Submitted:
23 January 2024
Posted:
24 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
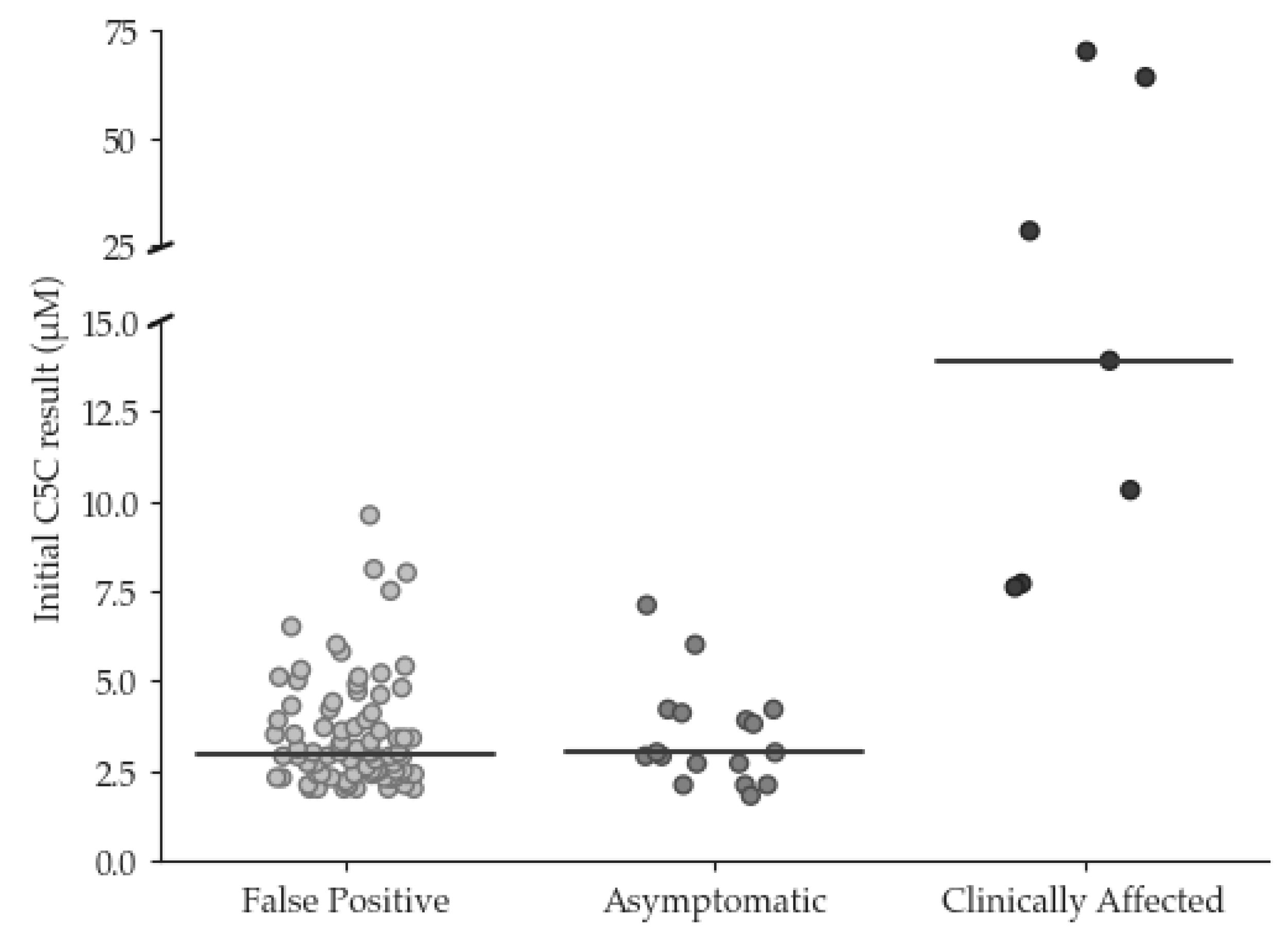
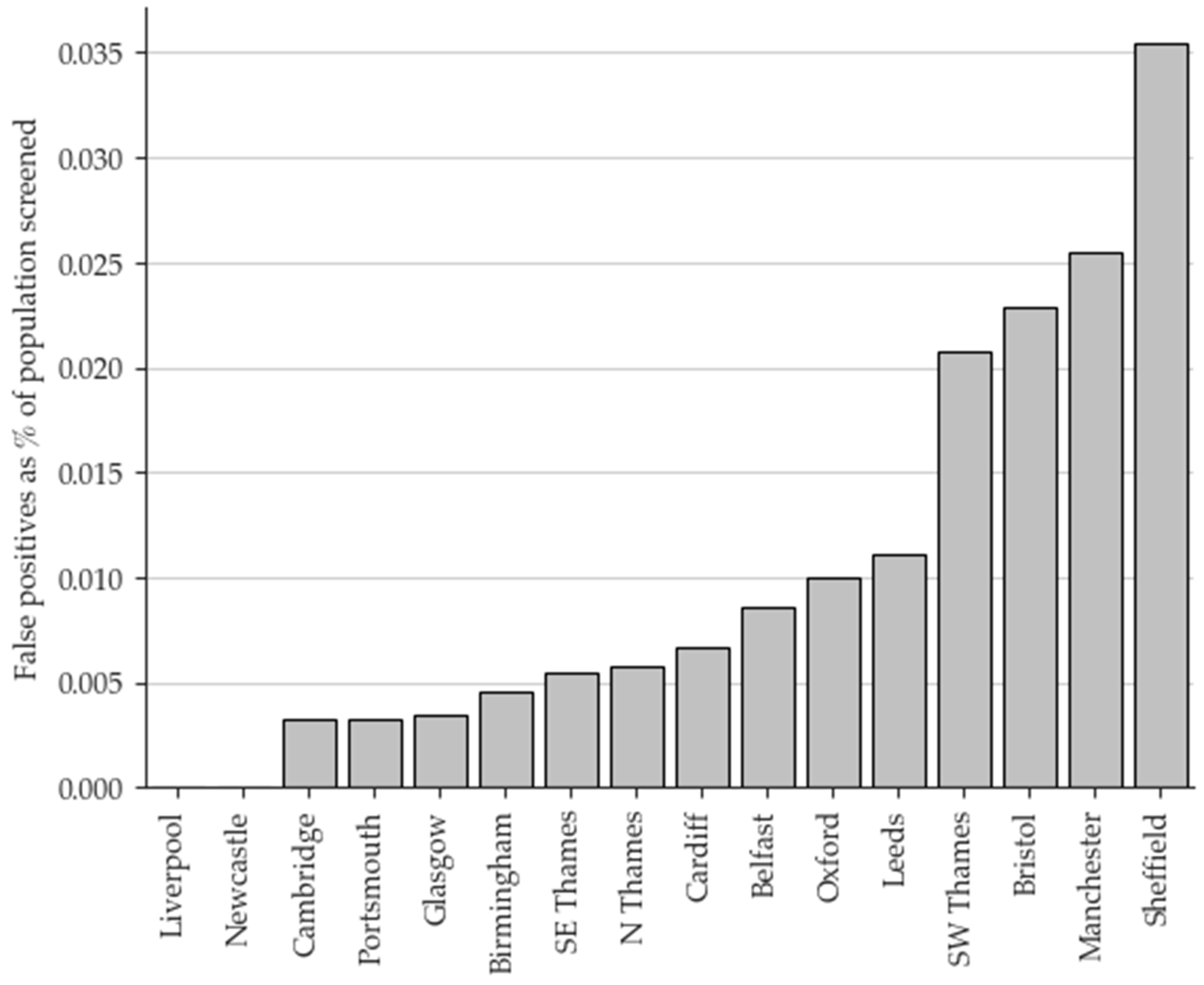
3.1. Retrospective review of condition suspected IVA results
3.2. Clinical outcome questionnaire
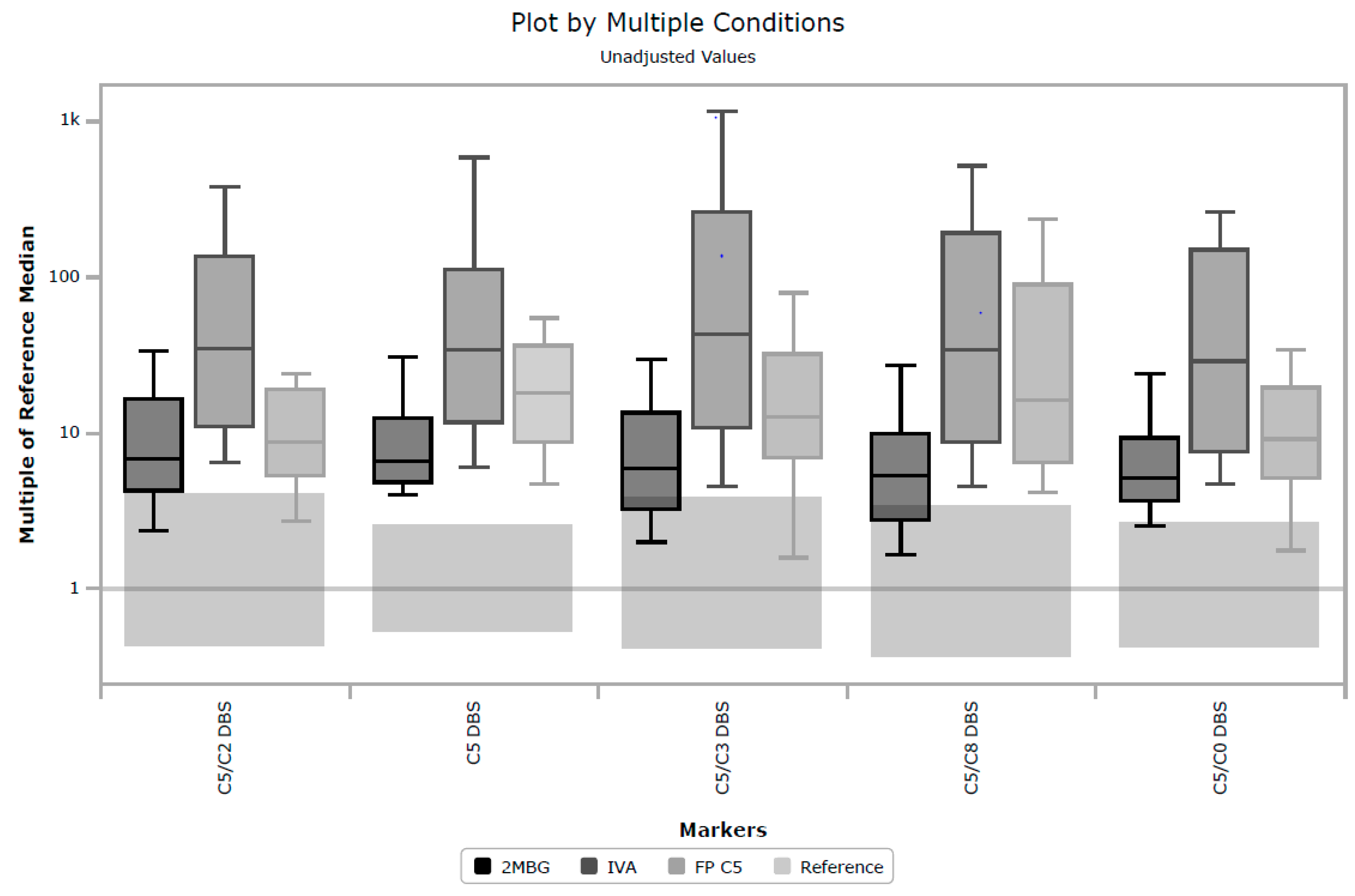
3.3. Evaluation of precision newborn screening
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
References
- Abdenur, J.E.; Chamoles, N.A.; Guinle, A.E.; Schenone, A.B.; Fuertes, A.N. False positive result due to pivaloylcarnitine in a newborn screening programme. J Inherit Metab Dis 1998, 21, 624–630. [Google Scholar] [CrossRef] [PubMed]
- Boemer, F.; Schoos, R.; de Halleux, V.; Kalenga, M.; Debray, F. Surprising causes of C5-carnitine false positive results in newborn screening. Mol Gen Metab 2014, 111, 52–54. [Google Scholar] [CrossRef] [PubMed]
- Bonham, J.R.; Carling, R.S.; Lindner, M.; Franzson, L.; Zetterstrom, R.; Boemer, F.; Cerone, R.; Eyskens, F.; Vilarinho, L.; Hougaard, D.M.; Schielen, P.C.J.I. Raising awareness of false positive newborn screening results arising from pivalate-containing creams and antibiotics in Europe when screening for isovaleric acidemia. Int Journ Neonatal Screen. 2018, 4, 8. [Google Scholar] [CrossRef] [PubMed]
- Carling, R.S.; Burden, D.; Hutton, I.; Randle, R.; John, C.; Bonham, J.R. Introduction of a simple second tier screening test for C5 isobars in dried blood spots: Reducing the false positive rate for isovaleric acidemia in expanded newborn screening. J Inherit Meta. Dis 2018, 38, 75–80. [Google Scholar] [CrossRef]
- OpenPrescribing. Available online: https://openprescribing.net/ (accessed on 4th December 2023).
- Mütze, U.; Henze, L.; Gleich, F.; Lindner, M.; Grünert S., C.; Spiekerkoetter, U.; Santer, R.; Blessing, H.; Thimm, E.; Ensenauer, R.; Weigel, J.; et al. Newborn screening and disease variants predict neurological outcome in isovaleric aciduria. J Inherit Metab Dis 2021, 44, 857–870. [Google Scholar] [CrossRef] [PubMed]
- Hall, P.L.; Marquardt, G.; McHugh, D.M.S.; Currier, R.J.; Tang, H.; Stoway, S.D.; Rinaldo, P. Postanalytical tools improve performance of newborn screening by tandem mass spectrometry. Genet Med 2014, 16, 889–895. [Google Scholar] [CrossRef] [PubMed]
- Rowe, A.D.; Stoway, S.D.; Åhlman, H.; Arora, V.; Caggana, M.; Fornari, A.; Hagar, A.; Hall, P.L.; Marquardt, G.C.; Miller, B.J.; et al. A Novel Approach to Improve Newborn Screening for Congenital Hypothyroidism by Integrating Covariate-Adjusted Results of Different Tests into CLIR Customized Interpretive Tools. Int J Neonatal Screen 2021, 7, 23. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.A.M.; Patnick, J. Blanks R.G. Maximising benefit and minimising harm of screening. BMJ 2008, 336, 480-3. [Google Scholar] [CrossRef] [PubMed]
- Forni, S.; Fu, X.; Palmer, S.E.; Sweetman, L. Rapid determination of C4-acylcarnitine and C5-acylcarnitine isomers in plasma and dried blood spots by UPLC-MS/MS as a second-tier test following flow-injection MS/MS acylcarnitine profile analysis. Mol Genet Metab. 2010, 101, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Cloppenborg, T.; Janzen, N.; Wagner, H.; Steuerwald, U.; Peter, M.; Das, A. Application of a second-tier newborn screening assay for C5 isoforms. JIMD Rep 2014, 13, 23-6. [Google Scholar] [CrossRef]
- Minkler, P.E.; Stoll, M.S.K.; Ingalls, S.T.; Hoppel, C.L. Selective and accurate C5 acylcarnitine quantitation by UHPLC–MS/MS: Distinguishing true isovaleric acidemia from pivalate derived interference. J Chromatogr B 2017, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Murko, S.; Aseman, A.D.; Reinhardt, F.; Gramer, G.; Okun, J.G.; Mütze, U.; Santer, R. Neonatal screening for isovaleric aciduria: Reducing the increasingly high false-positive rate in Germany. JIMD Rep. 2022, 64, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Matern, D.; Tortorelli, S.; Oglesbee, D.; Gavrilov, D.; Rinaldo, P. Reduction of the false-positive rate in newborn screening by implementation of MS/MS-based second-tier tests: The Mayo Clinic experience (2004–2007). J Inherit Metab Dis. 2007, 30, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Oglesbee, D.; Sanders, K.A.; Lacey, J.M.; Magera, M.J.; Casetta, B.; Strauss, K.A.; Tortorelli, S.; Rinaldo, P.; Matern, D. Second-tier test for quantification of alloisoleucine and branched-chain amino acids in dried blood spots to improve newborn screening for maple syrup urine disease (MSUD). Clin Chem 2008, 54, 542–549. [Google Scholar] [CrossRef] [PubMed]
- Rossi, C.; Calton, L.; Hammond, G.; Brown, H.A.; Wallace, A.M.; Sacchetta, P.; Morris, M. Serum steroid profiling for congenital adrenal hyperplasia using liquid chromatography–tandem mass spectrometry. Clin Chim Acta. 2010, 411, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, G.B.; Ester, M.; Horvath, G.; Karnebeek, C.D.; van Stockler-Ipsirogu, S.; Vallance, H. Integrated Multianalyte Second-Tier Testing for Newborn Screening for MSUD, IVA, and GAMT Deficiencies. J. Inborn Errors Metab Screen. 2016, 4. [Google Scholar] [CrossRef]
- Kilgore, MB.; Platis, D.; Lim, T.; Isenberg, s.; Pickens, C.A.; Cuthbert, c.; Petritis, K. Development of a Universal Second-Tier Newborn Screening LC–MS/MS Method for Amino Acids, Lysophosphatidylcholines, and Organic Acids. Anal Chem 2023, 95, 3187–3194. [Google Scholar] [CrossRef] [PubMed]
- Mütze, U.; Henze, L.; Schröter, J.; Gleich, F.; Lindner, M.; Grunert, S.C.; et al. Isovaleric aciduria identified by newborn screening: Strategies to predict disease severity and stratify treatment. J Inherit Metab Dis 2023, 46, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



