Submitted:
14 July 2025
Posted:
15 July 2025
You are already at the latest version
Abstract
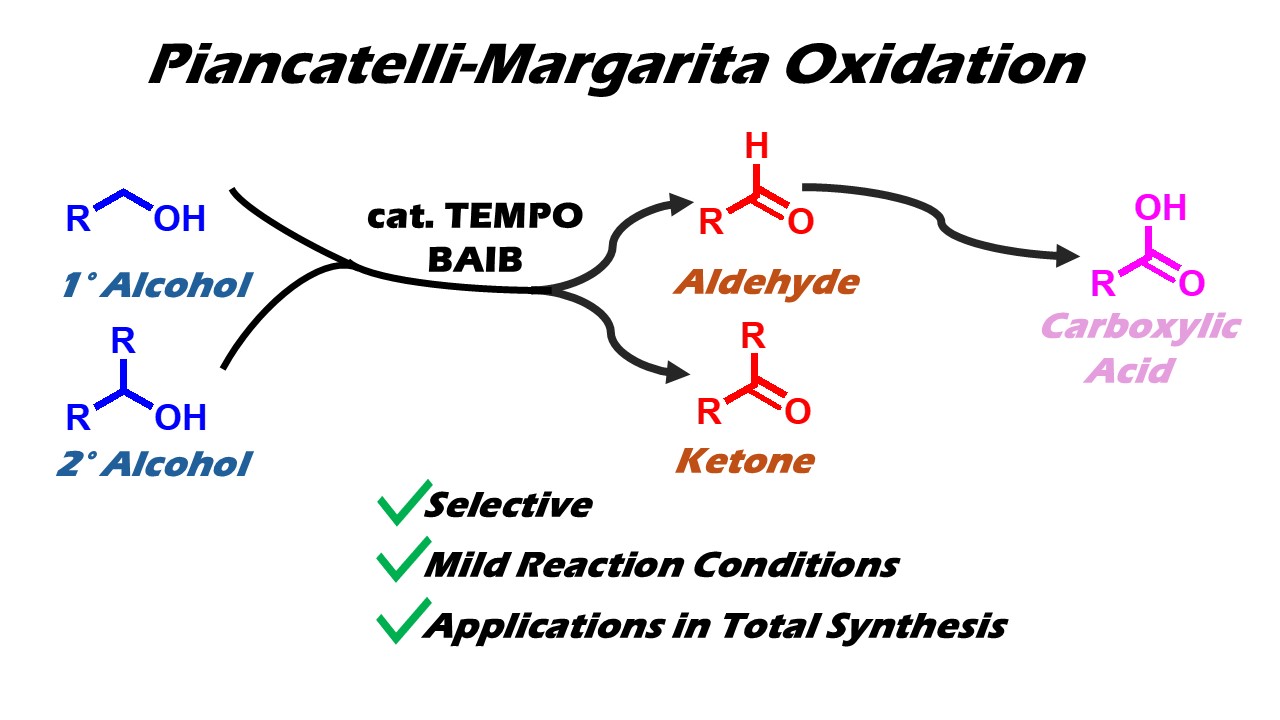
Keywords:
1. Introduction.
2. The Original Piancatelli-Margarita Oxidation.
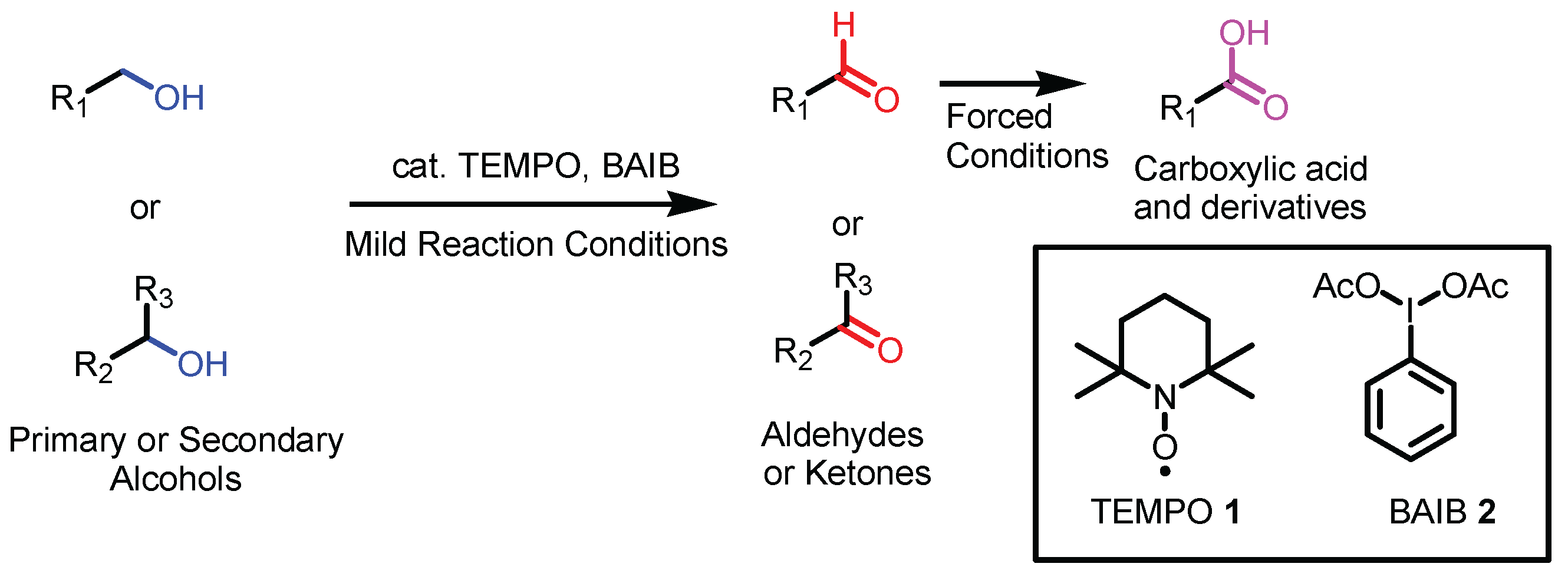
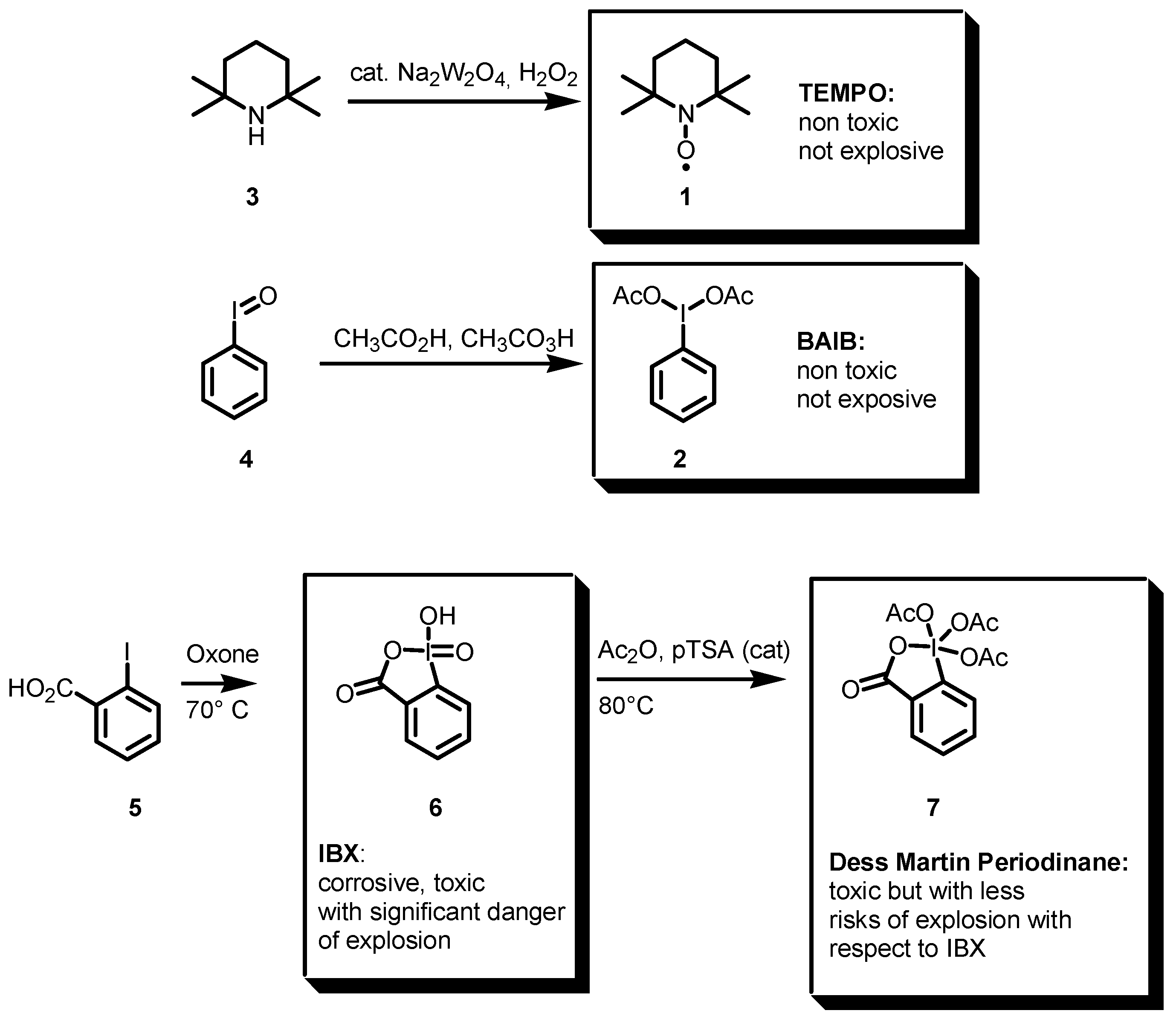
2.1. TEMPO, BAIB, and comparison with other hypervalent iodine compounds as oxidants.
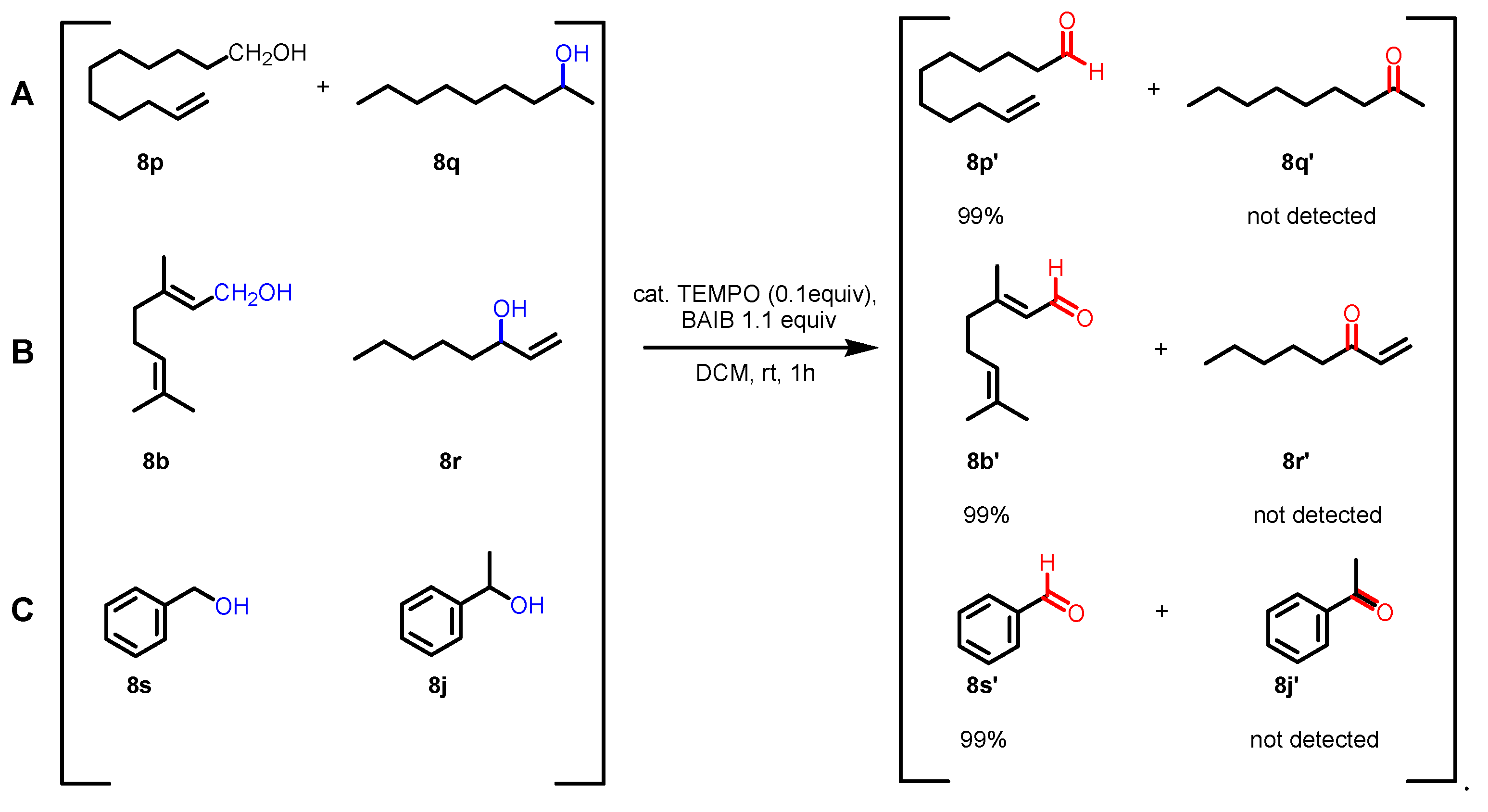
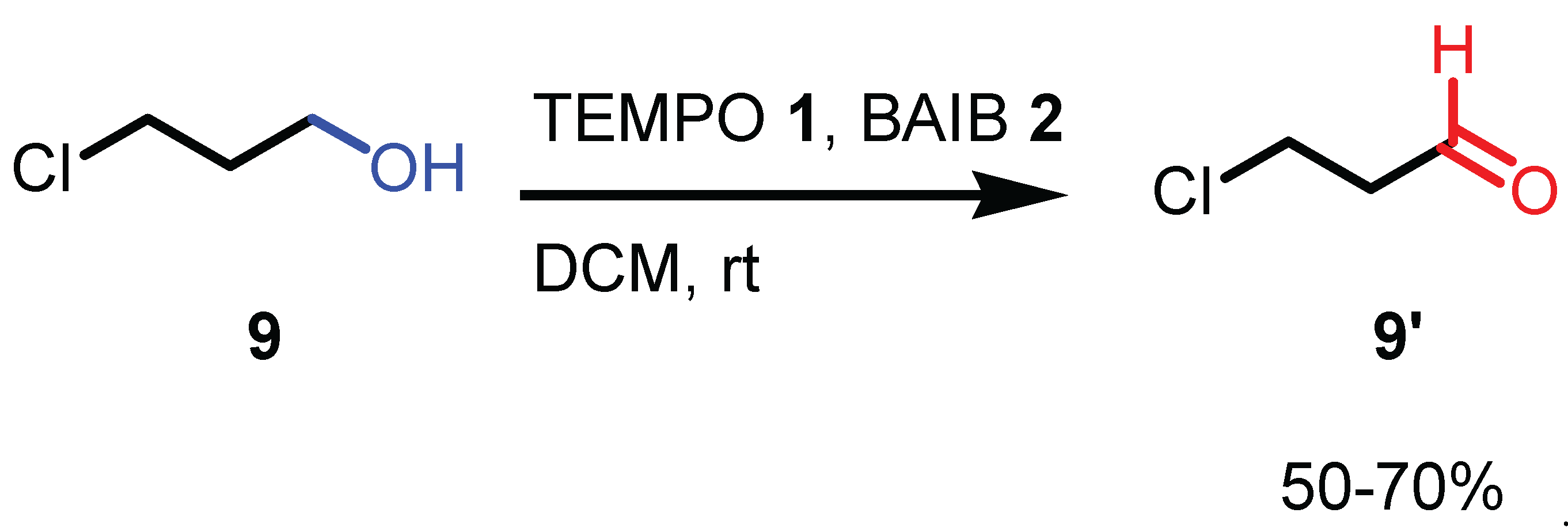
2.2. Reaction conditions and substrate scope in the original paper
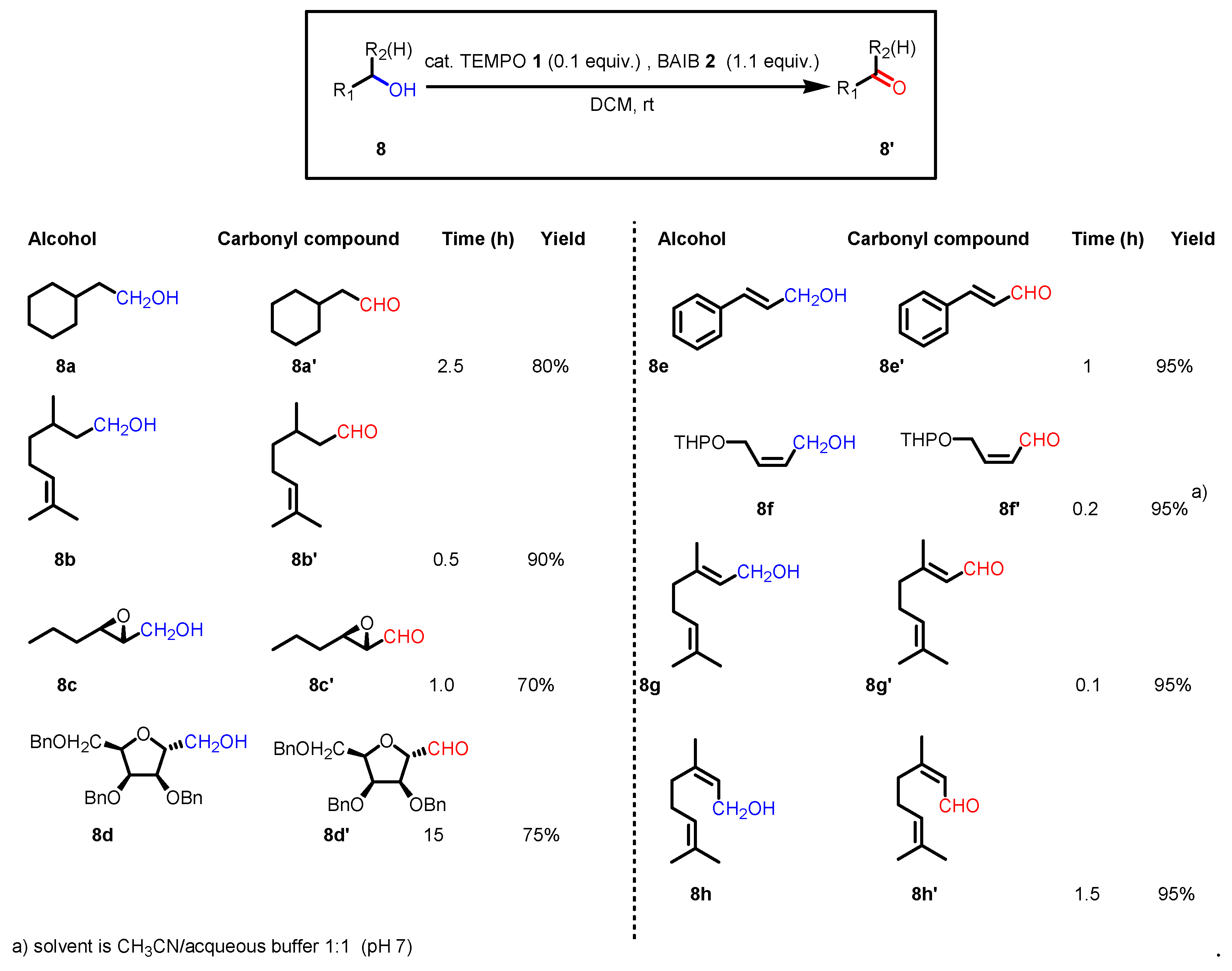
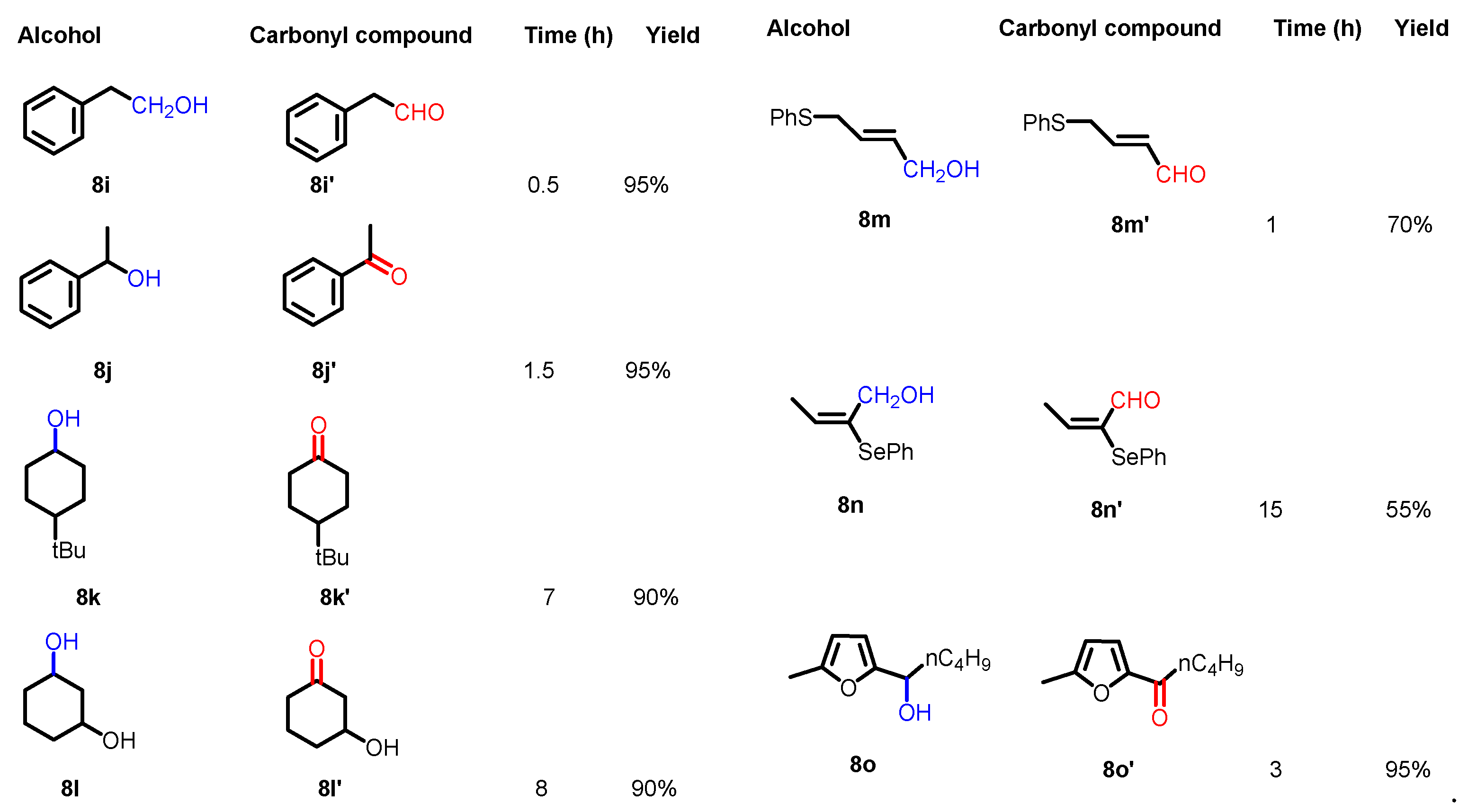
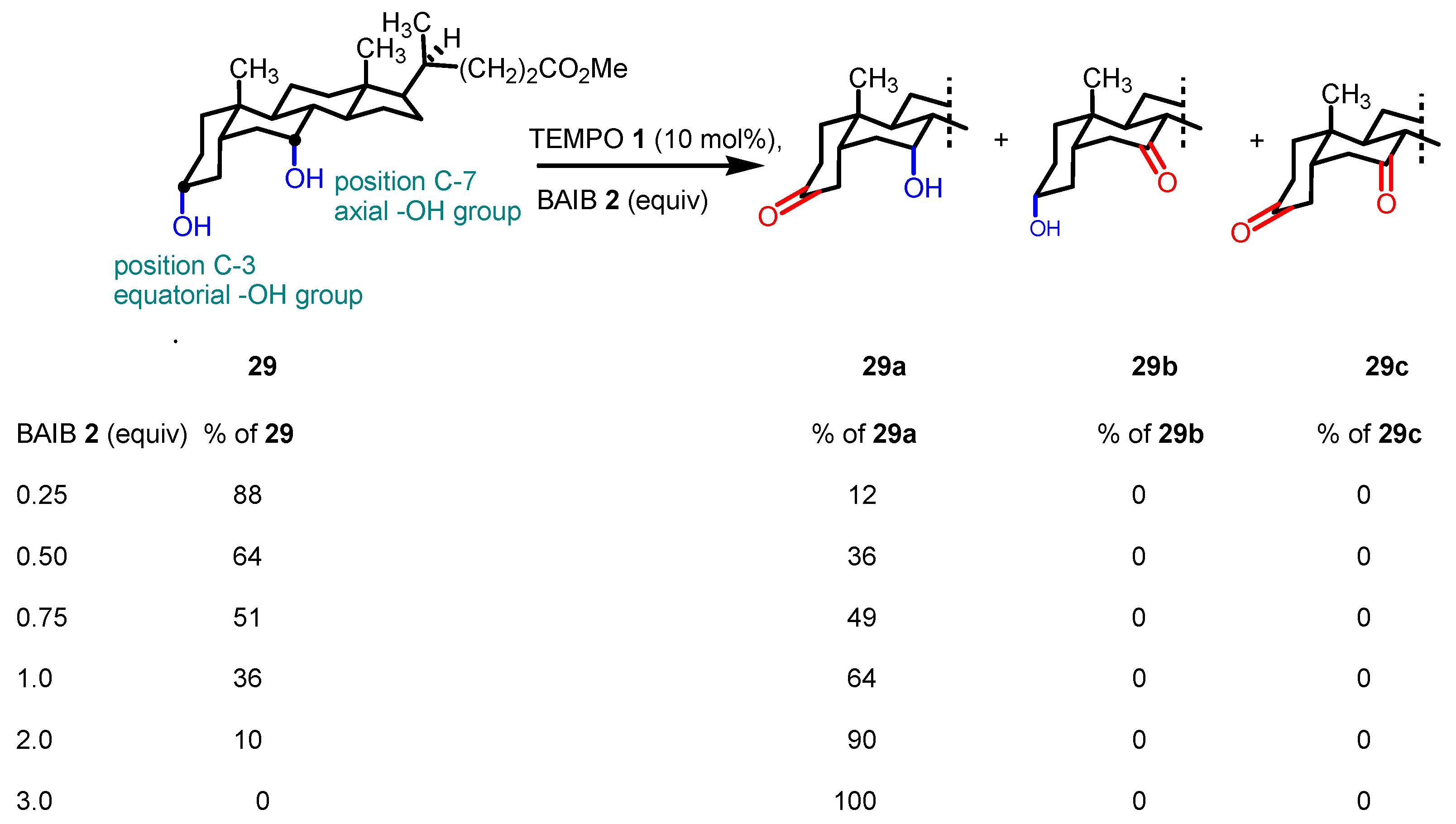
2.3. Key features of the Piancatelli-Margarita oxidation.
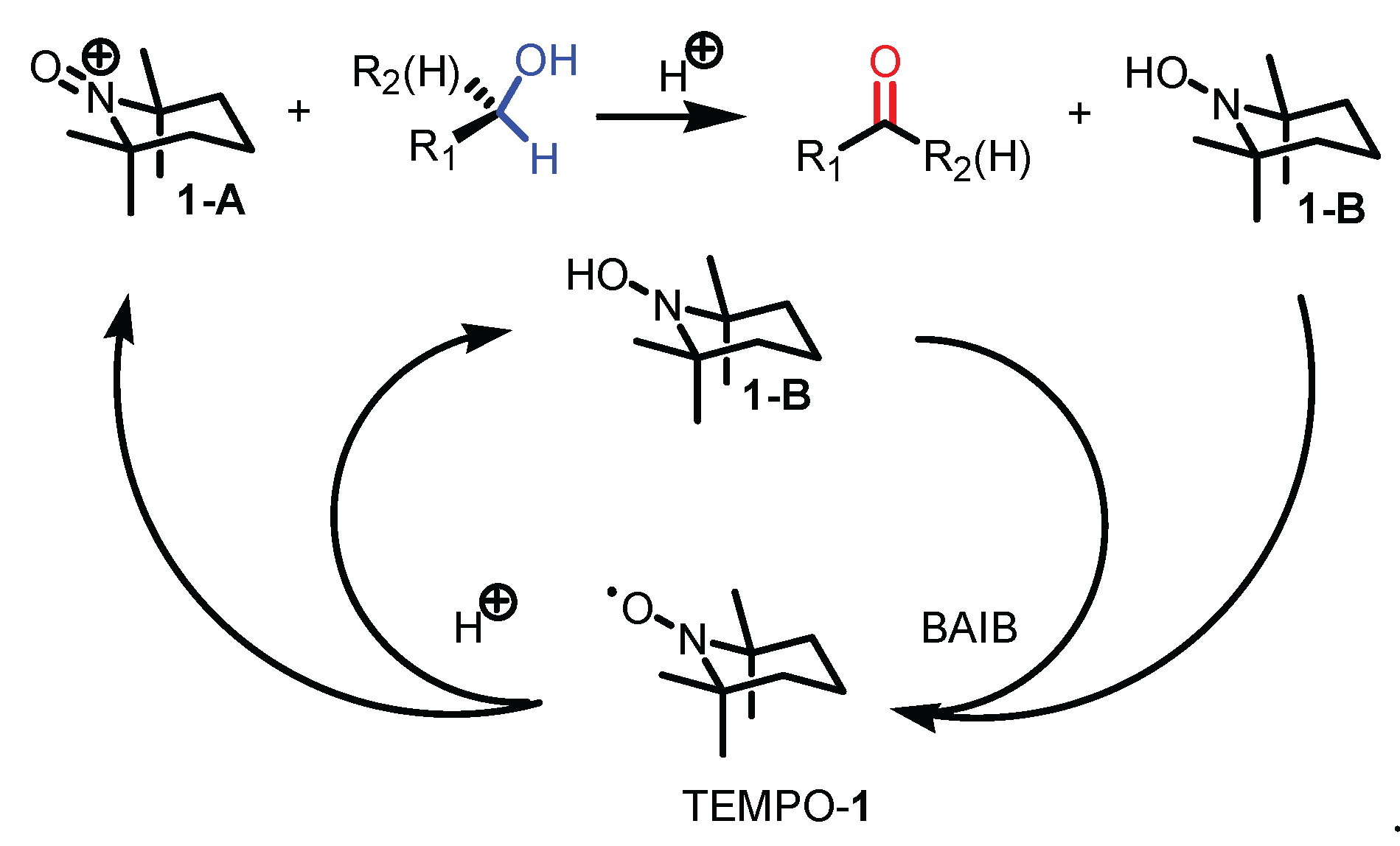
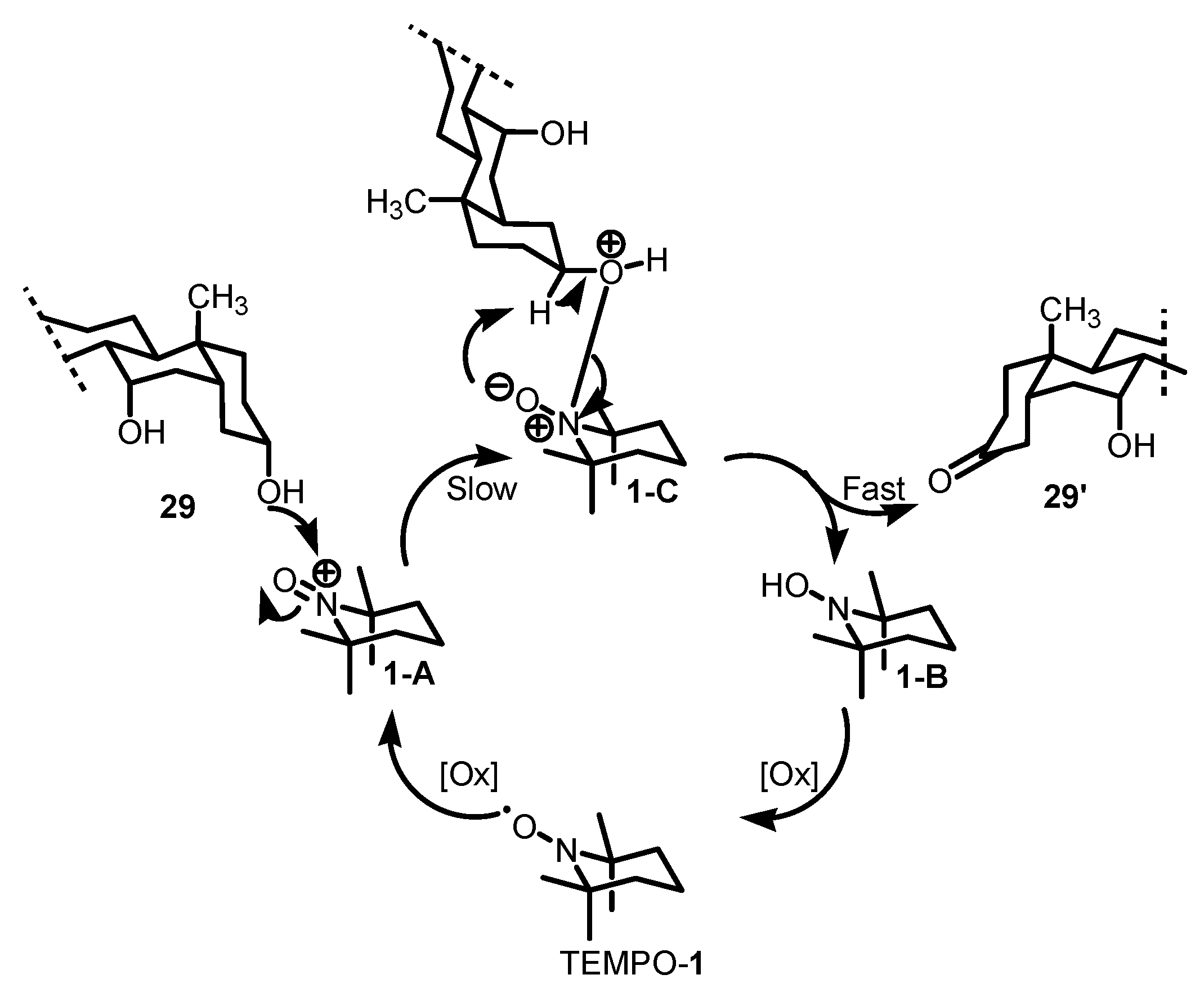
2.4. Proposed reaction mechanism
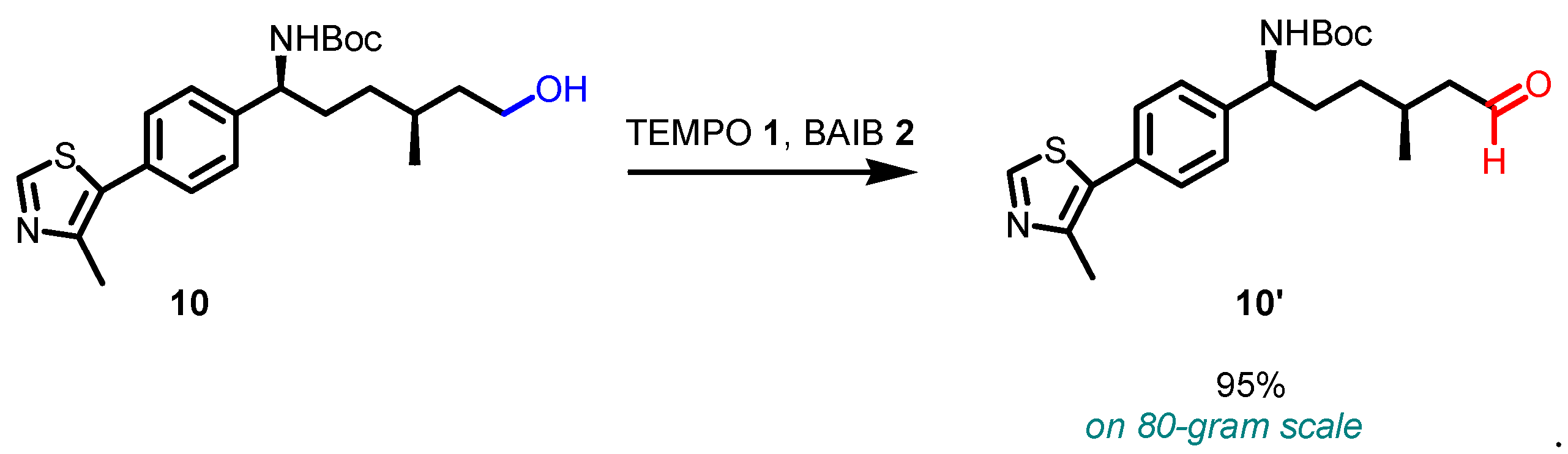
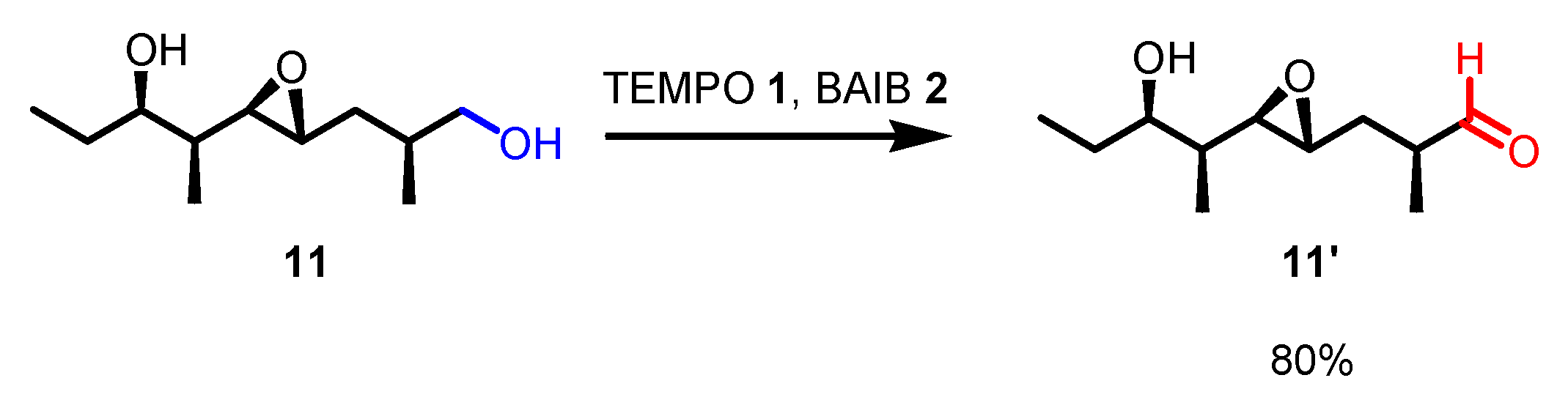
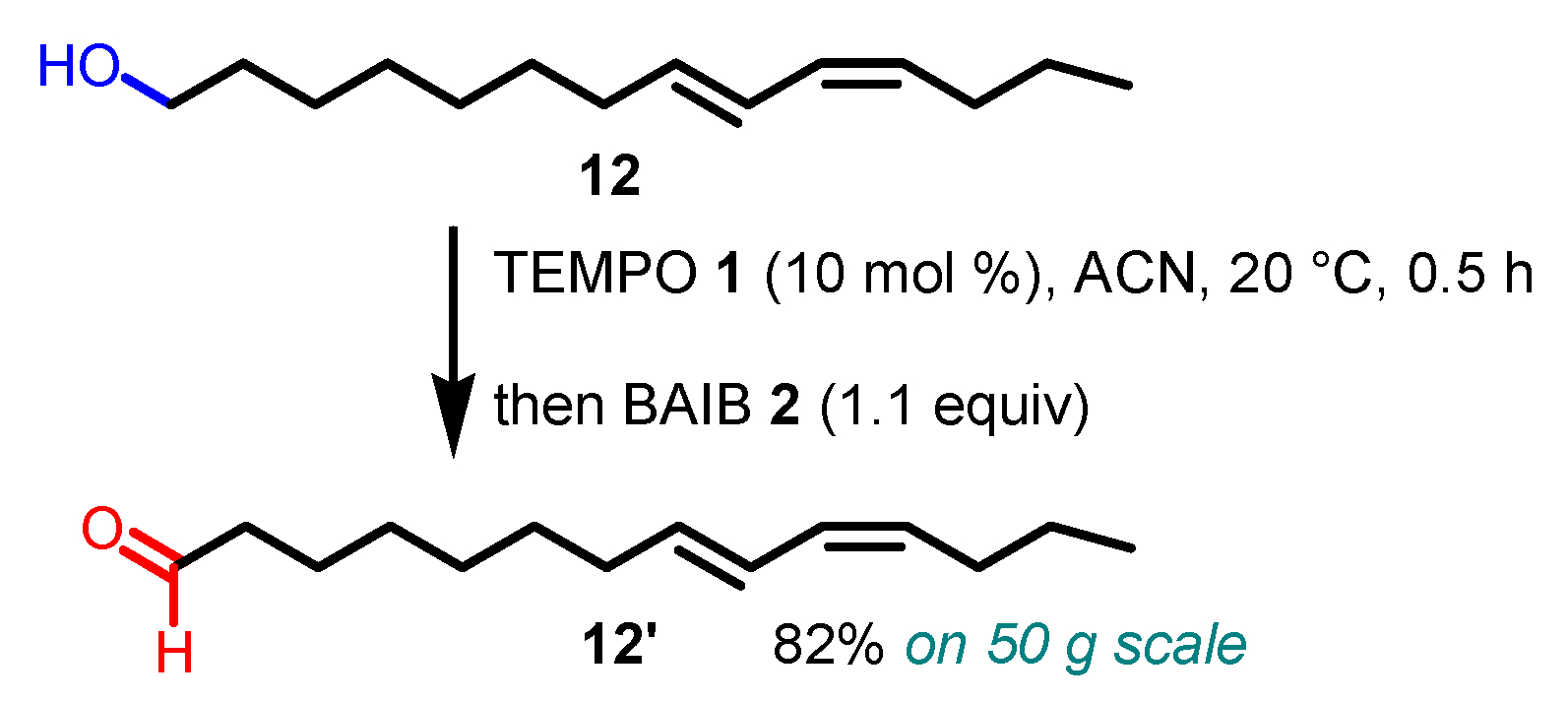
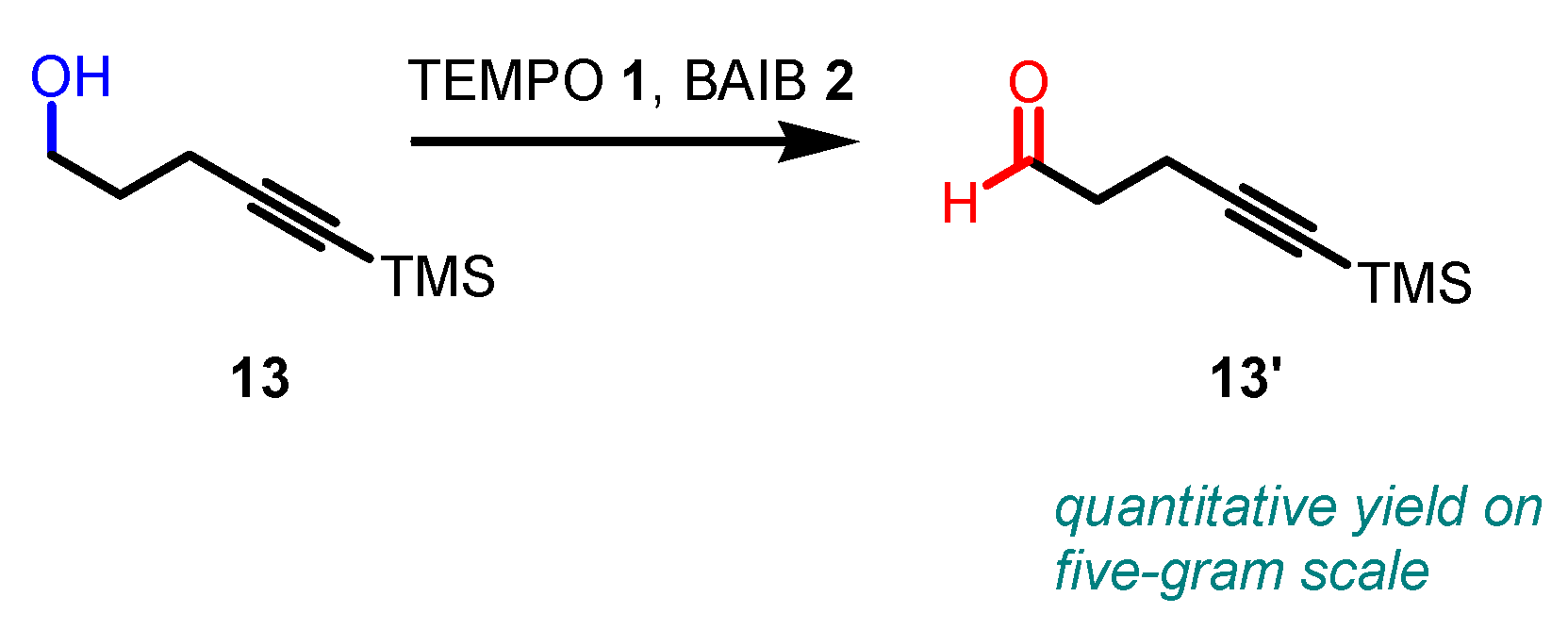
3. Selected examples of Piancatelli-Margarita Oxidation applications in synthesis: large-scale reactions and small molecules.
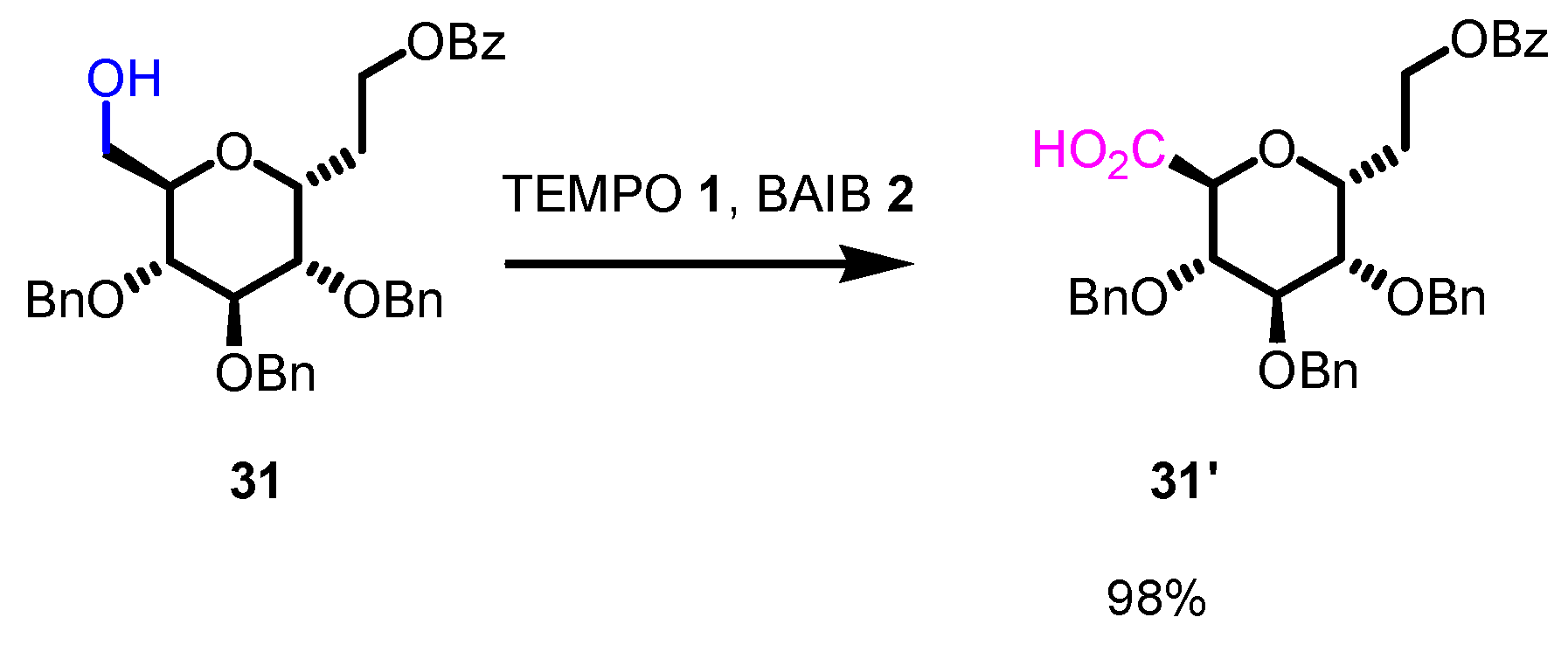
4. Noteworthy applications of Piancatelli-Margarita oxidation in carbohydrate chemistry: examples from the recent literature.
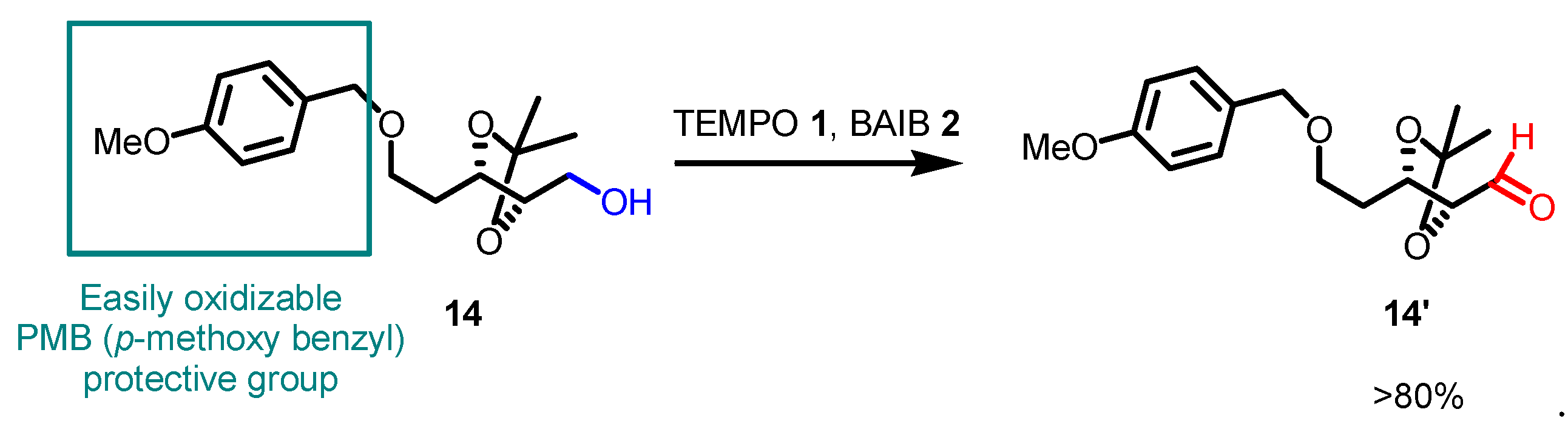
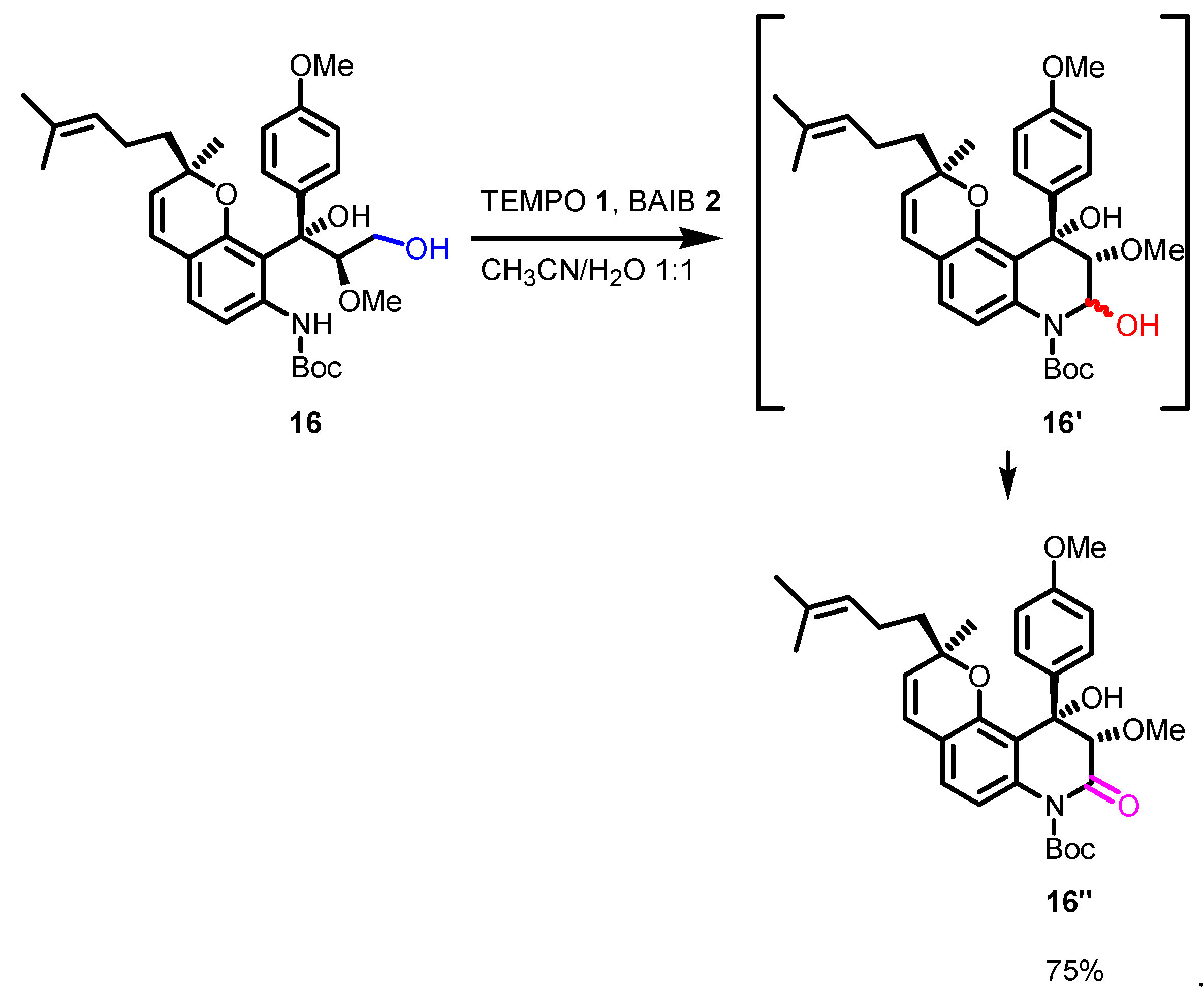
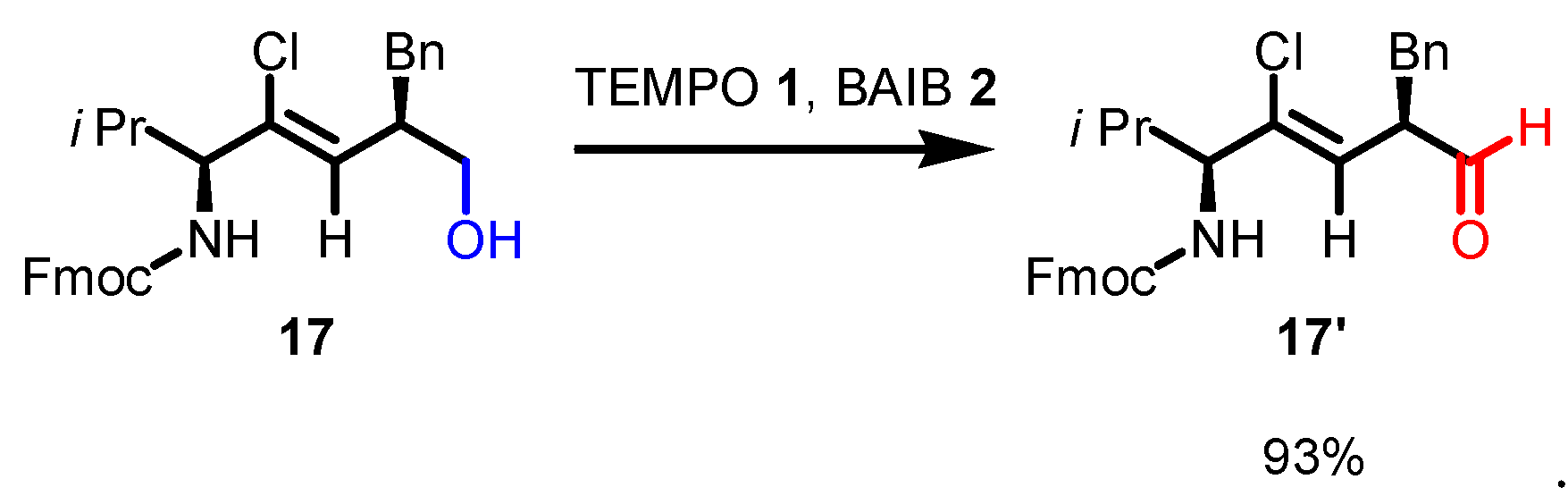
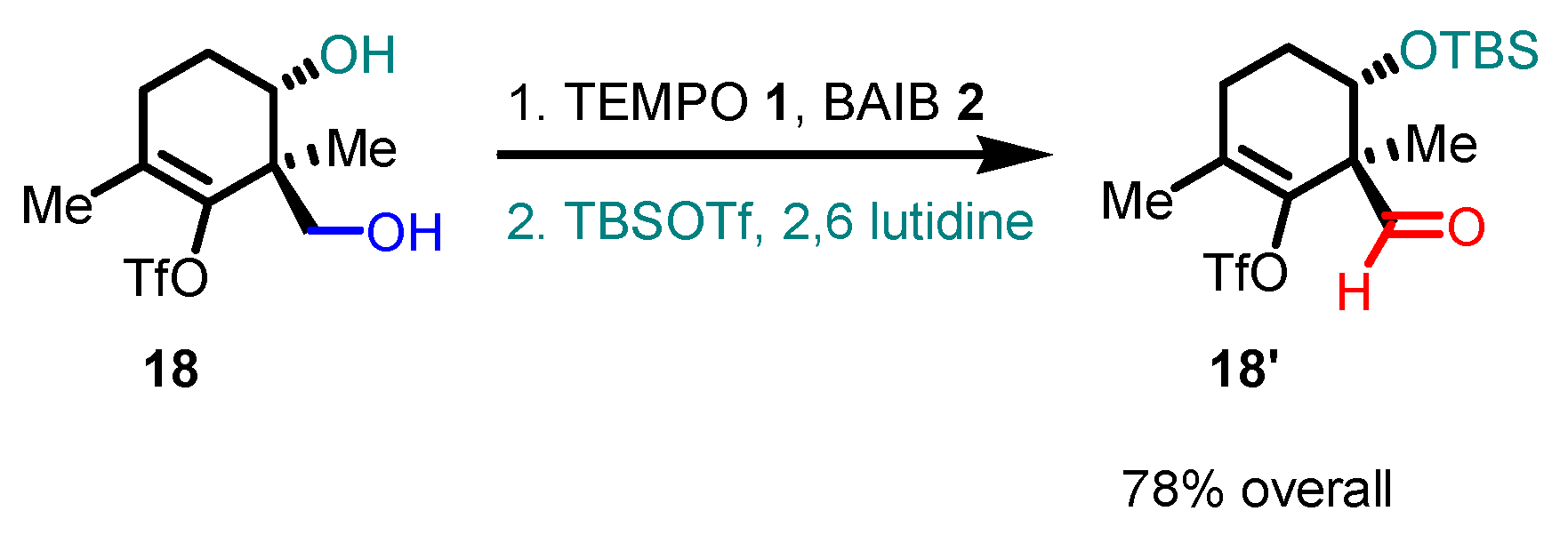
5. Selected examples of Piancatelli-Margarita Oxidation applications in total synthesis: late-stage intermediates and endgame.
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| TEMPO | (2,2,6,6-tetramethylpiperidin-1-yl)oxyl |
| BAIB | bis(acetoxy)iodobenzene |
References
- De Mico, A.; Margarita, R.; Parlanti, L.; Vescovi, A.; Piancatelli, G. A Versatile and Highly Selective Hypervalent Iodine (III)/2,2,6,6-Tetramethyl-1-Piperidinyloxyl-Mediated Oxidation of Alcohols to Carbonyl Compounds. J. Org. Chem. 1997, 62, 6974–6977. [Google Scholar] [CrossRef]
- Piancatelli, G.; Scettri, A.; Barbadoro, S. A useful preparation of 4-substituted 5-hydroxy-3-oxocyclopentene. Tetrahedron Lett. 1976, 39, 3555–3558. [Google Scholar] [CrossRef]
- Piancatelli, G.; Leonelli, F. Oxidation of nerol to neral with iodobenzene and TEMPO. Org. Synth. 2006, 83, 18–23. [Google Scholar] [CrossRef]
- Leonelli, F.; Margarita, R.; Piancatelli, G. ; Discussion addendum for: oxidation of Nerol to Neral with iodosobenzene and TEMPO. Org. Synth. 2012, 89, 311–322. [Google Scholar] [CrossRef]
- Montanari, F.; Quici, S.; Henry-Riyad, H.; Tidwell, T. T. 2,2,6,6-Tetramethylpiperidin-1-oxyl. In Encyclopedia of Reagents for Organic Synthesis, John Wiley & Sons, 2005. https; ISBN 0471936235. [CrossRef]
- Anelli, P. L.; Biffi, C.; Montanari, F.; Quici, S. Fast and selective oxidation of primary alcohols to aldehydes or to carboxylic acids and of secondary alcohols to ketones mediated by oxoammonium salts under two-phase conditions, J. Org. Chem. 1987, 52, 2559–2562. [Google Scholar] [CrossRef]
- Ciriminna, R.; Pagliaro, M. Industrial Oxidations with Organocatalyst TEMPO and Its Derivatives. Org. Process Res. Dev. 2010, 14, 245–251. [Google Scholar] [CrossRef]
- https://www.sigmaaldrich.com/. Available online: https://www.sigmaaldrich.com/US/en/substance/tempo156252564832 (accessed on 12 July 2025).
- Moriarty R., M.; Chany, C. J.; Kosmeder, J. W.; Du Bois, J. Diacetoxyiodo)benzene. In Encyclopedia of Reagents for Organic Synthesis, John Wiley & Sons, 2001. ISBN 9780470842898. [CrossRef]
- Frigerio, M.; Santagostino, M.; Sputore, S. A User-Friendly Entry to 2-Iodoxybenzoic Acid (IBX). J. Org. Chem. 1999, 64, 4537–4538. [Google Scholar] [CrossRef]
- Boeckman, R. J.; George, K. M. 1,1,1-Triacetoxy-1,1-dihydro-1,2-benziodoxol-3(1H)-one. In Encyclopedia of Reagents for Organic Synthesis, 2009. ISBN 978-0471936237. [CrossRef]
- https://www.sigmaaldrich.com/. Available online: https://www.sigmaaldrich.com/US/en/search/ibx?focus=products&page=1&perpage=30&sort=relevance&term=ibx&type=product (accessed on 12 07 2025).
- https://www.sigmaaldrich.com/. Available online: https://www.sigmaaldrich.com/US/en/search/dess-martin-periodinane?focus=products&page=1&perpage=30&sort=relevance&term=Dess-Martin%20periodinane&type=product (accessed on 12 July 2025).
- https://www.sigmaaldrich.com/. Available online: https://www.sigmaaldrich.com/US/en/sds/aldrich/178721?userType=anonymous (accessed on 12 July 2025).
- https://www.sigmaaldrich.com/. Available online: https://www.sigmaaldrich.com/US/en/search/baib?focus=products&page=1&perpage=30&sort=relevance&term=BAIB&type=product (accessed on 12 July 2025).
- Zelch, D.; Russo, C. M.; Ruud, K. J.; O’Reilly, M. C. A General and Scalable Method toward Enantioenriched C2-Substituted Azetidines Using Chiral Tert-Butanesulfinamides. J. Org. Chem. 2024, 89, 15137–15144. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Wu, J.-P.; Lee, J.; Wang, J.; White, J. A. H.; Rugg, K. W.; Sienkiewicz, A.; Lorenz, J. C.; Greb, P.; Bunner, M. H.; Hirsh, D. A.; Gonnella, N. C.; Nordstrom, F. L.; Kofink, C.; Weinstabl, H.; Reeves, J. T. A Chiral Pool Strategy for the Synthesis of a SMARCA2 Degrading PROTAC. Org. Process Res. Dev. 2024, 28, 1239–1252. [Google Scholar] [CrossRef]
- Rhoades, D.; Rheingold, A. L.; O’Malley, B. W.; Wang, J. Expedient Total Syntheses of Pladienolide-Derived Spliceosome Modulators. J. Am. Chem. Soc. 2021, 143, 4915–4920. [Google Scholar] [CrossRef] [PubMed]
- Chourreu, P.; Guerret, O.; Guillonneau, L.; Gayon, E.; Lefèvre, G. Short and Easily Scalable Synthesis of the Sex Pheromone of the Horse-Chestnut Leaf Miner (Cameraria Ohridella) Relying on a Key Ligand- and Additive-Free Iron-Catalyzed Cross-Coupling. Org. Process Res. Dev. 2020, 24, 1335–1340. [Google Scholar] [CrossRef]
- Kaden, F.; Nowotni, S.; Höfner, F.; Lorenz, M.; Barthel, A.; Jäger, A.; Hennersdorf, F.; Weigand, J. J.; Metz, P. Asymmetric Total Synthesis of (−)-Dehydrocostus Lactone by Domino Metathesis. Eur J Org Chem 2021, 2021, 3579–3586. [Google Scholar] [CrossRef]
- Ghosh, A. K.; Hsu, C.-S. Enantioselective Total Synthesis of (+)-EBC-23, a Potent Anticancer Agent from the Australian Rainforest. J. Org. Chem. 2021, 86, 6351–6360. [Google Scholar] [CrossRef] [PubMed]
- Movahhed, S.; Westphal, J.; Kempa, A.; Schumacher, C. E.; Sperlich, J.; Neudörfl, J.; Teusch, N.; Hochgürtel, M.; Schmalz, H. Total Synthesis of (+)-Erogorgiaene and the Pseudopterosin A−F Aglycone via Enantioselective Cobalt-Catalyzed Hydrovinylation. Chemistry A European J 2021, 27, 11574–11579. [Google Scholar] [CrossRef] [PubMed]
- Vece, V.; Jakkepally, S.; Hanessian, S. Total Synthesis and Absolute Stereochemical Assignment of the Insecticidal Metabolites Yaequinolones J1 and J2. Org. Lett. 2018, 20, 4277–4280. [Google Scholar] [CrossRef] [PubMed]
- Kobayakawa, T.; Azuma, C.; Watanabe, Y.; Sawamura, S.; Taniguchi, A.; Hayashi, Y.; Tsuji, K.; Tamamura, H. Development of Methods for Convergent Synthesis of Chloroalkene Dipeptide Isosteres and Its Application. J. Org. Chem. 2021, 86, 5091–5101. [Google Scholar] [CrossRef] [PubMed]
- Perea, M. A.; Wang, B.; Wyler, B. C.; Ham, J. S.; O’Connor, N. R.; Nagasawa, S.; Kimura, Y.; Manske, C.; Scherübl, M.; Nguyen, J. M.; Sarpong, R. General Synthetic Approach to Diverse Taxane Cores. J. Am. Chem. Soc. 2022, 144, 21398–21407. [Google Scholar] [CrossRef] [PubMed]
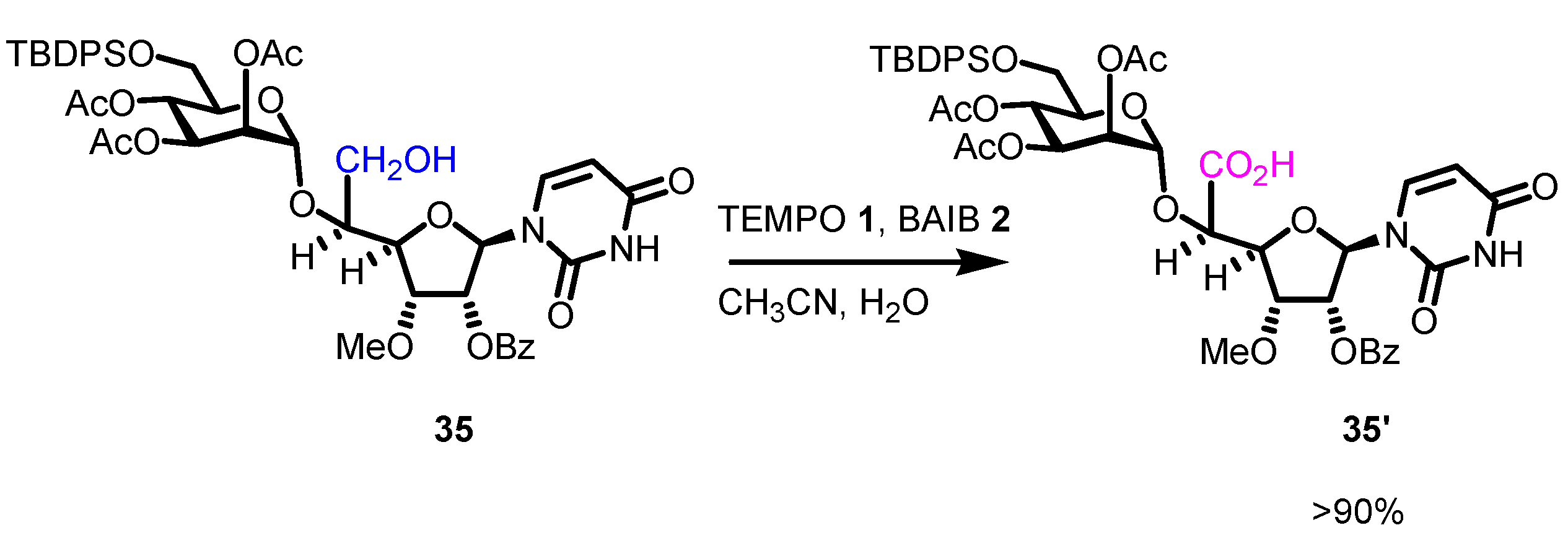
- Jeanneret, R. A.; Dalton, C. E.; Gardiner, J. M. Synthesis of Heparan Sulfate- and Dermatan Sulfate-Related Oligosaccharides via Iterative Chemoselective Glycosylation Exploiting Conformationally Disarmed [2.2.2] l-Iduronic Lactone Thioglycosides. J. Org. Chem. 2019, 84, 15063–15078. [Google Scholar] [CrossRef] [PubMed]
- Lusi, R. F.; Sennari, G.; Sarpong, R. Strategy Evolution in a Skeletal Remodeling and C–H Functionalization-Based Synthesis of the Longiborneol Sesquiterpenoids. J. Am. Chem. Soc. 2022, 144, 17277–17294. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, P.; Li, T.; Zhang, M.; Liu, W.; Li, J.; Wang, L.; Chen, Y. Synthesis of the ABC Ring System of Kadlongilactones. Org. Biomol. Chem. 2023, 21, 1704–1708. [Google Scholar] [CrossRef] [PubMed]
- López-Mendoza, P.; Porras-Santos, L. F.; Pérez-Bautista, J. A.; Quintero, L.; Bautista-Nava, J.; León-Rayo, D. F.; Cordero-Vargas, A.; Sartillo-Piscil, F. En Route to Furan-Fused Naphthopyrones: Formal Synthesis of the (+)-Lasionectrin and Its C12-Epimer. J. Org. Chem. 2023, 88, 17409–17419. [Google Scholar] [CrossRef] [PubMed]
- Takamura, H.; Sugitani, Y.; Morishita, R.; Yorisue, T.; Kadota, I. Total Synthesis and Structure–Antifouling Activity Relationship of Scabrolide F. Org. Biomol. Chem. 2024, 22, 5739–5747. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Shimokawa, J.; Yorimitsu, H. The Dioxasilepanyl Group as a Versatile Organometallic Unit: Studies on Stability, Reactivity, and Utility. Chem. Sci. 2021, 12, 9546–9555. [Google Scholar] [CrossRef] [PubMed]
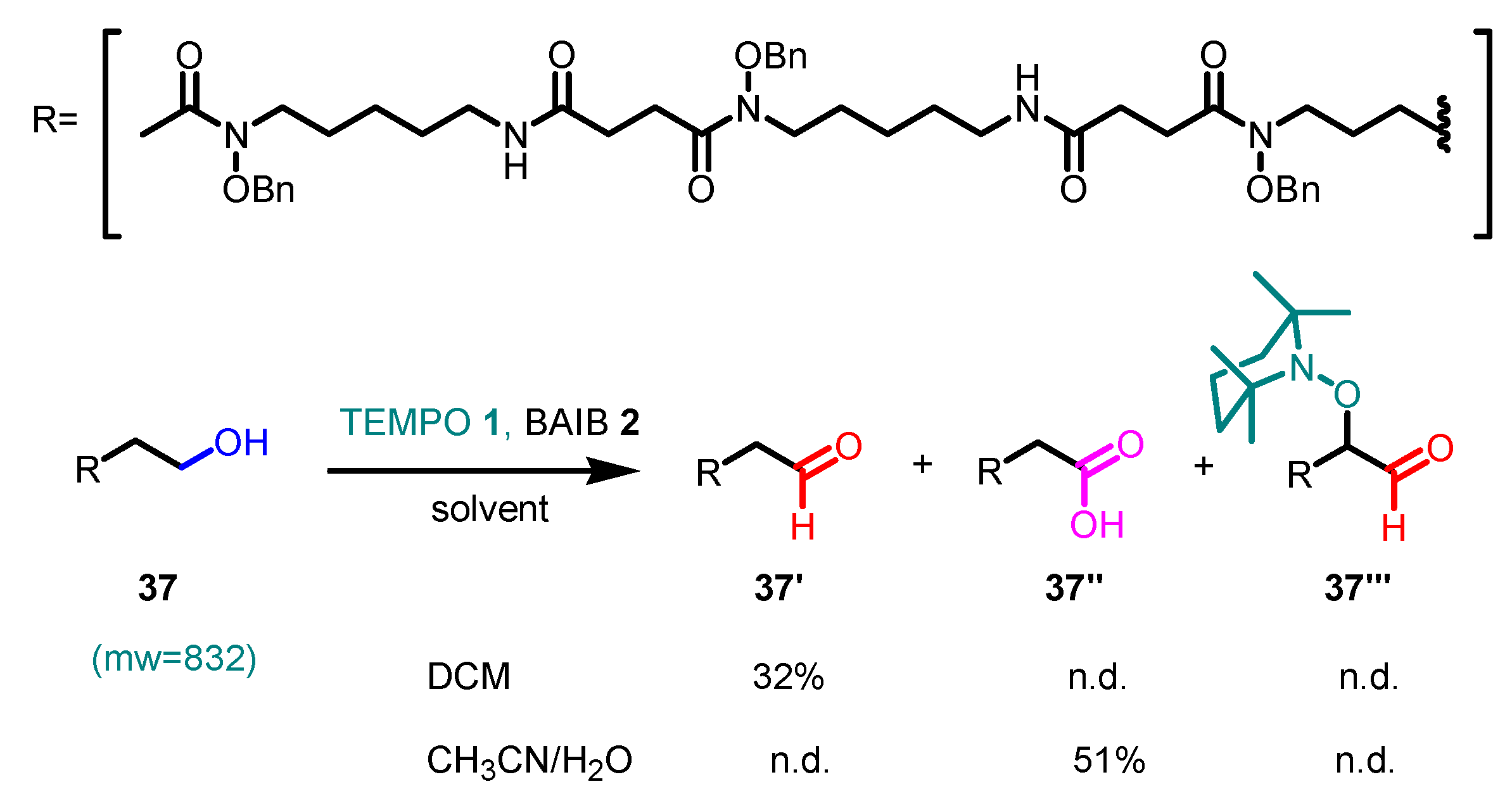
- Kaspar, M.; Kudova, E. Selectivity of Oxidizing Agents toward Axial and Equatorial Hydroxyl Groups. J. Org. Chem. 2022, 87, 9157–9170. [Google Scholar] [CrossRef] [PubMed]
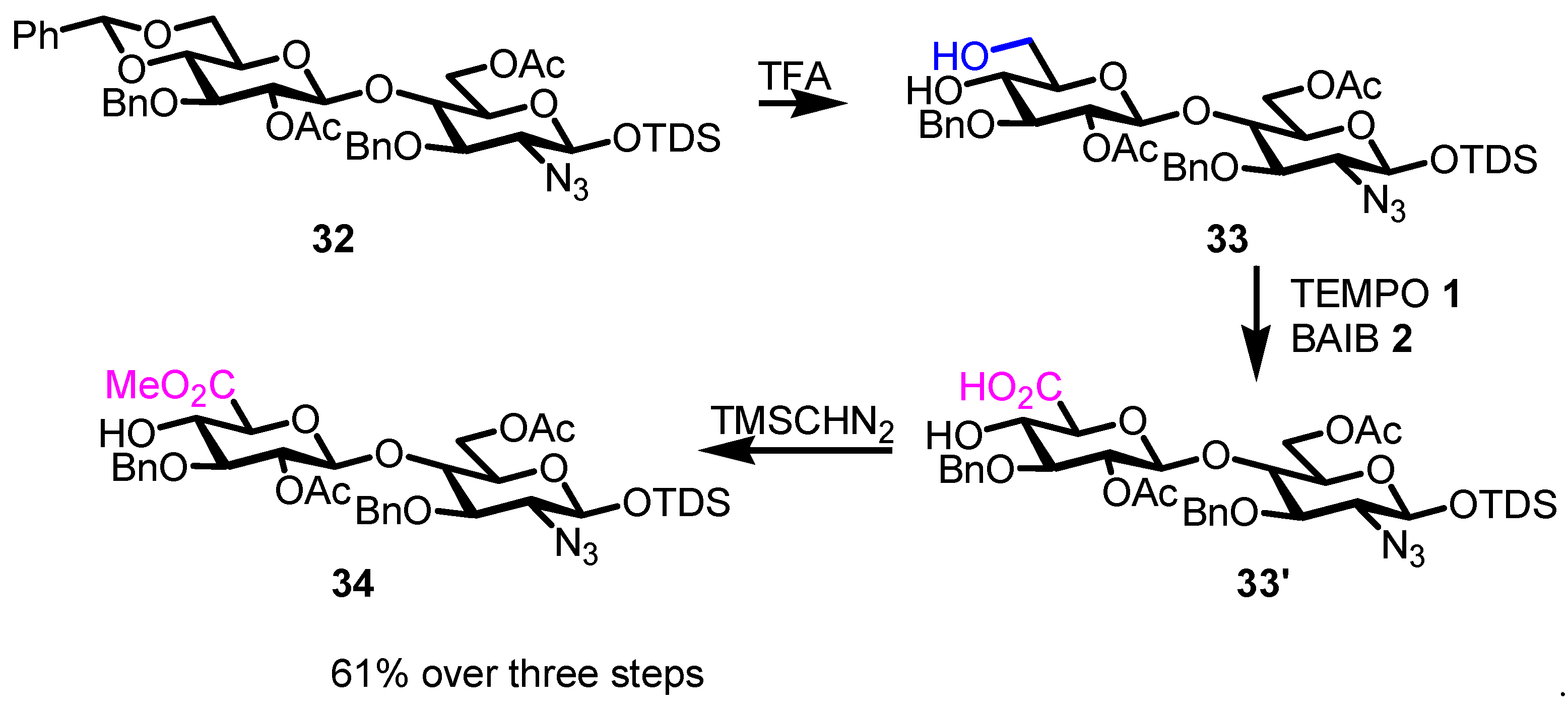
- Li, T.; Wang, J.; Zhu, X.; Zhou, X.; Sun, S.; Wang, P.; Cao, H.; Yu, G.; Li, M. Synthesis of Rare 6-Deoxy-d-/l-Heptopyranosyl Fluorides: Assembly of a Hexasaccharide Corresponding to Campylobacter Jejuni Strain CG8486 Capsular Polysaccharide. J. Am. Chem. Soc. 2021, 143, 11171–11179. [Google Scholar] [CrossRef] [PubMed]
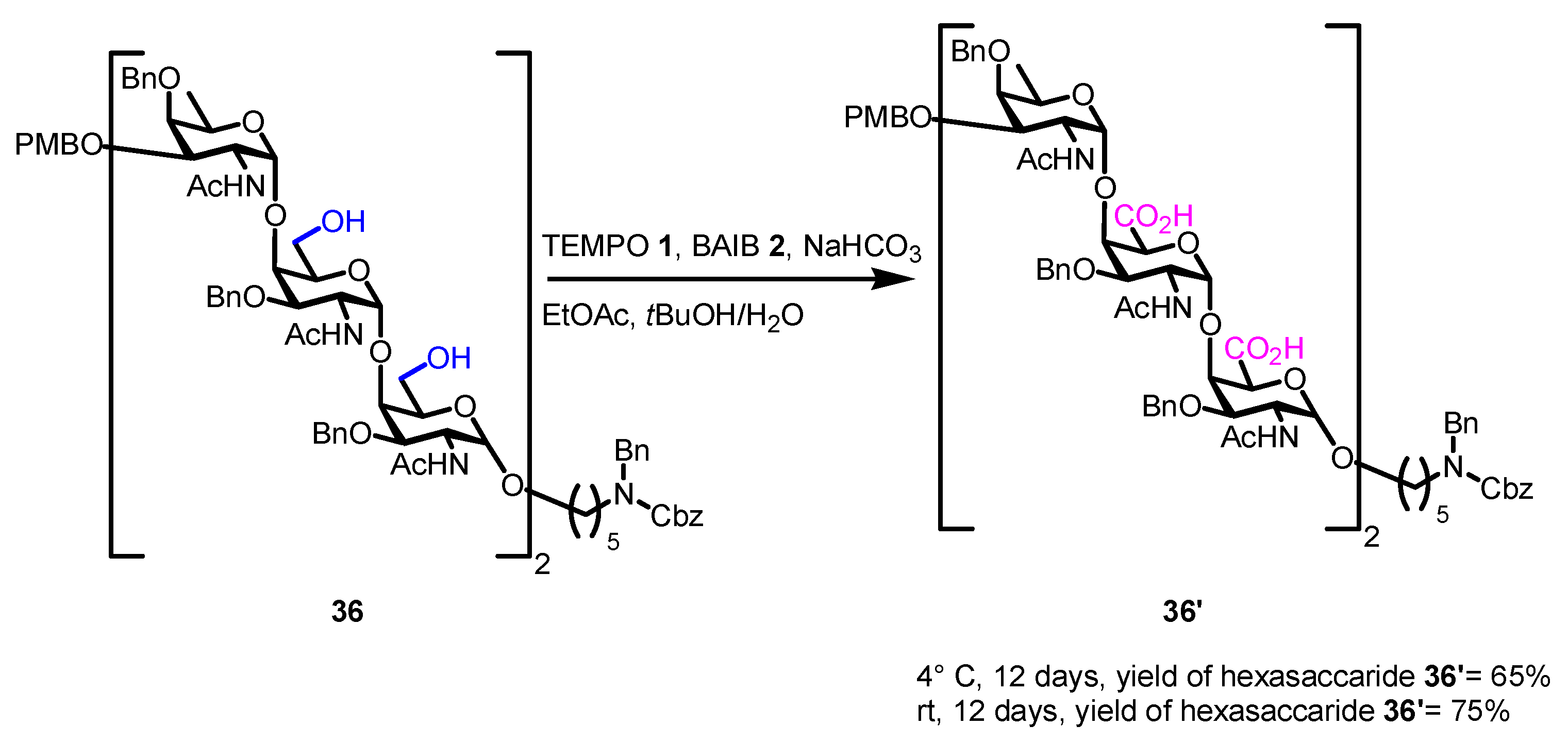
- Sun, L.; Chopra, P.; Boons, G.-J. Modular Synthesis of Heparan Sulfate Oligosaccharides Having N-Acetyl and N-Sulfate Moieties. J. Org. Chem. 2020, 85, 16082–16098. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Xu, L.; Sun, R.; Zhang, Y.; Huang, Y.; Chen, Z.; Li, P.; Yang, R.; Xiao, G. An Orthogonal and Reactivity-Based One-Pot Glycosylation Strategy for Both Glycan and Nucleoside Synthesis: Access to TMG-Chitotriomycin, Lipochitooligosaccharides and Capuramycin. Chem. Sci. 2021, 12, 5143–5151. [Google Scholar] [CrossRef] [PubMed]
- Østerlid, K. E.; Cergano, R.; Overkleeft, H. S.; Van Der Marel, G. A.; Codée, J. D. C. Synthesis of a Set of Staphylococcus Aureus Capsular Polysaccharide Type 1 Oligosaccharides Carrying Taurine Esters. Chemistry A European J 2025, 31. [Google Scholar] [CrossRef] [PubMed]
- Zujew, L.; Raibaut, L.; Chambron, J. From Desferrioxamine B Umpolung to an Enantiopure Bifunctional Chelator for 89Zr-immunoPET. Chemistry A European J 2025, 31. [Google Scholar] [CrossRef] [PubMed]
- Fadel, M.; Carreira, E. M. Enantioselective Total Synthesis of (+)-Pedrolide. J. Am. Chem. Soc. 2023, 145, 8332–8337. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Hu, J.; Chen, R.; Xiong, F.; Xie, H.; Ding, H. Divergent Total Syntheses of (−)-Crinipellins Facilitated by a HAT-Initiated Dowd–Beckwith Rearrangement. J. Am. Chem. Soc. 2022, 144, 2495–2500. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Flaxman, H. A.; Woo, C. M. Enantioselective Synthesis and Biological Evaluation of Sanglifehrin A and B and Analogs. Angew Chem Int Ed 2021, 60, 17045–17052. [Google Scholar] [CrossRef] [PubMed]
- Xin, Z.; Wang, H.; He, H.; Zhao, X.; Gao, S. Asymmetric Total Synthesis of Norzoanthamine. Angew Chem Int Ed 2021, 60, 12807–12812. [Google Scholar] [CrossRef] [PubMed]
- Anketell, M. J.; Sharrock, T. M.; Paterson, I. Total Synthesis of the Actinoallolides and a Designed Photoaffinity Probe for Target Identification. Org. Biomol. Chem. 2020, 18, 8109–8118. [Google Scholar] [CrossRef] [PubMed]
- Anketell, M. J.; Sharrock, T. M.; Paterson, I. A Unified Total Synthesis of the Actinoallolides, a Family of Potent Anti-Trypanosomal Macrolides. Angew Chem Int Ed 2020, 59, 1572–1576. [Google Scholar] [CrossRef] [PubMed]
- Moore, M. J.; Qu, S.; Tan, C.; Cai, Y.; Mogi, Y.; Jamin Keith, D.; Boger, D. L. Next-Generation Total Synthesis of Vancomycin. J. Am. Chem. Soc. 2020, 142, 16039–16050. [Google Scholar] [CrossRef] [PubMed]
- Moore, M. J.; Qin, P.; Yamasaki, N.; Zeng, X.; Keith, D. J.; Jung, S.; Fukazawa, T.; Graham-O’Regan, K.; Wu, Z.-C.; Chatterjee, S.; Boger, D. L. Tetrachlorovancomycin: Total Synthesis of a Designed Glycopeptide Antibiotic of Reduced Synthetic Complexity. J. Am. Chem. Soc. 2023, 145, 21132–21141. [Google Scholar] [CrossRef] [PubMed]


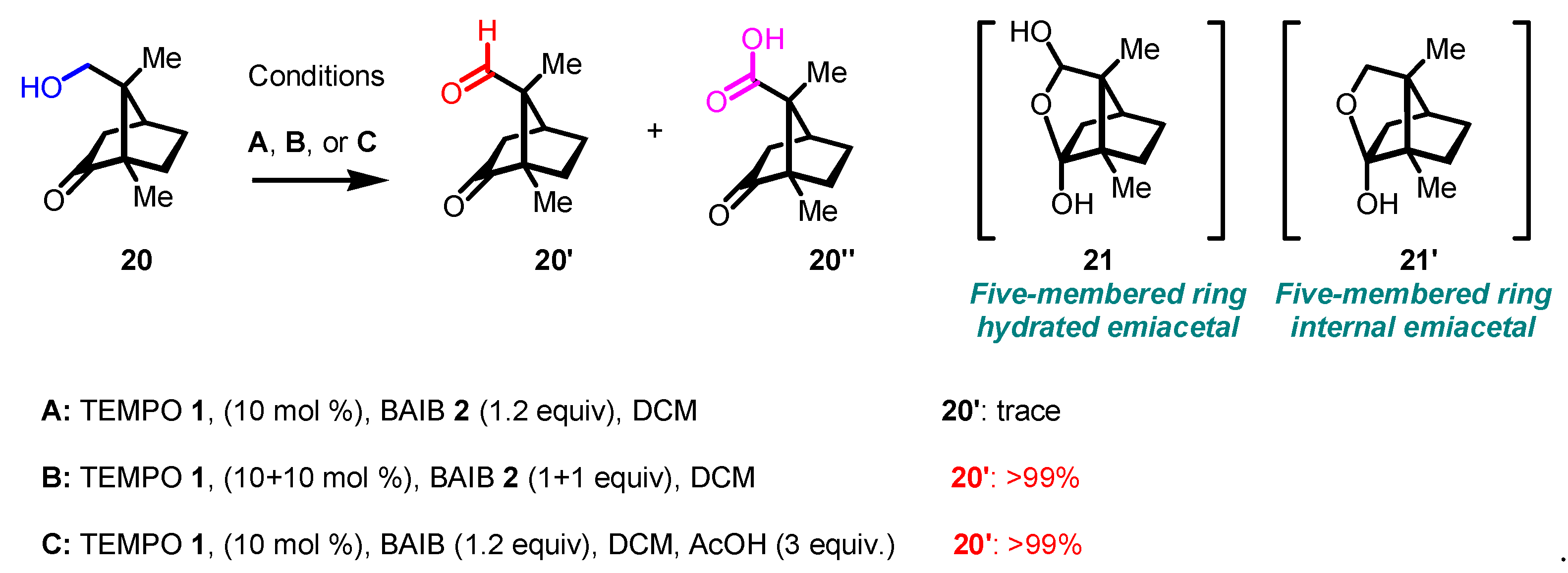
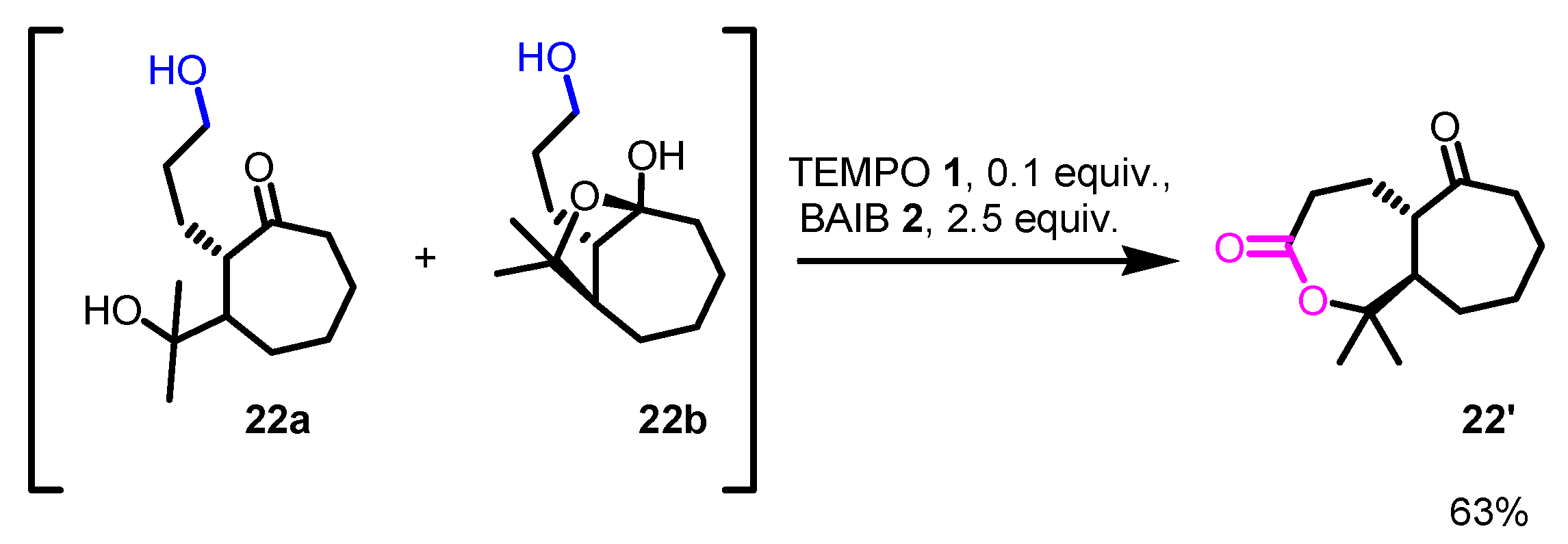
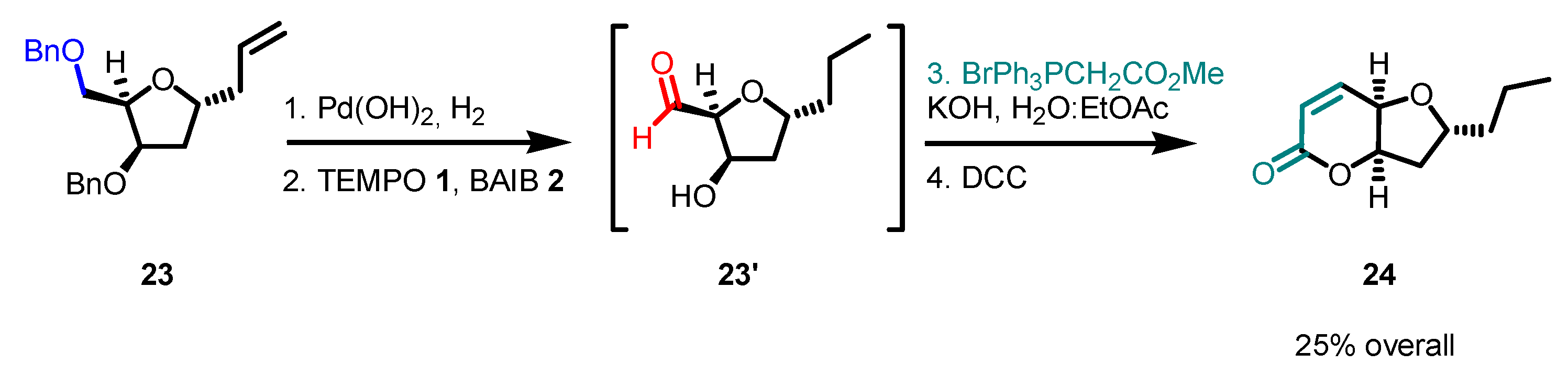
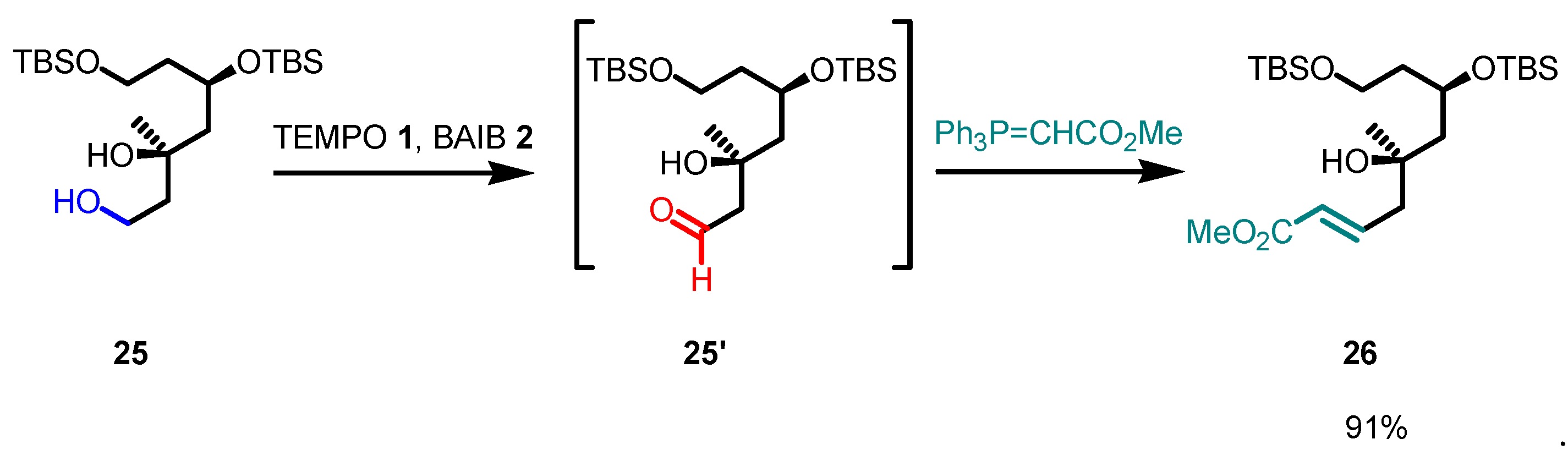

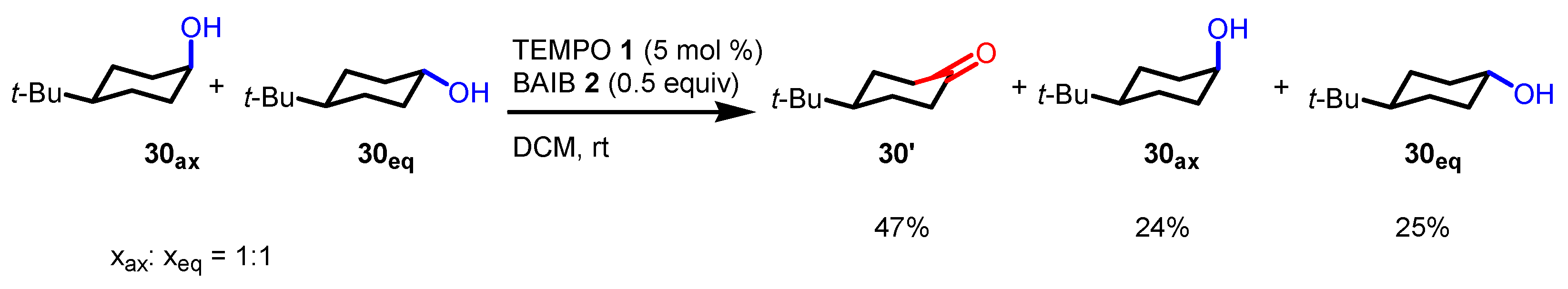
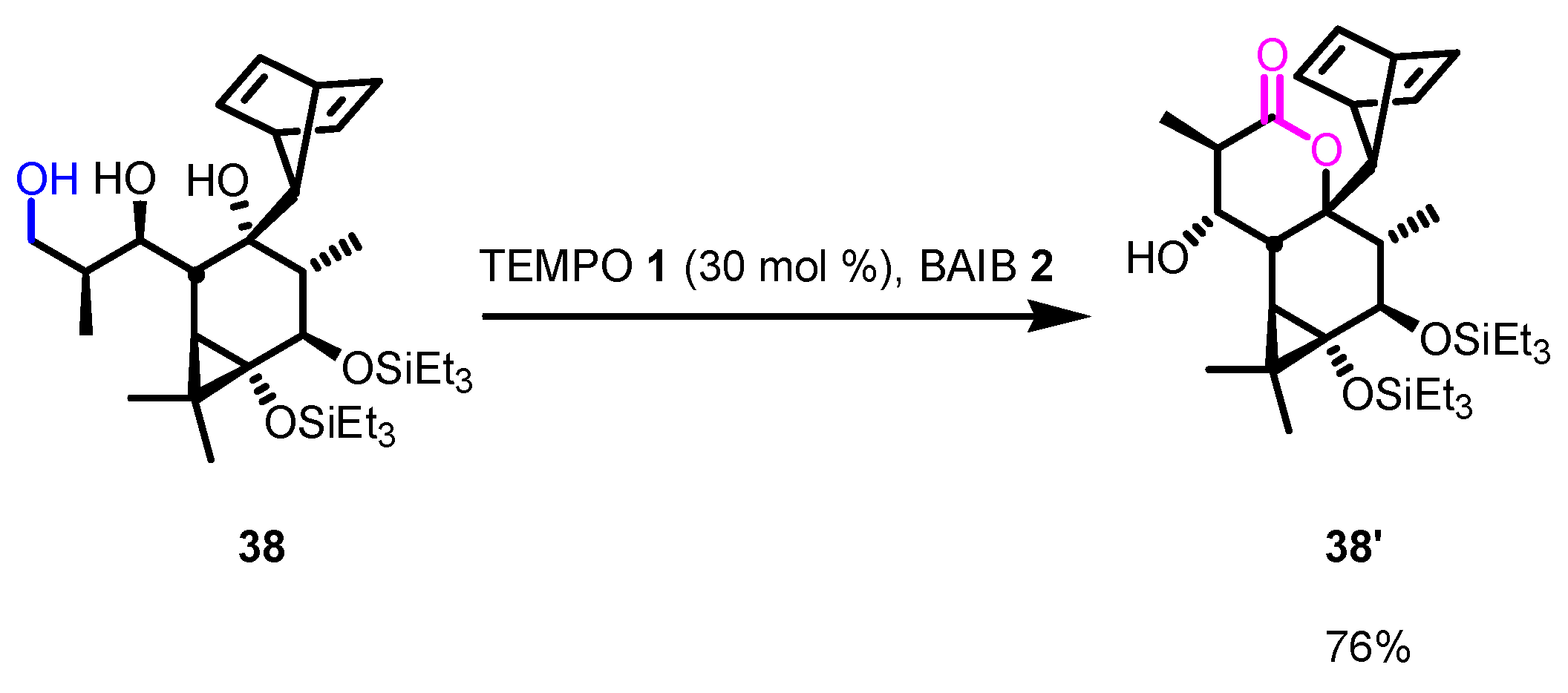
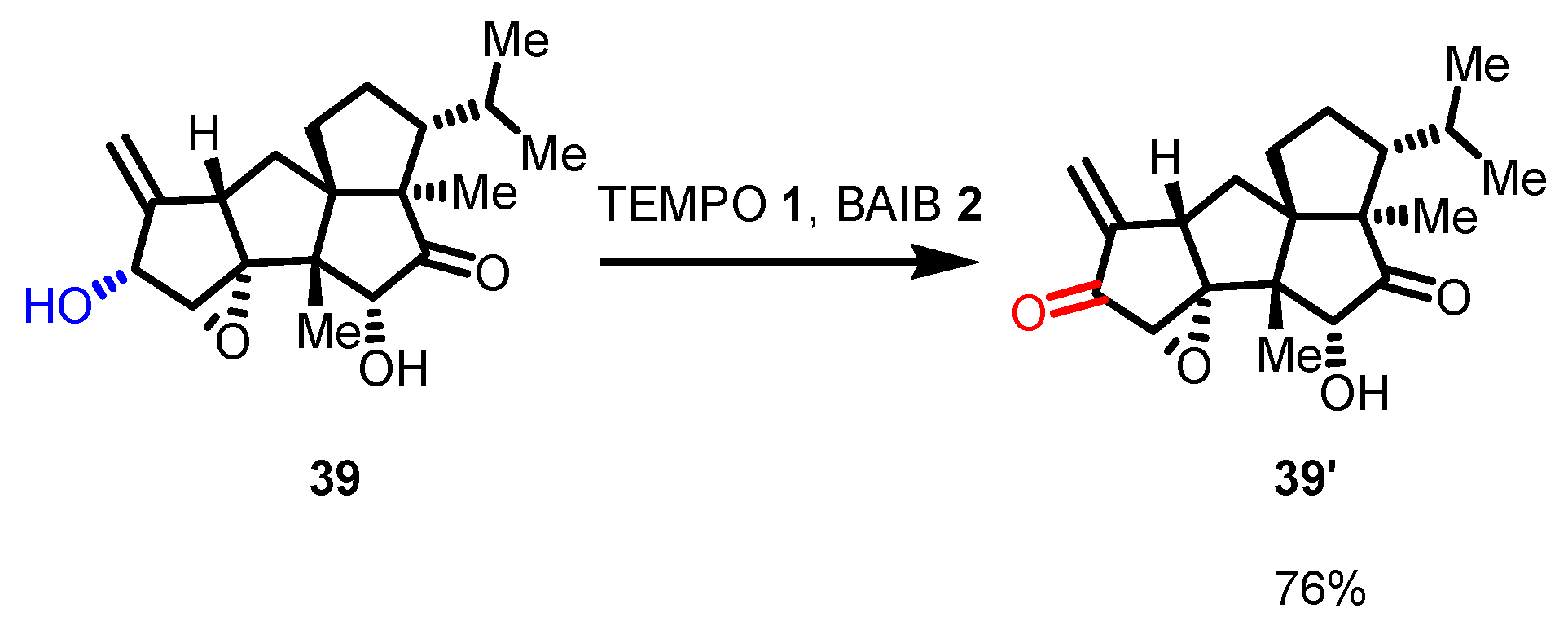

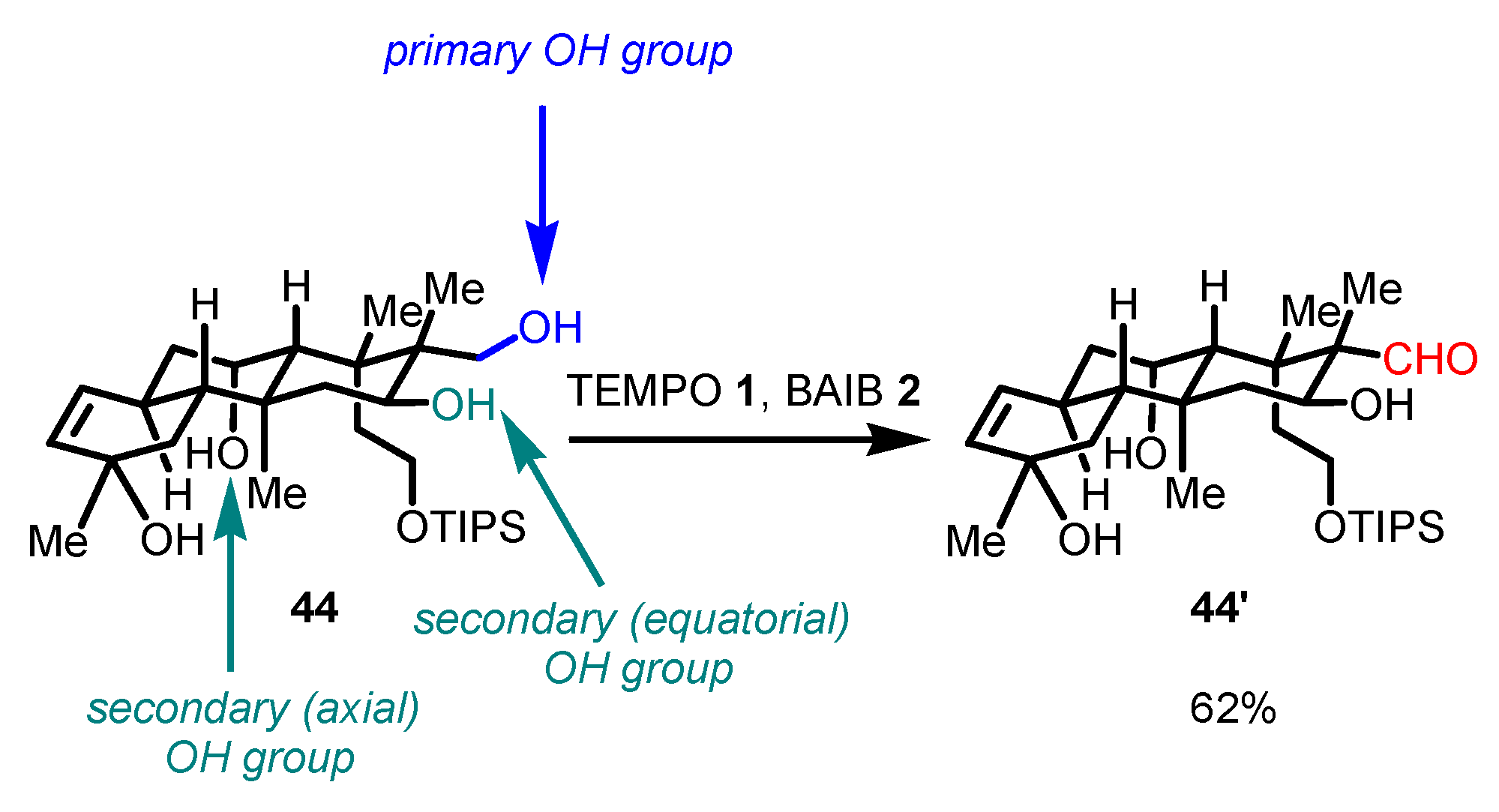

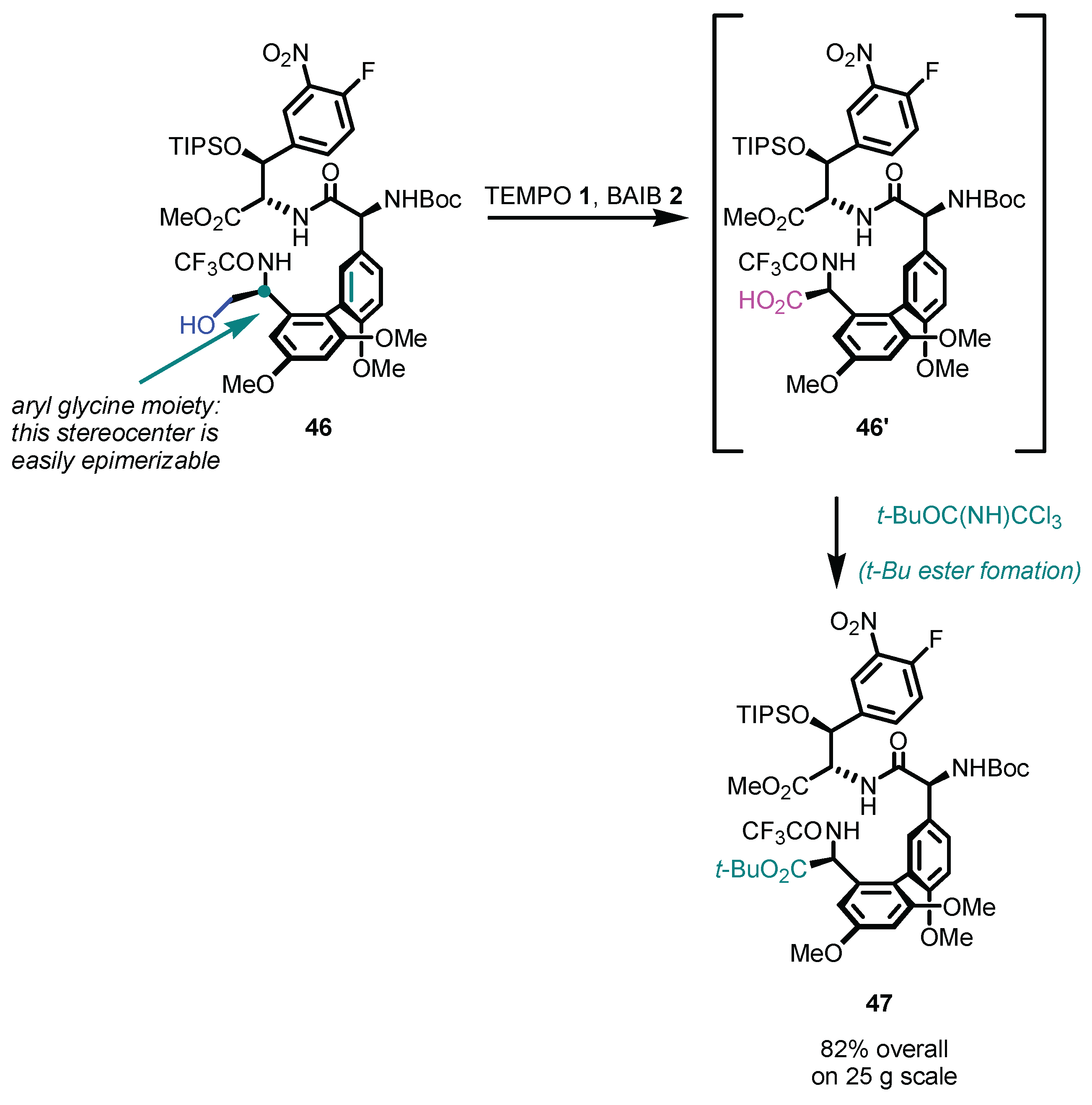
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



