Submitted:
06 June 2025
Posted:
06 June 2025
You are already at the latest version
Abstract
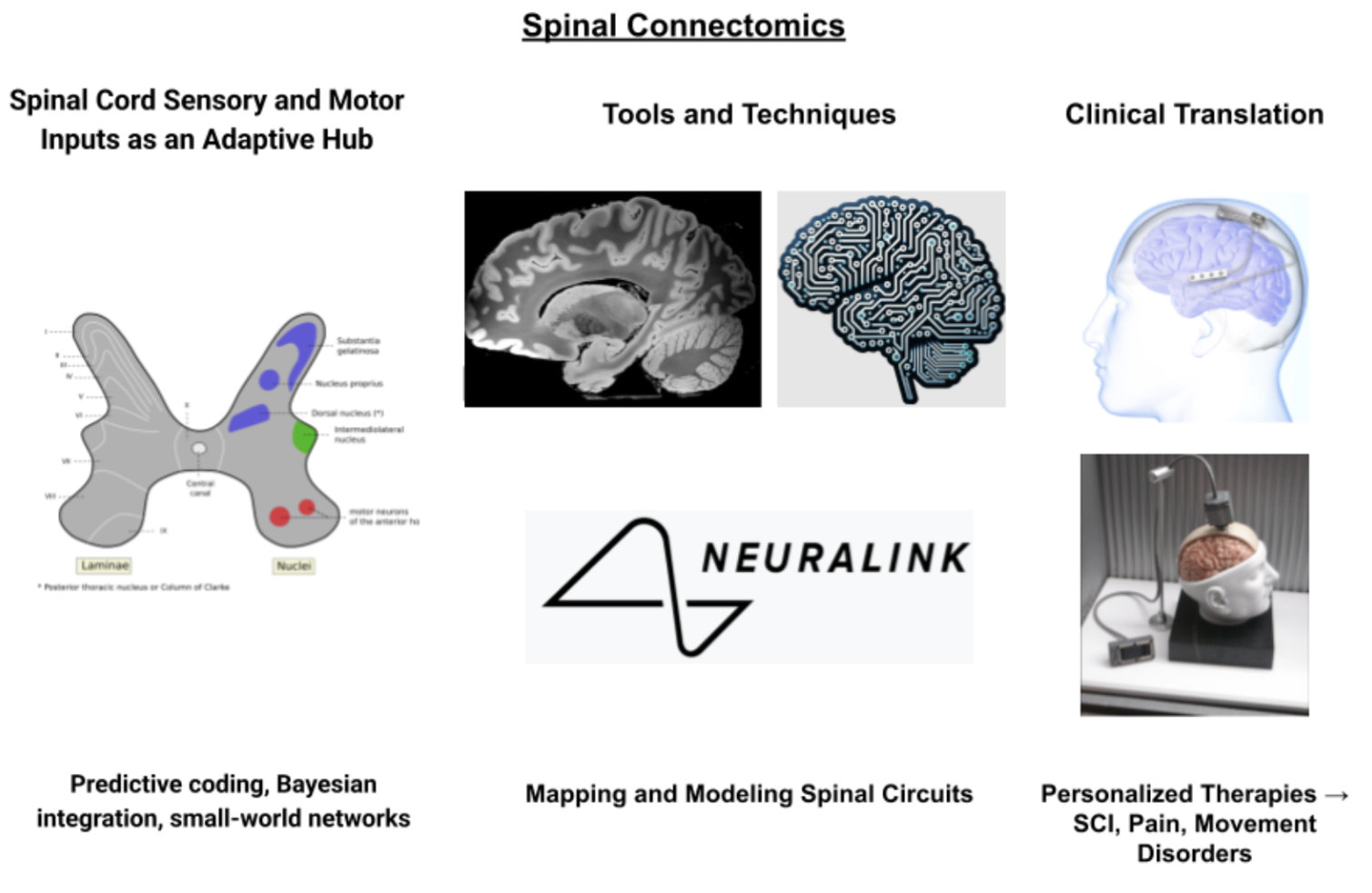
Keywords:
1. Introduction
2. Spinal Connectomics's Historical Development
2.1. From Reflex Arcs Through Computational Networks
2.2. Integration of Downstream Control Systems
2.3. Emergence of Computational Models
3. Neuroanatological Basis of Spinal Connectomics
3.1. Microcircuit Architectonics and Laminar Organization
3.2. Intersegmental Integration and Propriospinal Networks
4. Computational Systems for Spinal Processing
4.1. Error Calculation and Predictive Coding
4.2. Bayesian Uncertainty Quantification and Integration
4.3. Context Modulation and Adaptive Gain Control
4.4. Synchronization and Network Dynamics
5. Advances in Methodology in Spinal Connectomics
5.1. High Density Electrophysiology and Laminar Recording
5.2. Advanced Imaging and Connectivity Mapping
5.3. Computational Modeling and Simulation
6. Brain-Machine Interactions and Neuralink Technologies
6.1. Signal Processing and Architectural Design of Neural Interfaces
6.2. Two-Sided Neural Communication
6.3. Therapeutic Uses and Clinical Translation
7. Molecular Mechanisms and Genetic Foundations
7.1. Spinal Circuit Assembly Transcriptual Regulation
7.2. Epigenetic Control and Circuit Plasticity
7.3. Genetic Strategies for Circuit Modification
8. Therapeutic Interventions and Clinical Applications
8.1. Spinal Cord Damage and Neurorehabilitation
8.2. Sensory Problems and Long-Term Pain
8.3. Movement Disorders and Motor Control
9. Future Routes and New Technologies
9.1. Computer Discovery and Artificial Intelligence
9.2. Personalized Therapy and Precision Therapeutics
9.3. Second-Generation Neural Interfaces
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Courtine, G.; Sofroniew, M. V. Spinal cord repair: advances in biology and technology. Nat. Med. 2019, 25, 898–908. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, L. R.; Bacher, D.; Jarosiewicz, B. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature 2012, 485, 372–375. [Google Scholar] [CrossRef] [PubMed]
- Greicius, M. D.; Krasnow, B.; Reiss, A. L. Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc. Natl. Acad. Sci. U. S. A. 2003, 100, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Friston, K. J.; Shiner, T.; FitzGerald, T. Dopamine, affordance and active inference. PLoS Comput. Biol. 2012, 8, e1002327. [Google Scholar] [CrossRef] [PubMed]
- Clark, A. Whatever next? Predictive brains, situated agents, and the future of cognitive science. Behav. Brain Sci. 2013, 36, 181–204. [Google Scholar] [CrossRef] [PubMed]
- Angeli, C. A.; Boakye, M.; Morton, R. A. Recovery of over-ground walking after chronic motor complete spinal cord injury. N. Engl. J. Med. 2018, 379, 1244–1250. [Google Scholar] [CrossRef] [PubMed]
- Sherrington, C. S. Reflex inhibition as a factor in the co-ordination of movements and postures. Q. J. Exp. Physiol. 1913, 6, 251–310. [Google Scholar] [CrossRef] [PubMed]
- Grillner, S.; Wallén, P. Central pattern generators for locomotion, with special reference to vertebrates. Annu. Rev. Neurosci. 1985, 8, 233–261. [Google Scholar] [CrossRef] [PubMed]
- Brown, T. G. The intrinsic factors in the act of progression in the mammal. Proc. R. Soc. Lond. B 1911, 84, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Eccles, J. C.; Fatt, P.; Koketsu, K. Cholinergic and inhibitory synapses in a pathway from motor-axon collaterals to motoneurones. J. Physiol. 1954, 126, 524–562. [Google Scholar] [CrossRef] [PubMed]
- Bizzi, E.; Cheung, V. C.; d'Avella, A. Combining modules for movement. Brain Res. Rev. 2008, 57, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, E.; Roberts, W. J. Synaptic actions of single interneurones mediating reciprocal Ia inhibition of motoneurones. J. Physiol. 1972, 222, 623–642. [Google Scholar] [CrossRef] [PubMed]
- Stein, P. S.; Smith, J. L. Neural and biomechanical control strategies for different forms of vertebrate hindlimb locomotion. Adv. Neurol. 1997, 74, 61–73. [Google Scholar] [PubMed]
- Rossignol, S.; Dubuc, R.; Gossard, J. P. Dynamic sensorimotor interactions in locomotion. Physiol. Rev. 2006, 86, 89–154. [Google Scholar] [CrossRef] [PubMed]
- Flynn, J. R.; Graham, B. A.; Galea, M. P. The role of propriospinal neuronal networks in locomotor recovery after spinal cord injury. Neuroscience 2011, 192, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Alstermark, B.; Isa, T. Circuits for skilled reaching and grasping. Annu. Rev. Neurosci. 2012, 35, 559–578. [Google Scholar] [CrossRef] [PubMed]
- Häring, M.; Zeisel, A.; Hochgerner, H. Neuronal atlas of the dorsal horn defines its architecture and links sensory input to transcriptional cell types. Nat. Neurosci. 2018, 21, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Jessell, T. M. Neuronal specification in the spinal cord: inductive signals and transcriptional codes. Nat. Rev. Genet. 2000, 1, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Sporns, O.; Tononi, G.; Kötter, R. The human connectome: a structural description of the human brain. PLoS Comput. Biol. 2005, 1, e42. [Google Scholar] [CrossRef] [PubMed]
- Lemon, R. N. Descending pathways in motor control. Annu. Rev. Neurosci. 2008, 31, 195–218. [Google Scholar] [CrossRef] [PubMed]
- Kuypers, H. G.; Martin, G. F. Descending pathways to the spinal cord. Prog. Brain Res. 1982, 57, 1–419. [Google Scholar] [CrossRef] [PubMed]
- Drew, T.; Prentice, S.; Schepens, B. Cortical and brainstem control of locomotion. Prog. Brain Res. 2004, 143, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Jordan, L. M.; Liu, J.; Hedlund, P. B. New developments in the control of locomotion: modulation of spinal circuits by supraspinal systems. Prog. Brain Res. 2008, 171, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Adams, R. A.; Shipp, S.; Friston, K. J. Predictions not commands: active inference in the motor system. Brain Struct. Funct. 2013, 218, 611–643. [Google Scholar] [CrossRef] [PubMed]
- Wolpert, D. M.; Flanagan, J. R. Motor prediction. Curr. Biol. 2001, 11, R729–R732. [Google Scholar] [CrossRef] [PubMed]
- Shadmehr, R.; Krakauer, J. W. A computational neuroanatomy for motor control. Exp. Brain Res. 2008, 185, 359–381. [Google Scholar] [CrossRef] [PubMed]
- Schoffelen, J. M.; Oostenveld, R.; Fries, P. Neuronal coherence as a mechanism of effective corticospinal interaction. Science 2005, 308, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Kilavik, B. E.; Zaepffel, M.; Brovelli, A. The ups and downs of beta oscillations in sensorimotor cortex. Exp. Neurol. 2013, 245, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Heckman, C. J.; Lee, R. H.; Brownstone, R. M. Hyperexcitable dendrites in motoneurons and their neuromodulatory control during motor behavior. Trends Neurosci. 2003, 26, 688–695. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, B. L.; Fornal, C. A. Serotonin and motor activity. Curr. Opin. Neurobiol. 1997, 7, 820–825. [Google Scholar] [CrossRef] [PubMed]
- Hultborn, H.; Nielsen, J. B. Spinal control of locomotion—from cat to man. Acta Physiol. 2007, 189, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Viemari, J. C.; Ramirez, J. M. Norepinephrine differentially modulates different types of respiratory pacemaker neurons. J. Neurosci. 2006, 26, 9280–9289. [Google Scholar] [CrossRef] [PubMed]
- Ijspeert, A. J. Central pattern generators for locomotion control in animals and robots: a review. Neural Netw. 2008, 21, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Getting, P. A. Emerging principles governing the operation of neural networks. Annu. Rev. Neurosci. 1989, 12, 185–204. [Google Scholar] [CrossRef] [PubMed]
- Rybak, I. A.; Shevtsova, N. A.; Lafreniere-Roula, M. Modelling spinal circuitry involved in locomotor pattern generation: insights from deletions during fictive locomotion. J. Physiol. 2006, 577, 617–639. [Google Scholar] [CrossRef] [PubMed]
- Eliasmith, C.; Anderson, C. H. Neural engineering: computation, representation, and dynamics in neurobiological systems. IEEE Trans. Neural Netw. 2004, 15, 528–529. [Google Scholar] [CrossRef] [PubMed]
- Sussillo, D.; Abbott, L. F. Generating coherent patterns of activity from chaotic neural networks. Neuron 2009, 63, 544–557. [Google Scholar] [CrossRef] [PubMed]
- Todorov, E.; Jordan, M. I. Optimal feedback control as a theory of motor coordination. Nat. Neurosci. 2002, 5, 1226–1235. [Google Scholar] [CrossRef] [PubMed]
- Körding, K. P.; Wolpert, D. M. Bayesian integration in sensorimotor learning. Nature 2004, 427, 244–247. [Google Scholar] [CrossRef] [PubMed]
- Franklin, D. W.; Wolpert, D. M. Computational mechanisms of sensorimotor control. Neuron 2011, 72, 425–442. [Google Scholar] [CrossRef] [PubMed]
- Dayan, P.; Daw, N. D. Decision theory, reinforcement learning, and the brain. Cogn. Affect. Behav. Neurosci. 2008, 8, 429–453. [Google Scholar] [CrossRef] [PubMed]
- Bullmore, E.; Sporns, O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 2009, 10, 186–198. [Google Scholar] [CrossRef] [PubMed]
- Watts, D. J.; Strogatz, S. H. Collective dynamics of 'small-world' networks. Nature 1998, 393, 440–442. [Google Scholar] [CrossRef] [PubMed]
- Meunier, D.; Lambiotte, R.; Bullmore, E. T. Modular and hierarchically modular organization of brain networks. Front. Neurosci. 2010, 4, 200. [Google Scholar] [CrossRef] [PubMed]
- Rubinov, M.; Sporns, O. Complex network measures of brain connectivity: uses and interpretations. Neuroimage 2010, 52, 1059–1069. [Google Scholar] [CrossRef] [PubMed]
- Rexed, B. The cytoarchitectonic organization of the spinal cord in the cat. J. Comp. Neurol. 1952, 96, 414–495. [Google Scholar] [CrossRef] [PubMed]
- Todd, A. J. Neuronal circuitry for pain processing in the dorsal horn. Nat. Rev. Neurosci. 2010, 11, 823–836. [Google Scholar] [CrossRef] [PubMed]
- Craig, A. D. Pain mechanisms: labeled lines versus convergence in central processing. Annu. Rev. Neurosci. 2003, 26, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Prescott, S. A.; De Koninck, Y. Integration of developing and adult pain pathways. Prog. Neurobiol. 2009, 88, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Willis, W. D.; Westlund, K. N. Neuroanatomy of the pain system and of the pathways that modulate pain. J. Clin. Neurophysiol. 1997, 14, 2–31. [Google Scholar] [CrossRef] [PubMed]
- Brown, A. G. Organization in the spinal cord: the anatomy and physiology of identified neurones. Trends Neurosci. 1982, 5, 188–191. [Google Scholar] [CrossRef] [PubMed]
- Le Bars, D.; Dickenson, A. H.; Besson, J. M. Diffuse noxious inhibitory controls (DNIC). I. Effects on dorsal horn convergent neurones in the rat. Pain 1979, 6, 283–304. [Google Scholar] [CrossRef] [PubMed]
- Baldissera, F.; Hultborn, H.; Illert, M. Integration in spinal neuronal systems. Handb. Physiol. 1981, 2, 509–595. [Google Scholar] [CrossRef] [PubMed]
- Goulding, M. Circuits controlling vertebrate locomotion: moving in a new direction. Nat. Rev. Neurosci. 2009, 10, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Kiehn, O. Locomotor circuits in the mammalian spinal cord. Annu. Rev. Neurosci. 2006, 29, 279–306. [Google Scholar] [CrossRef] [PubMed]
- Arber, S. Motor circuits in action: specification, connectivity, and function. Neuron 2012, 74, 975–989. [Google Scholar] [CrossRef] [PubMed]
- Juvin, L.; Simmers, J.; Morin, D. Propriospinal circuitry underlying interlimb coordination in mammalian quadrupedal locomotion. J. Neurosci. 2005, 25, 6025–6035. [Google Scholar] [CrossRef] [PubMed]
- Cowley, K. C.; Schmidt, B. J. Regional distribution of the locomotor pattern-generating network in the adult spinal cord. J. Neurophysiol. 1997, 77, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Reed, W. R.; Shum-Siu, A.; Magnuson, D. S. Reticulospinal pathways in the ventrolateral funiculus with terminations in the cervical and lumbar enlargements of the adult rat spinal cord. Neuroscience 2008, 151, 505–517. [Google Scholar] [CrossRef] [PubMed]
- Dietz, V. Proprioception and locomotor disorders. Nat. Rev. Neurosci. 2002, 3, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Pierrot-Deseilligny, E.; Burke, D. The circuitry of the human spinal cord: its role in motor control and movement disorders. Clin. Neurophysiol. 2005, 116, 2243–2244. [Google Scholar] [CrossRef] [PubMed]
- Deliagina, T. G.; Beloozerova, I. N.; Zelenin, P. V. Supraspinal and propriospinal control of posture and locomotion in quadrupeds. Neuroscience 2008, 153, 1047–1064. [Google Scholar] [CrossRef] [PubMed]
- McCrea, D. A.; Rybak, I. A. Organization of mammalian locomotor rhythm and pattern generation. Brain Res. Rev. 2008, 57, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Bareyre, F. M.; Kerschensteiner, M.; Raineteau, O. The injured spinal cord spontaneously forms a new intraspinal circuit in adult rats. Nat. Neurosci. 2004, 7, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Raineteau, O.; Schwab, M. E. Plasticity of motor systems after incomplete spinal cord injury. Nat. Rev. Neurosci. 2001, 2, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Fouad, K.; Tetzlaff, W. Rehabilitative training and plasticity following spinal cord injury. Exp. Neurol. 2012, 235, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Levine, A. J.; Hinckley, C. A.; Hilde, K. L. Identification of a cellular node for motor control pathways. Nat. Neurosci. 2014, 17, 586–593. [Google Scholar] [CrossRef] [PubMed]
- Tripodi, M.; Stepien, A. E.; Arber, S. Motor antagonism exposed by spatial segregation and timing of neurogenesis. Nature 2011, 479, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Basbaum, A. I.; Bautista, D. M.; Scherrer, G. Cellular and molecular mechanisms of pain. Cell 2009, 139, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Wall, P. D.; Woolf, C. J. Muscle but not cutaneous C-afferent input produces prolonged increases in the excitability of the flexion reflex in the rat. J. Physiol. 1984, 356, 443–458. [Google Scholar] [CrossRef] [PubMed]
- Marder, E.; Bucher, D. Central pattern generators and the control of rhythmic movements. Curr. Biol. 2001, 11, R986–R996. [Google Scholar] [CrossRef] [PubMed]
- Brownstone, R. M.; Wilson, J. M. Strategies for delineating spinal locomotor rhythm-generating networks and the possible role of Hb9 interneurones in rhythmogenesis. Brain Res. Rev. 2008, 57, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Marder, E.; Calabrese, R. L. Principles of rhythmic motor pattern generation. Physiol. Rev. 1996, 76, 687–717. [Google Scholar] [CrossRef] [PubMed]
- Grillner, S.; Jessell, T. M. Measured motion: searching for simplicity in spinal locomotor networks. Curr. Opin. Neurobiol. 2009, 19, 572–586. [Google Scholar] [CrossRef] [PubMed]
- Abraira, V. E.; Ginty, D. D. The sensory neurons of touch. Neuron 2013, 79, 618–639. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Wickramasinghe, S. R.; Savitt, J. M. A hierarchical NGF signaling cascade controls Ret-dependent and Ret-independent events during development of nonpeptidergic DRG neurons. Neuron 2007, 54, 739–754. [Google Scholar] [CrossRef] [PubMed]
- Melzack, R.; Wall, P. D. Pain mechanisms: a new theory. Science 1965, 150, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Perl, E. R. Ideas about pain, a historical view. Nat. Rev. Neurosci. 2007, 8, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Rao, R. P.; Ballard, D. H. Predictive coding in the visual cortex: a functional interpretation of some extra-classical receptive-field effects. Nat. Neurosci. 1999, 2, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Bastos, A. M.; Usrey, W. M.; Adams, R. A. Canonical microcircuits for predictive coding. Neuron 2012, 76, 695–711. [Google Scholar] [CrossRef] [PubMed]
- Spratling, M. W. A review of predictive coding algorithms. Brain Cogn. 2017, 112, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Egner, T.; Monti, J. M.; Summerfield, C. Expectation and surprise determine neural population responses in the ventral visual stream. J. Neurosci. 2010, 30, 16601–16608. [Google Scholar] [CrossRef] [PubMed]
- Fries, P. A mechanism for cognitive dynamics: neuronal communication through neuronal coherence. Trends Cogn. Sci. 2005, 9, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Engel, A. K.; Fries, P.; Singer, W. Dynamic predictions: oscillations and synchrony in top-down processing. Nat. Rev. Neurosci. 2001, 2, 704–716. [Google Scholar] [CrossRef] [PubMed]
- Buzsáki, G.; Draguhn, A. Neuronal oscillations in cortical networks. Science 2004, 304, 1926–1929. [Google Scholar] [CrossRef] [PubMed]
- von Stein, A.; Sarnthein, J. Different frequencies for different scales of cortical integration: from local gamma to long range alpha/theta synchronization. Int. J. Psychophysiol. 2000, 38, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Denève, S.; Machens, C. K. Efficient codes and balanced networks. Nat. Neurosci. 2016, 19, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Keller, G. B.; Mrsic-Flogel, T. D. Predictive processing: a canonical cortical computation. Neuron 2018, 100, 424–435. [Google Scholar] [CrossRef] [PubMed]
- Shipp, S. Neural elements for predictive coding. Front. Psychol. 2016, 7, 1792. [Google Scholar] [CrossRef] [PubMed]
- Barrett, L. F.; Simmons, W. K. Interoceptive predictions in the brain. Nat. Rev. Neurosci. 2015, 16, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Ernst, M. O.; Banks, M. S. Humans integrate visual and haptic information in a statistically optimal fashion. Nature 2002, 415, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Angelaki, D. E.; Gu, Y.; DeAngelis, G. C. Multisensory integration: psychophysics, neurophysiology, and computation. Curr. Opin. Neurobiol. 2009, 19, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Fetsch, C. R.; DeAngelis, G. C.; Angelaki, D. E. Bridging the gap between theories of sensory cue integration and the physiology of multisensory neurons. Nat. Rev. Neurosci. 2013, 14, 429–442. [Google Scholar] [CrossRef] [PubMed]
- Parise, C. V.; Ernst, M. O. Correlation detection as a general mechanism for multisensory integration. Nat. Commun. 2016, 7, 11543. [Google Scholar] [CrossRef] [PubMed]
- Stein, B. E.; Stanford, T. R. Multisensory integration: current issues from the perspective of the single neuron. Nat. Rev. Neurosci. 2008, 9, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Knill, D. C.; Pouget, A. The Bayesian brain: the role of uncertainty in neural coding and computation. Trends Neurosci. 2004, 27, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Ma, W. J.; Beck, J. M.; Latham, P. E. Optimal integration of sensory evidence: theory and neural implications. Neuron 2006, 51, 539–541. [Google Scholar] [CrossRef] [PubMed]
- Körding, K. P.; Tenenbaum, J. B.; Shadmehr, R. The dynamics of memory as a consequence of optimal adaptation to a changing body. Nat. Neurosci. 2007, 10, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Bays, P. M.; Wolpert, D. M. Computational principles of sensorimotor control that minimize uncertainty and variability. J. Physiol. 2007, 578, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Harris, C. M.; Wolpert, D. M. Signal-dependent noise determines motor planning. Nature 1998, 394, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Rudomin, P.; Schmidt, R. F. Presynaptic inhibition in the vertebrate spinal cord revisited. Exp. Brain Res. 1999, 129, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Sillar, K. T.; Picton, L. D.; Heitler, W. J. The neuroethology of predation and escape. J. Exp. Biol. 2016, 219, 310–311. [Google Scholar] [CrossRef] [PubMed]
- Clarac, F.; Cattaert, D.; Le Ray, D. Central control components of a 'simple' stretch reflex. Trends Neurosci. 2000, 23, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Nusbaum, M. P.; Blitz, D. M.; Marder, E. Functional consequences of neuropeptide and small-molecule co-transmission. Nat. Rev. Neurosci. 2017, 18, 389–403. [Google Scholar] [CrossRef] [PubMed]
- Miles, G. B.; Sillar, K. T. Neuromodulation of vertebrate locomotor control networks. Physiology 2011, 26, 393–411. [Google Scholar] [CrossRef] [PubMed]
- Sharples, S. A.; Koblinger, K.; Humphreys, J. M. Dopamine: an unexpected neuromodulator of spinal circuits. Front. Neural Circuits 2014, 8, 116. [Google Scholar] [CrossRef] [PubMed]
- Perrier, J. F.; Cotel, F. Serotonergic modulation of spinal motor control. Curr. Opin. Neurobiol. 2015, 33, 1–7. [Google Scholar] [CrossRef] [PubMed]
- El Manira, A.; Kyriakatos, A.; Nanou, E. Neuromodulation of spinal networks: from synapses to behavior. Curr. Opin. Physiol. 2019, 8, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Abbott, L. F.; Regehr, W. G. Synaptic computation. Nature 2004, 431, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Turrigiano, G. G.; Nelson, S. B. Homeostatic plasticity in the developing nervous system. Nat. Rev. Neurosci. 2004, 5, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Yuste, R.; MacLean, J. N.; Smith, J. The cortex as a central pattern generator. Nat. Rev. Neurosci. 2005, 6, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Schnitzler, A.; Gross, J. Normal and pathological oscillatory communication in the brain. Nat. Rev. Neurosci. 2005, 6, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Uhlhaas, P. J.; Singer, W. Neural synchrony in brain disorders: relevance for cognitive dysfunctions and pathophysiology. Neuron 2006, 52, 155–168. [Google Scholar] [CrossRef] [PubMed]
- Kopell, N.; Ermentrout, G. B.; Whittington, M. A. Gamma rhythms and beta rhythms have different synchronization properties. Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 1867–1872. [Google Scholar] [CrossRef] [PubMed]
- Connors, B. W.; Long, M. A. Electrical synapses in the mammalian brain. Annu. Rev. Neurosci. 2004, 27, 393–418. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M. V.; Zukin, R. S. Electrical coupling and neuronal synchronization in the mammalian brain. Neuron 2004, 41, 495–511. [Google Scholar] [CrossRef] [PubMed]
- Shew, W. L.; Yang, H.; Yu, S. Information capacity and transmission are maximized in balanced cortical networks with neuronal avalanches. J. Neurosci. 2011, 31, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Beggs, J. M.; Plenz, D. Neuronal avalanches in neocortical circuits. J. Neurosci. 2003, 23, 11167–11177. [Google Scholar] [CrossRef] [PubMed]
- Chialvo, D. R. Emergent complex neural dynamics. Nat. Phys. 2010, 6, 744–750. [Google Scholar] [CrossRef] [PubMed]
- Hesse, J.; Gross, T. Self-organized criticality as a fundamental property of neural systems. Front. Syst. Neurosci. 2014, 8, 166. [Google Scholar] [CrossRef] [PubMed]
- Kinouchi, O.; Copelli, M. Optimal dynamical range of excitable networks at criticality. Nat. Phys. 2006, 2, 348–351. [Google Scholar] [CrossRef] [PubMed]
- Jun, J. J.; Steinmetz, N. A.; Siegle, J. H. Fully integrated silicon probes for high-density recording of neural activity. Nature 2017, 551, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Steinmetz, N. A.; Aydin, C.; Lebedeva, A. Neuropixels 2. 0: A miniaturized high-density probe for stable, long-term brain recordings. Science 2021, 372, eabf4588. [Google Scholar] [CrossRef] [PubMed]
- Pachitariu, M.; Steinmetz, N. A.; Kadir, S. N. Fast and accurate spike sorting of high-channel count probes with KiloSort. Adv. Neural Inf. Process. Syst. 2016, 29, 4448–4456. [Google Scholar] [PubMed]
- Einevoll, G. T.; Franke, F.; Hagen, E. Local field potential: biophysical origin and analysis. J. Neurophysiol. 2012, 108, 2539–2563. [Google Scholar] [CrossRef] [PubMed]
- Buzsáki, G.; Anastassiou, C. A.; Koch, C. The origin of extracellular fields and currents—EEG, ECoG, LFP and spikes. Nat. Rev. Neurosci. 2012, 13, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Friston, K. J.; Harrison, L.; Penny, W. Dynamic causal modelling. Neuroimage 2003, 19, 1273–1302. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, T. Measuring information transfer. Phys. Rev. Lett. 2000, 85, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Kriegeskorte, N.; Douglas, P. K. Cognitive computational neuroscience. Nat. Neurosci. 2018, 21, 1148–1160. [Google Scholar] [CrossRef] [PubMed]
- Boyden, E. S.; Zhang, F.; Bamberg, E. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 2005, 8, 1263–1268. [Google Scholar] [CrossRef] [PubMed]
- Deisseroth, K. Optogenetics: 10 years of microbial opsins in neuroscience. Nat. Neurosci. 2015, 18, 1213–1225. [Google Scholar] [CrossRef] [PubMed]
- Histed, M. H.; Bonin, V.; Reid, R. C. Direct activation of sparse, distributed populations of cortical neurons by electrical microstimulation. Neuron 2009, 63, 508–522. [Google Scholar] [CrossRef] [PubMed]
- Krook-Magnuson, E.; Armstrong, C.; Oijala, M. On-demand optogenetic control of spontaneous seizures in temporal lobe epilepsy. Nat. Commun. 2013, 4, 1376. [Google Scholar] [CrossRef] [PubMed]
- Helmchen, F.; Denk, W. Deep tissue two-photon microscopy. Nat. Methods 2005, 2, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Grienberger, C.; Konnerth, A. Imaging calcium in neurons. Neuron 2012, 73, 862–885. [Google Scholar] [CrossRef] [PubMed]
- Chen, T. W.; Wardill, T. J.; Sun, Y. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 2013, 499, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Dana, H.; Chen, T. W.; Hu, A. Thy1-GCaMP6 transgenic mice for neuronal population imaging in vivo. PLoS One 2014, 9, e108697. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Tillberg, P. W.; Boyden, E. S. Optical imaging. Expansion microscopy. Science 2015, 347, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Wassie, A. T.; Zhao, Y.; Boyden, E. S. Expansion microscopy: principles and uses in biological research. Nat. Methods 2019, 16, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Lichtman, J. W.; Denk, W. The big and the small: challenges of imaging the brain’s circuits. Science 2011, 334, 618–623. [Google Scholar] [CrossRef] [PubMed]
- Morgan, J. L.; Lichtman, J. W. Why not connectomics? Nat. Methods 2013, 10, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Wickersham, I. R.; Lyon, D. C.; Barnard, R. J. Monosynaptic restriction of transsynaptic tracing from single, genetically targeted neurons. Neuron 2007, 53, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Callaway, E. M.; Luo, L. Monosynaptic circuit tracing with glycoprotein-deleted rabies viruses. J. Neurosci. 2015, 35, 8979–8985. [Google Scholar] [CrossRef] [PubMed]
- Fenno, L. E.; Ramakrishnan, C.; Kim, Y. S. Comprehensive dual- and triple-feature intersectional single-vector delivery of combinatorial therapeutics. Nat. Methods 2020, 17, 789–793. [Google Scholar] [CrossRef] [PubMed]
- Osakada, F.; Callaway, E. M. Design and generation of recombinant rabies virus vectors. Nat. Protoc. 2013, 8, 1583–1601. [Google Scholar] [CrossRef] [PubMed]
- Markram, H.; Muller, E.; Ramaswamy, S. Reconstruction and simulation of neocortical microcircuitry. Cell 2015, 163, 456–492. [Google Scholar] [CrossRef] [PubMed]
- Druckmann, S.; Banitt, Y.; Gidon, A. A novel multiple objective optimization framework for constraining conductance-based neuron models by experimental data. Front. Neurosci. 2007, 1, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Destexhe, A.; Mainen, Z. F.; Sejnowski, T. J. Synthesis of models for excitable membranes, synaptic transmission and neuromodulation using a common kinetic formalism. J. Comput. Neurosci. 1994, 1, 195–230. [Google Scholar] [CrossRef] [PubMed]
- Lytton, W. W.; Sejnowski, T. J. Simulations of cortical pyramidal neurons synchronized by inhibitory interneurons. J. Neurophysiol. 1991, 66, 1059–1079. [Google Scholar] [CrossRef] [PubMed]
- Glaser, J. I.; Benjamin, A. S.; Farhoodi, R. The roles of supervised machine learning in systems neuroscience. Prog. Neurobiol. 2019, 175, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Yamins, D. L.; DiCarlo, J. J. Using goal-driven deep learning models to understand sensory cortex. Nat. Neurosci. 2016, 19, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Kriegeskorte, N. Deep neural networks: a new framework for modeling biological vision and brain information processing. Annu. Rev. Vis. Sci. 2015, 1, 417–446. [Google Scholar] [CrossRef] [PubMed]
- Scarselli, F.; Gori, M.; Tsoi, A. C. The graph neural network model. IEEE Trans. Neural Netw. 2009, 20, 61–80. [Google Scholar] [CrossRef] [PubMed]
- Evensen, G. The ensemble Kalman filter: theoretical formulation and practical implementation. Ocean Dyn. 2003, 53, 343–367. [Google Scholar] [CrossRef] [PubMed]
- Schiff, S. J.; Sauer, T. Kalman filter control of a model of spatiotemporal cortical dynamics. J. Neural Eng. 2008, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Van Geit, W.; De Schutter, E.; Achard, P. Automated neuron model optimization techniques: a review. Biol. Cybern. 2008, 99, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Huys, Q. J.; Paninski, L. Smoothing of, and parameter estimation from, noisy biophysical recordings. PLoS Comput. Biol. 2009, 5, e1000379. [Google Scholar] [CrossRef] [PubMed]
- Donoghue, J. P. Bridging the brain to the world: a perspective on neural interface systems. Neuron 2008, 60, 511–521. [Google Scholar] [CrossRef] [PubMed]
- Fetz, E. E. Volitional control of neural activity: implications for brain-computer interfaces. J. Physiol. 2007, 579, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Polikov, V. S.; Tresco, P. A.; Reichert, W. M. Response of brain tissue to chronically implanted neural electrodes. J. Neurosci. Methods 2005, 148, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Lebedev, M. A.; Nicolelis, M. A. Brain-machine interfaces: past, present and future. Trends Neurosci. 2006, 29, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Hatsopoulos, N. G.; Donoghue, J. P. The science of neural interface systems. Annu. Rev. Neurosci. 2009, 32, 249–266. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, A. B.; Cui, X. T.; Weber, D. J. Brain-controlled interfaces: movement restoration with neural prosthetics. Neuron 2006, 52, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Kipke, D. R.; Shain, W.; Buzsáki, G. Advanced neurotechnologies for chronic neural interfaces: new horizons and clinical opportunities. J. Neurosci. 2008, 28, 11830–11838. [Google Scholar] [CrossRef] [PubMed]
- Grill, W. M.; Norman, S. E.; Bellamkonda, R. V. Implanted neural interfaces: biochallenges and engineered solutions. Annu. Rev. Biomed. Eng. 2009, 11, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Pandarinath, C.; Nuyujukian, P.; Blabe, C. H. High performance communication by people with paralysis using an intracortical brain-computer interface. Elife 2017, 6, e18554. [Google Scholar] [CrossRef] [PubMed]
- O’Doherty, J. E.; Lebedev, M. A.; Ifft, P. J. Active tactile exploration using a brain-machine-brain interface. Nature 2011, 479, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Ethier, C.; Oby, E. R.; Bauman, M. J. Restoration of grasp following paralysis through brain-controlled stimulation of muscles. Nature 2012, 485, 368–371. [Google Scholar] [CrossRef] [PubMed]
- Moritz, C. T.; Perlmutter, S. I.; Fetz, E. E. Direct control of paralysed muscles by cortical neurons. Nature 2008, 456, 639–642. [Google Scholar] [CrossRef] [PubMed]
- Nicolelis, M. A.; Lebedev, M. A. Principles of neural ensemble physiology underlying the operation of brain-machine interfaces. Nat. Rev. Neurosci. 2009, 10, 530–540. [Google Scholar] [CrossRef] [PubMed]
- Venkatraman, S.; Carmena, J. M. Active sensing of target location encoded by cortical microstimulation. IEEE Trans. Neural Syst. Rehabil. Eng. 2011, 19, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Yizhar, O.; Fenno, L. E.; Davidson, T. J. Optogenetics in neural systems. Neuron 2011, 71, 9–34. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, J. G.; Boyden, E. S. Optogenetic tools for analyzing the neural circuits of behavior. Trends Cogn. Sci. 2011, 15, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Tye, K. M.; Deisseroth, K. Optogenetic investigation of neural circuits underlying brain disease in animal models. Nat. Rev. Neurosci. 2012, 13, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Sohal, V. S.; Zhang, F.; Yizhar, O. Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 2009, 459, 698–702. [Google Scholar] [CrossRef] [PubMed]
- Cardin, J. A.; Carlén, M.; Meletis, K. Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 2009, 459, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Siegle, J. H.; López, A. C.; Patel, Y. A. Open Ephys: an open-source, plugin-based platform for multichannel electrophysiology. J. Neural Eng. 2017, 14, 045003. [Google Scholar] [CrossRef] [PubMed]
- Collinger, J. L.; Wodlinger, B.; Downey, J. E. High-performance neuroprosthetic control by an individual with tetraplegia. Lancet 2013, 381, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Ajiboye, A. B.; Willett, F. R.; Young, D. R. Restoration of reaching and grasping movements through brain-controlled muscle stimulation in a person with tetraplegia: a proof-of-concept demonstration. Lancet 2017, 389, 1821–1830. [Google Scholar] [CrossRef] [PubMed]
- Capogrosso, M.; Milekovic, T.; Borton, D. A brain-spine interface alleviating gait deficits after spinal cord injury in primates. Nature 2016, 539, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Flesher, S. N.; Collinger, J. L.; Foldes, S. T. Intracortical microstimulation of human somatosensory cortex. Sci. Transl. Med. 2016, 8, 361ra141. [Google Scholar] [CrossRef] [PubMed]
- Wagner, F. B.; Mignardot, J. B.; Le Goff-Mignardot, C. G. Targeted neurotechnology restores walking in humans with spinal cord injury. Nature 2018, 563, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Harkema, S.; Gerasimenko, Y.; Hodes, J. Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: a case study. Lancet 2011, 377, 1938–1947. [Google Scholar] [CrossRef] [PubMed]
- Gill, M. L.; Grahn, P. J.; Calvert, J. S. Neuromodulation of lumbosacral spinal networks enables independent stepping after complete paraplegia. Nat. Med. 2018, 24, 1677–1682. [Google Scholar] [CrossRef] [PubMed]
- Rejc, E.; Angeli, C. A.; Atkinson, D. Motor recovery after activity-based training with spinal cord epidural stimulation in a chronic motor complete paraplegic. Sci. Rep. 2017, 7, 13476. [Google Scholar] [CrossRef] [PubMed]
- Tan, D. W.; Schiefer, M. A.; Keith, M. W. A neural interface provides long-term stable natural touch perception. Sci. Transl. Med. 2014, 6, 257ra138. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T. C.; Janik, J. J.; Grill, W. M. Mechanisms and models of spinal cord stimulation for the treatment of neuropathic pain. Brain Res. 2014, 1569, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Catalan, M.; Håkansson, B.; Brånemark, R. An osseointegrated human-machine gateway for long-term sensory feedback and motor control of artificial limbs. Sci. Transl. Med. 2014, 6, 257re6. [Google Scholar] [CrossRef] [PubMed]
- Bensmaia, S. J.; Miller, L. E. Restoring sensorimotor function through intracortical interfaces: progress and looming challenges. Nat. Rev. Neurosci. 2014, 15, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Shanechi, M. M.; Orsborn, A. L.; Carmena, J. M. Robust brain-machine interface design using optimal feedback control modeling and adaptive point process filtering. PLoS Comput. Biol. 2016, 12, e1004730. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, K. V.; Carmena, J. M. Combining decoder design and neural adaptation in brain-machine interfaces. Neuron 2014, 84, 665–680. [Google Scholar] [CrossRef] [PubMed]
- Willett, F. R.; Avansino, D. T.; Hochberg, L. R. High-performance brain-to-text communication via handwriting. Nature 2021, 593, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Little, S.; Pogosyan, A.; Neal, S. Adaptive deep brain stimulation in advanced Parkinson disease. Ann. Neurol. 2013, 74, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Philippidou, P.; Dasen, J. S. Hox genes: choreographers in neural development, architects of circuit organization. Neuron 2013, 80, 12–34. [Google Scholar] [CrossRef] [PubMed]
- Dasen, J. S.; Jessell, T. M. Hox networks and the origins of motor neuron diversity. Curr. Top. Dev. Biol. 2009, 88, 169–200. [Google Scholar] [CrossRef] [PubMed]
- Soshnikova, N.; Duboule, D. Epigenetic regulation of vertebrate Hox genes: a dynamic equilibrium. Epigenetics 2009, 4, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, A.; Hochgerner, H.; Lönnerberg, P. Molecular architecture of the mouse nervous system. Cell 2018, 174, 999–1014. [Google Scholar] [CrossRef] [PubMed]
- Bikoff, J. B.; Gabitto, M. I.; Rivard, A. F. Spinal inhibitory interneuron diversity delineates variant motor microcircuits. Cell 2016, 165, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, L. B.; Bikoff, J. B.; Gabitto, M. I. Origin and segmental diversity of spinal inhibitory interneurons. Neuron 2018, 97, 341–355. [Google Scholar] [CrossRef] [PubMed]
- Delile, J.; Rayon, T.; Melchionda, M. Single cell transcriptomics reveals spatial and temporal dynamics of gene expression in the developing mouse spinal cord. Development 2019, 146, dev173807. [Google Scholar] [CrossRef] [PubMed]
- Flavell, S. W.; Greenberg, M. E. Signaling mechanisms linking neuronal activity to gene expression and plasticity of the nervous system. Annu. Rev. Neurosci. 2008, 31, 563–590. [Google Scholar] [CrossRef] [PubMed]
- West, A. E.; Greenberg, M. E. Neuronal activity-regulated gene transcription in synapse development and cognitive function. Cold Spring Harb. Perspect. Biol. 2011, 3, a005744. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Greenberg, M. E. Communication between the synapse and the nucleus in neuronal development, plasticity, and disease. Annu. Rev. Cell Dev. Biol. 2008, 24, 183–209. [Google Scholar] [CrossRef] [PubMed]
- Yap, E. L.; Greenberg, M. E. Activity-regulated transcription: bridging the gap between neural activity and behavior. Neuron 2018, 100, 330–348. [Google Scholar] [CrossRef] [PubMed]
- Sweatt, J. D. The emerging field of neuroepigenetics. Neuron 2013, 80, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Lister, R.; Mukamel, E. A.; Nery, J. R. Global epigenomic reconfiguration during mammalian brain development. Science 2013, 341, 1237905. [Google Scholar] [CrossRef] [PubMed]
- Day, J. J.; Sweatt, J. D. DNA methylation and memory formation. Nat. Neurosci. 2010, 13, 1319–1323. [Google Scholar] [CrossRef] [PubMed]
- Halder, R.; Hennion, M.; Vidal, R. O. DNA methylation changes in plasticity genes accompany the formation and maintenance of memory. Nat. Neurosci. 2016, 19, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Zovkic, I. B.; Guzman-Karlsson, M. C.; Sweatt, J. D. Epigenetic regulation of memory formation and maintenance. Learn. Mem. 2013, 20, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Strahl, B. D.; Allis, C. D. The language of covalent histone modifications. Nature 2000, 403, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, E.; Nestler, E. J.; Allis, C. D. Decoding the epigenetic language of neuronal plasticity. Neuron 2008, 60, 961–974. [Google Scholar] [CrossRef] [PubMed]
- Palmisano, I.; Danzi, M. C.; Hutson, T. H. Epigenomic signatures underpin the axonal regenerative ability of dorsal root ganglia sensory neurons. Nat. Neurosci. 2019, 22, 1913–1924. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D. P. MicroRNAs: target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Fabian, M. R.; Sonenberg, N.; Filipowicz, W. Regulation of mRNA translation and stability by microRNAs. Annu. Rev. Biochem. 2010, 79, 351–379. [Google Scholar] [CrossRef] [PubMed]
- Krol, J.; Loedige, I.; Filipowicz, W. The widespread regulation of microRNA biogenesis, function and decay. Nat. Rev. Genet. 2010, 11, 597–610. [Google Scholar] [CrossRef] [PubMed]
- Fenno, L.; Yizhar, O.; Deisseroth, K. The development and application of optogenetics. Annu. Rev. Neurosci. 2011, 34, 389–412. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Gradinaru, V.; Adamantidis, A. R. Optogenetic interrogation of neural circuits: technology for probing mammalian brain functions. Nat. Protoc. 2010, 5, 439–456. [Google Scholar] [CrossRef] [PubMed]
- Kim, T. I.; McCall, J. G.; Jung, Y. H. Injectable, cellular-scale optoelectronics with applications for wireless optogenetics. Science 2013, 340, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Häusser, M. Optogenetics: the age of light. Nat. Methods 2014, 11, 1012–1014. [Google Scholar] [CrossRef] [PubMed]
- Roth, B. L. DREADDs for neuroscientists. Neuron 2016, 89, 683–694. [Google Scholar] [CrossRef] [PubMed]
- Armbruster, B. N.; Li, X.; Pausch, M. H. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc. Natl. Acad. Sci. U. S. A. 2007, 104, 5163–5168. [Google Scholar] [CrossRef] [PubMed]
- Alexander, G. M.; Rogan, S. C.; Abbas, A. I. Remote control of neuronal activity in transgenic mice expressing evolved G protein-coupled receptors. Neuron 2009, 63, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Sternson, S. M.; Roth, B. L. Chemogenetic tools to interrogate brain functions. Annu. Rev. Neurosci. 2014, 37, 387–407. [Google Scholar] [CrossRef] [PubMed]
- Cong, L.; Ran, F. A.; Cox, D. Multiplex genome engineering using CRISPR/Cas systems. Science 2013, 339, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Ran, F. A.; Hsu, P. D.; Wright, J. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 2013, 8, 2281–2308. [Google Scholar] [CrossRef] [PubMed]
- Hilton, I. B.; D’Ippolito, A. M.; Vockley, C. M. Epigenome editing by a CRISPR-Cas9-based acetyltransferase activates genes from promoters and enhancers. Nat. Biotechnol. 2015, 33, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Jinek, M.; Chylinski, K.; Fonfara, I. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Edgerton, V. R.; Courtine, G.; Gerasimenko, Y. P. Training locomotor networks. Brain Res. Rev. 2008, 57, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Dietz, V.; Fouad, K. Restoration of sensorimotor functions after spinal cord injury. Brain 2014, 137, 654–667. [Google Scholar] [CrossRef] [PubMed]
- Behrman, A. L.; Bowden, M. G.; Nair, P. M. Neuroplasticity after spinal cord injury and training: an emerging paradigm shift in rehabilitation and walking recovery. Phys. Ther. 2006, 86, 1406–1425. [Google Scholar] [CrossRef] [PubMed]
- Dobkin, B.; Apple, D.; Barbeau, H. Weight-supported treadmill vs over-ground training for walking after acute incomplete SCI. Neurology 2006, 66, 484–493. [Google Scholar] [CrossRef] [PubMed]
- van den Brand, R.; Heutschi, J.; Barraud, Q. Restoring voluntary control of locomotion after paralyzing spinal cord injury. Science 2012, 336, 1182–1185. [Google Scholar] [CrossRef] [PubMed]
- Grahn, P. J.; Lavrov, I. A.; Sayenko, D. G. Enabling task-specific volitional motor functions via spinal cord neuromodulation in a human with paraplegia. Mayo Clin. Proc. 2017, 92, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Formento, E.; Minassian, K.; Wagner, F. Electrical spinal cord stimulation must preserve proprioception to enable locomotion in humans with spinal cord injury. Nat. Neurosci. 2018, 21, 1728–1741. [Google Scholar] [CrossRef] [PubMed]
- Courtine, G.; Micera, S.; DiGiovanna, J. Neuromodulation of lower limb motor control in restorative neurology. Clin. Neurol. Neurosurg. 2012, 114, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Lempka, S. F.; McIntyre, C. C. Theoretical analysis of the local field potential in deep brain stimulation applications. PLoS One 2013, 8, e59839. [Google Scholar] [CrossRef] [PubMed]
- Rosin, B.; Slovik, M.; Mitelman, R. Closed-loop deep brain stimulation is superior in ameliorating parkinsonism. Neuron 2011, 72, 370–384. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.; Fetz, E. E. Compact movable microwire array for long-term chronic unit recording in cerebral cortex of primates. J. Neurophysiol. 2007, 98, 3109–3118. [Google Scholar] [CrossRef] [PubMed]
- Latremoliere, A.; Woolf, C. J. Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J. Pain 2009, 10, 895–926. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C. J.; Salter, M. W. Neuronal plasticity: increasing the gain in pain. Science 2000, 288, 1765–1769. [Google Scholar] [CrossRef] [PubMed]
- Kuner, R. Central mechanisms of pathological pain. Nat. Med. 2010, 16, 1258–1266. [Google Scholar] [CrossRef] [PubMed]
- Melzack, R.; Wall, P. D. Pain mechanisms: a new theory. Science 1965, 150, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T. C.; Janik, J. J.; Grill, W. M. Modeling effects of spinal cord stimulation on wide-dynamic range dorsal horn neurons: influence of stimulation frequency and GABAergic inhibition. J. Neurophysiol. 2014, 112, 552–567. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M. P.; Gertz, K. J.; Kupper, A. E. Steps toward developing an EEG biofeedback treatment for chronic pain. Appl. Psychophysiol. Biofeedback 2013, 38, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Russo, J. F.; Sheth, S. A. Deep brain stimulation of the dorsal anterior cingulate cortex for the treatment of chronic neuropathic pain. Neurosurg. Focus 2015, 38, E11. [Google Scholar] [CrossRef] [PubMed]
- Knotkova, H.; Hamani, C.; Sivanesan, E. Neuromodulation for chronic pain. Lancet 2021, 397, 2111–2124. [Google Scholar] [CrossRef] [PubMed]
- Büchel, C.; Geuter, S.; Sprenger, C. Placebo analgesia: a predictive coding perspective. Neuron 2014, 81, 1223–1239. [Google Scholar] [CrossRef] [PubMed]
- Wager, T. D.; Atlas, L. Y. The neuroscience of placebo effects: connecting context, learning and health. Nat. Rev. Neurosci. 2015, 16, 403–418. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Ahmad, M.; Al-Ahmadi, A. Virtual reality for pain management: a systematic review. Pain Res. Manag. 2020, 2020, 3427402. [Google Scholar] [CrossRef] [PubMed]
- Eccleston, C.; Crombez, G. Pain demands attention: a cognitive-affective model of the interruptive function of pain. Psychol. Bull. 1999, 125, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Hallett, M. Neurophysiology of dystonia: the role of inhibition. Neurobiol. Dis. 2011, 42, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Trompetto, C.; Marinelli, L.; Mori, L. Pathophysiology of spasticity: implications for neurorehabilitation. Biomed Res. Int. 2014, 2014, 354906. [Google Scholar] [CrossRef] [PubMed]
- Crone, C.; Nielsen, J.; Petersen, N. Reciprocal inhibition between ankle dorsiflexors and plantarflexors in man. J. Physiol. 1987, 389, 163–185. [Google Scholar] [CrossRef] [PubMed]
- Knikou, M. Neural control of locomotion and training-induced plasticity after spinal and cerebral lesions. Clin. Neurophysiol. 2010, 121, 1655–1668. [Google Scholar] [CrossRef] [PubMed]
- Lohse, A.; Meder, B.; Nielsen, J. B. Precision medicine in the treatment of spasticity: a Delphi study. Front. Neurol. 2020, 11, 837. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.; Westenberger, A. Genetics of Parkinson’s disease. Cold Spring Harb. Perspect. Med. 2012, 2, a008888. [Google Scholar] [CrossRef] [PubMed]
- Palfi, S.; Gurruchaga, J. M.; Ralph, G. S. Long-term safety and tolerability of ProSavin, a lentiviral vector-based gene therapy for Parkinson’s disease: a dose escalation, open-label, phase 1/2 trial. Lancet 2014, 383, 1138–1146. [Google Scholar] [CrossRef] [PubMed]
- Spear, B. B.; Heath-Chiozzi, M.; Huff, J. Clinical application of pharmacogenetics. Trends Mol. Med. 2001, 7, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Kühn, A. A.; Volkmann, J. Innovations in deep brain stimulation methodology. Mov. Disord. 2017, 32, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Arlotti, M.; Rosa, M.; Marceglia, S. The adaptive deep brain stimulation challenge. Parkinsonism Relat. Disord. 2016, 28, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Priori, A.; Foffani, G.; Rossi, L. Adaptive deep brain stimulation (aDBS) controlled by local field potential oscillations. Exp. Neurol. 2013, 245, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, R.; Petersson, P.; Siesser, W. B. Spinal cord stimulation restores locomotion in animal models of Parkinson’s disease. Science 2009, 323, 1578–1582. [Google Scholar] [CrossRef] [PubMed]
- Richards, B. A.; Lillicrap, T. P.; Beaudoin, P. A deep learning framework for neuroscience. Nat. Neurosci. 2019, 22, 1761–1770. [Google Scholar] [CrossRef] [PubMed]
- Sussillo, D.; Churchland, M. M.; Kaufman, M. T. A neural network that finds a naturalistic solution for the production of muscle activity. Nat. Neurosci. 2015, 18, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Barak, O. Recurrent neural networks as versatile tools of neuroscience research. Curr. Opin. Neurobiol. 2017, 46, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hassabis, D.; Kumaran, D.; Summerfield, C. Neuroscience-inspired artificial intelligence. Neuron 2017, 95, 245–258. [Google Scholar] [CrossRef] [PubMed]
- Yang, G. R.; Wang, X. J. Artificial neural networks for neuroscientists: a primer. Neuron 2020, 107, 1048–1070. [Google Scholar] [CrossRef] [PubMed]
- Vinyals, O.; Toshev, A.; Bengio, S. Show and tell: a neural image caption generator. Proc. IEEE Comput. Soc. Conf. Comput. Vis. Pattern Recognit. 2015, 3156–3164. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Cui, G.; Hu, S. Graph neural networks: a review of methods and applications. AI Open 2020, 1, 57–81. [Google Scholar] [CrossRef] [PubMed]
- LeCun, Y.; Bengio, Y.; Hinton, G. Deep learning. Nature 2015, 521, 436–444. [Google Scholar] [CrossRef] [PubMed]
- King, R. D.; Rowland, J.; Oliver, S. G. The automation of science. Science 2009, 324, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Kitano, H. Artificial intelligence to win the Nobel Prize and beyond: creating the engine for scientific discovery. AI Mag. 2016, 37, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Sparkes, A.; Aubrey, W.; Byrne, E. Towards robot scientists for autonomous scientific discovery. Autom. Exp. 2010, 2, 1. [Google Scholar] [CrossRef] [PubMed]
- Settles, B. Active learning literature survey. Univ. Wis. Madison Comput. Sci. Tech. Rep. 2010, 1648, 1–67. [Google Scholar] [PubMed]
- Hamburg, M. A.; Collins, F. S. The path to personalized medicine. N. Engl. J. Med. 2010, 363, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Jameson, J. L.; Longo, D. L. Precision medicine—personalized, problematic, and promising. N. Engl. J. Med. 2015, 372, 2229–2234. [Google Scholar] [CrossRef] [PubMed]
- Ashley, E. A. Towards precision medicine. Nat. Rev. Genet. 2016, 17, 507–522. [Google Scholar] [CrossRef] [PubMed]
- Collins, F. S.; Varmus, H. A new initiative on precision medicine. N. Engl. J. Med. 2015, 372, 793–795. [Google Scholar] [CrossRef] [PubMed]
- Denny, J. C.; Collins, F. S. Precision medicine in 2030—seven ways to transform healthcare. Cell 2021, 184, 1415–1419. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, G. S.; Phillips, K. A. Precision medicine: from science to value. Health Aff. 2018, 37, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Topol, E. J. High-performance medicine: the convergence of human and artificial intelligence. Nat. Med. 2019, 25, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Obermeyer, Z.; Emanuel, E. J. Predicting the future—big data, machine learning, and clinical medicine. N. Engl. J. Med. 2016, 375, 1216–1219. [Google Scholar] [CrossRef] [PubMed]
- Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 2001, 69, 89–95. [CrossRef] [PubMed]
- Strimbu, K.; Tavel, J. A. What are biomarkers? Curr. Opin. HIV AIDS 2010, 5, 463–466. [Google Scholar] [CrossRef] [PubMed]
- Aronson, J. K.; Ferner, R. E. Biomarkers—a general review. Curr. Protoc. Pharmacol. 2017, 76, 9. [Google Scholar] [CrossRef] [PubMed]
- Califf, R. M. Biomarker definitions and their applications. Exp. Biol. Med. 2018, 243, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Adewole, D. O.; Serruya, M. D.; Wolf, J. A. Bioactive neuroelectronic interfaces. Front. Neurosci. 2019, 13, 269. [Google Scholar] [CrossRef] [PubMed]
- Hong, G.; Lieber, C. M. Novel electrode technologies for neural recordings. Nat. Rev. Neurosci. 2019, 20, 330–345. [Google Scholar] [CrossRef] [PubMed]
- Won, S. M.; Song, E.; Zhao, J. Recent advances in materials, devices, and systems for neural interfaces. Adv. Mater. 2018, 30, e1800534. [Google Scholar] [CrossRef] [PubMed]
- Sahyouni, R.; Chen, J. An overview of neural interface technology: current state and future directions. J. Neural Eng. 2018, 15, 021001. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Canales, A.; Anikeeva, P. Neural recording and modulation technologies. Nat. Rev. Mater. 2017, 2, 16093. [Google Scholar] [CrossRef] [PubMed]
- Park, D. W.; Schendel, A. A.; Mikael, S. Graphene-based carbon-layered electrode array technology for neural imaging and optogenetic applications. Nat. Commun. 2014, 5, 5258. [Google Scholar] [CrossRef] [PubMed]
- Rogers, J. A.; Someya, T.; Huang, Y. Materials and mechanics for stretchable electronics. Science 2010, 327, 1603–1607. [Google Scholar] [CrossRef] [PubMed]
- Viventi, J.; Kim, D. H.; Vigeland, L. Flexible, foldable, actively multiplexed, high-density electrode array for mapping brain activity in vivo. Nat. Neurosci. 2011, 14, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Patil, A. C.; Thakor, N. V. Implantable neurotechnologies: a review of micro- and nanoelectrodes for neural recording. Med. Biol. Eng. Comput. 2016, 54, 23–44. [Google Scholar] [CrossRef] [PubMed]
- Anikeeva, P.; Koppes, R. A. Restoring the sense of touch. Science 2015, 350, 274–275. [Google Scholar] [CrossRef] [PubMed]
- Khodagholy, D.; Gelinas, J. N.; Buzsáki, G. Learning-enhanced coupling between ripple oscillations in association cortices and hippocampus. Science 2017, 358, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Obien, M. E.; Deligkaris, K.; Bullmann, T. Revealing neuronal function through microelectrode array recordings. Front. Neurosci. 2015, 8, 423. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



