Submitted:
18 January 2024
Posted:
25 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Theoretical Background and Working Principles of Nanoplasmonic Biosensors
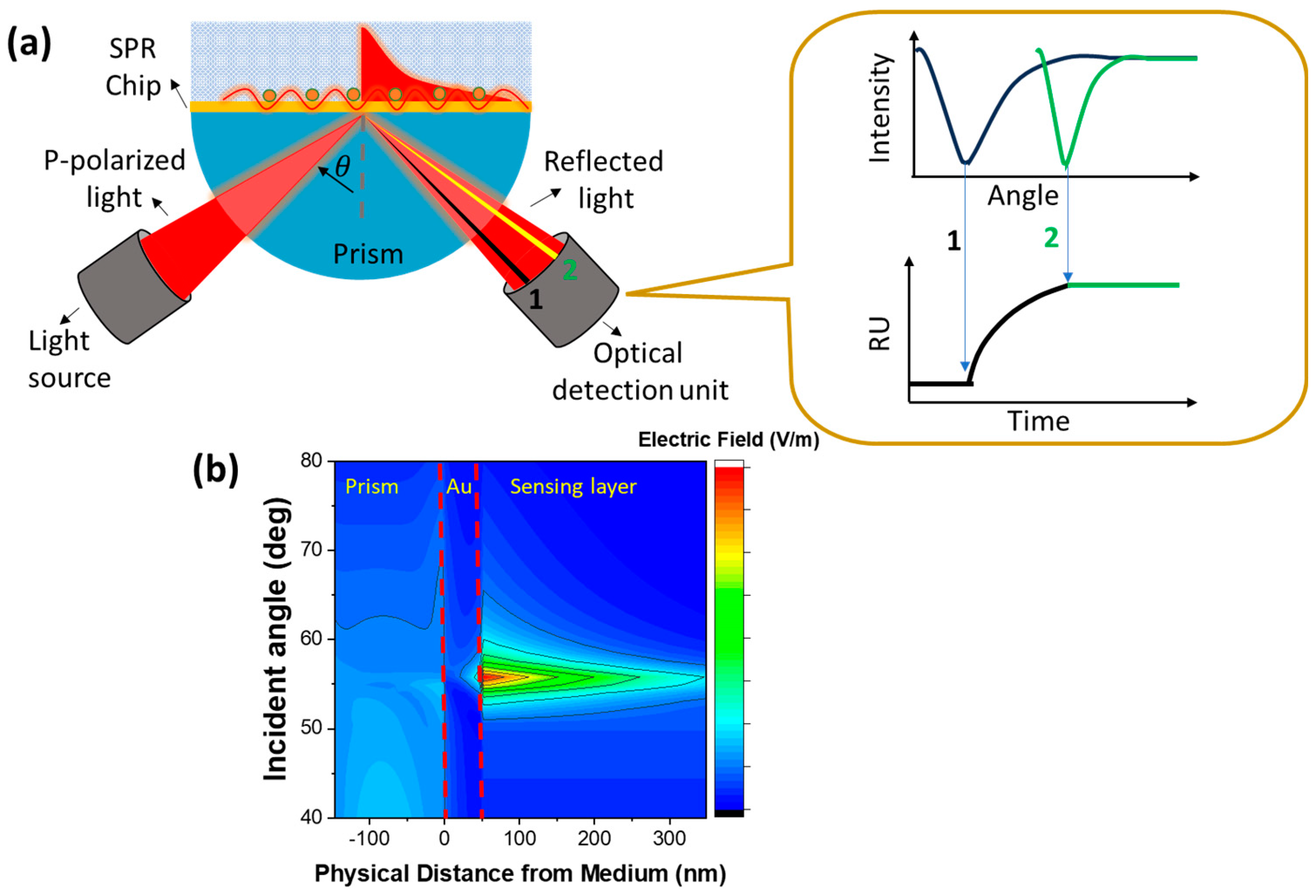
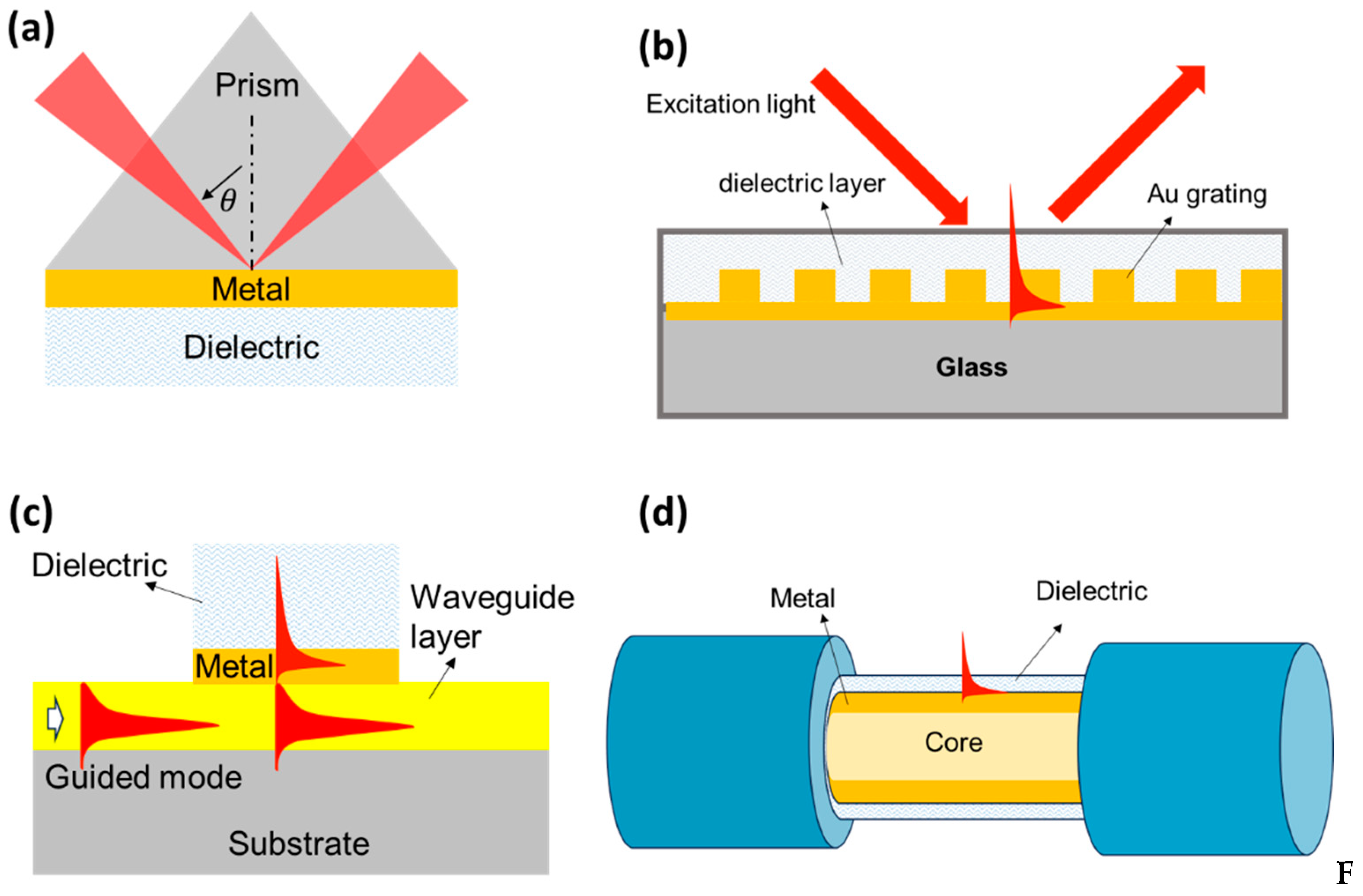
2.1. SPR Based Biosensor
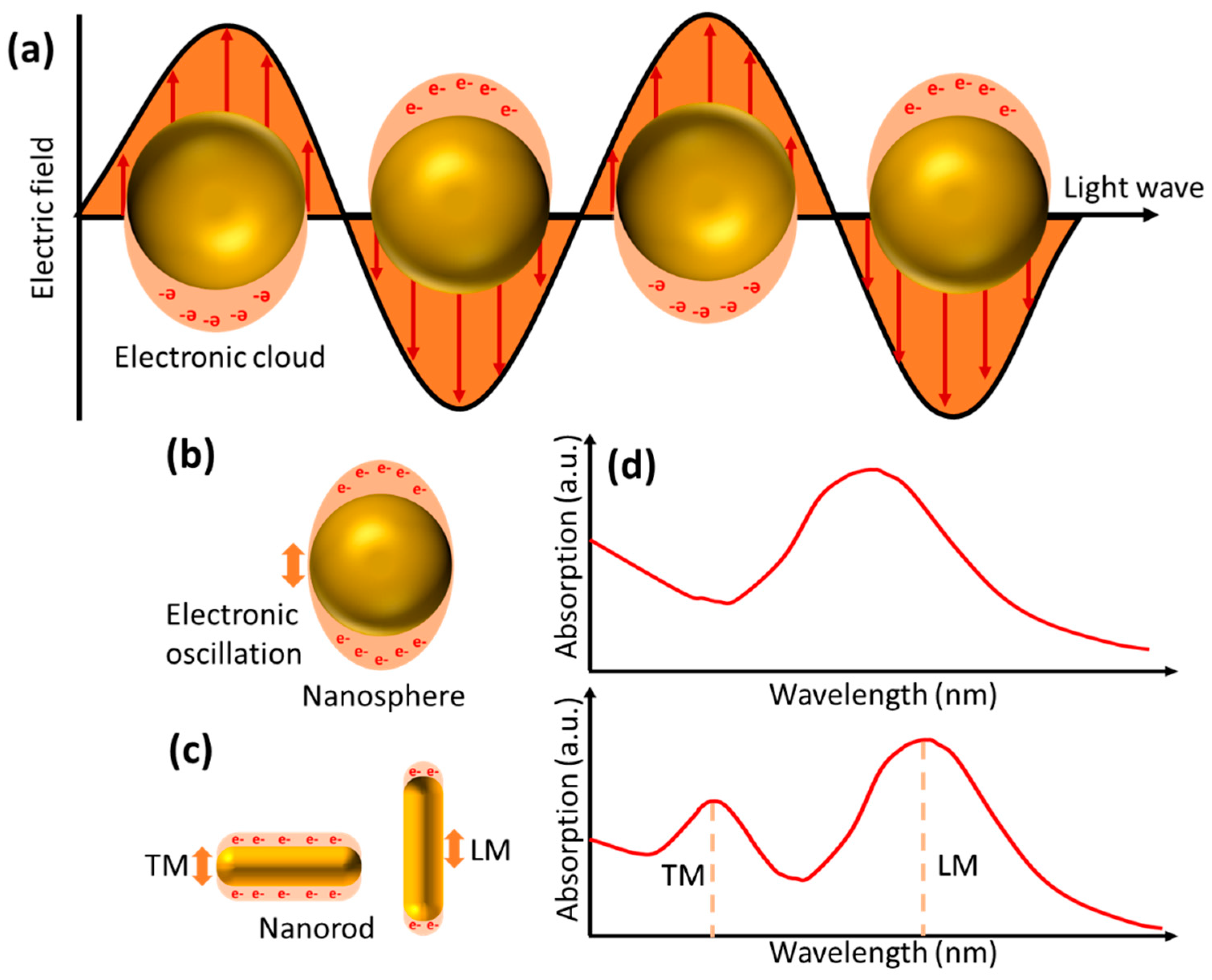
2.2. LSPR Based Biosensor
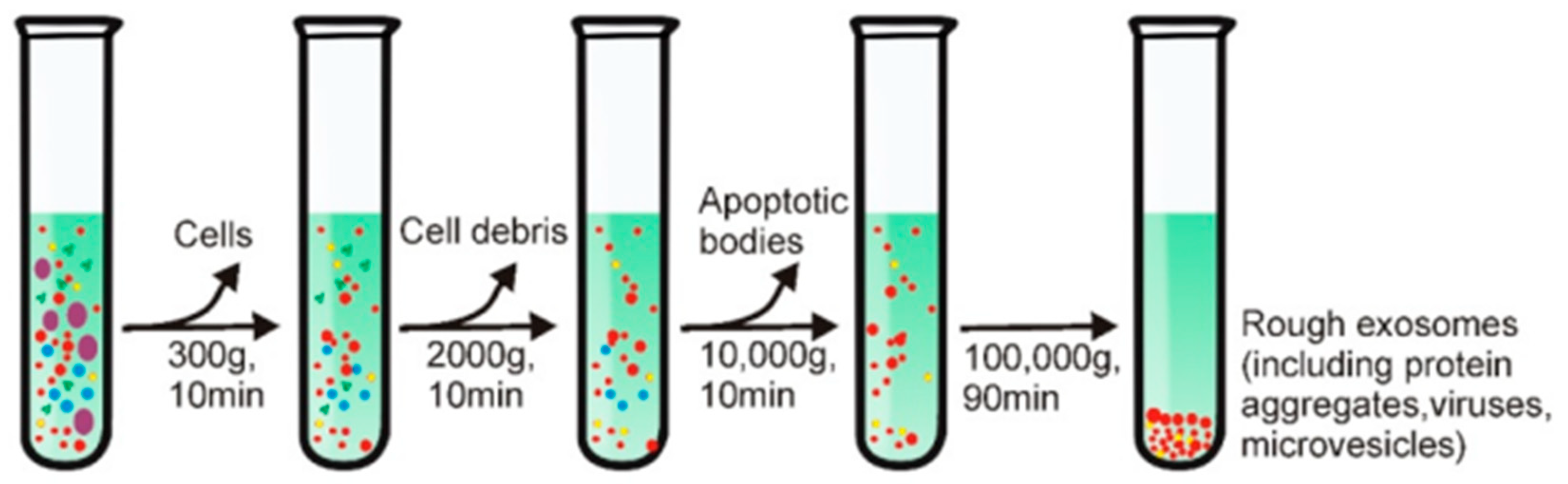
3. Exosomes Isolation Methods
4. Development of Nanoplasmonic Biosensor for Exosome Detection
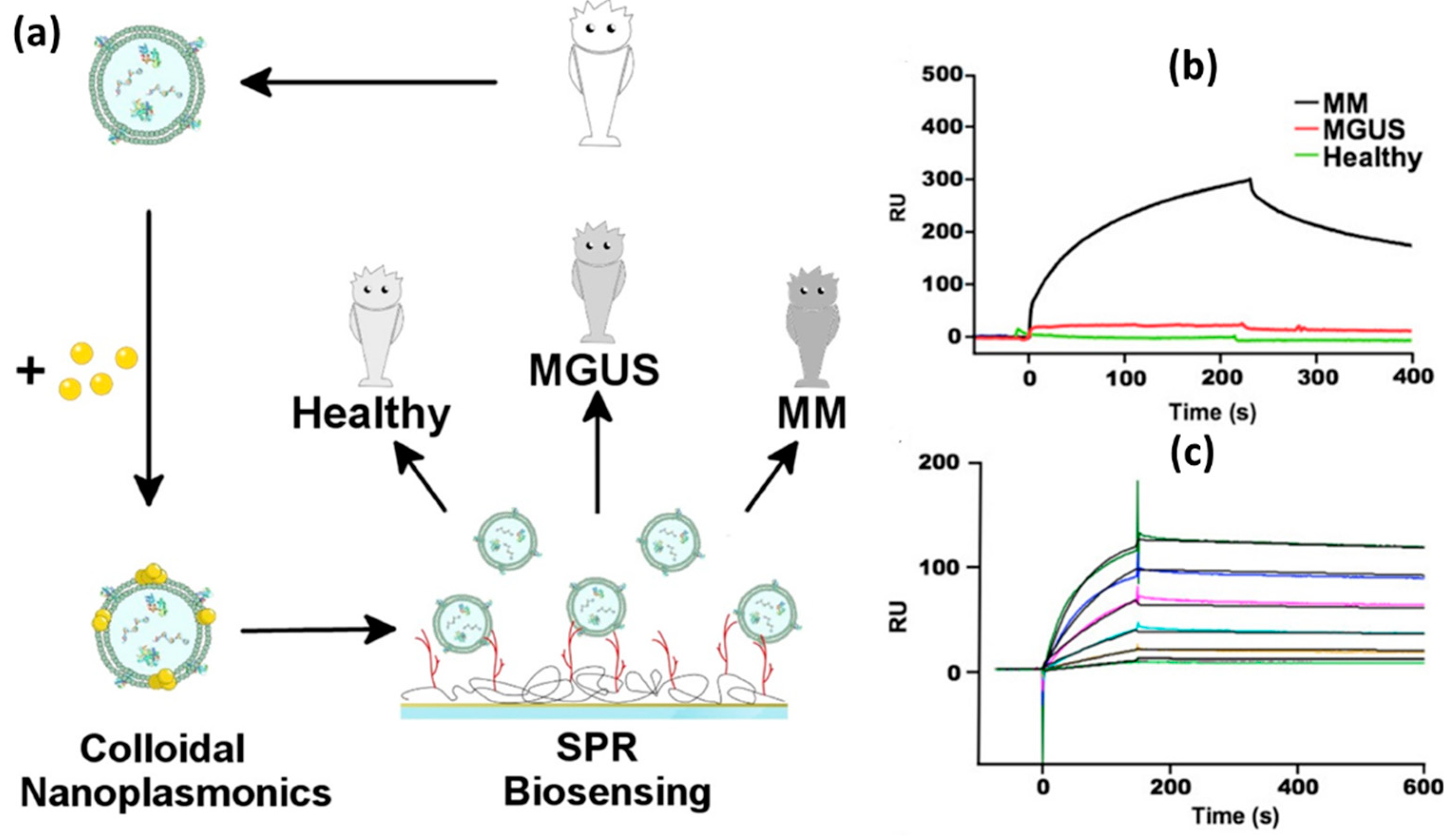
4.1. SPR Based Biosensor
4.2. LSPR Based Biosensor
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cheng, N.; Du, D.; Wang, X.; Liu, D.; Xu, W.; Luo, Y.; Lin, Y. Recent Advances in Biosensors for Detecting Cancer-Derived Exosomes. Trends Biotechnol. 2019, 37, 1236–1254. [Google Scholar] [CrossRef] [PubMed]
- Bari, S.M.I.; Hossain, F.B.; Nestorova, G.G. Advances in Biosensors Technology for Detection and Characterization of Extracellular Vesicles. Sensors 2021, 21, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Shafiei, M.; Ansari, M.N.M.; Razak, S.I.A.; Khan, M.U.A. A Comprehensive Review on the Applications of Exosomes and Liposomes in Regenerative Medicine and Tissue Engineering. Polymers (Basel). 2021, 13, 1–27. [Google Scholar] [CrossRef]
- Sałaga-Zaleska, K.; Kuchta, A.; Bzoma, B.; Chyła-Danił, G.; Safianowska, A.; Płoska, A.; Kalinowski, L.; Dębska-Ślizień, A.; Jankowski, M. Nanoparticle Tracking Analysis of Urinary Extracellular Vesicle Proteins as a New Challenge in Laboratory Medicine. Int. J. Mol. Sci. 2023, 24, 1–14. [Google Scholar] [CrossRef]
- Morales-Kastresana, A.; Jones, J.C. Flow Cytometric Analysis of Extracellular Vesicles. Methods Mol. Biol. 2017, 1545, 215–225. [Google Scholar] [CrossRef]
- Szatanek, R.; Baj-Krzyworzeka, M.; Zimoch, J.; Lekka, M.; Siedlar, M.; Baran, J. The Methods of Choice for Extracellular Vesicles (EVs) Characterization. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef]
- Liu, C.; Qie, Y.; Qin, W.; Zhao, K.; Zhu, J.; Zhao, L.; Li, M.; Guo, L.H. Emerging Immunoassay Technologies for the Rapid Detection of Exosomes. Sensors Actuators, B Chem. 2021, 345, 130336. [Google Scholar] [CrossRef]
- Justino, C.I.L.; Duarte, A.C.; Rocha-Santos, T.A.P. Critical Overview on the Application of Sensors and Biosensors for Clinical Analysis. TrAC Trends Anal. Chem. 2016, 85, 36–60. [Google Scholar] [CrossRef]
- Fathi, F.; Jalili, R.; Amjadi, M.; Rashidi, M.R. SPR Signals Enhancement by Gold Nanorods for Cell Surface Marker Detection. BioImpacts 2019, 9, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Chiu, N.F.; Tai, M.J.; Nurrohman, D.T.; Lin, T.L.; Wang, Y.H.; Chen, C.Y. Immunoassay-Amplified Responses Using a Functionalized Mos2-Based Spr Biosensor to Detect Papp-A2 in Maternal Serum Samples to Screen for Fetal down’s Syndrome. Int. J. Nanomedicine 2021, 16, 2715–2733. [Google Scholar] [CrossRef]
- Nurrohman, D.T.; Wang, Y.-H.; Chiu, N.-F. Exploring Graphene and MoS2 Chips Based Surface Plasmon Resonance Biosensors for Diagnostic Applications. Front. Chem. 2020, 8. [Google Scholar] [CrossRef]
- Chiu, N.F.; Kuo, C.T.; Lin, T.L.; Chang, C.C.; Chen, C.Y. Ultra-High Sensitivity of the Non-Immunological Affinity of Graphene Oxide-Peptide-Based Surface Plasmon Resonance Biosensors to Detect Human Chorionic Gonadotropin. Biosens. Bioelectron. 2017, 94, 351–357. [Google Scholar] [CrossRef]
- Kumar, R.; Agarwal, S.; Pal, S.; Verma, A.; Kumar Prajapati, Y. Refractive Index Sensing Using MXene Mediated Surface Plasmon Resonance Sensor in Visible to near Infrared Regime. Measurement 2024, 224, 113682. [Google Scholar] [CrossRef]
- Shrivastav, A.M.; Cvelbar, U.; Abdulhalim, I. A Comprehensive Review on Plasmonic-Based Biosensors Used in Viral Diagnostics. Commun. Biol. 2021, 4, 1–12. [Google Scholar] [CrossRef]
- Chiu, N.F.; Yang, C. Du; Chen, C.C.; Kuo, C.T. Stepwise Control of Reduction of Graphene Oxide and Quantitative Real-Time Evaluation of Residual Oxygen Content Using EC-SPR for a Label-Free Electrochemical Immunosensor. Sensors Actuators, B Chem. 2018, 258, 981–990. [Google Scholar] [CrossRef]
- Wang, D.S.; Fan, S.K. Microfluidic Surface Plasmon Resonance Sensors: From Principles to Point-of-Care Applications. Sensors (Switzerland) 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Nurrohman, D.T.; Chiu, N.F. Interaction Studies of Localized Surface Plasmon Resonance Immunosensor Based on Gold Nanoparticles. IEEE Sens. J. 2023, 23, 19262–19271. [Google Scholar] [CrossRef]
- Farooq, S.; de Araujo, R.E. Engineering a Localized Surface Plasmon Resonance Platform for Molecular Biosensing. Open J. Appl. Sci. 2018, 08, 126–139. [Google Scholar] [CrossRef]
- Pellas, V.; Hu, D.; Mazouzi, Y.; Mimoun, Y.; Blanchard, J.; Guibert, C.; Salmain, M.; Boujday, S. Gold Nanorods for LSPR Biosensing: Synthesis, Coating by Silica, and Bioanalytical Applications. Biosensors 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Bansal, A.; Sekhon, J.S.; Verma, S.S. Scattering Efficiency and LSPR Tunability of Bimetallic Ag, Au, and Cu Nanoparticles. Plasmonics 2014, 9, 143–150. [Google Scholar] [CrossRef]
- Unser, S.; Bruzas, I.; He, J.; Sagle, L. Localized Surface Plasmon Resonance Biosensing: Current Challenges and Approaches. Sensors (Switzerland) 2015, 15, 15684–15716. [Google Scholar] [CrossRef]
- Prajna, N.D.; Santhosh, C.; Tom, D.; Rajeev, K.S. LSPR-Based Sensing of Insulin Using Gold Nanostars. AIP Conf. Proc. 2023, 2901. [Google Scholar] [CrossRef]
- Gao, J.; Li, A.; Hu, J.; Feng, L.; Liu, L.; Shen, Z. Recent Developments in Isolating Methods for Exosomes. Front. Bioeng. Biotechnol. 2023, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Kaslan, M.; Lee, S.H.; Yao, J.; Gao, Z. Progress in Exosome Isolation Techniques. Theranostics 2017, 7, 789–804. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Zhang, W.; Zhang, H.; Zhang, F.; Chen, L.; Ma, L.; Larcher, L.M.; Chen, S.; Liu, N.; Zhao, Q.; et al. Progress, Opportunity, and Perspective on Exosome Isolation - Efforts for Efficient Exosome-Based Theranostics. Theranostics 2020, 10, 3684–3707. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.L.; Zhu, J.; Liu, J.X.; Jiang, F.; Ni, W.K.; Qu, L.S.; Ni, R.Z.; Lu, C.H.; Xiao, M.B. A Comparison of Traditional and Novel Methods for the Separation of Exosomes from Human Samples. Biomed Res. Int. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Liu, W. zhao; Ma, Z. jun; Kang, X. wen Current Status and Outlook of Advances in Exosome Isolation. Anal. Bioanal. Chem. 2022, 414, 7123–7141. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Li, Z.; Cheng, W.; Wu, T.; Li, J.; Li, X.; Liu, L.; Bai, H.; Ding, S.; Li, X.; et al. Surface Plasmon Resonance Biosensor for Exosome Detection Based on Reformative Tyramine Signal Amplification Activated by Molecular Aptamer Beacon. J. Nanobiotechnology 2021, 19, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhu, Y.; Guan, M.; Liu, Y.; Lv, M.; Zhang, C.; Zhang, H.; Zhang, Z. Isolation of Circulating Exosomes and Identification of Exosomal PD-L1 for Predicting Immunotherapy Response. Nanoscale 2022, 14, 8995–9003. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.; Zhao, J.; Chen, J.; Hu, X.; Koh, K.; Chen, H. A Simple and Direct SPR Platform Combining Three-in-One Multifunctional Peptides for Ultra-Sensitive Detection of PD-L1 Exosomes. Sensors Actuators B Chem. 2021, 346, 130496. [Google Scholar] [CrossRef]
- Di Noto, G.; Bugatti, A.; Zendrini, A.; Mazzoldi, E.L.; Montanelli, A.; Caimi, L.; Rusnati, M.; Ricotta, D.; Bergese, P. Merging Colloidal Nanoplasmonics and Surface Plasmon Resonance Spectroscopy for Enhanced Profiling of Multiple Myeloma-Derived Exosomes. Biosens. Bioelectron. 2016, 77, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zou, L.; Yang, X.; Liu, X.; Nie, W.; Zheng, Y.; Cheng, Q.; Wang, K. Direct Quantification of Cancerous Exosomes via Surface Plasmon Resonance with Dual Gold Nanoparticle-Assisted Signal Amplification. Biosens. Bioelectron. 2019, 135, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Liu, Y.; Wang, Y.; Hu, X.; Koh, K.; Chen, H. Tunable Au@SiO2/Au Film Metasurface as Surface Plasmon Resonance Enhancer for Direct and Ultrasensitive Detection of Exosomes. Anal. Chem. 2023, 95, 9663–9671. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Wang, X.; Li, F.; Xie, Y.; Shen, J.; Wang, X.; Huang, Y.; Lin, S.; Chen, J.; Zhang, L.; et al. Label-Free Plasmonic Metasensing of PSA and Exosomes in Serum for Rapid High-Sensitivity Diagnosis of Early Prostate Cancer. Biosens. Bioelectron. 2023, 235, 115380. [Google Scholar] [CrossRef]
- Zhang, P.; Jiang, J.; Zhou, X.; Kolay, J.; Wang, R.; Wan, Z.; Wang, S. Label-Free Imaging and Biomarker Analysis of Exosomes with Plasmonic Scattering Microscopy. Chem. Sci. 2022, 367, 12760–12768. [Google Scholar] [CrossRef]
- Sina, A.A.I.; Vaidyanathan, R.; Wuethrich, A.; Carrascosa, L.G.; Trau, M. Label-Free Detection of Exosomes Using a Surface Plasmon Resonance Biosensor. Anal. Bioanal. Chem. 2019, 411, 1311–1318. [Google Scholar] [CrossRef]
- Liao, G.; Liu, X.; Yang, X.; Wang, Q.; Geng, X.; Zou, L.; Liu, Y.; Li, S.; Zheng, Y.; Wang, K. Surface Plasmon Resonance Assay for Exosomes Based on Aptamer Recognition and Polydopamine-Functionalized Gold Nanoparticles for Signal Amplification. Microchim. Acta 2020, 187, 1–9. [Google Scholar] [CrossRef]
- Wang, Y.; Mao, Z.; Chen, Q.; Koh, K.; Hu, X.; Chen, H. Rapid and Sensitive Detection of PD-L1 Exosomes Using Cu-TCPP 2D MOF as a SPR Sensitizer. Biosens. Bioelectron. 2022, 201, 113954. [Google Scholar] [CrossRef] [PubMed]
- Qiu, G.; Thakur, A.; Xu, C.; Ng, S.; Lee, Y.; Wu, C.L. Detection of Glioma-Derived Exosomes with the Biotinylated Antibody-Functionalized Titanium Nitride Plasmonic Biosensor. Adv. Funct. Mater. 2019, 29. [Google Scholar] [CrossRef]
- Chen, W.; Li, J.; Wei, X.; Fan, Y.; Qian, H.; Li, S.; Xiang, Y.; Ding, S. Surface Plasmon Resonance Biosensor Using Hydrogel-AuNP Supramolecular Spheres for Determination of Prostate Cancer-Derived Exosomes. Microchim. Acta 2020, 187. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.M.; Park, J.S.; Jung, S.W.; Yeom, J.; Yoo, S.M. =Biosensing Applications Using Nanostructure-Based Localized Surface Plasmon Resonance Sensors. Sensors 2021, 21, 1–27. [Google Scholar] [CrossRef]
- Song, S.; Lee, J.U.; Jeon, M.J.; Kim, S.; Sim, S.J. Detection of Multiplex Exosomal MiRNAs for Clinically Accurate Diagnosis of Alzheimer’s Disease Using Label-Free Plasmonic Biosensor Based on DNA-Assembled Advanced Plasmonic Architecture. Biosens. Bioelectron. 2022, 199. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Huang, C.-H.; Gao, Z.; Shen, J.; He, J.; MacLachlan, A.; Ma, C.; Chang, Y.; Yang, W.; Cai, Y.; et al. Nanoplasmonic Sandwich Immunoassay for Tumor-Derived Exosome Detection and Exosomal PD-L1 Profiling. ACS Sensors 2021, 6, 3308–3319. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiao, J.; Wei, Y.; Wang, D.; Yang, C.; Xu, Z. Plasmonic Colorimetric Biosensor for Sensitive Exosome Detection via Enzyme-Induced Etching of Gold Nanobipyramid@MnO 2 Nanosheet Nanostructures. Anal. Chem. 2020, 92, 15244–15252. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, D.; Yue, S.; Lu, Y.; Yang, C.; Fang, J.; Xu, Z. Sensitive Multicolor Visual Detection of Exosomes via Dual Signal Amplification Strategy of Enzyme-Catalyzed Metallization of Au Nanorods and Hybridization Chain Reaction. ACS Sensors 2019, 4, 3210–3218. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.; Geng, Z.; Su, Y.; Fan, Z.; Wang, S.; Fang, W.; Chen, H. Label-Free Exosome Detection Based on a Low-Cost Plasmonic Biosensor Array Integrated with Microfluidics. Langmuir 2019, 35, 9816–9824. [Google Scholar] [CrossRef]
- Wang, Q.L.; Huang, W.X.; Zhang, P.J.; Chen, L.; Lio, C.K.; Zhou, H.; Qing, L. Sen; Luo, P. Colorimetric Determination of the Early Biomarker Hypoxia-Inducible Factor-1 Alpha (HIF-1α) in Circulating Exosomes by Using a Gold Seed-Coated with Aptamer-Functionalized Au@Au Core-Shell Peroxidase Mimic. Microchim. Acta 2020, 187, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Thakur, A.; Qiu, G.; NG, S.P.; Guan, J.; Yue, J.; Lee, Y.; Wu, C.M.L. Direct Detection of Two Different Tumor-Derived Extracellular Vesicles by SAM-AuNIs LSPR Biosensor. Biosens. Bioelectron. 2017, 94, 400–407. [Google Scholar] [CrossRef]
- Li, H.; Huang, T.; Lu, L.; Yuan, H.; Zhang, L.; Wang, H.; Yu, B. Ultrasensitive Detection of Exosomes Using an Optical Microfiber Decorated with Plasmonic MoSe2-Supported Gold Nanorod Nanointerfaces. ACS Sensors 2022, 7, 1926–1935. [Google Scholar] [CrossRef]
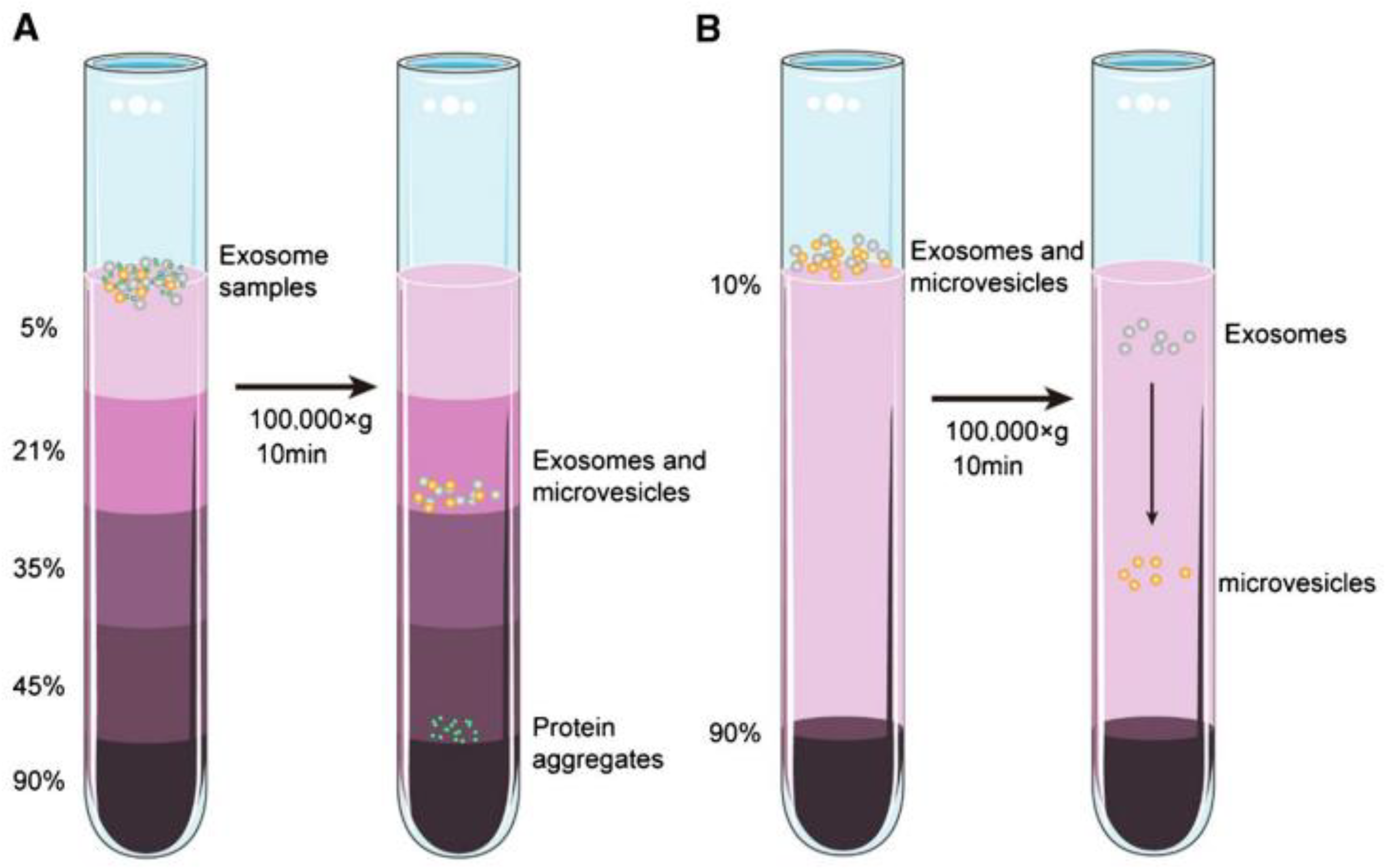
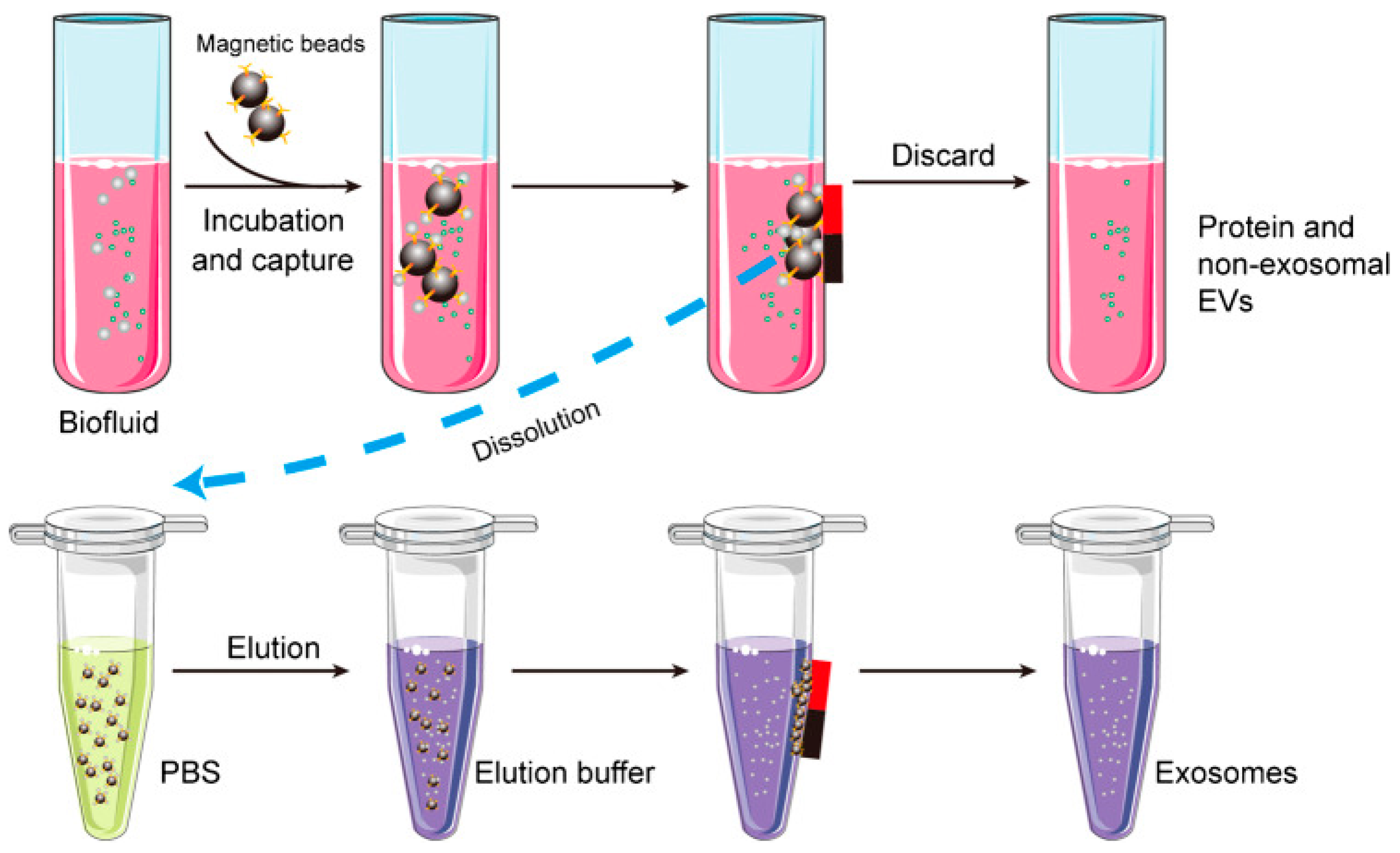
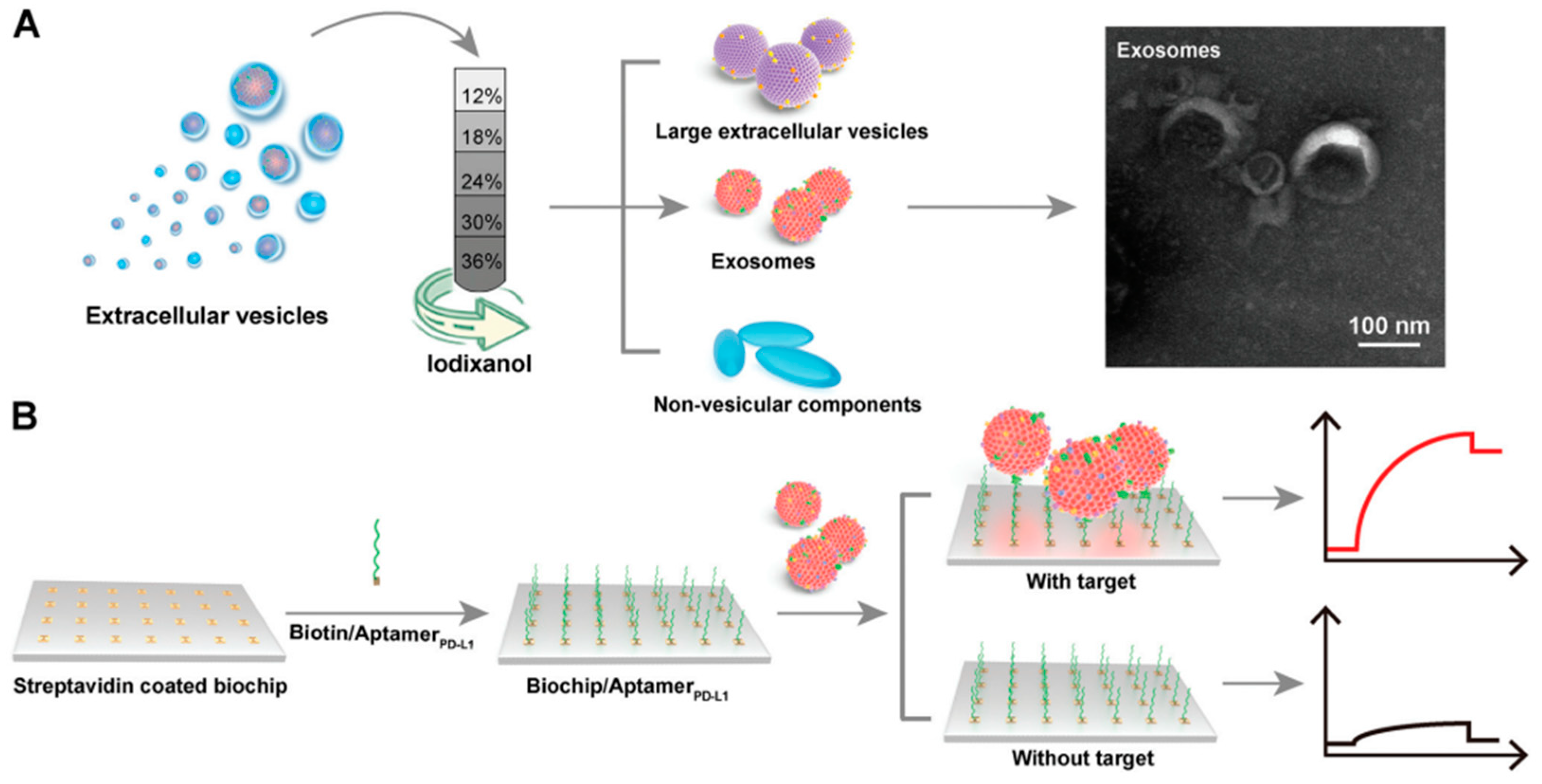
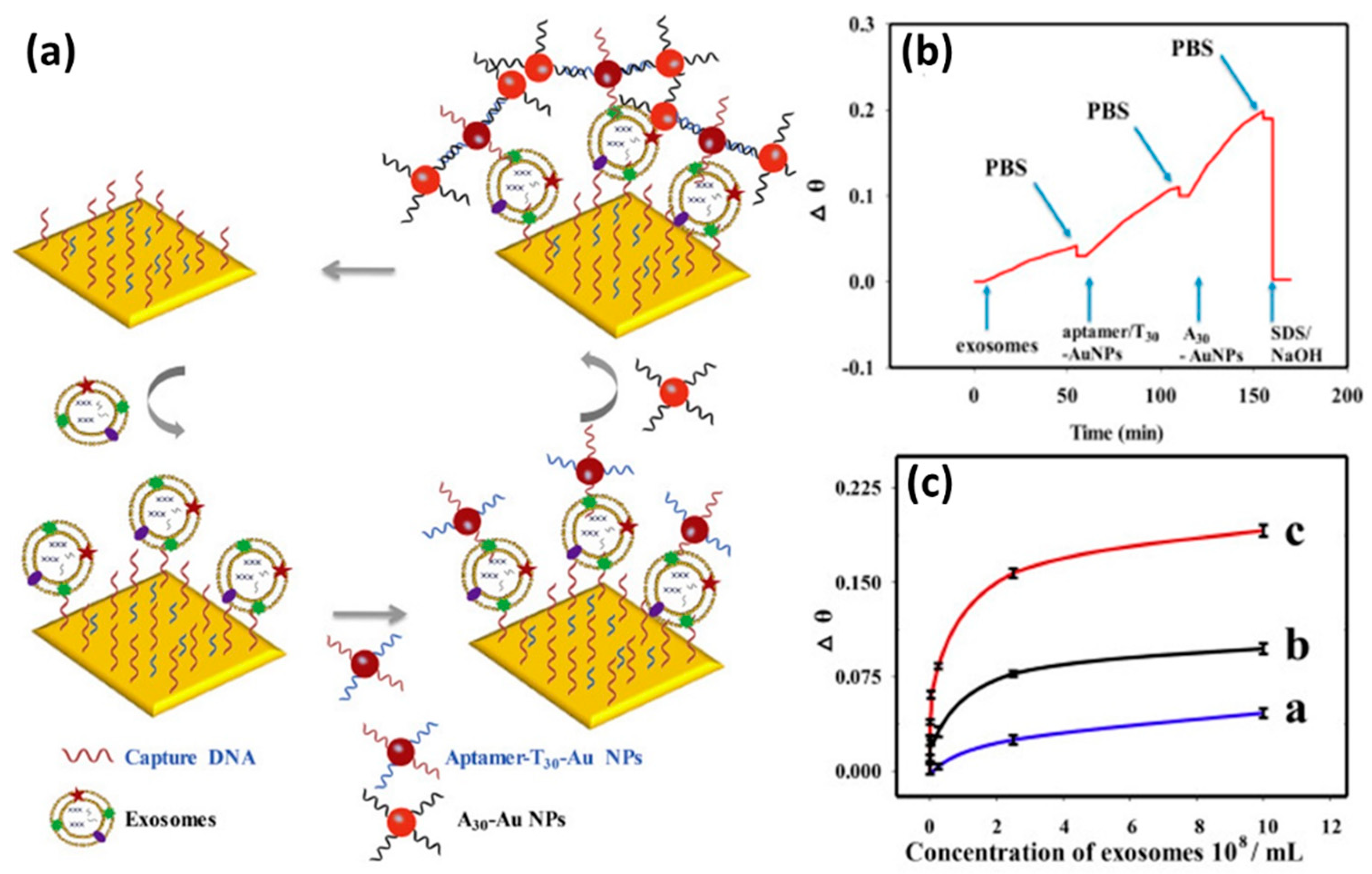
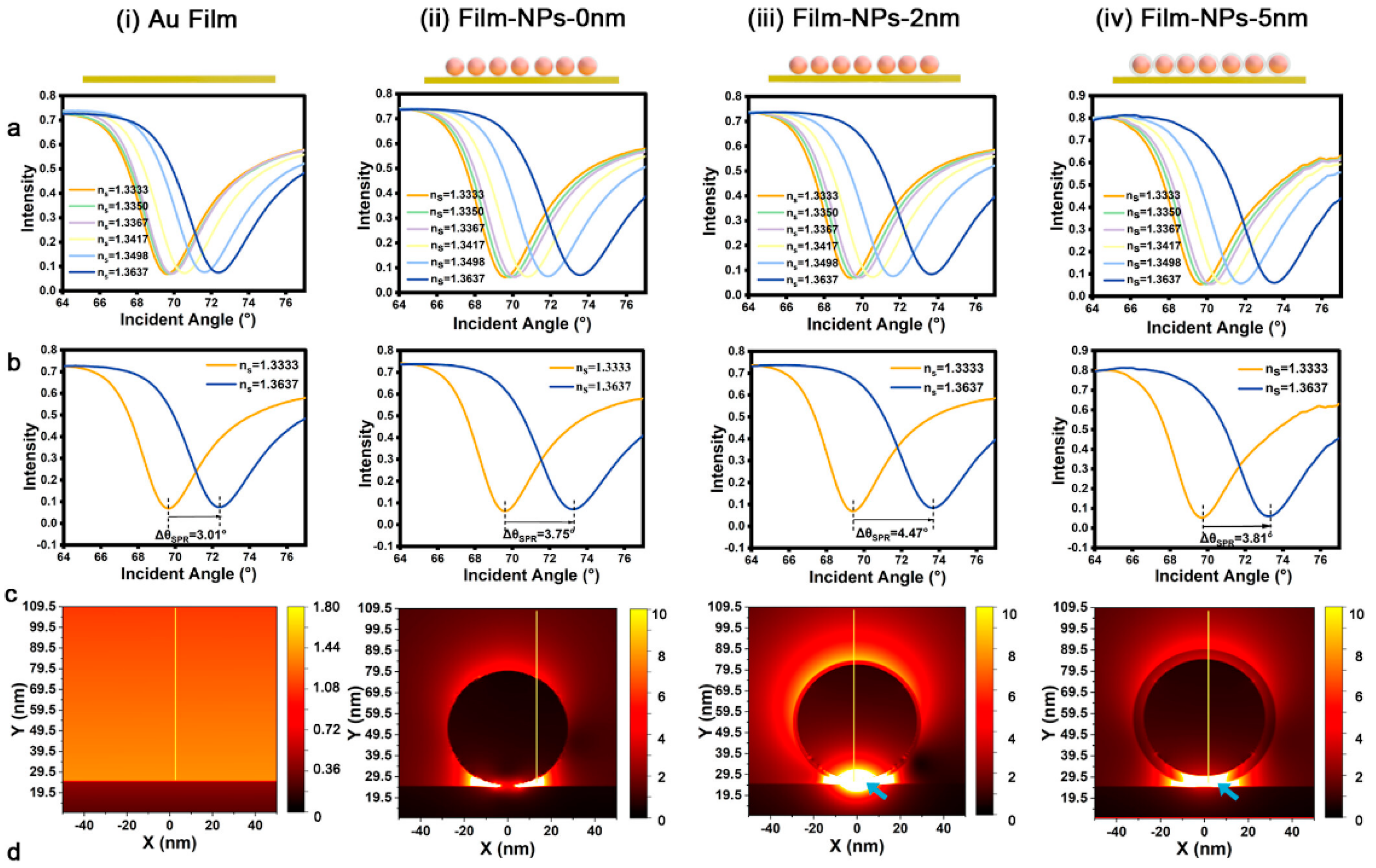
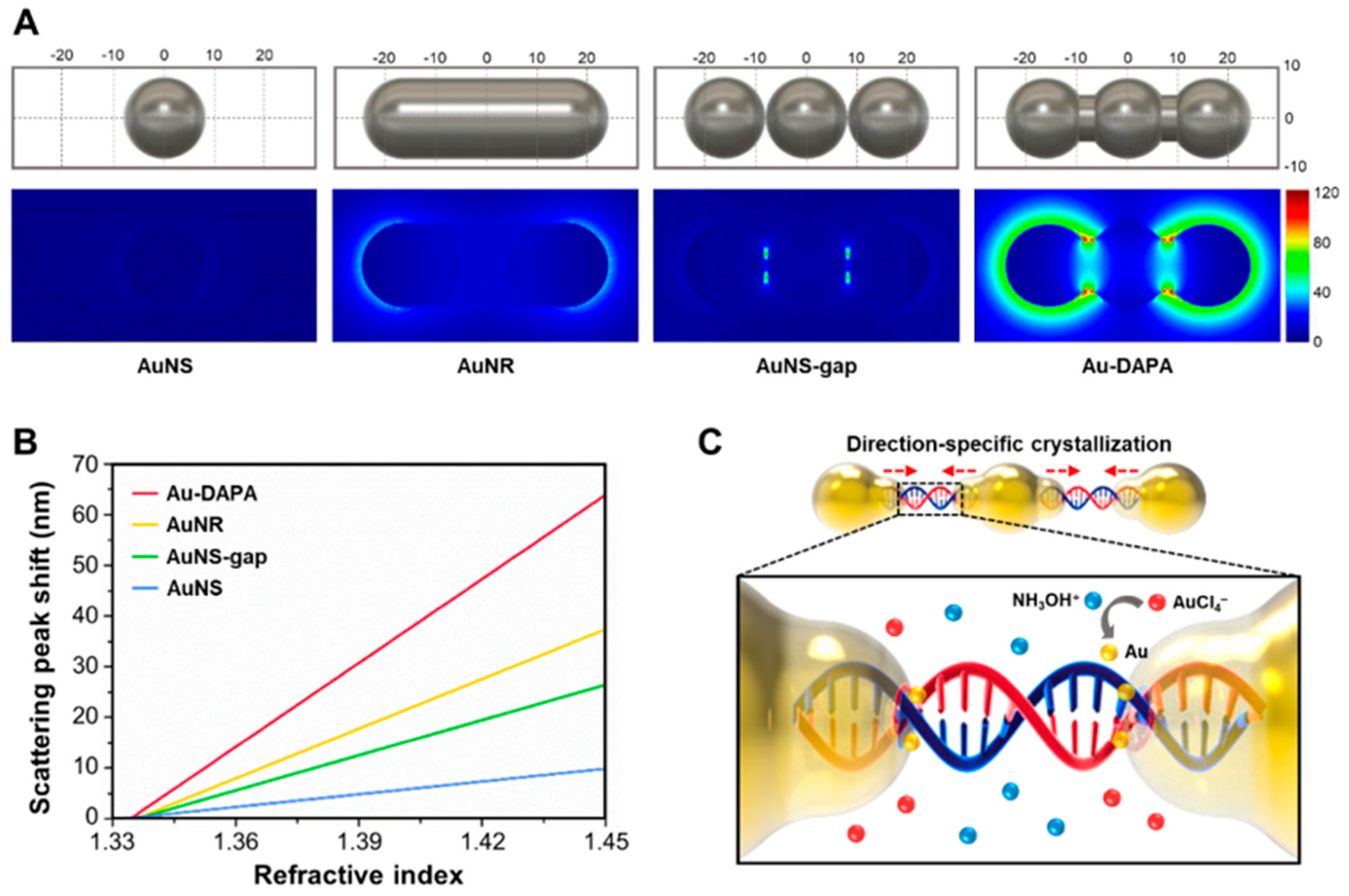
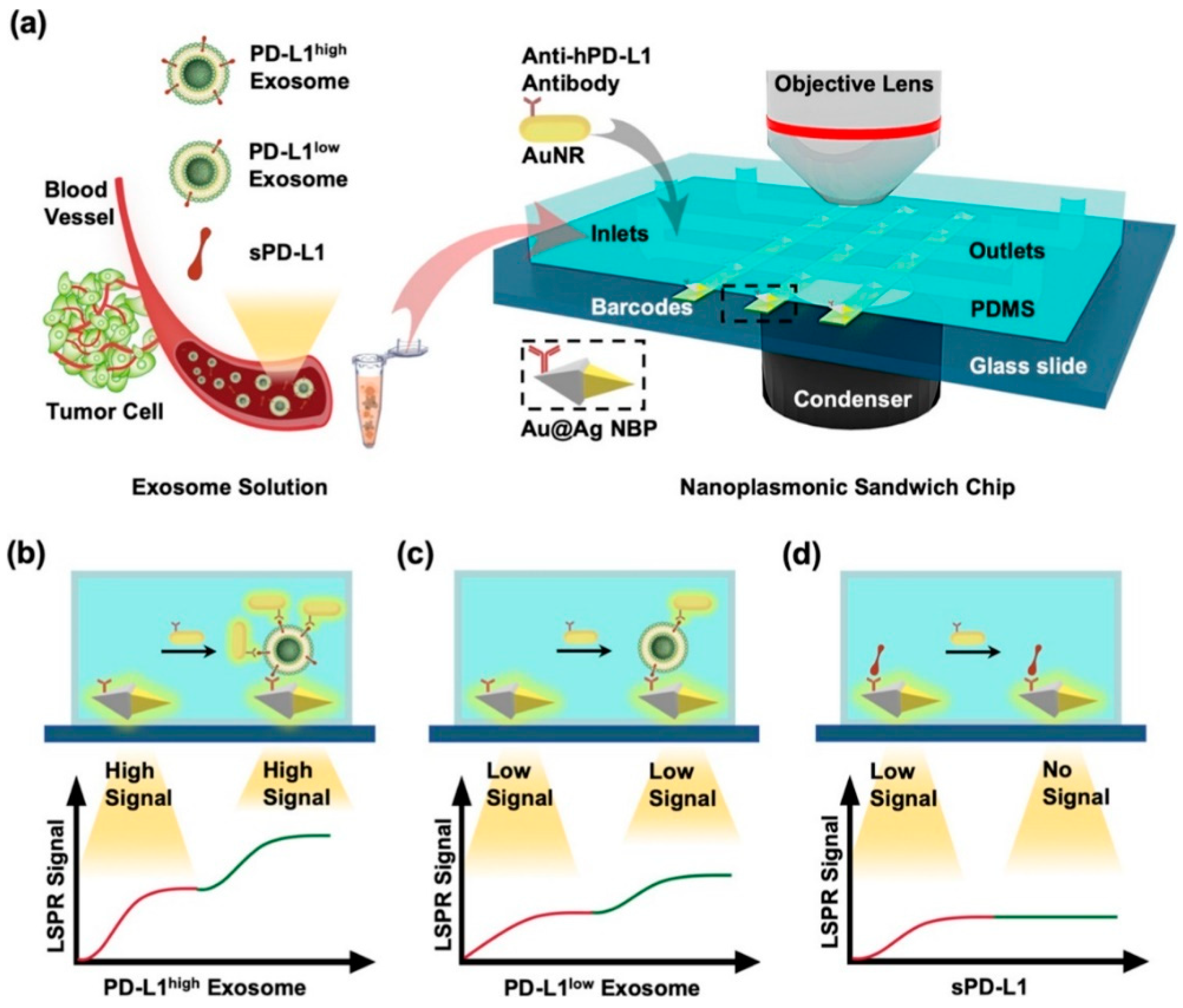
| Isolation method | Principle | Advantages | Disadvantages |
|---|---|---|---|
| differential ultracentrifugation | Size and density | Relatively high purity, widely used, large sample capacity | Time consuming, exosome damage, large sample volumes, expensive instrumentation |
| gradient density ultracentrifugation | Size and density | High purity, high recovery rate | Time consuming, exosome damage, large sample volumes |
| Chromatography | Size | Cost and time saving, Fast and simple, Maintain the biological activity and integrity of exosomes | Specialized instrumentation, Nanoscale contaminants (e.g., lipoproteins) |
| Immunoaffinity capture | Specific binding | High purity, Sub populations can be isolated, Can be integrated with detection systems | Expensive, Low yield, Nanoscale contaminants, Intermolecular cross reactions |
| microfluidics | Specific binding, size and density | Fast, low cost, portable, easy automation and integration, high portability. | Lack of standardization and large scale tests on clinical samples, lack of method validation, moderate to low sample capacity |
| Recognition Element |
Specific target | Developed biosensor system | Detection limit | Ref. |
|---|---|---|---|---|
| Anti-HER2 | HER2 (+) Exosome | Conventional SPR chip was functionalized with anti-HER2 | 0.828×104 exosomes/μL | [36] |
| Aptamer | exosome PD-L1 | Exosome detection was carried out by utilizing the interaction of streptavidin and biotin using a conventional SPR chip | 44.50 pM | [29] |
| Peptide | PD-L1 exosomes | Gold-based SPR chips deposited with graphene | 20 particles/mL | [30] |
| Heparin | multiple myeloma (MM) |
The SPR signal was amplified with Au NPs | 0.06 nM | [31] |
| Aptamer | hepatic carcinoma SMMC-7721 | The SPR signal was amplified using AuNPs coated with polydopamine | 5.6×105 particles/mL | [37] |
| molecular aptamer beacon (MAB) | HER2-positive exosomes | The SPR signal was amplified with AuNPs coated with tyramine | 1×104 particles/mL | [28] |
| Peptide | PD-L1 exosomes | SPR chip was deposited with a 2D metal organic framework | 16.7 particles/mL | [38] |
| DNA | MCF-7 breast cancer cells | SPR biosensor with dual AuNP-assisted signal amplification | 5×103 exosomes/mL | [32] |
| Biotinylated antibody | epidermal growth factor receptor variant-III | SPR Chip based on Titanium nitride (TiN) | 2.75 × 10−3 µg/mL | [39] |
| aptamer-DNA linker | LNCaP | SPRi with signal amplification with hydrogel-AuNP supramolecular sphere | 1×105 particles/mL | [40] |
| Recognition Element |
Specific target | Developed biosensor system | Detection limit | Ref. |
|---|---|---|---|---|
| CD63 aptamer | CD63 | Colorimetric biosensor where exosome quantification is based on metallization of Au NRs and hybridization chain reaction (HCR) | 1.6 × 102 particles/Μl by UV−vis spectroscopy and 9 × 103 particles/μL by naked eyes | [45] |
| anti-CD63 | exosome transmembrane protein CD63 | LSPR biosensor based on gold nano-ellipsoid arrays integrated with microfluidics | 1 ng/mL | [46] |
| HIF-1α- aptamer | HIF-1α | Au NPs with a diameter of 13 nm were functionalized with aptamer. The bond between the ligand and the analyte results in changes in the absorbance intensity. | 0.2 ng/L | [47] |
| - | A-549 and SH-SY5Y cells | LSPR biosensor with self-assembly gold nanoislands (SAM-AuNIs) | 0.194 µg/ml | [48] |
| locked nucleic acid (LNA) | exo-miR-125b | DNA-assembled advanced plasmonic architecture (DAPA)-based plasmonic biosensor | 10.54 aM | [42] |
| anti-hPD-L1 antibody | PD-L1 exosomes | nanoplasmonic sandwich composed of Au@Ag core shell nanobipyramid (NBP) and AuNR | 1.2 × 103 particles/μL | [43] |
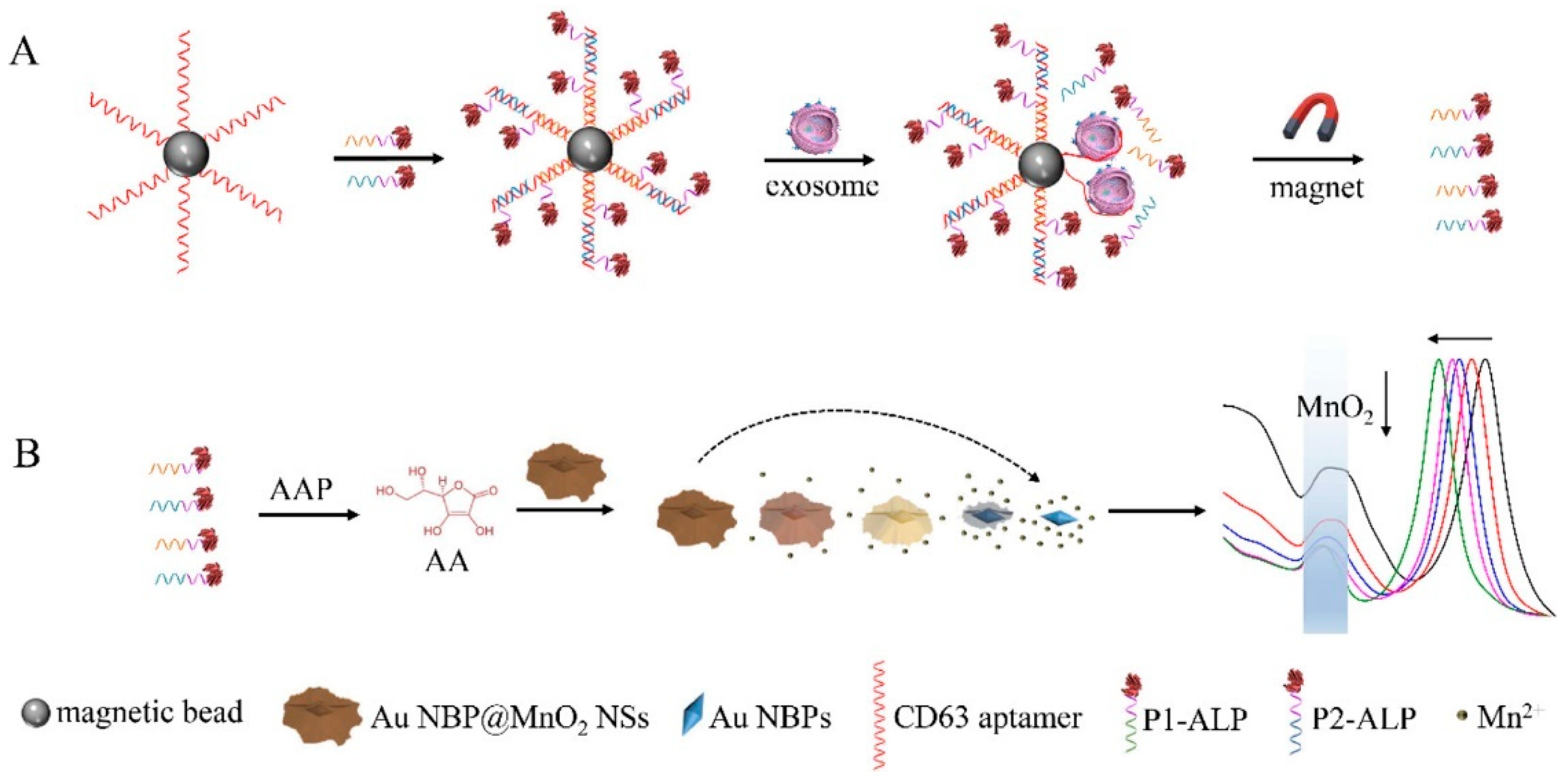
| CD63 aptamer | CD63 | colorimetric biosensors based on Au NBP@MnO2 nanostructures | 1.35 × 102 particles/μL | [44] |
| CA9 Aptamer | Clear Cell Renal Cancer Exosome | optical microfiber integrated with -supported Au NRs | 9.32 particles/mL | [49] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




