Submitted:
29 November 2024
Posted:
30 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
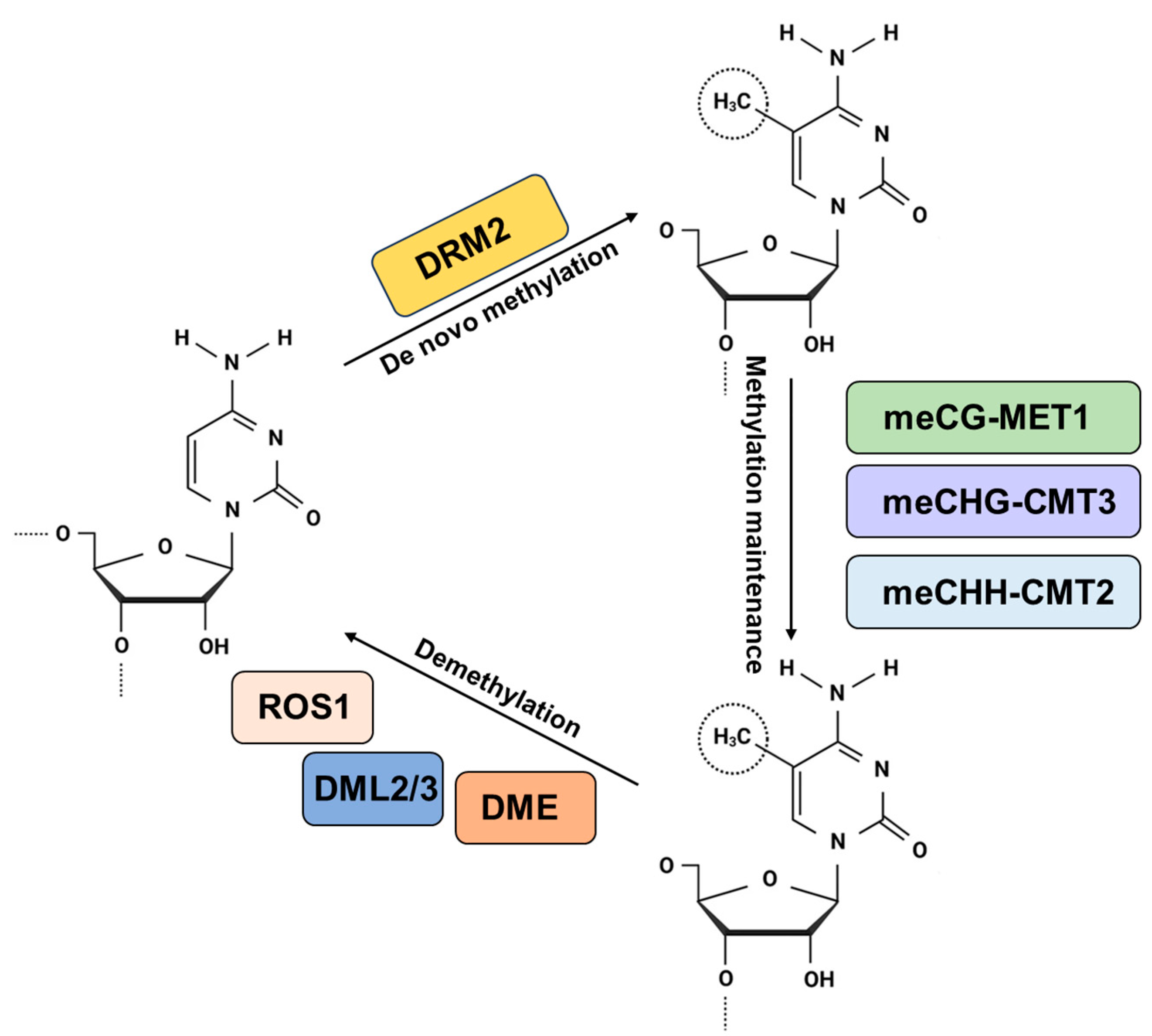
2. DNA Methylation Kinetics
2.1. Establishment of DNA Methylation
2.2. Maintenance of DNA Methylation
2.3. Demethylation of Active DNA
3. Molecular Functions of Plant DNA Methylation
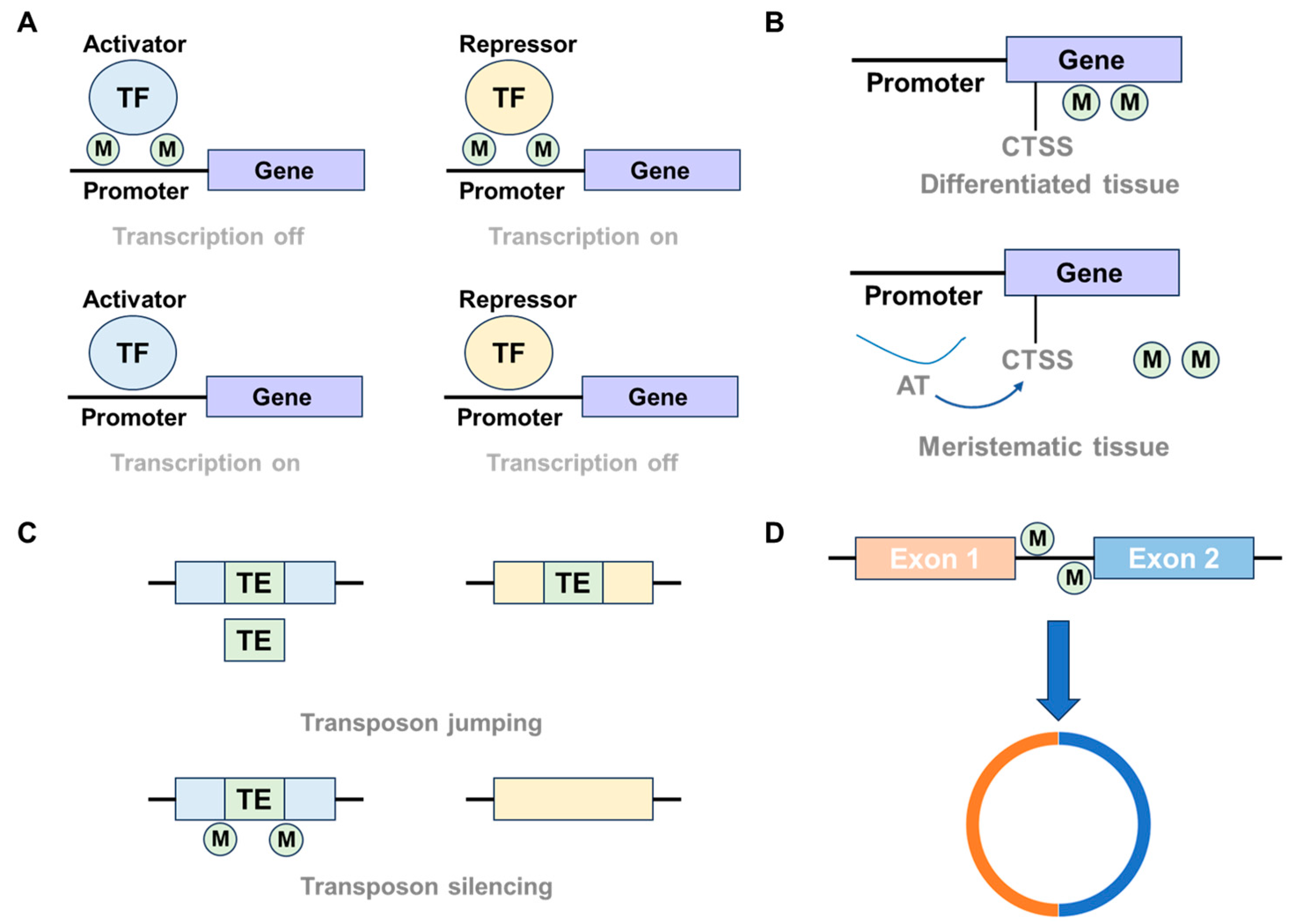
3.1. Regulation of Gene Expression
3.2. Transposon Silence
3.3. Chromosome Interaction
4. The Role of DNA Methylation in Plant Development
4.1. Genomic Imprinting and Seed Development
4.2. Role of DNA Methylation in Plant Meristem and Leaf Epidermal Development
4.3. Role of DNA Methylation in Flower Development
4.4. Role of DNA Methylation in Fruit Ripening
5. The role of Plant DNA Methylation in Abiotic Stress
5.1. High Temperature Stress
5.2. Drought Stress
5.3. Salt Stress
6. The Role of Plant DNA Methylation in Biological Stress
6.1. Dynamic Changes of DNA Methylation and Response to Biological Stress
6.2. DNA Methylation Regulation of Disease Resistance Genes
6.3. Synergistic Effect of DNA Methylation with Plant Hormone Signaling
7. Prospects of DNA Methylation in Crop Breeding
7. Conclusion and Prospect
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Berger, S.L.; Kouzarides, T.; Shiekhattar, R.; Shilatifard, A. An operational definition of epigenetics. Genes Dev. 2009, 23, 781–783.
- Henderson, I.R.; Jacobsen, S.E. Epigenetic inheritance in plants. Nature 2007, 447, 418–424.
- Maeji, H.; Nishimura, T. Epigenetic Mechanisms in Plants. Adv. Bot. Res. 2018, 88, 21–47.
- Fransz, P.F.; de Jong, J.H. Chromatin dynamics in plants. Curr. Opin. Plant Biol. 2002, 5, 560–567.
- Zilberman, D.; Gehring, M.; Tran, R.K.; Ballinger, T.; Henikoff, S. Genome-wide analysis of Arabidopsis thaliana DNA methylation uncovers an interdependence between methylation and transcription. Nat. Genet. 2007, 39, 61–69.
- Zhang, M.; Kimatu, J.N.; Xu, K.; Liu, B. DNA cytosine methylation in plant development. J. Genet. Genom. 2010, 37, 1–12.
- Ashapkin, V.V.; Kutueva, L.I.; Aleksandrushkina, N.I.; Vanyushin, B.F. Epigenetic Mechanisms of Plant Adaptation to Biotic and Abiotic Stresses. Int. J. Mol. Sci. 2020, 21, 7457.
- Li, E., Bestor, T. H. & Jaenisch, R. Targeted mutation of the DNA methyltransferase gene results in embryonic lethality. Cell 69, 915–926 (1992).
- Okano, M., Bell, D. W., Haber, D. A. & Li, E. DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 99, 247–257 (1999).
- Alleman, M. et al. An RNA-dependent RNA polymerase is required for paramutation in maize. Nature 442, 295–298 (2006).
- Wyatt, G. R. Occurrence of 5-methylcytosine in nucleic acids. Nature 166, 237–238 (1950).
- Vidalis, A. et al. Methylome evolution in plants. Genome Biol. 17, 264 (2016).
- Zhu, J. K. Active DNA demethylation mediated by DNA glycosylases. Annu. Rev. Genet. 43, 143–166 (2009).
- Wu, S. C. & Zhang, Y. Active DNA demethylation: many roads lead to Rome. Nat. Rev. Mol. Cell Biol. 11, 607–620 (2010).
- Law, J. A. & Jacobsen, S. E. Establishing, maintaining and modifying DNA methylation patterns in plants and animals. Nat. Rev. Genet. 11, 204–220 (2010).
- He, X. J., Chen, T. & Zhu, J. K. Regulation and function of DNA methylation in plants and animals. Cell Res. 21, 442–465 (2011).
- Law, J. A. & Jacobsen, S. E. Establishing, maintaining and modifying DNA methylation patterns in plants and animals. Nat. Rev. Genet. 11, 204–220 (2010).
- Stroud, H., Greenberg, M. V., Feng, S., Bernatavichute, Y. V. & Jacobsen, S. E. Comprehensive analysis of silencing mutants reveals complex regulation of the Arabidopsis methylome. Cell 152, 352–364 (2013).
- Stroud, H. et al. Non-CG methylation patterns shape the epigenetic landscape in Arabidopsis. Nat. Struct. Mol. Biol. 21, 64–72 (2014).
- Matzke, M. A. & Mosher, R. A. RNA-directed DNA methylation: an epigenetic pathway of increasing complexity. Nat. Rev. Genet. 15, 394–408 (2014).
- Zhang, H. & Zhu, J. K. RNA-directed DNA methylation. Curr. Opin. Plant Biol. 14, 142–147 (2011).
- Pikaard, C. S., Haag, J. R., Pontes, O. M., Blevins, T. & Cocklin, R. A transcription fork model for Pol IV and Pol V-dependent RNA-directed DNA methylation. Cold Spring Harb. Symp. Quant. Biol. 77, 205–212 (2012).
- Zhong, X. et al. Molecular mechanism of action of plant DRM de novo DNA methyltransferases. Cell 157, 1050–1060 (2014).
- Gao, Z. et al. An RNA polymerase II- and AGO4-associated protein acts in RNA-directed DNA methylation. Nature 465, 106–109 (2010).
- Vaucheret, H. & Voinnet, O. The plant siRNA landscape. Plant Cell 36, 246–275 (2023).
- Creasey, K. M. et al. miRNAs trigger widespread epigenetically activated siRNAs from transposons in Arabidopsis. Nature 508, 411–415 (2014).
- Cuerda-Gil, D. & Slotkin, R. K. Non-canonical RNA-directed DNA methylation. Nat. Plants 2, 16163 (2016).
- He, X. J. et al. An effector of RNA-directed DNA methylation in Arabidopsis is an ARGONAUTE 4-and RNA-binding protein. Cell 137, 498–508 (2009).
- Bies-Etheve, N. et al. RNA-directed DNA methylation requires an AGO4-interacting member of the SPT5 elongation factor family. EMBO Rep. 10, 649–654 (2009).
- Zhang, H. et al. An Rrp6-like protein positively regulates noncoding RNA levels and DNA methylation in Arabidopsis. Mol. Cell 54, 418–430 (2014).
- Ausin, I., Mockler, T. C., Chory, J. & Jacobsen, S. E. IDN1 and IDN2 are required for de novo DNA methylation in Arabidopsis thaliana. Nat. Struct. Mol. Biol. 16, 1325–1327 (2009).
- Zheng, Z. et al. An SGS3-like protein functions in RNA-directed DNA methylation and transcriptional gene silencing in Arabidopsis. Plant J. 62, 92–99 (2010).
- Ausin, I. et al. INVOLVED IN DE NOVO 2-containing complex involved in RNA-directed DNA methylation in Arabidopsis. Proc. Natl Acad. Sci. USA 109, 8374–8381 (2012).
- Zhang, C. J. et al. IDN2 and its paralogs form a complex required for RNA-directed DNA methylation. PLoS Genet. 8, e1002693 (2012).
- Zhang, H. et al. DTF1 is a core component of RNA-directed DNA methylation and may assist in the recruitment of Pol IV. Proc. Natl Acad. Sci. USA 110, 8290–8295 (2013).
- Law, J. A. et al. Polymerase IV occupancy at RNA-directed DNA methylation sites requires SHH1. Nature 498, 385–389 (2013).
- Wang, Y. et al. ZMP recruits and excludes Pol IV-mediated DNA methylation in a site-specific manner. Sci. Adv. 8, eadc9454 (2022).
- Zhou, M. et al. The CLASSY family controls tissue-specific DNA methylation patterns in Arabidopsis. Nat. Commun. 13, 244 (2022).
- Zhou, M., Palanca, A. M. S. & Law, J. A. Locus-specific control of the de novo DNA methylation pathway in Arabidopsis by the CLASSY family. Nat. Genet. 50, 865–873 (2018).
- Kanno, T. et al. Involvement of putative SNF2 chromatin remodeling protein DRD1 in RNA-directed DNA methylation. Curr. Biol. 14, 801–805 (2004).
- Kanno, T. et al. A structural-maintenance-of-chromosomes hinge domain-containing protein is required for RNA-directed DNA methylation. Nat. Genet. 40, 670–675 (2008).
- Law, J. A. et al. A protein complex required for polymerase V transcripts and RNA- directed DNA methylation in Arabidopsis. Curr. Biol. 20, 951–956 (2010).
- Zhong, X. H. et al. DDR complex facilitates global association of RNA polymerase V to promoters and evolutionarily young transposons. Nat. Struct. Mol. Biol. 19, 870–875 (2012).
- Liu, Z. W. et al. The SET domain proteins SUVH2 and SUVH9 are required for Pol V occupancy at RNA-directed DNA methylation loci. PLoS Genet. 10, e1003948 (2014).
- Johnson, L. M. et al. SRA- and SET-domain-containing proteins link RNA polymerase V occupancy to DNA methylation. Nature 507, 124–128 (2014). References 35 and 37 reveal how POL V may be recruited to some RdDM loci.
- Wierzbicki, A. T., Haag, J. R. & Pikaard, C. S. Noncoding transcription by RNA polymerase Pol IVb/Pol V mediates transcriptional silencing of overlapping and adjacent genes. Cell 135, 635–648 (2008).
- levins, T. et al. Identification of Pol IV and RDR2-dependent precursors of 24 nt siRNAs guiding de novo DNA methylation in Arabidopsis. eLife 4, e09591 (2015).
- Li, S. F. et al. Detection of Pol IV/RDR2-dependent transcripts at the genomic scale in Arabidopsis reveals features and regulation of siRNA biogenesis. Genome Res. 25, 235–245 (2015).
- Zhai, J. et al. A one precursor one siRNA model for Pol IV-dependent siRNA biogenesis. Cell 163, 445–455 (2015).
- Yang, D. L. et al. Dicer-independent RNA-directed DNA methylation in Arabidopsis. Cell Res. 26, 1264 (2016).
- Ye, R. Q. et al. A Dicer-independent route for biogenesis of siRNAs that direct DNA methylation in Arabidopsis. Mol. Cell 61, 222–235 (2016).
- Zheng, B. et al. Intergenic transcription by RNA polymerase II coordinates Pol IV and Pol V in siRNA-directed transcriptional gene silencing in Arabidopsis. Genes Dev. 23, 2850–2860 (2009).
- Duan, C. G. et al. Specific but interdependent functions for Arabidopsis AGO4 and AGO6 in RNA-directed DNA methylation. EMBO J. 34, 581–592 (2015).
- McCue, A. D. et al. ARGONAUTE 6 bridges transposable element mRNA-derived siRNAs to the establishment of DNA methylation. EMBO J. 34, 20–35 (2015).
- Mari-Ordonez, A. et al. Reconstructing de novo silencing of an active plant retrotransposon. Nat. Genet. 45, 1029–1039 (2013).
- Huang, C. F. et al. A pre-mRNA-splicing factor is required for RNA-directed DNA methylation in Arabidopsis. PLoS Genet. 9, e1003779 (2013).
- Dou, K. et al. The PRP6-like splicing factor STA1 is involved in RNA-directed DNA methylation by facilitating the production of Pol V-dependent scaffold RNAs. Nucleic Acids Res. 41, 8489–8502 (2013).
- Du, J., Johnson, L. M., Jacobsen, S. E. & Patel, D. J. DNA methylation pathways and their crosstalk with histone methylation. Nat. Rev. Mol. Cell Biol. 16, 519–532 (2015).
- Riggs, A. D. X inactivation, differentiation, and DNA methylation. Cytogenet. Cell Genet. 14, 9–25 (1975).
- Holliday, R. & Pugh, J. E. DNA modification mechanisms and gene activity during development. Science 187, 226–232 (1975).
- Bashtrykov, P. et al. Specificity of Dnmt1 for methylation of hemimethylated CpG sites resides in its catalytic domain. Chem. Biol. 19, 572–578 (2012).
- Song, J., Teplova, M., Ishibe-Murakami, S. & Patel, D. J. Structure-based mechanistic insights into DNMT1-mediated maintenance DNA methylation. Science 335, 709–712 (2012).
- Adam, S., Klingel, V., Radde, N. E., Bashtrykov, P. & Jeltsch, A. On the accuracy of the epigenetic copy machine: comprehensive specificity analysis of the DNMT1 DNA methyltransferase. Nucleic Acids Res. 51, 6622–6633 (2023).
- Bostick, M. et al. UHRF1 plays a role in maintaining DNA methylation in mammalian cells. Science 317, 1760–1764 (2007).
- Sharif, J. et al. The SRA protein Np95 mediates epigenetic inheritance by recruiting Dnmt1 to methylated DNA. Nature 450, 908–912 (2007).
- Woo, H. R., Dittmer, T. A. & Richards, E. J. Three SRA-domain methylcytosine-binding proteins cooperate to maintain global CpG methylation and epigenetic silencing in Arabidopsis. PLoS Genet. 4, e1000156 (2008).
- Petryk, N., Bultmann, S., Bartke, T. & Defossez, P. A. Staying true to yourself: mechanisms of DNA methylation maintenance in mammals. Nucleic Acids Res. 49, 3020–3032 (2021).
- Rocha, P. S. et al. The Arabidopsis HOMOLOGY-DEPENDENT GENE SILENCING1 gene codes for an S-adenosyl-L-homocysteine hydrolase required for DNA methylation-dependent gene silencing. Plant Cell 17, 404–417 (2005).
- Zhang, H. et al. Sulfamethazine suppresses epigenetic silencing in Arabidopsis by impairing folate synthesis. Plant Cell 24, 1230–1241 (2012).
- Zhou, H. R. et al. Folate polyglutamylation is involved in chromatin silencing by maintaining global DNA methylation and histone H3K9 dimethylation in Arabidopsis. Plant Cell 25, 2545–2559 (2013).
- Gong, Z. et al. ROS1, a repressor of transcriptional gene silencing in Arabidopsis, encodes a DNA glycosylase/lyase. Cell 111, 803–814 (2002). This study provides a genetic evidence for base excision repair-mediated active DNA demethylation.
- Gehring, M. et al. DEMETER DNA glycosylase establishes MEDEA polycomb gene self-imprinting by allele-specific demethylation. Cell 124, 495–506 (2006).
- Wu, X. & Zhang, Y. TET-mediated active DNA demethylation: mechanism, function and beyond. Nat. Rev. Genet. 18, 517–534 (2017).
- Agius, F., Kapoor, A. & Zhu, J. K. Role of the Arabidopsis DNA glycosylase/lyase ROS1 in active DNA demethylation. Proc. Natl Acad. Sci. USA 103, 11796–11801 (2006).
- Morales-Ruiz, T. et al. DEMETER and REPRESSOR OF SILENCING 1 encode 5-methylcytosine DNA glycosylases. Proc. Natl Acad. Sci. USA 103, 6853–6858 (2006).
- Penterman, J. et al. DNA demethylation in the Arabidopsis genome. Proc. Natl Acad. Sci. USA 104, 6752–6757 (2007).
- Huh, J. H., Bauer, M. J., Hsieh, T. F. & Fischer, R. L. Cellular programming of plant gene imprinting. Cell 132, 735–744 (2008).
- Martinez-Macias, M. I. et al. A DNA 3 ‘ phosphatase functions in active DNA demethylation in Arabidopsis. Mol. Cell 45, 357–370 (2012).
- Lee, J. et al. AP endonucleases process 5-methylcytosine excision intermediates during active DNA demethylation in Arabidopsis. Nucleic Acids Res. 42, 11408–11418 (2014).
- Raja P, Sanville BC, Buchmann RC, Bisaro DM (2008) Viral genome methylation as an epigenetic defense against geminiviruses. J Virol 82:8997–9007.
- Zhang, H. & Zhu, J. K. Active DNA demethylation in plants and animals. Cold Spring Harb. Symp. Quant. Biol. 77, 161–173 (2012).
- Lei, M. G. et al. Regulatory link between DNA methylation and active demethylation in Arabidopsis. Proc. Natl Acad. Sci. USA 112, 3553–3557 (2015).
- Williams, B. P., Pignatta, D., Henikoff, S. & Gehring, M. Methylation-sensitive expression of a DNA demethylase gene serves as an epigenetic rheostat. PLOS Genet. 11, e1005142 (2015).
- Domcke, S. et al. Competition between DNA methylation and transcription factors determines binding of NRF1. Nature 528, 575–579 (2015).
- Zhu, H., Wang, G. H. & Qian, J. Transcription factors as readers and effectors of DNA methylation. Nat. Rev. Genet. 17, 551–565 (2016).
- Tang, K., Lang, Z., Zhang, H. & Zhu, J. K. The DNA demethylase ROS1 targets genomic regions with distinct chromatin modifications. Nat. Plants 2, 16169 (2016).
- Lei, M. G. et al. Regulatory link between DNA methylation and active demethylation in Arabidopsis. Proc. Natl Acad. Sci. USA 112, 3553–3557 (2015).
- Feng, Z. et al. Genetic analysis implicates a molecular chaperone complex in regulating epigenetic silencing of methylated genomic regions. J. Integr. Plant Biol. 63, 1451–1461 (2021).
- Qian, W. et al. Regulation of active DNA demethylation by an alpha-crystallin domain protein in Arabidopsis. Mol. Cell 55, 361–371 (2014).
- Boone, B. A. et al. ACD15, ACD21, and SLN regulate the accumulation and mobility of MBD6 to silence genes and transposable elements. Sci. Adv. 9, eadi9036 (2023).
- Duan, C. G. et al. A pair of transposon-derived proteins function in a histone acetyltransferase complex for active DNA demethylation. Cell Res. 27, 226–240 (2017).
- Haslbeck, M., Weinkauf, S. & Buchner, J. Small heat shock proteins: simplicity meets complexity. J. Biol. Chem. 294, 2121–2132 (2019).
- Cokus, S. J. et al. Shotgun bisulphite sequencing of the Arabidopsis genome reveals DNA methylation patterning. Nature 452, 215–219 (2008).
- Du, J. M. et al. Dual binding of chromomethylase domains to H3K9me2-containing nucleosomes directs DNA methylation in plants. Cell 151, 167–180 (2012).
- Li, Q. et al. RNA-directed DNA methylation enforces boundaries between heterochromatin and euchromatin in the maize genome. Proc. Natl Acad. Sci. USA 112, 14728–14733 (2015).
- Zakrzewski, F., Schmidt, M., Van Lijsebettens, M. & Schmidt, T. DNA methylation of retrotransposons, DNA transposons and genes in sugar beet (Beta vulgaris L.). Plant J. 90, 1156–1175 (2017).
- Gouil, Q. & Baulcombe, D. C. DNA methylation signatures of the plant chromomethyltransferases. PLoS Genet. 12, e1006526 (2016).
- Grob, S., Schmid, M. W. & Grossniklaus, U. Hi-C analysis in Arabidopsis identifies the KNOT, a structure with similarities to the flamenco Locus of Drosophila. Mol. Cell 55, 678–693 (2014).
- Feng, S. et al. Genome-wide Hi-C analyses in wild-type and mutants reveal high-resolution chromatin interactions in Arabidopsis. Mol. Cell 55, 694–707 (2014).
- Rowley, M. J., Rothi, M. H., Bohmdorfer, G., Kucinski, J. & Wierzbicki, A. T. Long-range control of gene expression via RNA-directed DNA methylation. PLoS Genet. 13, e1006749 (2017).
- Gehring, M., Bubb, K. L. & Henikoff, S. Extensive demethylation of repetitive elements during seed development underlies gene imprinting. Science 324, 1447–1451 (2009).
- Hsieh, T. F. et al. Genome-wide demethylation of Arabidopsis endosperm. Science 324, 1451–1454 (2009).
- Ibarra, C. A. et al. Active DNA demethylation in plant companion cells reinforces transposon methylation in gametes. Science 337, 1360–1364 (2012).
- Park, K. et al. DNA demethylation is initiated in the central cells of Arabidopsis and rice. Proc. Natl Acad. Sci. USA 113, 15138–15143 (2016).
- Slotkin, R. K. et al. Epigenetic reprogramming and small RNA silencing of transposable elements in pollen. Cell 136, 461–472 (2009).
- Martínez, G., Panda, K., Köhler, C. & Slotkin, R. K. Silencing in sperm cells is directed by RNA movement from the surrounding nurse cell. Nat. Plants. 2, 16030 (2016).
- Ingouff, M. Live-cell analysis of DNA methylation during sexual reproduction in Arabidopsis reveals context and sex-specific dynamics controlled by noncanonical RdDM. Genes Dev. 31, 72–83 (2017).
- Walker, J. Sexual-lineage-specific DNA methylation regulates meiosis in Arabidopsis. Nat. Genet. 50, 130–137 (2018).
- Zhang, M. et al. Genome-wide high resolution parental-specific DNA and histone methylation maps uncover patterns of imprinting regulation in maize. Genome Res. 24, 167–176 (2014).
- Klosinska, M., Picard, C. L. & Gehring, M. Conserved imprinting associated with unique epigenetic signatures in the Arabidopsis genus. Nat. Plants 2, 16145 (2016).
- Rodrigues, J. A. et al. Imprinted expression of genes and small RNA is associated with localized hypomethylation of the maternal genome in rice endosperm. Proc. Natl Acad. Sci. USA 110, 7934–7939 (2013).
- Kinoshita, T. et al. One-way control of FWA imprinting in Arabidopsis endosperm by DNA methylation. Science 303, 521–523 (2004).
- Hsieh, T. F. et al. Regulation of imprinted gene expression in Arabidopsis endosperm. Proc. Natl Acad. Sci. USA 108, 1755–1762 (2011).
- Kinoshita, T. et al. One-way control of FWA imprinting in Arabidopsis endosperm by DNA methylation. Science 303, 521–523 (2004).
- Bratzel, F. et al. Regulation of the new Arabidopsis imprinted gene AtBMI1C requires the interplay of different epigenetic mechanisms. Mol. Plant 5, 260–269 (2012).
- Moreno-Romero, J., Jiang, H., Santos-Gonzalez, J. & Kohler, C. Parental epigenetic asymmetry of PRC2-mediated histone modifications in the Arabidopsis endosperm. EMBO J. 35, 1298–1311 (2016).
- Dong, X. M. et al. Dynamic and antagonistic allele-Specific epigenetic modifications controlling the expression of imprinted genes in maize endosperm. Mol. Plant 10, 442–455 (2017).
- Baubec, T., Finke, A., Scheid, O. M. & Pecinka, A. Meristem-specific expression of epigenetic regulators safeguards transposon silencing in Arabidopsis. EMBO Rep. 15, 446–452 (2014).
- Kawakatsu, T. et al. Unique cell-type-specific patterns of DNA methylation in the root meristem. Nat. Plants 2, 16058 (2016). This study demonstrates diversified epigenomes in different somatic cell types in A. thaliana roots.
- Zhou, S. L. et al. Cooperation between the H3K27me3 chromatin mark and non-CG methylation in epigenetic regulation. Plant Physiol. 172, 1131–1141 (2016).
- Candaele, J. et al. Differential methylation during maize leaf growth targets developmentally regulated genes. Plant Physiol. 164, 1350–1364 (2014).
- Yamamuro, C. et al. Overproduction of stomatal lineage cells in Arabidopsis mutants defective in active DNA demethylation. Nat. Commun. 5, 4062 (2014).
- Wang, Y. H., Xue, X. Y., Zhu, J. K. & Dong, J. Demethylation of ERECTA receptor genes by IBM1 histone demethylase affects stomatal development. Development 143, 4452–4461 (2016).
- Hieber, A.D.; Mudalige-Jayawickrama, R.G.; Kuehnle, A.R. Color genes in the orchid Oncidium Gower Ramsey: Identification, expression, and potential genetic instability in an interspecific cross. Planta 2006, 223, 521–531.
- Chiou, C.Y.; Pan, H.A.; Chuang, Y.N.; Yeh, K.W. Differential expression of carotenoid-related genes determines diversified carotenoid coloration in floral tissues of Oncidium cultivars. Planta 2010, 232, 937–948.
- Liu, X.J.; Chuang, Y.N.; Chiou, C.Y.; Chin, D.C.; Shen, F.Q.; Yeh, K.W. Methylation effect on chalcone synthase gene expression determines anthocyanin pigmentation in floral tissues of two Oncidium orchid cultivars. Planta 2012, 236, 401–409.
- Han, M.L.; Yin, J.; Zhao, Y.H.; Sun, X.W.; Meng, J.X.; Zhou, J.; Shen, T.; Li, H.H.; Zhang, F. How the Color Fades from Malus halliana Flowers: Transcriptome Sequencing and DNA Methylation Analysis. Front. Plant Sci. 2020, 11, 576054.
- Yuan, X.; Ma, K.; Zhang, M.; Wang, J.; Zhang, Q. Integration of Transcriptome and Methylome Analyses Provides Insight Into the Pathway of Floral Scent Biosynthesis in Prunus mume. Front. Genet. 2021, 12, 779557.
- Zhong, S. et al. Single-base resolution methylomes of tomato fruit development reveal epigenome modifications associated with ripening. Nat. Biotechnol. 31, 154–159 (2013).
- Liu, R. et al. A DEMETER-like DNA demethylase governs tomato fruit ripening. Proc. Natl Acad. Sci. USA 112, 10804–10809 (2015).
- Lang, Z. et al. Critical roles of DNA demethylation in the activation of ripening-induced genes and inhibition of ripening-repressed genes in tomato fruit. Proc. Natl Acad. Sci. USA 114, E4511–E4519 (2017).
- Telias, A. et al. Apple skin patterning is associated with differential expression of MYB10. BMC Plant Biol. 11, 93 (2011).
- El-Sharkawy, I., Liang, D. & Xu, K. N. Transcriptome analysis of an apple (Malus x domestica) yellow fruit somatic mutation identifies a gene network module highly associated with anthocyanin and epigenetic regulation. J. Exp. Bot. 66, 7359–7376 (2015).
- Daccord, N. et al. High-quality de novo assembly of the apple genome and methylome dynamics of early fruit development. Nat. Genet. 49, 1099–1106 (2017).
- Tollefson J (2020) How hot will earth get by 2100? Nature 580:443–445.
- Zandalinas SI, Mittler R, Balfagón D, Arbona V, Gómez-Cadenas A (2018) Plant adaptations to the combination of drought and high temperatures. Physiol Plant 162:2–12.
- Gao G, Li J, Li H et al (2014) Comparison of the heat stress induced variations in DNA methylation between heat-tolerant and heat-sensitive rapeseed seedlings. Breed Sci 64:125–133.
- Singh RK, Jaishankar J, Muthamilarasan M, Shweta S, Dangi A, Prasad M (2016) Genome-wide analysis of heat shock proteins in C4 model, foxtail millet identifies potential candidates for crop improvement under abiotic stress. Sci Rep 6:32641.
- Qian Y, Hu W, Liao J, Zhang J, Ren Q (2019) The dynamics of DNA methylation in the maize (Zea mays L.) inbred line B73 response to heat stress at the seedling stage. Biochem Biophys Res Commun 512:742–749.
- Lamelas L, Valledor L, Escandón M, Pinto G, Cañal MJ, Meijón M (2020) Integrative analysis of the nuclear proteome in Pinus radiata reveals thermopriming coupled to epigenetic regulation. J Exp Bot 71:2040–2057.
- Liu H, Able AJ, Able JA (2021a) Priming crops for the future: rewiring stress memory. Trends Plant Sci 11:1360–1385.
- Yu A, Lepère G, Jay F, Wang J, Bapaume L, Wang Y, Abraham AL, Penterman J, Fischer RL, Voinnet O, Navarro L (2013) Dynamics and biological relevance of DNA demethylation in Arabidopsis antibacterial defense. Proc Natl Acad Sci USA 110:2389–2394.
- Lafon-Placette C, Le Gac AL, Chauveau D et al (2018) Changes in the epigenome and transcriptome of the poplar shoot apical meristem in response to water availability affect preferentially hormone pathways. J Exp Bot 69:537–551.
- Arora H, Singh RK, Sharma S, Sharma N, Panchal A, Das T, Prasad A, Prasad M. DNA methylation dynamics in response to abiotic and pathogen stress in plants. Plant Cell Rep. 2022 Oct;41(10):1931-1944.
- Herman, J. J., & Sultan, S. E. (2016). DNA methylation mediates stress-induced transgenerational plasticity in plants. Proceedings of the National Academy of Sciences, 113(32), 8804–8809.
- Cai Q, Qiao L, Wang M, He B, Lin FM, Palmquist J, Huang SD, Jin H (2018) Plants send small RNAs in extracellular vesicles to fungal pathogen to silence virulence genes. Science 360:1126–1129.
- Ventouris, Y., et al. (2020). Adaptive epigenetic memory in alfalfa subjected to recurrent drought. Environmental and Experimental Botany, 179, 104218.
- Sallam, H., & Moussa, M. (2021). Amplified polymorphic DNA-based insights into drought tolerance in maize. Molecular Biology Reports, 48, 5647–5658.
- Wang, M., et al. (2015). Epigenetic basis of plant responses to salinity stress. Journal of Experimental Botany, 66(3), 809–820.
- Kumar, S., et al. (2020). DNA methylation dynamics under salt stress in crop plants. Plant Physiology and Biochemistry, 157, 192–202.
- Song, Q., et al. (2019). Role of epigenetics in plant stress responses and adaptation. Plant, Cell & Environment, 42(3), 583–589.
- Dowen, R. H., Pelizzola, M., Schmitz, R. J., Lister, R., Dowen, J. M., Nery, J. R., Dixon, J. E., & Ecker, J. R. (2012). Widespread dynamic DNA methylation in response to biotic stress. Proceedings of the National Academy of Sciences, 109(32), E2183–E2191.
- Yu, A., Lepère, G., Jay, F., Wang, J., Bapaume, L., Wang, Y., ... & Navarro, L. (2013). Dynamics and biological relevance of DNA demethylation in Arabidopsis antibacterial defense. Proceedings of the National Academy of Sciences, 110(6), 2389–2394.
- López A, Ramírez V, García-Andrade J, Flors V, Vera P (2011) The RNA silencing enzyme RNA polymerase V is required for plant immunity. PLoS Genet 7:e1002434.
- Wang, W., Pan, Y., Zhao, X., Zhu, L., Fu, B., & Li, Z. (2016). DNA methylation is critical for defense against the fungal pathogen Verticillium dahliae in Arabidopsis. The Plant Cell, 28(5), 1237–1251.
- Kou, H. P., Li, Y., Song, X. X., Ou, X. F., Xing, S. C., Ma, J., ... & Liu, B. (2011). Heritable alteration in DNA methylation induced by nitrogen-deficiency stress accompanies enhanced tolerance in rice (Oryza sativa L.). The Plant Journal, 65(5), 655–664.
- Boyko, A., & Kovalchuk, I. (2010). Transgenerational response to stress in plants and its application for breeding. Journal of Experimental Botany, 62(3), 633–644.
- Cubas, P., Vincent, C. & Coen, E. An epigenetic mutation responsible for natural variation in floral symmetry. Nature 401, 157–161 (1999).
- Zhang, H., Lang, Z. & Zhu, J. K. Dynamics and function of DNA methylation in plants. Nat. Rev. Mol. Cell Biol. 19, 489–506 (2018).
- Le TN, Schumann U, Smith NA, Tiwari S, Au PC, Zhu QH, Taylor JM, Kazan K, Llewellyn DJ, Zhang R, Dennis ES, Wang MB (2014) DNA demethylases target promoter transposable elements to positively regulate stress responsive genes in Arabidopsis. Genome Biol 15:1–18.
- Manning, K. et al. A naturally occurring epigenetic mutation in a gene encoding an SBP-box transcription factor inhibits tomato fruit ripening. Nat. Genet. 38, 948–952 (2006).
- Song, Q., Zhang, T., Stelly, D. M. & Chen, Z. J. Epigenomic and functional analyses reveal roles of epialleles in the loss of photoperiod sensitivity during domestication of allotetraploid cottons. Genome Biol. 18, 99 (2017).
- Pilu, R. et al. A paramutation phenomenon is involved in the genetics of maize low phytic acid1-241 (lpa1-241) trait. Heredity 102, 236–245 (2009).
- Kawanabe, T. et al. Role of DNA methylation in hybrid vigor in Arabidopsis thaliana. Proc. Natl Acad. Sci. USA 113, E6704–E6711 (2016).
- Zhang, Q. et al. The chromatin remodeler DDM1 promotes hybrid vigor by regulating salicylic acid metabolism. Cell Discov. 2, 16027 (2016).
- Brachi, B., Morris, G. P. & Borevitz, J. O. Genome-wide association studies in plants: the missing heritability is in the field. Genome Biol. 12, 232 (2011).
- Kawakatsu, T. et al. Epigenomic diversity in a global collection of Arabidopsis thaliana accessions. Cell 166, 492–505 (2016).
- Kawakatsu, T. et al. Epigenomic diversity in a global collection of Arabidopsis thaliana accessions. Cell 166, 492–505 (2016).
- Eichten, S. R. et al. Epigenetic and genetic influences on DNA methylation variation in maize populations. Plant Cell 25, 2783–2797 (2013).
- Xu, G. et al. Evolutionary and functional genomics of DNA methylation in maize domestication and improvement. Nat. Commun. 11, 5539 (2020).
- Shen, Y. et al. DNA methylation footprints during soybean domestication and improvement. Genome Biol. 19, 128 (2018).
- Zhao, L. et al. Integrative analysis of reference epigenomes in 20 rice varieties. Nat. Commun. 11, 2658 (2020).
- Gardiner, L. J. et al. Hidden variation in polyploid wheat drives local adaptation. Genome Res. 28, 1319–1332 (2018).
- Xu, J. et al. Population-level analysis reveals the widespread occurrence and phenotypic consequence of DNA methylation variation not tagged by genetic variation in maize. Genome Biol. 20, 243 (2019).
- Schmitz, R. J. et al. Patterns of population epigenomic diversity. Nature 495, 193–198 (2013).
- Gohlke J, Scholz CJ, Kneitz S, Weber D, Fuchs J, Hedrich R, Deeken R (2013) DNA methylation mediated control of gene expression is critical for development of crown gall tumors. PLoS Genet 9:e1003267.
- Gouil, Q. & Baulcombe, D. C. Paramutation-like features of multiple natural epialleles in tomato. BMC Genomics 19, 203 (2018).
- Cao, S. et al. Small RNAs mediate transgenerational inheritance of genome-wide trans-acting epialleles in maize. Genome Biol. 23, 53 (2022).
- Cortijo, S. et al. Mapping the epigenetic basis of complex traits. Science 343, 1145–1148 (2014).
- Zhang, Y. Y., Latzel, V., Fischer, M. & Bossdorf, O. Understanding the evolutionary potential of epigenetic variation: a comparison of heritable phenotypic variation in epiRILs, RILs, and natural ecotypes of Arabidopsis thaliana. Heredity 121, 257–265 (2018).
- Schmidt, M. et al. Methylome and epialleles in rice epilines selected for energy use efficiency. Agronomy 8, 163 (2018).
- Hauben, M. et al. Energy use efficiency is characterized by an epigenetic component that can be directed through artificial selection to increase yield. Proc. Natl Acad. Sci. USA 106, 20109–20114 (2009).
- Zhan, X., Lu, Y., Zhu, J. K. & Botella, J. R. Genome editing for plant research and crop improvement. J. Integr. Plant Biol. 63, 3–33 (2021).
- Pan, C. et al. Boosting plant genome editing with a versatile CRISPR-Combo system. Nat. Plants 8, 513–525 (2022).
- Gallego-Bartolome, J. et al. Targeted DNA demethylation of the Arabidopsis genome using the human TET1 catalytic domain. Proc. Natl Acad. Sci. USA 115, E2125–E2134 (2018).
- Ji, L. et al. TET-mediated epimutagenesis of the Arabidopsis thaliana methylome. Nat. Commun. 9, 895 (2018).
- Papikian, A., Liu, W., Gallego-Bartolome, J. & Jacobsen, S. E. Site-specific manipulation of Arabidopsis loci using CRISPR-Cas9 SunTag systems. Nat. Commun. 10, 729 (2019).
- Ghoshal, B., Picard, C. L., Vong, B., Feng, S. & Jacobsen, S. E. CRISPR-based targeting of DNA methylation in Arabidopsis thaliana by a bacterial CG-specific DNA methyltransferase. Proc. Natl Acad. Sci. USA 118, e2125016118 (2021).
- Galonska, C. et al. Genome-wide tracking of dCas9-methyltransferase footprints. Nat. Commun. 9, 597 (2018).
- Pflueger, C. et al. A modular dCas9-SunTag DNMT3A epigenome editing system overcomes pervasive off-target activity of direct fusion dCas9-DNMT3A constructs. Genome Res. 28, 1193–1206 (2018).
- Chan, W. F. et al. Activation of stably silenced genes by recruitment of a synthetic de-methylating module. Nat. Commun. 13, 5582 (2022).
- Swain, T. et al. A modular dCas9-based recruitment platform for combinatorial epigenome editing. Nucleic Acids Res. 52, 474–491 (2024).
| Arabidopsis mutants | Pathogen | Phenotype | Defense response | References |
|---|---|---|---|---|
| DNA Hypomethylation | ||||
| drd1 | P. syringae pv. tomato DC3000 (Pst) | Resistant | Enhancement of SA-dependent defense | Dowen et al. [152] |
| P. cucumerina | Susceptible | Suppression of JA-dependent defense | López et al. [154] | |
| ago4 | B. cinerea | Susceptible | Suppression of JA-dependent defense | López et al. [154] |
| rdr2 | P. cucumerina | Susceptible | Suppression of JA-dependent defense | López et al. [154] |
| Pst | Resistant | Enhancement of SA-dependent defense | Dowen et al. [152] | |
| rdr6 | B. cinerea | Susceptible | Loss of transfer siRNAs that target pathogen genes | Cai et al. [146] |
| Pst | Resistant | − | Dowen et al. [152] | |
| nrpd1 | Pst | Resistant | Enhancement of SA-dependent defense | Dowen et al. [152] |
| nrpe1 | P. cucumerina | Susceptible | Suppression of JA-dependent defense | López et al. [154] |
| B. cinerea | Susceptible | Suppression of JA-dependent defense | López et al. [154] | |
| Pst | Resistant | Enhancement of SA-dependent defense | López et al. [154] | |
| nrpd2 | P. cucumerina | Susceptible | Suppression of JA-dependent defense | López et al. [154] |
| B. cinerea | Susceptible | Suppression of JA-dependent defense | López et al. [154] | |
| Pst | Resistant | Enhancement of SA-dependent defense | López et al. [154] | |
| nrpd1/nrpe1 | P. cucumerina | Susceptible | Suppression of JA-dependent defense | López et al. [154] |
| Pst | Resistant | Enhancement of SA-dependent defense | López et al. [154] | |
| drm1/drm2 | P. cucumerina | Susceptible | Suppression of JA-dependent defense | López et al. [154] |
| Pst | Resistant | Enhancement of SA-dependent defensePrimed state of defense response | Yu et al. [142] | |
| CaLCuV | Susceptible | − | Raja et al. [80] | |
| BCTV | Susceptible | − | Raja et al. [80] | |
| drm1/drm2/cmt3 (ddc) | A. tumefaciens | Susceptible | Enhancement of ABA-dependent response | Gohlke et al. [176] |
| Pst | Resistant | Enhancement of SA-dependent defense | Dowen et al. [152] | |
| dcl2/3/4 | B. cinerea | Susceptible | Loss of siRNAs that move into fungal cells and suppress virulence genes | Cai et al. [146] |
| Pst | Resistant | Enhancement of SA-dependent defense | Dowen et al. [152] | |
| CaLCuV | Susceptible | − | Raja et al. [80] | |
| BCTV | Susceptible | − | Raja et al. [80] | |
| DNA Hypermethylation | ||||
| ros1 | Pst | Susceptible | Methylation at the promoter of RMG1 and RLP43 | Yu et al. [142] |
| ros1/dml2/dml3 (rdd) | F. oxysporum | Susceptible | Suppression of defense-related genes | Le et al. [160] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




