Submitted:
04 June 2024
Posted:
10 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
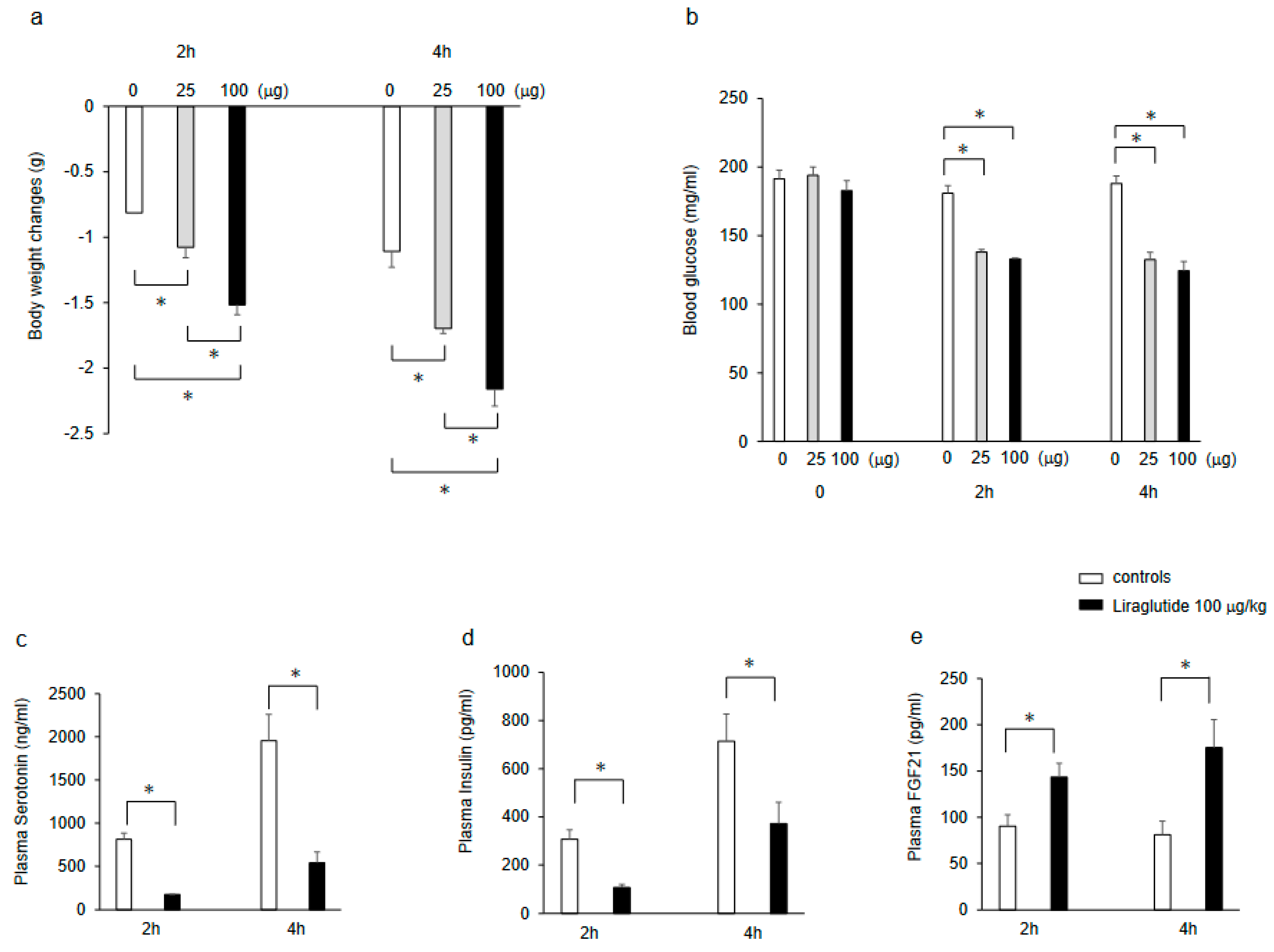
2.1. Effects of Liraglutide on Body Weight, Blood Glucose, Plasma Insulin, Serotonin, FGF21 Levels in Mice Deprived of Food after Intraperitonal Administration of the Drug
2.2. Effects of Liraglutide on Tryptophan Metabolites in the Colon of Mice
2.3. Effects of Liraglutide on Bile Acids in the Feces and Liver of Mice
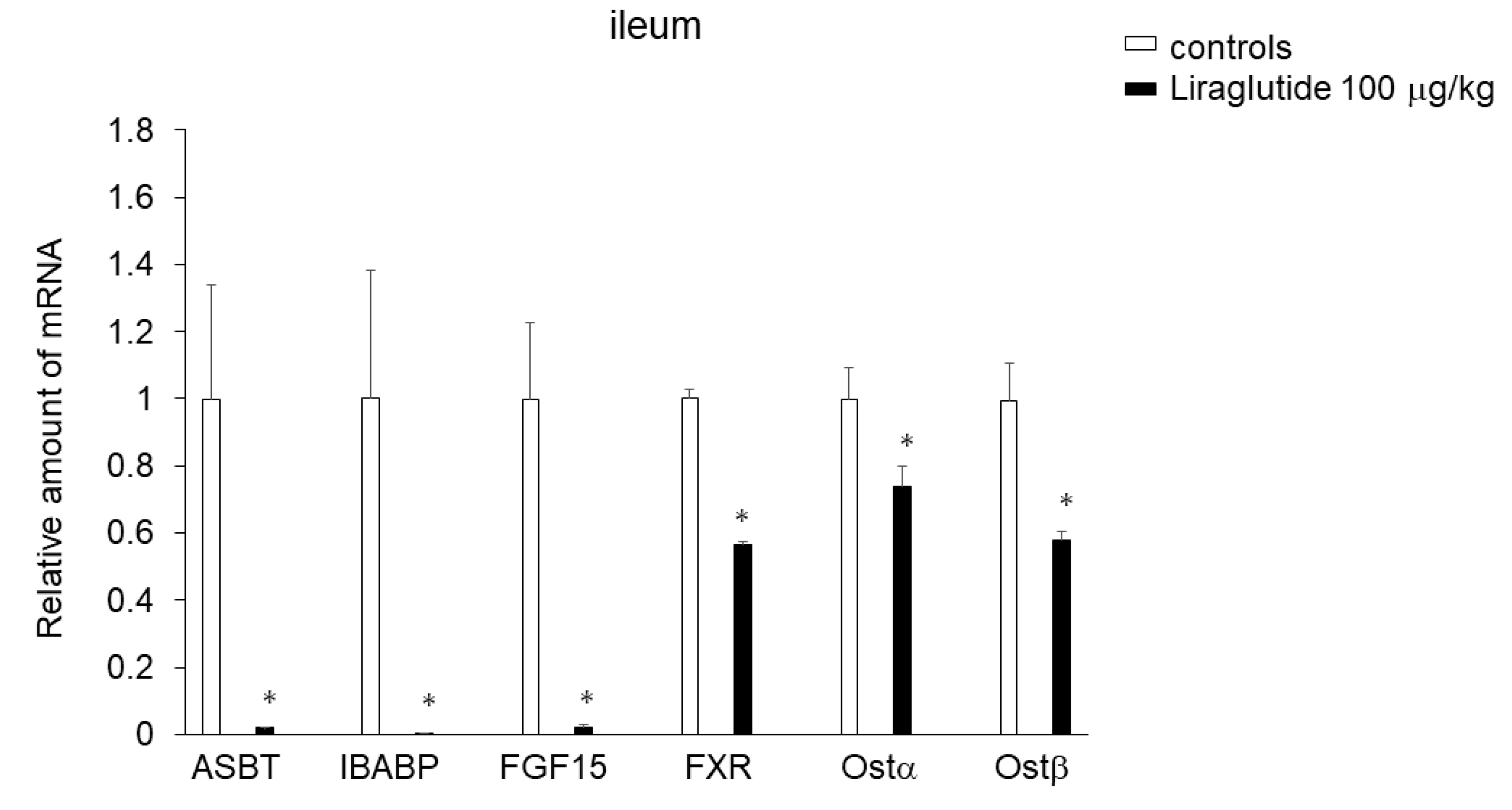
2.4. Effects of Liraglutide on Expression of Genes Involved in BA Transport in the Ileum
3. Discussion
4. Materials and Methods
4.1. General Procedures
4.2. Blood Chemistry.
4.3. BA Analysis
4.4. Tryptophan and Its Metabolites Analysis
4.5. Real-Time Quantitative Reverse Transcription–Polymerase Chain Reaction (RT–PCR)
4.6. Statistical Methods
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Campbell, J.E.; Drucker, D.J. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metab. 2013, 17, 819–837. [Google Scholar] [CrossRef] [PubMed]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Meier, J.J. GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Mol. Metab. 2021, 46, 101102. [Google Scholar] [CrossRef] [PubMed]
- Popoviciu, M.S.; Păduraru, L.; Yahya, G.; Metwally, K.; Cavalu, S. Emerging Role of GLP-1 Agonists in Obesity: A Comprehensive Review of Randomised Controlled Trials. Int. J. Mol. Sci. 2023, 24, 10449. [Google Scholar] [CrossRef] [PubMed]
- Kårhus, M.L.; Brønden, A.; Forman, J.L.; Haaber, A.; Knudsen, E.; Langholz, E.; Dragsted, L.O.; Hansen, S.H.; Krakauer, M.; Vilsbøll, T.; et al. Safety and efficacy of liraglutide versus colesevelam for the treatment of bile acid diarrhoea: a randomised, double-blind, active-comparator, non-inferiority clinical trial. Lancet Gastroenterol. Hepatol. 2022, 7, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Nonogaki, K. The Regulatory Role of the Central and Peripheral Serotonin Network on Feeding Signals in Metabolic Diseases. Int. J. Mol. Sci. 2022, 23, 1600. [Google Scholar] [CrossRef] [PubMed]
- Koopman, N.; Katsavelis, D.; Hove, A.S.T.; Brul, S.; Jonge, W.J.; Seppen, J. The Multifaceted Role of Serotonin in Intestinal Homeostasis. Int. J. Mol. Sci. 2021, 22, 9487. [Google Scholar] [CrossRef] [PubMed]
- Chojnacki, J.; Konrad, P.; Mędrek-Socha, M.; Kaczka, A.; Błońska, A.; Zajdel, R.; Chojnacki, C.; Gasiorowska, A. The Variability of Tryptophan Metabolism in Patients with Mixed Type of Irritable Bowel Syndrome. Int. J. Mol. Sci. 2024, 25, 2550. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, J.; Wu, X.; Jing, H.; Zhang, S.; Hu, Z.; Rao, L.; Chang, Q.; Wang, L.; Zhang, Z. Gut microbiota alteration after cholecystectomy contributes to post-cholecystectomy diarrhea via bile acids stimulating colonic serotonin. Gut. Microbes 2023, 15, 2168101. [Google Scholar] [CrossRef] [PubMed]
- Martin, A., M.; Yabut, J.M.; Choo, J.M.; Page, A.J.; Sun, E.W.; Jessup, C.F.; Wesselingh, S.L.; Khan, W.I.; Rogers, G.B.; Steinberg, G.R.; et al. The gut microbiome regulates host glucose homeostasis via peripheral serotonin. Proc. Natl. Acad. Sci. USA. 2019, 116, 19802–19804. [Google Scholar] [CrossRef]
- Borup, C.; Vinter-Jensen, L.; Jørgensen, S.P.G.; Wildt, S.; Graff, J.; Gregersen, T.; Zaremba, A.; Borup Andersen, T.; Nøjgaard, C.; Timm, H.B.; et al. Efficacy and safety of colesevelam for the treatment of bile acid diarrhoea: a double-blind, randomised, placebo-controlled, phase 4 clinical trial. Lancet Gastroenterol. Hepatol. 2023, 8, 321–331. [Google Scholar] [CrossRef]
- Lund, M.L.; Egerod, K.L.; Engelstoft, M.S.; Dmytriyeva, O.; Theodorsson, E.; Patel, B.A.; Schwartz, T.W. Enterochromaffin 5-HT cells - A major target for GLP-1 and gut microbial metabolites. Mol. Metab. 2018, 11, 70–83. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, H.; Kolodziejczyk, A.A.; Halstuch, D.; Elinav, E.J. Bile acids in glucose metabolism in health and diseases. Exp. Med. 2018, 215, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Lundåsen, T.; Andersson, E.-M.; Snaith, M.; Lindmark, H.; Lundberg, J.; Östlund-Lindqvist, A.-M. Inhibition of intestinal bile acid transporter Slc10a2 improves triglyceride metabolism and normalizes elevated plasma glucose levels in mice. PLoS One 2012, 7, e37787. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.; Sonne, D.P.; Mikkelsen, K.H.; Gluud, L.L.; Vilsbøll, T.; Knop, F.K. Bile acid sequestrants for glycemia control in patients with type 2 diabetes: A systematic review with meta-analysis of randomized controlled trials. J. Diabetes Complications 2017, 31, 918–927. [Google Scholar] [PubMed]
- Ge, M.-X.; Niu, W.-X.; Ren, J.-F.; Cai, S.-Y.; Yu, D.-K.; Liu, H.-T.; Zhang, N.; Zhang, Y.X.; Wang, Y.C.; Shao, R.G.; et al. A novel ASBT inhibitor, IMB17-15, repressed nonalcoholic fatty liver disease development in high-fat diet-fed Syrian golden hamsters. Acta Pharmacol. Sin. 2019, 40, 895–907. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.; Haywood, J.; Craddock, A.L.; Belinsky, M.G.; Kruh, G.D.; Dawson, P.A. The organic solute transporter alpha-beta, Ostalpha-Ostbeta, is essential for intestinal bile acid transport and homeostasis. Proc. Natl. Acad. Sci. USA. 2008, 105, 3891–3896. [Google Scholar] [CrossRef]
- Wikoff, W.R.; Anfora, A.T.; Liu, J.; Schultz, P.G.; Lesley, S.A.; Peters, E.C.; Siuzdak, G. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc. Natl. Acad. Sci. USA. 2009, 106, 3698–3703. [Google Scholar] [CrossRef]
- Tuomainen, M.; Lindström, J.; Lehtonen, M.; Auriola, S.; Pihlajamäki, J.; Peltonen, M.; Tuomilehto. J.; Uusitupa, M.; de Mello, V.D.; Hanhineva, K. Associations of serum indolepropionic acid, a gut microbiota metabolite, with type 2 diabetes and low-grade inflammation in high-risk individuals. Nutr. Diabetes 2018, 8, 35. [Google Scholar] [CrossRef]
- de Mello, V.D.; Paananen, J.; Lindström, J.; Lankinen, M.A.; Shi, L.; Kuusisto, J.; Pihlajamäki, J.; Auriola, S.; Lehtonen, M.; Rolandsson, O.; et al. Indolepropionic acid and novel lipid metabolites are associated with a lower risk of type 2 diabetes in the Finnish Diabetes Prevention Study. Sci. Rep. 2017, 7, 46337. [Google Scholar] [CrossRef]
- Chen, L.; Yang, Y.; Sun, S.; Xie, Y.; Pan, C.; Li, M.; Li, C.; Liu, Y.; Xu, Z.; Liu, W.; et al. Indolepropionic acid reduces obesity-induced metabolic dysfunction through colonic barrier restoration mediated via tuft cell-derived IL-25. FEBS J. 2022, 289, 5985–6004. [Google Scholar] [CrossRef]
- Nonogaki, K.; Suzuki, M.; Sanuki, M.; Wakameda, M.; Tamari, T. The contribution of serotonin 5 HT2C and melanocortin-4 receptors to the satiety signaling of glucagon-like peptide 1 and liraglutide, a glucagon-like peptide 1 receptor agonist, in mice. Biochem. Biophysic. Res. Communi. 2011, 411, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Nonogaki, K.; Kaji, T. The acute anorexic effect of liraglutide, a GLP-1 receptor agonist, does not require functional leptin receptor, serotonin, and hypothalamic POMC and CART activities in mice. Diabetes Res. Clin. Practice 2016, 120, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Nonogaki, K.; Kaji, T. Whey protein isolate inhibits hepatic FGF21 production, which precedes weight gain, hyperinsulinemia and hyperglycemia in mice fed a high-fat diet. Sci. Rep. 2020, 10, 15784. [Google Scholar] [CrossRef]
- Nonogaki, K.; Kaji, T. Ingestion of whey protein and β-conglycinin exerts opposite effects on intestinal FGF15 and serotonin secretion in mice. Frontier Endocrinol. 2023, 14, 1080790. [Google Scholar] [CrossRef] [PubMed]
| (ng/g) | Control | Liraglutide | p-Value |
|---|---|---|---|
| Trp | 15620±4608.09 | 3027±220.4 | P<0.05 |
| 5-HT | 1367±120.7 | 385±13.66 | P<0.05 |
| 5-HIAA | 462±66.0 | 266±5.42 | P<0.05 |
| KYN | 311±56.93 | 46±0.82 | P<0.05 |
| XA | 417±100.68 | 72.1±7.72 | P<0.05 |
| IPA | 29.9±1.91 | 652.5±119.94 | P<0.05 |
| (nmol/g) | Control | Liraglutide | p-Value |
|---|---|---|---|
| CA | 291.18±68.59 | 16.53±1.79 | P<0.05 |
| TCA | 70.65±23.95 | 5.42±2.3 | P<0.05 |
| TCDCA | 2.55±0.75 | 0.39±0.12 | P<0.05 |
| GCA | 0.57±0.11 | 0.07±0.01 | P<0.05 |
| αMCA | 37.13±11.25 | 21.48±4.21 | NS |
| βMCA | 176.93±62.51 | 70.60±17.04 | P<0.05 |
| DCA | 382.08±108.4 | 407.5±72.23 | NS |
| UDCA | 28.16±9.10 | 31.6±8.73 | NS |
| TDCA | 4.54±1.50 | 1.3795±0.41 | P<0.05 |
| LCA | 11.88±4.37 | 20.89±3.89 | NS |
| GDCA | 0.27±0.06 | 0.37±0.10 | NS |
| TBA | 1008.78±155.75 | 439.81±87.28 | P<0.05 |
| (nmol/g) | Control | Liraglutide | p-Value |
|---|---|---|---|
| CA | 2.72±0.14 | 1.45±0.41 | P<0.05 |
| TCA | 131.57±23.57 | 39.55±0.47 | P<0.05 |
| TCDCA | 2.36±0.15 | 2.01±0.46 | NS |
| GCA | 0.18±0.03 | 0.16±0.05 | NS |
| CDCA | 0.31±0.02 | 0.24±0.05 | NS |
| αMCA | 0.91±0.12 | 0.78±0.18 | NS |
| βMCA | 5.24±0.26 | 3.57±0.19 | P<0.05 |
| DCA | 0.21±0.004 | 0.20±0.01 | NS |
| UDCA | 0.25±0.02 | 0.18±0.08 | P<0.05 |
| TDCA | 10.28±0.99 | 8.57±1.82 | NS |
| LCA | 0.06±0.02 | 0.08±0.06 | NS |
| TBA | 154.1±23.46 | 43.54±6.57 | P<0.05 |
| GENES | Sense | Antisense |
|---|---|---|
| FGF15 | ACGGGCTGATTCGCTACTC | TGTAGCCTAAACAGTCCATTTCCT |
| FXR | CCCCTGCTTGATGTGCTAC | CGTGGTGATGGTTGAATGTC |
| ASBT | TGGGTTTCTTCCTGGCTAGACT | TGTTCTGCATTCCAGTTTCCAA |
| IBABP | CAGGAGACGTGATTGAAAGGG | GCCCCCAGAGTAAGACTGGG |
| Ostα | TACAAGAACACCCTTTGCCC | CGAGGAATCCAGAGACCAAA |
| Ostβ | GTATTTTCGTGCAGAAGATGCG | TTTCTGTTTGCCAGGATGCTC |
| β-actin | TTGTAACCAACTGGGACGATATGG | GATCTTGATCTTCATGGTGCTAGG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




