Submitted:
30 July 2025
Posted:
06 August 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
- Coefficients of the CM’s characteristic polynomial have the same sign.
- Hurwitz determinants are all positive.
Results and Discussion
Initial Considerations of Ecological Control
External and Internal Drivers
One Species Systems
Self-Regulation and Internal Dynamics
Keystone Species and Node Relevance
Two Species Systems
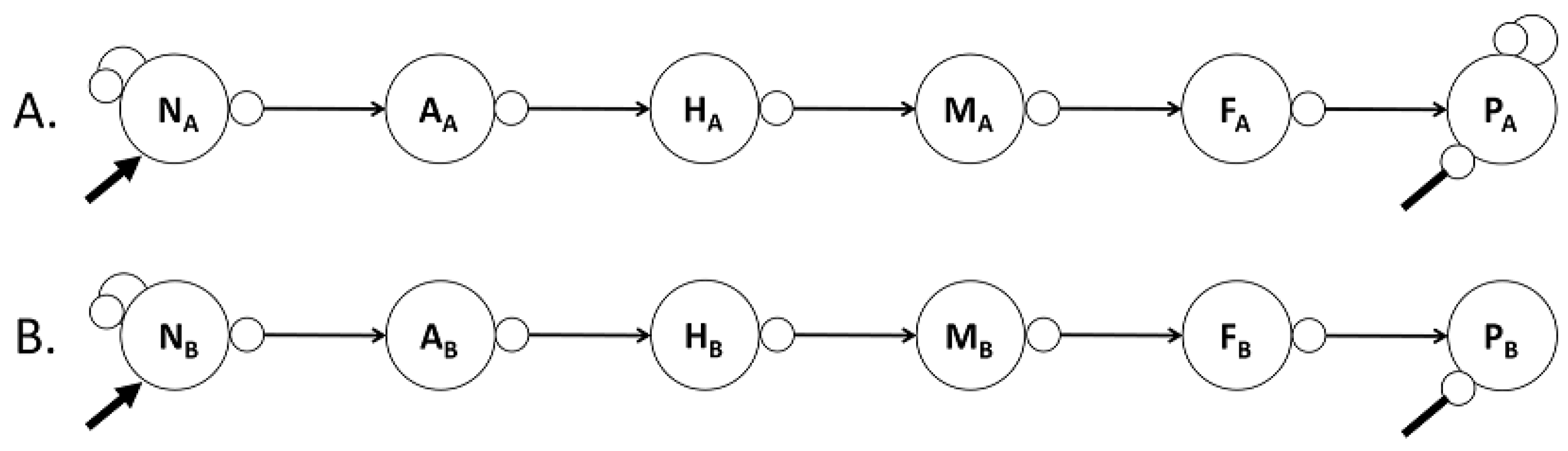
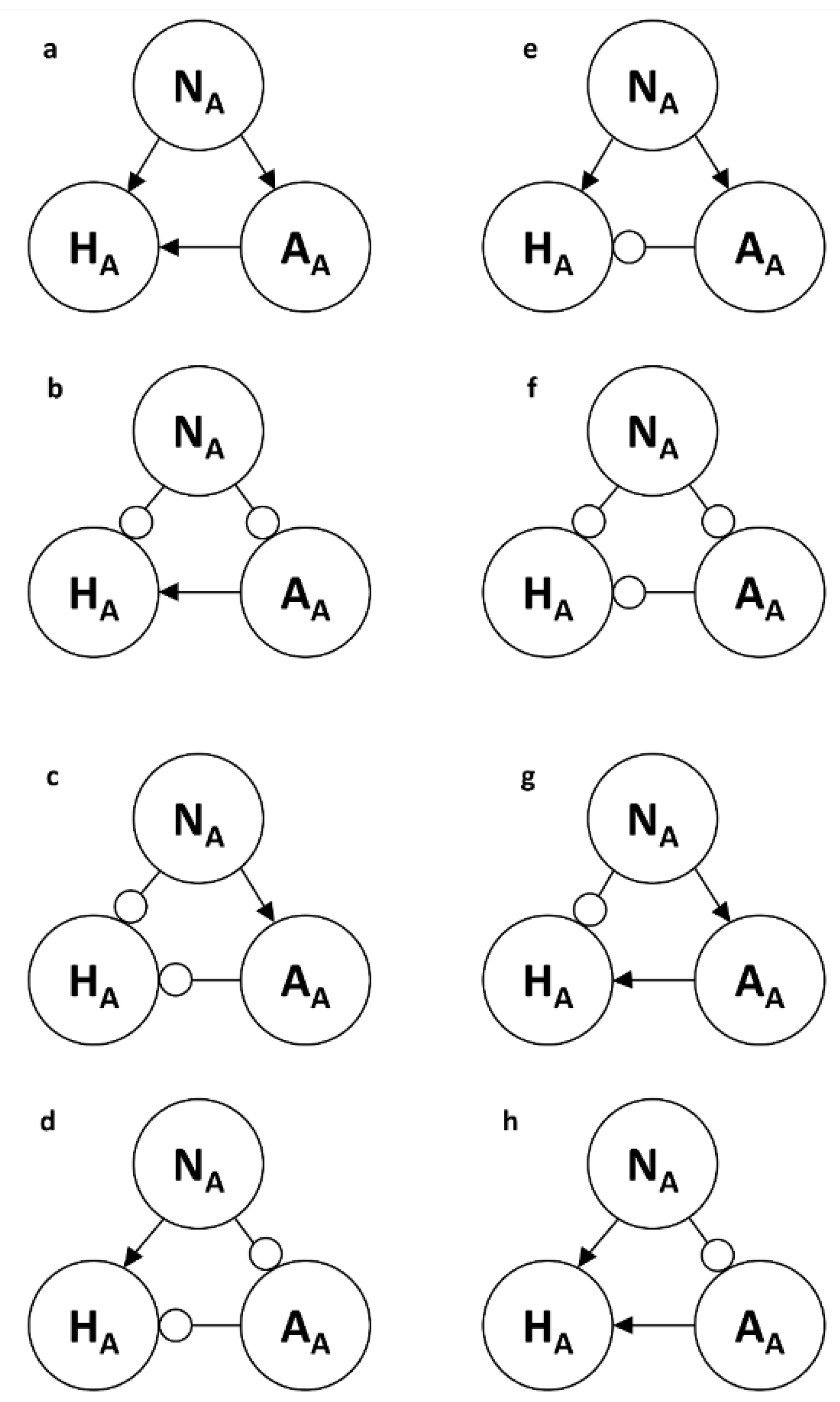
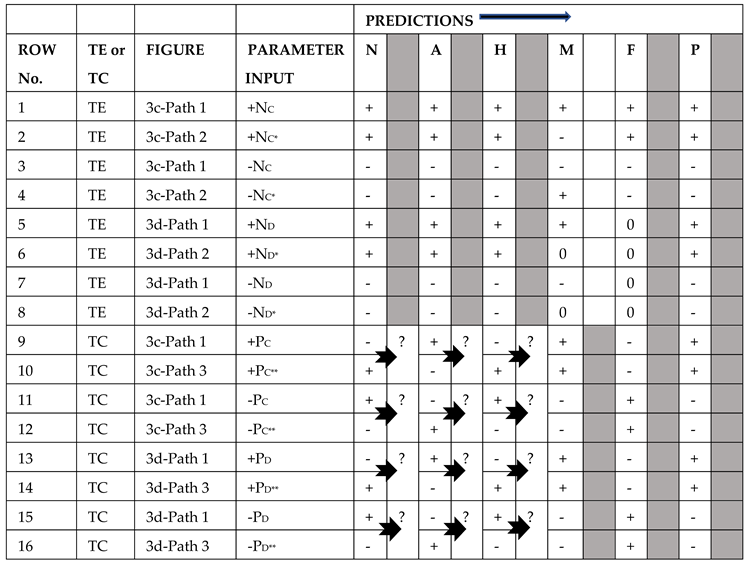
Food Chains
Are Trophic Cascades and Escalades Controllers?
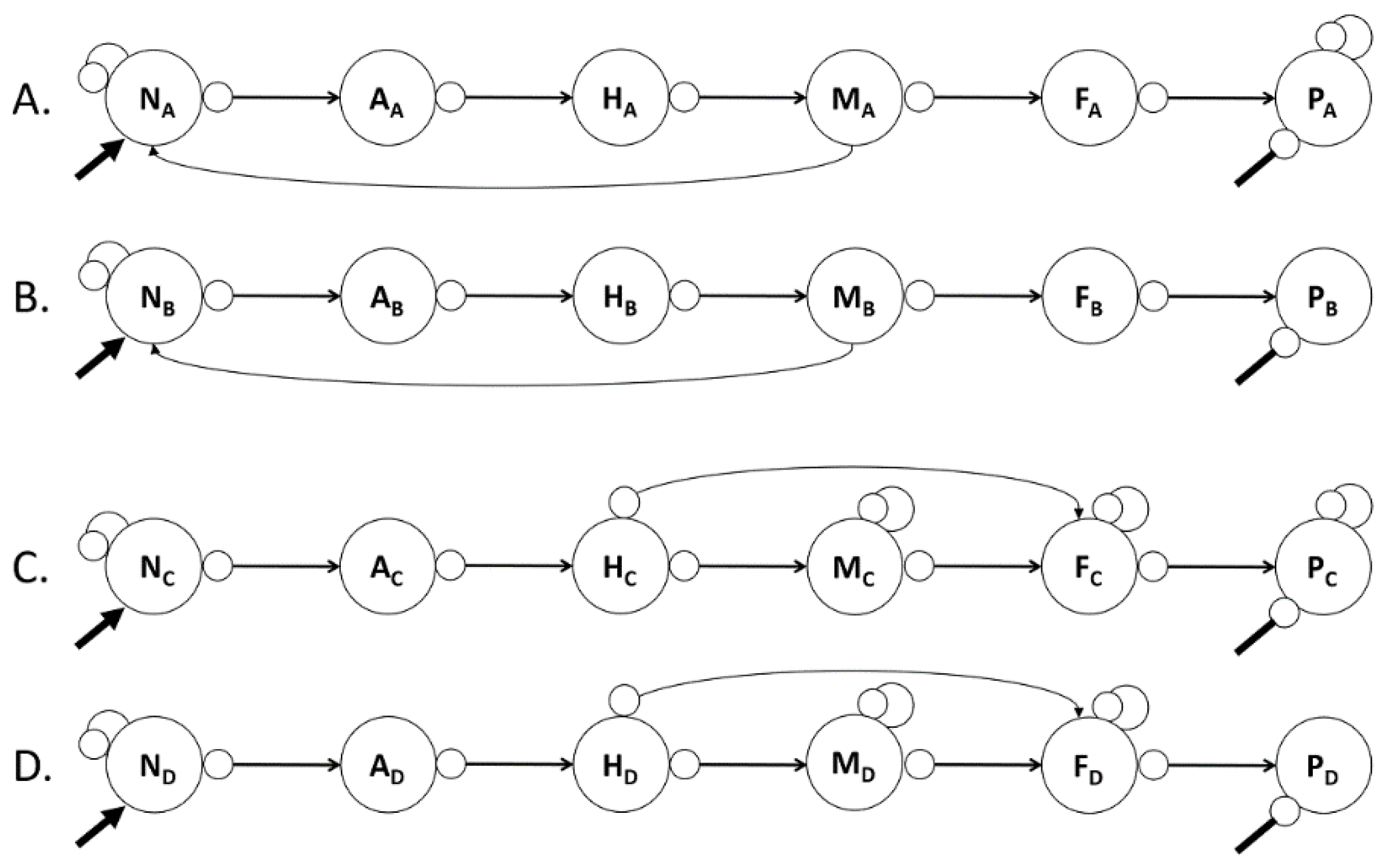
A Comparative Loop Analysis of Six Food Chains
- Nitrogen excretion of meso-predator to the nutrient pool and the top predator is self-damped.
- Nitrogen excretion of meso-predator to the nutrient pool and the top predator is not self-damped.
- Herbivores have two predators, and the top predator is self-damped.
- Herbivores have two predators, and the top predator is not self-damped.
Food Webs
Control Versus Constraint in Networks
Control
Constraints
- (1)
- Physical-Chemical Laws and Constants: The experience of existence includes many types of external constraints, limitations, boundaries, and other influences related to physical-chemical laws and constants like the nature of matter, fundamental forces such as gravity, electromagnetism, strong and weak nuclear forces; universal constants like the speed of light, the gravitational constant, the proton to electron mass constant, the cosmological constant etc., and laws of nature such as the laws of thermodynamics, law of gravitation, and law of mass action. For example, the speed of light is seen as a constraint on momentum. Living systems, as combinations of matter and energy, are part of the physical universe; however, they are proportionally a small percentage of all matter and energy. Nevertheless, living systems are subject to the same physical and chemical laws as nonliving matter. For this paper, we assume these external, universe-wide constraints and laws provide a backdrop or landscape for life. Patten et al. (2011) detailed the roles of several physical-chemical constraints operative in ecosystems. Additionally, Patten’s Environs Theory provides a deeper understanding of the interconnectedness and wholeness of ecosystems, encompassing their physical, chemical, and biological features.
- (2)
- Spatial-Topological-Temporal Constraints: Food webs exhibit constraints, including functional boundaries, which are distributed in both space and time. A set of interacting whole-system constraints is much more challenging to identify and understand than a predator decreasing the abundance of its prey. Mobus and Kalton (2015) pointed out, “One does not easily see the mutual web of constraint, i.e., the limits inherent in these interdependencies, until something unfitting transforms the dynamic.” Climate change appears to be one of those ‘unfitting’ circumstances (Lane, 2026). Boundary conditions are a type of spatial-temporal constraint. When we change boundaries, we either constrict or broaden both the feedback and the constraint potential. Sometimes, boundaries are as simple as a physical shoreline; others are virtual, such as a food web configuration, yet they are nonetheless real. Constructing a system model, such as a loop diagram or a carbon flow diagram, involves inserting a formal boundary between the system and its environment, thereby creating an inside and an outside that remain intimately connected, as ecosystems are thermodynamically open systems. Patten et al. (2011) concluded, “Boundary constraints and network constraints combine to give results not obtainable with empirical measurements alone…Environments may operate autonomously within systems, but in the end, they are virtual and empirically immeasurable”. Boundaries are also temporal and can be observed, for example, in phenology, time lags, feedforward mechanisms, bifurcations, loop lengths, stability dynamics, and many other ecological phenomena.
- (3)
- Environmental Factors and Their Distributional Patterns: External drivers were discussed in an earlier section and are not considered further here except to remark that Patten et al. (2011) concluded that, “forcing functions breach boundaries and carry external constraints explicitly into the systems they enter”. This is termed ‘network enfolding’ in Environ Theory. Forcing functions or drivers are equivalent to parameter inputs in Loop Analysis.
- (4)
- Self-Imposed Biological Constraints: The rest of this paper focuses on this fourth category of distributed, self-made constraints constructed within ecosystem networks. They are created internally without any external constructing agents. They arise from the network configuration itself and all have both spatial and temporal dimensions. As distributed and diffuse constraints, they operate holistically and in intricate synchrony to ensure the ecosystem achieves its immediate objectives of securing nutrients and energy, discarding waste, gathering information, repair and replacement, organizational integrity (coherence), and responding to external perturbations, as well as its long-term goal of safeguarding persistence (Lane, 2018a). Bodini et al. (2017), using Loop Analysis, concluded that “the locus of control in the ecological community of the Black Sea is diffuse and that the behavior of the system depends on the structure of its interaction network”. Bechtel (2016) reported that “states of whole systems often constrain the behavior of their parts”. He pointed out that “any networks in which the edges [links] are not all in one direction [such as the signed digraphs of Loop Analysis, see below] are subject to complex dynamical behavior, often as oscillations in…other parts of the network”.
Network Context: The Ecosystem as a Complex Chimera
- (1)
- The ecosystem chimera captures energy and nutrients, distributing them throughout the food web, much like metabolic cellular pathways. Open dissipative systems require a continual, secure supply of nutrients and energy to maintain their system structure and function, to dispatch wastes, and to keep ahead of unrelenting increases in entropy. Life runs uphill as fast as it can struggling against entropy like omnipresent friction, and before the crest can be realized, the living fall eventually back down into the primordial dust. The very term' food web' emphasizes this material dependency.
- (2)
- Modular integrity is required to ensure the function-enabling structure of the food web to ensure the survival of its identity, its ‘selfness’ is kept within certain limits so that food and energy can reach all contributing members in the right amounts and times as well as enzyme across hierarchical levels and maintaining resilience under the continuous onslaught of external drivers. Rosen (1991, 2000) believed life is a process realized by system functionalities. He did not engage in endless chicken-and-egg deliberations about which came first. Function came first to him, and relational structure determined how successfully functionality is enabled. If he is correct, this concept provides a new perspective when analyzing food web structure. Mobus and Kalton (2015) concluded, modules are beneficial when specifications are changing, and goals are changing. “Modules [exist] for each sub-problem of the goal; thus, modules give more adaptive flexibilities and provide structural simplicity…” to the overall system. Modular integrity is similar, but not identical, to Juarrero’s (2023) concept of coherence.
- (3)
- Time management is critical, and ecosystem chimeras like cells and organisms have evolved diverse functionalities to manage and manipulate time. For example, self-regulation, other feedback, and feedforward are processes found across biological systems that utilize similar structures to contribute to this functionality. (See Network Motif subsection below.) Mobus and Kalton (2015) believed “Timing is crucial for success,” and they outlined a set of timing components that are associated with biological systems. Considering how a marine food web is structured in the following subsection, it is helpful to consider how all three chimera functions are embedded in and unfolded from within these unique configurations.
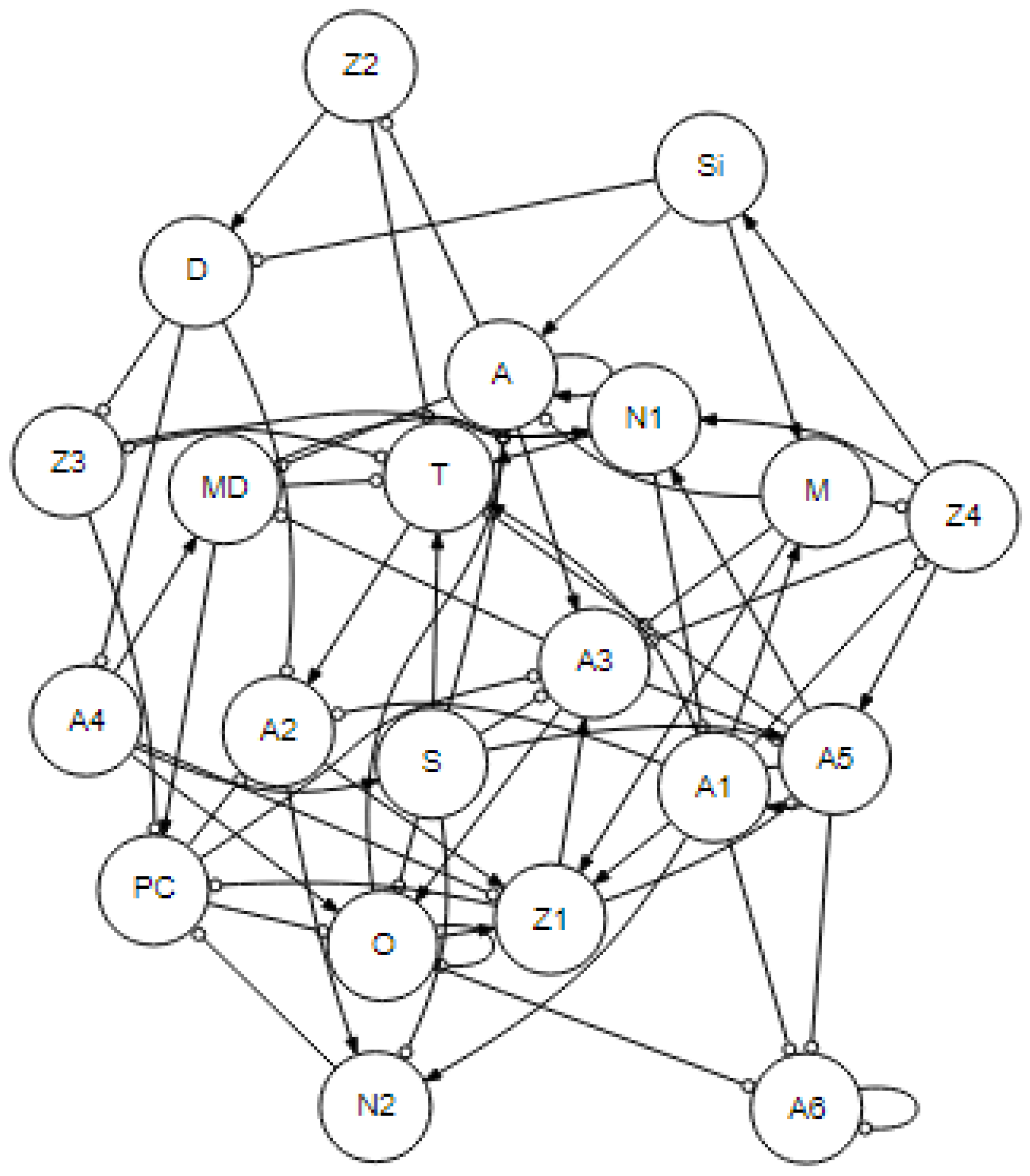
Self-Organization and Constraint: Ecological Skeletons in Marine Food Webs
Purpose
3. -Tier Lattice Structure
Valid Complement Rule
Stability Rules
Nonlinear Focal Points
Satellite Nodes
Network Motifs: Comparison of Biologically Reasonable Versus Random Food Webs
| Core Model No. 16 | Mean Random Network | Z Scores | |||||||||
| N | - | + | Total | N | - | + | Total | N | - | + | Total |
| 1 | 15 | 0 | 15 | 1 | 1.55 | 1.65 | 3.20 | 1 | 11.30 | -1.45 | 9.85 |
| 2 | 25 | 0 | 25 | 2 | 2.45 | 2.40 | 4.85 | 2 | 14.55 | -1.62 | 12.93 |
| 3 | 5 | 3 | 8 | 3 | 4.96 | 5.04 | 10.00 | 3 | 0.02 | -0.86 | -0.84 |
| 4 | 0 | 10 | 10 | 4 | 9.72 | 10.04 | 19.76 | 4 | -2.42 | -0.01 | -2.43 |
| Motif | Model 16 | Mean Random Network (500) | Z-Score | 0.10 | 0.05 | 0.01 |
| Feedforward | 24 | 29.68 | -0.96 | NS | NS | NS |
| Bi-Parallel | 40 | 80.23 | -2.53 | S | S | NS |
| Bi-fan | 20 | 40.69 | -1.59 | NS | NS | NS |
| Core Model No. 16 | Mean Random Network | Z Scores | ||||||||||
| Type | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 |
| Coherent | 5 | 3 | 5 | 0 | 3.61 | 3.85 | 3.67 | 3.67 | 0.57 | -0.41 | 0.67 | -1.86 |
| Incoherent | 3 | 3 | 0 | 5 | 3.67 | 3.73 | 3.67 | 3.80 | -0.35 | -0.29 | -2.01 | 0.57 |
Network Motif: Auto-regulation
Network Motifs with 3 Nodes (Feedforward)
Network Motifs with 4 Nodes (Bi-Fan & Bi-Parallel)
Some Benefits of Using Loop Analysis for Identifying Internal Constraints of Food Webs
Conclusions
Funding
Acknowledgments
| 1 |
https://dictionary.cambridge.org/dictionary/english/control (accessed May 18, 2025). |
| 2 | Ibid. |
References
- Allen, C., M. Bekoff, G. Lauder. (1998). Nature’s Purposes: Analysis of Function and Design in Biology. MIT Press. Cambridge, MA.
- Alon, U. (2007a). An Introduction to Systems Biology: Design Principles of Biological Circuits. Chapman and Hall. Boca Raton, FL.
- Alon, U. (2007b). Network motifs: theory and experimental approaches. Nature Review Genetics 8:450-461. [CrossRef]
- Andersen, K. H., M. Pedersen. (2010). Damped trophic cascades driven by fishing and model marine ecosystems. Proceedings Royal Society London B 277: 795–802. [CrossRef]
- Arditi, R., L. R. Ginzburg. (2012). How Species Interact, Altering the Standard View of Trophic Ecology. Oxford University Press. Oxford, U.K.
- Ashby, W. R. (1956). An Introduction to Cybernetics. Chapman & Hall. London, UK.
- Ashby, W. R. (1952). Design for a Brain. Chapman & Hall. London, UK.
- Atkinson, A., S. L. Hill, M. Barange, E. A. Pakhomov, D. Raubenheuimer, K. Schmidt, S. Simpson, et al. (2014). Sardine cycles, krill declines, and locust plagues: revisiting wasp-waist food webs. Trends in Ecology & Evolution 29(6):309-316.
- Babcock, R., J.M. Dambacher, E.B. Morello, E.E. Plagányi, K.R. Hayes, H. P. A. Sweatman, M. S. Pratchett. (2016). Assessing different causes of crown-of-thorns starfish outbreaks and appropriate responses for management on the Great Barrier Reef. PLOS One. [CrossRef]
- DOI:10.1371/journal pone.0169048.johnson. [CrossRef]
- Bakun,A., (2006). Wasp-waist populations and marine ecosystem dynamics: navigating the ‘predator pit’ topographies. Progress in Oceanography 68:271-288. [CrossRef]
- Ball, P. (1999). The Self-Made Tapestry: Pattern Formation in Nature. Oxford University Press. Oxford, U.K.
- Barabás, G., M.J. Michalska, S. Allesina. (2017). Self-regulation and the stability of large ecological networks. Nature Ecology & Evolution 1:1870-1875. [CrossRef]
- Barabási, A. L. (2016). Network Science. Cambridge University Press. Cambridge, U.K.
- Barbier, M., M. Loreau. (2018). Pyramids and cascades: a synthesis of food chain functioning and stability. Ecology Letters 22:405-419. [CrossRef]
- Barrios-O’Neill, D., C. Bertolini, P. C. Collins. (2017). Trophic cascades and the transient keystone concept. Biological Conservation 212:191-195. [CrossRef]
- Bechtel, W. (2016). Explicating top-down causation using networks and dynamics. Philosophy of science 84(2):253–274. [CrossRef]
- Bechtel, W., L. Bich, (2021). Grounding cognition: heterarchical control mechanisms in biology. Philosophical Transactions Royal Society B 376: 20190751. [CrossRef]
- Bejan, A., J. P. Zane. (2012). Design in Nature: How the Constructional Law Governs Evolution in Biology, Physics, Technology, and Social Organizations. Doubleday/Random House. N.Y., New York.
- Benton, T. G., E. Ranta, V. Kaitala, A. P. Beckerman. (2001). Maternal effects in the stability of population dynamics in noisy environments. 70:590-599. [CrossRef]
- Bodini, A. M. Rocchi, M. Scott. (2017). Insights into the ecology of the Black Sea through the qualitative Loop Analysis of the community structure. Limnology and Oceanography 63(2):968–984.
- Bongard, J. and M. Levin. (2021). Living Things Are Not (20th Century) Machines: Updating Mechanism Metaphors in Light of the Modern Science of Machine Behavior. [CrossRef]
- Borrett, S. R., S.J. Whipple, B.C.Patten, R.C. Christian. (2006). Indirect effects of distributed control in ecosystems: temporal variation of indirect effects in a seven-compartment model of nitrogen flow in the Neuse River Estuary, USA—time series analysis. Ecological Modelling 194: 178-188.
- Bossier, S., J. R. Nielsen, S. Neuenfeldt. (2020). Exploring trophic interactions and cascades in the Baltic Sea using a complex end-to-end ecosystem model with extensive food web integration. Ecological Modelling 436. [CrossRef]
- Botsford, L. W., M. D. Holland, J. C. Field, A. Hastings. (2014). Cohort resonance: a significant component of fluctuations in recruitment, egg production, and catch of fish populations. ICES Journal of Marine Science 71:2158-2170. [CrossRef]
- Bruder, A., A. Frainer, T. Rota, R. Primicerio. (2019). The importance of ecological networks in multiple-stressor research and management. Frontiers in Environmental Science 7, Article 59. [CrossRef]
- Bruni, L.E., F, Giorgi. (2015). Towards a herterarchical approach to biology and cognition. Progress in Biophysics and Molecular Biology. [CrossRef]
- Bruno, J. F., M. I. O’Connor. (2005). Cascading effects of predator diversity and omnivory in a marine food web. Ecology Letters 8(4):1048–1056. [CrossRef]
- Burgstaller, W. (2024). About organisms, a teaser. Archives of Biology & Life Sciences 1(1):1-7.
- Cagua, E. F., K. L. Wootton, D. B. Stouffer. (2019). Keystoneness, centrality, and the structural controllability of ecological networks. Journal of Ecology 107:1779-1790. [CrossRef]
- Carey, M. P., P. S. Levin, H. Townsend, T. J. Minello, G. R. Sutton, T. B. Francis, C. J. Harvey, et al. (2013). Characterizing coastal food webs with qualitative links to bridge the gap between the theory and the practice of ecosystem-based management. ICES Journal of Marine Science 71(3): 713–724. [CrossRef]
- Cazzolla-Gatti, R., B. Fath. W. Hordijk, S. Kauffman, R. Ulanowicz. (2018). Niche emergence as an autocatalytic process in the evolution of ecosystems. Journal of Theoretical Biology 454:110-117. [CrossRef]
- Choi, J., D. Lee. (2018). Topological motifs populate complex networks through grouped attachment. Scientific Reports 8:12670. [CrossRef]
- Coale, T. H., V. Loconte, K.A. Turk-Kubo, B. Vanslembrouck, W.K.E. Mak, S. Cheung, A. Ekman, J-H. Chen, K. Hagino, J.P. Zehr, + 5 Authors. (2024). Nitrogen-fixing organelle in a marine alga. Science 384 (6692): 217–222. April 12, 2024. [CrossRef]
- Coll, M., M. Alblo-Puigserver, J. Navarro, I. Palomera, J.M. Dambacher. (2019). Who is to blame? Possible pressures on small pelagic fish population changes in the northwestern Mediterranean Sea. Marine Ecology Progress Series. [CrossRef]
- Cosentino, C., D. Bates. (2019). Feedback Control in Biology. CRC Press. New York, NY.
- Cottam, R., W. Ranson. (2017). Bridging the Gap between Life and Reality. Springer International Publishing. Cham, Switzerland.
- Csárdi G, Nepusz T. (2006). The igraph software package for complex network research. InterJournal, Complex Systems 1695. http://igraph.org.
- Cumming, G.S. (2016). Heterarchies: Reconciling networks and hierarchies. Trends in Ecology and Evolution. [CrossRef]
- Cury, P. (2018). Obstinate nature. ICES Journal of Marine Science. [CrossRef]
- Cury, P., A. Bakun, R.J. M. Crawford, Q. A. Jarre, R.A. Quinoes, L.J. Shannon, H. M. Verheye. (2000). Small pelagics in upwelling systems: patterns of interaction and structural changes in “wasp-waist” ecosystems. ICES Journal of Marine Sciences 57: 603–618. [CrossRef]
- Dambacher, J. M., Gaughan, D.J., Rochet, M.J., Rossignol, P.A., Trenkei, V. (2009). Qualitative modelling and indicators of exploited ecosystems. Fish and Fisheries 10 (1): 305–322. [CrossRef]
- Dambacher, J. M., R. Ramos-Jiliberto. (2007). Understanding and predicting effects of modified interactions through a qualitative analysis of community structure. The Quarterly Review of Biology 82:227-250. [CrossRef]
- Dambacher, J. M., H.W. Li, P.A. Rossignol. (2003a). Qualitative predictions and model ecosystems. Ecological Modelling 161:79-93. [CrossRef]
- Dambacher, J. M., H.K. Luh, H.W. Li, P.A. Rossignol. (2003b). Qualitative stability and ambiguity in model ecosystems. American Naturalist 161:876-888. [CrossRef]
- Dambacher, J. M., H.W. Li, J. O. Wolff, P.A. Rossignol. (1999). Parsimonious interpretation of the impact of vegetation, food, and predation on snowshoe hare. Oikos 84: 530–532. [CrossRef]
- Dey, A. K., Y. R. Gel, H. V. Poor. (2019). What network motifs tell us about the resilience and reliability of complex networks? Proceedings of the National Academy of Sciences 116:19368-19373. [CrossRef]
- Dupré, J. & D. J. Nicholson. (2018). A manifesto for a processual philosophy of biology. In: Nicholson, D. J., and J. Dupré (eds.) Everything Flows Towards a Processual Philosophy of Biology. Oxford University Press, Oxford, U.K.
- Estes, J. A., A. Burdin, D. F. Doak. (2016). Sea otters, kelp forests, and the extinction of Steller’s sea cow. Proceedings of the National Academy of Sciences 113:880-885. [CrossRef]
- Estes, J. A., J. Terborgh, J. S. Brashares, M. E. Power, W. J. Berger, W. J. Bond, S. R. Carpenter, et al. (2011). Trophic Downgrading of Planet Earth. Science 333:301-306. [CrossRef]
- Estrada, E. (2011). The Structure of Complex Networks: Theory and Applications. Oxford University Press, Oxford, U.K.
- Estrada, E. (2007). Characterization of topological keystone species: local, global, and meso-scale. Ecological Complexity 4: 48–57. [CrossRef]
- Fath, B. D. (2004). Distributed control and ecological networks. Ecological Modelling 179:235-245.
- Fath, B.D., B.C. Patten. (1998). Network synergism: emergence of positive relations in ecological systems. Ecological Modelling 107:127-143. [CrossRef]
- Fauchald, P., H. Skov, M. Skern-Mauritzen, D. Johns, T. Tvera. (2011). Wasp-waist interactions in the North Sea ecosystem. PLoS ONE. [CrossRef]
- Fox Keller, E. (2005). Ecosystems, organisms, and machines. Bioscience 55:1069-1074.
- Frank, K. T., B. Petri, J.S. Choi, W.C. Leggett. (2005). Trophic cascades in a formerly cod-dominated ecosystem. Science 308:1621-1623. [CrossRef]
- Frank, K. T., B. Petri, N. L. Shackell, J.S. Choi. (2006). Reconciling differences in trophic control in mid-latitude marine ecosystems. Ecology Letters 9: 1096–1105. [CrossRef]
- Furla, P., D. Allemand, J.M. Shick, C. Ferrier-Pages, S. Richier, A. Plantivaux, S. Tambutte, (2005). The symbiotic anthozoan: a physiological chimera between alga and animal. Integr. Comparative Biol. 45 (4), 595–604. http://www.jstor.org/stable/4485835. [CrossRef]
- Garay-Narvez, L. R. Ramos-Jiliberto. (2009). Induced defences within food webs: the role of community tradeoffs, delayed responses, and defense specificity. Ecological Complexity. [CrossRef]
- Garvey, J. E., M. Whiles. (2016). Trophic Ecology. CRC Press. New York, N.Y.
- Gause, G. F. (1935). The Struggle for Existence. Reprinted 2019. Dover Publications. Mineola, NY.
- Gawne, R., M. Levin, M. (2023). Using tissue chimeras to probe regenerative patterning and physiological controls in Planaria. Integrative And Comparative Biology 63: S156-S156.
- Getz, W. M., H. W. Westerhoff, J. H. S. Hoffmyer, J. L. Snoep. (2003). Control analysis of trophic chains. Ecological Modelling 168: 153-171. [CrossRef]
- Gibert, J.P. (2019). Temperature has a direct and indirect influence on food web structure. Scientific Reports 9:5312. [CrossRef]
- Ginzburg, L., M. Colyvan. (2004). Ecological Orbits: How Planets Move and Populations Grow. Oxford University Press, New York, NY.
- Hall, C. A. S. (2020). Systems ecology and limits to growth: history, models and present status. In: Gary S. Metcalf & Kyoichi Kijima & Hiroshi Deguchi (ed.), Handbook of Systems Sciences, Springer. ISBN 978-981-15-0719-9. [CrossRef]
- Hall, C. A. S., J. A. Stanford, R. Hauer. (1992). The distribution and abundance of organisms as a consequence of energy balances along multiple environmental gradients. Oikos 65:377-390. [CrossRef]
- Harvey, C.J., G.D. Harvey, P.S. Levin. (2012). Food web structure and trophic control in Central Puget Sound. Estuaries and Coasts 35:821-838. [CrossRef]
- Hastings, A. (2001). Transient dynamics and persistence of ecological systems. Ecology Letters 4: 215–220. [CrossRef]
- Haswell, P. M., J. Kusak, M. W. Hayward. (2016). Large carnivore impacts are context-dependent. Food webs 12:3-13. [CrossRef]
- Heath, M. R., D. C. Speirs, J. H. Steele. (2014). Understanding patterns and processes in models of trophic cascades. Ecology Letters 17:101-114. [CrossRef]
- Heithaus, M. R., A. Frid, A. J. Wirsing, B. Worm. (2008). Predicting ecological consequences of marine top predator declines. Trends in Ecology and Evolution 23(4):202-20. [CrossRef]
- Henning, B., Scarfe, A. C. (Eds.). (2013). Beyond Mechanism: Putting Life Back into Biology. Lexington Books. Plymouth, UK.
- Hessen, E. O., S. Kaardvedt. (2014). Top-down cascades in lakes and oceans: different perspectives but same story? Journal Plankton Research 36(4): 914-924.
- Higashi, M. & B.C. Patten. Dominance of indirect causality in ecosystems. American Naturalist 1989, 133, 288–302. [Google Scholar] [CrossRef]
- Hofmyer, J.H.S. 2021. A biochemically-realisable relational model of the self System-manufacturing cell. Biosystems 207, 104463.
- Hofmyer, J.H.S. The harmony of the cell: the regulatory design of cellular processes. Essays in Biochemistry 2008, 45, 57–66. [Google Scholar]
- Holling, C. S., G. K. Meffe. (1996). Command-and-control and the pathology of natural resource management. Conservation Biology 10(2): 328–337. [CrossRef]
- Hordijk, W. (2013). Autocatalytic sets: from the origin of life to the ecology. Bioscience 63:877-881.
- Hunt, J. G. L., S. McKinnell. (2006). Interplay between top-down, bottom-up, and wasp-waist control in marine ecosystems. Progress in Oceanography 68:115-124.
- Hurwitz A. (1895) 1964. On the conditions under which an equation has only roots with negative real parts. Translated from the German by H. G. Bergman. Pages 72–82 in R. E. Bellman and R. Kalaba, eds. Selected Papers on Mathematical Trends in Control Theory. Dover, New York.
- Iniguez, G.; Battiston, F.; Karsai, M. Bridging the gap between graphs and networks. Nature Communications Physics 2020, 3, 88. [Google Scholar] [CrossRef]
- Jacob, F. (1977). Evolution and tinkering. Science 196:1161-1166.
- Jacobs, J. (2001). The Nature of Economies. Vintage Canada. Toronto, ON.
- Juarrero, A. (2023). Context Changes Everything. How Constraints Create Coherence. MIT Press. Cambridge, MA.
- Johannessen, T. (2014). From an antagonistic to a synergistic predator-prey perspective: bifurcations in marine ecosystems. Academress. New York, N. Y.
- Jonsson, T., F. Berg, M. Emerson, A. Pimenov. (2015). The context-dependent status of species as keystone during food web disassembly. Food webs 5:1-10.
- Jordán, F., T. A. Okey, B. Bauer, S. Libralato. (2008). Identifying important species: linking structure and function in ecological networks. Ecological Modelling 216:75-80. [CrossRef]
- Joslyn, C. (2001). The semiotics of control in modelling relations in complex systems. Biosystems 60: 131-148. 10.1016/s0303-2647(01)00113-7.
- Kauffman, S.A. (2019). The World Beyond Physics. The Emergence and Evolution of Life. Oxford University Press. Oxford: UK.
- Kauffman, S.A. (1995). At Home in the Universe. The Search for the Laws of Self-Organization and Complexity. Oxford University Press. Oxford: UK.
- Kauffman, S.A. (1993). Self-Organization and Selection in Evolution. Oxford University Press. London, UK.
- Kondoh. M. (2005). Chapter 11: Does food web complexity maintain biodiversity? - The Adaptive Food Web Hypothesis. Pp. 130–142. In: Belgrano, A., U.M. Scharler, J. Dunne, and R.E. Ulanowicz. (Eds.) Aquatic food webs: An Ecosystem Approach. Oxford University Press.
- Kondoh, M., K. Ninomiya. (2009). Food-chain length and adaptive foraging. Proceedings B of the Royal Society 276(1670): 3113-3121. [CrossRef]
- Kuhn, T. S. (1970). The Structure of Scientific Revolutions. University of Chicago Press, Chicago, IL.
- Lane, P. A. (2026). Understanding the Ecological Complexity of Marine-Mediated Climate Change in Biosphere Security: In Complex and Security - Theorizing Within and Beyond Borders. Edited by: E. Charalampaki, C. Mesjasz, and L. Tome. Invited chapter for a book to be published by Routledge. (In press).
- Lane, P.A. 2025. Evolutionary Implications of Chimeras Throughout the Biological Hierarchy. March 24, 2025. Michael Levin’s Laboratory, Tufts University. (Invited talk). https://www.youtube.com/watch?v=QFQNTv8lGFw.
- Lane, P.A., (2024a). Using Relational Biology with Loop Analysis to Study the North Atlantic Biological Carbon Pump in a ‘Hybrid’ Non-Algorithmic Manner. Mathematics 12, 3972. https://doi.org/10.3390/math12243972. [CrossRef]
- Lane, P. A. (2024b). Robert Rosen’s Relational Biology Theory and His Emphasis on Non-Algorithmic Approaches to Living Systems. Mathematics, 12(22), 3529. [CrossRef]
- Lane, P.A., (2021). "Putting More “System” into Ecosystem-Based Management Using Qualitative Analysis," Pp. 917-942. In: Gary S. Metcalf & Kyoichi Kijima & Hiroshi Deguchi (ed.), Handbook of Systems Sciences, chapter 34, pages 917-942, Springer. ISBN 978-981-15-0719-9. [CrossRef]
- Lane, P.A. (2018a). Ecosystems as Chimeras: A thought experiment in Rosennean Complexity. Ecological Complexity 35: 51–75. [CrossRef]
- Lane, P.A. (2018b). Ecological Skeletons: excavating the bare bones of community structure using Loop Analysis. Pp 201–219. In: The Truth is the Whole: on the Unity and Dynamic Complexity of Life. T. Awerbach, M. S. Clark, P.J. Taylor (Eds.). Proceedings of a Symposium at the Harvard University School of Public Health, Boston, Massachusetts, May 21-23, 2015. The Pumping Station Press.
- Lane, P.A. (2017a). A review of the trophic cascade concept using the lens of Loop Analysis: “The truth is the whole” Food Webs 13: 1–11.
- Lane, P.A. (2017b). Assumptions about trophic cascades: The inevitable collision between reductionist simplicity and ecological complexity. Food Webs 13: 12–26.
- Lane, P.A. (2016). A Cautionary Tale: The Goldilocks Effect in measuring ecological complication in food webs. Food Webs 8:1-11.
- Lane, P.A. (1998). Assessing cumulative health effects in ecosystems. Pp. 129–153. In: Rapport, D., Costanza, R., Epstein, P., Gaudet, C., Levins, R. (Eds.), Ecosystem Health. Blackwell Science, Malden, Massachusetts.
- Lane, P.A. (1986). Symmetry, change, perturbation and observing mode in natural communities. Ecology 67: 223–239.
- Lane, P.A., R. Levins. (1977). Dynamics of aquatic systems. II. The effects of nutrient enrichment on model plankton communities. Limnology and Oceanography 21:454-471.
- Lassalle, G., J. Lobry, F. Le Loćh, S. Mackinson, F. Sanchez, M. P. Tomczak, N. Niquel. (2012). Ecosystem status and functioning: searching for rules of thumb using an inter-site comparison of food web models of Northeast Atlantic continental shelves. ICES Journal of Marine Science 70(1): 135–149. [CrossRef]
- Lau, M. K., S. R. Borret, B. Baiser, N. J. Gotelli, A. M. Ellison. (2017). Ecological network metrics: opportunities for synthesis. Ecosphere 8: Article E01900. [CrossRef]
- Lear, J. (1988). Aristotle: The Desire to Understand. Cambridge University Press. New York, NY.
- Leonard, A., T. Scholte, K. Shepherd, J Truss. (2021). Cybernetic approaches and models. In: Gary S. Metcalf & Kyoichi Kijima & Hiroshi Deguchi (ed.), Handbook of Systems Sciences, Springer. ISBN 978-981-15-0719-9. [CrossRef]
- Leroux, S. J., M. Loreau. (2015). Chapter 1: Theoretical perspectives on bottom-up and top-down interactions across ecosystems. Pp. 3–30. In: Hanley, T. C. and K.J. LaPierre. (Eds.) Trophic Ecology: Bottom-Up and Top-Down Interactions across Aquatic and Terrestrial Systems. Ecological Reviews. Cambridge University Press. Cambridge, UK.
- Lissack, M. (2021). Cybernetics and control. In: Gary S. Metcalf & Kyoichi Kijima & Hiroshi Deguchi (ed.), Handbook of Systems Sciences. Springer. ISBN 978-981-15-0719-9. [CrossRef]
- Levin, M., & Resnik, D. (2025). Technological Approach to Mind Everywhere: A Framework for Conceptualizing Goal-Directedness in Biology and Other Domains. [CrossRef]
- Levins, R., (1998). Qualitative mathematics for understanding, prediction, and intervention in complex ecosystems. Pp. 178–204. In: Rapport, D., Costanza, R., Epstein, P., Gaudet, C., Levins, R. (Eds.), Ecosystem Health. Blackwell Science, Malden, Massachusetts.
- Levins, R., (1975). Evolution in communities near equilibrium. Pp.16–50. In M. Cody, M., Diamond, J., (eds.), Ecology & Evolution of Natural Communities. Belknap Press, Boston, Massachusetts.
- Levins, R., (1974). The qualitative analysis of partially specified systems. Annals of the New York Academy of Science 231:123–138.
- Lindstörm, J. & H. Kokko. (2002). Cohort effects and population dynamics. Ecology Letters 5(3). [CrossRef]
- Loeuille, N., M. Loreau. (2004). Nutrient enrichment and food chains: can evolution buffer top-down ‘control’? Theoretical Population Biology 65:285-298.
- Lyon, P. (2025). Fundamental principles of cognitive biology 2.0. Biological Theory. [CrossRef]
- Ma, Q., C. Kazanci. (2013). Analysis of indirect effects within ecosystem models using pathway- based methodology. Ecological Modelling 252: 238-245. [CrossRef]
- Mangan, S. & U. Alon. (2003). Structure and function of the feed-forward loop network motif. Proceedings of the National Academy of Sciences 100:11980-11985.
- Margulis, L., C. A. Asikainen, W. E. Krumbein. (2011). Chimeras and consciousness: evolution of the sensory self. The MIT Press. Cambridge, MA.
- Martone, R. G., A. Bodini, F. Micheli. (2017). Identifying potential consequences of natural perturbations and management decisions on a coastal fisheries social-ecological system using qualitative Loop Analysis. Ecology and Society 22:34-48. [CrossRef]
- Maturana, H. R. & F.J. Varela. (1980). Autopoiesis and cognition: the realization of the living. Springer, Netherlands.
- Metcalfe, S. J., J. M. Dambacher, P. Rogers, N. Lonergan, D. J. Gaughan. (2013). Identifying key dynamics and ideal governance structures for successful ecological management. Environmental Science & Policy 37:34-49. [CrossRef]
- Mills, L., M. E. Soulé, & D. F. Doak. (1993). The keystone-species concept in ecology and conservation. BioScience 43:219-224. [CrossRef]
- Milo, R., S. Shen-Orr, S. Itzkovitz, et al. (2002). Network motif: simple building blocks of complex networks. Science 298 (5594): 824-827. [CrossRef]
- Mittelbach, G. G. 2012. Community Ecology. Sinuaer Associates, Inc. 400 pp.
- Mobus, G. E. & M. C. Kalton. (2015). Principles of System Science. Springer. New York, New York.
- Modlmeier, A. P., C. M. Keiser, J. V. Watters, et al. (2014). The keystone individual concept: an ecological and evolutionary overview. Animal Behaviour 89:53-62. [CrossRef]
- Moñtano-Moctezuma, G., H. W. Li, P. A. Rossignol. (2008). Variability of community interaction networks in marine reserves in adjacent exploited areas. Fisheries Research 94:99-108. [CrossRef]
- Montevil, M., M. Mossio. (2015). Biological organization as closure of constraints. Journal of Theoretical Biology 372:179-191. [CrossRef]
- Moody, E.R.R., S. Álvarez-Carretero, T.A. Mahendrarajah, et al. The nature of the last universal common ancestor and its impact on the early Earth system. Nat Ecol Evol 8, 1654–1666 (2024). [CrossRef]
- Mougi, A., M. Kondoh. (2014). Adaptation in a hybrid world with multiple interaction types: a new mechanism for species coexistence. Ecological Research 29:113-119. [CrossRef]
- Moreno, A., M. Mossio. (2015). Biological Autonomy: A Philosophical and Theoretical Enquiry. Springer. New York, N.Y.
- Mossio, M., A. Moreno, (2010). Organizational closure in biological organisms. History and Philosophy of the Life Sciences 32: 269-288.
- Mossio, M., M. Montevil, G. Longo. (2016). Theoretical principles for biology: organization. Progress in Biophysics and Molecular Biology 122: 24-35.
- Newman, M. E. J. (2010). Networks: An Introduction, Oxford University Press. Oxford, U.K.
- Nielsen, S.N., B.D. Fath, S, Bastianoni, S., J.C. Marques, F. Muller, B.C. Patten, R.E. Ulanowicz, S.E. Jorgensen, E. Tiezzi. (2020). A New Ecology Systems Perspective. 2nd. Ed. Elsevier, Amsterdam, The Netherlands.
- Noble, R., D. Noble (2023). Physiology restores purpose to evolutionary biology. Biological Journal of the Linnean Society 139 (4):357–369. [CrossRef]
- Odling-Smee, J. (2024). Niche Construction. How Life Contributes to Its Own Evolution. MIT Press. Cambridge, MA.
- Odum, E. P., G. W. Barrett. (2005). Fundamentals of Ecology. Thomas Brooks/Cole. Belmont, CA.
- Ortiz, M., R. Levins. (2017). Self-feedbacks determine the sustainability of human interventions in eco-social complex systems: impacts on biodiversity and ecosystem health. PLoS One. [CrossRef]
- Palsson, B. O. (2015). Systems Biology Constraint-Based Reconstruction and Analysis. Cambridge University Press. Cambridge, U.K.
- Patten, B.C. (1998). Network orientors: steps toward a cosmography of ecosystems: orientors for directional development, self-organization, and autoevolution. In: Miller, F., M. Leupelt. (Eds.) Eco-Targets, Goal Functions, and Orientors. Springer. Berlin, Germany.
- Patten, B. C., (1984). Toward a theory of the quantitative dominance of indirect effects in ecosystems. Verh. Gesellschaft für Ökologie. 13:271–284.
- Patten, B.C. (1978). Systems approach to the concept of environment. Ohio J. Science 78:206-222.
- Patten, B. C. (1959). An introduction to the cybernetics of the ecosystem: the trophic-dynamic aspect. Ecology 40:221-231. [CrossRef]
- Patten, B. C., M.Straskraba, S.E, Jorgensen. (2011). Ecosystems emerging. 5: Constraints. Ecological Modelling 222: 2945-2972.
- Patten, B.C. & M. Higashi. (1991). Indirect effects in complex ecology II. The quantitative Theory. In B.C. Patten and S. E. Jorgensen. Complex Ecology: The Parts-Whole Relation in Ecology. Prentice Hall. Englewood Cliffs, New Jersey.
- Patten, B. C., E. P. Odum. (1981). The cybernetic nature of ecosystems. The American Naturalist 118(6): 886-895.
- Patten, B.C. & M. Witcamp. (1967). Systems analysis of 134Cesium kinetics in terrestrial microcosms Ecology 48:813-824.
- Payo-Payo, A., M. Genovart, A. Bertolera, et al. (2016). Consecutive cohort effects driven by density-dependence and climate influence early-life survival in the long-lived bird. Proceedings of the Royal Society B. [CrossRef]
- Pershing, A. J., M. A. Alexander, C. M. Hernandez, L. A. Kerr, A. Le Bris, et al. (2015). Slow adaptation in the face of rapid warming leads to the collapse of the Gulf of Maine cod fishery. Science 350(6262): 809–812. [CrossRef]
- Pezzulo, G., M. Levin. (2016). Top- down models in biology: exclamation explanation and control of compacts living systems above the molecular level. Interface. [CrossRef]
- Piovia-Scott, J., L. H. Yang, A.N. Wright. (2017). Temporal variation in trophic cascades. Annual Review of Ecology 48:281-300. [CrossRef]
- Power, M. E., D. Tilman, J. A. Estes, B. A. Menge, W. J. Bond, L. S. Mills, G. Daily, et al. (1996). Challenges in the quest for keystones: identifying keystone species is difficult, but essential to understanding how loss of species will affect ecosystems. BioScience 46(8):609–620.
- R Core Team. (2020). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
- Raginsky, M. (2023). Biological autonomy. Biological Theory 18: 303-308.
- Ratajczak, Z., P. D’Odorico, S.L. Collins, B.T. Bestelmeyer, et al. (2017). The interactive effects of press/pulse intensity and duration on regime shifts at multiple scales. Ecological Monographs 87:198-218. [CrossRef]
- Reiners, W. A., J. A. Lockwood, D. S. Reiners, S. D. Praeger. (2017). 100 years of ecology: what are our concepts and are they useful? Ecological Monographs 87:260-277.
- Reum, J. C. P., P. S. McDonald, B. E. Ferriss, D. M. Farrell, C. J. Harvey, P.S. Levin. (2015). Qualitative Network Models in Support of Ecosystem Approaches to Bivalve Aquaculture. ICES Journal of Marine Science, 72: 2278–2288. [CrossRef]
- Rip, J. M. K., K. S. McCain, D. H. Lynn, S. Fawcett. (2010). An experimental test of a fundamental food web motif. Proceedings of the Royal Society B. [CrossRef]
- Ripple, W.J., J. A. Estes, O. J. Schmitz, V. Constant, M. J. Kaylor, A. Lenz, J. L. Motley, et al. (2016). What is a trophic cascade? Trends in Ecology and Evolution 31(11): 842–849.
- Rosen, R. (2012). Anticipatory Systems. Springer. New York, NY.
- Rosen, R. (2000). Essays on Life Itself. Columbia University Press. New York, NY.
- Rosen, R. (1995). Cooperation and chimera. Pp. 343–358. In Casti, J.L., Karlqvist, A., 1995. Cooperation & Conflict in General Evolutionary Processes. John Wiley & Sons, Inc., New York, N.Y.
- Rosen, R. (1991). Life Itself. A Comprehensive Inquiry into the Nature, Origin, and Fabrication of Life. Rosen, R., 1958a. The relational theory of biological systems. Bulletin Math. Biophys. 20:245–260. [CrossRef]
- Rosen, R., (1963). Some results in graph theory and their application to abstract relational biology. Bull. Math. Biophys. 25:231–241. [CrossRef]
- Rosen, R., (1959). Ontological paradox implicit in the notion of a self-reproducing automata. Bull. Math. Biophys. 21:387–394. [CrossRef]
- Rosen, R., (1958a). The relational theory of biological systems. Bull. Math. Biophys. 20:317–341. [CrossRef]
- Rosen, R., (1958b). The representation of biological systems from the standpoint of the.
- theory of categories. Bull. Math. Biophys. 20, 317–341.
- Rovelli, C. (2018). The Order of Time. Riverhead Books. Penguin Random House. New York, NY.
- Rovelli, C. (2014). Reality Is Not What It Seems. The Journey to Quantum Gravity. Riverhead Books. Penguin Random House. New York, NY.
- Ruths, J., D. Ruths. (2014). Control profiles of complex networks. Science 343(6177): 1373–1376.
- Sacco, F., D. A. R. Sakthivadivel, M. Levin. 2025. Topological constraints on self-organization in locally interacting systems. ArXiv:2501.13188v1.
- Salomon, A. K., S. K. Gaichas, N. T. Shears, J. E. Smith, E. M. P. Madden, S. D. Gaines. (2010). Key fisheries and context-dependence of fishery—induced trophic cascades. Conservation Biology 24(2): 382–394.
- Shurety, A.L., O. Bodin, G.S. Cumming. (2022). A comparative approach to quantify the heterarchical structures of complex systems. Ecology and Society 27 ( 3: 38. [CrossRef]
- Solé, R., B. Goodwin. (2000). Signs of Life: How Complexity Pervades Biology. Basic Books. New York, NY.
- Solé, R., S. Valverde. (2019). Evolving complexity: how tinkering shape cells, software and ecological networks. Philosophical Transactions of the Royal Society B. [CrossRef]
- Terborgh, J., J.A. Estes. (2010). (Eds.)Trophic Cascades: Predators, Prey, and the Changing Dynamics of Nature. Island Press.
- Terborgh, J., R. D. Holt, & J.A. Estes. (2010). Terborgh, J. and J.A. Estes. (Eds.) Trophic cascades: what they are, how they work, and why they matter. Pp. 1-18 In: Trophic Cascades: Predators, Prey, and the Changing Dynamics of Nature. Island Press. 464 pp.
- Tiselius, P., L. F. Møller. (2017). Community cascades in the marine pelagic food web controlled by the non-visual apex predator Mnemiopsis leidyi. Journal of Plankton Research 39(2):271-279. [CrossRef]
- Trosvik, P., E. J. de Muinck. (2015). Ecology of bacteria in the human gastrointestinal tract – identification of keystone and foundation taxa. Microbiome 3:44.
- Ulanowicz, R. E. (2019). The tripartite nature of causalities and ecosystem dynamics. Current Opinion in Systems Biology 13:129-135. [CrossRef]
- Ulanowicz, R. E. (2008). Autocatalysis.pp. 288-290.
- Ulanowicz, R. E. (1995). Utricularia’s secret: the advantages of positive feedback in oligotrophic environments. Ecological Modelling 79: 49-57. [CrossRef]
- Varela, F.J. (2025). Principles of Biological Autonomy. A New Annotated Edition by DiPaolo, E. & Thompson, E. MIT Press. Cambridge, MA.
- Veldhuis, M. P., M. P. Berg, M. Loreau, and H. Olff. (2018). Ecological autocatalysis: a central principle of ecosystem organization. [CrossRef]
- Voit, E. O. (2013). A First Course in Systems Biology. Garland Science. N. Y., New York.
- Whipple, S. J., B. C. Patten, S.R. Borrett. (2014). Interactive effects and distributed control in ecosystems. Comparative network environment analysis of a seven-compartment model of nitrogen storage in the Neuse River Estuary, USA: time series analysis. Ecological Modeling 293: 161–186.
- Widder, S., R. Solé, J. Macía. (2012). Evolvability of feed-forward loop architecture biases its abundance in transcription networks. BMC Systems Biology 6: Article No. 7. [CrossRef]
- Wiener, N. (1961). Cybernetics: Or Control and Communication in the Animal and the Machine. 2nd Ed. MIT Press. Cambridge, MA.
- Wildermuth, R. P., G. Fay, S. Gaichas. (2018). Structural uncertainty and qualitative models for ecosystem-based management of Georges Bank. Canadian Journal of Fisheries and Aquatic Science 75:1635-1643. [CrossRef]
- Wilkinson, D. M. (2003). The fundamental process in ecology: a thought experiment on extraterrestrial biospheres. Biological Review 78:171-179. [CrossRef]
- Winemiller, K. O., C. A. Layman. (2005). Pattern, process, and scale in the food web paradigm: moving on the path from abstraction to prediction. Pp 10–23. In: P.C. de Ruiter, V. Wolters, and J.C. Moore (Eds.), Dynamic Food webs: Multi-Species Assemblages, Ecosystem Development, and Environmental Change. Elsevier/Academic Press. Amsterdam, the Netherlands.
- Winning, J., W. Bechtel. (2018). Rethinking causality and biological in neural mechanisms: constraints and control. Minds & Machines 28:287-310. [CrossRef]
- Wolfram, S. (2002). A New Kind of Science. Wolfram Media Inc. Champaign, IL.
- Woodger, J. H. (1929). A Critical Study. Biological Principles. Rutledge & Kagan Paul Ltd. London, U.K.
- Worm, B., Myers, R., (2003). Meta-analysis of cod–shrimp interactions reveals top-down control in oceanic food webs. Ecology Volume 84(1):162-173.
- Wright, J.A., P.A. Lane. (1986). The theory of Loop Analysis. Ecology 67. Supplementary Publication Source Document No. 8525B.
- Yu, S., J. Xu, C. Zhang, F. Xia, Z. Almakhadmeh, A. Tolba. (2019). Motifs in big networks: methods and applications. IEEE Access 7: 183322-183338. [CrossRef]
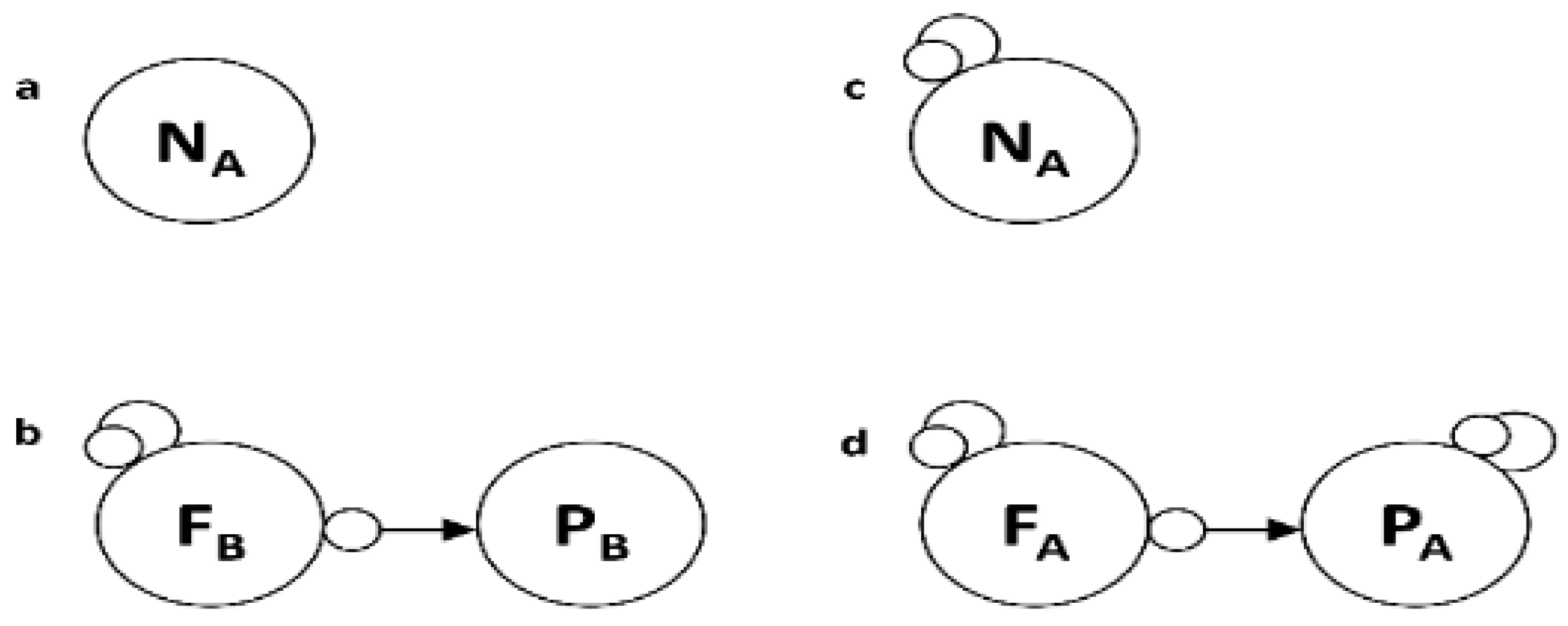

| Controller (C1) |
Controlee (C2) |
Change (C3) |
Process (C4) |
|
|---|---|---|---|---|
| (1)Single Species A on Self | An external driver to A or A changing internally due to gene turning on or off, developmental process, etc. |
Species A | Species A exhibits changes in density with concurrent changes in birth or death rates. | Species A is activated or deactivated to cause morphological, physiological, behavioral, population etc. changes. |
|
(2) Two Species Interactions |
An external driver to A or A changing internally |
Species B: Prey |
Predator decreases prey density. | Predator A consumes Prey B (Predator ‘b’ increases) (Prey ‘d’ increases). |
| An external driver to B or B changing internally |
Species A: Predator | Prey increases predator density. | Prey B is consumed by Predator A. (Predator ‘b’ increases). |
|
|
(3) Trophic Pathway -Trophic Cascade (top-down) |
An external driver to Predator A or A changing internally A = Species or Functional Group A |
Nodes B & C or Functional Groups B & C on the pathway: A-B-C |
Densities change as: 0 0 0, + - + or - + - + - + or - + - |
Coupled biological interactions, especially predator-prey pairs of 3 or more nodes. |
| -Trophic Escalade (bottom-up) | An external driver to Prey C or C changing internally C = Species or Functional Group C |
Nodes C & B or Functional Groups C & B on the pathway: C-B-A |
Densities change as: 0 0 0, + + + or - - - 0 + 0 or 0 - 0 + 0 + or - 0 - |
Coupled biological interactions, especially predator-prey pairs of 3 or more nodes. |
| (4) Food web | Externally Perturbed Species or Functional Group or internally changing |
Other Food Web Components |
Densities of one or more nodes may change, or the whole food web structure can be altered. | External driver to one node can change all others through multiple pathways and feedback loops. A food web may undergo self-reorganization. |
| EXTERNAL DRIVERS | INTERNAL DRIVERS | |
|---|---|---|
| CONTEXT INDEPENDENT | ||
| One Species | A top predator experiences a favourable temperature change, causing it to increase its reproduction in a Q10 response. | |
| Two Species | An algal species poisons an herbivorous copepod, decreasing its abundance but not its position in the food web. | |
| Pathways: ≥ 3 Species | Rising light levels increase photosynthetic rates of phytoplankton, initiating a trophic escalade. | A top predator consumes several prey species, whose pattern of relative abundance is not maintained in the absence of the predator, resulting in a trophic cascade; however, the nodes and links remain intact. |
| Food Web | Abundances of food web nodes might change, but system identity and overall network configuration remains intact although system exhibits various behaviors. | |
| CONTEXT DEPENDENT | ||
| One Species | A pH change that exceeds a species’ tolerance level causes it to go locally extinct, thereby removing that node and all its links. | As some copepods develop from eggs to adults, their feeding habits change from herbivory to carnivory, and new food webs (or links) emerge. |
| Two Species | An invasive predator species becomes established in the food web, creating a new node with new links and local extinction of the original predator. | A top predator consumes a prey species to such low levels that it becomes locally extinct, and the system changes. |
| Pathways: ≥ 3 Species | Decreasing temperatures cause a decline in fish egg production, initiating a trophic cascade. |
|
| Food Web | Nodes and links are added or subtracted, thus changing food web structure. System identity does not remain intact. |
|
PREDICTIONS 
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|
| ROW NUMBER | TE or TC | FIGURE | PARAMETER INPUT | N | A | H | M | F | P |
| 1 | TE | 1a | +NA | + | + | + | + | + | + |
| 2 | TE | 1a | -NA | - | - | - | - | - | - |
| 3 | TE | 1b | +NB | 0 | + | 0 | + | 0 | + |
| 4 | TE | 1b | -NB | 0 | - | 0 | - | 0 | - |
| 5 | TC | 1a | -PA | + | - | + | - | + | - |
| 6 | TC | 1a | +PA | - | + | - | + | - | + |
| 7 | TC | 1b | -PB | + | - | + | - | + | - |
| 8 | TC | 1b | +PB | - | + | - | + | - | + |

| Loop Length | Operating Paths |
Non-operating Paths |
Total Paths |
N | Negative Feedback Loops |
Positive Feedback Loops | Total Feedback Loops |
|---|---|---|---|---|---|---|---|
| 1 | 54 | 0 | 54 | 1 | 15 | 0 | 15 |
| 2 | 137 | 4 | 141 | 2 | 25 | 0 | 25 |
| 3 | 297 | 10 | 307 | 3 | 5 | 3 | 8 |
| 4 | 552 | 27 | 579 | 4 | 0 | 10 | 10 |
| 5 | 933 | 44 | 977 | 5 | 1 | 7 | 8 |
| 6 | 1376 | 53 | 1429 | 6 | 8 | 7 | 15 |
| 7 | 1682 | 40 | 1722 | 7 | 12 | 2 | 14 |
| 8 | 1631 | 38 | 1669 | 8 | 3 | 3 | 6 |
| 9 | 1322 | 20 | 1342 | 9 | 0 | 2 | 2 |
| 10 | 802 | 9 | 811 | 10 | 0 | 0 | 0 |
| 11 | 378 | 0 | 378 | 11 | 0 | 0 | 0 |
| 12 | 121 | 0 | 121 | 12 | 0 | 0 | 0 |
| 13 | 28 | 0 | 28 | 13 | 0 | 0 | 0 |
| 14 | 0 | 0 | 0 | 14 | 0 | 0 | 0 |
| 15 | 0 | 0 | 0 | 15 | 0 | 0 | 0 |
| 16 | 0 | 0 | 0 | 16 | 0 | 0 | 0 |
| 17 | 0 | 0 | 0 | 17 | 0 | 0 | 0 |
| 18 | 0 | 0 | 0 | 18 | 0 | 0 | 0 |
| 19 | 0 | 0 | 0 | 19 | 0 | 0 | 0 |
| 20 | 0 | 0 | 0 | 20 | 0 | 0 | 0 |
| 21 | 0 | 0 | 0 | 21 | 0 | 0 | 0 |
| Total | 9313 | 245 | 9558 | Total | 69 | 34 | 103 |
| Si | N1 | N2 | A1 | A2 | A3 | A4 | A5 | A6 | Z1 | Z2 | Z3 | Z4 | A | D | M | MD | O | PC | S | T | |
| +Si | + | - | + | - | - | + | - | - | + | + | + | + | - | - | - | - | + | - | - | + | + |
| +N1 | - | + | - | + | + | - | + | + | - | - | - | - | + | + | + | + | - | + | + | - | - |
| +N2 | - | - | + | - | + | + | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - |
| +A1 | ? | ? | ? | + | ? | - | ? | ? | ? | ? | ? | ? | ? | ? | ? | ? | ? | ? | + | ? | ? |
| +A2 | - | - | - | - | + | + | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - |
| +A3 | - | - | + | - | - | + | - | - | - | + | + | + | - | - | - | - | + | - | - | + | + |
| +A4 | + | - | + | - | - | + | + | - | + | + | + | + | - | - | - | - | + | + | - | + | + |
| +A5 | + | - | + | - | - | + | - | + | + | + | + | + | + | - | - | - | + | - | - | + | + |
| +A6 | - | + | - | + | + | - | + | + | + | - | - | - | + | + | + | + | - | + | + | - | - |
| +Z1 | + | + | - | + | + | - | + | + | + | + | - | - | + | + | + | + | - | + | + | - | + |
| +Z2 | + | + | + | - | - | + | + | + | + | + | + | + | + | + | + | - | + | + | - | + | + |
| +Z3 | + | + | + | + | - | - | + | + | + | + | + | + | + | + | + | - | + | + | + | + | + |
| +Z4 | - | + | - | + | + | - | + | - | - | - | - | - | + | + | + | + | - | + | + | - | - |
| +A | ? | + | ? | + | ? | - | - | + | + | - | + | - | + | + | - | - | - | - | + | - | - |
| +D | + | + | + | + | - | - | + | + | + | + | + | + | + | - | + | - | + | + | + | + | + |
| +M | - | - | + | - | - | + | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - |
| +MD | - | - | - | - | + | + | - | - | - | - | - | - | - | - | - | + | + | - | - | - | - |
| +O | - | + | - | + | + | - | - | + | - | - | - | - | + | + | + | + | - | + | + | - | - |
| +PC | - | - | - | + | + | + | - | - | - | - | - | - | - | + | + | - | - | - | + | - | - |
| +S | - | - | - | - | + | + | - | - | - | - | - | - | - | - | - | + | - | - | - | + | - |
| +T | - | - | + | - | - | + | - | - | - | - | + | + | - | - | - | - | + | - | - | + | + |
| Potential Ecosystem Chimera Functions | |||
|---|---|---|---|
| Structure & Function | Secure NEI: Nutrients, Energy, Information | Maintain Modular Identity/Stability | Manage Time |
| 3-Tier Lattice | Built around nutrient input nodes, contains nine nodes most likely to receive environmental drivers and occur as high-frequency links and paths. Accommodates a complete set of feeding types. |
Helps form modular patterns and shapes; lattices are robust structures common in in biological hierarchies. Could be 3-dimensional in nature. Lattice structures are known to be robust in other contexts. |
Lattice structure shortens the time for energy, nutrient, and information flows among the nine key nodes. Promotes a scaffolding in terms of key temporal relationships. |
|
Valid Complement Rule |
Determines the flows of NEI by distinguishing operating from non-operating paths |
Constraints the behavior of nodes not on the path into a set of disjoint loops. | All nodes are slotted into temporal paths and cycles synchronously for each time dominated by an external driver. |
| Stability Rules- Routh-Hurwitz Criteria | Closed loops facilitate recycling and material economies. | Overall pattern of feedback loops, including their lengths and signs, ensures network stability when there is a prevalence of short negative loops versus long positive ones. |
Each feedback loop takes a given amount of time to traverse all its nodes and return to the starting node. Generally, longer loops take longer than short ones. |
| Non-linear Focal Points | Assists in the choice of pathways on which to move nutrients, energy, and information. | Provides the flexibility so that the main lattice module can undergo topological stretching without breaking. |
Gives time management flexibility through the coordination of life history stages. |
|
Satellite Nodes |
Turn pathways on and off as a function of the satellite's self-damping. | Self-damping adds to overall network stability. | As a result of the time given, the satellite will only be self-damped at high densities in an annual cycle. |
|
Network Motif: Auto-regulation |
Keeps nutrients in a steady state, which is often observed in marine environments. |
Central stabilizer in food webs. Helps balance longer positive loops. | Significantly affects temporal relationships. |
|
Network Motif: Feedforward 3 nodes |
Could provide security as well as change NEI rates as conditions warrant. |
Provides path choice, which has temporal ramifications. | It is the primary source of anticipation and can speed up critical interactions. |
|
Network Motif: Bi-fan/Parallel 4 nodes |
Provides pathway flexibility, perhaps increasing security. |
It could help in maintaining stability. | Unclear |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




