Submitted:
17 January 2024
Posted:
19 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Extraction of gelatin
Polymer (G-CH-M) construction
Antibacterial activity of G-CH-M biopolymer
Liquid medium microdilution assay
Crystal violet biofilm assay
Motility assay
Antioxidant activities
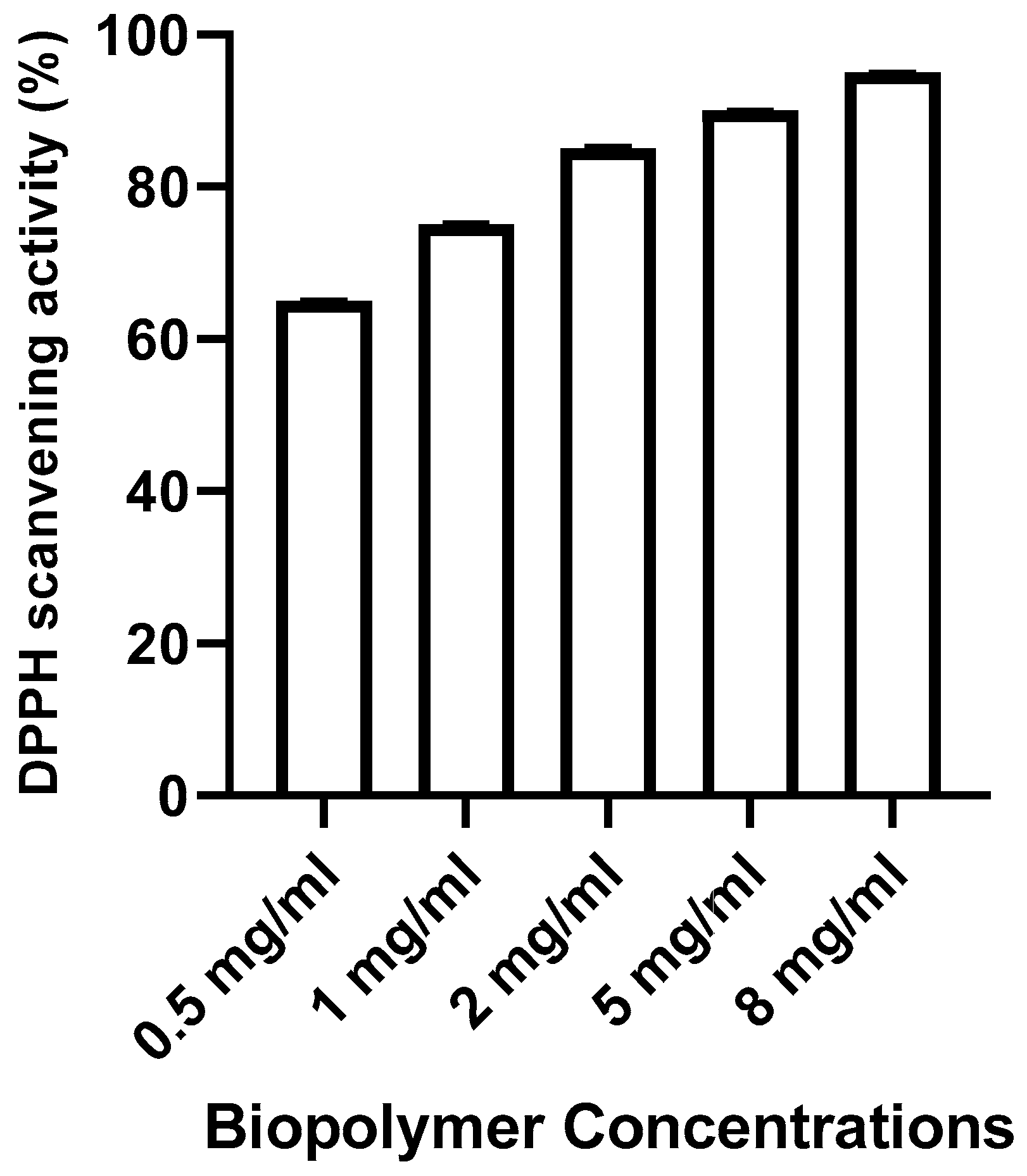
DPPH radical-scavenging activity
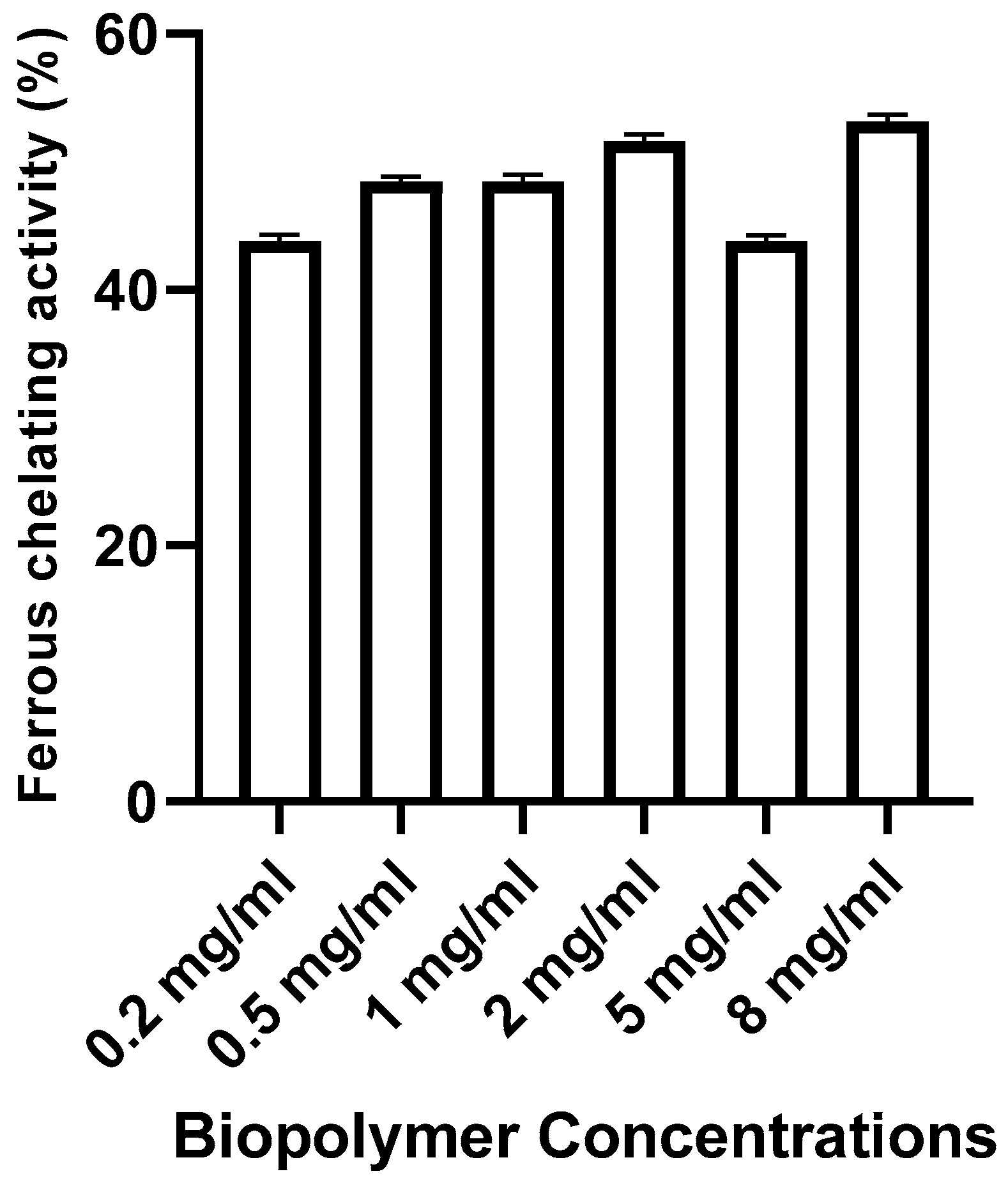
Ferrous chelating activity
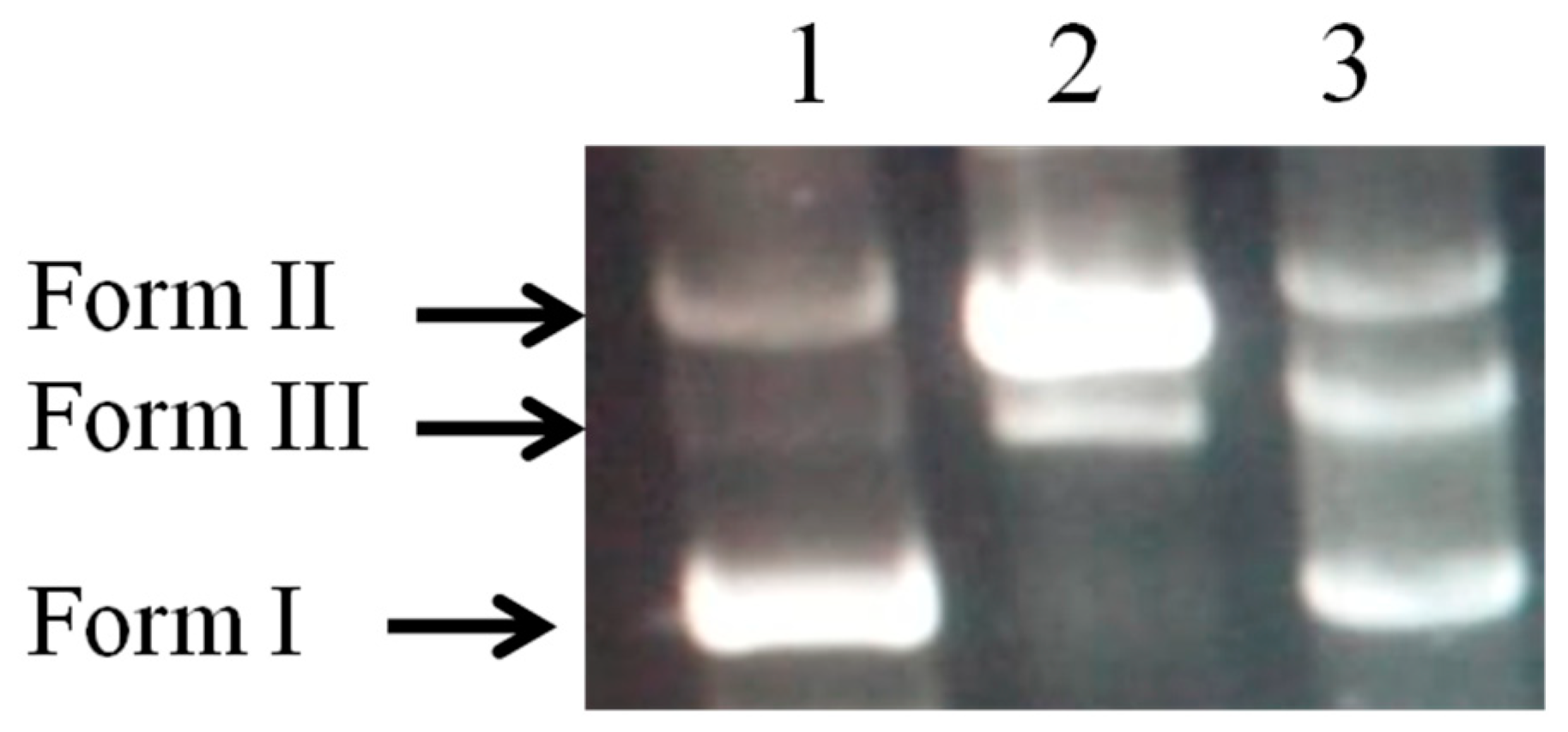
DNA Damage Protective Effect assay
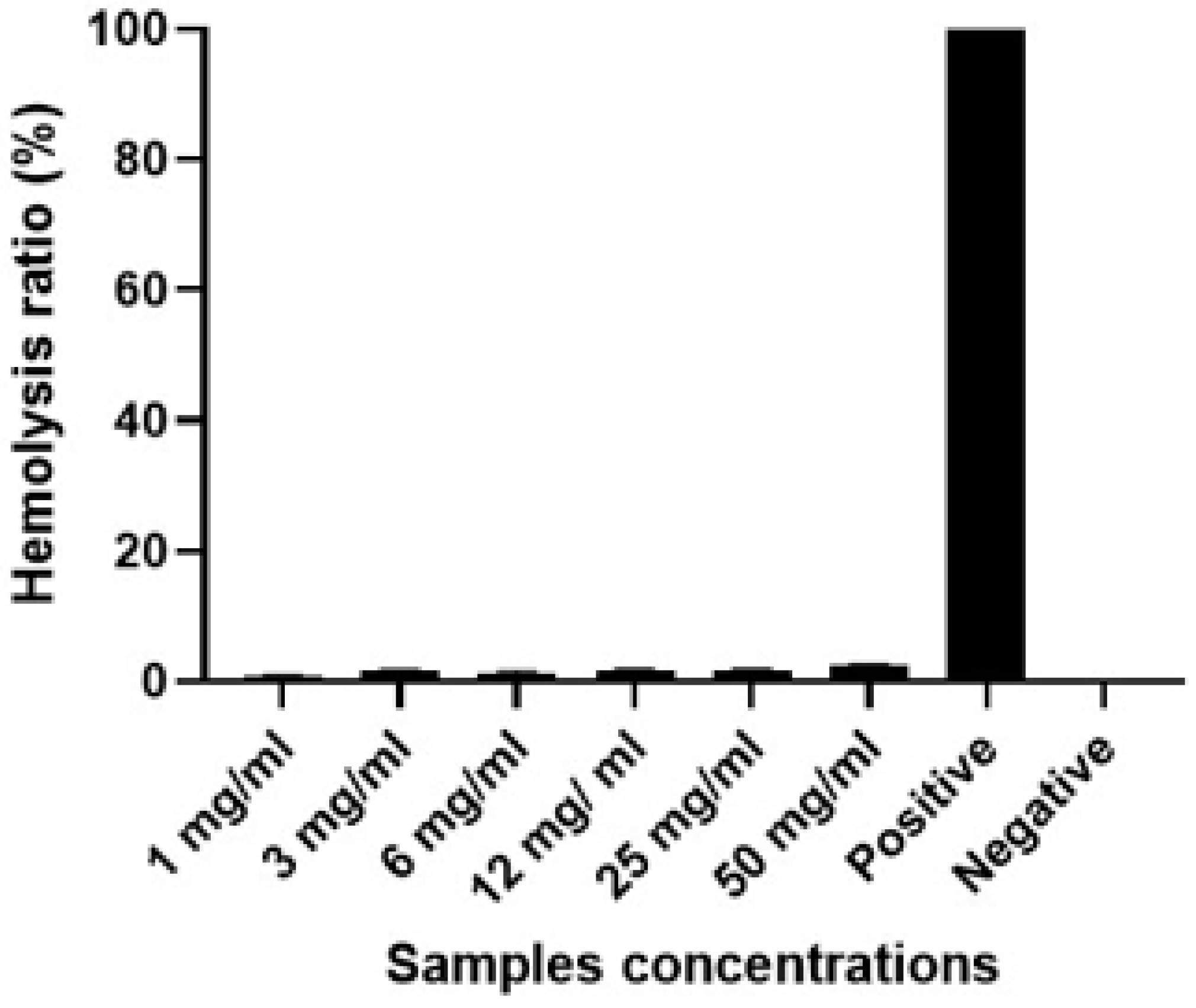
Biocompatibility
Anti-inflammatory activity
Results
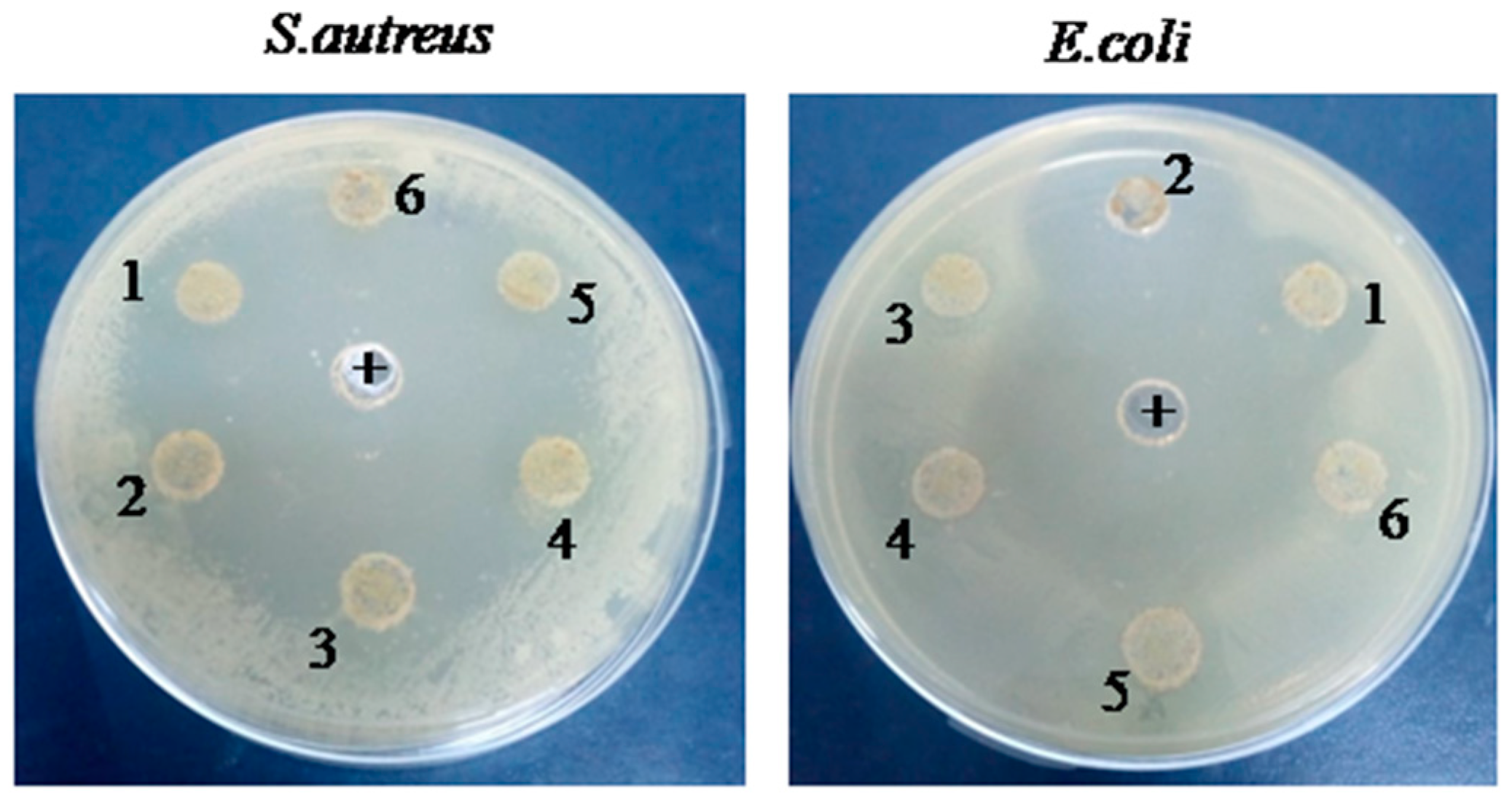
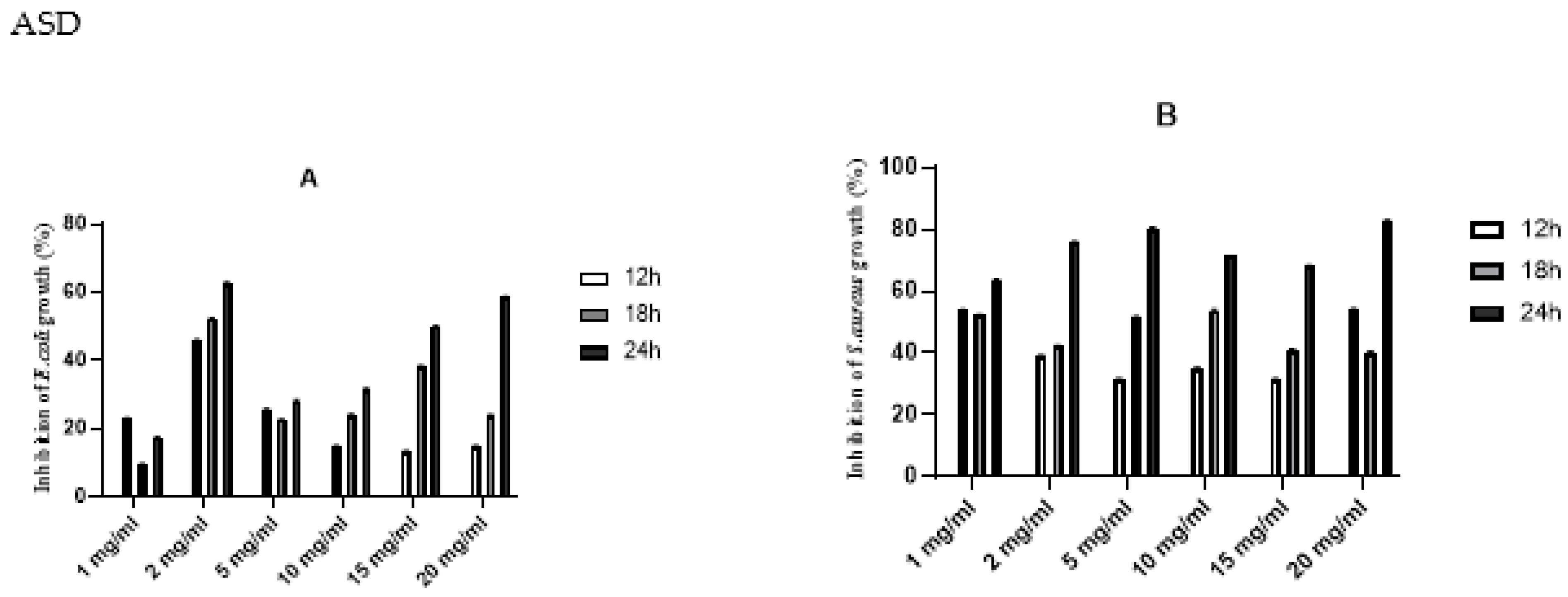
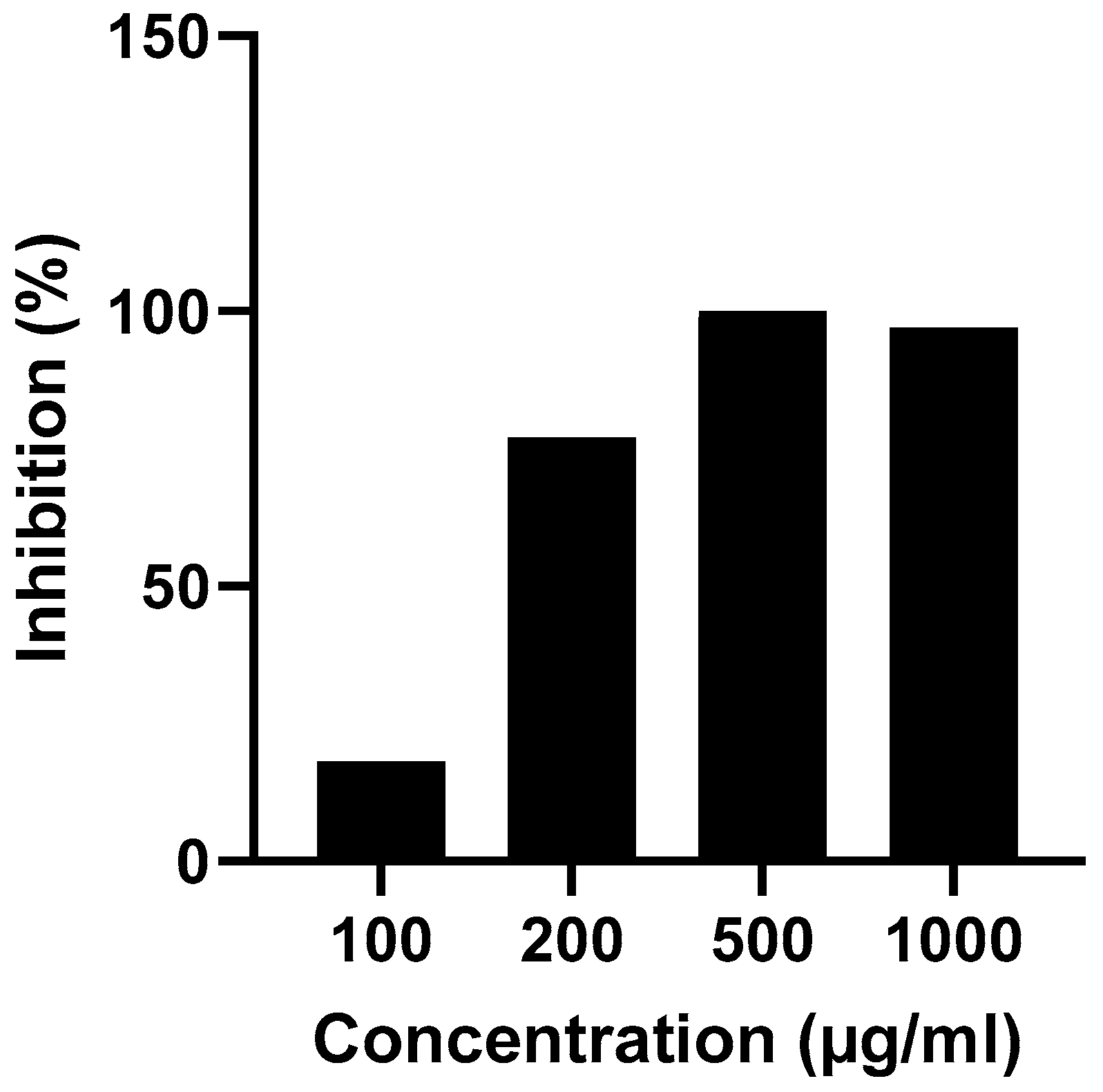
Antibacterial activities
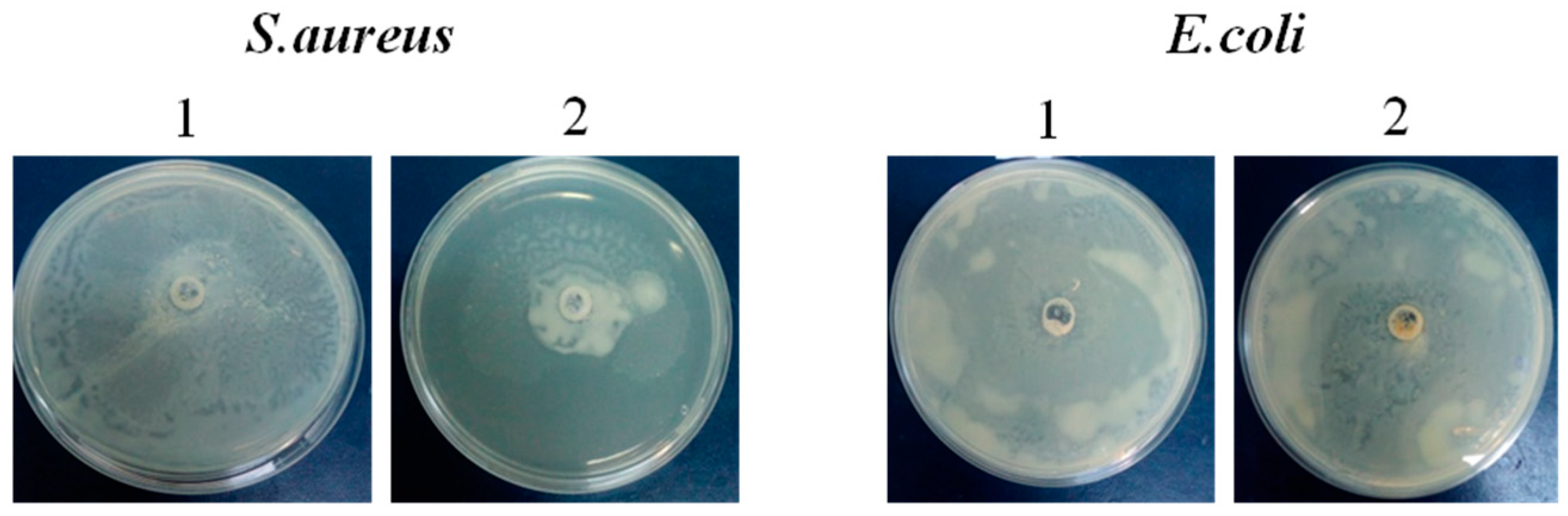
Modulation of bacteria motility
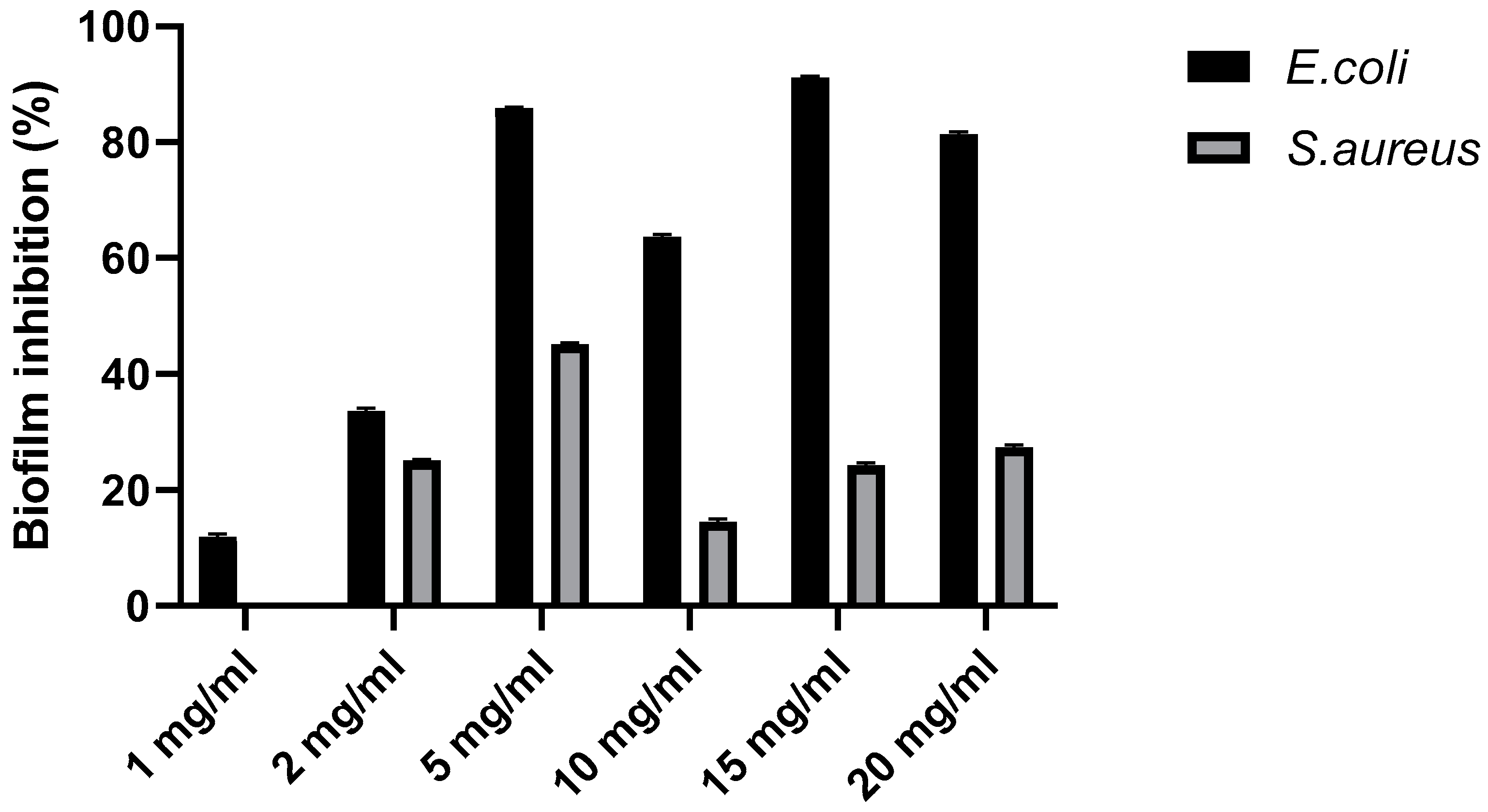
Anti-biofilm activity
Antioxidant activities
DPPH assay
Ferrous chelating activity
DNA Damage Protective Effect assay
Biocompatibility test
Anti-inflammatory activity
Discussion
Conclusions
Author Contributions
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflict of interest
References
- Arfat, Y.A., Benjakul, S., Prodpran, T., Sumpavapol, P and Songtipya, P., “Properties and antimicrobial activity of fish protein isolate/fish skin gelatin film containing basil leaf essential oil and zinc oxide nanoparticles.” Food Hydrocoll., 2014, 41, 265-273. [CrossRef]
- Kuijpers, A.J., Van Wachem, P.B., van Luyn, M.J., Plantinga, J.A., Engbers, G.H., Krijgsveld, J., Zaat, S.A., Dankert, J., Feijen, J., “In vivo compatibility and degradation of cross linked gelatin gels incorporated in knitted Dacron.” J. Biomed. Mater. Res., 2000,51, 136-145.
- Gómez-Guillén, M.C., Giménez, B., López-Caballero, M.A., Montero, M.P., “Functional and bioactive properties of collagen and gelatin from alternative sources: A review.” Food Hydrocoll., 2011, 25, 1813–1827. [CrossRef]
- Al-Hassan, A. A., “Gelatin from camel skins: Extraction and characterizations.” Food Hydrocolloids, 2020, 101, 105457. [CrossRef]
- Suganya, S., Senthil Ram, T., Lakshmi, B., Giridev, V., “Herbal drug incorporated antibacterial nanofibrous mat fabricated by electrospinning: an excellent matrix for wound dressings.” J. App. Polymer Science., 2011, 121, 2893-2899. [CrossRef]
- Shan, Y.-H., Peng, L.-H., Liu, X., Chen, X., Xiong, J., Gao, J.-Q., “Silk fibroin/gelatin electrospun nanofibrous dressing functionalized with astragaloside IV induces healing and anti-scar effects on burn wound.” Inter J pharma., 2015, 479, 291-301. [CrossRef]
- Mbikay, M. “Therapeutic potential of Moringa oleifera leaves in chronic hyperglycemia and dyslipidemia: a review.” Frontiers in Pharmacology, 2012, 3, 24. [CrossRef]
- Gothai, S., Arulselvan, P., Tan, W.S., Fakurazi, S., “Wound healing properties of ethyl acetate fraction of Moringa oleifera in normal human dermal fbroblasts.” J Intercult Ethnopharmacol., 2016, 51–6. [CrossRef]
- Muhammad, AA., Pauzi, N.A., Arulselvan, P., Abas, F., Fakurazi, S., “In vitro wound healing potential and identifcation of bioactive compounds from Moringa oleifera Lam.” BioMed Res Int., 2013, ID974580, 1–10. [CrossRef]
- Saini, R. K., Sivanesan, I., and Keum, Y. S., “Phytochemicals of Moringa oleifera: a review of their nutritional, therapeutic and industrial significance.” 3 Biotech., 2016, 6, 203. [CrossRef]
- [11]. Ma, Z. F., Ahmad, J., Zhang, H., Khan, I., & Muhammad, S., “Evaluation of phytochemical and medicinal properties of Moringa (Moringa oleifera) as a potential functional food. South African Journal of Botany, 2020, 129, 40-46. [CrossRef]
- Rubio-Elizalde, I., Bernáldez-Sarabia, J., Moreno-Ulloa, A., Vilanova, C., Juárez, P., Licea- Navarro, A., Castro-Ceseña, A.B., “Scaffolds based on alginate-PEG methyl ether methacrylate-Moringa oleifera-Aloe vera for wound healing applications.” Carbohydr. Polym., 2019, 206, 455–467. [CrossRef]
- Chin, C.Y., Ng, P.Y., Ng, S.F., “Moringa oleifera standardised aqueous leaf extractloaded hydrocolloid film dressing: in vivo dermal safety and wound healing evaluation in STZ/HFD diabetic rat model.” Drug Deliv. Transl. Res., 2019, 9, 453–468. [CrossRef]
- Singh, B., Kumar, A., “Graft and crosslinked polymerization of polysaccharide gum to form hydrogel wound dressings for drug delivery applications.” Carbohydr. Res., 2020,489, 107949. [CrossRef]
- Bessalah, S., Jebahi, S., Faraz, A., Raoufi, A., Tırınk, C., Dridi, W., andMohamed Hammadi, A. W. “Effect of gamma radiation on novel gelatin extracted from camel skin for pharmaceutical application.” Pakistan J of Zoology, 2023,55(2), 819. [CrossRef]
- Berghe, V. A., and Vlietinck, A. J. “Screening methods for antibacterial and antiviral agents from higher plants.” Methods in Plant Biochemistry, 1991,6(3),47-68.
- Wang, S., Zheng, F., Huang, Y., Fang, Y., Shen, M., Zhu, M., Shi, X., “Encapsulation of amoxicillin within laponite-doped poly (lactic-co-glycolic acid) nanofibers: preparation, characterization, and antibacterial activity.” ACS Applied Materials & Interfaces., 2012, 6393-6401. [CrossRef]
- O’Toole, G.A., Kolter, R., “Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development.” Mol. Microbiol., 1998, 30, 295–304. [CrossRef]
- O’May, C., Tufenkji, N., “The Swarming omtility of Pseudomonas aeruginosa is blocked by cranberry proanthocyanidins and other tannin-containing materials.” Appl. Environ. Microbiol. 2011, 77, 3061−3067. [CrossRef]
- Bersuder, P., Hole, M., & Smith, G. “Antioxidants from a heated histidine-glucose model system. I: Investigation of the antioxidant role of histidine and isolation of antioxidants by high-performance liquid chromatography.” Journal of the American Oil Chemists' Society, 1998, 75, 181-187. [CrossRef]
- Hus, B., Coupar, I.M., Ng,K., “Antioxidant activity of hot water extract from the fruit of the Doum palm Hyphaene thebaica.” Food Chem., 2006, 98(2),317–328. [CrossRef]
- Lee, J.C., Kim, H.R., Kim, J., Jang, Y.S., 2002. “Antioxidant property of an ethanol extract of the stem of Opuntia ficus-indica var. Saboten.” J. Agric. Food Chem., 50, 6490–6496. [CrossRef]
- International, Standard Practice for Assessment of Hemolytic Properites of Materials, 2000 (n.d.).
- Williams, LAD., Connar, AO, Latore, L, Dennis O, Ringer S, Whittaker, JA, Conrad J, Vogler B, Rosner H, Kraus, W. “The in vitro anti-denaturation effects induced by natural products and non-steroidal compounds in heat treated (immunogenic) Bovine Serum Albumin (BSA) is proposed as a screening assay for the detection of anti-inflammatory compounds, without the use of animals in the early stages of the drug discovery process.” West Indian Med. J., 2008,58, 327-331.
- Thaya, R., Vaseeharan, B., Sivakamavalli, J., Iswarya, A., Govindarajan, M., Alharbi, N. S., Kadaikunnan, S., Al-Anbr, M. N., Khaled, J. M., & Benelli, G. “Synthesis of chitosan-alginate microspheres with high antimicrobial and antibiofilm activity against multi-drug resistant microbial pathogens.” Microbial Pathogenesis, 2018, 114, 17-24. [CrossRef]
- Divya, M., Vaseeharan, B., Abinaya, M., Vijayakumar, S., Govindarajan, M., Alharbi, N. S., Kadaikunnan, S., Khaled, J. M., & Benelli, G., “Biopolymer gelatin-coated zinc oxide nanoparticles showed high antibacterial, antibiofilm and anti-angiogenic activity.” Journal of Photochemistry and Photobiology B: Biology.,2018, 178, 211-218. [CrossRef]
- Matica, M. A., Aachmann, F. L., Tøndervik, A., Sletta, H., & Ostafe, V., “Chitosan as a wound dressing starting material: Antimicrobial properties and mode of action.” International Journal of Molecular Sciences., 2019, 20, 5889. [CrossRef]
- Bukar A, Uba A, Oyeyi T “Antimicrobial profile of Moringa oleifera Lam. extracts against some food-borne microorganisms.” Bayero J Pure Appl Sci., 2010, 3,43–48. [CrossRef]
- Ajose DJ, Oluwarinde BO, Abolarinwa TO, Fri J, Montso KP, Fayemi OE, Aremu AO, Ateba CN. Combating bovine mastitis in the dairy sector in an era of antimicrobial resistance: ethno-veterinary medicinal option as a viable alternative approach. Frontiers in veterinary science. 2022 Apr 4;9:800322. [CrossRef]
- Butler, M. T., Q. F. Wang, and R. M. Harshey., “Cell density and mobility protect swarming bacteria against antibiotics.” Proc. Natl. Acad. Sci. U. S. A., 2010, 107,3776–3781. [CrossRef]
- O’Toole, G., H. B. Kaplan, and R. Kolter. Biofilm formation as microbial development. Annu. Rev.” Microbiol., 2000, 54,49–79. [CrossRef]
- Vaseeharan, B., Sivakamavalli, J., & Thaya, R., “Synthesis and characterization of chitosan-ZnO composite and its antibiofilm activity against aquatic bacteria.” Journal of Composite Materials., 2015,49, 177-184. [CrossRef]
- Siddhuraju, P., Becker, K., “Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves.” J Agric Food Chem., 2003, 51,2144–2155. [CrossRef]
- Shibata, H., Sakamoto, Y., OKA, M., & KONO, Y., “Natural antioxidant, chlorogenic acid, protects against DNA breakage caused by monochloramine.” Bioscience, Biotechnology, and Biochemistry., 1999, 63, 1295-1297. [CrossRef]
- Alhakmani, F., Kumar, S., & Khan, S. A., “Estimation of total phenolic content, in–vitro antioxidant and anti–inflammatory activity of flowers of Moringa oleifera.” Asian Pacific Journal of Tropical Biomedicine., 2013, 3, 623-627. [CrossRef]
- Fard, M. T., Arulselvan, P., Karthivashan, G., Adam, S. K., Fakurazi, S., “Bioactive extract from Moringa oleifera inhibits the pro-inflammatory mediators in lipopolysaccharide stimulated macrophages.” Pharmacognosy Magazine., 2015,11, S556. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



