Submitted:
03 August 2025
Posted:
05 August 2025
You are already at the latest version
Abstract
Keywords:
Introduction: Regulation of Infant Formula and Operation Stork Speed
Regulatory Processes for New Formulas
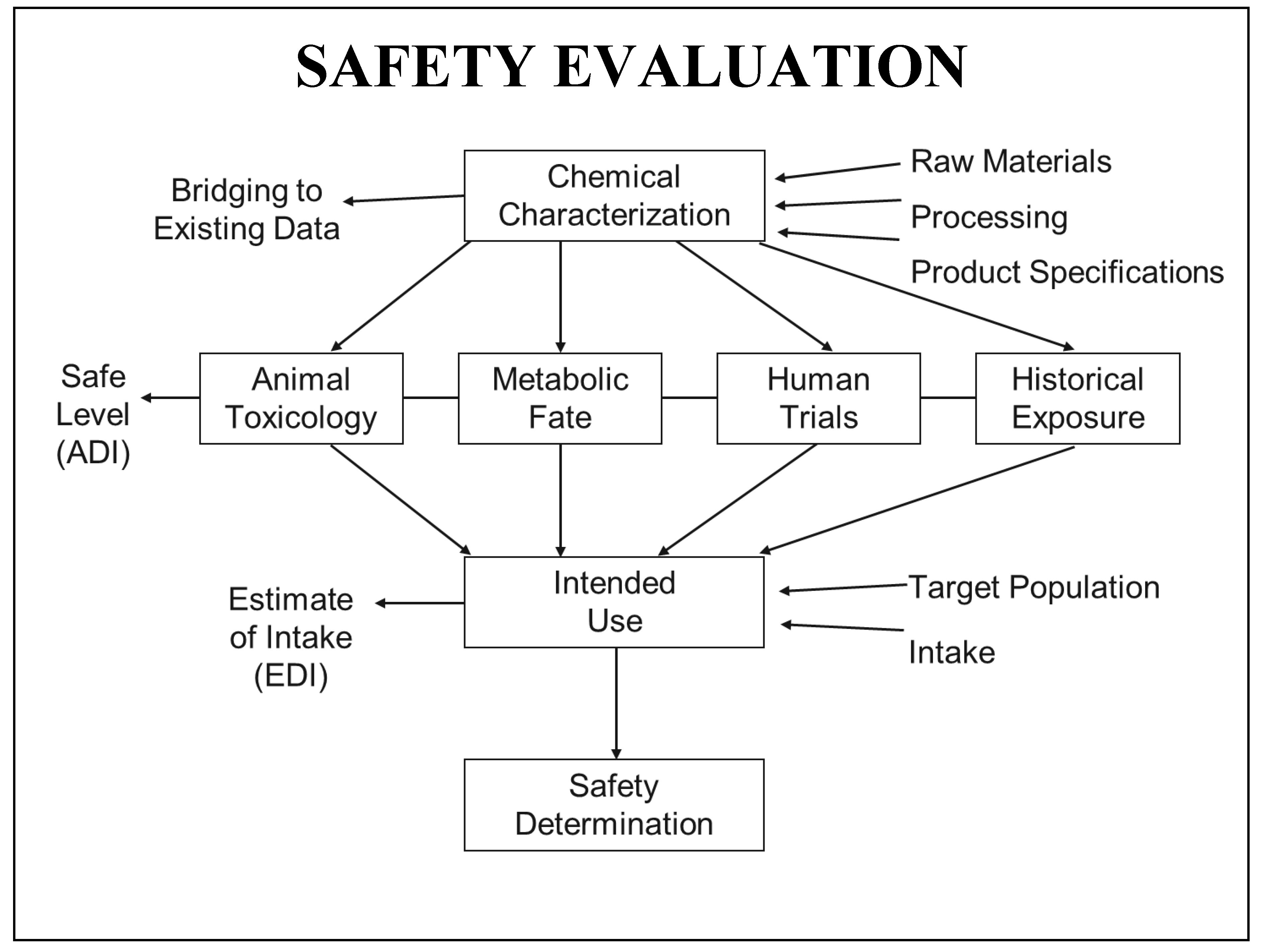
Safety Components and GRAS Process
Impact of Environmental Chemicals on Infant Development
Heavy Metals
International Guidelines
New Approach Methods (NAMs) for Basic Research and Discovery in Risk Science
Safety by Design
Conclusion
Acknowledgments
Financial Support
Abbreviations
References
- Video of FDA panel on infant formula, June 4, 2025, https://www.youtube.com/live/MmE6rlMJdwA).
- National Academies of Sciences, Engineering, and Medicine (NASEM). 2024. Challenges in Supply, Market Competition, and Regulation of Infant Formula in the United States. The National Academies Press. Washington, DC. [CrossRef]
- U.S. FDA, Long-Term National Strategy to Increase the Resiliency of the U.S. Infant Formula Market. https://www.fda.gov/food/infant-formula-guidance-documents-regulatory-information/long-term-national-strategy-increase-resiliency-us-infant-formula-market. Posted January 10, 2025; Accessed June 4, 2025.
- National Academies of Sciences, Engineering, and Medicine. 2025. Protein Quality and Growth Monitoring Studies: Quality Factor Requirements for Infant Formula. Washington, DC: The National Academies Press. [CrossRef]
- U.S. FDA, Docket FDA-2020-D-1922. 2023. Guidance for Industry: Recommendations for Submission of Chemical and Technological Data for Direct Food Additive Petitions. Update from March 2009. Posted October 17, 2023.
- Federal Register 81(159). 2016. 54960-55055. https://www.govinfo.gov/content/pkg/FR-2016-08-17/pdf/2016-19164.pdf.
- Institute of Medicine. Conflict of Interest in Medical Research, Education, and Practice. National Academies Press. 2009. Washington, DC.
- Neltner T, Maffini M. Generally Recognized as Secret: Chemicals Added to Food in the United States. National Research Defense Council, 2014.
- Redbook 2000. Guidance for Industry and Other Stakeholders: Toxicological Principles for the Safety Assessment of Food Ingredients. https://www.fda.gov/files/food/published/Toxicological-Principles-for-the-Safety-Assessment-of-Food-Ingredients.pdf.
- International Conference on Harmonization. (ICH M3 (R2) Non-clinical safety studies for the conduct of human clinical trials for pharmaceuticals – Scientific guideline. 2013. https://www.ema.europa.eu/en/ich-m3-r2-non-clinical-safety-studies-conduct-human-clinical-trials-pharmaceuticals-scientific-guideline#current-effective-version-9045.
- Organization for Economic Cooperation and Development (OECD). Guidelines for the Testing of Chemicals. Updated 2025. https://www.oecd.org/en/topics/sub-issues/testing-of-chemicals/test-guidelines.
- U.S. FDA. GRAS Notices. https://www.hfpappexternal.fda.gov/scripts/fdcc/index.cfm?set=GRASNotices.
- Rodríguez-Barranco M, Lacasaña M, Aguilar-Garduño C, Alguacil J, Gil F, González-Alzaga, B et al. Association of arsenic, cadmium and manganese exposure with neurodevelopment and behavioural disorders in children: a systematic review and meta-analysis. Sci. Total Environ. 454-455 (2013) 562-77. [CrossRef]
- Sanders AP, Claus Henn B, Wright RO. Perinatal and Childhood Exposure to Cadmium, Manganese, and Metal Mixtures and Effects on Cognition and Behavior: A Review of Recent Literature. Curr. Environ. Health Rep. 2(3) (2015) 284-94. [CrossRef]
- Rahman A, Granberg C, Persson LA. Early life arsenic exposure, infant and child growth, and morbidity: a systematic review. Arch. Toxicol. 91 (2017) 3459-3467. [CrossRef]
- Shea, KM. American Academy of Pediatrics Committee on Environmental Health. Pediatric exposure and potential toxicity of phthalate plasticizers. Pediatrics. 111(6 Pt 1) (2003) 1467-74. [CrossRef]
- van Beijsterveldt IALP, van Zelst BD, de Fluiter KS, van den Berg SAA, van der Steen M, Hokken-Koelega ACS. Poly- and perfluoroalkyl substances (PFAS) exposure through infant feeding in early life. Environ. Int. 164 (2022) 107274. [CrossRef]
- Li D, Shi Y, Yang L, Xiao L, Kehoe DK, Gun'ko YK, Boland JJ, et al., Microplastic release from the degradation of polypropylene feeding bottles during infant formula preparation. Nat. Food. 1(11) (2020)746-754. [CrossRef]
- Pajurek M, Mikolajczyk S, Warenik-Bany M. Occurrence and dietary intake of dioxins, furans (PCDD/Fs), PCBs, and flame retardants (PBDEs and HBCDDs) in baby food and infant formula. Sci. Total Environ. 903 (2023) 166590. [CrossRef]
- Hatzidaki E, Pagkalou M, Katsikantami I, Vakonaki E, Kavvalakis M, Tsatsakis AM, et al. Endocrine-Disrupting Chemicals and Persistent Organic Pollutants in Infant Formulas and Baby Food: Legislation and Risk Assessments. Foods. 12 (2023) 1697. [CrossRef]
- Soni S, Kurian JW, Kurian C, Chakraborty P, Paari KA. Food additives and contaminants in infant foods: a critical review of their health risk, trends and recent developments. Food Prod. Process Nutr. 6 (2024) 63. [CrossRef]
- Consumer Reports. (2025) https://www.consumerreports.org/babies-kids/baby-formula/baby-formula-contaminants-test-results-a7140095293. Accessed July 30, 2025.
- U.S. FDA. Closer to Zero: Reducing Childhood Exposure to Contaminants from Foods. (2025) https://www.fda.gov/food/environmental-contaminants-food/closer-zero-reducing-childhood-exposure-contaminants-foods. Accessed , 2025.
- Bair, EC. A Narrative Review of Toxic Heavy Metal Content of Infant and Toddler Foods and Evaluation of United States Policy. Front Nutr. 9 (2022) 919913. [CrossRef]
- European Commission. (2023) Regulation (EU) 2023/915 of 25 April 2023 on maximum levels for certain contaminants in food and repealing Regulation (EC) No 1881/2006. Official Journal of the European Union\ 2023;L 119\(103\-57\. https://eur-lex.europa.eu/eli/reg/2023/915/oj/eng. (Amended July 16, 2024; Accessed July 9, 2025).
- Food Standards Australia New Zealand. (2023) P1028 - Infant Formula Products. https://www.foodstandards.gov.au/consumer/special-purpose-foods/infant-formula-products Published July 26, 2023; Accessed July 19, 2025.
- Government of Canada. (2024) List of Contaminants and Other Adulterating Substances in Foods. In: F. A. N. D. Health Canada's Bureau of Chemical Safety. ed. Ottawa, Canada\: Government of Canada\; 2024. https://www.canada.ca/en/health-canada/services/food-nutrition/food-safety/chemical-contaminants/contaminants-adulterating-substances-foods.html. (Published December 2024; Modified February 2025; Accessed July 9, 2025).
- Food and Agriculture Organization of the United Nations and World Health Organization. (2024) General Standard For Contaminants And Toxins In Food And Feed (Codex Stan 193-1995). Revised 2024. https://www.fao.org/fao-who-codexalimentarius/sh-proxy/fr/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCXS%2B193-1995%252FCXS_193e.pdf. (Accessed July 9, 2025).
- Andrade VM, Aschner M, Marreilha Dos Santos AP. Neurotoxicity of metal mixtures. Adv. Neurobiol. 18 (2017) 227-265. [CrossRef]
- Martín-Carrasco I, Carbonero-Aguilar P, Dahiri B, Moreno IM, Hinojosa M. Comparison between pollutants found in breast milk and infant formula in the last decade: A review. Sci. Total Environ. 875 (2023) 162461. [CrossRef]
- U.S. EPA. (2020) New Approach Methods Work Plan. https://www.epa.gov/chemical-research/epa-new-approach-methods-work-plan-reducing-use-vertebrate-animals-chemical (updated December 2021; Accessed July 9, 2025).
- U.S. FDA. (2017) FDA’S Predictive Toxicology Roadmap. https://www.fda.gov/media/109634/download. (content current as of March 5, 2024; Accessed July 9, 2025).
- Toxicology in the 21st Century (Tox21). (2023) https://tox21.gov/wp-content/uploads/2024/02/Tox21_FactSheet_Jan2023.pdf (Accessed July 9, 2025).
- Knudsen TB, Spencer RM, Pierro JD, Baker NC. Computational biology and in silico toxicodynamics. Curr. Opin. Toxicol. 23 (2020)1 19-126. [CrossRef]
- European Directive 2006/141/EC. https://eur-lex.europa.eu/eli/dir/2006/141/oj/eng.
- Dietrich R, Jessberger N, Ehling-Schulz M, Märtlbauer E, Granum PE. The Food Poisoning Toxins of Bacillus cereus. Toxins (Basel). 13(2) (2021) 98. [CrossRef]
- Ling N, Forsythe S, Wu Q, Ding Y,. Zhang J, Zeng H, et al., Insights into Cronobacter sakazakii Biofilm Formation and Control Strategies in the Food Industry. Engineering. 6(4) (2020) 393-405. [CrossRef]
- U.S. FDA. Infant Formula, Posted May 13, 2025; https://www.fda.gov/food/resources-you-food/infant-formula. Accessed July 28, 2025.
| USA | Canada | Australia/New Zealand | European Union |
| **Lead (0.01ppm for baby foods, no level for formula) | Lead (0.01 ppm for formula) | Lead (0.02 mg/kg for formula) | Lead (0.01mg/kg for liquid formula, 0.02 mg/kg for powder) |
| **Cadmium( Action levels pending for baby food, not formula) | Tin (250 mg/kg, canned food) | Tin (50 mg/kg, canned formula) | |
| **Arsenic (Action levels pending for baby food, not formula) | Vinyl Chloride(0.01 mg/kg) | Cadmium (0.01mg/kg for liquid formula, 0.005 mg/kg for powder) | |
| Aluminum (0.05 mg/100 ml) | Arsenic (0.01 mg/kg for liquid formula, 0.020mg/kg for powder) | ||
| Acrylonitrile (0.02 mg/kg) | Polycyclic aromatic hydrocarbons (1 ug/kg) | ||
| Melamine (1 mg/kg, 0.15 mg/kg) | |||
| Mycotoxins(0.025ug/kg) |
| Regulatory Reform | Operation Stork Speed launched to modernize infant formula oversight after 2022 shortages. Current FDA processes are slow and complex, making it difficult for new formulas to enter the market. |
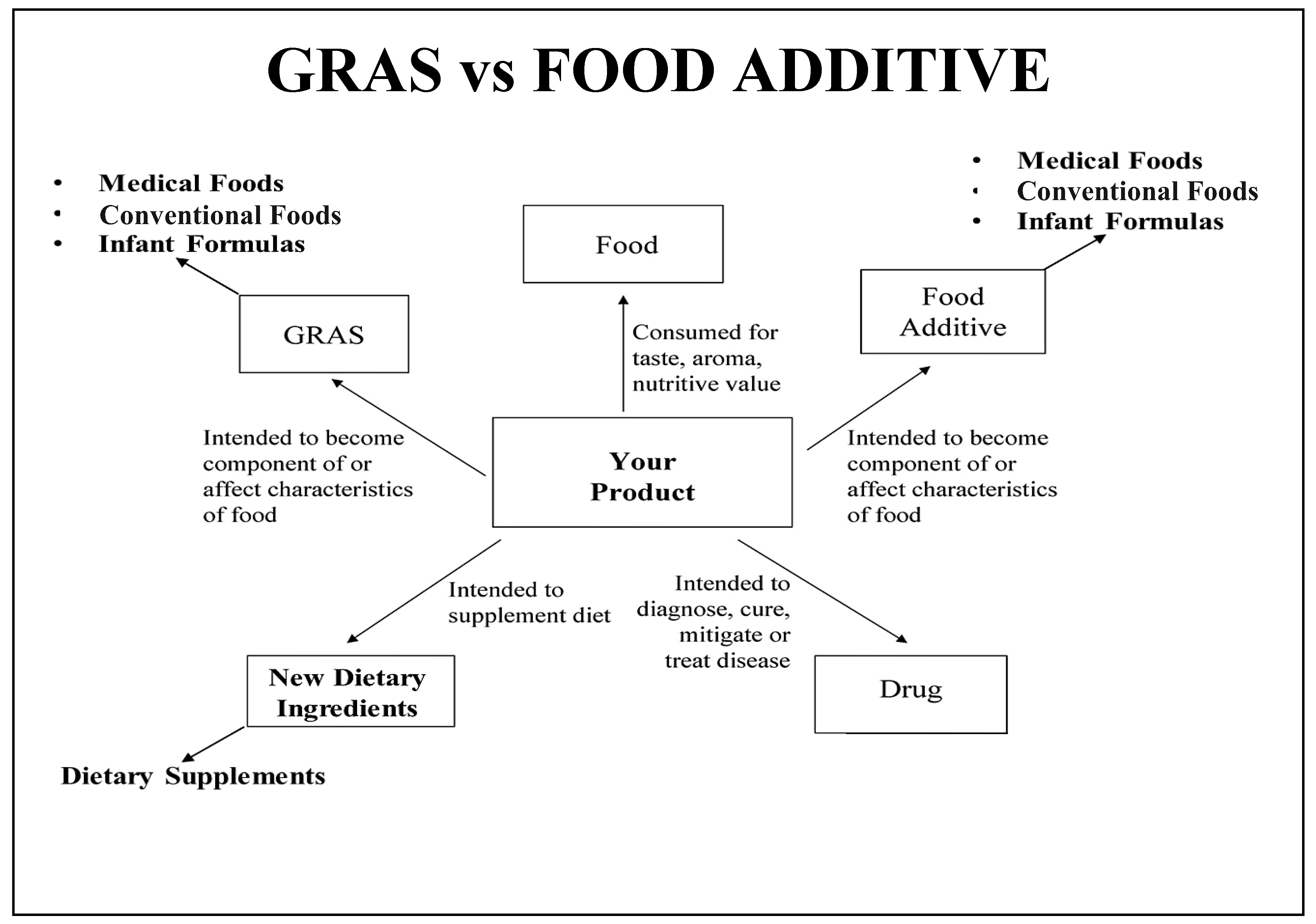
| Ingredient Safety | GRAS and food additive pathways require safety data, but food additive petitions require more safety information and can not be marketed until FDA approval is granted. |
| Contaminant Risks | Heavy metals, PFAS and other toxins can be found in formulas and infants can be at increased risk of effects. US lacks enforceable limits, unlike EU, Canadian and Australian counterparts. |
| Testing Innovations | NAMS may be a helpful non-animal toxicology method |
| Call for Action | Legislative updates, supply chain transparency and alignment with global safety standards are needed. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




