Submitted:
25 March 2024
Posted:
26 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. What Are Lipopolysaccharides?
3. LPS in Food and Diets
3.1. Cooking and LPS
3.2. LPS in Milk
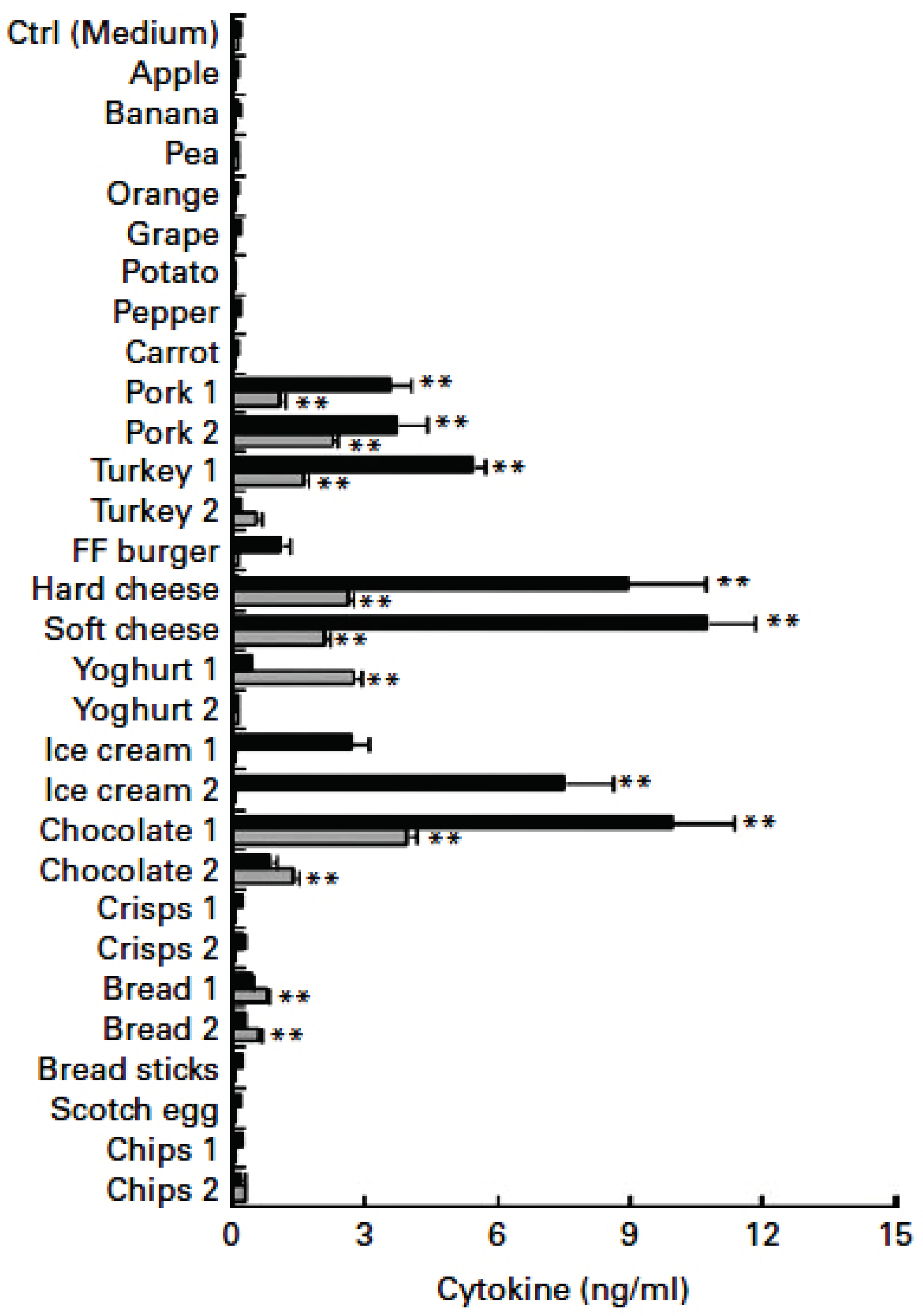
3.3. LPS in Meat and Processed Foods
3.4. LPS in Diets
3.5. LPS after a Test Meal
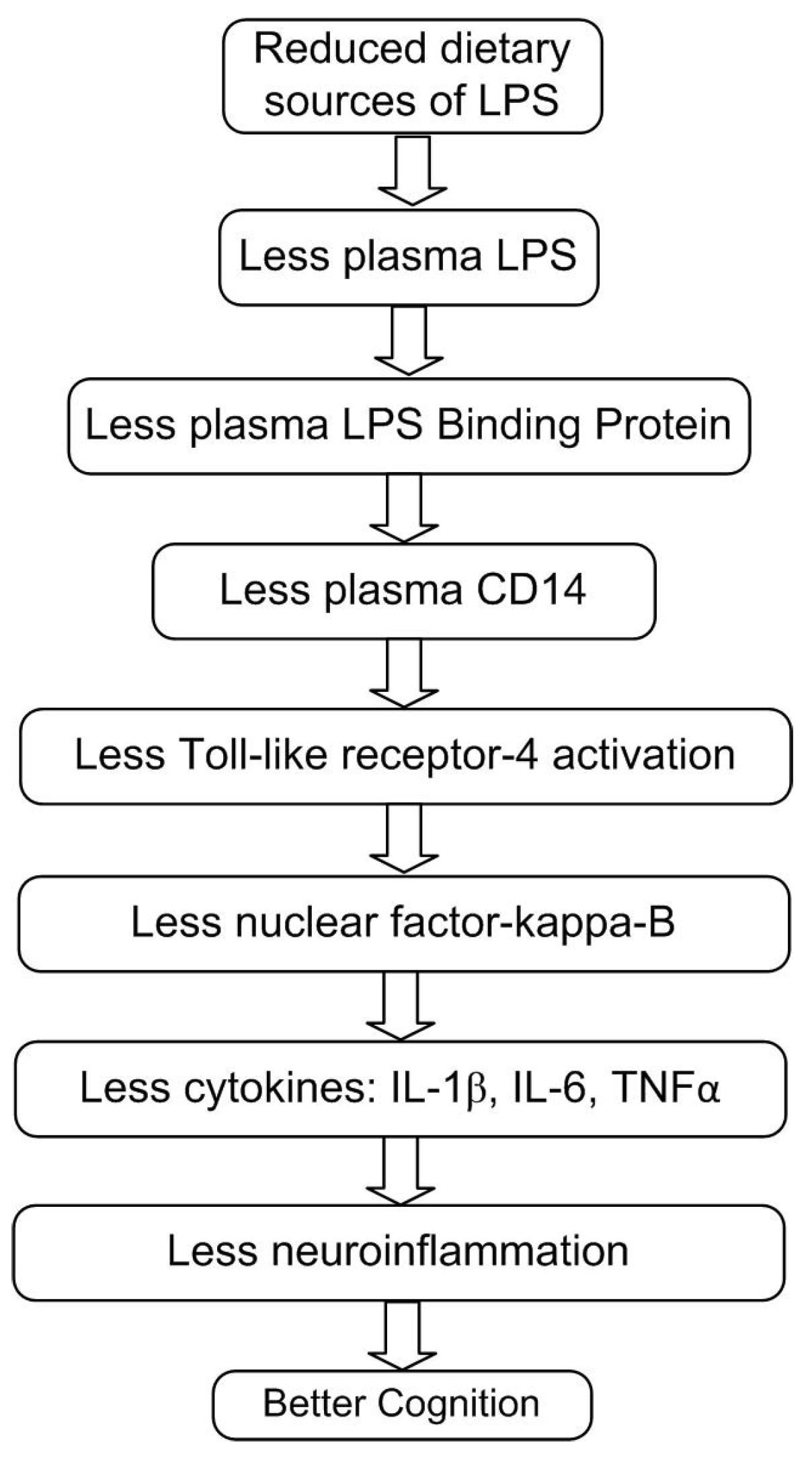
4. Dietary LPS Enters the Bloodstream and Triggers Inflammation
4.1. Colonic Sources of Plasma LPS
4.2. Small Intestine Microbiota Versus LPS in Food
4.3. Transport of LPS into the Blood by Saturated Fats and Chylomicrons
4.4. High-Fat Diets Can Increase Endotoxemia
5. Association of High Plasma LPS with AD and Related Dementias
5.1. LPS Increases Risk of Cognitive Decline and AD
5.2. LPS Is Elevated in AD
5.3. LPS Stimulates Brain Inflammation
5.4. LPS, Neuroinflammation, and Amyloid
5.5. Saturated Fatty Acids Can Raise Serum LPS and Impact Cognition
5.6. Injecting LPS Impairs Cognition
5.7. Injecting LPS Increases Neuroinflammation
6. Association of High Cytokine Levels with AD and Cognitive Impairment
6.1. Excess Inflammation Increases Cognitive Decline
6.2. Increased IL-6 and Activities of Daily Living
6.3. Autopsy Findings of Higher Cytokines in AD
6.4. Cytokine Therapy Can Impair Cognition
7. LPS Can Represent a Risk Factor for Atherosclerosis and Reduced Brain Perfusion
7.1. Brain Perfusion
7.2. Atheroslerosis, LPS, and LBP
7.3. BBB Damage and Cytokines
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Dementia. Availabe online: https://www.who.int/news-room/fact-sheets/detail/dementia (accessed on March 17, 2024).
- Rohr, M.W.; Narasimhulu, C.A.; Rudeski-Rohr, T.A.; Parthasarathy, S. Negative Effects of a High-Fat Diet on Intestinal Permeability: A Review. Adv Nutr 2020, 11, 77-91. [CrossRef]
- Laugerette, F.; Vors, C.; Peretti, N.; Michalski, M.C. Complex links between dietary lipids, endogenous endotoxins and metabolic inflammation. Biochimie 2011, 93, 39-45. [CrossRef]
- Saji, N.; Saito, Y.; Yamashita, T.; Murotani, K.; Tsuduki, T.; Hisada, T.; Sugimoto, T.; Niida, S.; Toba, K.; Sakurai, T. Relationship Between Plasma Lipopolysaccharides, Gut Microbiota, and Dementia: A Cross-Sectional Study. J Alzheimers Dis 2022, 86, 1947-1957. [CrossRef]
- Vors, C.; Drai, J.; Pineau, G.; Laville, M.; Vidal, H.; Laugerette, F.; Michalski, M.C. Emulsifying dietary fat modulates postprandial endotoxemia associated with chylomicronemia in obese men: a pilot randomized crossover study. Lipids Health Dis 2017, 16, 97. [CrossRef]
- Brown, G.C. The endotoxin hypothesis of neurodegeneration. J Neuroinflammation 2019, 16, 180. [CrossRef]
- Andre, P.; Laugerette, F.; Feart, C. Metabolic Endotoxemia: A Potential Underlying Mechanism of the Relationship between Dietary Fat Intake and Risk for Cognitive Impairments in Humans? Nutrients 2019, 11. [CrossRef]
- Grant, W.B.; Blake, S.M. Diet’s Role in Modifying Risk of Alzheimer’s Disease: History and Present Understanding. J Alzheimers Dis 2023, 96, 1353-1382. [CrossRef]
- Andreasen, A.S.; Krabbe, K.S.; Krogh-Madsen, R.; Taudorf, S.; Pedersen, B.K.; Moller, K. Human endotoxemia as a model of systemic inflammation. Curr Med Chem 2008, 15, 1697-1705. [CrossRef]
- Wu, H.; Wang, Y.; Li, H.; Meng, L.; Zheng, N.; Wang, J. Effect of Food Endotoxin on Infant Health. Toxins (Basel) 2021, 13. [CrossRef]
- Erridge, C. Accumulation of stimulants of Toll-like receptor (TLR)-2 and TLR4 in meat products stored at 5 degrees C. J Food Sci 2011, 76, H72-79. [CrossRef]
- Jay, J.M.; Margitic, S.; Shereda, A.L.; Covington, H.V. Determining endotoxin content of ground beef by the Limulus amoebocyte lysate test as a rapid indicator of microbial quality. Appl Environ Microbiol 1979, 38, 885-890. [CrossRef]
- Gehring, U.; Spithoven, J.; Schmid, S.; Bitter, S.; Braun-Fahrlander, C.; Dalphin, J.C.; Hyvarinen, A.; Pekkanen, J.; Riedler, J.; Weiland, S.K. et al. Endotoxin levels in cow’s milk samples from farming and non-farming families - the PASTURE study. Environ Int 2008, 34, 1132-1136. [CrossRef]
- Venter, P. Endotoxin residues in food: a review. Interim: Interdisciplinary J. 2010, 9, 106-126.
- Ferguson, J.F.; Patel, P.N.; Shah, R.Y.; Mulvey, C.K.; Gadi, R.; Nijjar, P.S.; Usman, H.M.; Mehta, N.N.; Shah, R.; Master, S.R. et al. Race and gender variation in response to evoked inflammation. J Transl Med 2013, 11, 63. [CrossRef]
- Hansen, K.; Mikkelsen, T.; Moller-Madsen, A. Use of the Limulus test to determine the hygienic status of milk products as characterized by levels of gram-negative bacterial lipopolysaccharide present. J Dairy Res 1982, 49, 323-328. [CrossRef]
- Faraj, T.A. Regulation of cardiometabolic risk factors by dietary Toll-like receptor stimulants. PhD, University of Leicester, Leicester, 2017.
- Prester, L.; Orct, T.; Macan, J.; Vukusic, J.; Kipcic, D. Determination of biogenic amines and endotoxin in squid, musky octopus, Norway lobster, and mussel stored at room temperature. Arh Hig Rada Toksikol 2010, 61, 389-397. [CrossRef]
- Herieka, M.; Faraj, T.A.; Erridge, C. Reduced dietary intake of pro-inflammatory Toll-like receptor stimulants favourably modifies markers of cardiometabolic risk in healthy men. Nutr Metab Cardiovasc Dis 2016, 26, 194-200. [CrossRef]
- Brown, B.I. Nutritional Management of Metabolic Endotoxemia: A Clinical Review. Altern Ther Health Med 2017, 23, 42-54.
- Pastori, D.; Carnevale, R.; Nocella, C.; Novo, M.; Santulli, M.; Cammisotto, V.; Menichelli, D.; Pignatelli, P.; Violi, F. Gut-Derived Serum Lipopolysaccharide is Associated With Enhanced Risk of Major Adverse Cardiovascular Events in Atrial Fibrillation: Effect of Adherence to Mediterranean Diet. J Am Heart Assoc 2017, 6. [CrossRef]
- Estadella, D.; da Penha Oller do Nascimento, C.M.; Oyama, L.M.; Ribeiro, E.B.; Damaso, A.R.; de Piano, A. Lipotoxicity: effects of dietary saturated and transfatty acids. Mediators Inflamm 2013, 2013, 137579. [CrossRef]
- Pendyala, S.; Walker, J.M.; Holt, P.R. A high-fat diet is associated with endotoxemia that originates from the gut. Gastroenterology 2012, 142, 1100-1101 e1102. [CrossRef]
- Mo, Z.; Huang, S.; Burnett, D.J.; Rutledge, J.C.; Hwang, D.H. Endotoxin May Not Be the Major Cause of Postprandial Inflammation in Adults Who Consume a Single High-Fat or Moderately High-Fat Meal. J Nutr 2020, 150, 1303-1312. [CrossRef]
- Meessen, E.C.E.; Warmbrunn, M.V.; Nieuwdorp, M.; Soeters, M.R. Human Postprandial Nutrient Metabolism and Low-Grade Inflammation: A Narrative Review. Nutrients 2019, 11. [CrossRef]
- Ahola, A.J.; Lassenius, M.I.; Forsblom, C.; Harjutsalo, V.; Lehto, M.; Groop, P.H. Dietary patterns reflecting healthy food choices are associated with lower serum LPS activity. Sci Rep 2017, 7, 6511. [CrossRef]
- Andre, P.; Pais de Barros, J.P.; Mj Merle, B.; Samieri, C.; Helmer, C.; Delcourt, C.; Feart, C. Mediterranean diet and prudent diet are both associated with low circulating esterified 3-hydroxy fatty acids, a proxy of LPS burden, among older adults. Am J Clin Nutr 2021, 114, 1080-1091. [CrossRef]
- Ghanim, H.; Abuaysheh, S.; Sia, C.L.; Korzeniewski, K.; Chaudhuri, A.; Fernandez-Real, J.M.; Dandona, P. Increase in plasma endotoxin concentrations and the expression of Toll-like receptors and suppressor of cytokine signaling-3 in mononuclear cells after a high-fat, high-carbohydrate meal: implications for insulin resistance. Diabetes Care 2009, 32, 2281-2287. [CrossRef]
- Szczechowiak, K.; Diniz, B.S.; Leszek, J. Diet and Alzheimer’s dementia - Nutritional approach to modulate inflammation. Pharmacol Biochem Behav 2019, 184, 172743. [CrossRef]
- Henning, A.L.; Venable, A.S.; Vingren, J.L.; Hill, D.W.; McFarlin, B.K. Consumption of a high-fat meal was associated with an increase in monocyte adhesion molecules, scavenger receptors, and Propensity to Form Foam Cells. Cytometry B Clin Cytom 2018, 94, 606-612. [CrossRef]
- Hawkesworth, S.; Moore, S.E.; Fulford, A.J.; Barclay, G.R.; Darboe, A.A.; Mark, H.; Nyan, O.A.; Prentice, A.M. Evidence for metabolic endotoxemia in obese and diabetic Gambian women. Nutr Diabetes 2013, 3, e83. [CrossRef]
- Al-Disi, D.A.; Al-Daghri, N.M.; Khan, N.; Alfadda, A.A.; Sallam, R.M.; Alsaif, M.; Sabico, S.; Tripathi, G.; McTernan, P.G. Postprandial Effect of a High-Fat Meal on Endotoxemia in Arab Women with and without Insulin-Resistance-Related Diseases. Nutrients 2015, 7, 6375-6389. [CrossRef]
- Gonzalez, F.; Considine, R.V.; Abdelhadi, O.A.; Acton, A.J. Saturated Fat Ingestion Promotes Lipopolysaccharide-Mediated Inflammation and Insulin Resistance in Polycystic Ovary Syndrome. J Clin Endocrinol Metab 2019, 104, 934-946. [CrossRef]
- Vors, C.; Pineau, G.; Drai, J.; Meugnier, E.; Pesenti, S.; Laville, M.; Laugerette, F.; Malpuech-Brugere, C.; Vidal, H.; Michalski, M.C. Postprandial Endotoxemia Linked With Chylomicrons and Lipopolysaccharides Handling in Obese Versus Lean Men: A Lipid Dose-Effect Trial. J Clin Endocrinol Metab 2015, 100, 3427-3435. [CrossRef]
- Harte, A.L.; Varma, M.C.; Tripathi, G.; McGee, K.C.; Al-Daghri, N.M.; Al-Attas, O.S.; Sabico, S.; O’Hare, J.P.; Ceriello, A.; Saravanan, P. et al. High fat intake leads to acute postprandial exposure to circulating endotoxin in type 2 diabetic subjects. Diabetes Care 2012, 35, 375-382. [CrossRef]
- Lyte, J.M.; Gabler, N.K.; Hollis, J.H. Postprandial serum endotoxin in healthy humans is modulated by dietary fat in a randomized, controlled, cross-over study. Lipids Health Dis 2016, 15, 186. [CrossRef]
- Laugerette, F.; Alligier, M.; Bastard, J.P.; Drai, J.; Chanseaume, E.; Lambert-Porcheron, S.; Laville, M.; Morio, B.; Vidal, H.; Michalski, M.C. Overfeeding increases postprandial endotoxemia in men: Inflammatory outcome may depend on LPS transporters LBP and sCD14. Mol Nutr Food Res 2014, 58, 1513-1518. [CrossRef]
- White, A.J.; Wijeyekoon, R.S.; Scott, K.M.; Gunawardana, N.P.; Hayat, S.; Solim, I.H.; McMahon, H.T.; Barker, R.A.; Williams-Gray, C.H. The Peripheral Inflammatory Response to Alpha-Synuclein and Endotoxin in Parkinson’s Disease. Front Neurol 2018, 9, 946. [CrossRef]
- Netto Candido, T.L.; Bressan, J.; Alfenas, R.C.G. Dysbiosis and metabolic endotoxemia induced by high-fat diet. Nutr Hosp 2018, 35, 1432-1440. [CrossRef]
- Erridge, C. Diet, commensals and the intestine as sources of pathogen-associated molecular patterns in atherosclerosis, type 2 diabetes and non-alcoholic fatty liver disease. Atherosclerosis 2011, 216, 1-6. [CrossRef]
- Bowser, S.M.; McMillan, R.P.; Boutagy, N.E.; Tarpey, M.D.; Smithson, A.T.; Osterberg, K.L.; Neilson, A.P.; Davy, B.M.; Davy, K.P.; Hulver, M.W. Serum endotoxin, gut permeability and skeletal muscle metabolic adaptations following a short term high fat diet in humans. Metabolism 2020, 103, 154041. [CrossRef]
- Wan, Y.; Wang, F.; Yuan, J.; Li, J.; Jiang, D.; Zhang, J.; Li, H.; Wang, R.; Tang, J.; Huang, T. et al. Effects of dietary fat on gut microbiota and faecal metabolites, and their relationship with cardiometabolic risk factors: a 6-month randomised controlled-feeding trial. Gut 2019, 68, 1417-1429. [CrossRef]
- Michalski, M.C.; Le Barz, M.; vors, C. Metabolic impact of dietary lipids: towards a role of unabsorbed lipid residues? OCL 2021, 28, 9. [CrossRef]
- Lakhan, S.E.; Kirchgessner, A. Gut inflammation in chronic fatigue syndrome. Nutr Metab (Lond) 2010, 7, 79. [CrossRef]
- Burkitt, D.P.; Walker, A.R.; Painter, N.S. Effect of dietary fibre on stools and the transit-times, and its role in the causation of disease. Lancet 1972, 2, 1408-1412. [CrossRef]
- Erridge, C. The capacity of foodstuffs to induce innate immune activation of human monocytes in vitro is dependent on food content of stimulants of Toll-like receptors 2 and 4. Br J Nutr 2011, 105, 15-23. [CrossRef]
- Moreira, A.P.; Texeira, T.F.; Ferreira, A.B.; Peluzio Mdo, C.; Alfenas Rde, C. Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia. Br J Nutr 2012, 108, 801-809. [CrossRef]
- Kastl, A.J. Jr.; Terry, N.A.; Wu, G.D.; Albenberg, L.G. The Structure and Function of the Human Small Intestinal Microbiota: Current Understanding and Future Directions. Cell Mol Gastroenterol Hepatol 2020, 9, 33-45. [CrossRef]
- Reichenberg, A.; Yirmiya, R.; Schuld, A.; Kraus, T.; Haack, M.; Morag, A.; Pollmacher, T. Cytokine-associated emotional and cognitive disturbances in humans. Arch Gen Psychiatry 2001, 58, 445-452. [CrossRef]
- Cohen, O.; Reichenberg, A.; Perry, C.; Ginzberg, D.; Pollmacher, T.; Soreq, H.; Yirmiya, R. Endotoxin-induced changes in human working and declarative memory associate with cleavage of plasma “readthrough” acetylcholinesterase. J Mol Neurosci 2003, 21, 199-212. [CrossRef]
- Ghoshal, S.; Witta, J.; Zhong, J.; de Villiers, W.; Eckhardt, E. Chylomicrons promote intestinal absorption of lipopolysaccharides. J Lipid Res 2009, 50, 90-97. [CrossRef]
- Madison, A.A.; Belury, M.A.; Andridge, R.; Shrout, M.R.; Renna, M.E.; Malarkey, W.B.; Bailey, M.T.; Kiecolt-Glaser, J.K. Afternoon distraction: a high-saturated-fat meal and endotoxemia impact postmeal attention in a randomized crossover trial. Am J Clin Nutr 2020, 111, 1150-1158. [CrossRef]
- Amar, J.; Burcelin, R.; Ruidavets, J.B.; Cani, P.D.; Fauvel, J.; Alessi, M.C.; Chamontin, B.; Ferrieres, J. Energy intake is associated with endotoxemia in apparently healthy men. Am J Clin Nutr 2008, 87, 1219-1223. [CrossRef]
- Erridge, C.; Attina, T.; Spickett, C.M.; Webb, D.J. A high-fat meal induces low-grade endotoxemia: evidence of a novel mechanism of postprandial inflammation. Am J Clin Nutr 2007, 86, 1286-1292. [CrossRef]
- Moieni, M.; Irwin, M.R.; Jevtic, I.; Breen, E.C.; Eisenberger, N.I. Inflammation impairs social cognitive processing: A randomized controlled trial of endotoxin. Brain Behav Immun 2015, 48, 132-138. [CrossRef]
- Grigoleit, J.S.; Kullmann, J.S.; Wolf, O.T.; Hammes, F.; Wegner, A.; Jablonowski, S.; Engler, H.; Gizewski, E.; Oberbeck, R.; Schedlowski, M. Dose-dependent effects of endotoxin on neurobehavioral functions in humans. PLoS One 2011, 6, e28330. [CrossRef]
- Andre, P.; Samieri, C.; Buisson, C.; Dartigues, J.F.; Helmer, C.; Laugerette, F.; Feart, C. Lipopolysaccharide-Binding Protein, Soluble CD14, and the Long-Term Risk of Alzheimer’s Disease: A Nested Case-Control Pilot Study of Older Community Dwellers from the Three-City Cohort. J Alzheimers Dis 2019, 71, 751-761. [CrossRef]
- DellaGioia, N.; Hannestad, J. A critical review of human endotoxin administration as an experimental paradigm of depression. Neurosci Biobehav Rev 2010, 34, 130-143. [CrossRef]
- Melo, H.M.; Santos, L.E.; Ferreira, S.T. Diet-Derived Fatty Acids, Brain Inflammation, and Mental Health. Front Neurosci 2019, 13, 265. [CrossRef]
- Arnesen, P.M.; Wettergreen, M.; You, P.; Fladby, T. Alzheimer’s disease risk genes are differentially expressed upon Lipopolysaccharide stimulation in a myelogenic cell model. Alzheimer’s and Dementia 2023, 19, e080112.
- Gayle, D.A.; Ling, Z.; Tong, C.; Landers, T.; Lipton, J.W.; Carvey, P.M. Lipopolysaccharide (LPS)-induced dopamine cell loss in culture: roles of tumor necrosis factor-alpha, interleukin-1beta, and nitric oxide. Brain Res Dev Brain Res 2002, 133, 27-35. [CrossRef]
- Heppner, F.L.; Ransohoff, R.M.; Becher, B. Immune attack: the role of inflammation in Alzheimer disease. Nat Rev Neurosci 2015, 16, 358-372. [CrossRef]
- Moreno-Navarrete, J.M.; Blasco, G.; Puig, J.; Biarnes, C.; Rivero, M.; Gich, J.; Fernandez-Aranda, F.; Garre-Olmo, J.; Ramio-Torrenta, L.; Alberich-Bayarri, A. et al. Neuroinflammation in obesity: circulating lipopolysaccharide-binding protein associates with brain structure and cognitive performance. Int J Obes (Lond) 2017, 41, 1627-1635. [CrossRef]
- Zhao, Y.; Sharfman, N.M.; Jaber, V.R.; Lukiw, W.J. Down-Regulation of Essential Synaptic Components by GI-Tract Microbiome-Derived Lipopolysaccharide (LPS) in LPS-Treated Human Neuronal-Glial (HNG) Cells in Primary Culture: Relevance to Alzheimer’s Disease (AD). Front Cell Neurosci 2019, 13, 314. [CrossRef]
- Yirmiya, R.; Goshen, I. Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain Behav Immun 2011, 25, 181-213. [CrossRef]
- Fiebich, B.L.; Hull, M.; Lieb, K.; Schumann, G.; Berger, M.; Bauer, J. Potential link between interleukin-6 and arachidonic acid metabolism in Alzheimer’s disease. J Neural Transm Suppl 1998, 54, 268-278.
- Berk, M.; Williams, L.J.; Jacka, F.N.; O’Neil, A.; Pasco, J.A.; Moylan, S.; Allen, N.B.; Stuart, A.L.; Hayley, A.C.; Byrne, M.L. et al. So depression is an inflammatory disease, but where does the inflammation come from? BMC Med 2013, 11, 200. [CrossRef]
- Zhan, X.; Hakoupian, M.; Jin, L.W.; Sharp, F.R. Lipopolysaccharide, Identified Using an Antibody and by PAS Staining, Is Associated With Corpora amylacea and White Matter Injury in Alzheimer’s Disease and Aging Brain. Front Aging Neurosci 2021, 13, 705594. [CrossRef]
- Zhao, Y.; Jaber, V.; Lukiw, W.J. Gastrointestinal Tract Microbiome-Derived Pro-inflammatory Neurotoxins in Alzheimer’s Disease. J Aging Sci 2021, 9.
- Zhang, R.; Miller, R.G.; Gascon, R.; Champion, S.; Katz, J.; Lancero, M.; Narvaez, A.; Honrada, R.; Ruvalcaba, D.; McGrath, M.S. Circulating endotoxin and systemic immune activation in sporadic amyotrophic lateral sclerosis (sALS). J Neuroimmunol 2009, 206, 121-124. [CrossRef]
- Sanchez-Tapia, M.; Mimenza-Alvarado, A.; Granados-Dominguez, L.; Flores-Lopez, A.; Lopez-Barradas, A.; Ortiz, V.; Perez-Cruz, C.; Sanchez-Vidal, H.; Hernandez-Acosta, J.; Avila-Funes, J.A. et al. The Gut Microbiota-Brain Axis during Aging, Mild Cognitive Impairment and Dementia: Role of Tau Protein, beta-Amyloid and LPS in Serum and Curli Protein in Stool. Nutrients 2023, 15. [CrossRef]
- Wilson, C.J.; Finch, C.E.; Cohen, H.J. Cytokines and cognition--the case for a head-to-toe inflammatory paradigm. J Am Geriatr Soc 2002, 50, 2041-2056. [CrossRef]
- Niehaus, I.; Lange, J.H. Endotoxin: is it an environmental factor in the cause of Parkinson’s disease? Occup Environ Med 2003, 60, 378. [CrossRef]
- Cunningham, C.; Wilcockson, D.C.; Campion, S.; Lunnon, K.; Perry, V.H. Central and systemic endotoxin challenges exacerbate the local inflammatory response and increase neuronal death during chronic neurodegeneration. J Neurosci 2005, 25, 9275-9284. [CrossRef]
- Creely, S.J.; McTernan, P.G.; Kusminski, C.M.; Fisher f, M.; Da Silva, N.F.; Khanolkar, M.; Evans, M.; Harte, A.L.; Kumar, S. Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. Am J Physiol Endocrinol Metab 2007, 292, E740-747. [CrossRef]
- McGrattan, A.M.; McGuinness, B.; McKinley, M.C.; Kee, F.; Passmore, P.; Woodside, J.V.; McEvoy, C.T. Diet and Inflammation in Cognitive Ageing and Alzheimer’s Disease. Curr Nutr Rep 2019, 8, 53-65. [CrossRef]
- Eikelenboom, P.; van Exel, E.; Hoozemans, J.J.; Veerhuis, R.; Rozemuller, A.J.; van Gool, W.A. Neuroinflammation - an early event in both the history and pathogenesis of Alzheimer’s disease. Neurodegener Dis 2010, 7, 38-41. [CrossRef]
- Fassbender, K.; Walter, S.; Kuhl, S.; Landmann, R.; Ishii, K.; Bertsch, T.; Stalder, A.K.; Muehlhauser, F.; Liu, Y.; Ulmer, A.J. et al. The LPS receptor (CD14) links innate immunity with Alzheimer’s disease. FASEB J 2004, 18, 203-205. [CrossRef]
- Lee, J.W.; Lee, Y.K.; Yuk, D.Y.; Choi, D.Y.; Ban, S.B.; Oh, K.W.; Hong, J.T. Neuro-inflammation induced by lipopolysaccharide causes cognitive impairment through enhancement of beta-amyloid generation. J Neuroinflammation 2008, 5, 37. [CrossRef]
- Zhan, X.; Stamova, B.; Jin, L.W.; DeCarli, C.; Phinney, B.; Sharp, F.R. Gram-negative bacterial molecules associate with Alzheimer disease pathology. Neurology 2016, 87, 2324-2332. [CrossRef]
- Marizzoni, M.; Cattaneo, A.; Mirabelli, P.; Festari, C.; Lopizzo, N.; Nicolosi, V.; Mombelli, E.; Mazzelli, M.; Luongo, D.; Naviglio, D. et al. Short-Chain Fatty Acids and Lipopolysaccharide as Mediators Between Gut Dysbiosis and Amyloid Pathology in Alzheimer’s Disease. J Alzheimers Dis 2020, 78, 683-697. [CrossRef]
- Kalmijn, S.; Launer, L.J.; Ott, A.; Witteman, J.C.; Hofman, A.; Breteler, M.M. Dietary fat intake and the risk of incident dementia in the Rotterdam Study. Ann Neurol 1997, 42, 776-782. [CrossRef]
- Krabbe, K.S.; Reichenberg, A.; Yirmiya, R.; Smed, A.; Pedersen, B.K.; Bruunsgaard, H. Low-dose endotoxemia and human neuropsychological functions. Brain Behav Immun 2005, 19, 453-460. [CrossRef]
- Salardini, A.; Hillmer, A.T.; AP, M.; A, H.-A.; E, L.; SE, S.; RS, O.D.; JE, H.; TA, G.; NB, N. et al. PBR28 brain PET imaging with lipopolysaccharide challenge for the study of microglia function in Alzheimer’s disease. In Proceedings of 2020 Alzheimer’s Association International Conference July 28, 2020.
- Sandiego, C.M.; Gallezot, J.D.; Pittman, B.; Nabulsi, N.; Lim, K.; Lin, S.F.; Matuskey, D.; Lee, J.Y.; O’Connor, K.C.; Huang, Y. et al. Imaging robust microglial activation after lipopolysaccharide administration in humans with PET. Proc Natl Acad Sci U S A 2015, 112, 12468-12473. [CrossRef]
- Holmes, C.; Cunningham, C.; Zotova, E.; Woolford, J.; Dean, C.; Kerr, S.; Culliford, D.; Perry, V.H. Systemic inflammation and disease progression in Alzheimer disease. Neurology 2009, 73, 768-774. [CrossRef]
- Ferrucci, L.; Harris, T.B.; Guralnik, J.M.; Tracy, R.P.; Corti, M.C.; Cohen, H.J.; Penninx, B.; Pahor, M.; Wallace, R.; Havlik, R.J. Serum IL-6 level and the development of disability in older persons. J Am Geriatr Soc 1999, 47, 639-646. [CrossRef]
- Cunningham, C. Microglia and neurodegeneration: the role of systemic inflammation. Glia 2013, 61, 71-90. [CrossRef]
- Engelhart, M.J.; Geerlings, M.I.; Meijer, J.; Kiliaan, A.; Ruitenberg, A.; van Swieten, J.C.; Stijnen, T.; Hofman, A.; Witteman, J.C.; Breteler, M.M. Inflammatory proteins in plasma and the risk of dementia: the rotterdam study. Arch Neurol 2004, 61, 668-672. [CrossRef]
- Cunningham, C.; Campion, S.; Lunnon, K.; Murray, C.L.; Woods, J.F.; Deacon, R.M.; Rawlins, J.N.; Perry, V.H. Systemic inflammation induces acute behavioral and cognitive changes and accelerates neurodegenerative disease. Biol Psychiatry 2009, 65, 304-312. [CrossRef]
- Cohen, H.J.; Pieper, C.F.; Harris, T.; Rao, K.M.; Currie, M.S. The association of plasma IL-6 levels with functional disability in community-dwelling elderly. J Gerontol A Biol Sci Med Sci 1997, 52, M201-208. [CrossRef]
- Zhao, Y.; Jaber, V.; Lukiw, W.J. Secretory Products of the Human GI Tract Microbiome and Their Potential Impact on Alzheimer’s Disease (AD): Detection of Lipopolysaccharide (LPS) in AD Hippocampus. Front Cell Infect Microbiol 2017, 7, 318. [CrossRef]
- Licinio, J.; Kling, M.A.; Hauser, P. Cytokines and brain function: relevance to interferon-alpha-induced mood and cognitive changes. Semin Oncol 1998, 25, 30-38.
- Arvin, B.; Neville, L.F.; Barone, F.C.; Feuerstein, G.Z. The role of inflammation and cytokines in brain injury. Neurosci Biobehav Rev 1996, 20, 445-452. [CrossRef]
- Asby, D.; Boche, D.; Allan, S.; Love, S.; Miners, J.S. Systemic infection exacerbates cerebrovascular dysfunction in Alzheimer’s disease. Brain 2021, 144, 1869-1883. [CrossRef]
- Moller, K.; Strauss, G.I.; Qvist, J.; Fonsmark, L.; Knudsen, G.M.; Larsen, F.S.; Krabbe, K.S.; Skinhoj, P.; Pedersen, B.K. Cerebral blood flow and oxidative metabolism during human endotoxemia. J Cereb Blood Flow Metab 2002, 22, 1262-1270. [CrossRef]
- Wiedermann, C.J.; Kiechl, S.; Dunzendorfer, S.; Schratzberger, P.; Egger, G.; Oberhollenzer, F.; Willeit, J. Association of endotoxemia with carotid atherosclerosis and cardiovascular disease: prospective results from the Bruneck Study. J Am Coll Cardiol 1999, 34, 1975-1981. [CrossRef]
- Funari, E.; Testai, E. Human health risk assessment related to cyanotoxins exposure. Crit Rev Toxicol 2008, 38, 97-125. [CrossRef]
- Serrano, M.; Moreno-Navarrete, J.M.; Puig, J.; Moreno, M.; Guerra, E.; Ortega, F.; Xifra, G.; Ricart, W.; Fernandez-Real, J.M. Serum lipopolysaccharide-binding protein as a marker of atherosclerosis. Atherosclerosis 2013, 230, 223-227. [CrossRef]
| Food | ng/g of LPS | Serving Size g | LPS per Serving in ng |
|---|---|---|---|
| Macaroni cheese | 6,500 | 340 | 2,200,000 |
| Minced turkey | 7800 | 230 | 1,800,000 |
| Cheese and onion Rolls | 17,000 | 74 | 1,300,000 |
| Minced pork | 10,000 | 110 | 1,100,000 |
| Minced beef | 7000 | 98 | 690,000 |
| Pork sausage rolls | 4,200 | 2 rolls 120 | 520,000 |
| Turkey | 300 | 230 | 510,000 |
| Hamburger patty | 3090 | 98 | 300,000 |
| Infant formula milk powder | 2800 | 100 | 280,000 |
| Lobster | 1200 | 145 | 170,000 |
| Pork | 1100 | 110 | 120,000 |
| Spaghetti bolognese | 220 | 400 | 90,000 |
| Minced beef and onion pie | 330 | 310 | 69,000 |
| Skim milk, one cup | 75 ng/ml | 240 | 18,000 |
| Milk, one cup | 50 | 240 | 12,000 |
| Exposure to LPS | Amount | Cognitive Test Result |
|---|---|---|
| LPS injection | 0.8 ng/kg of body weight | Impairment of social cognitive processing [55]. |
| LPS injection | 0.4 ng/kg or 0.8 ng/kg body weight | Long-term memory performance was impaired, low dose only [56]. |
| LPS injection | 0.2 ng/kg body weight | Degraded performance in declarative memory [57]. |
| LPS-induced cytokine secretion | 49 ng Injection of LPS | Impairments in verbal and nonverbal declarative memory functions and decrease immediate and delayed recall [50]. |
| LPS injection | 0.2 ng/kg body weight | Significant impairment in declarative memory and word-list learning [7]. |
| LPS injection | 0.8 ng/kg body weight | Negative effects on memory function [9]. |
| Levels of circulating LBP | Higher | Impaired cognitive performance [57]. |
| Levels of circulating LBP | Higher | Poorer working memory/short-term verbal memory and the Digit Span Test Puig, 2017}. |
| LPS administration | Low-dose | Impaired both immediate and delayed recall [58]. |
| Plasma LPS | Elevated | Performed worse on the post-meal cognitive test and had poorer attention during testing Belury, 2020}. |
| Cholesterol and SFAs | High intake | Increased the risk of impaired cognitive function and the development of dementia, including AD [59]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




