Submitted:
29 January 2024
Posted:
30 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. LyeTx I mn∆K Shows Good In Vitro Activity against MRSA
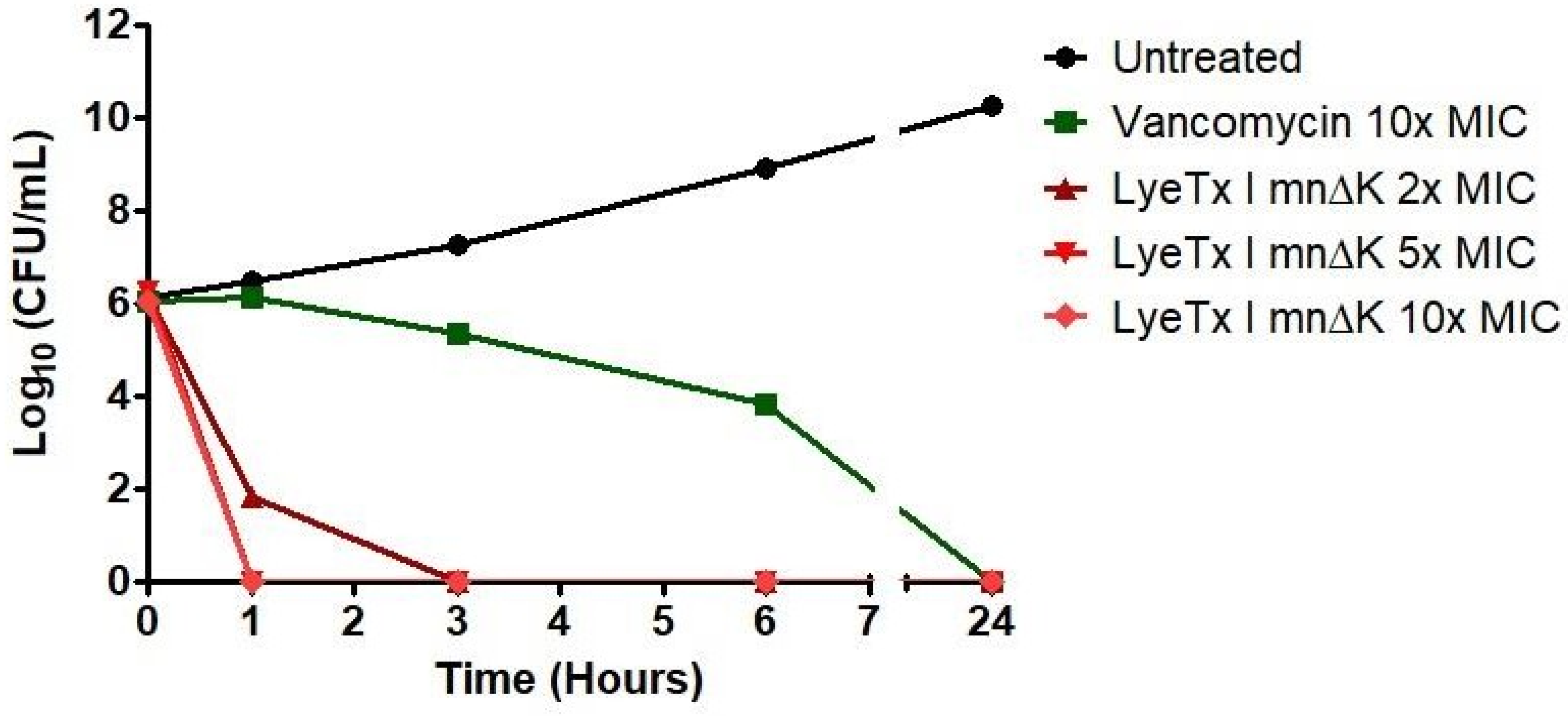
2.2. LyeTx I mn∆K Exhibits Rapid Bactericidal Effects on Cells in Logarithmic Growth of MRSA
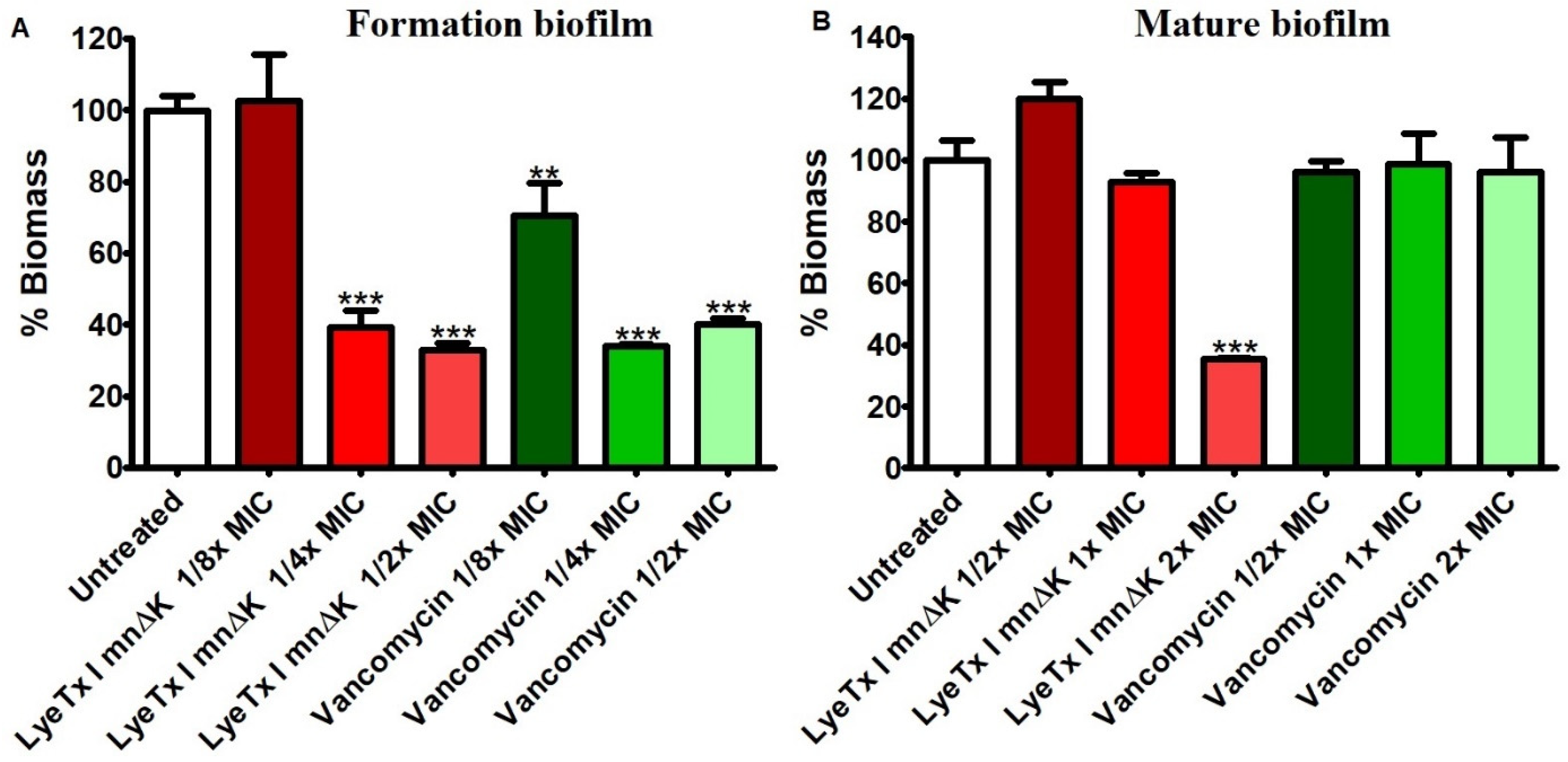
2.3. LyeTx I mn∆K Reduces Preformed Biofilms of MRSA
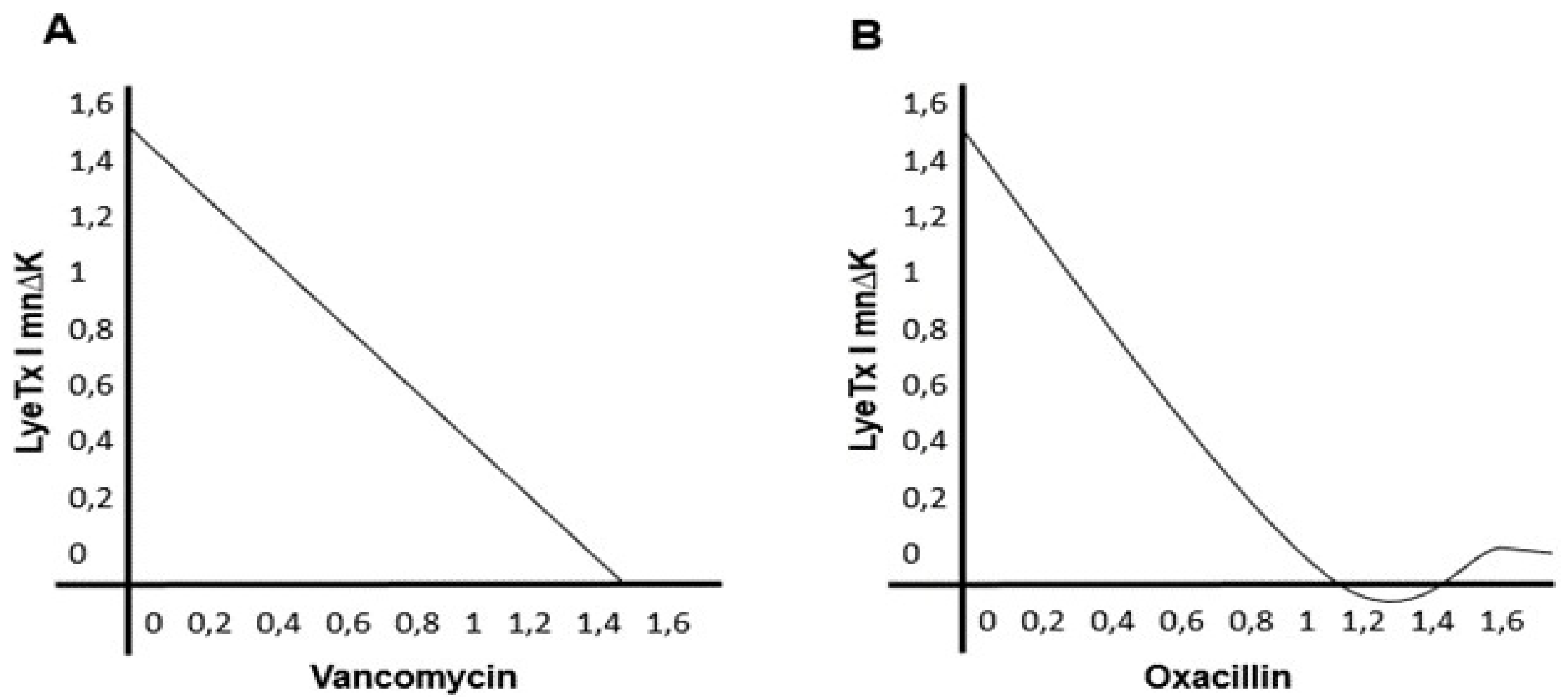
2.4. Behavior of LyeTx I mnΔK after Combination with Conventional Antimicrobial Agents against MRSA
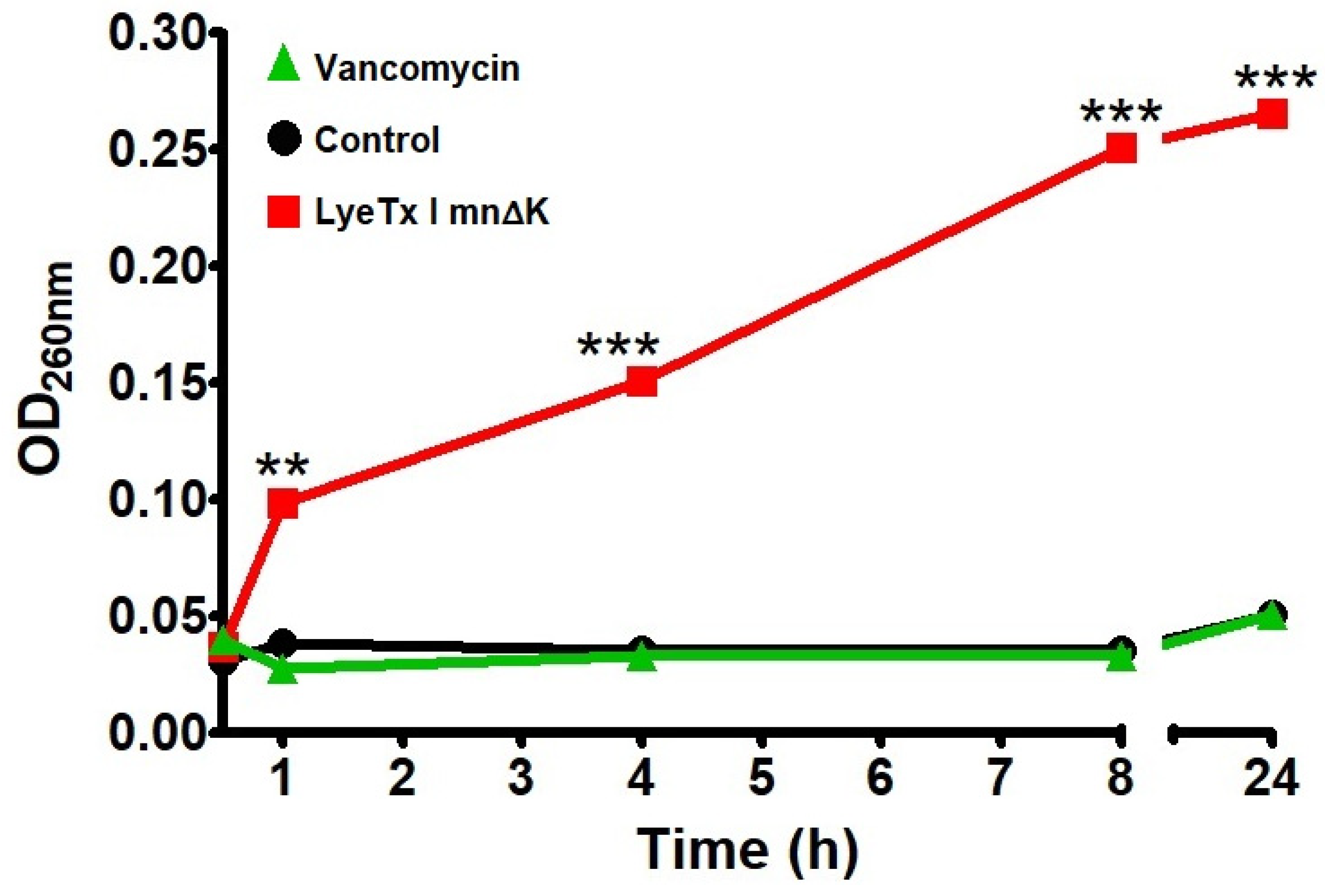
2.5. LyeTx I mn∆K Induces Membranolytic Effect on MRSA Cells
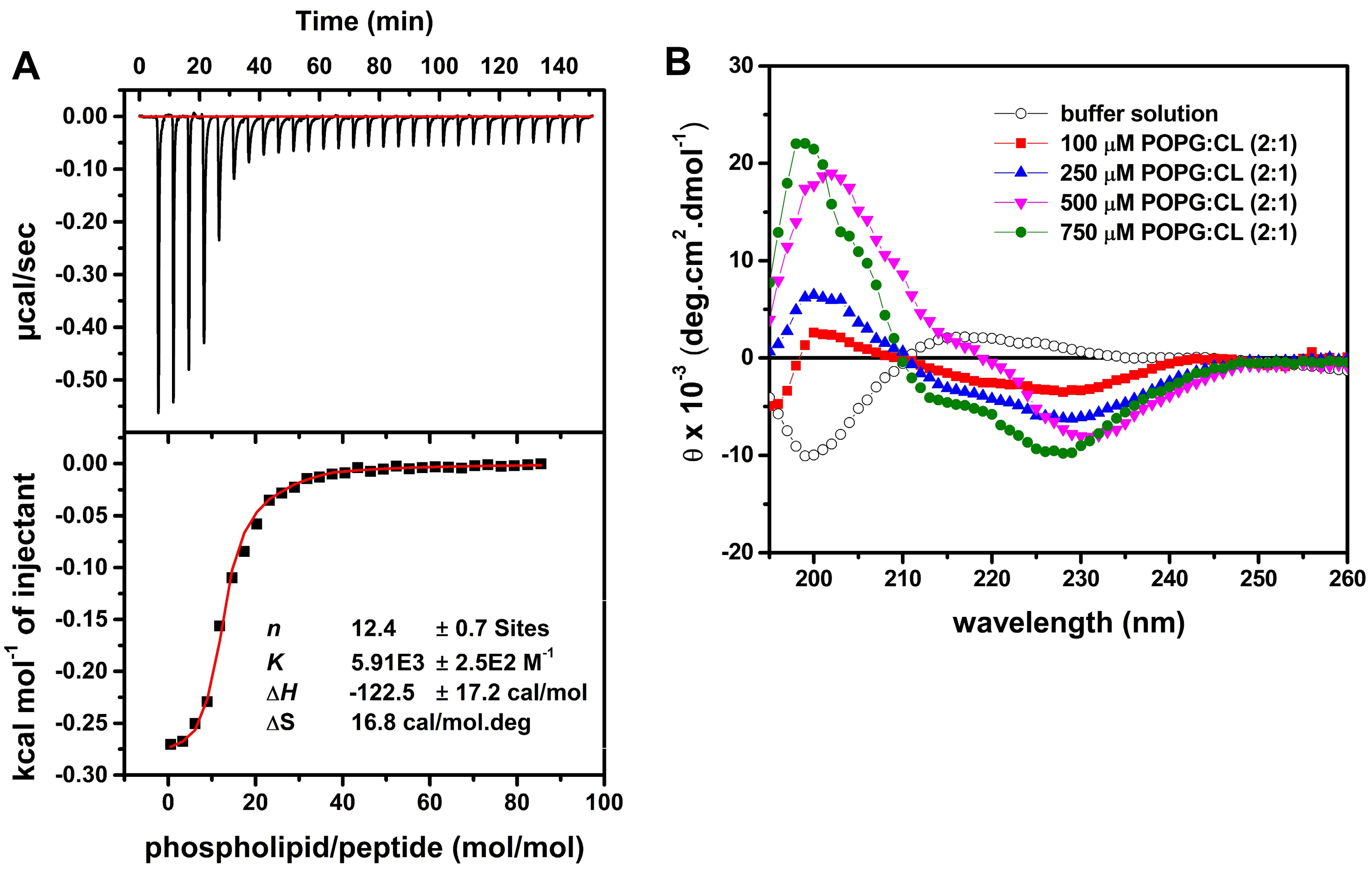
2.6. LyeTx I mn∆K Interacts with POPG:CL Membranes
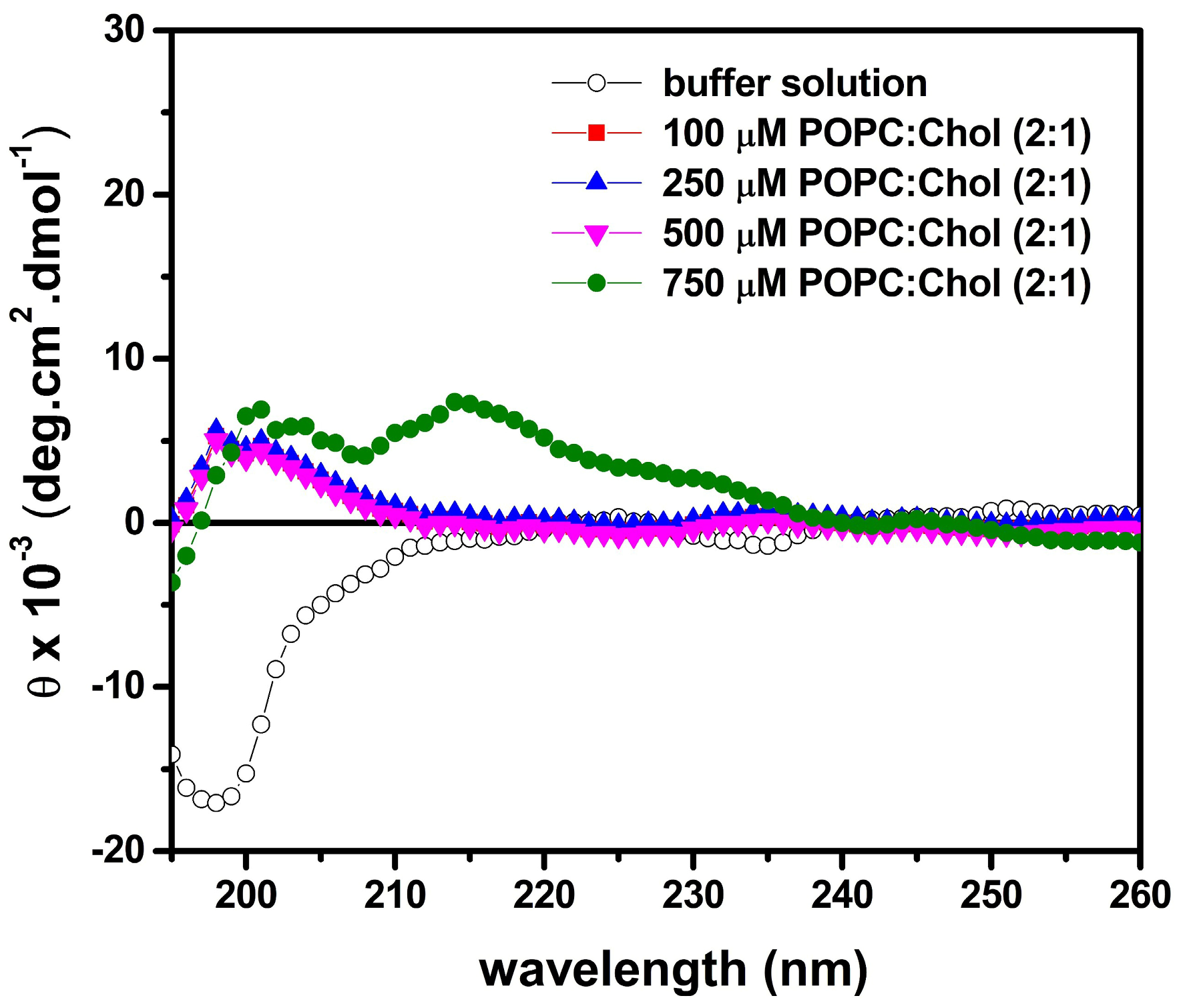
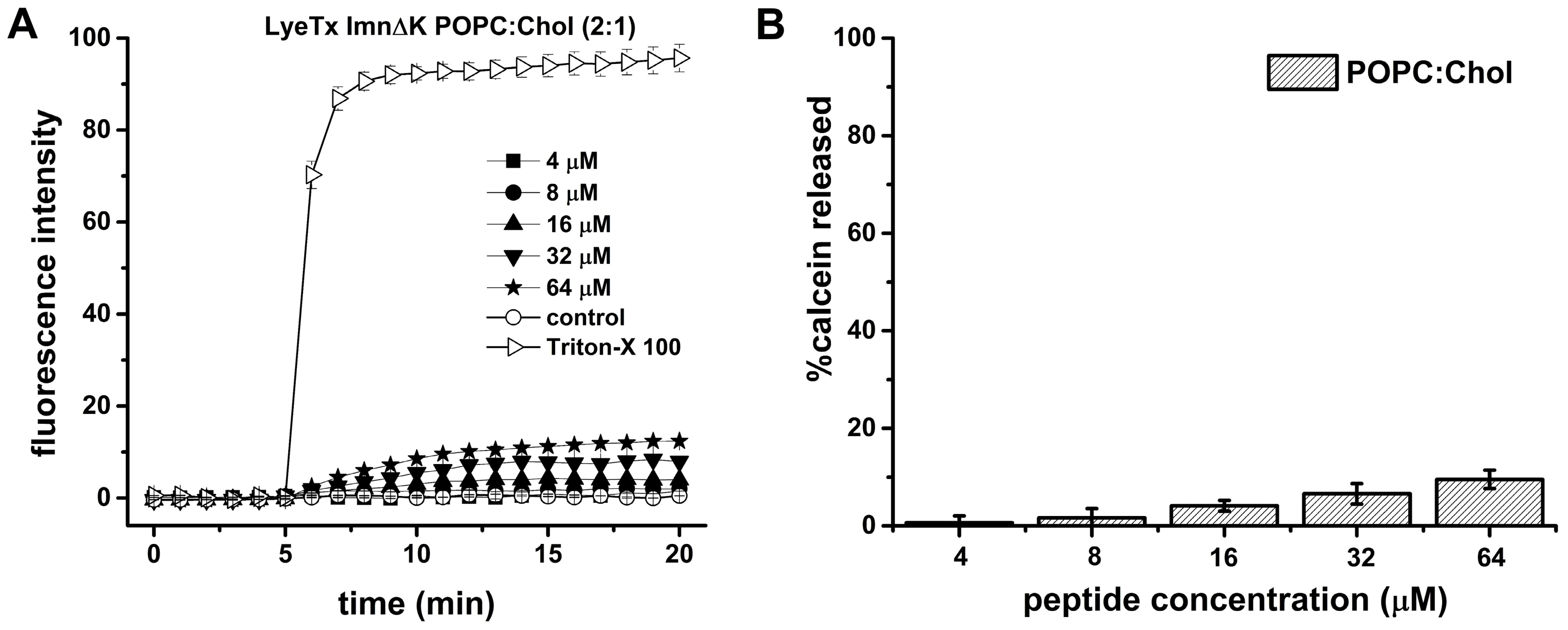
2.7. LyeTx I mn∆K Has Low Interaction with Artificial Vesicles That Mimicking Eukaryotic Membranes
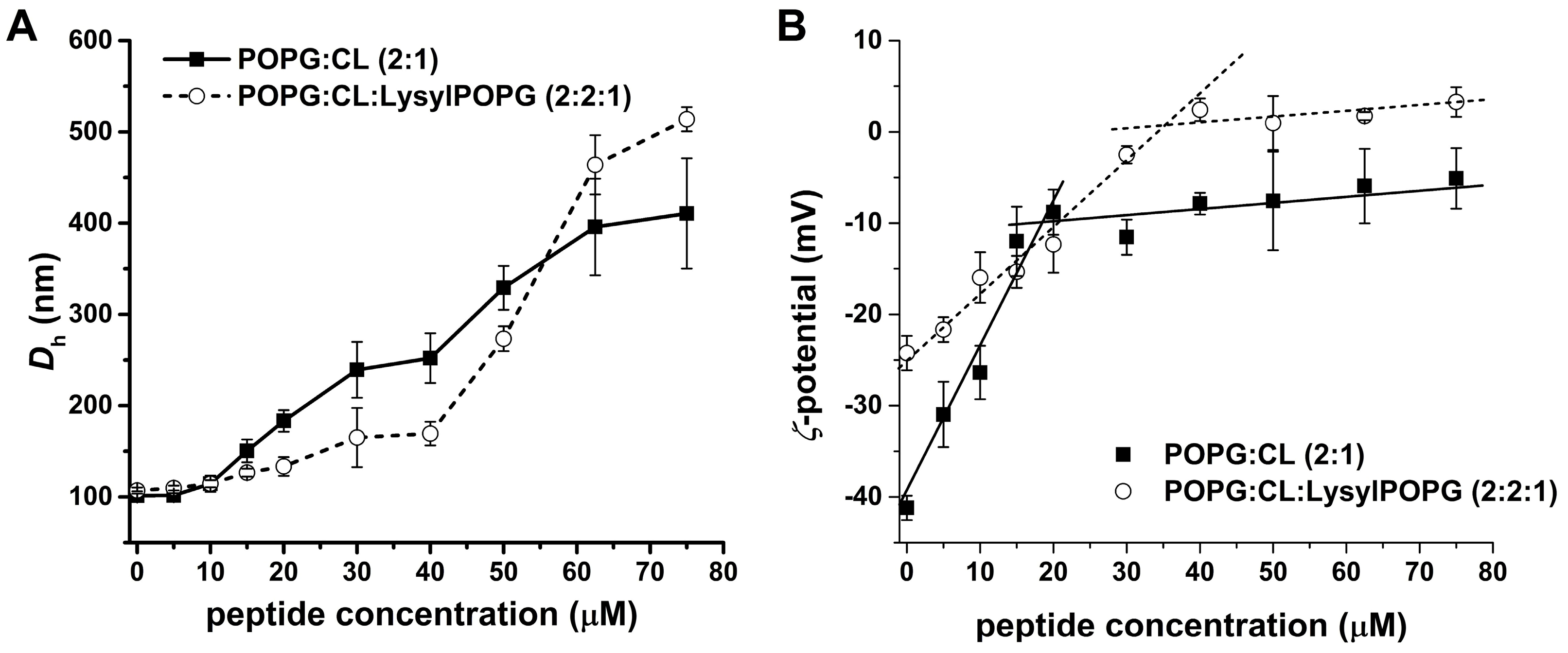
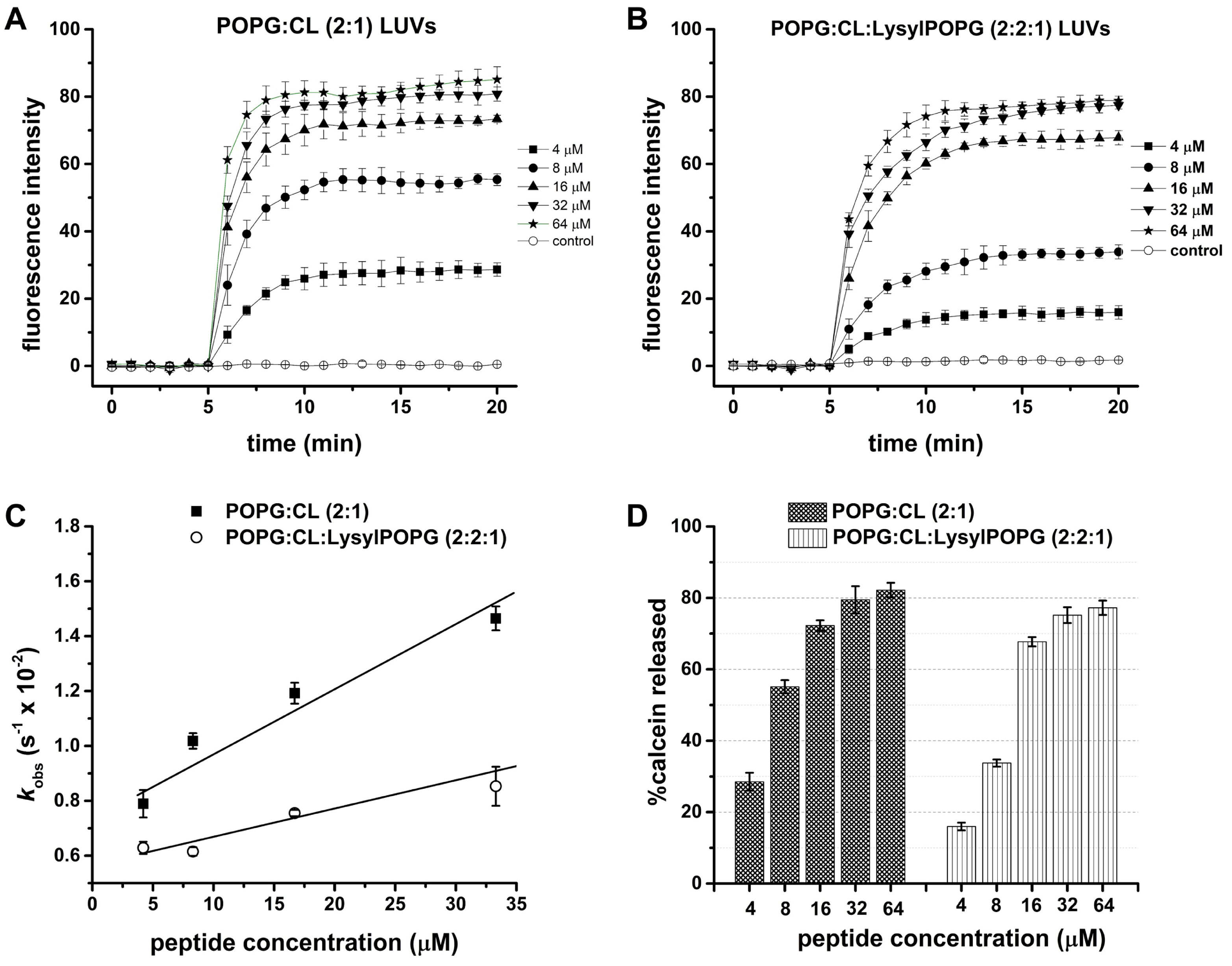
2.8. Effect of LysylPOPG on the Peptide-Membrane Interaction
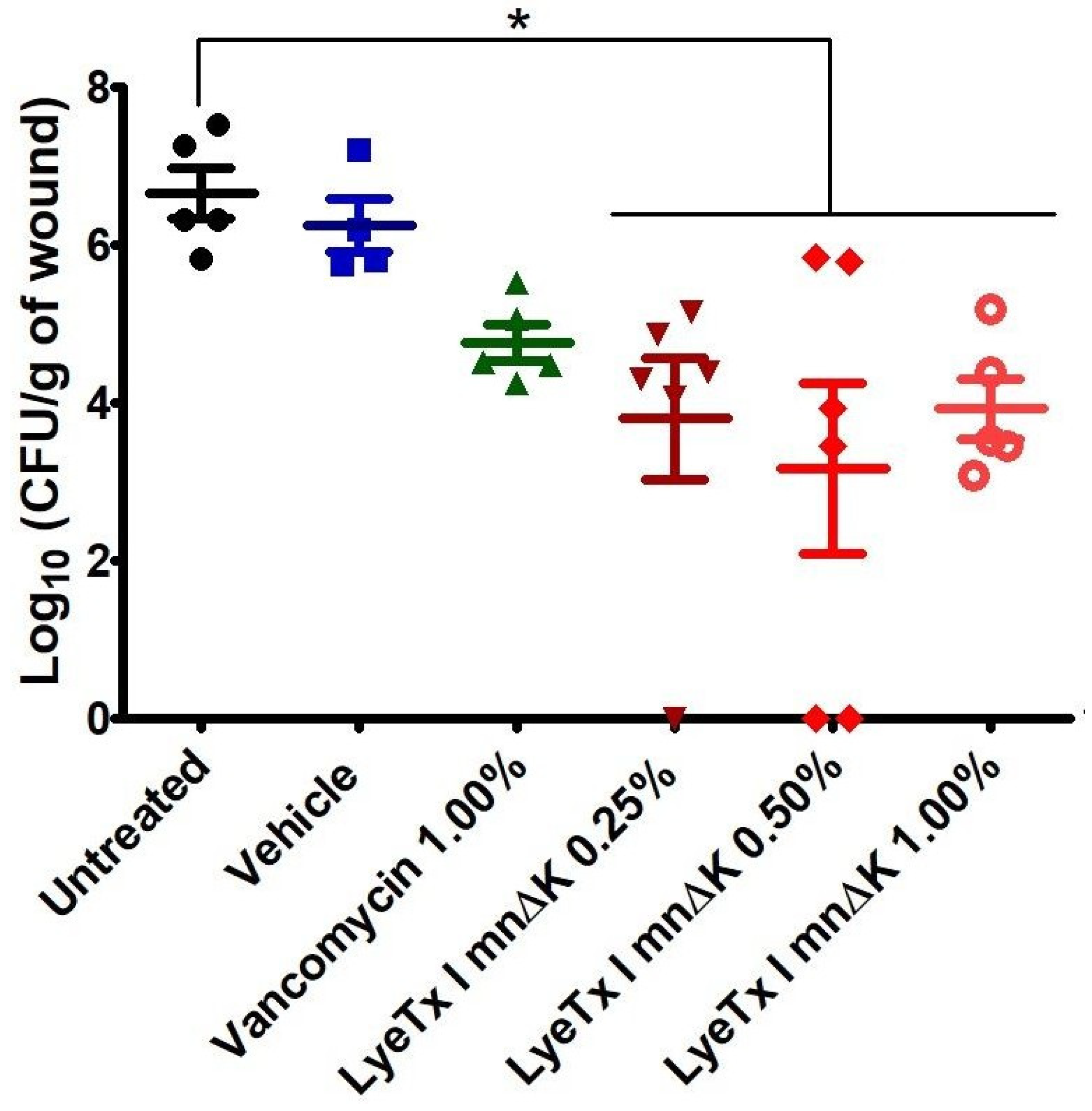
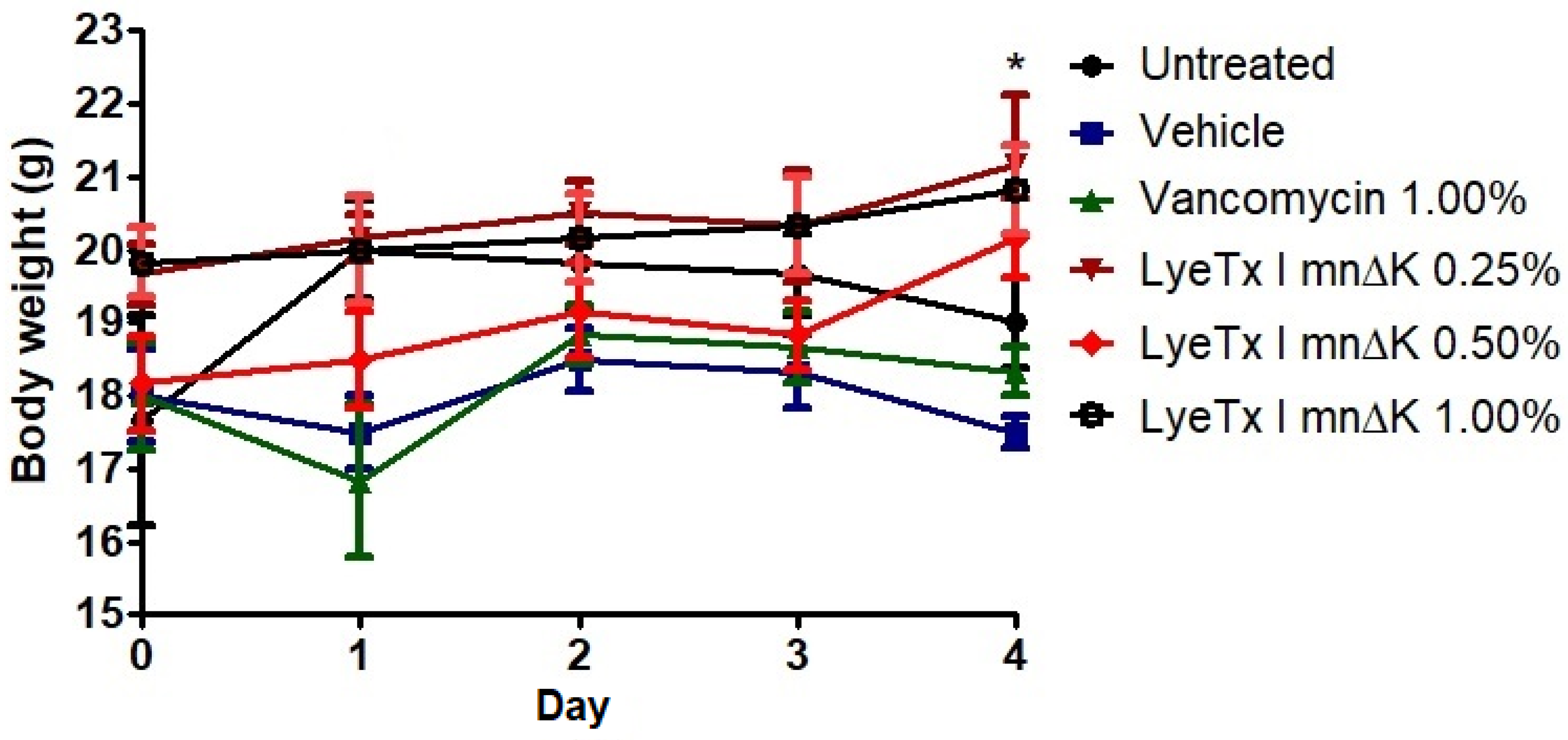
2.9. Gel Containing LyeTx I mnΔK is Effective in MRSA-Induced Wounds in Mice
3. Discussion
4. Materials and Methods
4.1. Microorganisms
4.2. Reagents
4.3. Antimicrobial Activity
4.3.1. Preparation of the Inoculum
4.3.2. Determination of Minimum Inhibitory Concentration (MIC)
4.3.3. Determination of Minimum Bactericidal Concentration (MBC)
4.4. Time-Kill Curve
4.5. Anti-Biofilm Activity
4.6. Membranolytic Effect
4.7. Combination Assays
4.7.1. Synergism
4.7.2. Resensitization
4.8. Preparation of Large Unilamellar Vesicles (LUVS)
4.9. Isothermal Titration Calorimetry (ITC)
4.10. Dynamic Light Scattering (DLS)
4.11. Calcein Release
4.12. Peptide Formulation
4.13. In Vivo assays
4.13.1. Animals
4.13.2. Murine Model of Non-Surgical MRSA-Infected Wounds
4.13.3. Treatment of Animals
4.14. Statistical Analysis
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Sharma, N.K.; Garg, R.; Baliga, S.; Bhat, G.J.J.o.c.; JCDR, d.r. Nosocomial infections and drug susceptibility patterns in methicillin sensitive and methicillin resistant Staphylococcus aureus. 2013, 7, 2178.
- Turner, N.A.; Sharma-Kuinkel, B.K.; Maskarinec, S.A.; Eichenberger, E.M.; Shah, P.P.; Carugati, M.; Holland, T.L.; Fowler Jr, V.G.J.N.R.M. Methicillin-resistant Staphylococcus aureus: an overview of basic and clinical research. 2019, 17, 203-218.
- Ventola, C.L.J.P.; therapeutics. The antibiotic resistance crisis: part 1: causes and threats. 2015, 40, 277.
- Mahjabeen, F.; Saha, U.; Mostafa, M.N.; Siddique, F.; Ahsan, E.; Fathma, S.; Tasnim, A.; Rahman, T.; Faruq, R.; Sakibuzzaman, M.J.C. An Update on Treatment Options for Methicillin-Resistant Staphylococcus aureus (MRSA) Bacteremia: A Systematic Review. 2022, 14.
- Oliveira, C.F.d.; Morey, A.T.; Biasi-Garbin, R.P.; Perugini, M.R.E.; Yamauchi, L.M.; Yamada-Ogatta, S.F. Emergência de Staphylococcus aureus resistentes aos antimicrobianos: um desafio contínuo. 2014.
- Mba, I.E.; Nweze, E.I.J.T.Y.j.o.b.; medicine. Focus: Antimicrobial Resistance: Antimicrobial Peptides Therapy: An Emerging Alternative for Treating Drug-Resistant Bacteria. 2022, 95, 445.
- Mwangi, J.; Hao, X.; Lai, R.; Zhang, Z.-Y.J.Z.r. Antimicrobial peptides: new hope in the war against multidrug resistance. 2019, 40, 488.
- Vale, N.; Aguiar, L.; Gomes, P.J.F.i.P. Antimicrobial peptides: a new class of antimalarial drugs? 2014, 5, 275.
- Bechinger, B.; Gorr, S.-U.J.J.o.d.r. Antimicrobial peptides: mechanisms of action and resistance. 2017, 96, 254-260.
- Chen, C.H.; Lu, T.K.J.A. Development and challenges of antimicrobial peptides for therapeutic applications. 2020, 9, 24.
- Brogden, K.A.J.N.r.m. Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? 2005, 3, 238-250.
- Huang, H.W.; Charron, N.E.J.Q.r.o.b. Understanding membrane-active antimicrobial peptides. 2017, 50, e10.
- Espeche, J.C.; Varas, R.; Maturana, P.; Cutro, A.C.; Maffía, P.C.; Hollmann, A.J.P.S. Membrane permeability and antimicrobial peptides: Much more than just making a hole. 2023, e24305.
- Marr, A.K.; Gooderham, W.J.; Hancock, R.E.J.C.o.i.p. Antibacterial peptides for therapeutic use: obstacles and realistic outlook. 2006, 6, 468-472.
- Liu, Y.; Shi, J.; Tong, Z.; Jia, Y.; Yang, K.; Wang, Z.J.M. Potent broad-spectrum antibacterial activity of amphiphilic peptides against multidrug-resistant bacteria. 2020, 8, 139.
- Santos, D.; Verly, R.; Piló-Veloso, D.; De Maria, M.; De Carvalho, M.; Cisalpino, P.; Soares, B.; Diniz, C.; Farias, L.; Moreira, D.J.A.A. LyeTx I, a potent antimicrobial peptide from the venom of the spider Lycosa erythrognatha. 2010, 39, 135-144.
- Fuscaldi, L.L.; de Avelar Júnior, J.T.; Dos Santos, D.M.; Boff, D.; de Oliveira, V.L.S.; Gomes, K.A.G.G.; Cruz, R.d.C.; de Oliveira, P.L.; Magalhães, P.P.; Cisalpino, P.S.J.E.B., et al. Shortened derivatives from native antimicrobial peptide LyeTx I: In vitro and in vivo biological activity assessment. 2021, 246, 414-425.
- Cardoso, B.G.; de Lima, W.G.; Fernandes, S.O.A.; de Lima, M.E.; Cardoso, V.N.J.N.P.R. Antifungal activity of a shortened analogue of the natural peptide LyeTx I isolated from the venom of the spider Lycosa erythrognatha. 2023, 37, 759-763.
- Lima, W.G.; Brito, J.C.M.; de Lima, M.E.; Pizarro, A.C.S.T.; Vianna, M.A.M.d.M.; de Paiva, M.C.; de Assis, D.C.S.; Cardoso, V.N.; Fernandes, S.O.A.J.T.J.o.A. A short synthetic peptide, based on LyeTx I from Lycosa erythrognatha venom, shows potential to treat pneumonia caused by carbapenem-resistant Acinetobacter baumannii without detectable resistance. 2021, 74, 425-434.
- Santos Neto, N.A.d. United we stand, divided we fall: antibiofilm activity and mechanisms of action of synthetic peptides in combination with ciprofloxacin against Staphylococcus aureus biofilm. 2023.
- de Brito, J.C.M. Peptídeo antimicrobiano PEGuilado como potencial biofármaco para o tratamento de pneumonia causada por Acinetobacter baumannii. 2020.
- Wiseman, T.; Williston, S.; Brandts, J.F.; Lin, L.-N.J.A.b. Rapid measurement of binding constants and heats of binding using a new titration calorimeter. 1989, 179, 131-137.
- Seelig, J.J.B.e.B.A.-B. Thermodynamics of lipid–peptide interactions. 2004, 1666, 40-50.
- Abrunhosa, F.; Faria, S.; Gomes, P.; Tomaz, I.; Pessoa, J.C.; Andreu, D.; Bastos, M.J.T.J.o.P.C.B. Interaction and lipid-induced conformation of two cecropin− melittin hybrid peptides depend on peptide and membrane composition. 2005, 109, 17311-17319.
- Niu, Z.; Prade, E.; Malideli, E.; Hille, K.; Jussupow, A.; Mideksa, Y.G.; Yan, L.M.; Qian, C.; Fleisch, M.; Messias, A.C.J.A.C. Structural Insight into IAPP-Derived Amyloid Inhibitors and Their Mechanism of Action. 2020, 132, 5820-5830.
- Junior, E.F.; Guimarães, C.F.; Franco, L.L.; Alves, R.J.; Kato, K.C.; Martins, H.R.; de Souza Filho, J.D.; Bemquerer, M.P.; Munhoz, V.H.; Resende, J.M.J.A.a. Glycotriazole-peptides derived from the peptide HSP1: synergistic effect of triazole and saccharide rings on the antifungal activity. 2017, 49, 1389-1400.
- Hassoun, A.; Linden, P.K.; Friedman, B.J.C.c. Incidence, prevalence, and management of MRSA bacteremia across patient populations—a review of recent developments in MRSA management and treatment. 2017, 21, 1-10.
- Wijesekara, P.N.K.; Kumbukgolla, W.W.; Jayaweera, J.A.A.S.; Rawat, D.J.V.s. Review on usage of vancomycin in livestock and humans: maintaining its efficacy, prevention of resistance and alternative therapy. 2017, 4, 6.
- Lima, W.G.; Batista Filho, F.L.; Lima, I.P.; Simião, D.C.; Brito, J.C.M.; da Cruz Nizer, W.S.; Cardoso, V.N.; Fernandes, S.O.A.J.N.P.R. Antibacterial, anti-biofilm, and anti-adhesive activities of melittin, a honeybee venom-derived peptide, against quinolone-resistant uropathogenic Escherichia coli (UPEC). 2022, 36, 6381-6388.
- Percival, S.L.; McCarty, S.M.; Lipsky, B.J.A.i.w.c. Biofilms and wounds: an overview of the evidence. 2015, 4, 373-381.
- Tolker-Nielsen, T.J.M.B. Biofilm development. 2015, 51-66.
- Venkatesan, N.; Perumal, G.; Doble, M.J.F.m. Bacterial resistance in biofilm-associated bacteria. 2015, 10, 1743-1750.
- Hall, C.W.; Mah, T.-F.J.F.m.r. Molecular mechanisms of biofilm-based antibiotic resistance and tolerance in pathogenic bacteria. 2017, 41, 276-301.
- de Avelar Júnior, J.T. Estudo de três peptídeos sintéticos com atividade antimicrobiana, derivados da toxina LyeTx I da aranha Lycosa erythrognatha (Lucas, 1836). 2015.
- Wang, S.; Zeng, X.; Yang, Q.; Qiao, S.J.I.j.o.m.s. Antimicrobial peptides as potential alternatives to antibiotics in food animal industry. 2016, 17, 603.
- Lima, W.G.; de Brito, J.C.M.; Cardoso, V.N.; Fernandes, S.O.A.J.E.J.o.P.S. In-depth characterization of antibacterial activity of melittin against Staphylococcus aureus and use in a model of non-surgical MRSA-infected skin wounds. 2021, 156, 105592.
- Stogios, P.J.; Savchenko, A.J.P.S. Molecular mechanisms of vancomycin resistance. 2020, 29, 654-669.
- Hayami, M.; Okabe, A.; Kariyama, R.; Abe, M.; Kanemasa, Y.J.M.; immunology. Lipid composition of Staphylococcus aureus and its derived L-forms. 1979, 23, 435-442.
- Hernández-Villa, L.; Manrique-Moreno, M.; Leidy, C.; Jemioła-Rzemińska, M.; Ortíz, C.; Strzałka, K.J.B.c. Biophysical evaluation of cardiolipin content as a regulator of the membrane lytic effect of antimicrobial peptides. 2018, 238, 8-15.
- Perez, M.I.; Trier, S.M.; Bernal, A.; Vargas, J.C.; Herrfurth, C.; Feussner, I.; Gonzalez, J.M.; Leidy, C.J.B.J. S. Aureus adapt to growth conditions by changing membrane order. 2014, 106, 580a.
- Verly, R.M.; Rodrigues, M.A.; Daghastanli, K.R.P.; Denadai, A.M.L.; Cuccovia, I.M.; Bloch Jr, C.; Frézard, F.; Santoro, M.M.; Piló-Veloso, D.; Bemquerer, M.P.J.P. Effect of cholesterol on the interaction of the amphibian antimicrobial peptide DD K with liposomes. 2008, 29, 15-24.
- Moreira Brito, J.C.; Carvalho, L.R.; Neves de Souza, A.; Carneiro, G.; Magalhães, P.P.; Farias, L.M.; Guimarães, N.R.; Verly, R.M.; Resende, J.M.; Elena de Lima, M.J.F.i.M.B. PEGylation of the antimicrobial peptide LyeTx Ib maintains structure-related biological properties and improves selectivity. 2022, 9, 1001508.
- Greenfield, N.J.J.N.p. Using circular dichroism spectra to estimate protein secondary structure. 2006, 1, 2876-2890.
- Verly, R.M.; Resende, J.M.; Junior, E.F.; de Magalhães, M.T.; Guimarães, C.F.; Munhoz, V.H.; Bemquerer, M.P.; Almeida, F.C.; Santoro, M.M.; Piló-Veloso, D.J.S.r. Structure and membrane interactions of the homodimeric antibiotic peptide homotarsinin. 2017, 7, 40854.
- Rashid, R.; Cazenave-Gassiot, A.; Gao, I.H.; Nair, Z.J.; Kumar, J.K.; Gao, L.; Kline, K.A.; Wenk, M.R.J.P.o. Comprehensive analysis of phospholipids and glycolipids in the opportunistic pathogen Enterococcus faecalis. 2017, 12, e0175886.
- Van Bambeke, F.; Tulkens, P.M.; Brasseur, R.; Mingeot-Leclercq, M.-P.J.E.J.o.P.M.P. Aminoglycoside antibiotics induce aggregation but not fusion of negatively-charged liposomes. 1995, 289, 321-333.
- Van Bambeke, F.; Mingeot-Leclercq, M.-P.; Brasseur, R.; Tulkens, P.M.; Schanck, A.J.C.; lipids, p.o. Aminoglycoside antibiotics prevent the formation of non-bilayer structures in negatively-charged membranes. Comparative studies using fusogenic (bis (β-diethylaminoethylether) hexestrol) and aggregating (spermine) agents. 1996, 79, 123-135.
- Parmanik, A.; Das, S.; Kar, B.; Bose, A.; Dwivedi, G.R.; Pandey, M.M.J.C.m. Current treatment strategies against multidrug-resistant bacteria: a review. 2022, 79, 388.
- Tängdén, T.J.U.j.o.m.s. Combination antibiotic therapy for multidrug-resistant Gram-negative bacteria. 2014, 119, 149-153.
- Hatlen, T.J.; Miller, L.G.J.I.D.C. Staphylococcal skin and soft tissue infections. 2021, 35, 81-105.
- Simonetti, O.; Marasca, S.; Candelora, M.; Rizzetto, G.; Radi, G.; Molinelli, E.; Brescini, L.; Cirioni, O.; Offidani, A.J.A.m. Methicillin-resistant Staphylococcus aureus as a cause of chronic wound infections: Alternative strategies for management. 2022, 8, 125.
- Santana, H.J.A. Interação do glicopeptídeo Vancomicina em monocamadas de Langmuir como modelos de membrana celular. 2022.
- (BrCAST), C.B.D.T.D.S.A.A. Orientações do EUCAST para a detecção de mecanismos de resistência e resistências específicas de importância clínica e/ou epidemiológica. 2017.
- Herrera, K.M.; da Silva, F.K.; de Lima, W.G.; Barbosa, C.d.S.; Gonçalves, A.M.; Viana, G.H.; Soares, A.C.; Ferreira, J.M.J.M.C.R. Antibacterial and antibiofilm activities of synthetic analogs of 3-alkylpyridine marine alkaloids. 2020, 29, 1084-1089.
- Orhan, G.; Bayram, A.; Zer, Y.; Balci, I.J.J.o.c.m. Synergy tests by E test and checkerboard methods of antimicrobial combinations against Brucella melitensis. 2005, 43, 140-143.
- Oroojalian, F.; Kasra-Kermanshahi, R.; Azizi, M.J.P.M. Synergistic antibacterial activity of Bunium persicum and Cuminum cyminum essential oils. 2008, 74, PI36.
- Mohamed, M.F.; Abdelkhalek, A.; Seleem, M.N.J.S.r. Evaluation of short synthetic antimicrobial peptides for treatment of drug-resistant and intracellular Staphylococcus aureus. 2016, 6, 29707.
- Stewart, J.C.M.J.A.b. Colorimetric determination of phospholipids with ammonium ferrothiocyanate. 1980, 104, 10-14.
- Brazil. AGENCIA NACIONAL DE VIGILÂNCIA SANITÁRIA (ANVISA). 5ª ed.; Farmacopeia Brasileira, Brasilia: 2010b; Vol. 1.
|
Microorganism |
Clinical origin |
Antibacterial activity (µM) | |||
|---|---|---|---|---|---|
| LyeTx I mnΔK | Vancomycin | ||||
| MIC | MBC | MIC | MBC | ||
| S. aureus 11 | Infected wound | 8 | 32 | 1 | 8 |
| S. aureus 29 | Infected wound | 8 | 16 | 1 | 2 |
| S. aureus 130 | Infected wound | 8 | 32 | 1 | 2 |
| S. aureus 366 | Infected wound | 16 | 32 | 1 | 8 |
| S. aureus 524 | Infected wound | 16 | 32 | 1 | 2 |
| S. aureus 526 | Infected wound | 16 | 32 | 1 | 4 |
| MRSA USA300 | Infected wound | 8 | 16 | 1 | 2 |
| MIC50 | 8 | 1 | |||
| MBC50 | 32 | 2 | |||
|
Class |
Antimicrobials |
FIC | FICI (ΣFIC) |
Effect | |
|---|---|---|---|---|---|
| LyeTx I mnΔK | Antimicrobial | ||||
| Glycopeptide | Vancomycin | 1.00 | 0.50 | 1.50 | Indifferent |
| β-lactam | Oxacillin | 1.00 | 0.02 | 1.02 | Indifferent |
| Antibacterial | MICs (µg/mL) | Fold resensitization | |
|---|---|---|---|
| Not exposed to LyeTx I mnΔK | Exposed to LyeTx I mnΔK | ||
| Vancomycin | 1.0 | 0.5 | 2 |
| Oxacillin | 128 | 64 | 2 |
| LyeTx I mn∆K (mM) | POPG:CL | POPG:CL:Lysyl:POPG | ||||||
| %calcein released |
SD* | kobs (s-1) x10-2 | SD* x10-4 |
%calcein released |
SD* | kobs (s-1) x10-2 | SD* x10-4 |
|
| 4 | 28.51 | 2.50 | 0.78 | 5.01 | 15.98 | 1.06 | 0.629 | 2.23 |
| 8 | 55.11 | 1.83 | 1.01 | 2.82 | 33.74 | 1.01 | 0.615 | 1.56 |
| 18 | 72.23 | 1.49 | 1.19 | 3.81 | 67.73 | 1.30 | 0.755 | 1.13 |
| 32 | 79.45 | 3.82 | 1.46 | 4.36 | 75.17 | 2.20 | 0.853 | 7.10 |
| 64 | 82.17 | 2.04 | 2.16 | 12.5 | 77.23 | 1.99 | 1.225 | 4.89 |
| Sample | Vpep ( L) | VLUVs ( L) | Vbuffer ( L) | [LyeTx I mn K] ( M) |
| 1 | 0 | 400 | 400 | 0 |
| 2 | 4 | 400 | 396 | 5 |
| 3 | 8 | 400 | 392 | 10 |
| 4 | 12 | 400 | 388 | 15 |
| 5 | 16 | 400 | 384 | 20 |
| 6 | 24 | 400 | 376 | 30 |
| 7 | 32 | 400 | 368 | 40 |
| 8 | 40 | 400 | 360 | 50 |
| 9 | 50 | 400 | 350 | 62.5 |
| 10 | 60 | 400 | 340 | 75 |
| Sample | Vpep (μL) | VLUVs (μL) | Vbuffer (μL) | [LyeTx I mnΔK] (μM) |
| 1 | 0 | 150 | 150 | 0 |
| 2 | 5 | 150 | 145 | 4,2 |
| 3 | 10 | 150 | 140 | 8,3 |
| 4 | 20 | 150 | 130 | 16,7 |
| 5 | 40 | 150 | 110 | 33,3 |
| 6 | 80 | 150 | 70 | 66,7 |
| Component | Concentration |
|---|---|
| Hydroxyethylcellulose (Natrosol®) | 2.2% |
| Sodium metabisulfite | 0.6% |
| Methylparaben (Nipagin®) | 0.2% |
| Distilled water | q.s. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




