Submitted:
22 January 2024
Posted:
22 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Flavivirus infection in humans
2.1. Dengue Virus (DENV)
2.2. Japanese Encephalitis Virus (JEV)
2.3. Kyasanur Forest disease virus (KFDV)
2.4. Powassan virus (POWV)
2.5. Tick-borne encephalitis virus (TBEV)
2.6. West Nile virus (WNV)
2.7. Yellow fever virus (YFV)
2.8. Zika virus (ZIKV)
3. Animal models of flavivirus infection
3.1. Immunocompetent Mice
3.1.1. Antibody-Mediated Receptor-Blocked Mice
3.1.2. Neonatal Mice
3.1.3. Pregnant Mice
3.1.4. Elderly Mice
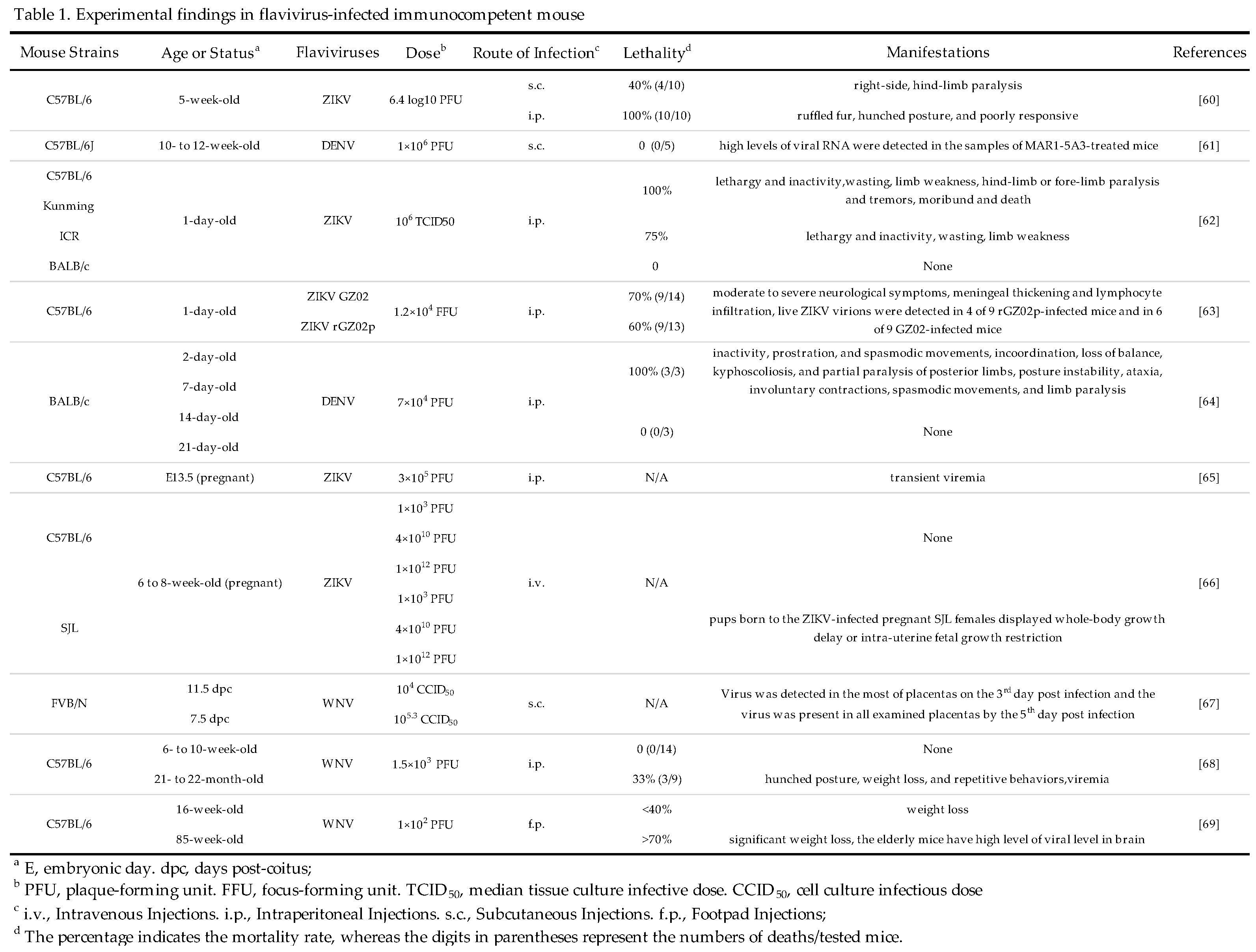
3.2. Immunodeficient Mice
3.3. Non-human primates (NHP)
3.3.1. Cynomolgus macaques
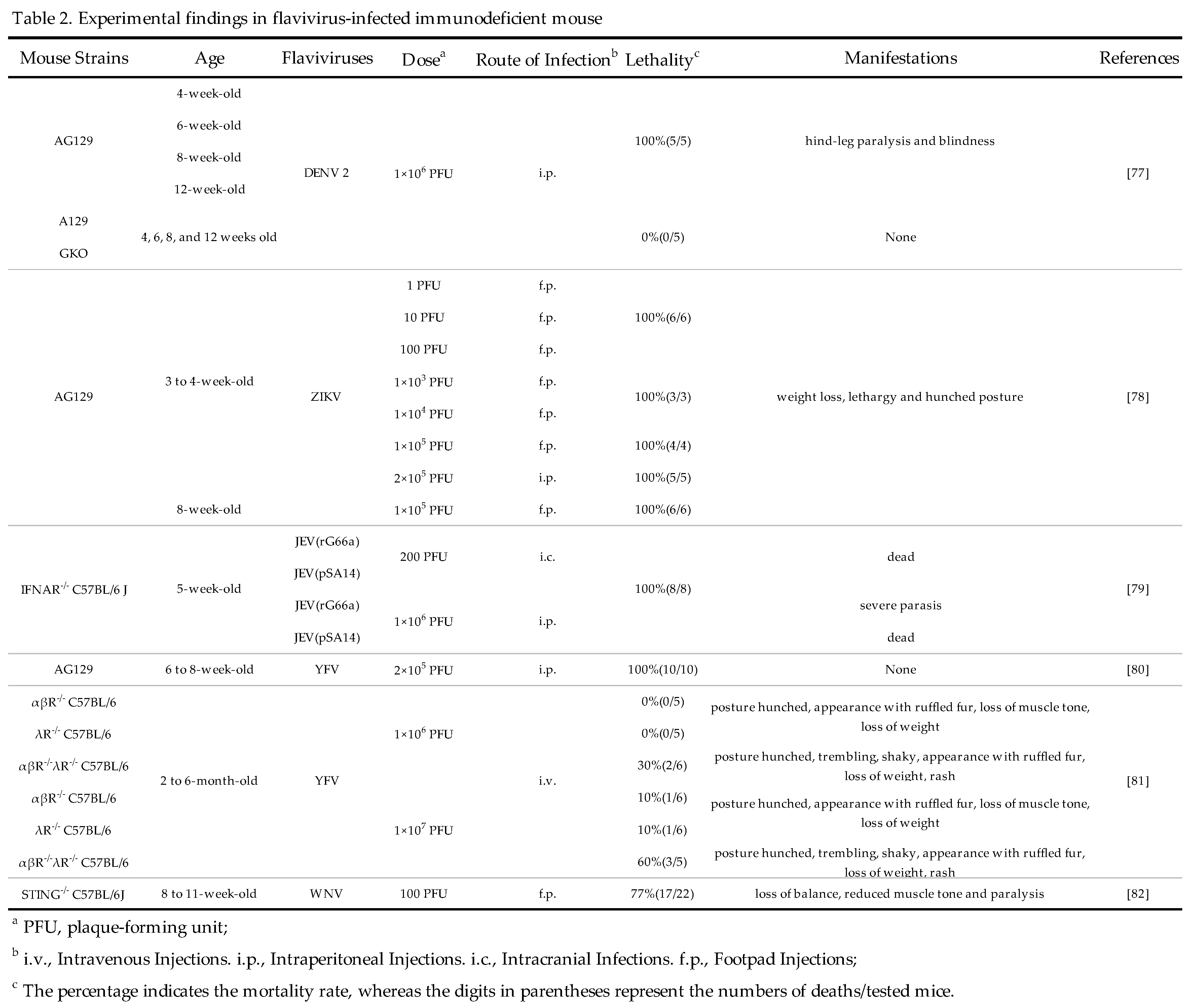
3.3.2. Rhesus macaques
3.3.3. Pigtailed macaques
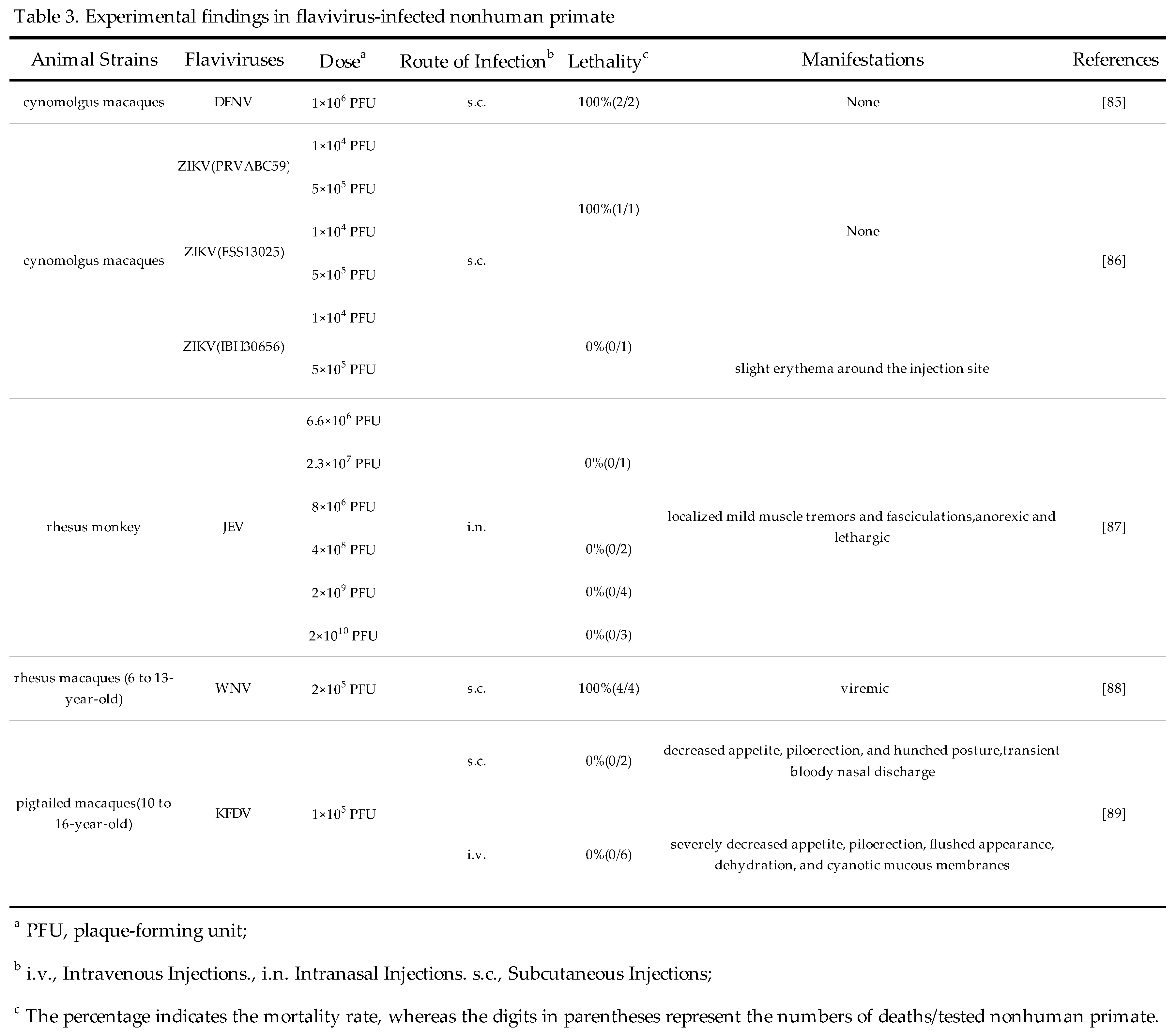
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kummerer BM: Establishment and Application of Flavivirus Replicons. Adv Exp Med Biol 2018, 1062:165-173. [CrossRef]
- van Leur SW, Heunis T, Munnur D, Sanyal S: Pathogenesis and virulence of flavivirus infections. Virulence 2021, 12(1):2814-2838. [CrossRef]
- Zaitseva E, Yang ST, Melikov K, Pourmal S, Chernomordik LV: Dengue virus ensures its fusion in late endosomes using compartment-specific lipids. PLoS Pathog 2010, 6(10):e1001131. [CrossRef]
- Neufeldt CJ, Cortese M, Acosta EG, Bartenschlager R: Rewiring cellular networks by members of the Flaviviridae family. Nat Rev Microbiol 2018, 16(3):125-142. [CrossRef]
- Cortese M, Goellner S, Acosta EG, Neufeldt CJ, Oleksiuk O, Lampe M, Haselmann U, Funaya C, Schieber N, Ronchi P et al: Ultrastructural Characterization of Zika Virus Replication Factories. Cell Rep 2017, 18(9):2113-2123. [CrossRef]
- Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, Drake JM, Brownstein JS, Hoen AG, Sankoh O et al: The global distribution and burden of dengue. Nature 2013, 496(7446):504-507. [CrossRef]
- Ngono AE, Shresta S: Immune Response to Dengue and Zika. Annu Rev Immunol 2018, 36:279-308. [CrossRef]
- Shepard DS, Undurraga EA, Halasa YA, Stanaway JD: The global economic burden of dengue: a systematic analysis. Lancet Infect Dis 2016, 16(8):935-941. [CrossRef]
- Diamond MS, Pierson TC: Molecular Insight into Dengue Virus Pathogenesis and Its Implications for Disease Control. Cell 2015, 162(3):488-492. [CrossRef]
- van den Hurk AF, Ritchie SA, Mackenzie JS: Ecology and geographical expansion of Japanese encephalitis virus. Annu Rev Entomol 2009, 54:17-35. [CrossRef]
- Pearce JC, Learoyd TP, Langendorf BJ, Logan JG: Japanese encephalitis: the vectors, ecology and potential for expansion. J Travel Med 2018, 25(suppl_1):S16-S26. [CrossRef]
- Chai C, Palinski R, Xu Y, Wang Q, Cao S, Geng Y, Zhao Q, Wen Y, Huang X, Yan Q et al: Aerosol and Contact Transmission Following Intranasal Infection of Mice with Japanese Encephalitis Virus. Viruses 2019, 11(1). [CrossRef]
- Garcia-Nicolas O, Braun RO, Milona P, Lewandowska M, Dijkman R, Alves MP, Summerfield A: Targeting of the Nasal Mucosa by Japanese Encephalitis Virus for Non-Vector-Borne Transmission. J Virol 2018, 92(24). [CrossRef]
- Ricklin ME, Garcia-Nicolas O, Brechbuhl D, Python S, Zumkehr B, Nougairede A, Charrel RN, Posthaus H, Oevermann A, Summerfield A: Vector-free transmission and persistence of Japanese encephalitis virus in pigs. Nat Commun 2016, 7:10832. [CrossRef]
- Chapagain S, Pal Singh P, Le K, Safronetz D, Wood H, Karniychuk U: Japanese encephalitis virus persists in the human reproductive epithelium and porcine reproductive tissues. PLoS Negl Trop Dis 2022, 16(7):e0010656. [CrossRef]
- Basumatary LJ, Raja D, Bhuyan D, Das M, Goswami M, Kayal AK: Clinical and radiological spectrum of Japanese encephalitis. J Neurol Sci 2013, 325(1-2):15-21. [CrossRef]
- Heffelfinger JD, Li X, Batmunkh N, Grabovac V, Diorditsa S, Liyanage JB, Pattamadilok S, Bahl S, Vannice KS, Hyde TB et al: Japanese Encephalitis Surveillance and Immunization - Asia and Western Pacific Regions, 2016. MMWR Morb Mortal Wkly Rep 2017, 66(22):579-583. [CrossRef]
- Xiang JY, Zhang YH, Tan ZR, Huang J, Zhao YW: Guillain-Barre syndrome associated with Japanese encephalitis virus infection in China. Viral Immunol 2014, 27(8):418-420. [CrossRef]
- Wang G, Li H, Yang X, Guo T, Wang L, Zhao Z, Sun H, Hou X, Ding X, Dou C et al: Guillain-Barre Syndrome Associated with JEV Infection. N Engl J Med 2020, 383(12):1188-1190. [CrossRef]
- Ooi MH, Lewthwaite P, Lai BF, Mohan A, Clear D, Lim L, Krishnan S, Preston T, Chieng CH, Tio PH et al: The epidemiology, clinical features, and long-term prognosis of Japanese encephalitis in central sarawak, malaysia, 1997-2005. Clin Infect Dis 2008, 47(4):458-468. [CrossRef]
- Shah SZ, Jabbar B, Ahmed N, Rehman A, Nasir H, Nadeem S, Jabbar I, Rahman ZU, Azam S: Epidemiology, Pathogenesis, and Control of a Tick-Borne Disease- Kyasanur Forest Disease: Current Status and Future Directions. Front Cell Infect Microbiol 2018, 8:149. [CrossRef]
- Holbrook MR: Kyasanur forest disease. Antiviral Res 2012, 96(3):353-362. [CrossRef]
- Work TH: Russian spring-summer virus in India: Kyasanur Forest disease. Prog Med Virol 1958, 1:248-279.
- Boshell J, Rajagopalan PK: Preliminary studies on experimental transmission of Kyasanur Forest disease virus by nymphs of Ixodes petauristae Warburton, 1933, infected as larvae on Suncus murinus and Rattus blanfordi. Indian J Med Res 1968, 56(4):589-593.
- Trapido H, Rajagopalan PK, Work TH, Varma MG: Kyasanur Forest disease. VIII. Isolation of Kyasanur Forest disease virus from naturally infected ticks of the genus Haemaphysalis. Indian J Med Res 1959, 47(2):133-138.
- Stone R: Monkey fever unbound. Science 2014, 345(6193):130-131, 133. [CrossRef]
- Munivenkatappa A, Sahay RR, Yadav PD, Viswanathan R, Mourya DT: Clinical & epidemiological significance of Kyasanur forest disease. Indian J Med Res 2018, 148(2):145-150. [CrossRef]
- Bhatia B, Meade-White K, Haddock E, Feldmann F, Marzi A, Feldmann H: A live-attenuated viral vector vaccine protects mice against lethal challenge with Kyasanur Forest disease virus. NPJ Vaccines 2021, 6(1):152. [CrossRef]
- Bhatia B, Feldmann H, Marzi A: Kyasanur Forest Disease and Alkhurma Hemorrhagic Fever Virus-Two Neglected Zoonotic Pathogens. Microorganisms 2020, 8(9). [CrossRef]
- Hermance ME, Thangamani S: Powassan Virus: An Emerging Arbovirus of Public Health Concern in North America. Vector Borne Zoonotic Dis 2017, 17(7):453-462. [CrossRef]
- Piantadosi A, Solomon IH: Powassan Virus Encephalitis. Infect Dis Clin North Am 2022, 36(3):671-688. [CrossRef]
- Hinten SR, Beckett GA, Gensheimer KF, Pritchard E, Courtney TM, Sears SD, Woytowicz JM, Preston DG, Smith RP, Jr., Rand PW et al: Increased recognition of Powassan encephalitis in the United States, 1999-2005. Vector Borne Zoonotic Dis 2008, 8(6):733-740. [CrossRef]
- Petri E, Gniel D, Zent O: Tick-borne encephalitis (TBE) trends in epidemiology and current and future management. Travel Med Infect Dis 2010, 8(4):233-245. [CrossRef]
- Lindquist L, Vapalahti O: Tick-borne encephalitis. Lancet 2008, 371(9627):1861-1871. [CrossRef]
- Beaute J, Spiteri G, Warns-Petit E, Zeller H: Tick-borne encephalitis in Europe, 2012 to 2016. Euro Surveill 2018, 23(45). [CrossRef]
- Kovalev SY, Mukhacheva TA: Tick-borne encephalitis virus subtypes emerged through rapid vector switches rather than gradual evolution. Ecol Evol 2014, 4(22):4307-4316. [CrossRef]
- Kaiser R: Tick-borne encephalitis. Infect Dis Clin North Am 2008, 22(3):561-575, x. [CrossRef]
- Suss J: Tick-borne encephalitis in Europe and beyond--the epidemiological situation as of 2007. Euro Surveill 2008, 13(26).
- Kaiser R: Tick-borne encephalitis: Clinical findings and prognosis in adults. Wien Med Wochenschr 2012, 162(11-12):239-243. [CrossRef]
- Colpitts TM, Conway MJ, Montgomery RR, Fikrig E: West Nile Virus: biology, transmission, and human infection. Clin Microbiol Rev 2012, 25(4):635-648. [CrossRef]
- Centers for Disease C, Prevention: Investigations of West Nile virus infections in recipients of blood transfusions. MMWR Morb Mortal Wkly Rep 2002, 51(43):973-974.
- Iwamoto M, Jernigan DB, Guasch A, Trepka MJ, Blackmore CG, Hellinger WC, Pham SM, Zaki S, Lanciotti RS, Lance-Parker SE et al: Transmission of West Nile virus from an organ donor to four transplant recipients. N Engl J Med 2003, 348(22):2196-2203. [CrossRef]
- Centers for Disease C, Prevention: Possible West Nile virus transmission to an infant through breast-feeding--Michigan, 2002. MMWR Morb Mortal Wkly Rep 2002, 51(39):877-878.
- Centers for Disease C, Prevention: Intrauterine West Nile virus infection--New York, 2002. MMWR Morb Mortal Wkly Rep 2002, 51(50):1135-1136.
- Martinez AA, Espinosa BA, Adamek RN, Thomas BA, Chau J, Gonzalez E, Keppetipola N, Salzameda NT: Breathing new life into West Nile virus therapeutics; discovery and study of zafirlukast as an NS2B-NS3 protease inhibitor. Eur J Med Chem 2018, 157:1202-1213. [CrossRef]
- Garske T, Van Kerkhove MD, Yactayo S, Ronveaux O, Lewis RF, Staples JE, Perea W, Ferguson NM, Yellow Fever Expert C: Yellow Fever in Africa: estimating the burden of disease and impact of mass vaccination from outbreak and serological data. PLoS Med 2014, 11(5):e1001638. [CrossRef]
- Paules CI, Fauci AS: Yellow Fever - Once Again on the Radar Screen in the Americas. N Engl J Med 2017, 376(15):1397-1399. [CrossRef]
- Cardoso Jda C, de Almeida MA, dos Santos E, da Fonseca DF, Sallum MA, Noll CA, Monteiro HA, Cruz AC, Carvalho VL, Pinto EV et al: Yellow fever virus in Haemagogus leucocelaenus and Aedes serratus mosquitoes, southern Brazil, 2008. Emerg Infect Dis 2010, 16(12):1918-1924. [CrossRef]
- Faria NR, Kraemer MUG, Hill SC, Goes de Jesus J, Aguiar RS, Iani FCM, Xavier J, Quick J, du Plessis L, Dellicour S et al: Genomic and epidemiological monitoring of yellow fever virus transmission potential. Science 2018, 361(6405):894-899. [CrossRef]
- Davis EH, Wang B, White M, Huang YS, Sarathy VV, Wang T, Bourne N, Higgs S, Barrett ADT: Impact of yellow fever virus envelope protein on wild-type and vaccine epitopes and tissue tropism. NPJ Vaccines 2022, 7(1):39. [CrossRef]
- Li C, Xu D, Ye Q, Hong S, Jiang Y, Liu X, Zhang N, Shi L, Qin CF, Xu Z: Zika Virus Disrupts Neural Progenitor Development and Leads to Microcephaly in Mice. Cell stem cell 2016, 19(1):120-126.
- Prasad VM, Miller AS, Klose T, Sirohi D, Buda G, Jiang W, Kuhn RJ, Rossmann MG: Structure of the immature Zika virus at 9 A resolution. Nat Struct Mol Biol 2017, 24(2):184-186. [CrossRef]
- Kostyuchenko VA, Lim EX, Zhang S, Fibriansah G, Ng TS, Ooi JS, Shi J, Lok SM: Structure of the thermally stable Zika virus. Nature 2016, 533(7603):425-428. [CrossRef]
- Hasan SS, Sevvana M, Kuhn RJ, Rossmann MG: Structural biology of Zika virus and other flaviviruses. Nat Struct Mol Biol 2018, 25(1):13-20. [CrossRef]
- Lanciotti RS, Lambert AJ, Holodniy M, Saavedra S, Signor Ldel C: Phylogeny of Zika Virus in Western Hemisphere, 2015. Emerg Infect Dis 2016, 22(5):933-935. [CrossRef]
- Tabata T, Petitt M, Puerta-Guardo H, Michlmayr D, Wang C, Fang-Hoover J, Harris E, Pereira L: Zika Virus Targets Different Primary Human Placental Cells, Suggesting Two Routes for Vertical Transmission. Cell Host Microbe 2016, 20(2):155-166. [CrossRef]
- Mlakar J, Korva M, Tul N, Popovic M, Poljsak-Prijatelj M, Mraz J, Kolenc M, Resman Rus K, Vesnaver Vipotnik T, Fabjan Vodusek V et al: Zika Virus Associated with Microcephaly. The New England journal of medicine 2016, 374(10):951-958.
- Martines RB, Bhatnagar J, Keating MK, Silva-Flannery L, Muehlenbachs A, Gary J, Goldsmith C, Hale G, Ritter J, Rollin D et al: Notes from the Field: Evidence of Zika Virus Infection in Brain and Placental Tissues from Two Congenitally Infected Newborns and Two Fetal Losses--Brazil, 2015. MMWR Morb Mortal Wkly Rep 2016, 65(6):159-160. [CrossRef]
- Lazear HM, Govero J, Smith AM, Platt DJ, Fernandez E, Miner JJ, Diamond MS: A Mouse Model of Zika Virus Pathogenesis. Cell Host Microbe 2016, 19(5):720-730. [CrossRef]
- Smith DR, Hollidge B, Daye S, Zeng X, Blancett C, Kuszpit K, Bocan T, Koehler JW, Coyne S, Minogue T et al: Neuropathogenesis of Zika Virus in a Highly Susceptible Immunocompetent Mouse Model after Antibody Blockade of Type I Interferon. PLoS Negl Trop Dis 2017, 11(1):e0005296. [CrossRef]
- Wilken L, Stelz S, Prajeeth CK, Rimmelzwaan GF: Transient Blockade of Type I Interferon Signalling Promotes Replication of Dengue Virus Strain D2Y98P in Adult Wild-Type Mice. Viruses 2023, 15(4). [CrossRef]
- Li S, Armstrong N, Zhao H, Hou W, Liu J, Chen C, Wan J, Wang W, Zhong C, Liu C et al: Zika Virus Fatally Infects Wild Type Neonatal Mice and Replicates in Central Nervous System. Viruses 2018, 10(1). [CrossRef]
- Ye X, Liu X, Shu T, Deng W, Liao M, Zheng Y, Zheng X, Zhang X, Li T, Fan W et al: A Live-Attenuated Zika Virus Vaccine with High Production Capacity Confers Effective Protection in Neonatal Mice. J Virol 2021, 95(14):e0038321. [CrossRef]
- Velandia-Romero ML, Acosta-Losada O, Castellanos JE: In vivo infection by a neuroinvasive neurovirulent dengue virus. J Neurovirol 2012, 18(5):374-387. [CrossRef]
- Wu KY, Zuo GL, Li XF, Ye Q, Deng YQ, Huang XY, Cao WC, Qin CF, Luo ZG: Vertical transmission of Zika virus targeting the radial glial cells affects cortex development of offspring mice. Cell Res 2016, 26(6):645-654. [CrossRef]
- Cugola FR, Fernandes IR, Russo FB, Freitas BC, Dias JL, Guimaraes KP, Benazzato C, Almeida N, Pignatari GC, Romero S et al: The Brazilian Zika virus strain causes birth defects in experimental models. Nature 2016, 534(7606):267-271. [CrossRef]
- Julander JG, Winger QA, Olsen AL, Day CW, Sidwell RW, Morrey JD: Treatment of West Nile virus-infected mice with reactive immunoglobulin reduces fetal titers and increases dam survival. Antiviral Res 2005, 65(2):79-85. [CrossRef]
- Xie G, Luo H, Pang L, Peng BH, Winkelmann E, McGruder B, Hesse J, Whiteman M, Campbell G, Milligan GN et al: Dysregulation of Toll-Like Receptor 7 Compromises Innate and Adaptive T Cell Responses and Host Resistance to an Attenuated West Nile Virus Infection in Old Mice. J Virol 2016, 90(3):1333-1344. [CrossRef]
- Funk KE, Arutyunov AD, Desai P, White JP, Soung AL, Rosen SF, Diamond MS, Klein RS: Decreased antiviral immune response within the central nervous system of aged mice is associated with increased lethality of West Nile virus encephalitis. Aging Cell 2021, 20(8):e13412. [CrossRef]
- Frank JC, Song BH, Lee YM: Mice as an Animal Model for Japanese Encephalitis Virus Research: Mouse Susceptibility, Infection Route, and Viral Pathogenesis. Pathogens 2023, 12(5). [CrossRef]
- Brinton MA, Perelygin AA: Genetic resistance to flaviviruses. Adv Virus Res 2003, 60:43-85. [CrossRef]
- Brien JD, Uhrlaub JL, Hirsch A, Wiley CA, Nikolich-Zugich J: Key role of T cell defects in age-related vulnerability to West Nile virus. J Exp Med 2009, 206(12):2735-2745. [CrossRef]
- Zellweger RM, Shresta S: Mouse models to study dengue virus immunology and pathogenesis. Front Immunol 2014, 5:151. [CrossRef]
- Beasley DW, McAuley AJ, Bente DA: Yellow fever virus: genetic and phenotypic diversity and implications for detection, prevention and therapy. Antiviral Res 2015, 115:48-70. [CrossRef]
- Morrison TE, Diamond MS: Animal Models of Zika Virus Infection, Pathogenesis, and Immunity. J Virol 2017, 91(8). [CrossRef]
- Na W, Yeom M, Choi IK, Yook H, Song D: Animal models for dengue vaccine development and testing. Clin Exp Vaccine Res 2017, 6(2):104-110. [CrossRef]
- Johnson AJ, Roehrig JT: New mouse model for dengue virus vaccine testing. J Virol 1999, 73(1):783-786. [CrossRef]
- Aliota MT, Caine EA, Walker EC, Larkin KE, Camacho E, Osorio JE: Characterization of Lethal Zika Virus Infection in AG129 Mice. PLoS Negl Trop Dis 2016, 10(4):e0004682. [CrossRef]
- Zhou D, Jia F, Li Q, Zhang L, Chen Z, Zhao Z, Cui M, Song Y, Chen H, Cao S et al: Japanese Encephalitis Virus NS1' Protein Antagonizes Interferon Beta Production. Virol Sin 2018, 33(6):515-523. [CrossRef]
- Calvert AE, Dixon KL, Piper J, Bennett SL, Thibodeaux BA, Barrett AD, Roehrig JT, Blair CD: A humanized monoclonal antibody neutralizes yellow fever virus strain 17D-204 in vitro but does not protect a mouse model from disease. Antiviral Res 2016, 131:92-99. [CrossRef]
- Douam F, Soto Albrecht YE, Hrebikova G, Sadimin E, Davidson C, Kotenko SV, Ploss A: Type III Interferon-Mediated Signaling Is Critical for Controlling Live Attenuated Yellow Fever Virus Infection In Vivo. mBio 2017, 8(4). [CrossRef]
- McGuckin Wuertz K, Treuting PM, Hemann EA, Esser-Nobis K, Snyder AG, Graham JB, Daniels BP, Wilkins C, Snyder JM, Voss KM et al: STING is required for host defense against neuropathological West Nile virus infection. PLoS Pathog 2019, 15(8):e1007899. [CrossRef]
- Koraka P, Benton S, van Amerongen G, Stittelaar KJ, Osterhaus AD: Characterization of humoral and cellular immune responses in cynomolgus macaques upon primary and subsequent heterologous infections with dengue viruses. Microbes Infect 2007, 9(8):940-946. [CrossRef]
- Yoshida T, Omatsu T, Saito A, Katakai Y, Iwasaki Y, Kurosawa T, Hamano M, Higashino A, Nakamura S, Takasaki T et al: Dynamics of cellular immune responses in the acute phase of dengue virus infection. Arch Virol 2013, 158(6):1209-1220. [CrossRef]
- Ong EZ, Budigi Y, Tan HC, Robinson LN, Rowley KJ, Winnett A, Hobbie S, Shriver Z, Babcock GJ, Ooi EE: Preclinical evaluation of VIS513, a therapeutic antibody against dengue virus, in non-human primates. Antiviral Res 2017, 144:44-47. [CrossRef]
- Koide F, Goebel S, Snyder B, Walters KB, Gast A, Hagelin K, Kalkeri R, Rayner J: Development of a Zika Virus Infection Model in Cynomolgus Macaques. Front Microbiol 2016, 7:2028. [CrossRef]
- Myint KS, Raengsakulrach B, Young GD, Gettayacamin M, Ferguson LM, Innis BL, Hoke CH, Jr., Vaughn DW: Production of lethal infection that resembles fatal human disease by intranasal inoculation of macaques with Japanese encephalitis virus. Am J Trop Med Hyg 1999, 60(3):338-342. [CrossRef]
- Poore EA, Slifka DK, Raue HP, Thomas A, Hammarlund E, Quintel BK, Torrey LL, Slifka AM, Richner JM, Dubois ME et al: Pre-clinical development of a hydrogen peroxide-inactivated West Nile virus vaccine. Vaccine 2017, 35(2):283-292. [CrossRef]
- Broeckel RM, Feldmann F, McNally KL, Chiramel AI, Sturdevant GL, Leung JM, Hanley PW, Lovaglio J, Rosenke R, Scott DP et al: A pigtailed macaque model of Kyasanur Forest disease virus and Alkhurma hemorrhagic disease virus pathogenesis. PLoS Pathog 2021, 17(12):e1009678. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




