Submitted:
05 August 2025
Posted:
07 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
- The actual level of PK activity in the reticulocytes of PKD patients and the impact of reticulocytosis on the total PK activity (activity in erythrocytes isolated from whole blood purified from leukocytes)
- The reliability of PK activity and the PK:HK ratio for differential diagnosis between PKD and other anemias with similar clinical presentations and reduced PK activity
- The critical level of residual PK activity associated with clinically relevant hemolysis and anemia
2. Results
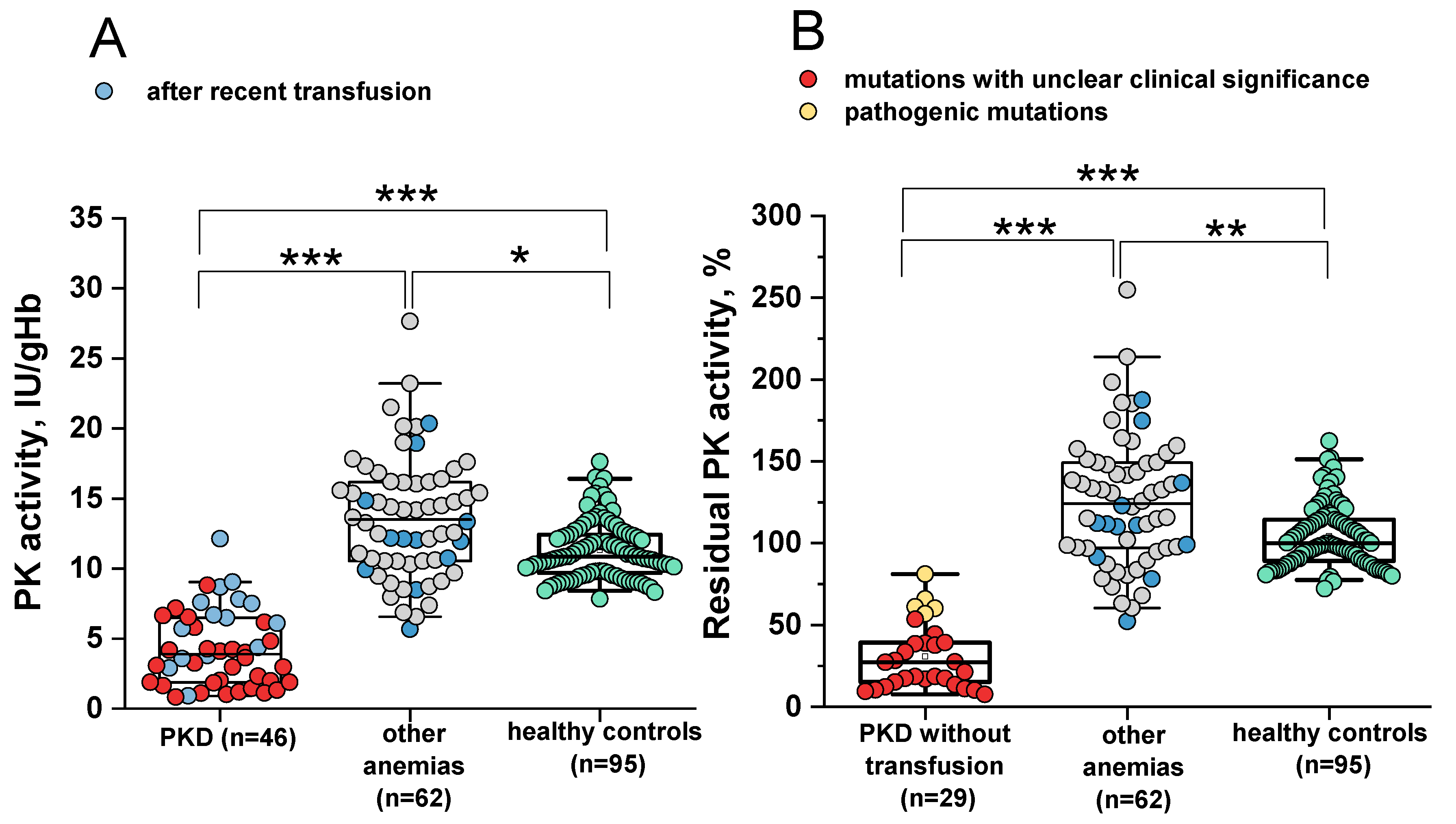
2.1. Sensitivity and Specificity of the PK Activity Assay for the Differential Diagnosis of PK Deficiency
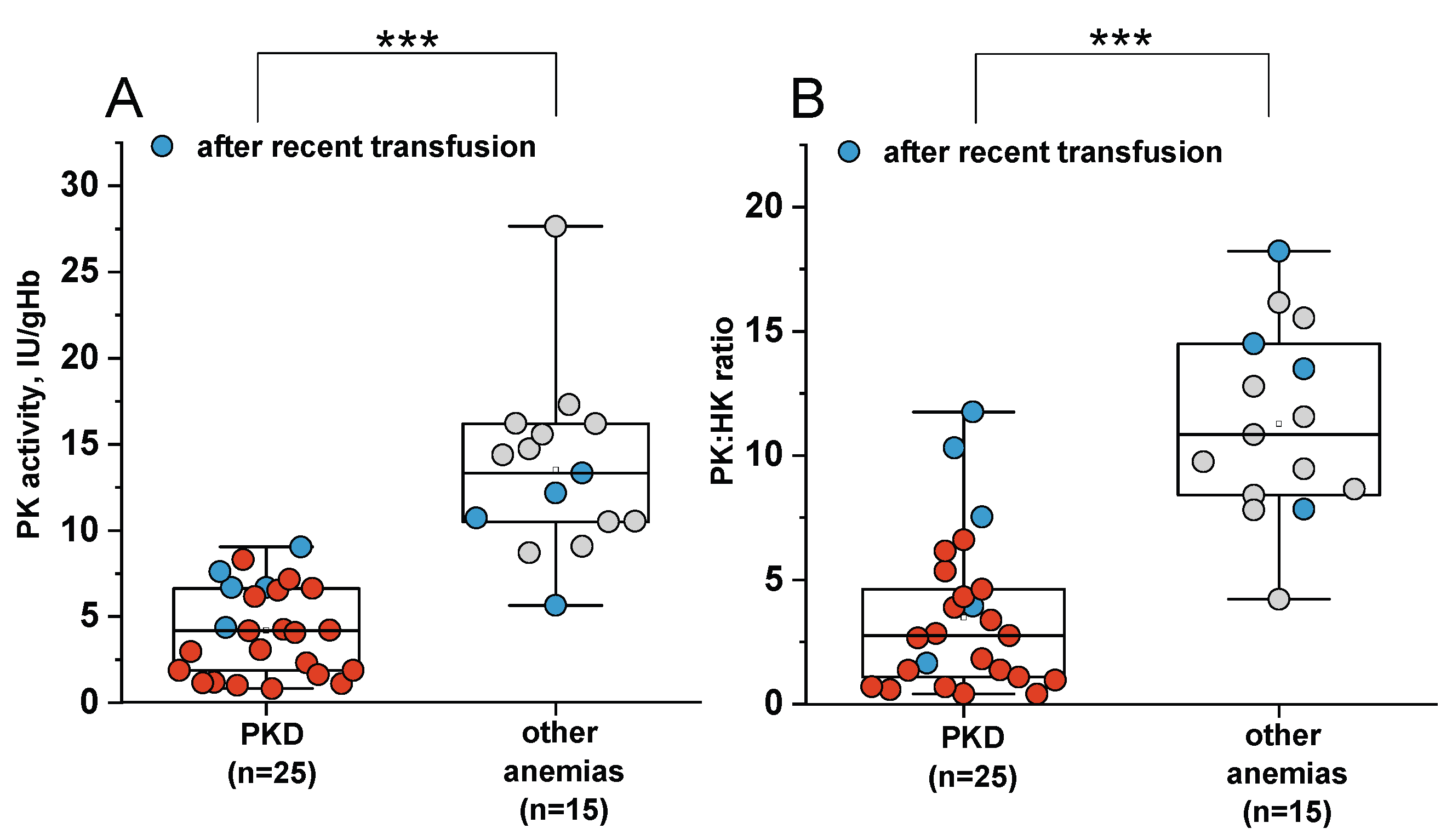
2.2. Comparison of Sensitivity and Specificity of PK Activity and the PK:HK Ratio in the Diagnosis of PKD
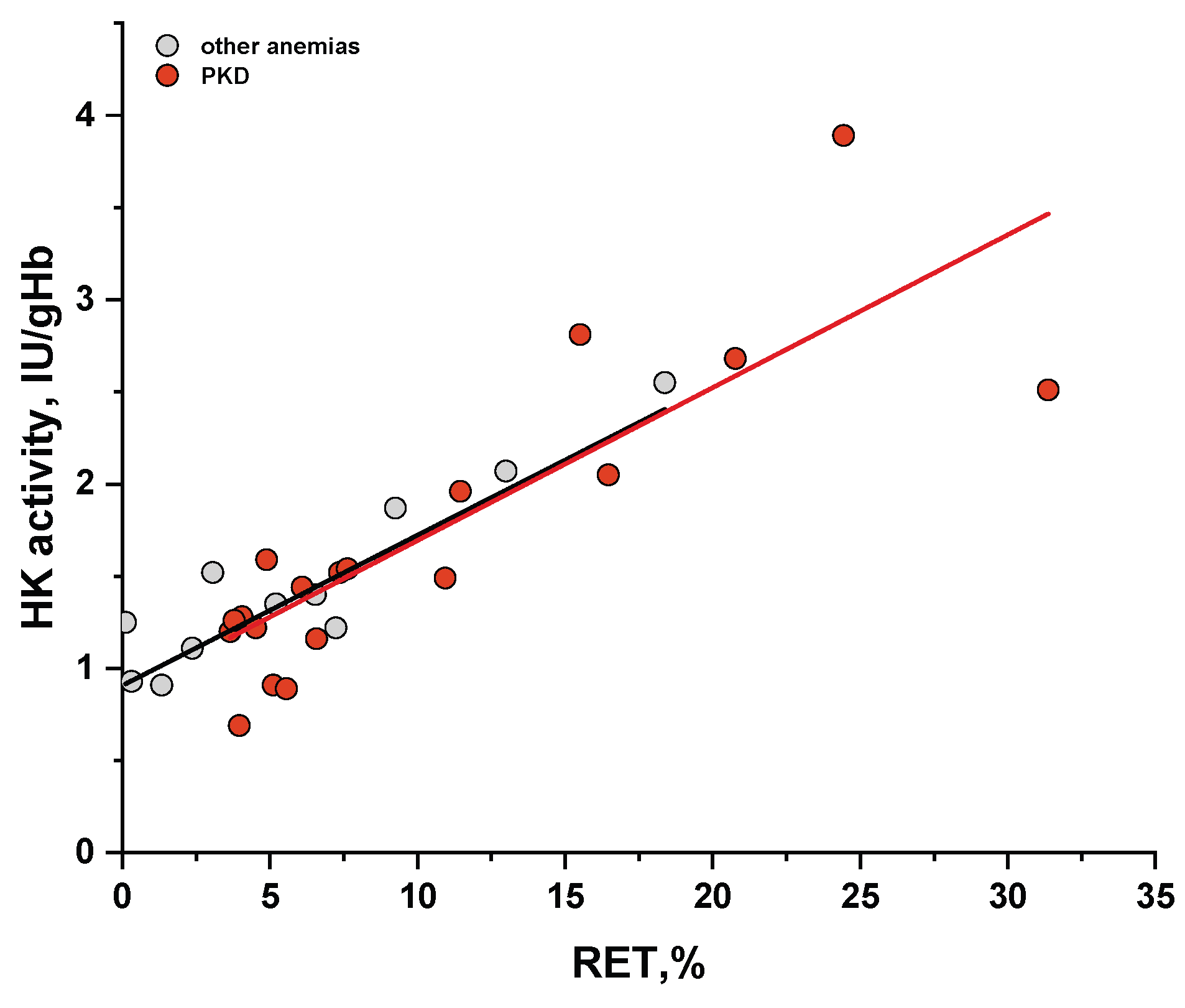
2.3. The Relationship Between Measured PK Activity and Reticulocyte Count in the Blood
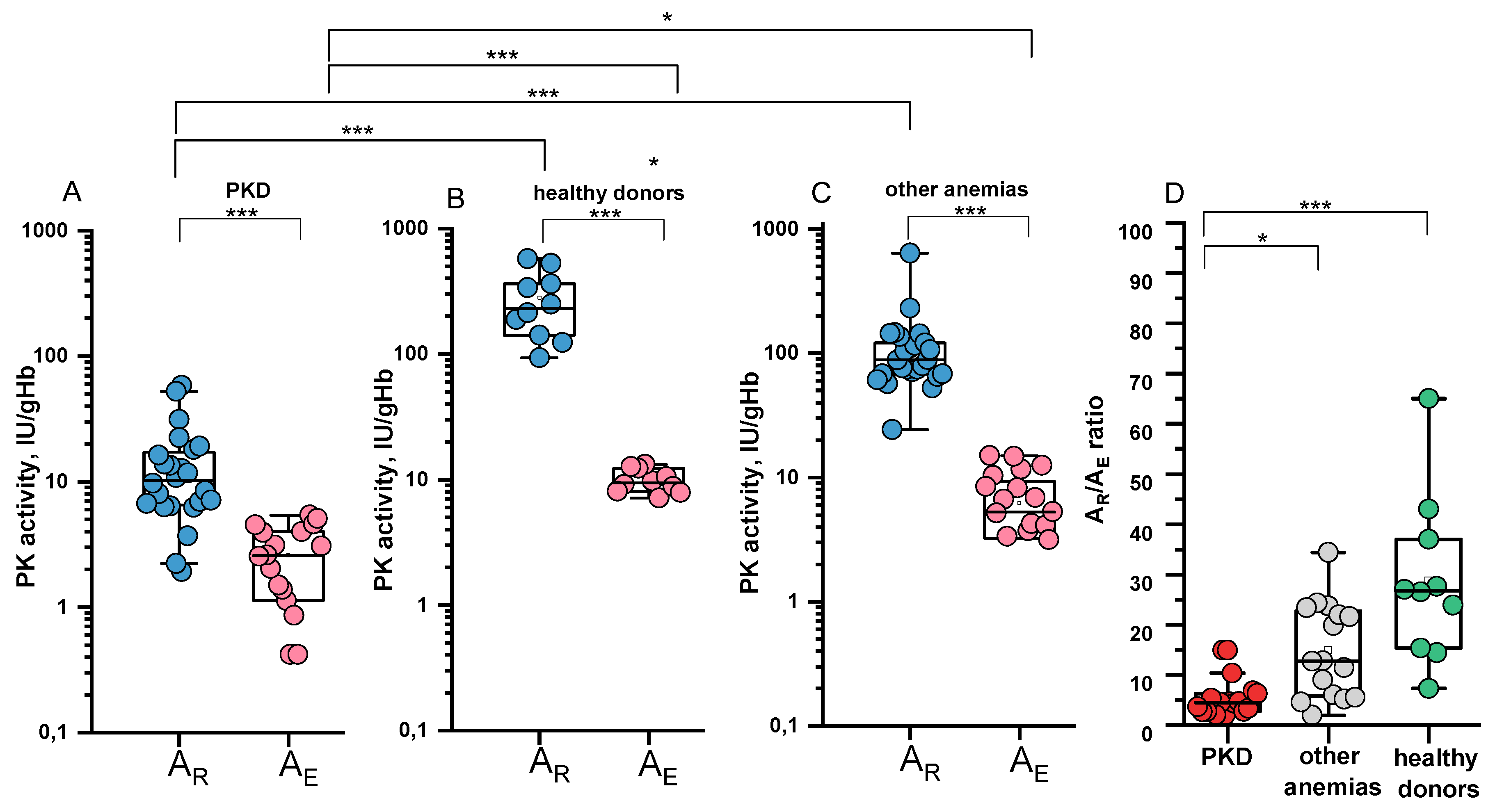
2.4. Specific PK Activities in the Reticulocyte and Erythrocyte and Their Ratio
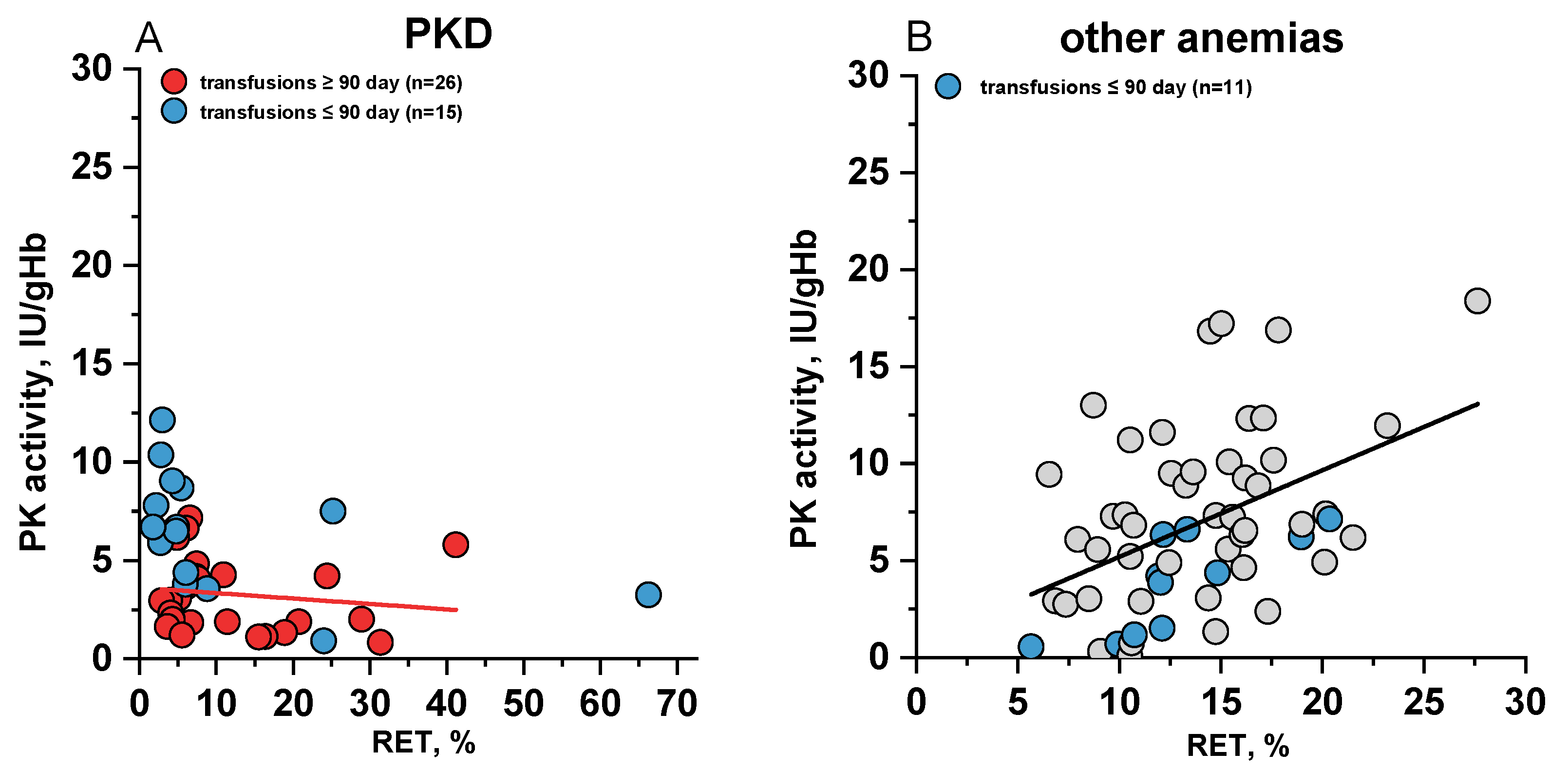
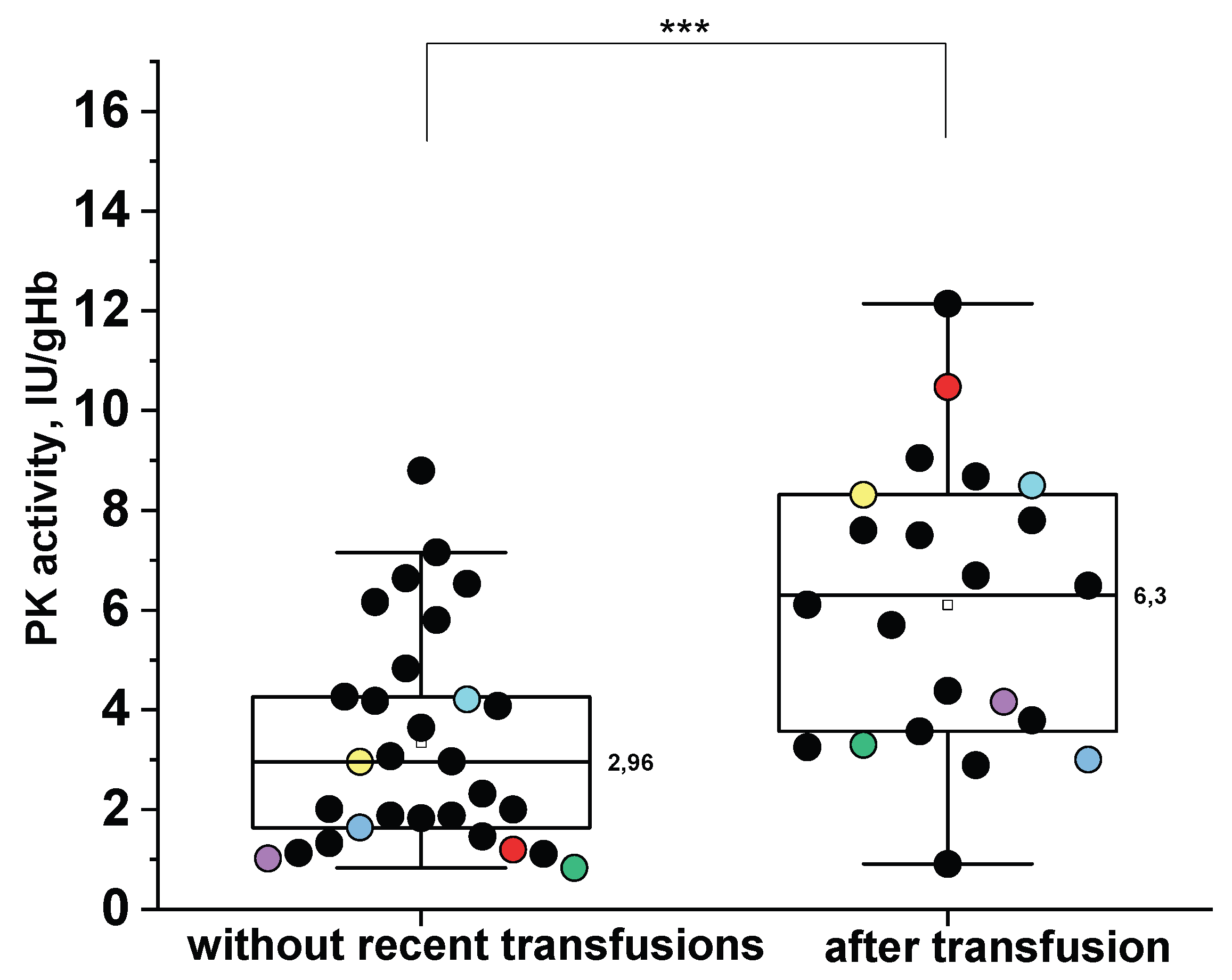
2.5. The Impact of Donor Red Blood Cells Transfusions on the PKD Diagnosis
3. Discussion
4. Conclusions
- The data from our study suggest that reticulocytosis does not significantly contribute to the observed total PK activity. This is due to the markedly reduced (23-fold) PK activity in reticulocytes of PKD patients compared to normal reticulocyte activity
- A distinct difference in the ratio of specific PK activities between reticulocytes and erythrocytes is observed in PKD patients and healthy donors. Healthy donors show a more pronounced difference in PK activity between these two cell types than patients
- Key factors influencing the sensitivity and specificity of PK activity include transfusions of donor erythrocytes and decreased PK activity in other types of anemia, including within reticulocytes
- In patients receiving regular transfusions (every 1-3 months) PK deficiency can be diagnosed by isolating reticulocytes and measuring specific PK activity in the reticulocytes
- Slightly decreased PK activity is insufficient for diagnosing PK deficiency. In 97% of cases, residual PK activity greater than 66% in patients without recent transfusions indicates anemia not related to PK deficiency
- The PK:HK ratio demonstrates higher sensitivity than PK activity when diagnosing PKD relative to healthy donors. However, it is less specific for the differential diagnosis of PKD and other anemias due to potential reduced PK activity and frequently observed reticulocytosis in other anemias. For this ratio to be used effectively, it is necessary to establish a reference range that accounts for the PK:HK ratio in patients with other types of anemia
5. Materials and Methods
5.1. Study Population
5.2. Reagents
5.3. Routine Hematological Studies
5.4. Genetic Analysis
5.5. Blood Preparation for Studies
5.6. Isolation of Reticulocytes
5.7. Measurement of Enzyme Activity in Erythrocytes
5.8. Determination of Specific
5.9. Statistical Analysis
Author Сontributions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Secrest MH, Storm M, Carrington C, et al. Prevalence of pyruvate kinase deficiency: A systematic literature review. Eur J Haematol. 2020, 105, 173–184.
- Beutler E, Forman L, Rios-Larrain E. Elevated pyruvate kinase activity in patients with hemolytic anemia due to red cell pyruvate kinase “deficiency”. Am J Med. 1987, 83, 899–904.
- Martinov M V, Plotnikov AG, Vitvitsky VM, Ataullakhanov FI. Deficiencies of glycolytic enzymes as a possible cause of hemolytic anemia. Biochim Biophys Acta - Gen Subj. 2000, 1474, 75–87.
- Grace RF, Bianchi P, van Beers EJ, et al. Clinical spectrum of pyruvate kinase deficiency: data from the pyruvate kinase deficiency natural history study. Blood. 2018, 131, 2183–2192.
- Fermo E, Bianchi P, Chiarelli LR, et al. Red cell pyruvate kinase deficiency: 17 new mutations of the PK-LR gene. Br J Haematol. 2005, 129, 839–846.
- Al-Samkari H, Shehata N, Lang-Robertson K, et al. Diagnosis and management of pyruvate kinase deficiency: international expert guidelines. Lancet Haematol. 2024, 11, e228–e239.
- Bianchi P, Fermo E, Glader B, et al. Addressing the diagnostic gaps in pyruvate kinase deficiency: Consensus recommendations on the diagnosis of pyruvate kinase deficiency. Am J Hematol. 2019, 94, 149–161.
- Yozgat AK, Erdem AY, Kaçar D, Özbek NY, Yaralı N. Pyruvate kinase deficiency mimicking congenital dyserythropoietic anemia type I. Turk J Pediatr. 2022, 64, 951–955.
- Addonizio K, Al-Samkari H, Glader B, et al. Pyruvate Kinase (PK) Protein and Enzyme Levels in the Diagnosis and Clinical Phenotype of PK Deficiency. Blood 2019, 1343515.
- Lezon-Geyda K, Rose MJ, McNaull MA, et al. Pklr Intron Splicing-Associated Mutations and Alternate Diagnoses Are Common in Pyruvate Kinase Deficient Patients with Single or No Pklr Coding Mutations. Blood 2018, 1323607.
- Fattizzo B, Cavallaro F, Marcello APML, Vercellati C, Barcellini W. Pyruvate Kinase Deficiency: Current Challenges and Future Prospects. J Blood Med 2022, 13461–471.
- Lakomek M, Schröter W, De Maeyer G, Winkler H. On the diagnosis of erythrocyte enzyme defects in the presence of high reticulocyte counts. Br J Haematol. 1989, 72, 445–451.
- Zimran A, Torem S, Beutler E. The in vivo ageing of red cell enzymes: direct evidence of biphasic decay from polycythaemic rabbits with reticulocytosis. Br J Haematol. 1988, 69, 67–70.
- Jansen G, Koenderman L, Rijksen G, Cats BP, Staal GE. Characteristics of hexokinase, pyruvate kinase, and glucose-6-phosphate dehydrogenase during adult and neonatal reticulocyte maturation. Am J Hematol. 1985, 20, 203–215.
- Zanella A, Fermo E, Bianchi P, Valentini G. Red cell pyruvate kinase deficiency: Molecular and clinical aspects. British Journal of Haematology. 2005, 130, 11–25. [Google Scholar] [CrossRef]
- Gök V, Leblebisatan G, Gürlek Gökçebay D, et al. Pyruvate kinase deficiency in 29 Turkish patients with two novel intronic variants. Br J Haematol. 2024, 205, 236–242.
- Al-Samkari H, Addonizio K, Glader B, et al. The pyruvate kinase (PK) to hexokinase enzyme activity ratio and erythrocyte PK protein level in the diagnosis and phenotype of PK deficiency. Br J Haematol. 2021, 192, 1092–1096.
- Andres O, Loewecke F, Morbach H, et al. Hereditary spherocytosis is associated with decreased pyruvate kinase activity due to impaired structural integrity of the red blood cell membrane. Br J Haematol. 2019, 187, 386–395.
- de Wilde JRA, Ruiter TJJ, Bos J, et al. Ex vivo activation of pyruvate kinase improves red blood cell metabolism and hydration in hereditary spherocytosis. Blood Red Cells Iron. 2025, 100005.
- Rab MAE, van Oirschot BA, van Straaten S, et al. Decreased Activity and Stability of Pyruvate Kinase in Hereditary Hemolytic Anemia: A Potential Target for Therapy By AG-348 (Mitapivat), an Allosteric Activator of Red Blood Cell Pyruvate Kinase. Blood. 2019, 134, 3506–3506.
- Traets MJM, Bos JF, van der Veen S, et al. Pyruvate Kinase Function Correlates With Red Blood Cell Properties and Clinical Manifestations in Sickle Cell Disease. Am J Hematol. 2025, 100, 785–796.
- Rab MAE, Bos J, van Oirschot BA, et al. Decreased activity and stability of pyruvate kinase in sickle cell disease: a novel target for mitapivat therapy. Blood. 2021, 137, 2997–3001.
- Fattizzo B, Vercellati C, Marcello A, et al. Glycolytic activity and in vitro effect of the pyruvate kinase activator AG-946 in red blood cells from low-risk myelodysplastic syndromes patients: A proof-of-concept study. American journal of hematology. 2024, 99, 1201–1204. [Google Scholar] [CrossRef] [PubMed]
- Utsugisawa T, Uchiyama T, Toki T, et al. Enzymatic changes in red blood cells of diamond-blackfan anemia. Tohoku J Exp Med. 2021, 255, 49–55.
- Grace RF, Barcellini W. Management of pyruvate kinase deficiency in children and adults. Blood. 2020, 136, 1241–1249.
- Jansen G, Koenderman L, Rijksen G, Cats BP, Staal GEJ. Characteristics of hexokinase, pyruvate kinase, and glucose-6-phosphate dehydrogenase during adult and neonatal reticulocyte maturation. Am J Hematol. 1985, 20, 203–215.
- Piomelli S, Seaman C, Corash L, Beutler E. How do red cell enzymes age? Hypothesis and facts. British Journal of Haematology. 1986, 64, 407–409. [Google Scholar] [CrossRef] [PubMed]
- Lakomek M, Neubauer B, Lühe A von d. , Hoch G, Schröter W, Winkler H. Erythrocyte pyruvate kinase deficiency: Relations of residual enzyme activity, altered regulation of defective enzymes and concentrations of high-energy phosphates with the severity of clinical manifestation. Eur J Haematol. 1992, 49, 82–92.
- van Dijk MJ, de Wilde JRA, Bartels M, et al. Activation of pyruvate kinase as therapeutic option for rare hemolytic anemias: Shedding new light on an old enzyme. Blood Rev;61.
- Lenzner C, Nürnberg P, Jacobasch G, Gerth C, Thiele B-J. Molecular Analysis of 29 Pyruvate Kinase–Deficient Patients From Central Europe With Hereditary Hemolytic Anemia. Blood. 1997, 89, 1793–1799.
- Rijksen G, Schipper-Kester GPM, Staal GEJ, Veerman AJP. Diagnosis of pyruvate kinase deficiency in a transfusion-dependent patient with severe hemolytic anemia. Am J Hematol. 1990, 35, 187–193.
- Beutler, E. Red cell metabolism. 2nd ed. Orlando: Gruen & Stratton Inc; 1984. 11 p.
- Russell B, Suwanarusk R, Borlon C, et al. A reliable ex vivo invasion assay of human reticulocytes by Plasmodium vivax. Blood. 2011, 118, e74–81.
| Patient | Gene mutation |
cDNA nucleotide substitution |
Total PK activity IU/gHb |
AR, IU/gHb | AE, IU/gHb | АR/АE | PK:HK ratio | |
|---|---|---|---|---|---|---|---|---|
| Allele1 | Allele2 | |||||||
| Patients with pyruvate kinase deficiency | ||||||||
| Severe condition | ||||||||
| 1. * | PKLR | hom c.101-1G>A | 7.8 | 14±5 | 7.7±0.7 | 1.8±0.7 | n/d | |
| 2. | PKLR | hom c.401T>A | 5.8±0.4 | n/d | n/d | n/d | n/d | |
| 3. | PKLR | hom c.695-2A>C | 2.01±0.07 | n/d | n/d | n/d | n/d | |
| 4. * | PKLR | hom c.1079G>A | 7.60 | 23±11 | 7.52±0.36 | 3.1±1.5 | 10.3 | |
| 5. * | PKLR | hom c.1269+1G>A | 12.1 | n/d | n/d | n/d | n/d | |
| 6. | PKLR | hom c.1529G>A | 1.65 | 6.2±0.7 | 1.39±0.09 | 4.5±0.6 | 1.37 | |
| 6. * | 3 | n/d | n/d | n/d | n/d | |||
| 7. | PKLR | hom c.1529G>A | 1.20 | 2.23±0.08 | 1.13±0.01 | 1.98±0.04 | 1.35 | |
| 7. * | 10.47 | n/d | n/d | n/d | n/d | |||
| 8. | PKLR | hom c.1529G>A | 1.83 | 7.0±0.3 | 1.50±0.03 | 4.64±0.11 | n/d | |
| 9. | PKLR | hom c.1529G>A | 1.13 | 3.7±2.3 | 0.86±0.43 | 4.3±3.4 | 0.39 | |
| 10. | PKLR | hom c.1529G>A | 1.88 | n/d | n/d | n/d | 0.96 | |
| 11. | PKLR | hom c.1529G>A | 1.88 | n/d | n/d | n/d | 0.70 | |
| 12. * | PKLR | c.101-1G>A | c.1318G>T | 5.7±0.2 | n/d | n/d | n/d | n/d |
| 13. * | PKLR | c.948C>A | c.848T>A | 6.11 | 53±7 | 5.08±0.17 | 10.4±1.4 | 7.53 |
| 14. | PKLR | c.1130T>C | c.1318G>T | 8.8±0.02 | n/d | n/d | n/d | n/d |
| 15. * | PKLR | c.1174G>A | c.1456C>T | 7.51±0.08 | n/d | n/d | n/d | n/d |
| 16. * | PKLR | c.1174G>A | c.1456C>T | 9.05 | 11.7±0.3 | 8.80±0.14 | 1.33±0.04 | 11.8 |
| 17. * | PKLR | c.1436G>A | c.487C>T | 2.89±0.2 | n/d | n/d | n/d | n/d |
| 18. | PKLR | c.1456C>T | c.1157C>T | 4.18 | 8±3 | 4.0±0.8 | 2.0±0.9 | 2.75 |
| 19. * | PKLR | Ex 1-2 del | c.1529G>A | 3.25±0.09 | n/d | n/d | n/d | n/d |
| 20. * | PKLR | Ex 1-2 del | c.1529G>A | 3.3 | 12.69±2.16 | 2.8±0.25 | 4.5±0.9 | n/d |
| 20. | 0.83 | n/d | n/d | n/d | 0.41 | |||
| 21. * | PKLR | Ex 1-2 del | c.1529G>A | 4.16 | 6.39±2.38 | 4.3±0.3 | 1.5±0.7 | n/d |
| 21. | 1.02 | n/d | n/d | n/d | 0.68 | |||
| 22. * | PKLR | c.-63G>A | c.1529G>A | 6.69 | n/d | n/d | n/d | 8.92 |
| 23. * | PKLR | с.101-1G>A | c.1529G>A | 3.6±0.2 | n/d | n/d | n/d | n/d |
| 24. * | PKLR | c.460G>A | c.1529G>A | 4.38 | n/d | n/d | n/d | 1.65 |
| 25. * | PKLR | c.994G>A | c.1529G>A | 3.8±0.4 | n/d | n/d | n/d | n/d |
| 26. | PKLR | c.1079G>A | c.1529G>A | 0.91 | 1.9±0.9 | 0.4±0.3 | 5±3 | n/d |
| 27. | PKLR | c.1223C>T | c.1529G>A | 4 | n/d | n/d | n/d | n/d |
| 28. * | PKLR | c.1583A>T | c.1436G>A | 6.49 | n/d | n/d | n/d | n/d |
| 29. * | PKLR | c.1637T>C | c.1529G>A | 8.5 | 13.5±0.12 | 8.27± 0.02 | 1.63 ± 0.015 | n/d |
| 29. | 4.21 | n/d | n/d | n/d | 1.08 | |||
| Moderate condition | ||||||||
| 30. | PKLR | hom c.1318G>A | 1.11 | 9.8±0.9 | 0 | n/d | 0.4 | |
| 31. | PKLR | c.932T>C | c.1456C>T | 2.97 | 7.1±0.96 | 2.6±0.096 | 2.7±0.4 | 4.31 |
| 32. | PKLR | c.1231G>T | c.1456C>T | 4.08 | 19.2±3.2 | 3.06±0.25 | 6.3±1.2 | 2.65 |
| 33. | PKLR | c.1231G>T | c.1456C>T | 4.26 | 8.46±4 | 3.1±0.6 | 2.9±1.4 | 2.84 |
| 34. * | PKLR | c.1130T>C | c.1456C>T | 8.7±0.5 | n/d | n/d | n/d | n/d |
| 35. | PKLR | c.1594C>T | c.1456C>T | 2.96 | n/d | n/d | n/d | 2.35 |
| 35. * | 8.31 | n/d | n/d | n/d | n/d | |||
| 36. | PKLR | c.1130T>C | c.1456C>T | 6.64 | 16±6 | 4.5±1.2 | 3.6±1.6 | n/d |
| Mild condition | ||||||||
| 37. | PKLR | c.932T>C | c.1456C>T | 1.46±0.17 | n/d | n/d | n/d | n/d |
| 38. | PKLR | c.1076G>A | c.1456C>T | 4.83±0.3 | n/d | n/d | n/d | n/d |
| 39. | PKLR | c.1181C>T | c.1456C>T | 6.16 | 31.3±1.0 | 4.61±0.19 | 6.8±0.4 | 3.88 |
| 40. | PKLR | c.1181C>T | c.1456C>T | 7.16 | 18±3 | 5.4±0.6 | 3.3±0.7 | 6.15 |
| 41. | PKLR | c.1195del | c.1456C>T | 3.07 | 7±6 | 2.6±0.4 | 2.6±1.0 | 3.38 |
| 42. | PKLR | c.1291G>A | c.1529G>A | 6.53 | 59±6 | 3.9±0.4 | 15±2 | 5.35 |
| 43. | PKLR | c.665G>A | c.1429A>G | 3.64 | n/d | n/d | n/d | n/d |
| 44. | PKLR | c.1072G>T | c.1529G>A | 1.33 | 6.27±0.5 | 0.42±0.2 | 15±8 | n/d |
| 45. | PKLR | c.1583A>T | c.1510C>T | 2.32 | 10.8±0.4 | 2.03±0.03 | 5.3±0.2 | 1.81 |
| 46. | PKLR | hom c.1529G>A | 2 | n/d | n/d | n/d | n/d | |
| Median | 3.9 | 10.27 | 2.57 | 4.5 | 2.75 | |||
| Patients with other anemias | ||||||||
| 47. | ALAD | het c.375del | 14.38 | n/d | n/d | n/d | 9.46 | |
| 48. | ANK1 | het c.5097-33G>A | 19 | 76.1 | 14.8 | 5.15 | n/d | |
| 49. | ANK1 | het c.596dup frameshift ter | 15.0 | 74.5±1.1 | 0 | n/d | n/d | |
| 50. | ANK1 | het c.4104+4A>G | 8.50 | 120±20 | 5.2±0.8 | 23±6 | n/d | |
| PIEZO1 | het c.3284A>C | |||||||
| 51. | ANK1 | het с.3329_3336 delinsACAAG | 16.13 | 71±9 | 11.7±1.6 | 6.1±1.1 | n/d | |
| 52. * | ANK1 | het c.3778T>C | 14.8 | 110±30 | 8±2 | 14±5 | n/d | |
| 53. * | ANK1 | het c.1814del | 13.3 | 65±17 | 8.7±1.5 | 7.5±1.3 | 14.50 | |
| 54. | ANK1 | het c.596dup frameshift ter | 15.4 | 61±4 | 5.3±0.9 | 11.5±2.1 | n/d | |
| 55. * | ANK1 | het c.4153C>T | 11.9 | 136±14 | 6.9±0.8 | 20±3 | n/d | |
| 56. | ANK1 | het c.2325dupG | 13.3 | 115 | 3.36 | 34.4 | n/d | |
| 57. | GPI | c.1039С>T | c.1612С>A | 27.7±0.5 | n/d | n/d | n/d | 10.8±0.4 |
| 58. | HBB | het c.193G>T | 10.5 | n/d | n/d | n/d | 8.41 | |
| 59. * | HK1 | c.1951G>A | c.2128G>A | 12.2 | 79.1±1.1 | 9.3±0.3 | 8.5±0.3 | 18.22 |
| 60. * | HK1 | homo c.34C>T | 5.66 | 144±3 | 5.5±0.4 | 26.2±2.0 | 14.2 | |
| HBB | homo c.316-106 C>G | |||||||
| 61. | KCNN4 | het c.940T>C | 16.2 | 130±40 | 8±4 | 17±10 | 11.6 | |
| 62. | PIEZO1 | het c.7483_7488dup | 9.60 | 68±3 | 3.2±1.0 | 21±7 | n/d | |
| 63. | PIEZO1 | het c.7483_7489dup | 10.5 | 53±2 | 4.1±0.7 | 13±2 | n/d | |
| 64. | PIGA | c.264delA | c.715+1G>A | 16.4 | 56.8 | 10,3 | 5.49 | n/d |
| 65. * | RPL11 | het c.45delT | 10.7 | n/d | n/d | n/d | 7.84 | |
| 66. * | RPS19 | het c.3G>A | 9.92 | 80±2 | 6.4±0.2 | 12.5±0.6 | n/d | |
| 67. | SLC4A1 | het c.1030C>T | 15.6 | 74±12 | 8.2±1.6 | 9±2 | 12.8 | |
| 68. | SLC4A1 | het c.1030C>T | 10.5 | 88.3±1.3 | 3.7±0.3 | 24±2 | 7.81 | |
| 69. | SLC4A1 | het c.749del | 20.2 | 230±50 | 0 | n/d | n/d | |
| 70. | SPTA1 | het c.82C>T | 16.1 | 600±400 | 0 | n/d | n/d | |
| 71. | SPTA1 | het c.2222A>T | 11.1 | 150±60 | 6.7±1.9 | 22±10 | n/d | |
| 72. | SPTA1 | het c.4019C>A | 14.7 | n/d | n/d | n/d | 16.2 | |
| NF1 | het c.4614G>A | |||||||
| KDM6A | het c.2308C>T | |||||||
| 73. | SPTA1 | het c.5645_5647del | 17.3 | n/d | n/d | n/d | 15.5 | |
| FLNA | het c.1790_1791delTT | |||||||
| COL3A1 | het c.689A>T | |||||||
| CFH | het c.2407T>A | |||||||
| 74. | SPTA1 | c.4339-99C>T | c.6421C>T | 13.6 | 24.1 | 12.6 | 1.91 | n/d |
| 75. | SPTA1 | het c.2671C>T | 23.2 | 68.8 | 15.2 | 4,54 | n/d | |
| HBB | het c.364G>C | |||||||
| 76. | SPTB | het c.566+1G>A | 17.1 | 104±3 | 4.3±0.8 | 24±5 | n/d | |
| 77. | SPTB | het c.1912C>T | 14.5 | 89±13 | 0 | n/d | n/d | |
| PKLR | het c.1456C>T | |||||||
| 78. | SPTB | het c.5800_5801insCAGG | 8.72 | 65±6 | 0 | n/d | 4.21 | |
| 79. | ANK1 | het c.4462C>T | 16.2 | n/d | n/d | n/d | 8.65 | |
| 80. | SF3B1 | n/d | 9.08 | 107±10 | 8.4±0.3 | 12.7±1.3 | 9.75 | |
| Median | 13.5 | 88.3 | 5.2 | 12.7 | 10.8 | |||
| Healthy donors (n=95) | ||||||||
| Median | 10.85 | 231.5 | 9.4 | 26.7 | 13.5 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




