Submitted:
07 August 2025
Posted:
07 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. A Primer on Liquid-Liquid Phase Separation in Biology
3. The Architects of the Granular Layer and Beyond
3.1. The Filaggrin Family: Master Scaffolds for LLPS
3.2. The Keratin Cytoskeleton: A Dynamic Cage
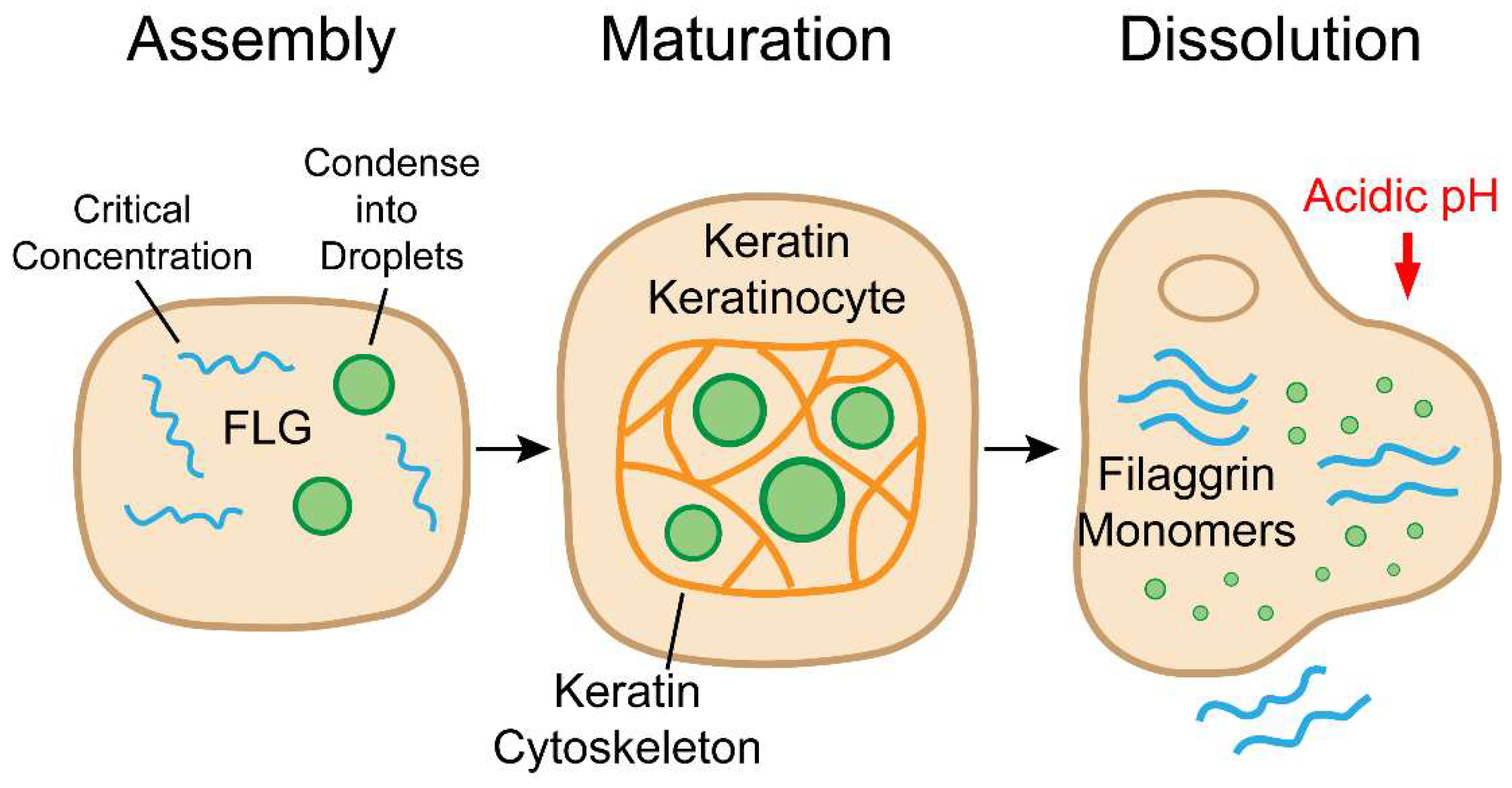
4. Keratohyalin Granules: An LLPS-Driven Lifecycle
4.1. Assembly: A Concentration- and Valency-Dependent Phase Transition
4.2. A Crowded, Structured Liquid: Maturation and Cytoplasmic Organization
4.3. Dissolution: A Multimodally Regulated Switch for Corneoptosis
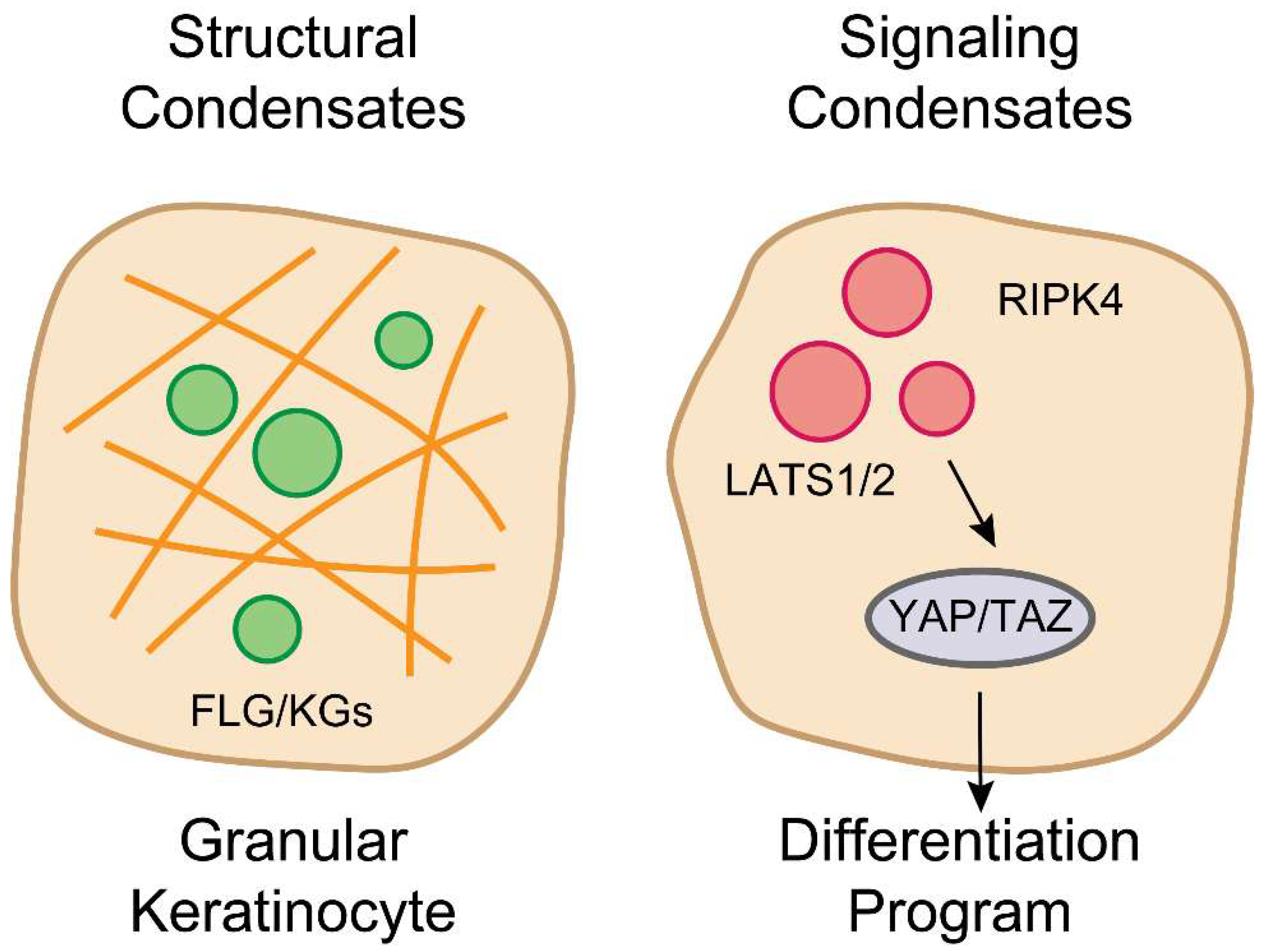
5. The RIPK4-Hippo Axis: A Parallel LLPS-Based Signaling Hub
6. Pathophysiology: When Phase Separation Fails
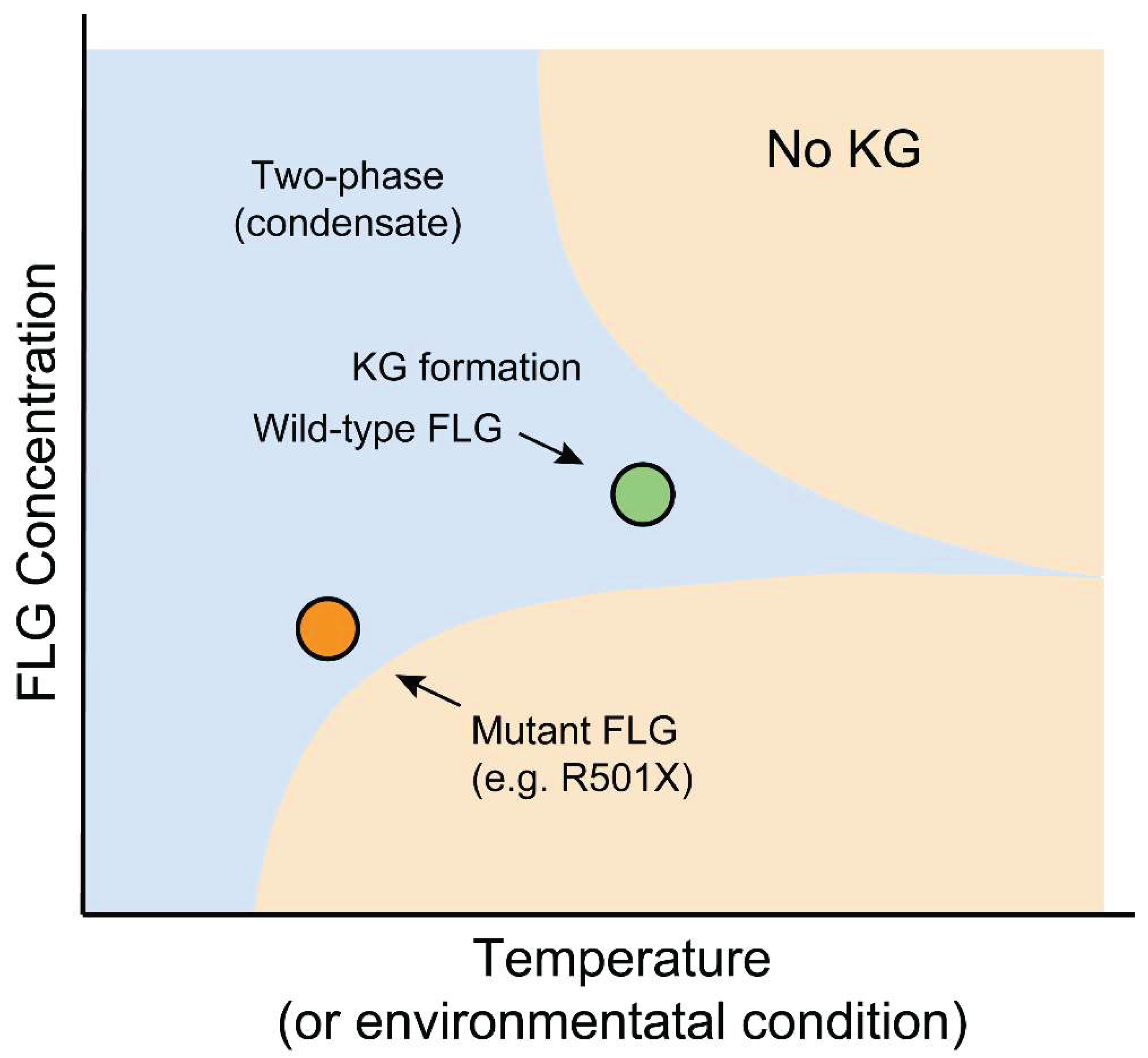
6.1. Atopic Dermatitis and Ichthyosis Vulgaris: Diseases of Altered Critical Concentration
6.2. Bartsocas-Papas Syndrome: A Disease of Defective Signaling Condensates
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Atopic Dermatitis |
| AFM | Atomic Force Microscopy |
| CNV | Copy Number Variation |
| EDC | Epidermal Differentiation Complex |
| FLG | Filaggrin |
| FLG2 | Filaggrin-2 |
| FRAP | Fluorescence Recovery After Photobleaching |
| IDP | Intrinsically Disordered Protein |
| IV | Ichthyosis Vulgaris |
| KG | Keratohyalin Granule |
| LLPS | Liquid-Liquid Phase Separation |
| NMF | Natural Moisturizing Factor |
| PTM | Post-Translational Modification |
| RIPK4 | Receptor-Interacting Serine/Threonine Kinase 4 |
| RPTN | Repetin |
| TCHH | Trichohyalin |
| TG | Trichohyalin Granule |
References
- Matsui, T.; Kadono-Maekubo, N.; Suzuki, Y.; Furuya, T.; Kido, Y.; Yoshida, Y.; Kakegawa, S.; Amagai, M.; Kubo, A. A unique mode of keratinocyte death requires intracellular acidification. Proc. Natl. Acad. Sci. USA 2021, 118, e2020722118. [Google Scholar] [CrossRef]
- Avecilla, A.R.C.; Quiroz, F.G. Cracking the Skin Barrier: Liquid-Liquid Phase Separation Shines under the Skin. JID Innov. 2021, 1, 100036. [Google Scholar] [CrossRef] [PubMed]
- Brody, I. An ultrastructural study on the role of the keratohyalin granules in the keratinization process. J. Ultrastruct. Res. 1959, 3, 84–104. [Google Scholar] [CrossRef] [PubMed]
- Sandilands, A.; Sutherland, C.; Irvine, A.D.; McLean, W.H. Filaggrin in the frontline: role in skin barrier function and disease. J. Cell Sci. 2009, 122, 1285–1294. [Google Scholar] [CrossRef]
- Palmer, C.N.; Irvine, A.D.; Terron-Kwiatkowski, A.; Zhao, Y.; Liao, H.; Lee, S.P.; Goudie, D.R.; Sandilands, A.; Campbell, L.E.; Smith, F.J.; et al. Common loss-of-function variants of the epidermal barrier protein filaggrin are a major predisposing factor for atopic dermatitis. Nat. Genet. 2006, 38, 441–446. [Google Scholar] [CrossRef]
- Smith, F.J.; Irvine, A.D.; Terron-Kwiatkowski, A.; Sandilands, A.; Campbell, L.E.; Zhao, Y.; Liao, H.; Evans, A.T.; Goudie, D.R.; Lewis-Jones, S.; et al. Loss-of-function mutations in the gene encoding filaggrin cause ichthyosis vulgaris. Nat. Genet. 2006, 38, 337–342. [Google Scholar] [CrossRef]
- Brown, S.J.; McLean, W.H.I. One remarkable molecule: filaggrin. J. Investig. Dermatol. 2012, 132, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Banani, S.F.; Lee, H.O.; Hyman, A.A.; Rosen, M.K. Biomolecular condensates: organizers of cellular biochemistry. Nat. Rev. Mol. Cell Biol. 2017, 18, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Quiroz, F.G.; Fiore, V.F.; Levorse, J.; Polak, L.; Wong, E.; Pasolli, H.A.; Fuchs, E. Liquid-liquid phase separation drives skin barrier formation. Science 2020, 367, eaax9554. [Google Scholar] [CrossRef]
- Rai, A.; Pelkmans, L. Liquid droplets in the skin. Science 2020, 367, 1193–1194. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Lu, Z.; Fang, Y.; et al. RIPK4 promotes epidermal differentiation through phase separation and activation of LATS1/2. Dev. Cell 2025, in press. [CrossRef]
- Brangwynne, C.P.; Eckmann, C.R.; Courson, D.S.; Rybarska, A.; Hoege, C.; Gharakhani, J.; Jülicher, F.; Hyman, A.A. Germline P granules are liquid droplets that localize by controlled dissolution/condensation. Science 2009, 324, 1729–1732. [Google Scholar] [CrossRef]
- Alberti, S.; Gladfelter, A.; Mittag, T. Considerations and Challenges in Studying Liquid-Liquid Phase Separation and Biomolecular Condensates. Cell 2019, 176, 419–434. [Google Scholar] [CrossRef] [PubMed]
- Uversky, V.N.; Gillespie, J.R.; Fink, A.L. Why are “natively unfolded” proteins unstructured under physiologic conditions? Proteins 2000, 41, 415–427. [Google Scholar] [CrossRef]
- Pappu, R.V.; Wang, X.; Vitalis, A.; Crick, S.L. A polymer physics perspective on driving forces and mechanisms for protein aggregation. Arch. Biochem. Biophys. 2008, 469, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Flory, P.J. Principles of Polymer Chemistry; Cornell University Press: Ithaca, NY, USA, 1953. [Google Scholar]
- Brangwynne, C.P.; Tompa, P.; Pappu, R.V. Polymer physics of intracellular phase transitions. Nat. Phys. 2015, 11, 899–904. [Google Scholar] [CrossRef]
- Rauscher, S.; Pomès, R. The liquid structure of elastin. eLife 2017, 6, e26526. [Google Scholar] [CrossRef] [PubMed]
- Söding, J.; Zwicker, D.; Sohrabi-Jahromi, S.; Boehning, M.; Kirschbaum, J. Phosphorylation as a switch for the all-or-none formation of membraneless organelles. Trends Cell Biol. 2020, 30, 4–16. [Google Scholar] [CrossRef]
- Choi, H.; Hong, Y.; Najafi, S.; et al. Spontaneous Transition of Spherical Coacervate to Vesicle-Like Compartment. Adv. Sci. 2024, 11, 2305978. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, P.; Zemke, F.; Wagermaier, W.; Linder, M.B. Interfacial Crystallization and Supramolecular Self-Assembly of Spider Silk Inspired Protein at the Water-Air Interface. Materials 2021, 14, 4239. [Google Scholar] [CrossRef]
- Mischke, D.; Korge, B.P.; Marenholz, I.; Volz, A.; Ziegler, A. Genes encoding structural proteins of epidermal cornification and S100 calcium-binding proteins form a gene complex (“epidermal differentiation complex”) on human chromosome 1q21. J. Investig. Dermatol. 1996, 106, 989–992. [Google Scholar] [CrossRef]
- Quiroz, F.G.; Chilkoti, A. Sequence heuristics to encode phase behaviour in intrinsically disordered protein polymers. Nat. Mater. 2015, 14, 1164–1171. [Google Scholar] [CrossRef]
- Rauscher, S.; Baud, S.; Miao, M.; Keeley, F.W.; Pomès, R. Proline and glycine control protein self-organization into elastomeric or amyloid fibrils. Structure 2006, 14, 1667–1676. [Google Scholar] [CrossRef]
- Mohamad, J.; Sarig, O.; Godsel, L.M.; et al. Filaggrin 2 deficiency results in abnormal cell-cell adhesion in the cornified cell layers and causes peeling skin syndrome type A. J. Investig. Dermatol. 2018, 138, 1736–1743. [Google Scholar] [CrossRef] [PubMed]
- Margolis, D.J.; Gupta, J.; Apter, A.J.; et al. Filaggrin-2 variation is associated with more persistent atopic dermatitis in African American subjects. J. Allergy Clin. Immunol. 2014, 133, 784–789. [Google Scholar] [CrossRef]
- Lee, S.C.; Kim, I.G.; Marekov, L.N.; et al. The structure of human trichohyalin. J. Biol. Chem. 1993, 268, 12164–12176. [Google Scholar] [CrossRef] [PubMed]
- Ü Basmanav, F.B.; Cau, L.; Tafazzoli, A.; et al. Mutations in three genes encoding proteins involved in hair shaft formation cause uncombable hair syndrome. Am. J. Hum. Genet. 2016, 99, 1292–1304. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Bouameur, J.E.; Bär, J.; et al. A keratin scaffold regulates epidermal barrier formation, mitochondrial lipid composition, and activity. J. Cell Biol. 2015, 211, 1057–1075. [Google Scholar] [CrossRef] [PubMed]
- Reichheld, S.E.; Muiznieks, L.D.; Keeley, F.W.; Sharpe, S. Direct observation of structure and dynamics during phase separation of an elastomeric protein. Proc. Natl. Acad. Sci. USA 2017, 114, E4408–E4415. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, K.; O'Sullivan, J.; Missero, C.; et al. Exome sequence identifies RIPK4 as the Bartsocas-Papas syndrome locus. Am. J. Hum. Genet. 2012, 90, 69–75. [Google Scholar] [CrossRef]
- Schlegelmilch, K.; Mohseni, M.; Kirak, O.; et al. Yap1 acts downstream of α-catenin to control epidermal proliferation. Cell 2011, 144, 782–795. [Google Scholar] [CrossRef]
- Yoneda, K.; Hohl, D.; McBride, O.W.; et al. The human loricrin gene. J. Biol. Chem. 1992, 267, 18060–18066. [Google Scholar] [CrossRef]
- Beutel, O.; Maraspini, R.; Pombo-García, K.; et al. Phase Separation of Zonula Occludens Proteins Drives Formation of Tight Junctions. Cell 2019, 179, 923–936.e11. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.L.; Huang, W.P.; Fang, Y.; et al. Fabrication of “Spongy Skin” on Diversified Materials Based on Surface Swelling Non-Solvent-Induced Phase Separation. ACS Appl. Mater. Interfaces 2021, 13, 57783–57792. [Google Scholar] [CrossRef]
- Ji, X.; Li, R.; Liu, G.; et al. Phase separation-based electrospun Janus nanofibers loaded with Rana chensinensis skin peptides/silver nanoparticles for wound healing. Mater. Des. 2021, 209, 109864. [Google Scholar] [CrossRef]
- Lee, H.; Kim, H.J.; Shin, Y.; Kim, D.H. Phase-separated stretchable conductive nanocomposite to reduce contact resistance of skin electronics. Sci. Rep. 2024, 14, 1393. [Google Scholar] [CrossRef]
- Wang, S.; Neufurth, M.; Schepler, H.; et al. Liquid–liquid phase transition as a basis for novel materials for skin repair and regeneration. J. Mater. Chem. B. 2024, 12, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Pei, S.; Zhang, P.; et al. Liquid-liquid phase separation throws novel insights into treatment strategies for skin cutaneous melanoma. BMC Cancer 2023, 23, 388. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



