Submitted:
18 January 2025
Posted:
21 January 2025
You are already at the latest version
Abstract
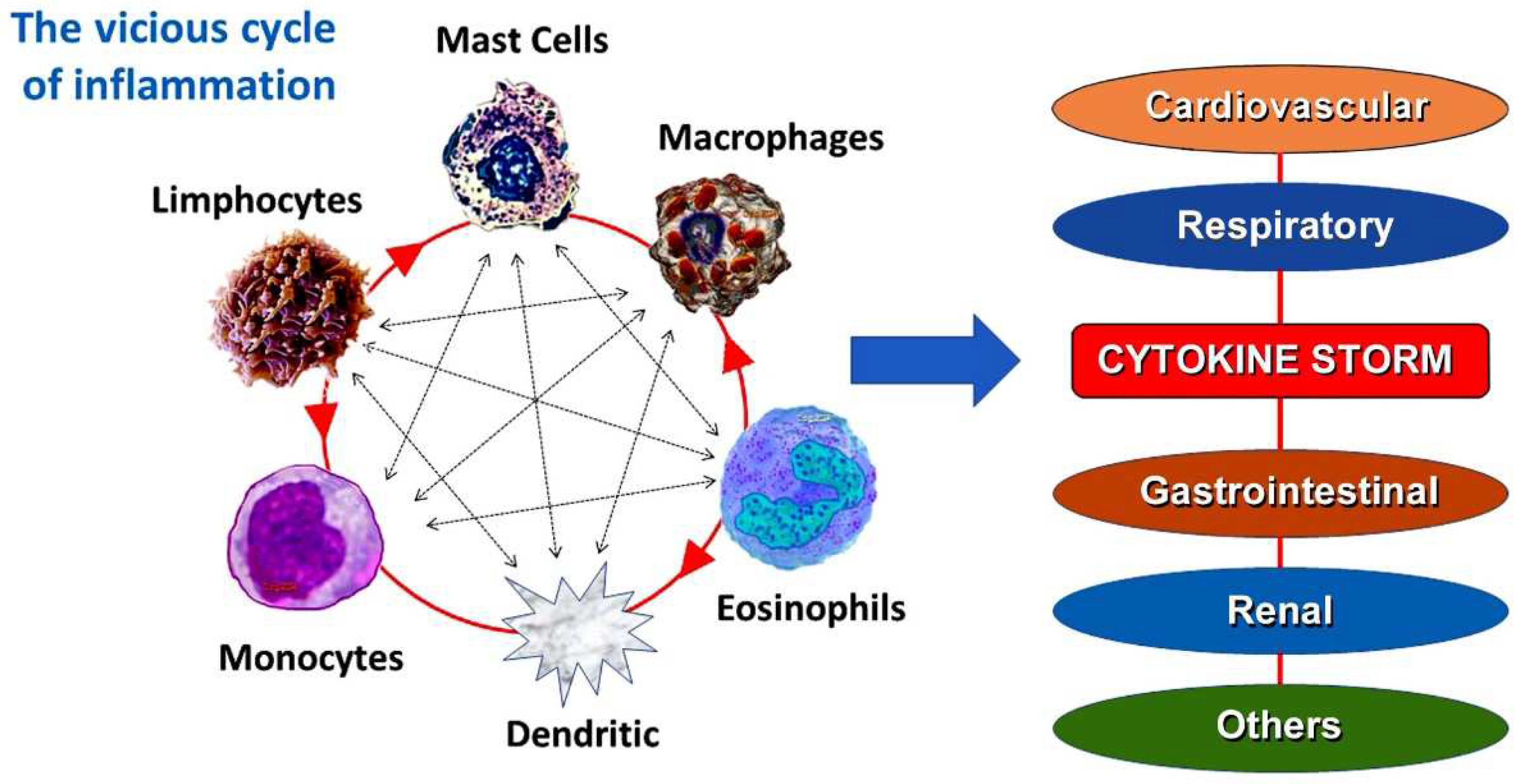
This narrative review explains the history of anaphylactic or hypersensitivity reactions, their connection to the cardiovascular system, and the Kounis syndrome, which is linked to hypersensitivity. Additional subjects discussed include immunoglobulin E and serum tryptase, common pathways of allergic and nonallergic cardiovascular events, current perspectives on Kounis syndrome, allergic myocardial infarction, allergic angina, and the impact of COVID-19 and its vaccination on Kounis syndrome. Kounis syndrome is a distinct kind of acute vascular disease that affects the coronary, cerebral, mesenteric, peripheral, and venous systems. Kounis syndrome is currently used to describe coronary symptoms linked to disorders involving mast cell activation and inflammatory cell interactions, such as those involving T-lymphocytes and macrophages, which further induce allergic, hypersensitive, anaphylactic, or anaphylactic insults. Platelet activating factor, histamine, neutral proteases like tryptase and chymase, arachidonic acid products, and a range of cytokines and chemokines released during the activation process are among the inflammatory mediators that cause it. Proinflammatory cytokines are primarily produced by mast cells in COVID-19 infections. Mast cell-derived proteases and eosinophil-associated mediators are also more prevalent in the lung tissues and sera of COVID-19 patients. Virus-associated molecular patterns can activate mast cells, but allergic triggers are typically the cause. By activating SARS-CoV-2 and other toll-like receptors, a variety of proinflammatory mediators, including IL-6 and IL-1β, are released, potentially contributing to the pathology of COVID-19.
Keywords:
1. Introduction
2. The antecedentes of Kounis syndrome
- a.
- Foreign proteins inducing acute carditis
- b.
- Rheumatic carditis and Morphologic cardiac reactions
3. Allergic angina and allergic myocardial infarction
4. Current views on Kounis syndrome
5. Common pathway between allergic and not allergic vascular events
6. Myocardial infarction, a preventable disease?
7. IgEs and Kounis Syndrome
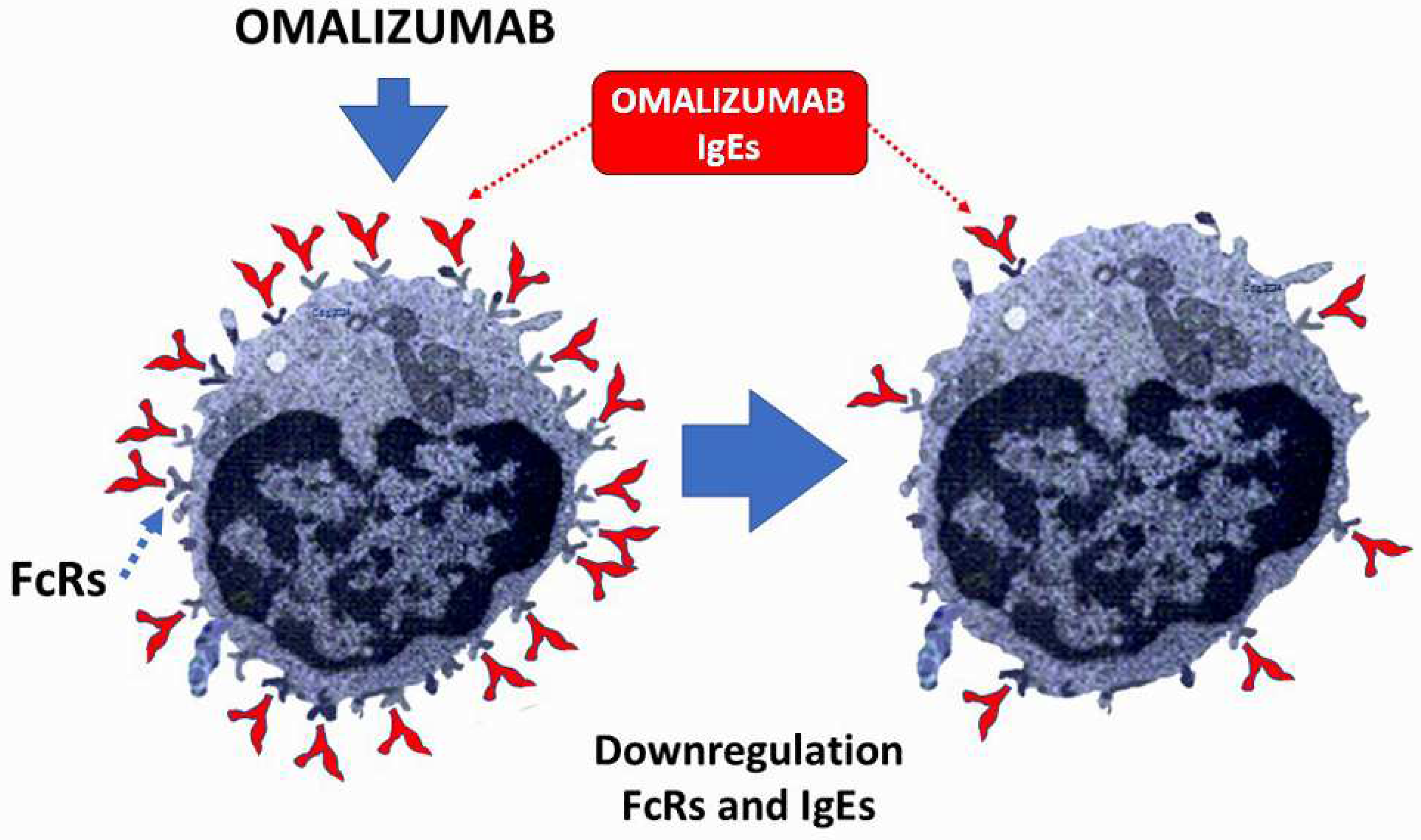
8. The lower IgEs the better for Kounis syndrome
9. Serum tryptase, a unique mast cel derived cytokine
10. Kounis syndrome, COVID-19 and vaccines
- a.
- Actual Kounis syndrome and COVID-19
- Patients with COVID-19 exhibit cytokine storm and abnormally high levels of inflammatory mediators, including IL-6, tumor necrosis factor-alpha (TNF-a), and C-reactive protein (CRP). Because these inflammatory mediators can cause coronary vasospasm, plaque destabilization, and thrombus formation, they have been linked to the pathophysiology of Kounis syndrome [66].
- Cytokines produced by mast cells, which are the main pathophysiological causes of Kounis syndrome can increase the blood–brain barriers’ permeability, which accounts for the SARS-CoV-2 “COVID-19 brain fog.” This can happen either directly by activating mast cells or indirectly by allowing cytokines to enter through a compromised blood–brain barriers [67].
- COVID-19 has an impact on the peripheral and coronary arteries. It may result in vascular or endothelial damage, hypoxic injury, cytokine storm, plaque rupture and microthrombi, coronary spasm, and an elevated risk of stent thrombosis. This is brought on by the underlying hypercoagulable disease, which clinically resembles the three primary forms of Kounis syndrome: stent thrombosis, coronary spasm, and acute myocardial infarction [29].
-
Activation of the immune system by COVID-19 in asymptomatic patients may raise the chance of developing Kounis syndrome, an unstable condition with susceptible plaques prone to thrombosis, from asymptomatic, subclinical, or atherosclerotic disease [68].
- b.
- COVID-19 vaccines and Kounis syndrome
11. Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| C3b | Complement component 3 |
| COVID-19 | Coronavirus disease 2019 |
| CRP | C-reactive protein |
| FcR | Fragment crystallisable region |
| F FcεRI | Fragment crystallisable epsilon region RI |
| FcεRII | Fragment crystallisable epsilon region RII |
| FcgRI | Fragment crystallisable gamma region RΙ |
| FcgRII | Fragment crystallisable gamma region ΙI |
| IgE | Immunoglobulin E |
| Ι IL-6 | Interleukin-6 |
| IgE | Immunoglobulin G |
| SCF | Stem cell factor |
| TNFα | Tumor necrosis factor-alpha |
References
- Krombach JW, Kampe S, Keller CA, Wright PM. Pharaoh Menes’ death after an anaphylactic reaction the end of a myth. Allergy 2004; 59: 1234-5. [CrossRef]
- Clark E. Serum carditis: morphologic cardiac alterations in man associated with serum disease. J Am Med Assoc 1938; 110: 1098–100. [CrossRef]
- Wadsworth GM, Brown CH. Serum reaction complicated by acute carditis. J Pediat 1940; 17: 801–5. [CrossRef]
- Rich AR, Gregory JE. Experimental evidence that lesions with basic characteristics of rheumatic carditis can result from anaphylactic hypersensitivity. Bull Johns Hopkins Hosp 1943; 73: 239–64. [CrossRef]
- Czickeli H. Contribution to the problem of the allergic etiology of angina pectoris and myocardial infarct. Klin Med Osterr Z Wiss Prakt Med 1950; 5: 364–7.
- Schultheiss E. Clinical aspects of allergic heart diseases. Dtsch Med J 1964;15:15–8.
- Auer J, Lewis PA. The physiology of the immediate reaction of anaphylaxis in the guinea-pig. J Exp Med 1910;12:151-75. [CrossRef]
- Nguyen SMT, Rupprecht CP, Haque A, Pattanaik D, Yusin J, Krishnaswamy G. Mechanisms Governing Anaphylaxis: Inflammatory Cells, Mediators, Endothelial Gap Junctions and Beyond. Int J Mol Sci 2021; 22: 7785. [CrossRef]
- Ribatti D, d’Amati A. Hematopoiesis and Mast Cell Development. Int J Mol Sci. 2023; 24: 10679. [CrossRef]
- Chorro L, Geissmann F. Development and homeostasis of ’resident’ myeloid cells: the case of the Langerhans cell. Trends Immunol 2010; 31: 438-45.
- Cline MJ. Histiocytes and histiocytosis. Blood 1994; 84: 2840-53.
- Swift HF. Rheumatic fever. JAMA 1929; 92: 2071.
- Gross I, Loewe I, Eliasoph B. Journal of Experimental Medicine 1929; 50: 41.
- Besterman EMM. Some notes on the history of rheumatic carditis. West Indian medical Journal 2001; 50: 180-182.
- Moukabary T. Willem Einthoven (1860-1927): Father of electrocardiography. Cardiol J. 2007;14(3):316-7.
- Pfister CW, Plice SG. Acute myocardial infarction during a prolonged allergic reaction to penicillin. Am Heart J 1950; 40: 945–947. [CrossRef]
- Kounis NG, Zavras GM. Histamine-induced coronary artery spasm: the concept of allergic angina. Br J Clin Pract 1991; 45: 121-128. [CrossRef]
- Constantinides P. Infiltrates of activated mast cells at the site ofcoronary atheromatous erosion or rupture in myocardial infarction. Circulatory 1995; 92: 1083.
- Brawnvald E. Unstable angina. An etiologic approach to management. Circulation 1998; 98:2 2.
- González-de-Olano D, Alvarez-Twose I, Matito A, Sánchez-Muñoz L, Kounis NG, Escribano L. Mast cell activation disorders presenting with cerebral vasospasm-related symptoms: a “Kounis-like” syndrome? Int J Cardiol 2011; 150: 210-1.
- Anastogiannis H, Litsardopoulos P, Anastopoulou GG, Petsas A, Tsigkas G, Kounis NG, Ravani I, Argyriou AA. Irreversible diffuse hypoxic-ischemic encephalopathy, secondary to type I Kounis syndrome. Int J Neurosci. 2020 Jul;130(7):746-748. [CrossRef]
- Peláez-Pérez JM, Sánchez Casado M, Álvarez-Twose I, Kounis NG. Amoxicillin-clavulanic acid-induced type II Kounis syndrome during general anaesthesia complicated with hypoxic-ischaemic encephalopathy. Rev Esp Anestesiol Reanim (Engl Ed). 2021; 68: 161-164. [CrossRef]
- Kounis N, Koniari I, Tsigkas G, Davlouros P. Rectosigmoid ischemia and cerebral coma following gadolinium induced anaphylaxis: A new manifestation of Kounis syndrome presented as devastating complication. Ann Ital Chir 2020; 91: 442-444.
- Goto M, Matsuzaki M, Fuchinoue A, Urabe N, Kawagoe N, Takemoto I, Tanaka H, Watanabe T, Miyazaki T, Takeuchi M, et al. Chronic athero-sclerotic mesenteric ischemia that started to develop symptoms just after anaphylaxis. Case Rep Gastroenterol 2012; 6:300-30.
- Adachi H, Ihara M, Nojima Y, Kurimoto T, Nanto S. Kounis syndrome caused by anaphylaxis without skin manifestations after cefazolin administration. J Allergy Clin Immunol Pract 2019 ;7: 317-319. [CrossRef]
- Kakarla P, Rijey J, Venugopal Reddy YC, Manechala UB. Central retinal vein occlusion with concurrent paracentral acute middle maculopathy secondary to honeybee sting. Indian J Ophthalmol 2024; 4: 475-477. [CrossRef]
- Kounis NG, Koniari I, Velissaris D, Tzanis G, Hahalis G. Kounis Syndrome—not a Single-organ Arterial Disorder but a Multisystem and Multidisciplinary Disease. Balkan Med J 2019; 36: 212-221. [CrossRef]
- Puri P, Kachhadia MP, Sardana P, Bhagat R, Dekowski S, Fohle E. Adrenaline, Takotsubo, Anaphylaxis, and Kounis Syndrome (ATAK) Complex Unveiled: Integrating Takotsubo and Kounis Syndromes in the Context of Chemotherapy-Related Anaphylaxis. Cureus 2024; 16: e53145. [CrossRef]
- Kounis NG, Mplani V, de Gregorio C, Koniari I. Attack the ATAK. A Challenging Contemporary Complex: Pathophysiologic, Therapeutic, and Preventive Considerations. Balkan Med J 2023; 40: 308–311.
- Van Klei WA, Szabo MD, Hesterberg P E. Case 22-2023: A 59-Year-OldWoman with Hypotension and Electrocardiographic Changes N Engl J Med 2023; 389: 263–272.
- Levick SP. Histamine receptors in heart failure. Heart Fail Rev 2022; 27: 1355-1372. [CrossRef]
- Kounis NG. Kounis syndrome (allergic angina and allergic myocardial infarction): a natural paradigm? Int J Cardiol 2006; 110: 7-14.
- Zhang Y, Liu Y, Sun J, Zhang W, Guo Z, Ma Q. Arachidonic acid metabolism in health and disease. MedComm 2023; 4: e363. [CrossRef]
- Deliargyris EN, Raymond RJ, Theoharides TC, Boucher WS, Tate DA, Dehmer GJ. Sites of interleukin-6 release in patients with acute coronary syndromes and in patients with congestive heart failure. Am J Cardiol 2000;86:913– 8. [CrossRef]
- Tsioufis P, Theofilis P, Tsioufis K, Tousoulis D. The Impact of Cytokines in Coronary Atherosclerotic Plaque: Current Therapeutic Approaches. Int J Mol Sci 2022; 23: 15937. [CrossRef]
- Kaartinen M, Penttila A, Kovanen PT. Accumulation of activated mast cells in the shoulder region of human coronary atheroma, the prediction site of atheromatous rupture. Circulation 1994; 90: 1669–78. [CrossRef]
- Kounis NG, Koniari I, Soufras GD, Chourdakis E, Despotopoulos S, Davlouros P, Hahalis G. The Humble Relation of Kounis Syndrome, MINOCA (Myocardial Infarction With Nonobstructive Coronary Arteries) and MACE (Major Adverse Cardiac Events). Can J Cardiol 2018; 34: 1089.e7. [CrossRef]
- Kounis NG, Mplani V, Koniari I. Kounis syndrome: A natural paradigm for preventing mast cell activation-degranulation. Int J Cardiol 2025; 419: 132704. [CrossRef]
- Wollam J, Solomon M, Villescaz C, Lanier M, Evans S, Bacon C, Freeman D, Vasquez A, Vest A, Napora J et al. Inhibition of mast cell degranulation by novel small molecule MRGPRX2 antagonists. J Allergy Clin Immunol 2024; 154: 1033-1043. [CrossRef]
- Takematsu E, Massidda M, Auster J, Chen PC, Im B, Srinath S, Canga S, Singh A, Majid M, Sherman M, et al. Transmembrane stem cell factor protein therapeutics enhance revascularization in ischemia without mast cell activation. Nat Commun 2022; 13: 249. [CrossRef]
- Nemmar A, Hoet PH, Vermylen J, Nemery B, Hoylaerts MF. Pharmacological stabilization of mast cells abrogates late thrombotic events induced by diesel exhaust particles in hamsters. Circulation. 2004; 110: 1670-7. [CrossRef]
- Kounis NG, Hahalis G. Serum IgE levels in coronary artery disease. Atherosclerosis 2016; 251: 498-500. [CrossRef]
- Kim J, Lee JY, Kim HG, Kwak MW, Kang TH. Fc Receptor Variants and Disease: A Crucial Factor to Consider in the Antibody Therapeutics in Clinic. Int J Mol Sci 2021; 22: 9489. [CrossRef]
- Kounis NG, Mazarakis A, Almpanis G, Gkouias K, Kounis GN, Tsigkas G. The more allergens an atopic patient is exposed to, the easier and quicker anaphylactic shock and Kounis syndrome appear: Clinical and therapeutic paradoxes. J Nat Sci Biol Med 2014; 5: 240-4. [CrossRef]
- Kounis NG, Koniari I, Velissaris D, Soufras G, Hahalis G. Aortic aneurysm and dissection in systemic lupus erythematosus-pathophysiologic and therapeutic considerations. Eur J Rheumatol 2018; 5: 209-211. [CrossRef]
- Morena D, Anta Y, Dbouk C. Hyper IgE Syndrome With Multiple Respiratory Infections. Review About a Clinical Case. Open Respir Arch 2023; 5: 100270. [CrossRef]
- Kovanen PT, Mänttäri M, Palosuo T, Manninen V, Aho K. Prediction of myocardial infarction in dyslipidemic men by elevated levels of immunoglobulin classes A, E, and G, but not M. Arch Intern Med 1998; 158: 1434-9. [CrossRef]
- Kovanen PT, Kaartinen M, Paavonen T. Infiltrates of activated mast cells at the sites of coronary atheromatous erosion or rupture in.
- myocardial infarction. Circulation 1995; 92: 1084–88.
- Smith SA, Chruszcz M, Chapman MD, Pomés A. Human Monoclonal IgE Antibodies-a Major Milestone in Allergy. Curr Allergy Asthma Rep 2023; 23: 53-65. [CrossRef]
- Vogel M, Engeroff P. A Comparison of Natural and Therapeutic Anti-IgE Antibodies. Antibodies (Basel) 2024; 13: 58. [CrossRef]
- Castells M, Irani AA, Scwarf LB. Evaluation of human peripheral. [CrossRef]
- blood leucocytes for mast cell tryptase. J Immunol 1987; 138: 2814-19.
- Madsen AT, Kristiansen HP, Winther-Larsen A. Short-term biological variation of serum tryptase. Clin Chem Lab Med 2023; 62: 713-719. [CrossRef]
- Mateja A, Wang Q, Chovanec J, Kim J, Wilson KJ, Schwartz LB, Glover SC, Carter MC, Metcalfe DD, Brittain E, et al. Defining baseline variability of serum tryptase levels improves accuracy in identifying anaphylaxis. J Allergy Clin Immunol 2022; 149: 1010-1017.e10. [CrossRef]
- Schwartz LB, Bradford TR, Rouse C, Irani AM, Rasp G, Van der Zwan JK, Van der Linden PW. Development of a new, more sensitive immunoassay for human tryptase: use in systemic anaphylaxis. J Clin Immunol 1994; 14: 190-204. [CrossRef]
- Baretto RL, Beck S, Heslegrave J, Melchior C, Mohamed O, Ekbote A, Huissoon AP, Krishna MT. Validation of international consensus equation for acute serum total tryptase in mast cell activation: A perioperative perspective. Allergy 2017; 72: 2031-2034. [CrossRef]
- Kounis NG. Serum tryptase levels and Kounis syndrome. Int J Cardiol 2007 18; 114: 407-8. [CrossRef]
- 59. Kervinen H, Kaartinen M, Makynen H, Palosuo T, Manttari M,.
- Kovanen PT. Serum tryptase levels in acute coronary syndromes. Int J. [CrossRef]
- Cardiol 2005; 104: 138–43.
- Khan S. Mast cell tryptase level should be checked in all patients with suspected Kounis syndrome. Eur Heart J 2020; 41: 3018. [CrossRef]
- Valent P, Hoermann G, Bonadonna P, Hartmann K, Sperr WR, Broesby-Olsen S, Brockow K, Niedoszytko M, Hermine O, Chantran Y, et al. The Normal Range of Baseline Tryptase Should Be 1 to 15 ng/mL and Covers Healthy Individuals With HαT. J Allergy Clin Immunol Pract 2023; 11: 3010-3020. [CrossRef]
- Nishiga M, Wang DW, Han Y, Lewis DB, Wu JC. COVID-19 and cardiovascular disease: from basic mechanisms to clinical perspectives. Nat Rev Cardiol 2020; 17: 543-558. [CrossRef]
- Wang X, Zhang P, Tang Y, Chen Y, Zhou E, Gao K. Mast cells: a double-edged sword in inflammation and fibrosis. Front Cell Dev Biol. 2024; 12: 1466491. [CrossRef]
- Budnevsky AV, Avdeev SN, Kosanovic D, Ovsyannikov ES, Savushkina IA, Alekseeva NG, Feigelman SN, Shishkina VV, Filin AA, Esaulenko DI, Perveeva IM. Involvement of Mast Cells in the Pathology of COVID-19: Clinical and Laboratory Parallels. Cells 2024; 13: 711. [CrossRef]
- Asaba CN, Ekabe CJ, Ayuk HS, Gwanyama BN, Bitazar R, Bukong TN. Interplay of TLR4 and SARS-CoV-2: Unveiling the Complex Mechanisms of Inflammation and Severity in COVID-19 Infections. J Inflamm Res 2024; 17 :5077-5091.
- Ribatti D, d’Amati A. Hematopoiesis and Mast Cell Development. Int J Mol Sci 2023;24: 10679. [CrossRef]
- Poto R, Marone G, Galli SJ, Varricchi G. Mast cells: a novel therapeutic avenue for cardiovascular diseases? Cardiovasc Res 2024; 120: 681-698.
- Balaji A, Kalurami VK. Kounis syndrome in the era of COVID-19: pathophysiology, clinical challenges, and therapeutic approaches. Frontiers in emergency medicine 2024; 8: e35.
- Kounis NG, Koniari I, de Gregorio C. COVID-19 and Kounis Syndrome: Deciphering Their Relationship. Balkan Med J 2021; 38: 145-149. [CrossRef]
- Saba L, Gerosa C, Wintermark M, Hedin U, Fanni D, Suri JS, Balestrieri A, Faa G. Can COVID19 trigger the plaque vulnerability-a Kounis syndrome warning for “asymptomatic subjects”. Cardiovasc Diagn Ther 2020; 10: 1352-1355.
- Zhao C, Lei R, Liu S, Zhao M. Kounis syndrome following COVID-19 vaccination: Clinical manifestations, mechanisms and management. Hum Vaccin Immunother 2024; 20: 23654. [CrossRef]
- Moghimi SM. Allergic Reactions and Anaphylaxis to LNP-Based COVID-19 Vaccines. Mol Ther 2021; 29: 898–900. [CrossRef]
- Awaya T, Hara H, Moroi M. Cytokine Storms and Anaphylaxis Following COVID-19 mRNA-LNP Vaccination: Mechanisms and Therapeutic Approaches. Diseases 2024; 12: 231.
- Fiolet T, Kherabi Y, MacDonald CJ, Ghosn J, Peiffer-Smadja N. Comparing COVID-19 vaccines for their characteristics, efficacy and effectiveness against SARS-CoV-2 and variants of concern: a narrative review. Clin Microbiol Infect 2022; 28: 202-221.
- Kounis NG, Koniari I, Mplani V, Kouni SN, Plotas P, Tsigkas G. Acute Myocardial Infarction Within 24 Hours After COVID-19 Vaccination: Is Kounis Syndrome the Culprit? Am J Cardiol 2022; 162: 207.
- Rabinowicz AL, Carrazana E, Maggio ET. Improvement of Intranasal Drug Delivery with Intravail® Alkylsaccharide Excipient as a Mucosal Absorption Enhancer Aiding in the Treatment of Conditions of the Central Nervous System. Drugs R D 2021; 21: 361-369.
- Jutel M, Torres MJ, Palomares O, Akdis CA, Eiwegger T, Untersmayr E, Barber D, Zemelka-Wiacek M, Kosowska A, Palmer E, et al. COVID-19 vaccination in patients receiving allergen immunotherapy (AIT) or biologicals-EAACI recommendations. Allergy 2022; 77: 2313-2336. [CrossRef]
| Tryptase | Chymase | Cathepsin D |
|---|---|---|
| Activates the zymogen forms of metalloproteinases such as interstitial collagenase, gelatinase and stromelysin and can promote plaque disruption or rupture | Converts angiotensin Ι to angiotensin II. Angiotensin II receptors are found in the medial muscle cells of human coronary arteries. Thus angiotensin II generated by chymase could act synergistically with histamine and aggravate the local spasm of the infracted coronary artery. Chymase can also remove cholesterol from HDL | Angiotensin II forming protease |
| Degrades the pericellular matrix components fibonectin and vitronectin and neuropeptides such as vasoactive intestinal peptide and calcitonin gene related peptide | Activates matrix metalloproteinases 1,2,9 and plays a major role in the physiologic degradation of fibronectin and thrombin | Degrades both fibronectin and vascular endothelial cadherin which are necessary for adhesion of endothelial cells to their basement membrane and to each other |
| Tryptase can degrade high-density lipoprotein, ( “good” cholesterol) | ||
| Activates neighboring cells by cleaving and activating protease-activated receptor (PAR)-2 and thrombin receptors |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




