Submitted:
16 January 2024
Posted:
16 January 2024
You are already at the latest version
Abstract
Keywords:
Introduction
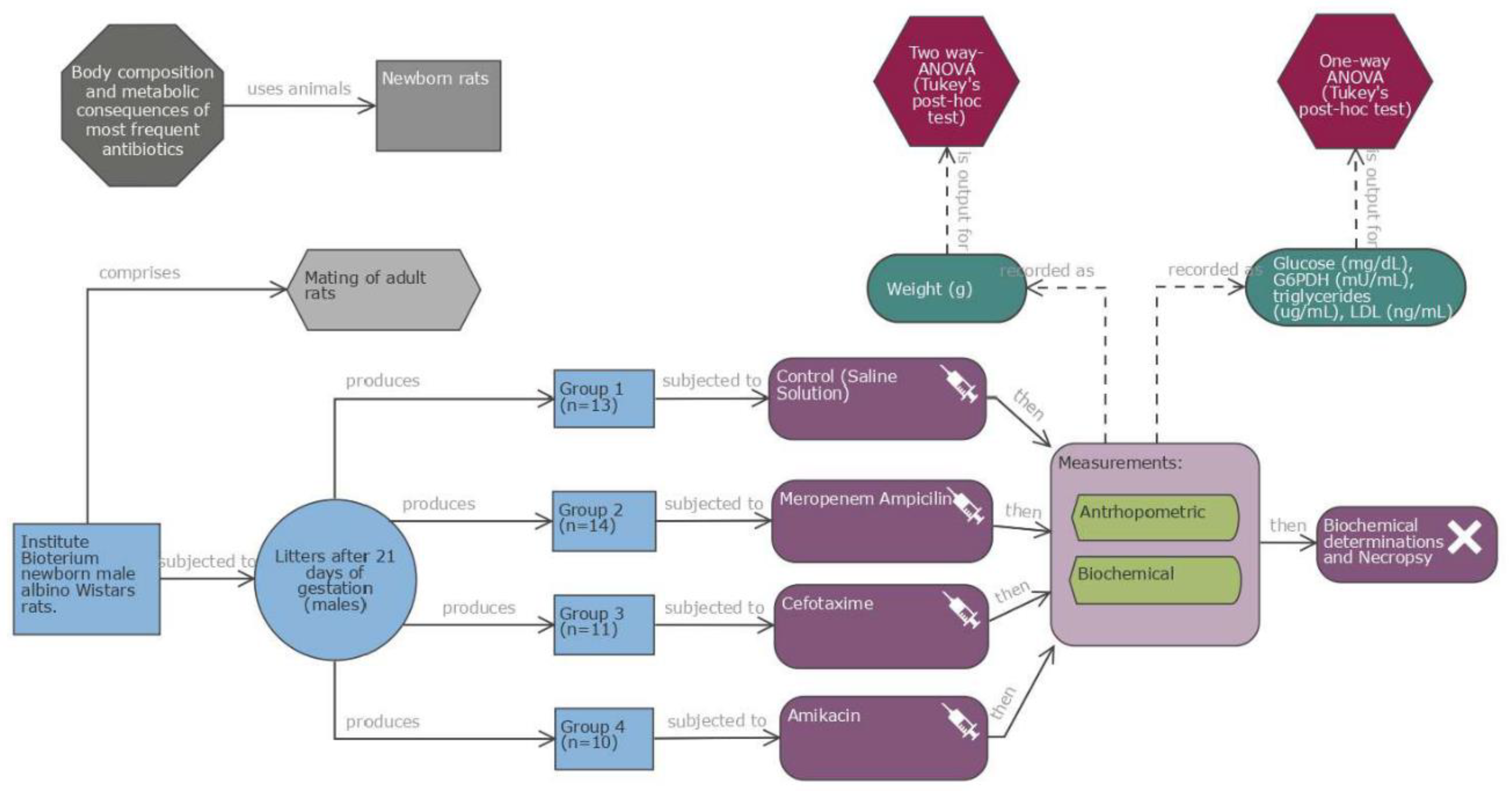
Material and Methods
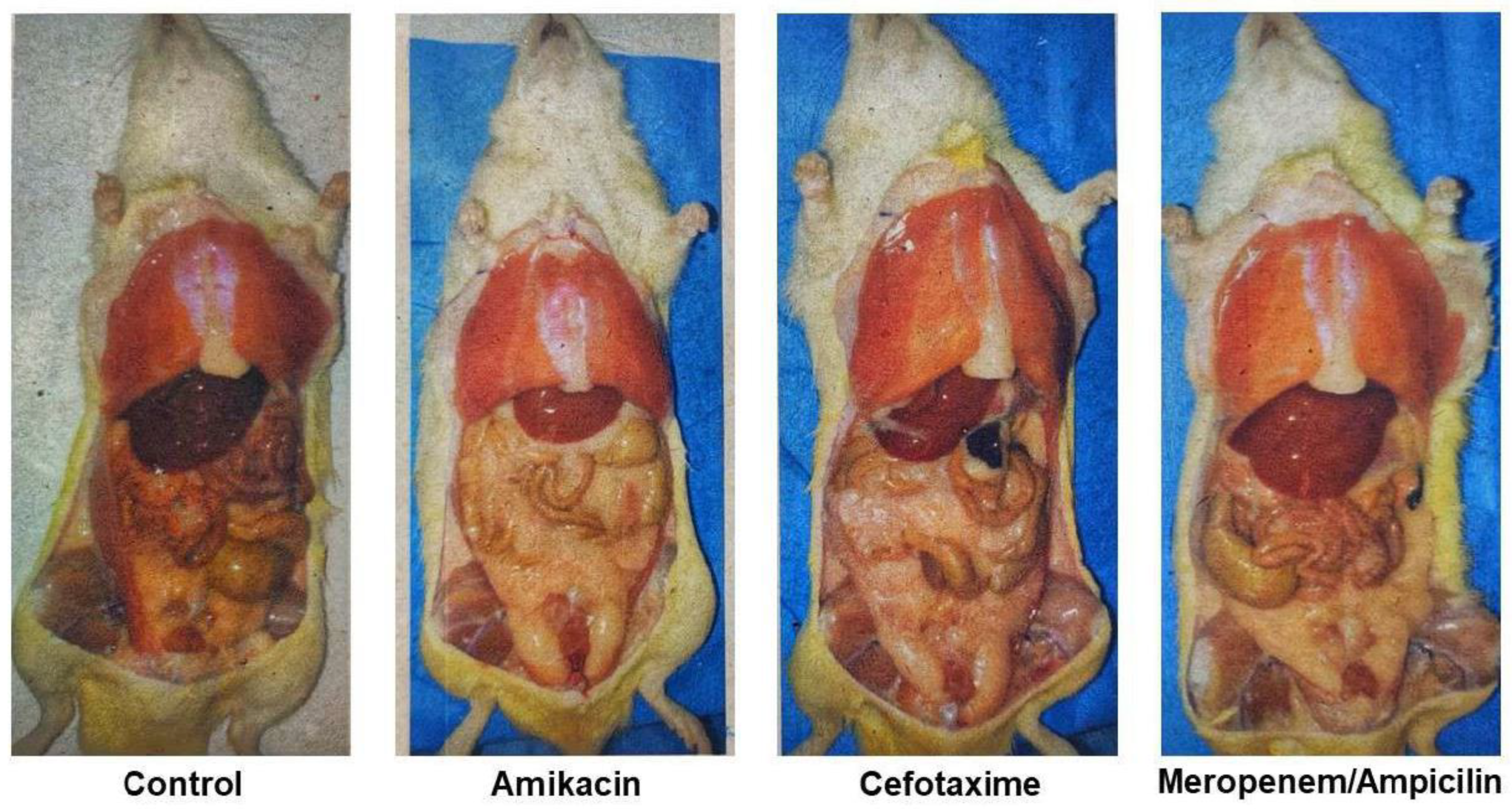
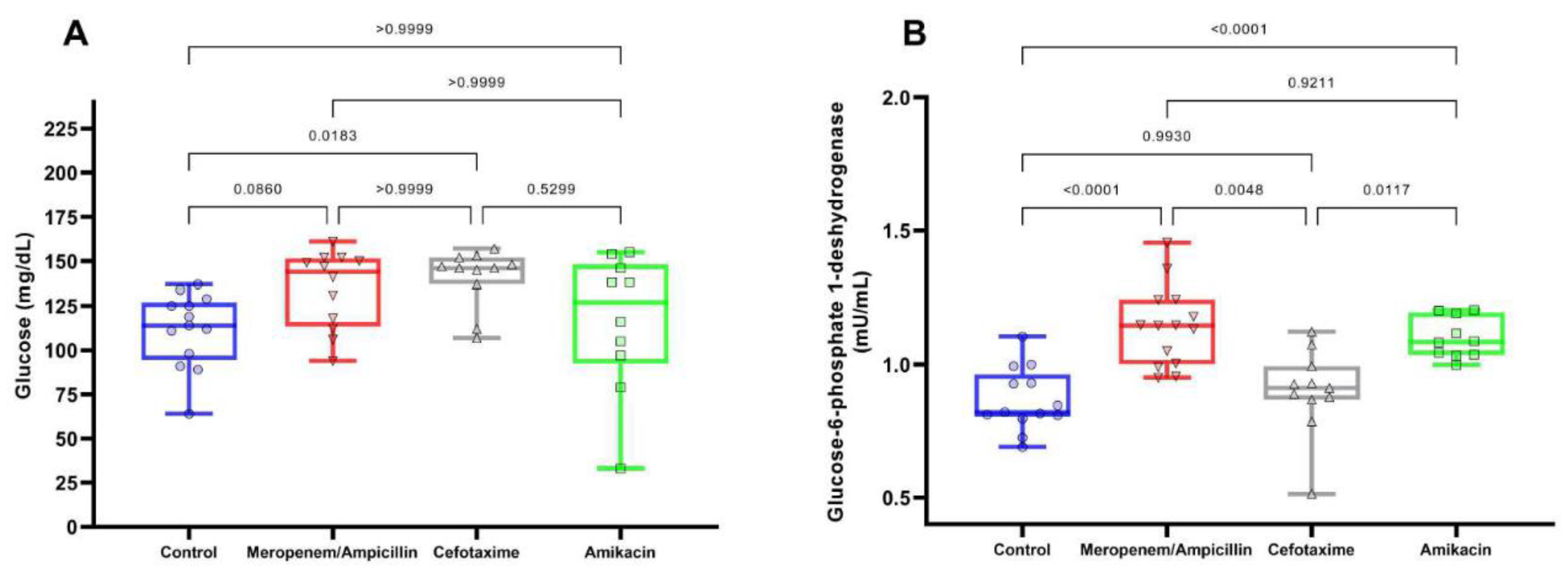
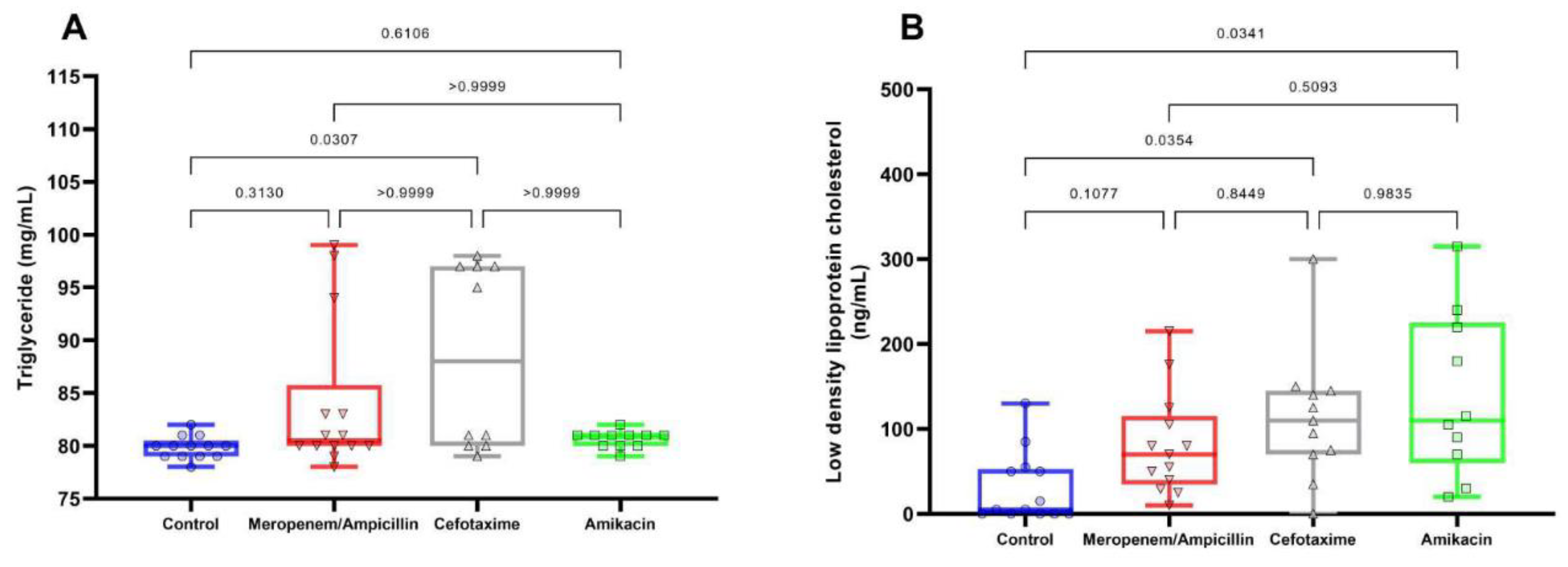
Discussion
Acknowledgments
References
- World Health Organization. Obesity and Overweight. World Health Organization (2021). https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed 24 October 2023).
- Turta O, Rautava S. Antibiotics, obesity and the link to microbes - what are we doing to our children? BMC Med. (2016) 14:57, 1-6. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed 24 October 2023). [CrossRef]
- Cox LM, & Blaser MJ. Antibiotics in early life and obesity. Nat Rev End (2015) 11:3, 182–190. [CrossRef]
- World Health Organization. Newborn health. World Health Organization (2023). https://www.who.int/teams/maternal-newborn-child-adolescent- health-and-ageing/newborn-health/newborn-infections (accessed 24 October 2023).
- Rahman AE, Hossain AT, Zaman S Bin, Salim N, K.C A, Day LT, et al. Antibiotic use for inpatient newborn care with suspected infection: EN-BIRTH multi-country validation study. BMC Pregnancy Childbirth. (2021) 1;21.
- Ting JY, & Shah PS. Antibiotic stewardship in neonates: Challenges and opportunities.Translational Pediatrics (2020) 9:198–201. [CrossRef]
- Segura-Cervantes E, Mancilla-Ramírez J, González-Canudas J, Alba E, Santillán-Ballesteros R, Morales-Barquet D, Sandoval-Plata G, et al. Inflammatory Response in Preterm and Very Preterm Newborns with Sepsis. Mediators of Inflammation 2016. [CrossRef]
- Prusakov P, Goff DA, Wozniak PS, Cassim A, Scipion CEA, Urzúa S, Ronchi A, et al. A global point prevalence survey of antimicrobial use in neonatal intensive care units: The no-more-antibiotics and resistance (NO-MAS-R) study. EClinicalMedicine (2021) 32. [CrossRef]
- Cortese F, Scicchitano P, Gesualdo M, Filaninno A, de Giorgi E, Schettini F, et al. Early and Late Infections in Newborns: Where Do We Stand? A Review. Pediatrics and Neonatology (2016). 57:265–273. [CrossRef]
- Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Critical Care Med (2006) 34:1589–1596. [CrossRef]
- Butranova OI, Ushkalova EA, Zyryanov SK, & Chenkurov MS. Developmental Pharmacokinetics of Antibiotics Used in Neonatal ICU: Focus on Preterm Infants. Biomedicines. (2023) 11:940. [CrossRef]
- Rivera-Chaparro ND, Cohen-Wolkowiez M and Greenberg RG. ‘Dosing antibiotics in neonates: Review of the pharmacokinetic data’. Future Microbiology. (2017) 12:1001–1016. [CrossRef]
- Kwon Y, Cho YS, Lee YM, Kim SJ, Bae J, & Jeong SJ. Changes to Gut Microbiota Following Systemic Antibiotic Administration in Infants. Antibiotics. 2022; 11, 470. [CrossRef]
- Reyman M, van Houten MA, Watson RL, Chu MLJN, Arp K, de Waal WJ, et al. Effects of early-life antibiotics on the developing infant gut microbiome and resistome: a randomized trial. Nature Communications. (2022) 13:1. [CrossRef]
- Cossio-Bolaños M, Campos RG, Vitoria RV, Hochmuller Fogaça RT, de Arruda M. Reference curves for assessing the physical growth of male Wistar rats. Nutr Hosp. (2013) 28:2151–2156.
- Cho I, Yamanishi S, Cox L, Methé BA, Zavadil J, Li K, et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature. (2012) 488:621–626. [CrossRef]
- Le Roy T, Lécuyer E, Chassaing B, Rhimi M, Lhomme M, Boudebbouze S, et al. The intestinal microbiota regulates host cholesterol homeostasis. BMC Biol. (2019) 17:1.
- Aberare OL, Okuonghae P, Mukoro N, et al. Triglycerides, total cholesterol, high density lipoprotein cholesterol and low density lipoprotein cholesterol in rats exposed to premium motor spirit fumes. N Am J Med Sci. 2011;3(6):277-280. [CrossRef]
- O’Dowd JF, & Stocker CJ. Endocrine pancreatic development: Impact of obesity and diet. Frontiers in Physiology. (2013) 4:170. :. [CrossRef]
- Bloomfield, FH. Impact of prematurity for pancreatic islet and beta-cell development. Journal of Endocrinology. 2018; 238, R161–R171. [Google Scholar] [CrossRef]
- Jacovetti C & Regazzi, R. Mechanisms Underlying the Expansion and Functional Maturation of β-Cells in Newborns: Impact of the Nutritional Environment. Int J Mol Sci. (2022) 23:2096. [CrossRef]
- Rehman HU, Ullah K, Rasool A, Manzoor R, Yuan Y, Tareen AM, et al. Comparative impact of streptozotocin on altering normal glucose homeostasis in diabetic rats compared to normoglycemic rats. Sci Rep. 2023 May 16;13(1):7921. [CrossRef]
- Hikmah Nuzulu, Shita AD, Maulana H. View of diabetic blood glucose level profile with stratified dose streptozotocin (SD-STZ) and multi low dose streptozotocin (MLD-STZ) induction methods. (s/f). The Journal of Tropical Life, 5(2015). doi:11594/jtls.5.1.%x.
- Quinn, R. Comparing rat’s to human’s age: How old is my rat in people years? Nutrition. (2005) 21:775–777). [CrossRef]
- Uzan-Yulzari A, Turta O, Belogolovski A, Ziv O, Kunz C, Perschbacher S, et al. ‘Neonatal antibiotic exposure impairs child growth during the first six years of life by perturbing intestinal microbial colonization’, Nature Communications. (2021) 12:443. doi:1038/s41467-020-20495-4.
- Kara A, Cetin H, Oktem F, Ciris IM, Altuntas I, & Kaya S. Amikacin induced renal damage and the role of the antioxidants on neonatal rats. Renal Failure. (2016) 38:671–677. [CrossRef]
- Conceição S, Queiroga MC, Laranjo M. Antimicrobial Resistance in Bacteria from Meat and Meat Products: A One Health Perspective. Microorganisms. 2023 Oct 17;11(10):2581. [CrossRef]
- Tang KL, Caffrey NP, Nóbrega DB, Cork SC, Ronksley PE, Barkema HW, Polachek AJ et al. Restricting the use of antibiotics in food-producing animals and its associations with antibiotic resistance in food-producing animals and human beings: a systematic review and meta-analysis. Lancet Planet Health. 2017 Nov;1(8):e316-e327. [CrossRef]
- Gerber JS, Bryan M, Ross RK, Daymont C, Parks EP, Localio AR, et. al. Antibiotic Exposure During the First 6 Months of Life and Weight Gain During Childhood. JAMA 2016, 315, 1258–1265. [CrossRef]
- Ternak, G. , Edel, Z., Feiszt, Z., Szekeres, J., Visegrady, B. and Wittmann, I. Selective Association between Cephalosporin, Quinolone, Macrolide, and Penicillin Antibiotic Consumption and Childhood Obesity in Europe. 2015, Health, 7, 1306-1314. [CrossRef]
- Ternák G, Németh M, Rozanovic M, Márovics G, Bogár L. “Growth-Promoting Effect" of Antibiotic Use Could Explain the Global Obesity Pandemic: A European Survey. Antibiotics (Basel). 2022 Sep 28;11(10):1321. [CrossRef]
- Becker J, Schüpbach-Regula G, Steiner A, Perreten V, Wüthrich D, Hausherr A, et al. Effects of the novel concept 'outdoor veal calf' on antimicrobial use, mortality and weight gain in Switzerland. (2020). Preventive veterinary medicine, 176, 104907. [CrossRef]
| ANTIBIOTIC | MEROPENEM | AMPICILLIN | CEFOTAXIME | AMIKACIN |
|---|---|---|---|---|
| FAMILY | Carbapenems | Beta-lactams | Third generation cephalosporins | Aminoglycoside |
| MECHANISM OF ACTION | Inhibits cell wall synthesis, binding to penicillin-binding proteins (PBPs) type 2, 3 and 4, thus facilitating bacterial lysis, bactericidal effect. | Inhibits the last stage of cell wall synthesis by binding to the inactivating transpeptidase, therefore preventing the cross-linking of the peptidoglycan chains that gives the cell wall structure, resulting in bacterial lysis and its subsequent death. | Inhibits the last stage of bacterial wall synthesis by binding to PBP’s, disrupting synthesis and causing cell lysis. | It binds to the 30S subunit of the bacterial ribosome, stopping bacterial DNA transcription and protein synthesis. |
| SPECIFIC ACTION | Gram +, Gram – and strict anaerobes. It is prescribed for complicated, intra-abdominal polymicrobial infections. Also used in cases of meningitis. | Group B Streptococcus, Listeria monocytogenes and Escherichia coli. | Gram +, Gram – broad spectrum. Streptococcus spp. and gram-negative bacilli (GNB). It has no activity against non-fermenting GNBs (Pseudomonas aeruginosa, Acinetobacter baumannii), L. monocytogenes, Enterococcus spp and strict anaerobes. | Gram – Pseudomonas, E. coli and species of Proteus, Providencia, Klebsiella, Enterobacter, Serratia y Acinetobacter |
| ELIMINATION | Renal elimination, including glomerular filtration and tubular secretion. | Renal, glomerular filtration and tubular secretion, with 10% hepatic metabolism. | Renal elimination | Renal elimination |
| TOXICITY | Interstitial nephritis, neurotoxicity and platelet disturbance. | Its use has been associated with an increased risk of necrotising enterocolitis (NEC). | It has risk of causing irreversible, bilateral, symmetrical ototoxicity. | |
| NEWBORN DOSE | Recommended dose 20 mg/kg every 12 h, for those under 32 gestational weeks and 30 mg/kg for those older than 32 weeks, every 8 h. | The commonly used dose is 200 mg/kg/day (range 100-350 mg/kg/day every 6-12 h, extrapolated from adults studies). Although by gestational age (GA) for premature babies, 50 mg/kg/dose every 12 h, or every 8 h if the baby is >34 weeks GA | Recommended dose 50 mg/kg/dose every 12 h for the first week of life | Dose according to weight of new-born 16 mg/kg every 36 to 48 h. |
| Control group (n=13) |
Meropenem/ Ampicillin group (n=14) |
Cefotaxime group (n=11) |
Amikacin group (n=10) |
|
|---|---|---|---|---|
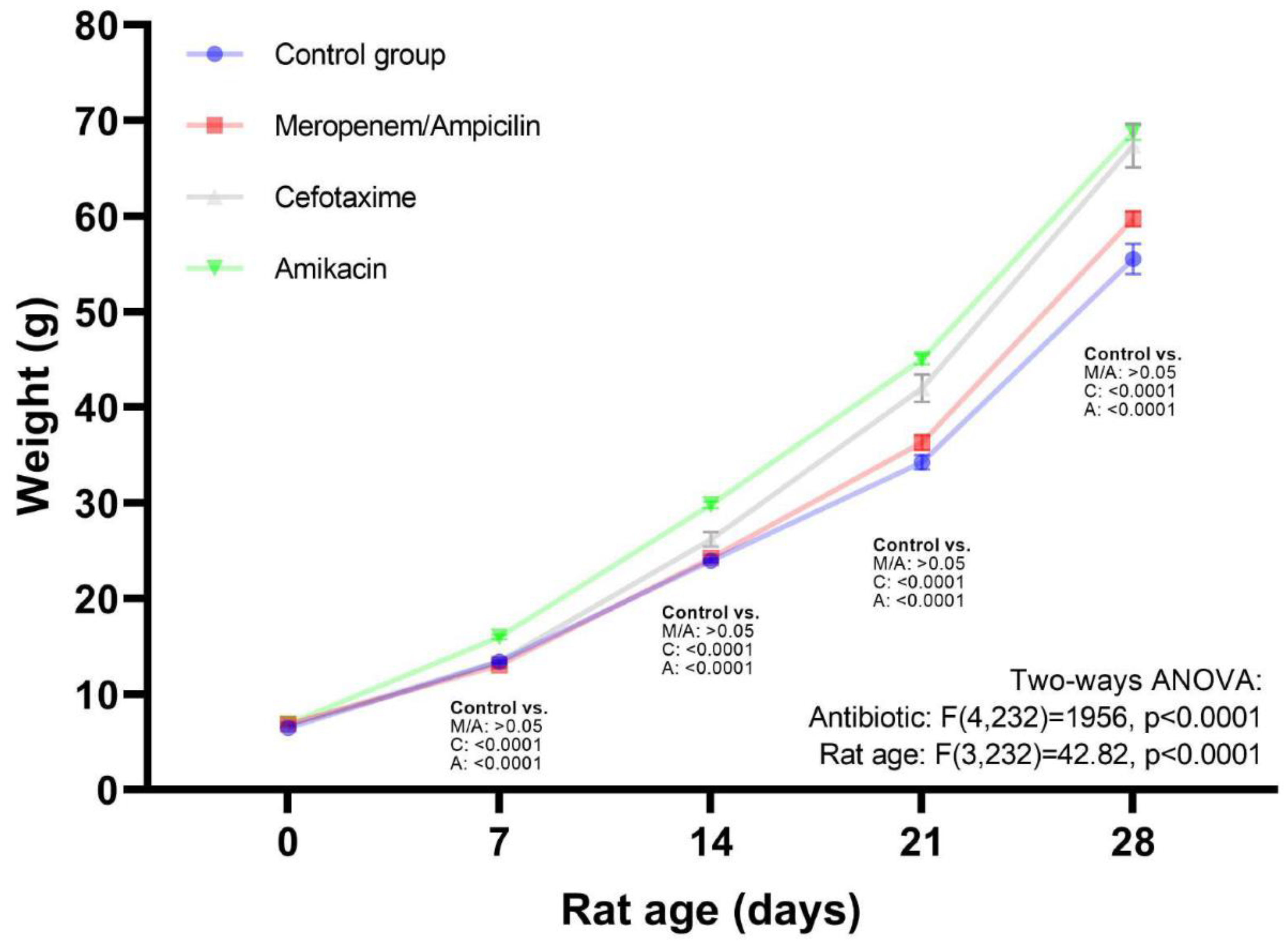
| Weight (g) | 6.48 (0.43) | 6.83 (0.47) | 6.81 (0.96) | 6.88 (0.43) |
| Rostro-caudal length (cm) | 7.11 (0.23) | 7.10 (0.24) | 7.32 (0.41) | 7.64 (0.32) |
| Thoracic diameter(cm) | 3.97 (0.27) | 4.21 (0.23) | 4.05 (0.29) | 4.16 (0.11) |
| Abdominal diameter (cm) | 4.10 (0.28) | 4.26 (0.30) | 4.05 (0.25) | 4.35 (0.12) |
| Control | M/A | Cefotaxime | Amikacin | |
|---|---|---|---|---|
| Baseline | ||||
| Mean (SD) | 6.48 (0.43) | 6.84 (0.48) | 6.81 (0.96) | 6.88 (0.43) |
| p values multi test comparison | ||||
| vs. Control | - | 0.79 | <0.0001 | <0.0001 |
| vs. M/A | 0.79 | - | <0.0001 | <0.0001 |
| Vs. Cefotaxime | <0.0001 | <0.0001 | - | 0.17 |
| vs. Amikacin | <0.0001 | <0.0001 | 0.17 | - |
| q values for controlling FDR | ||||
| vs. Control | - | 0.06 | 0.06 | 0.06 |
| vs. M/A | 0.06 | - | 0.08 | 0.08 |
| Vs. Cefotaxime | 0.06 | 0.08 | - | 0.07 |
| vs. Amikacin | 0.06 | 0.08 | 0.07 | - |
| 7 days | ||||
| Mean (SD) | 13.38 (0.65) | 13.02 (0.95) | 13.45 (1.59) | 15.98 (0.71) |
| p values multi test comparison | ||||
| vs. Control | - | 0.79 | <0.0001 | <0.0001 |
| vs. M/A | 0.79 | - | 0.0002 | <0.0001 |
| Vs. Cefotaxime | <0.0001 | 0.0002 | - | 0.17 |
| vs. Amikacin | <0.0001 | <0.0001 | 0.17 | - |
| q values for controlling FDR | ||||
| vs. Control | - | 0.06 | 0.07 | 0.002 |
| vs. M/A | 0.06 | - | 0.06 | 0.001 |
| Vs. Cefotaxime | 0.07 | 0.06 | - | 0.003 |
| vs. Amikacin | 0.002 | 0.001 | 0.003 | - |
| 14 days | ||||
| Mean (SD) | 23.97 (0.61) | 24.26 (1.22) | 26.19 (2.53) | 29.81 (1.12) |
| p values multi test comparison | ||||
| vs. Control | - | 0.79 | <0.0001 | <0.0001 |
| vs. M/A | 0.79 | - | 0.0002 | <0.0001 |
| Vs. Cefotaxime | <0.0001 | 0.0002 | - | 0.16 |
| vs. Amikacin | <0.0001 | <0.0001 | 0.16 | - |
| q values for controlling FDR | ||||
| vs. Control | - | 0.06 | 0.005 | <0.0001 |
| vs. M/A | 0.06 | - | 0.007 | <0.0001 |
| Vs. Cefotaxime | 0.005 | 0.007 | - | 0.0003 |
| vs. Amikacin | <0.0001 | <0.0001 | 0.0003 | - |
| 21 days | ||||
| Mean (SD) | 34.26 (2.71) | 36.34 (2.89) | 42 (4.71) | 45.03 (1.72) |
| p values multi test comparison | ||||
| vs. Control | - | 0.79 | <0.0001 | <0.0001 |
| vs. M/A | 0.79 | - | 0.0002 | <0.0001 |
| Vs. Cefotaxime | <0.0001 | 0.0002 | - | 0.16 |
| vs. Amikacin | <0.0001 | <0.0001 | 0.16 | - |
| q values for controlling FDR | ||||
| vs. Control | - | 0.005 | <0.0001 | <0.0001 |
| vs. M/A | 0.005 | - | <0.0001 | <0.0001 |
| Vs. Cefotaxime | <0.0001 | <0.0001 | - | 0.001 |
| vs. Amikacin | <0.0001 | <0.0001 | 0.001 | - |
| 28 days | ||||
| Mean (SD) | 55.51 (5.67) | 59.72 (2.99) | 67.40 (7.52) | 68.77 (2.49) |
| p values multi test comparison | ||||
| vs. Control | - | 0.78 | <0.0001 | <0.0001 |
| vs. M/A | 0.78 | - | 0.0002 | <0.0001 |
| Vs. Cefotaxime | <0.0001 | 0.0002 | - | 0.17 |
| vs. Amikacin | <0.0001 | <0.0001 | 0.17 | - |
| q values for controlling FDR | ||||
| vs. Control | - | <0.0001 | <0.0001 | <0.0001 |
| vs. M/A | <0.0001 | - | <0.0001 | <0.0001 |
| Vs. Cefotaxime | <0.0001 | <0.0001 | - | 0.02 |
| vs. Amikacin | <0.0001 | <0.0001 | 0.02 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




