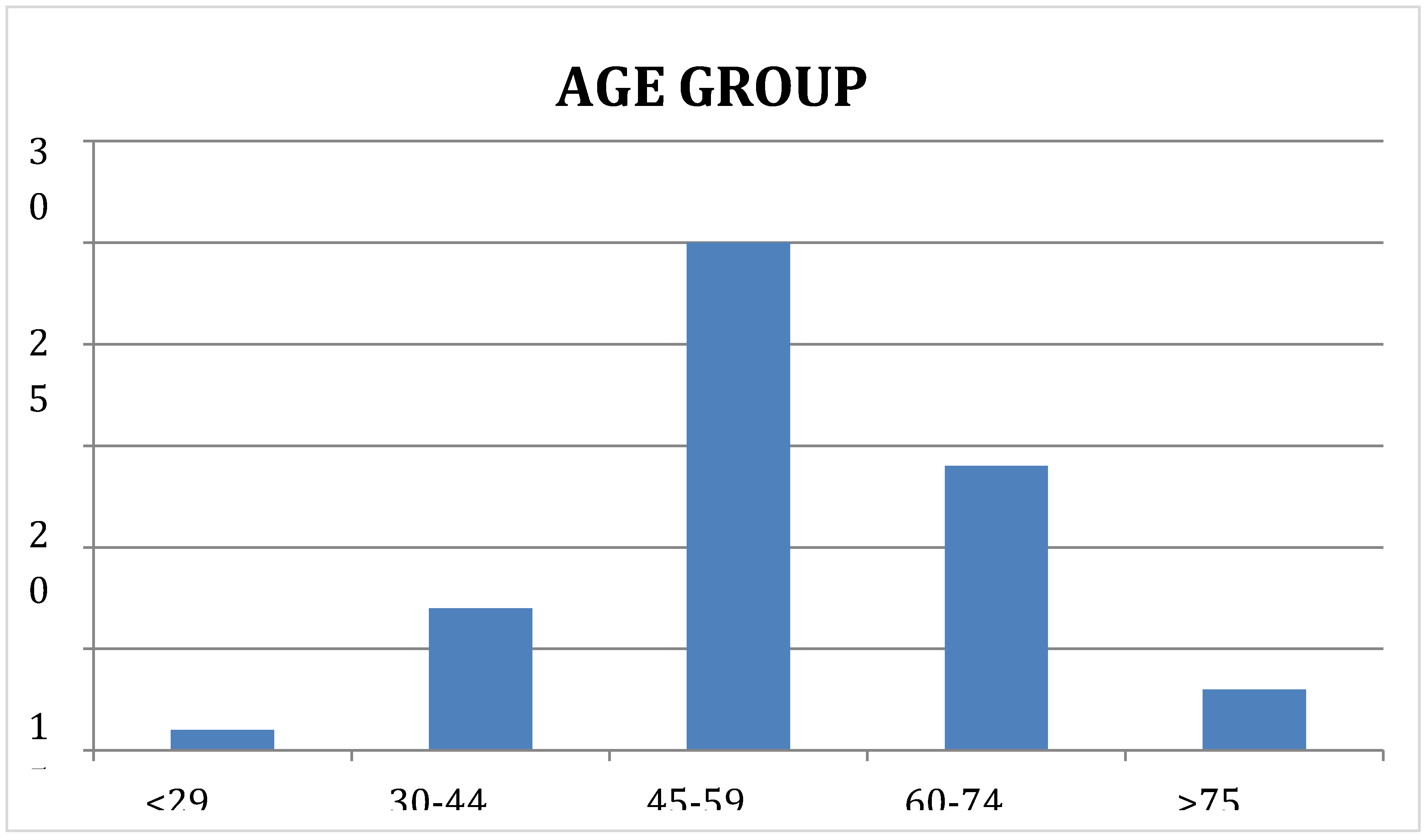4. Discussion
Several publications were found to be relevant to the topic of obesity and iron deficiency anemia. For instance, a review article by Zhao et al. (2015) reported a quantitative meta- analysis that showed a significant association between obesity and iron deficiency. Another study by Chowdhury et al. (2018) found a high prevalence of overweight and obesity among women of reproductive age in Bangladesh with iron deficiency anemia. Furthermore, a two- sample Mendelian randomization study by Chang et al. (2019) provided evidence of a potential causal association between obesity and iron deficiency anemia. Results of our study were similar to the results of Zimmermann et al. (2008), who studied the relationship between obesity and iron deficiency among healthy premenopausal women. They found that 22% of the women were considered overweight, and 20% were iron deficient. They measured the iron status indicators (hemoglobin and serum ferritin) and C-reactive protein (CRP) at pre- meal and two hours post-meal [
13,
14,
15,
16].
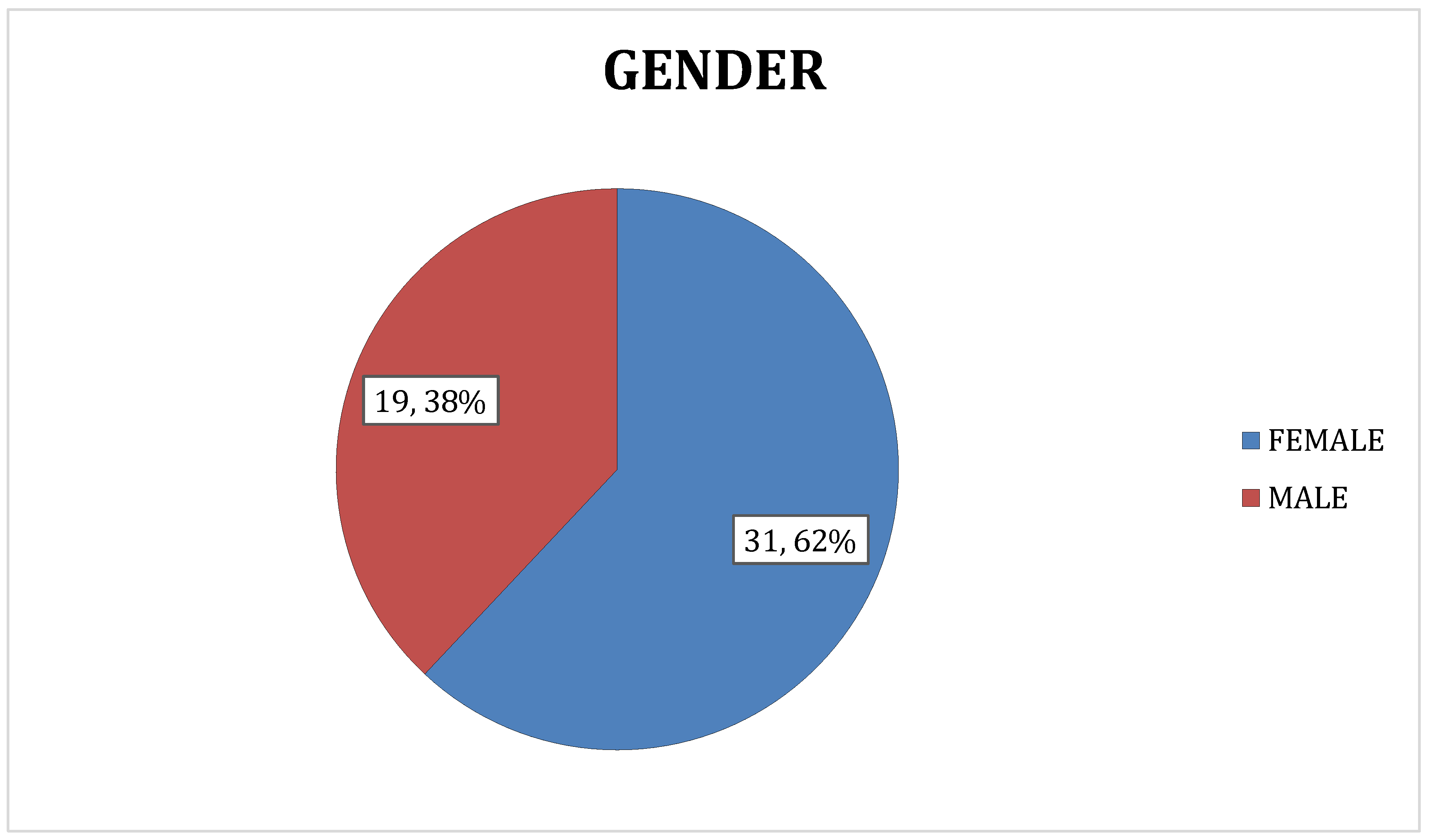
Most of the subjects in our study were middle to old aged, in the age group 45 to 59 years (n = 25, 50%). 14 subjects (28%) were between 60 to 74 years, 7 (14%) were between 30 and 44 years, 3 were older than 75 years and 1 (2%) was below the age of 30 years. The Median (IQR) was 55.0 (46.0 - 64.0) and the Mean age was 54.54 ± 11.2. Our study found that females constituted 62% of the study population (n = 31), while males represented only 38% of the study population (n = 19) Results are supported by the studies of Tanwi, Chakrabarty, & Hasanuzzaman, 2019, Adib Rad et al., 2019; Fongar, Godecke, & Qaim, 2019; Keding, 2016; Neymotin & Sen, 2011 [
17,
18,
19,
20,
21,
22].
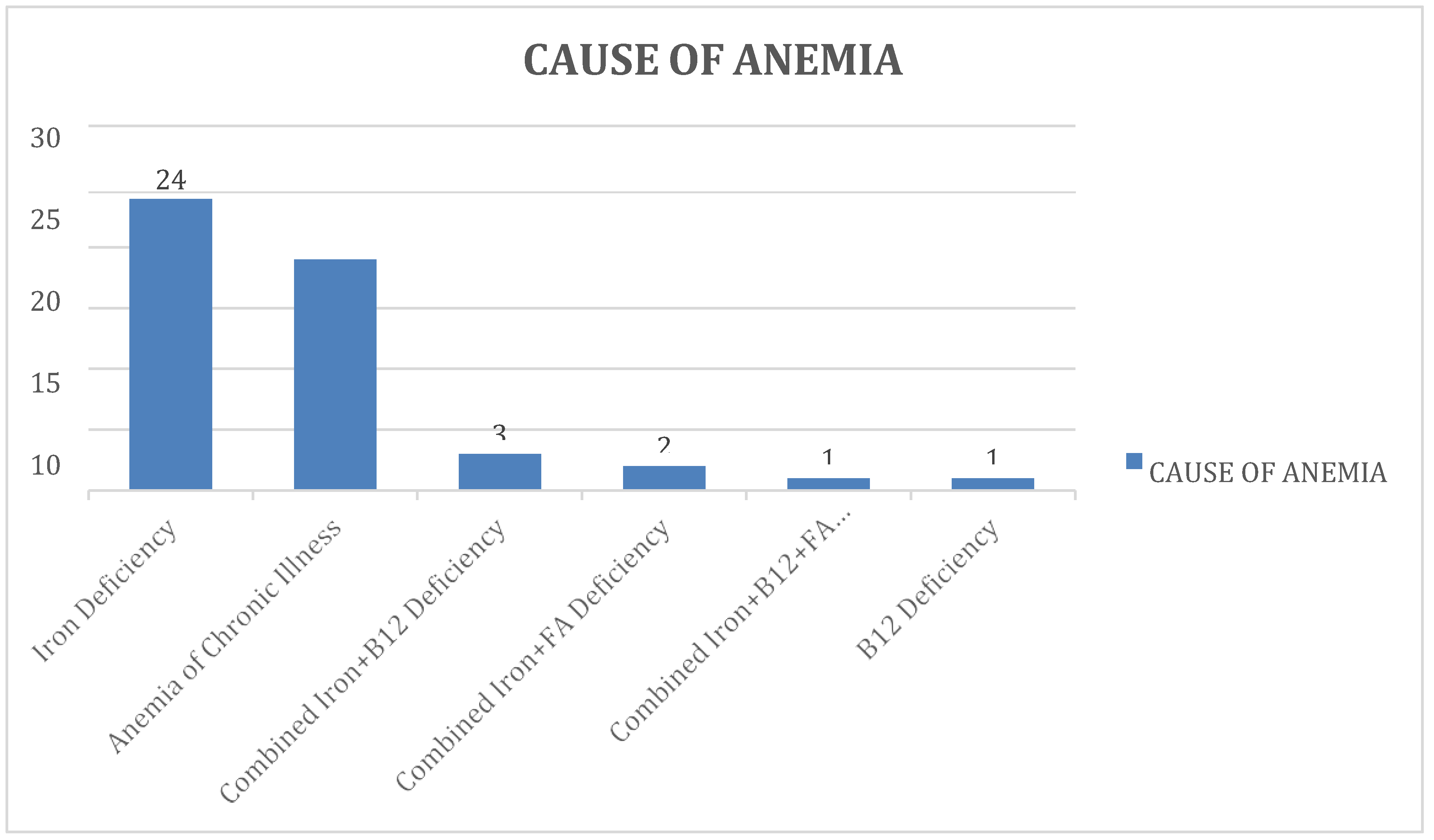
In our study, we found that iron deficiency was the most common etiology of anemia, present in 24 (48%) of subjects. Nutritional deficiencies were the most common cause of anemia, present in 31 (62%) of the subjects, while the remaining 19 subjects (38%) were found to have anemia of chronic illness. We used one essential criterion (hemoglobin less than 12 gm/dL in
females and less than 13 gm/dL in males) and three additional criteria (serum iron levels, serum ferritin levels, and TIBC) to diagnose iron deficiency anemia. If two criteria out of three were present, then it was labeled as iron deficiency anemia. Our study also found that 3 subjects (6%) had a combined deficiency of Iron and Vitamin B12, while another 2 subjects (4%) were found to have a combined iron and folate deficiency. One subject (2%) had an isolated Vitamin B12 deficiency, and the only remaining subject was found to have a combined deficiency of all three micronutrients - iron, folate, and vitamin B12. The study’s results were similar to those of other studies, such as Wang T, et al. (2023), which found evidence of a potential causal association between obesity and iron deficiency anemia, and Aguree and Reddy (2021), which reported elevated levels of inflammatory markers and hepcidin in obese women of reproductive age, which may contribute to iron deficiency anemia [
23,
24].
In our study, we found that the hemoglobin (g/dL) value ranged from a minimum of 5.6 to a maximum of 10.5, with a median (IQR) of 8.65 (7.8 - 9.4) and a mean ± standard deviation of 8.53 ± 1.23. The MCV (fL) values ranged from 53.0 through 99.0, with a median (IQR) of 78.0 (72.0 - 86.6) and a mean ± standard deviation of 78.75 ± 10.13. The average value of ESR (mm/Hour) was 20.56 ± 21.47, with a median (IQR) of 17.0 (12.0 - 23.0), a minimum value of 4.0, and a maximum of 155.0. Of the 50 subjects in the study, 20 (40%) had a CRP level of >5 mg/dL, with an average CRP of 4.71 ± 3.32 mg/dL and a median (IQR) of 4.0 (2.0 – 8.0) mg/dL. The results of our study were similar to the results of other studies, such as Zhao et al. (2015) and Aigner et al. (2014), Till and Grundman (2011), Chowdhuryb et al. (2018), Ahirwar, R., & Mondal, P. R. (2019) [
13,
14,
25,
26,
27].
Results of our study were similar to the findings of previous research. The serum iron levels in our study ranged from 10.0 to 65.0 µg/dL, with an average of 21.56 ± 12.22 µg/dL. This aligns with the importance of measuring serum ferritin levels for diagnosing iron deficiency anemia. The ferritin values in our study ranged from 8.03 to 516.0 ng/mL, with an average of 70.03 ± 109.59 ng/mL, reflecting the iron stores in the body. Additionally, the TIBC values ranged from 108.0 to 595.0 µg/dL, with an average of 333.66 ± 102.92 µg/dL, which is commonly used in evaluating anemia cases. The transferrin saturation levels in our study ranged from 1.68 to 27.43%, with an average of 7.31 ± 5.25%, reflecting the iron bound to transferrin. Furthermore, the B12 levels in our study ranged from 150.0 to 1000.0 pg/mL, with a median of 694.0 (IQR: 345.0 - 807.0) and a mean ± standard deviation of 606.54 ± 275.80 pg/mL. The folate levels averaged at 12.09 ± 5.23 ng/mL, with a median of 10.34 (IQR: 7.90 - 16.5) and values ranging from 3.06 to 20.0 ng/mL. Lastly, the hepcidin levels ranged from 240.15 to 1637.18, with a mean value of 825.36 ± 326.39. These results are in consensus with the many previous studies of Kumar A et al. (2022), Johnson-Wimbley TD & Graham (DY 2011) [
26,
28].
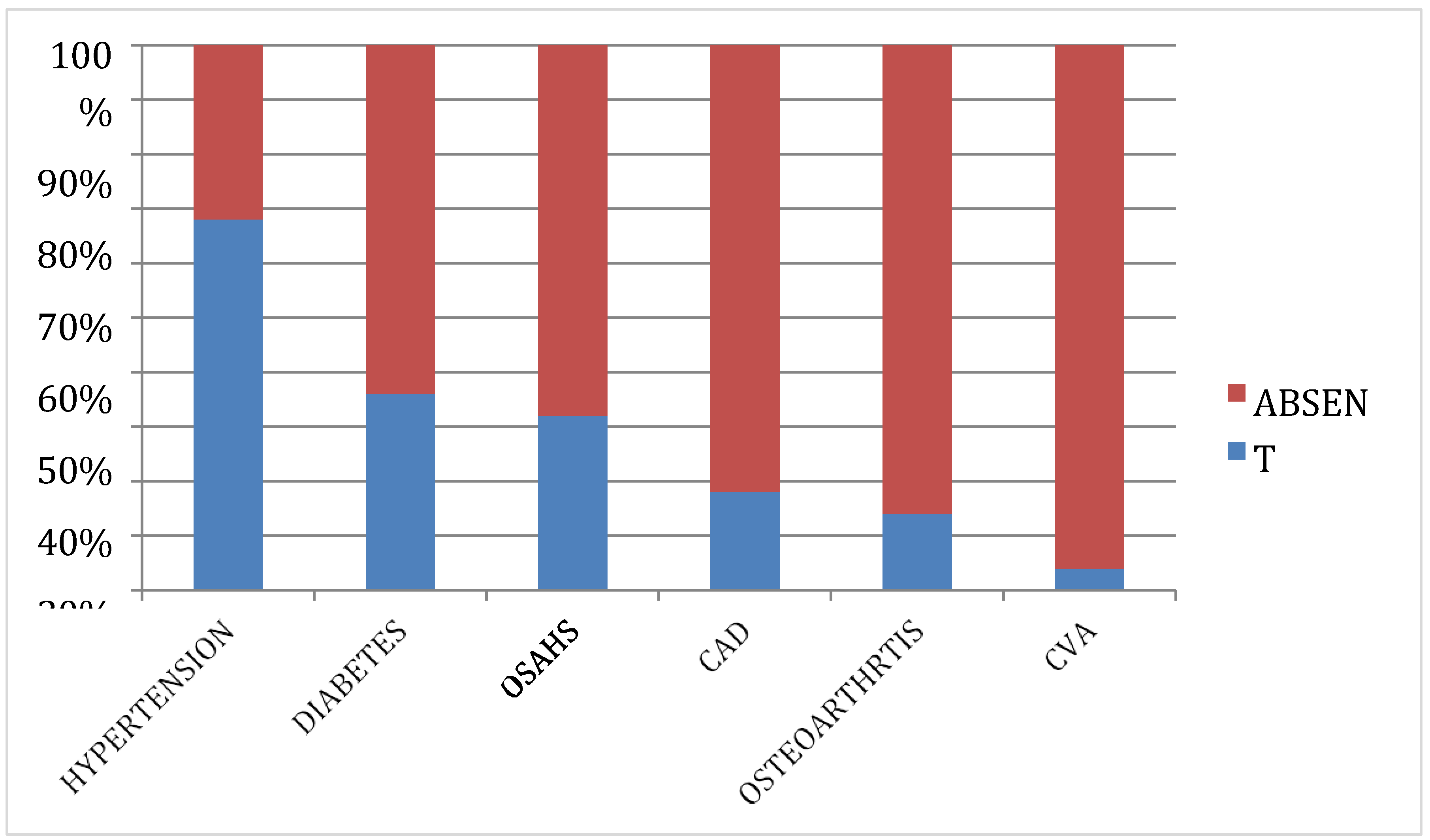
In our study we observed that among the 50 subjects, hypertension was the most common comorbidity, with 68.0% diagnosed as hypertensive and taking antihypertensive medicines. Diabetes was the second most common comorbidity, affecting 36% of the subjects. Additionally, 32% suffered from Obstructive Sleep Apnea Hypoventilation Syndrome (OSAHS), 18% had a history of coronary artery disease, 14% had osteoarthritis of the knees, and 4% had a past history of cerebrovascular accidents. Our results align with those of Safiri et al. (2021), who also reported a high prevalence of hypertension in their study. Further results align with the work of Zheng et al. (2021), emphasizing the importance of addressing OSAHS in overweight and obese individuals. Köseoğlu, F.D.; Özlek, B (2024) findings mirror those of Anemia and Iron Deficiency Predict All-Cause Mortality in Patients with Coronary artery disease, emphasizing the impact of comorbidities on mortality [
29,
30,
31].
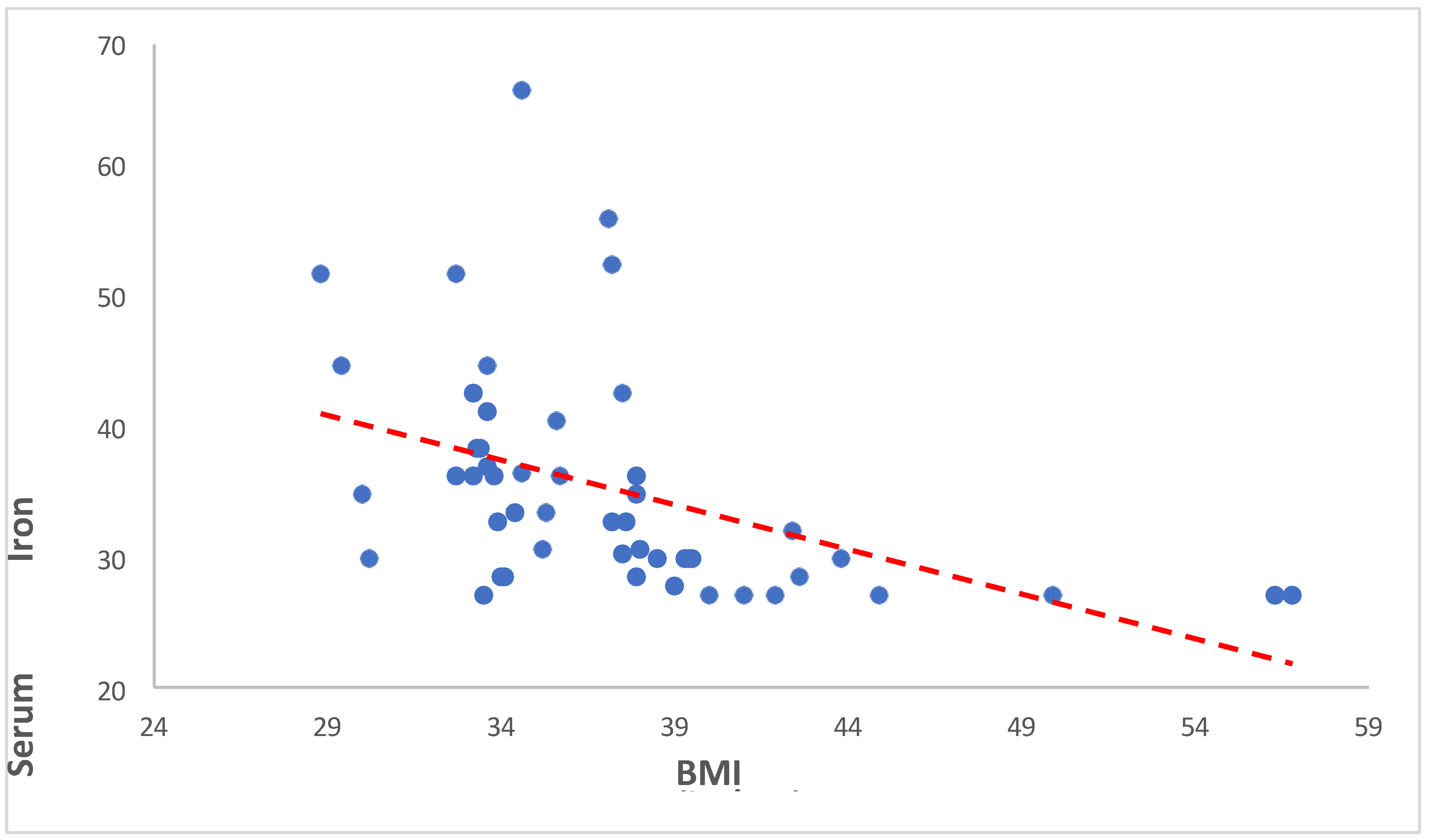
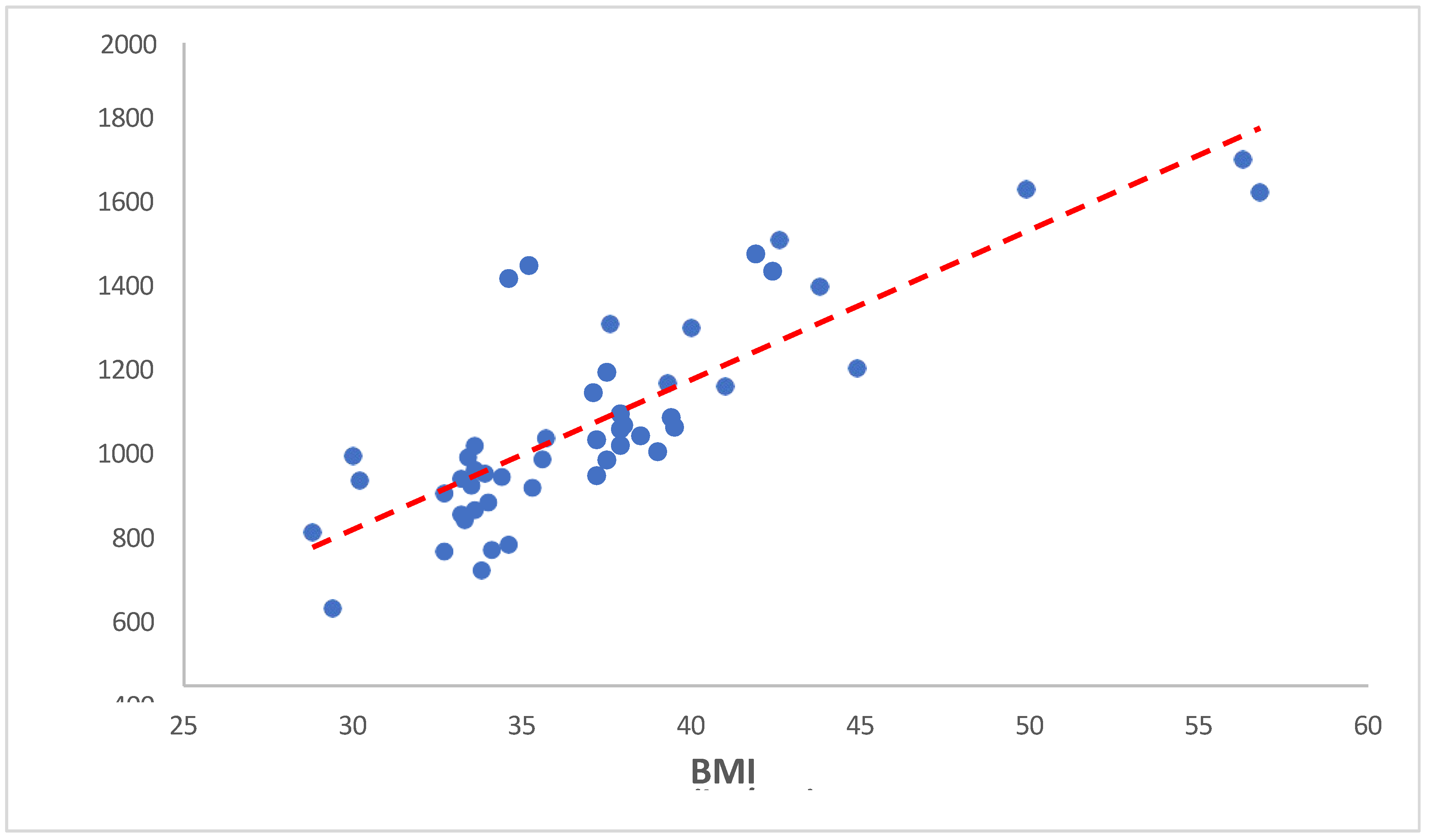
In our study, we investigated the relationship between BMI (Body Mass Index) and serum iron levels among anemic overweight and obese patients. Our findings revealed a significant negative correlation between BMI and serum iron levels (Spearman’s rho = -0.645, p < 0.001). This indicates that as BMI increases, serum iron levels tend to decrease. Such a robust negative correlation suggests a potentially important physiological relationship between adiposity and iron metabolism. Cepeda-Lopez and Baye (2020) discussed the complex relationship between obesity, iron deficiency, and anemia. They emphasized that obesity, characterized by chronic low-grade inflammation, can disrupt iron homeostasis and lead to systemic iron deficiency [
32].
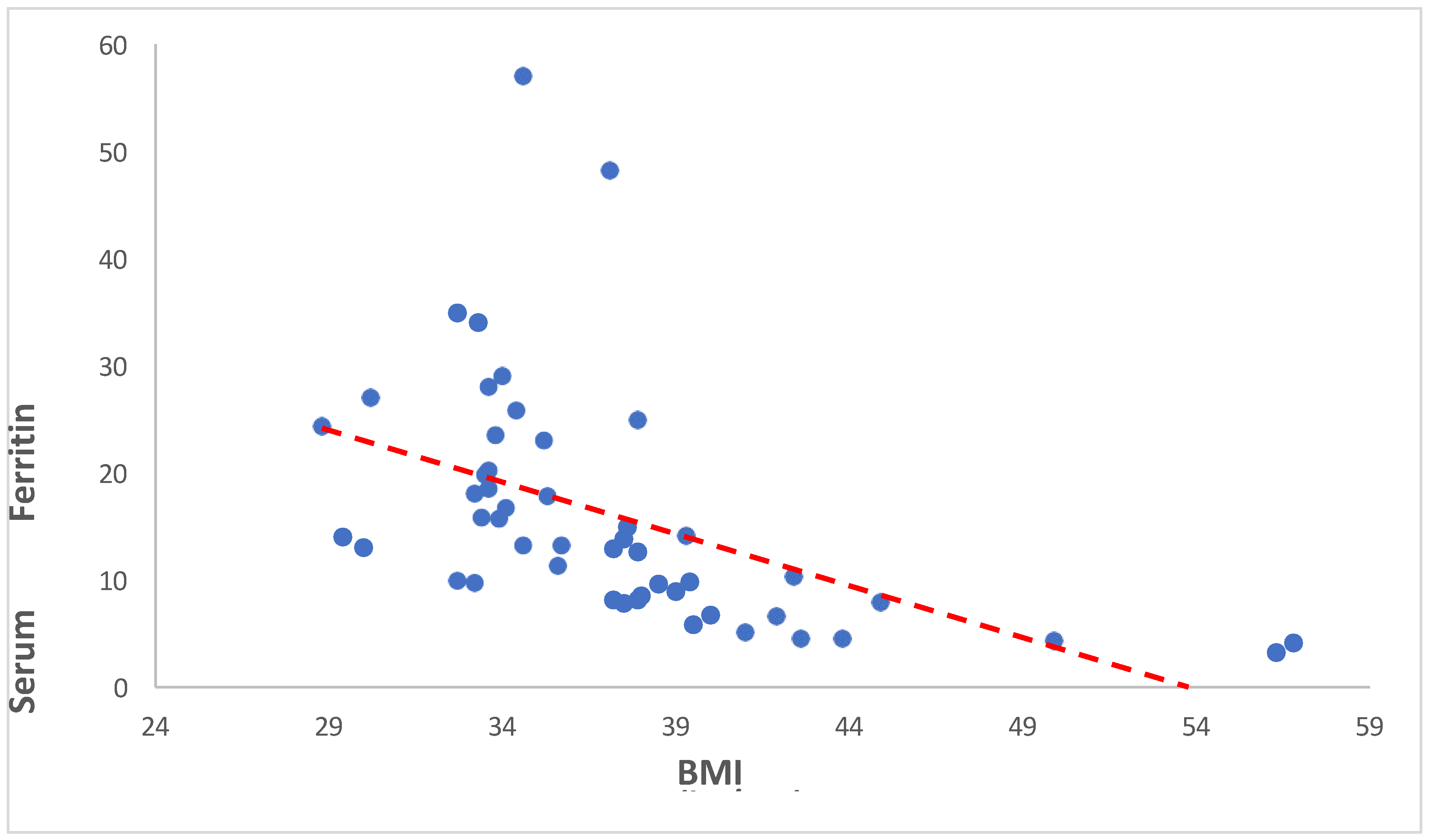
Our study shows the significant negative correlation between BMI and serum ferritin levels (Spearman’s rho = -0.723, p < 0.001). This indicates that as BMI increases, serum ferritin levels tend to decrease. Similarly study of Alam F et al., 2015 reveals that individuals with high BMI had high levels of Serum Ferritin despite low levels of iron with high levels of C- reactive protein. This might be caused due to inflammatory conditions prevailing in the presence of increased adipose tissue [
33].
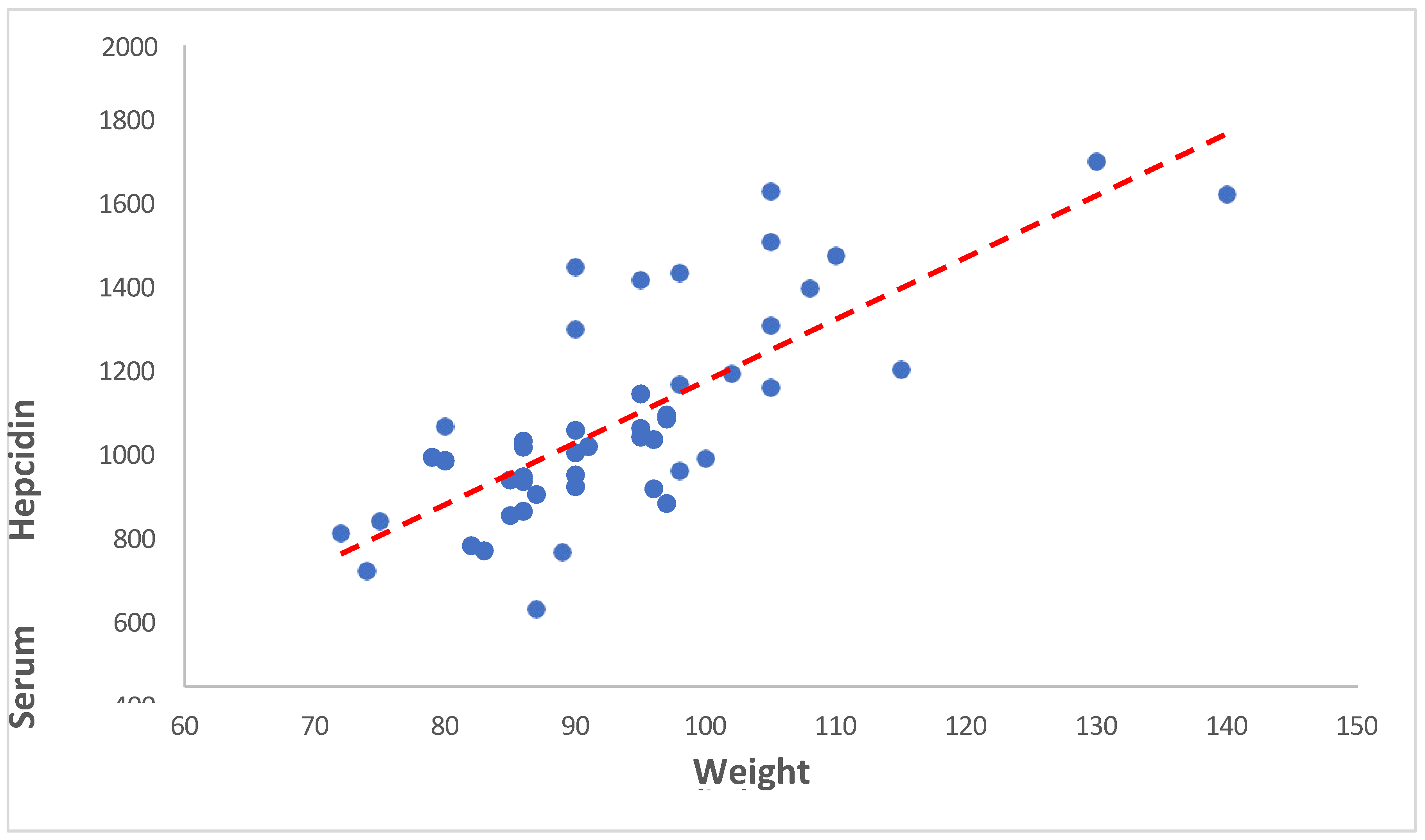
Our study found a significant positive correlation between weight and serum hepcidin concentrations (correlation coefficient = 0.728, p < 0.001). This suggests that as weight increases, serum hepcidin levels also tend to increase. The potential mechanisms underlying the correlation between weight and serum hepcidin concentrations in the study are not fully understood. However, studies have shown that hepcidin levels are disproportionately influenced by weight, with overweight patients with type 2 diabetes (T2D) having significantly elevated hepcidin levels compared to those with grade I obesity who have reduced levels. The sources of heterogeneity in the included studies remained high, and most studies did not control for confounders such as inflammation and obesity, which have an impact on hepcidin levels. Additionally, the quality of cumulative evidence in the study was low due to the observational nature of the included studies. Serum hepcidin levels have also been associated with obesity, with a high correlation between serum hepcidin and serum ferritin concentration in small samples of healthy persons Ndevahoma F et al., 2021, Vuppalanchi R et al., 2014 [
34,
35].
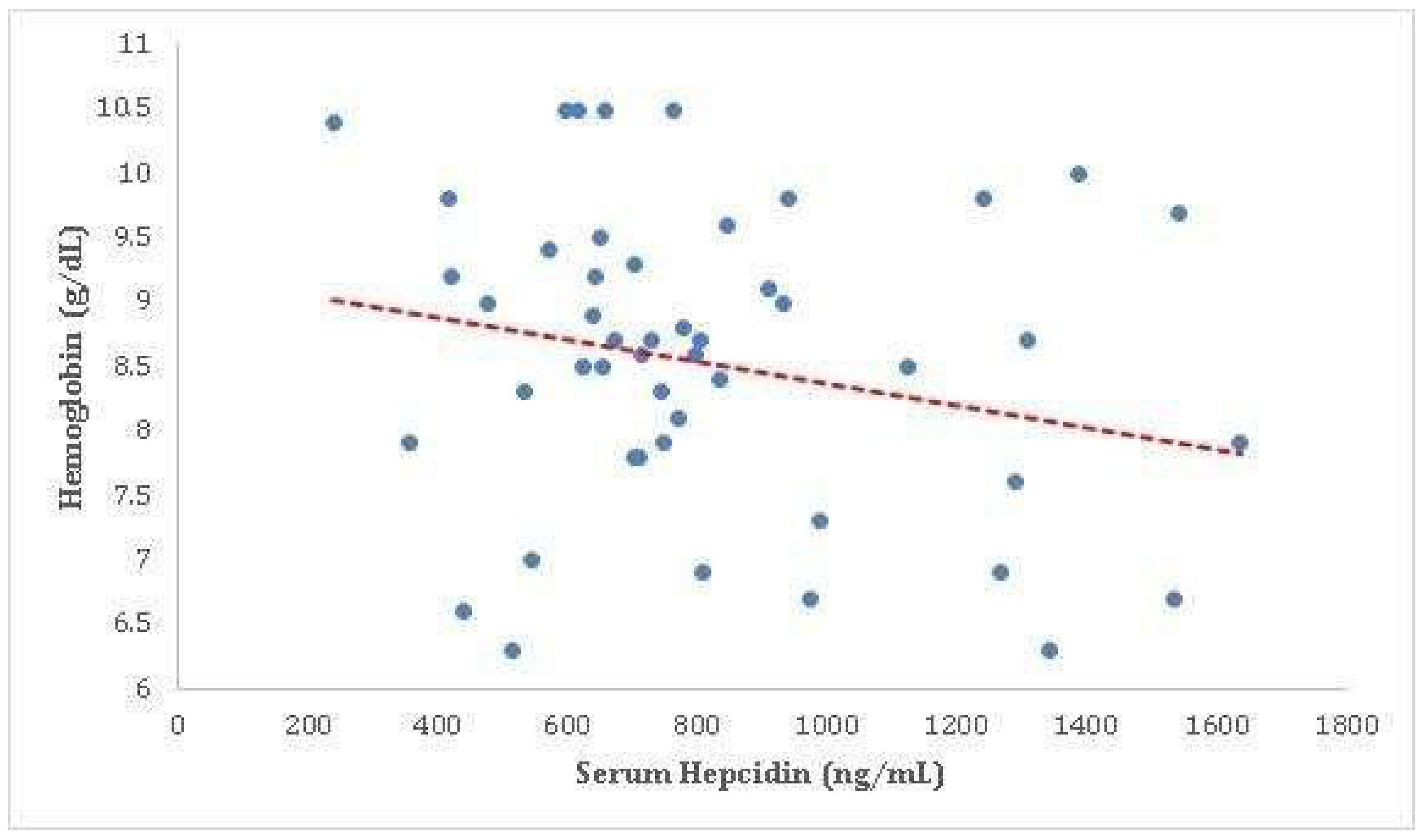
Our study found a weak negative correlation between serum hepcidin concentrations and serum hemoglobin levels (correlation coefficient = -0.203), although this did not reach statistical significance (p = 0.158). This suggests a trend towards a negative association between these two variables, meaning that as serum hepcidin levels increase, serum hemoglobin levels tend to decrease, and vice versa. Hemoglobin is a protein in your red blood cells that carries oxygen to your body’s organs and tissues and transports carbon dioxide from your organs and tissues back to your lungs. On the other hand, hepcidin is a hormone produced by the liver that regulates iron balance in the body. It inhibits the release of iron from cells, leading to decreased iron levels in the blood. The negative correlation between hemoglobin and hepcidin could suggest that as the body produces more hemoglobin (and therefore requires more iron), it may decrease the production of hepcidin to allow more iron to be released from cells. However, this correlation was not statistically significant in your study, indicating that the observed relationship could be due to chance. It’s important to note that this is a complex area and other factors could also be influencing the relationship between hemoglobin and hepcidin levels. Further research would be needed to fully understand the implications of these findings Ganz, T. (2003), Nemeth, E., & Ganz, T. (2006) and Tussing-Humphreys et al., (2012) [
36,
37,
38].