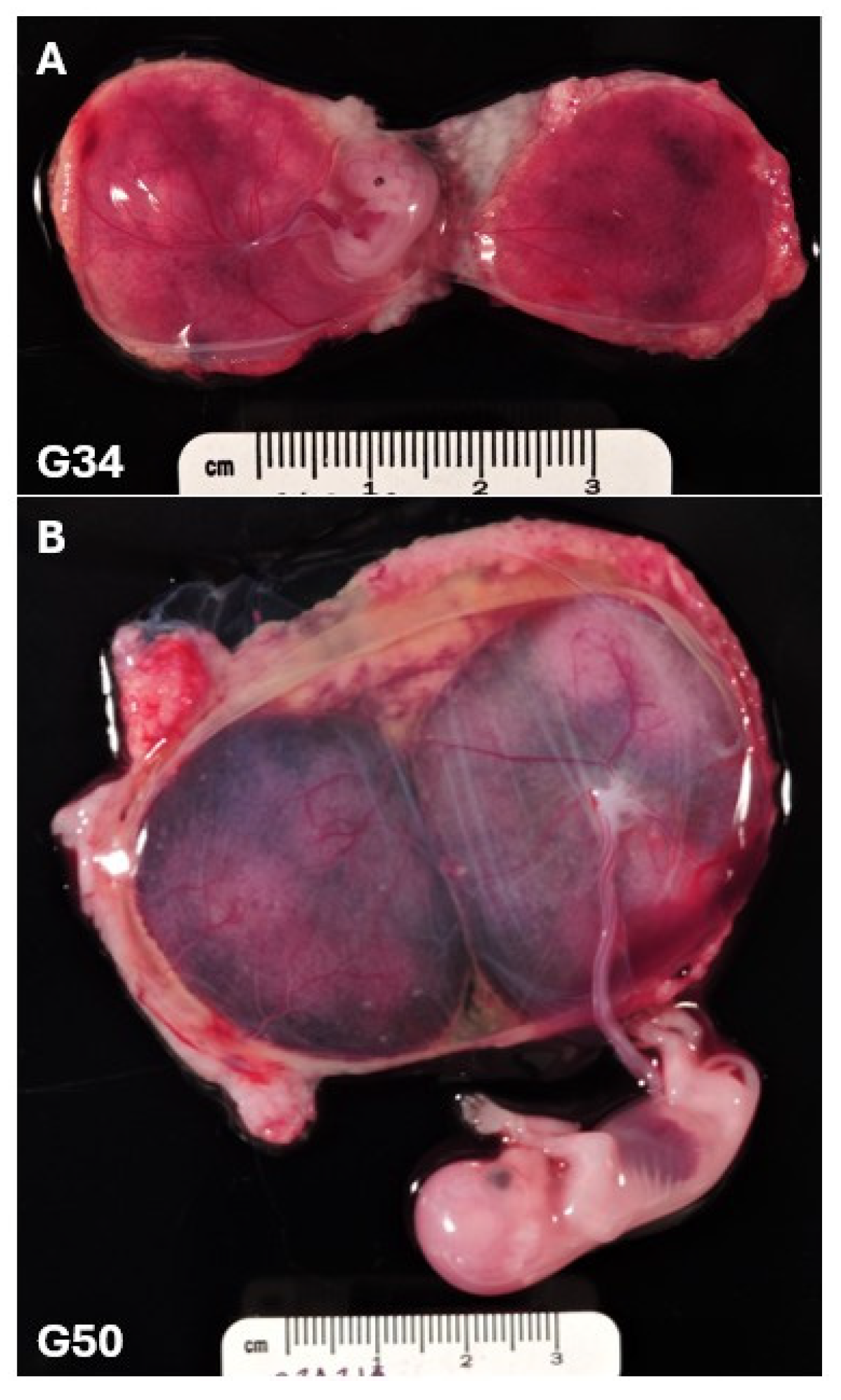
2.1. Pregnant Dams
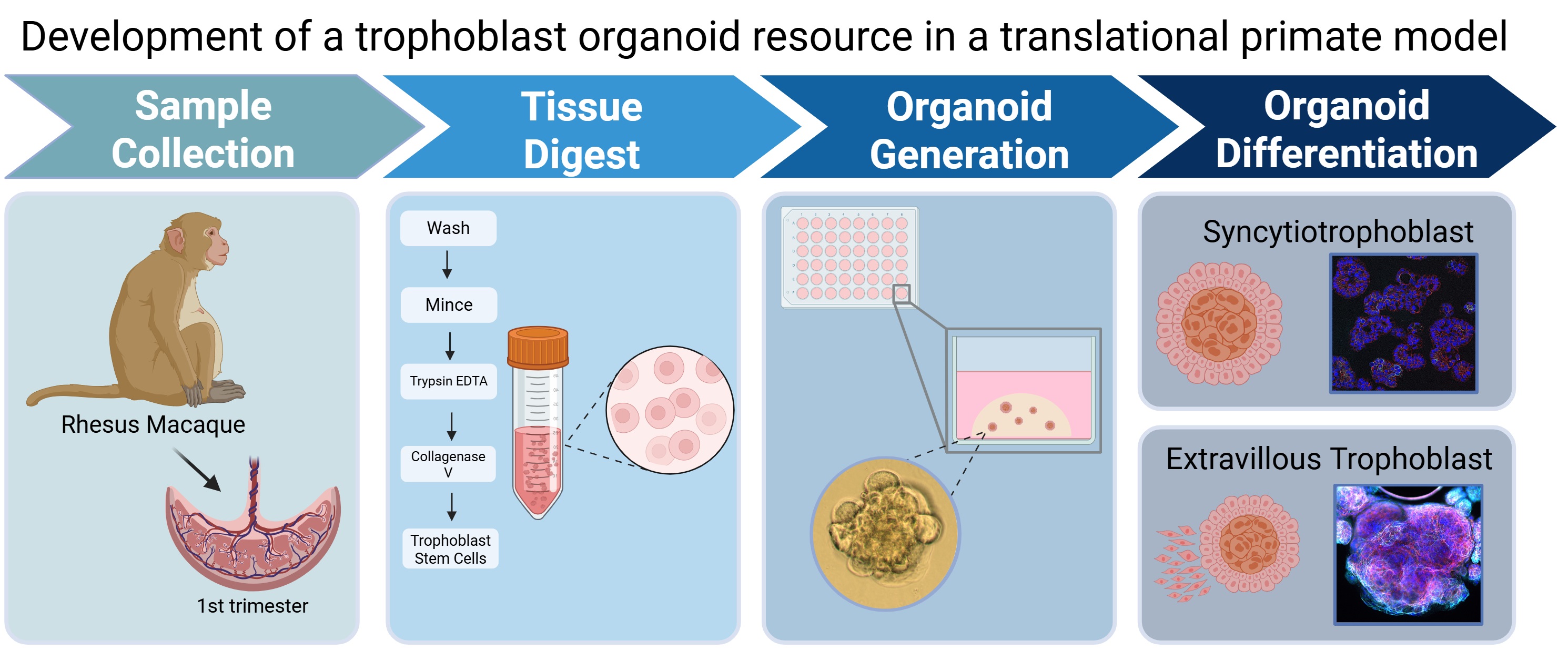
2.2. Organoid Isolation
2.3. Routine Maintenance and Passage
2.4. Cryopreservation and Re-Establishing in Culture
2.5. Differentiation of Trophoblast Organoids
2.6. Media Harvesting and Storage
2.7. Histology
| Media Component | Volume | Stock Concentration |
Final Concentration |
Manufacturer / Catalog Number |
|---|---|---|---|---|
| Advanced DMEM/F12 | 17.6mL | N/A | N/A | ThermoFisher 12634028 |
| L-glutamine | 200µl | 200mM | 2mM | Fisher Scientific 25-030-149 |
| 2-mercapthoethanol | 36.4µl | 55mM | 100µM | Gibco 21985023 |
| Penicillin/Streptomycin | 100µl | 100X | 0.5X | Fisher Scientific 15-140-148 |
| Bovine Serum Albumin | 1mL | 7.5% | 0.375% | Sigma Aldrich A8412-100ML |
| Insulin-Transferrin-Selenium Ethanolamine | 200µl | 100X | 1X | Gibco 15140148 |
| Neuraregulin-1* | 20µl | 100µg/ml | 100ng/ml | ThemoFisher 100-03-50UG |
| A83-01 | 30µl | 5mM | 5µM | Millipore Sigma SML0788-5MG |
| Knockout Serum Replacement | 800µl | N/A | 4% | Gibco 10828028 |
2.8. Immunostaining of Organoids
2.9. Immunostaining FFPE Tissue
2.10. Confocal Microscopy
2.11. RNA Isolation
2.12. RT-qPCR
2.13. Agarose Gel Electrophoresis
2.14. Protein Isolation
2.15. LDH Assay
2.16. GDF-15 ELISA
2.17. Organoid Polarity Manipulation
2.18. Image Processing
3. Results
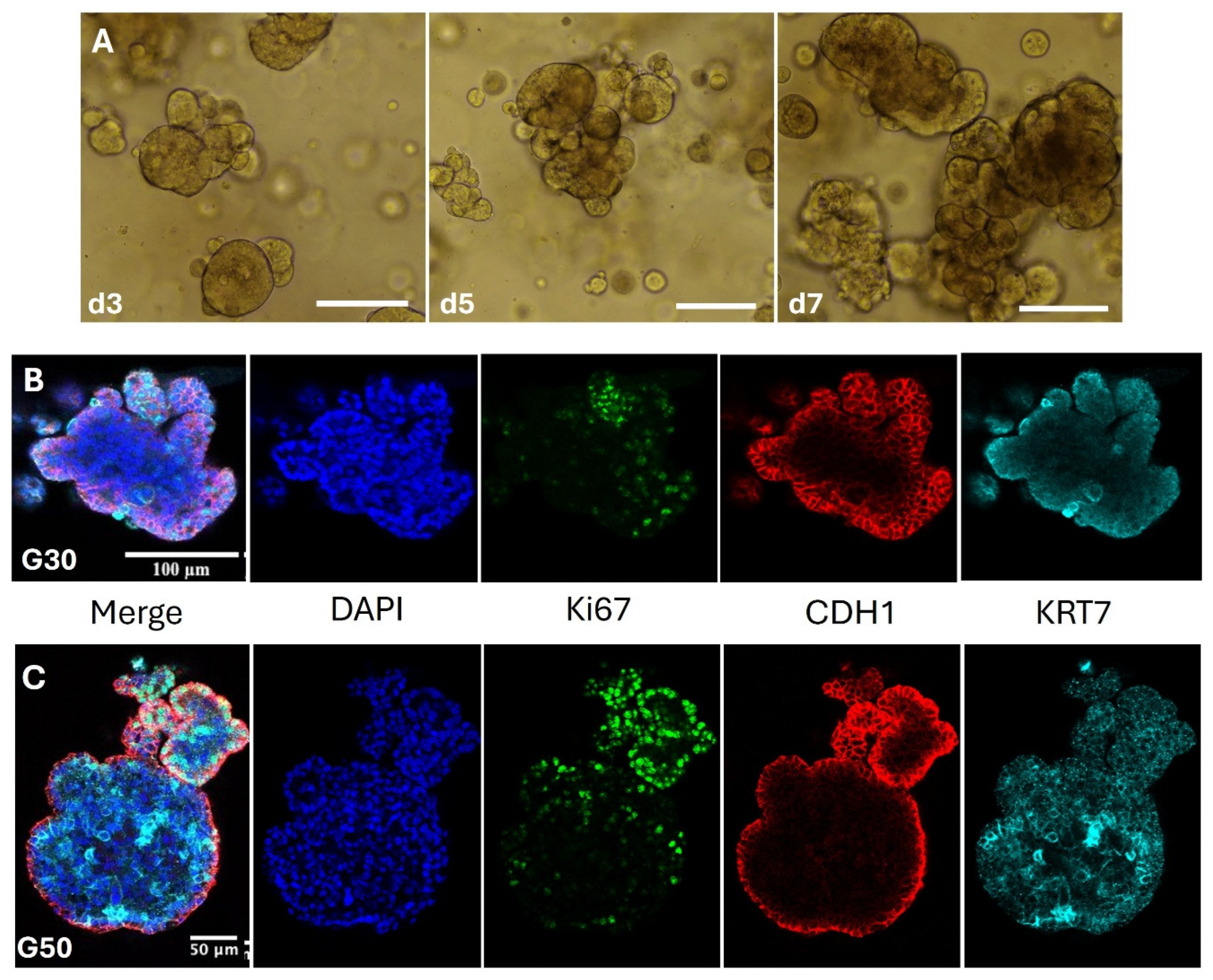
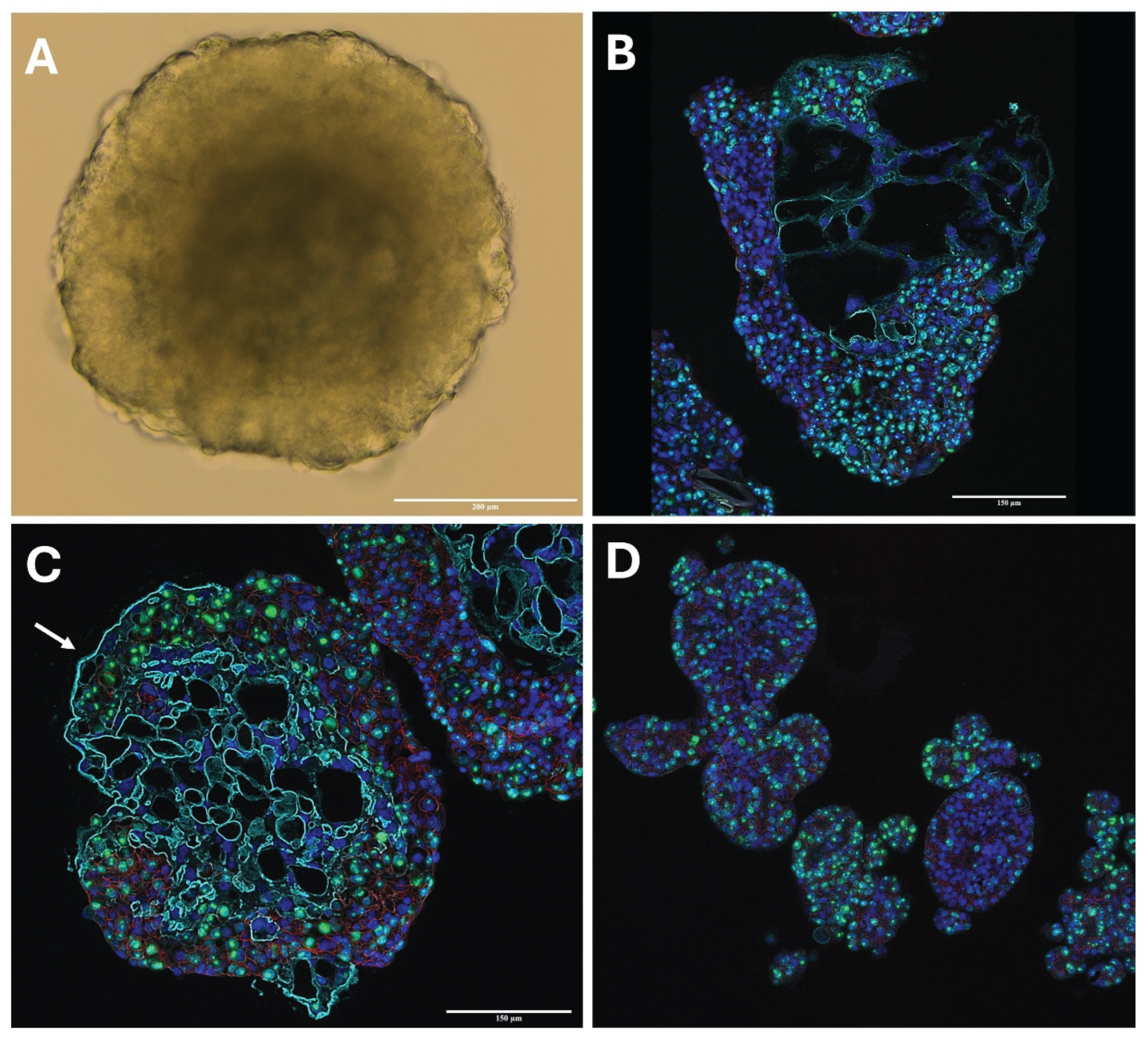
3.1. Generation of Rhesus Trophoblast Organoids
3.2. Characterization of Rhesus Trophoblast Organoids
3.3. Differentiation of Rhesus Trophoblast Organoids
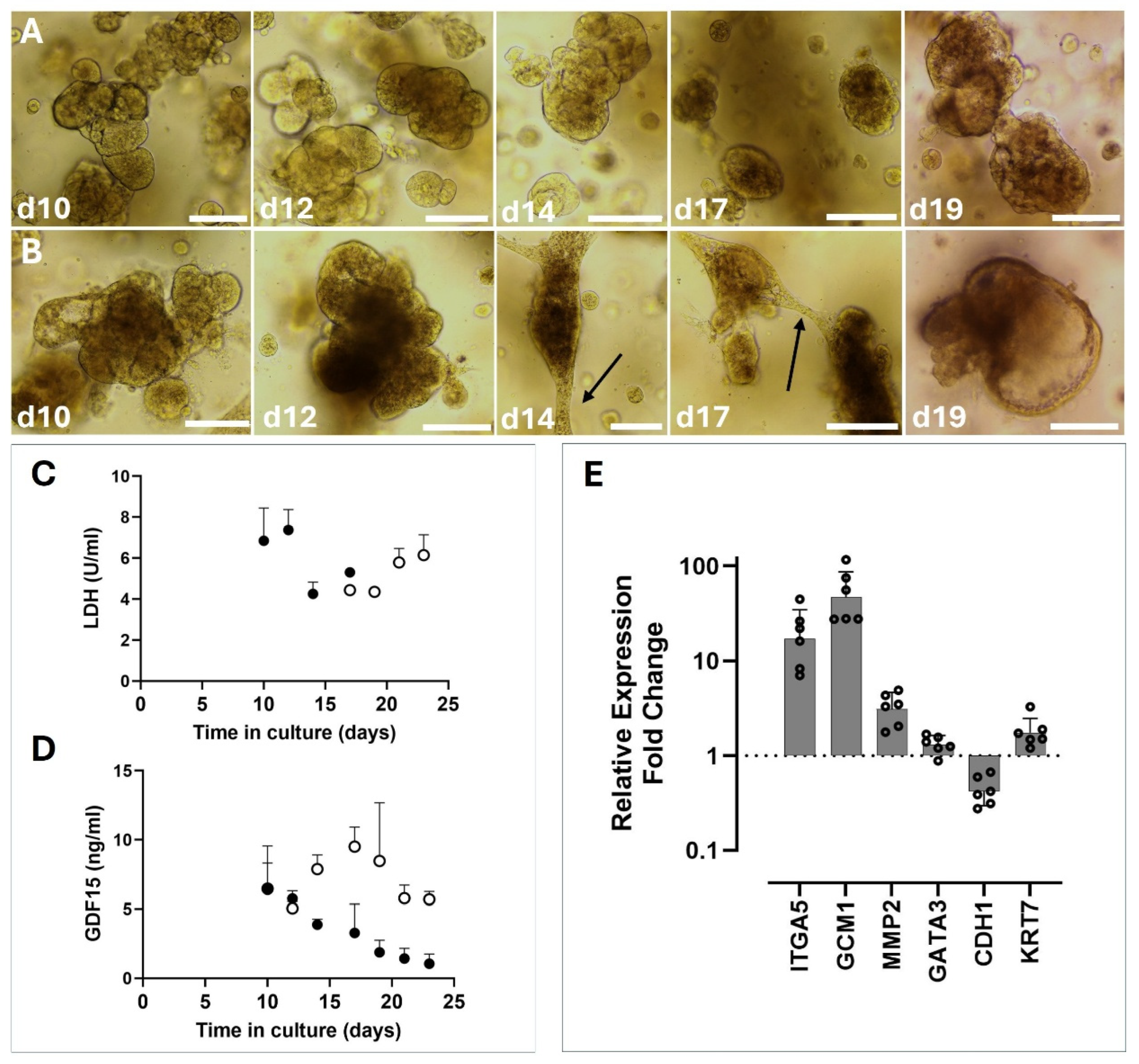
3.3. Differentiation of Rhesus Trophoblast Organoids
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Acronym | Definition |
| SYN | Syncytiotrophoblast |
| EVT | Extravillous Trophoblast |
| TSC | Trophoblast Stem Cell |
| NHP | Nonhuman Primate |
| TO | Trophoblast Organoid |
| RTO | Rhesus Trophoblast Organoid |
| HTO | Human Trophoblast Organoid |
| TOM | Trophoblast Organoid Media |
| TMB | Time-Mated Breeding |
| RBC | Red Blood Cell |
| CRS | Cell Recovery Solution |
| LDH | Lactate Dehydrogenase |
| IACUC | Institutional Animal Care and Use Committee |
Appendix A
| Buffer Name | Reagents | Manufacturer & Catalog Number |
|---|---|---|
| Phosphate Buffered Saline | NaCl 137mM, KCl 2.7mM, Na2HPO4 10mM, KH2PO4 1.8mM | Sigma Aldrich, S9888-1KG, P3911-500G, S9763-500G, P0662-500G |
| Permeabilization Buffer | PBS 0.5% Triton X-100 |
Sigma Aldrich, T8787-50ML |
| Blocking Buffer | PBS 5% Goat Serum, 0.1% Tween-20 |
Jackson Immunoresearch, 005-000-121 Thermo Fisher, J20605.AP |
| Citrate Buffer, pH 6.0 | 10mM sodium citrate, 0.05% Tween-20, pH 6.0 | Fisher Scientific, S279-500 |
| Tris-EDTA Buffer, pH 9.0 | 10mM Tris, 1mM EDTA, 0.05% Tween-20, pH 9.0 | Thermo Fisher, 15506017 Thermo Fisher, 15576028 |
| Mounting Media | 90% Glycerol, 10mM Tris | Fisher Scientific, G33-1 |
| Prolong Gold Antifade Mounting Media | Thermo Fisher, P36930 |
| Gene | Primer (5’-3’) | Amplicon Size (bp) |
|---|---|---|
| 18s rRNA | F:TAACGAACGAGACTCTGGCAT | 138 |
| R:CGGACATCTAAGGGCATCACAG | ||
| ITGA5 | F: TTACGGGACTCAACTGCACC | 149 |
| R: CTCCGGGCATTTCAGGATCT | ||
| ACTB | F: CTACCATGAGCTGCGTGTGG | 130 |
| R: GTACATGGCTGGGGTGTTGA | ||
| GCM1 | F: AAGCTGCCACAGAACGTGAA | 98 |
| R: CGCTGTGCATTCTTGTCCTC | ||
| GATA3 | F: CTCTTCGCTACCCAGGTGAC | 93 |
| R: ACTCTGGAATTCTGCGAGCC | ||
| NOTCH1 | F: GACGGCATCAATGGCTTCAC | 140 |
| R: CAGTCGCACTTGTACCCGTT | ||
| KRT7 | F: ACGGGAATGCAGTTGAGGAC | 134 |
| R: CATTACGGAGGTCCCTGGAAG | ||
| MMP2 | F: GAGAAGGCCAAGTGGTCCG | 171 |
| R: AGCCGTACTTGCCATCCTTC |
References
- R. N. Baergen, G. J. Burton, and C. G. Kaplan, Eds., Benirschke’s Pathology of the Human Placenta. Cham: Springer International Publishing, 2022. [CrossRef]
- C. M. Simán, C. P. Sibley, C. J. Jones, M. A. Turner, and S. L. Greenwood, “The functional regeneration of syncytiotrophoblast in cultured explants of term placenta,” Am. J. Physiol. Regul. Integr. Comp. Physiol., vol. 280, no. 4, pp. R1116-1122, Apr. 2001. [CrossRef]
- D. L. Bloxam, C. M. Bax, and B. E. Bax, “Culture of syncytiotrophoblast for the study of human placental transfer. Part I: Isolation and purification of cytotrophoblast,” Placenta, vol. 18, no. 2–3, pp. 93–98, 1997. [CrossRef]
- P. V. Mohanan, Microfluidics and Multi Organs on Chip. Springer Nature, 2022.
- M. Knöfler, S. Haider, L. Saleh, J. Pollheimer, T. K. J. B. Gamage, and J. James, “Human placenta and trophoblast development: key molecular mechanisms and model systems,” Cell. Mol. Life Sci. CMLS, vol. 76, no. 18, pp. 3479–3496, Sep. 2019. [CrossRef]
- A. E. Guttmacher, Y. T. Maddox, and C. Y. Spong, “The Human Placenta Project: placental structure, development, and function in real time,” Placenta, vol. 35, no. 5, pp. 303–304, May 2014. [CrossRef]
- A. E. Guttmacher and C. Y. Spong, “The human placenta project: it’s time for real time,” Am. J. Obstet. Gynecol., vol. 213, no. 4 Suppl, pp. S3-5, Oct. 2015. [CrossRef]
- N. Andescavage and C. Limperopoulos, “Emerging placental biomarkers of health and disease through advanced magnetic resonance imaging (MRI),” Exp. Neurol., vol. 347, p. 113868, Jan. 2022. [CrossRef]
- M. C. Schabel et al., “Quantitative longitudinal T2* mapping for assessing placental function and association with adverse pregnancy outcomes across gestation,” PloS One, vol. 17, no. 7, p. e0270360, 2022. [CrossRef]
- S. Haider et al., “Self-Renewing Trophoblast Organoids Recapitulate the Developmental Program of the Early Human Placenta,” Stem Cell Rep., vol. 11, no. 2, pp. 537–551, Aug. 2018. [CrossRef]
- S. Ramani, S. E. Crawford, S. E. Blutt, and M. K. Estes, “Human organoid cultures: Transformative New Tools for Human Virus Studies,” Curr. Opin. Virol., vol. 29, pp. 79–86, Apr. 2018. [CrossRef]
- M. Y. Turco et al., “Trophoblast organoids as a model for maternal-fetal interactions during human placentation,” Nature, vol. 564, no. 7735, pp. 263–267, Dec. 2018. [CrossRef]
- K. Benirschke, “Placentation,” J. Exp. Zool., vol. 228, no. 2, pp. 385–389, 1983. [CrossRef]
- E. M. Ramsey, M. L. Houston, and J. W. Harris, “Interactions of the trophoblast and maternal tissues in three closely related primate species,” Am. J. Obstet. Gynecol., vol. 124, no. 6, pp. 647–652, Mar. 1976. [CrossRef]
- A. E. Frias et al., “Using dynamic contrast-enhanced MRI to quantitatively characterize maternal vascular organization in the primate placenta,” Magn. Reson. Med., vol. 73, no. 4, pp. 1570–1578, Apr. 2015. [CrossRef]
- V. H. J. Roberts et al., “Adverse Placental Perfusion and Pregnancy Outcomes in a New Nonhuman Primate Model of Gestational Protein Restriction,” Reprod. Sci. Thousand Oaks Calif, vol. 25, no. 1, pp. 110–119, Jan. 2018. [CrossRef]
- V. H. J. Roberts, J. E. Gaffney, T. K. Morgan, and A. E. Frias, “Placental adaptations in a nonhuman primate model of gestational protein restriction,” J. Dev. Orig. Health Dis., vol. 12, no. 6, pp. 908–914, Dec. 2021. [CrossRef]
- M. C. Schabel et al., “Functional imaging of the nonhuman primate Placenta with endogenous blood oxygen level–dependent contrast,” Magn. Reson. Med., vol. 76, no. 5, pp. 1551–1562, 2016. [CrossRef]
- J. L. Rosenkrantz, J. E. Gaffney, V. H. J. Roberts, L. Carbone, and S. L. Chavez, “Transcriptomic analysis of primate placentas and novel rhesus trophoblast cell lines informs investigations of human placentation,” BMC Biol., vol. 19, no. 1, p. 127, Jun. 2021. [CrossRef]
- F. King, “Ultrastructural differentiation of stromal and vascular components in early macaque placental villi,” Am. J. Anat., vol. 178, no. 1, pp. 30–44, Jan. 1987. [CrossRef]
- M. A. Sheridan et al., “Establishment and differentiation of long-term trophoblast organoid cultures from the human placenta,” Nat. Protoc., vol. 15, no. 10, pp. 3441–3463, Oct. 2020. [CrossRef]
- P. Wolf et al., “In vitro fertilization and embryo transfer in the rhesus monkey,” Biol. Reprod., vol. 41, no. 2, pp. 335–346, Aug. 1989. [CrossRef]
- P. Wolf, S. Thormahlen, C. Ramsey, R. R. Yeoman, J. Fanton, and S. Mitalipov, “Use of assisted reproductive technologies in the propagation of rhesus macaque offspring,” Biol. Reprod., vol. 71, no. 2, pp. 486–493, Aug. 2004. [CrossRef]
- V. H. J. Roberts, J. N. Castro, B. M. Wessel, D. F. Conrad, A. D. Lewis, and J. O. Lo, “Rhesus macaque fetal and placental growth demographics: A resource for laboratory animal researchers,” Am. J. Primatol., vol. 85, no. 8, p. e23526, 2023. [CrossRef]
- P. S. Aranda, D. M. LaJoie, and C. L. Jorcyk, “Bleach gel: a simple agarose gel for analyzing RNA quality,” Electrophoresis, vol. 33, no. 2, pp. 366–369, Jan. 2012. [CrossRef]
- “Protocol for qPCR analysis that corrects for cDNA amplification efficiency.” Accessed: Aug. 07, 2025. [Online]. Available: https://star-protocols.cell.com/protocols/1813.
- J. Schindelin et al., “Fiji: an open-source platform for biological-image analysis,” Nat. Methods, vol. 9, no. 7, pp. 676–682, Jun. 2012. [CrossRef]
- J. Zhou et al., “Development of apical out trophoblast stem cell derived organoids to model early human pregnancy,” iScience, vol. 28, no. 3, p. 112099, Mar. 2025. [CrossRef]
- L. (杨利恒) Yang, P. Liang, H. Yang, and C. B. Coyne, “Trophoblast organoids with physiological polarity model placental structure and function,” J. Cell Sci., vol. 137, no. 5, p. jcs261528, Sep. 2023. [CrossRef]
- Y.-T. Zeng, W.-F. Liu, P.-S. Zheng, and S. Li, “GDF15 deficiency hinders human trophoblast invasion to mediate pregnancy loss through downregulating Smad1/5 phosphorylation,” iScience, vol. 26, no. 10, p. 107902, Sep. 2023. [CrossRef]
- M. A. Sheridan et al., “Characterization of primary models of human trophoblast,” Development, vol. 148, no. 21, p. dev199749, Nov. 2021. [CrossRef]
- J. K. Schmidt et al., “Placenta-derived macaque trophoblast stem cells: differentiation to syncytiotrophoblasts and extravillous trophoblasts reveals phenotypic reprogramming,” Sci. Rep., vol. 10, no. 1, Art. no. 1, Nov. 2020. [CrossRef]
- R. Vento-Tormo et al., “Single-cell reconstruction of the early maternal–fetal interface in humans,” Nature, vol. 563, no. 7731, pp. 347–353, Nov. 2018. [CrossRef]
- A. Arutyunyan et al., “Spatial multiomics map of trophoblast development in early pregnancy,” Nature, vol. 616, no. 7955, Art. no. 7955, Apr. 2023. [CrossRef]
- Baczyk, A. Satkunaratnam, B. Nait-Oumesmar, B. Huppertz, J. C. Cross, and J. C. P. Kingdom, “Complex patterns of GCM1 mRNA and protein in villous and extravillous trophoblast cells of the human placenta,” Placenta, vol. 25, no. 6, pp. 553–559, Jul. 2004. [CrossRef]
- J. Schreiber et al., “Placental Failure in Mice Lacking the Mammalian Homolog of Glial Cells Missing, GCMa,” Mol. Cell. Biol., vol. 20, no. 7, pp. 2466–2474, Apr. 2000. [CrossRef]
- M. J. Jeyarajah et al., “The multifaceted role of GCM1 during trophoblast differentiation in the human placenta,” Proc. Natl. Acad. Sci. U. S. A., vol. 119, no. 49, p. e2203071119, Dec. 2022. [CrossRef]
- C. H. Damsky et al., “Integrin switching regulates normal trophoblast invasion,” Development, vol. 120, no. 12, pp. 3657–3666, Dec. 1994. [CrossRef]
- T. Hori et al., “Trophoblast stem cell-based organoid models of the human placental barrier,” Nat. Commun., vol. 15, no. 1, p. 962, Feb. 2024. [CrossRef]
- B. B. van Rijn, D. Van Opstal, van Koetsveld, Nicole, Knapen, Maarten, J. Gribnau, and O. J. Schaffers, “Generation of Trophoblast Organoids from Chorionic Villus Sampling,” Organoids, vol. 3, no. 1, pp. 54–66. [CrossRef]
- S. Matsumoto et al., “Establishment of macaque trophoblast stem cell lines derived from cynomolgus monkey blastocysts,” Sci. Rep., vol. 10, no. 1, p. 6827, Apr. 2020. [CrossRef]
- R. M. Karvas et al., “Stem-cell-derived trophoblast organoids model human placental development and susceptibility to emerging pathogens,” Cell Stem Cell, vol. 29, no. 5, pp. 810-825.e8, May 2022. [CrossRef]
| Buffer Name | Reagents | Manufacturer & Catalog Number |
|---|---|---|
| Wash Buffer #1 (WB #1) | 500ml PBS, 5ml 100X Pen/Strep |
Gibco 10010023 Gibco 15140148 |
| Wash Buffer #2 (WB #2) | 500ml Ham F12, 5ml 100X Pen/Strep |
Gibco 11765054 Gibco 15140148 |
| Red Blood Cell Lysis Buffer | 10X RBC lysis buffer, sterile ddH2O | Biolegend 420301 |
| Digest Buffer | 60ml 0.25% Trypsin-EDTA, 15ml PBS |
Gibco 25200056 Gibco 15140148 |
| Digest Stop Buffer | 80ml WB #2, 20ml FBS |
Gibco A5670701 |
| Collagenase V Buffer | 13.5ml Ham F12, 1.5mL FBS, 15mg collagenase V |
Gibco 11765054 Gibco A5670701 Sigma-Aldrich C9263 |
| Freeze Buffer #1 | 200µl FBS, 300µl TOM | Gibco A5256701 |
| Freeze Buffer #2 | 100µl DMSO, 400µl TOM | Fisher Scientific D128-1 |
| Media Component | Volume | Stock Concentration |
Final Concentration |
Manufacturer / Catalog Number |
|---|---|---|---|---|
| Advanced DMEM/F-12 | 18.5mL | N/A | N/A | ThermoFisher 12634028 |
| N-2 Supplement | 200µl | 100X | 1X | Fisher Scientific A1370701 |
| B-27 Supplement | 400µl | 50X | 1X | Fisher Scientific 17504044 |
| L-Glutamine | 200µl | 200mM | 2mM | Fisher Scientific 25-030-149 |
| Antibiotic/Antimycotic | 200µl | 100X | 1X | Fisher Scientific 15-240-096 |
| NALC | 200µl | 125mM | 1.25mM | Millipore Sigma A9165-5G |
| A83-01 | 2µl | 5mM | 500nM | Millipore Sigma SML0788-5MG |
| CHIR99021 | 6.98µl | 4.3mM | 1.5µM | Millipore Sigma SML1046-5MG |
| EGF | 10µl | 0.1 µg/µl | 50 ng/mL | ThermoFisher AF-100-15-500UG |
| FGF2 | 200µl | 0.01 µg/µl | 100 ng/mL | ThermoFisher 100-18B-50UG |
| HGF | 100µl | 0.01 µg/µl | 50 ng/mL | ThermoFisher 100-39H-25UG |
| R-Spondin 1 | 20µl | 0.08 µg/µl | 80 ng/mL | ThermoFisher 120-38-20UG |
| Prostaglandin E2 | 20µl | 2.5 mM | 2.5µM | Cayman Chemical NC1775238 |
| Y-2 | 8µl for initial isolation 20µl for maintenance |
5 mM | 2.5µM initial 5µM maintenance |
STEMCELL Technologies 72304 |
| Antigen/Clone # (if applicable) |
Cell Marker | Dilution | Host Species |
Manufacturer & Catalog Number |
|---|---|---|---|---|
| SDC1/Polyclonal | SYN | 1:500-1000 | Rabbit | Proteintech, 10593-1-AP |
| CDH1/ 6B11F11 | CTB | 1:400-800 | Mouse | Proteintech, 60335-1-Ig |
| KI67/ B56 | CTB | 1:500 | Mouse | BD Biosciences, 550609 |
| KRT7/ D1E4 | Pan-Trophoblast | 1:500-1000 | Rabbit | Cell Signaling Technologies, 4465 |
| Phalloidin | Actin | 1:10,000 | N/A | ThermoFisher R415 |
| DAPI | Nuclei | 1ug/ml | N/A | Thermo Fisher D3571 |
| Anti-Mouse IgG1 CF488 | N/A | 1:1000 | Goat | Biotium, 20246 |
| Anti-Mouse IgG2b CF564 | N/A | 1:1000 | Goat | Biotium, 20268 |
| Anti-Rabbit IgG Coralite 488 | N/A | 1:1000 | Goat | Proteintech, SA00013-2 |
| Anti-Rabbit IgG CF637 | N/A | 1:1000 | Goat | Biotium, 20122 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




