Submitted:
16 January 2024
Posted:
17 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
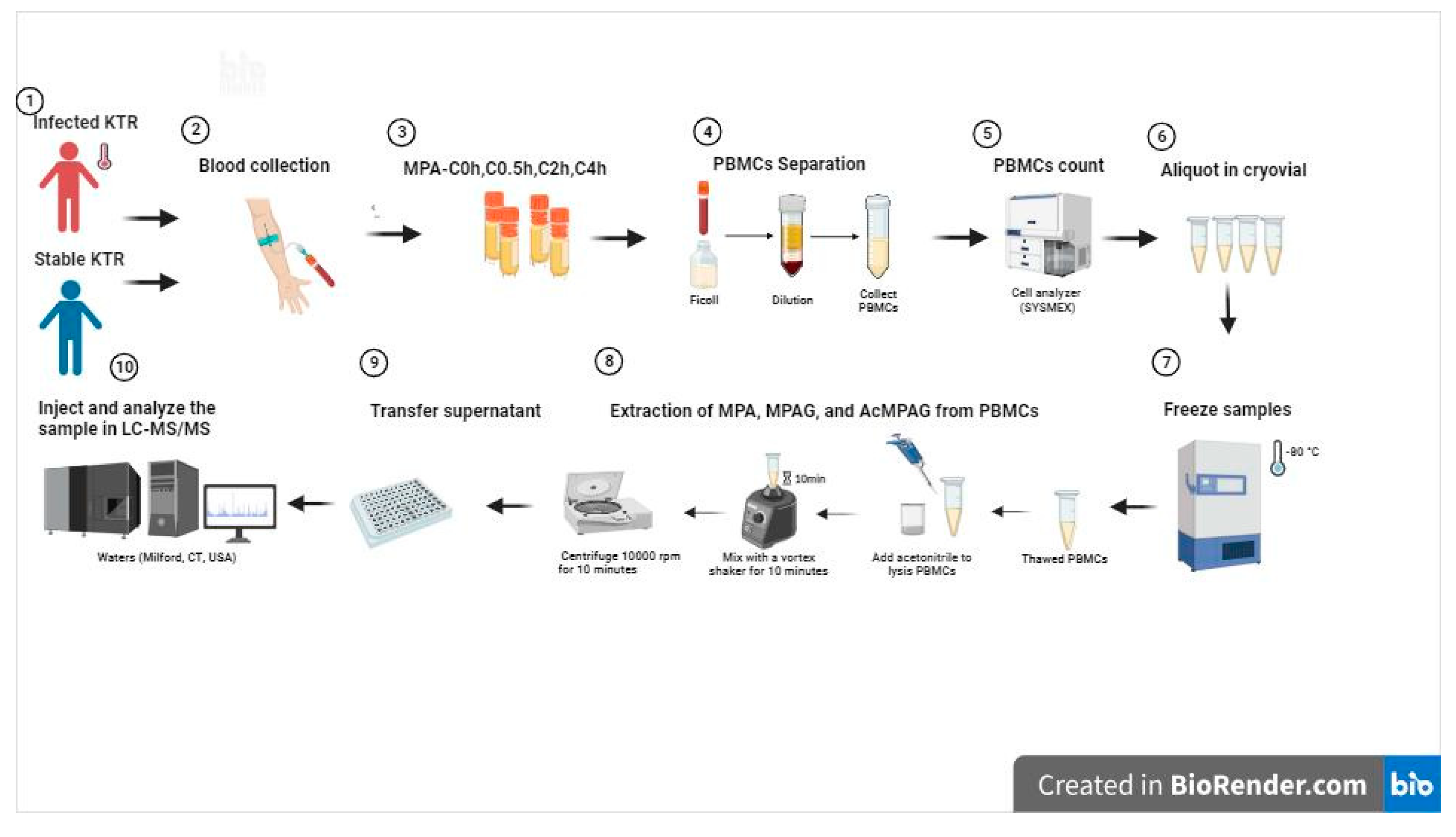
2.1. Sample collection
2.2. Reagents and Materials
2.3. Equipments and Conditions
2.4. Calibration Standards and Quality Control Samples
2.5. Sample pre-processing methods
2.6. Bioanalytical Validation
2.6.1. Standard Curve and Lower Limit of Quantitation
2.6.2. Selectivity and Carryover Effect
2.6.3. Matrix Effect and Extraction Recovery
2.6.4. Accuracy and precision
2.6.5. Stability
2.7. Clinical Validation
2.7.1. Patients
2.7.2. Sample and data collection
2.8. Statistical Analyses
3. Results
3.1. Bioanalytical Validation
3.1.1. Standard Curve and Lower Limit of Quantitation
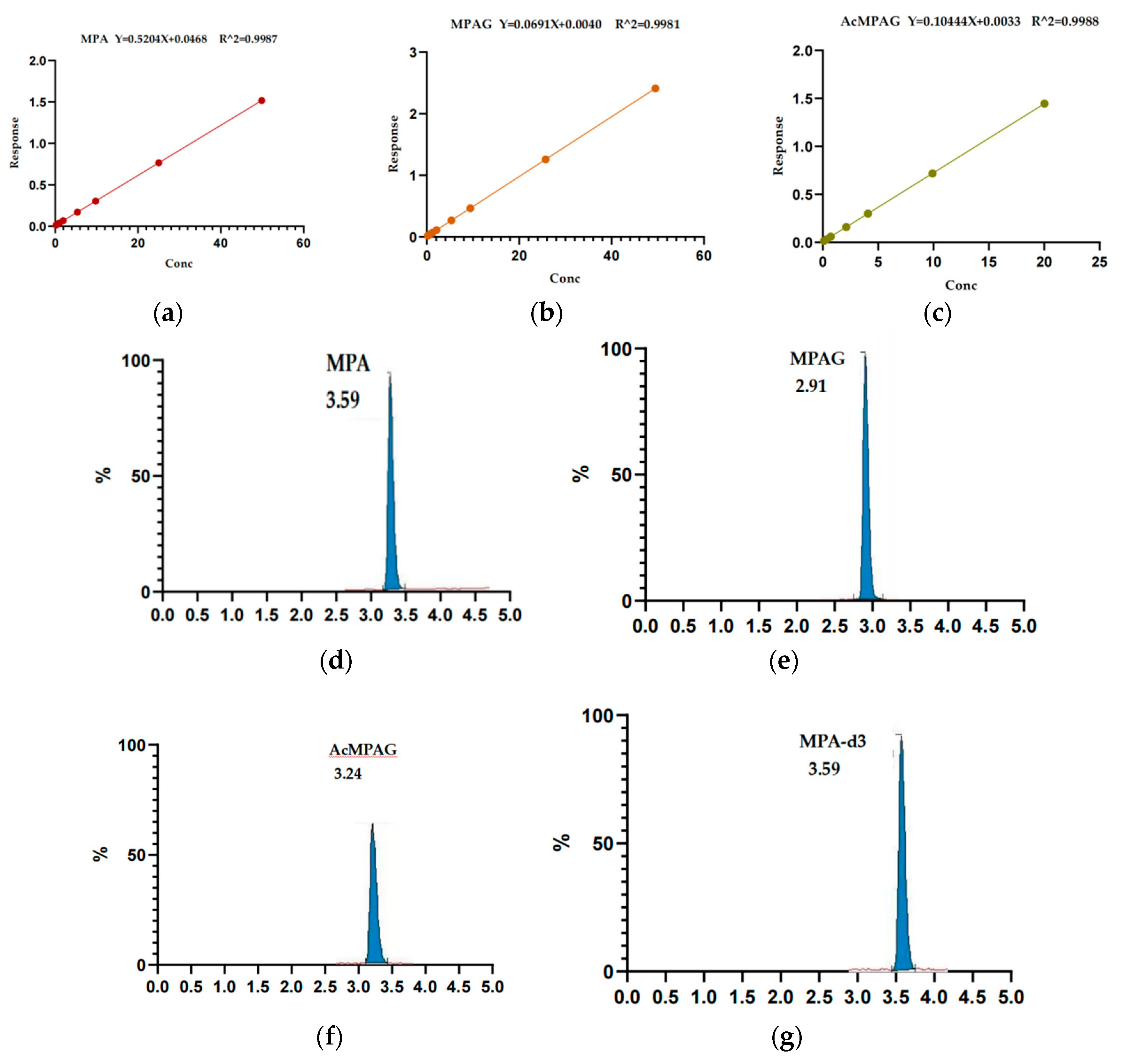
3.1.2. Selective and Carryover Effect
3.1.3. Matrix effect and recoveries
3.1.4. Accuracy and Precision
3.1.5. Stability
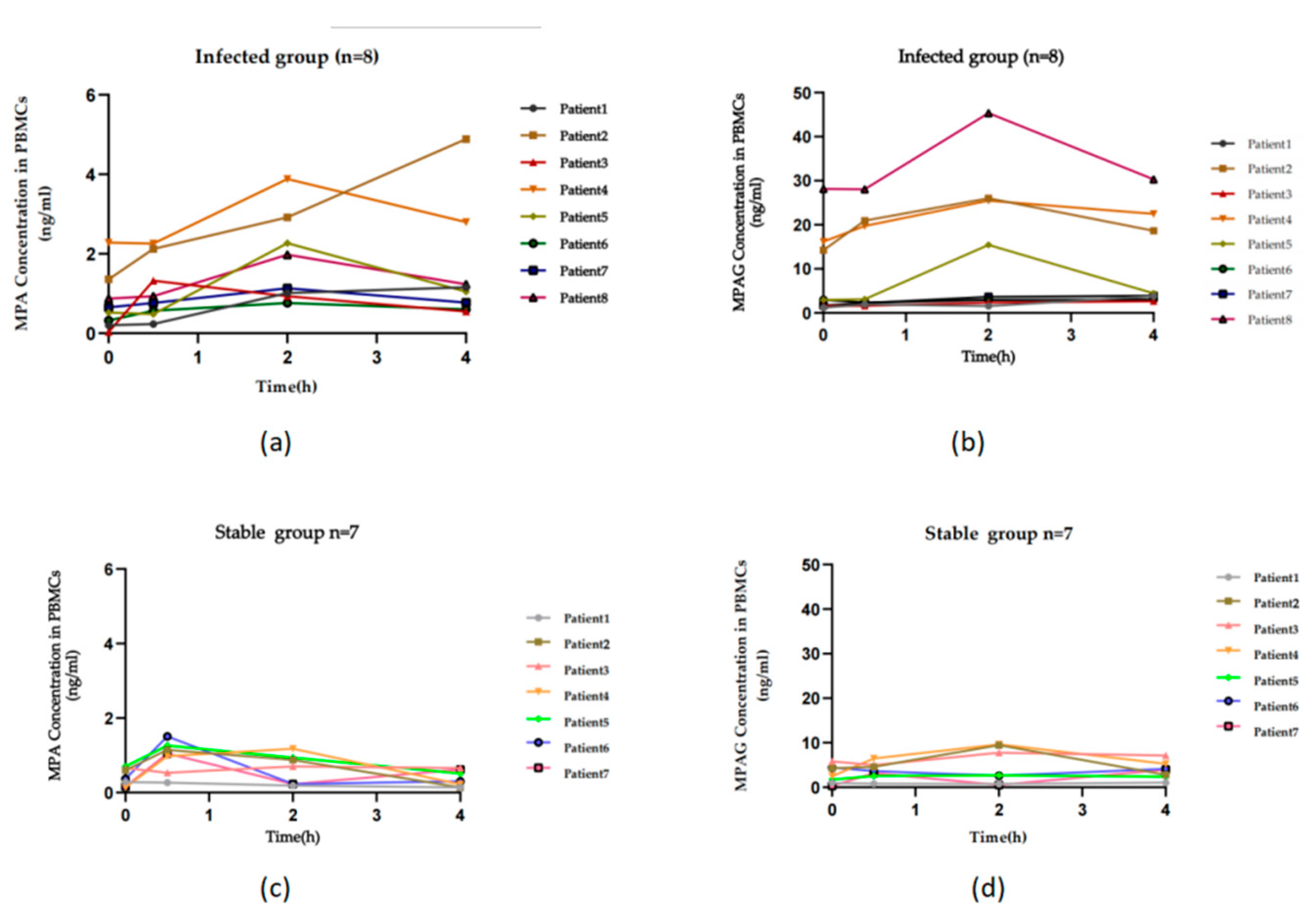
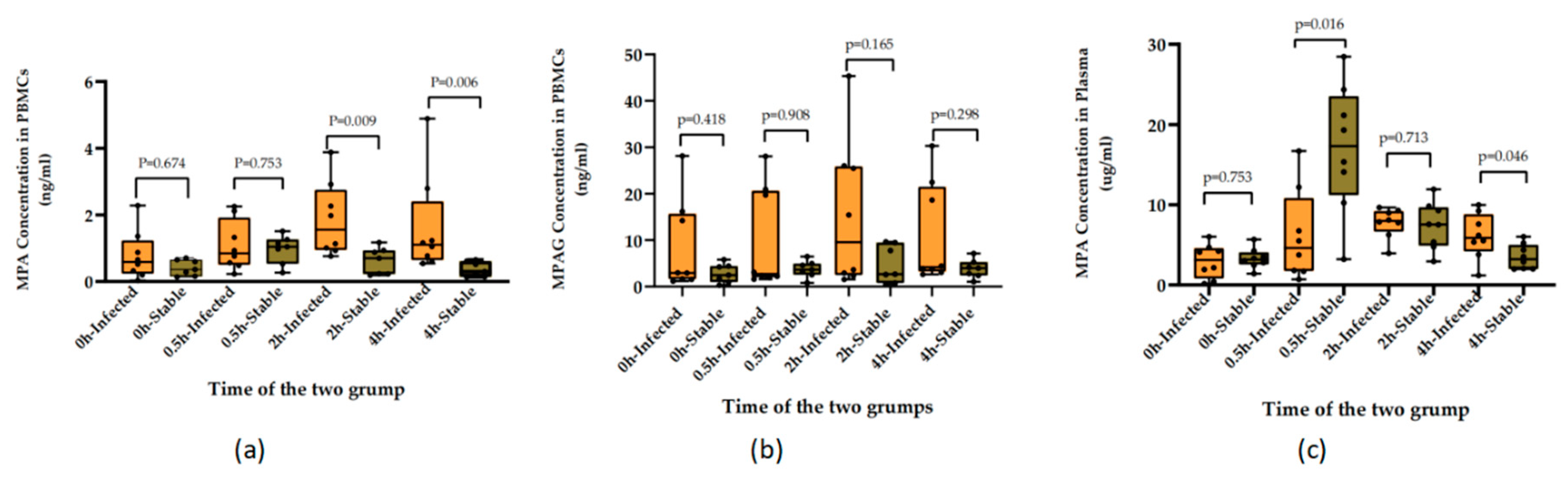
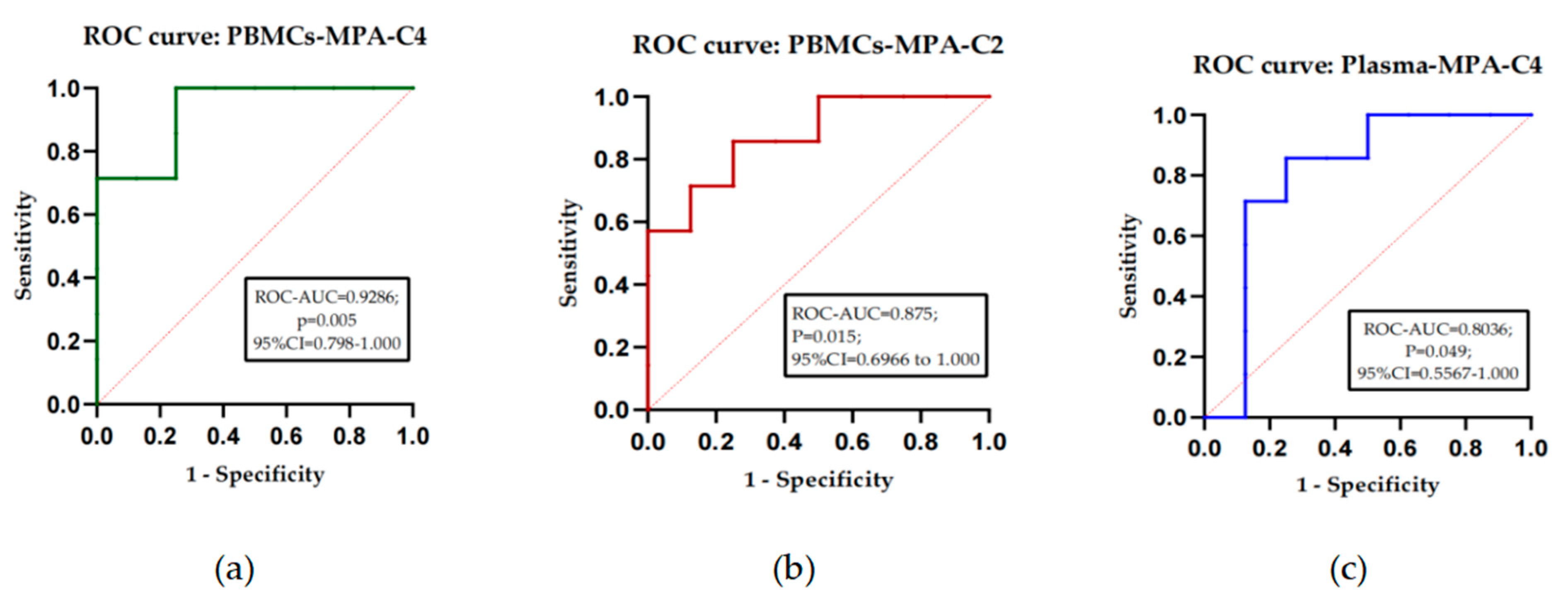
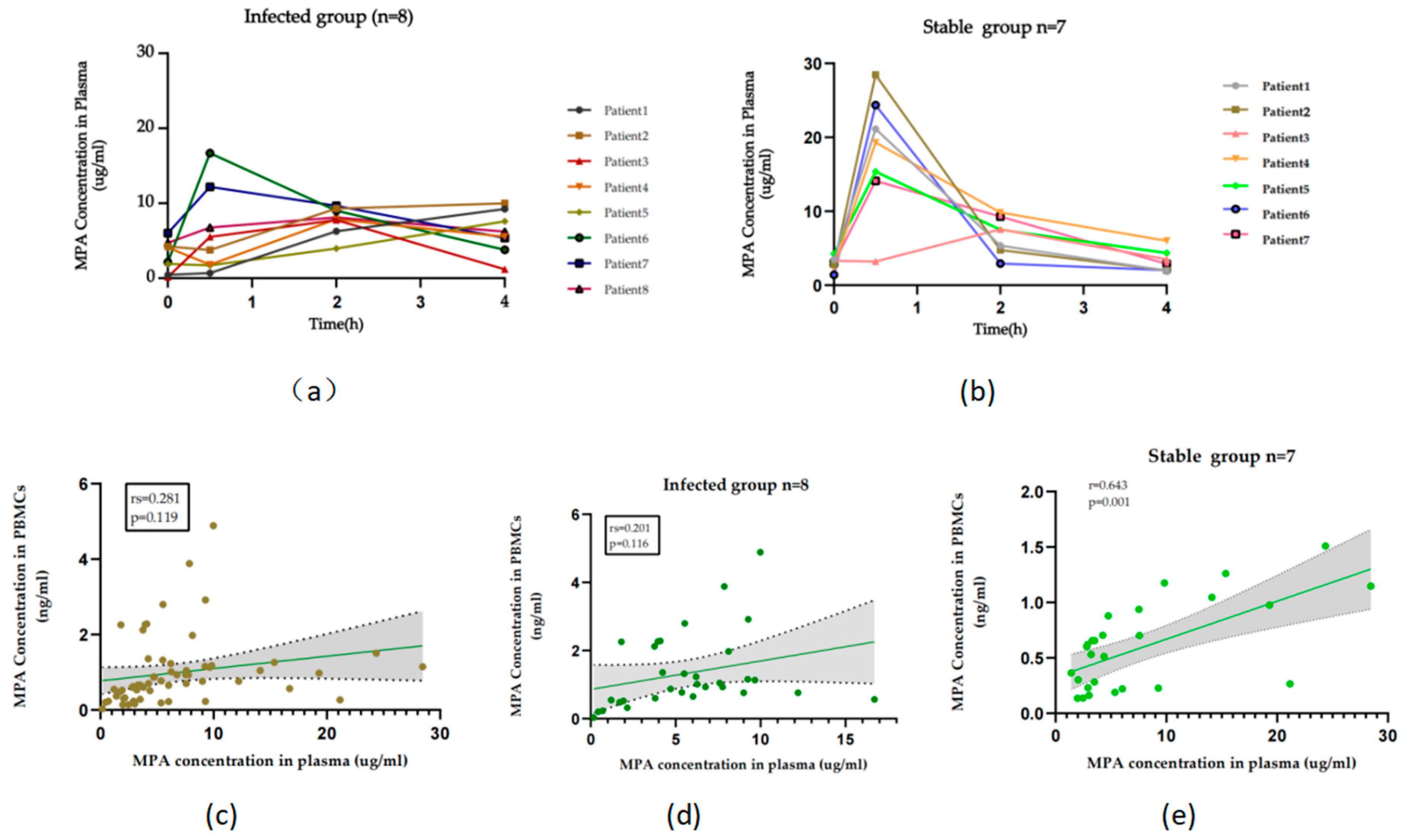
3.2. Clinical application assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Picard, N.; Ratanasavanh, D.; Prémaud, A.; Le Meur, Y.; Marquet, P. Identification of UDP glucuronosyltransferase isoforms involved in mycophenolic acid phase II metabolism. Drug Metab Dispos 2005, 33, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Kazuaki Yoshimura et al. Pharmacokinetic and Pharmacodynamic Markers of Mycophenolic Acid Associated with Effective Prophylaxis for Acute Graft-Versus-Host Disease and Neutrophil Engraftment in Cord Blood Transplant Patients .Biology of Blood and Marrow Transplantation.2018;S1083-8791. [CrossRef]
- Sobiak J et al.Effffect of mycophenolate mofetil on hematological side effffects incidence in renal transplant recipients.Clin Transplant 2013;E407–E414. [CrossRef]
- Grinyo JM, Ekberg H, Mamelok RD et al: The pharmacokinetics of mycophenolate mofetil in renal transplant recipients receiving standard-dose or low-dose cyclosporine, low-dose tacrolimus or low-dose sirolimus: the Symphony pharmacokinetic substudy. Nephrol Dial Transplant, 2009; 24: 2269–76. 2: Nephrol Dial Transplant, 2009; 24. [CrossRef]
- MASATOMO M, TAKENORI N, SHOUTARO K, et al. Monitoring of mycophenolic acid predose concentrations in the maintenance phase more than one year after renal transplantation.[J]. Therapeutic drug monitoring, 2011,33(3). [CrossRef]
- Zegarska J, Hryniewiecka E, Żochowska D, et al. Mycophenolic acid metabolites acyl-glucuronide and glucoside affect the occurrence of infectious complications and bone marrow dysfunction in liver transplant recipients. Ann Transplant. 2015;20:483-492. [CrossRef]
- Joseph Berthier et al.MRP4 is responsible for the efflux transport of mycophenolic acid β-D glucuronide(MPAG) from hepatocytes to blood .Xenobiotica 2020.1813352. [CrossRef]
- Sobiak J et al.Effffect of mycophenolate mofetil on hematological side effffects incidence in renal transplant recipients.Clin Transplant 2013;E407–E414. [CrossRef]
- Kaplan B.Decreased protein binding of mycophenolic acid associated with leukopenia in a pancreas transplant recipient with renal failure.Transplantation.1998 Apr 27;65(8):1127-9.
- David W Mudge.Severe toxicity associated with a markedly elevated mycophenolic acid free fraction in a renal transplant recipient.Ther Drug Monit. 2004 Aug;26(4):453-5.
- Kamińska, J. Pharmacokinetics of mycophenolic acid and its phenyl glucuronide metabolite in kidney transplant recipients with renal impairment Arch Med Sci. 2012 Feb 29;8(1):88-96.
- Sobiak, J. Effect of mycophenolate mofetil on hematological side effects incidence in renal transplant recipients.Clin Transplant. 2013 Jul-Aug;27(4):E407-14.
- Colom H.Pharmacokinetic modeling of enterohepatic circulation of mycophenolic acid in renal transplant recipients.Kidney Int. 2014 Jun;85(6):1434-43. [CrossRef]
- Olivier Gensburger.Effect of Mycophenolate Acyl-Glucuronide on Human Recombinant Type 2 Inosine Monophosphate Dehydrogenase.Clinical Chemistry. [CrossRef]
- M. Shipkova The Acyl Glucuronide Metabolite of Mycophenolic Acid Inhibits the Proliferation of Human Mononuclear Leukocytes. Elsevier Science Inc 2001.PII S0041-1345.
- Jolanta Zegarska Mycophenolic Acid Metabolites Acyl-Glucuronide and Glucoside Affect the Occurrence of Infectious Complications and Bone Marrow Dysfunction in Liver Transplant Recipients.© Ann Transplant, 2015; 20. [CrossRef]
- Wieland, E. Induction of Cytokine Release by the Acyl Glucuronide of Mycophenolic Acid: A Link to Side Effects? Clinical Biochemistry 2000, 33, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Zegarska J, Hryniewiecka E, Żochowska D, et al. Mycophenolic acid metabolites acyl-glucuronide and glucoside affect the occurrence of infectious complications and bone marrow dysfunction in liver transplant recipients. Ann Transplant. 2015;20:483-492. [CrossRef]
- Staatz CE, Tett SE. Clinical pharmacokinetics and pharmacodynamics of mycophenolate in solid organ transplant recipients. Clin Pharmacokinet 2007; 46: 14–58. [CrossRef]
- Nguyen Thi MT, Capron A, Mourad M, Wallemacq P. Mycophenolic acid quantification in human peripheral blood mononuclear cells using liquid chromatography-tandem mass spectrometry. Clin Biochem. 2013 Dec;46(18):1909-11. [CrossRef]
- Bénech H, Hascoët S, Furlan V, Pruvost A, Durrbach A. Development and validation of an LC/MS/MS assay for mycophenolic acid in human peripheral blood mononuclear cells. J Chromatogr B Analyt Technol Biomed Life Sci. 2007 Jun 15;853(1-2):168-74. [CrossRef]
- Bioanalytical Method Validation Guidance for Industry. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bioanalytical-method-validation-guidance-industry (accessed on 10 September 2022).
- Bioanalytical Method Validation. Available online: https://www.ema.europa.eu/en/bioanalytical-method-validation (accessed on 10 September 2022).
- Muzeeb Syed,and Nuggehally R. Srinivasb .A comprehensive review of the published assays for the quantitation of the immunosuppressant drug mycophenolic acid and its glucuronidated metabolites in biological fluids.Biomed. Chromatogr. 2016; 30: 721–748.
- Bing Chen, PhD, Jia-Qian Lu, MSc,Pei-Jun Zhou, MD, PhD et al.Establishment of a Liquid Chromatography-Tandem Mass Spectrometry Method for the Determination of Immunosuppressant Levels in the Peripheral Blood Mononuclear Cells of Chinese Renal Transplant Recipients.Ther Drug Monit 2020;42:686–694. [CrossRef]
- Henriette de Loor elt al.Stability of mycophenolic acid and glucuronide metabolites in human plasma and the impact of deproteinization methodology.Clinica Chimica Acta.2008;389 :87–92. [CrossRef]
- Elbarbry FA, Shoker AS.Therapeutic drug measurement of mycophenolic acid derivatives in transplant patients .Clin Biochem. 2007 Jul;40(11):752-64. [CrossRef]
- Halloran PF. Immunosuppressive drugs for kidney transplantation. N Engl J Med 2004; 351:2715–29. [CrossRef]
- Ferrara, J.L.; Levine, J.E.; Reddy, P.; Holler, E. Graft-versus-host disease. Lancet 2009, 373, 1550–1561. [Google Scholar] [CrossRef] [PubMed]
- DOM Z I M, COLLER J K, CARROLL R P, et al. Mycophenolic acid concentrations in peripheral blood mononuclear cells are associated with the incidence of rejection in renal transplant recipients[J]. British Journal of Clinical Pharmacology, 2018,84(10). [CrossRef]
| Analyte | ESI mode | Parent ion (m/z) |
Product ion (m/z) |
Cone voltage (V) |
Collision energy (eV) |
|---|---|---|---|---|---|
| MPA | + | 321.27 | 207.15 | 80.00 | 22.00 |
| MPAG | + | 514.27 | 321.20 | 8.00 | 10.00 |
| AcMPAG | + | 514.26 | 321.20 | 40.00 | 10.00 |
| MPA-d3 | + | 324.26 | 210.15 | 8.00 | 22.00 |
| QCs | Peak area in PBMCs | Peak area in Methanol | Matrix effect(%) | Recovery(%) | |
|---|---|---|---|---|---|
| MPA | LLOQ | 559.13 | 562.82 | -3.65 | 104.4 |
| LQC | 1129.28 | 1115.94 | 0.59 | 103.56 | |
| MQC | 44215.02 | 51203.82 | -9.22 | 100.24 | |
| HQC | 176298.91 | 184589.71 | -2.01 | 96.2 | |
| MPAG | LLOQ | 457.17 | 485.81 | -6.74 | 91.39 |
| LQC | 1015.68 | 942.62 | 10.45 | 96.71 | |
| MQC | 6703.8 | 6250.53 | 6.63 | 109.98 | |
| HQC | 25811.82 | 24554.00 | 7.21 | 107.13 | |
| AcMPAG | LLOQ | 82.43 | 92.06 | -7.49 | 86.6 |
| LQC | 235.01 | 261.15 | -11.42 | 91.62 | |
| MQC | 2962.55 | 3040.46 | -6.45 | 98.58 | |
| HQC | 11953.42 | 12424.06 | -3.66 | 94.95 | |
| Concentration (ng/mL) |
Intra-batch | Inter-batch | |||||
|---|---|---|---|---|---|---|---|
| Found (ng/mL) |
Imprecision CV (%) |
Accuracy (%) |
Found (ng/mL) |
Imprecision CV (%) |
Accuracy (%) |
||
| MPA * | LLOQ(0.05) | 0.04 | 6.15 | 93.86 | 0.04 | 0.92 | 99.08 |
| LQC(0.15) | 0.14 | 3.52 | 96.48 | 0.15 | 3.40 | 97.60 | |
| MQC(10.00) | 9.41 | 5.95 | 94.05 | 9.49 | 5.36 | 94.64 | |
| HQC(40.00) | 37.61 | 5.98 | 94.02 | 36.28 | 10.25 | 89.75 | |
| MPAG* | LLOQ(0.50) | 0.47 | 5.72 | 94.20 | 0.48 | 4.78 | 95.22 |
| LQC(1.50) | 1.47 | 2.14 | 97.87 | 1.48 | 1.27 | 99.63 | |
| MQC(10.00) | 9.74 | 2.56 | 97.44 | 10.82 | 7.56 | 92.44 | |
| HQC(40.00) | 38.31 | 4.23 | 95.77 | 40.92 | 2.24 | 97.76 | |
| LLOQ(0.10) | 0.09 | 3.76 | 96.00 | 0.09 | 9.39 | 91.61 | |
| AcMPAG | LQC(0.30) MQC(4.00) HQC(16.00) |
0.28 | 6.69 | 93.33 | 0.28 | 7.17 | 92.83 |
| 3.84 | 4.10 | 95.90 | 4.16 | 3.93 | 96.07 | ||
| 15.54 | 2.88 | 97.12 | 15.70 | 1.88 | 98.12 | ||
| QCs | Room temperature | Frozen(4℃) | Freeze-Thaw(Round1-3) | Autosampler | ||||
|---|---|---|---|---|---|---|---|---|
| 2h-RSD(%) | 8h-RSD (%) |
24h-RSD (%) |
R1-RSD(%) | R2-RSD(%) | R3-RSD(%) | 24h-RSD(%) | ||
| MPA | LLOQ | 8.40 | 6.33 | 3.31 | 0.90 | 2.33 | 6.66 | 2.33 |
| LQC | 10.67 | 10.67 | 5.45 | 10.67 | 10.68 | 10.66 | 10.67 | |
| MQC | 1.09 | 2.29 | 3.53 | 13.65 | 11.58 | 7.73 | 2.28 | |
| HQC | 7.85 | 2.15 | 9.38 | 10.63 | 12.12 | 12.43 | 7.85 | |
| MPAG | LLOQ | 10.56 | 14.4 | 5.51 | 5.20 | 3.50 | 5.13 | 5.19 |
| LQC | 1.55 | 6.06 | 1.14 | 4.61 | 0.13 | 3.31 | 1.15 | |
| MQC | 10.30 | 5.64 | 0.67 | 10.87 | 8.00 | 5.96 | 5.64 | |
| HQC | 1.88 | 9.92 | 9.76 | 7.58 | 8.48 | 7.04 | 8.47 | |
| AcMPAG | LLOQ | 11.66 | 4.10 | 6.95 | 3.50 | 2.15 | 36.50 | 6.95 |
| LQC | 5.16 | 6.80 | 2.46 | 1.44 | 2.05 | 8.05 | 1.43 | |
| MQC | 3.48 | 14.11 | 5.05 | 11.77 | 13.1 | 19.67 | 14.09 | |
| HQC | 4.32 | 8.44 | 4.58 | 1.75 | 0.76 | 2.42 | 0.76 | |
| QCs | 24h-RSD (%) |
48h-RSD (%) |
72h-RSD (%) |
7d-RSD (%) |
14d-RSD (%) |
||
|---|---|---|---|---|---|---|---|
| MPA | LLOQ | 4.00 | 2.00 | 2.00 | 12.00 | 2.00 | |
| LQC | 5.33 | 0.67 | 4.00 | 10.66 | 11.33 | ||
| MQC | 2.01 | 1.08 | 0.02 | 10.04 | 11.09 | ||
| HQC | 0.31 | 0.80 | 0.39 | 5.52 | 5.02 | ||
| MPAG | LLOQ | 2.60 | 0.20 | 13.60 | 4.20 | 4.60 | |
| LQC | 2.13 | 8.80 | 0.86 | 4.46 | 0.13 | ||
| MQC | 9.80 | 2.11 | 0.01 | 10.87 | 4.33 | ||
| HQC | 0.54 | 1.05 | 0.30 | 2.75 | 2.61 | ||
| AcMPAG | LLOQ | 6.00 | 12.16 | 19.33 | 49.83 | 56.83 | |
| LQC | 13.50 | 30.16 | 48.61 | 53.61 | 53.05 | ||
| MQC | 36.26 | 40.04 | 43.25 | 51.50 | 56.38 | ||
| HQC | 34.92 | 36.25 | 36.16 | 41.97 | 54.03 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




