Submitted:
05 January 2024
Posted:
08 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Embryology of the Aortic Valve
Normal outflow tract and valve formation
3. Transcriptional Regulation of Valvulogenesis
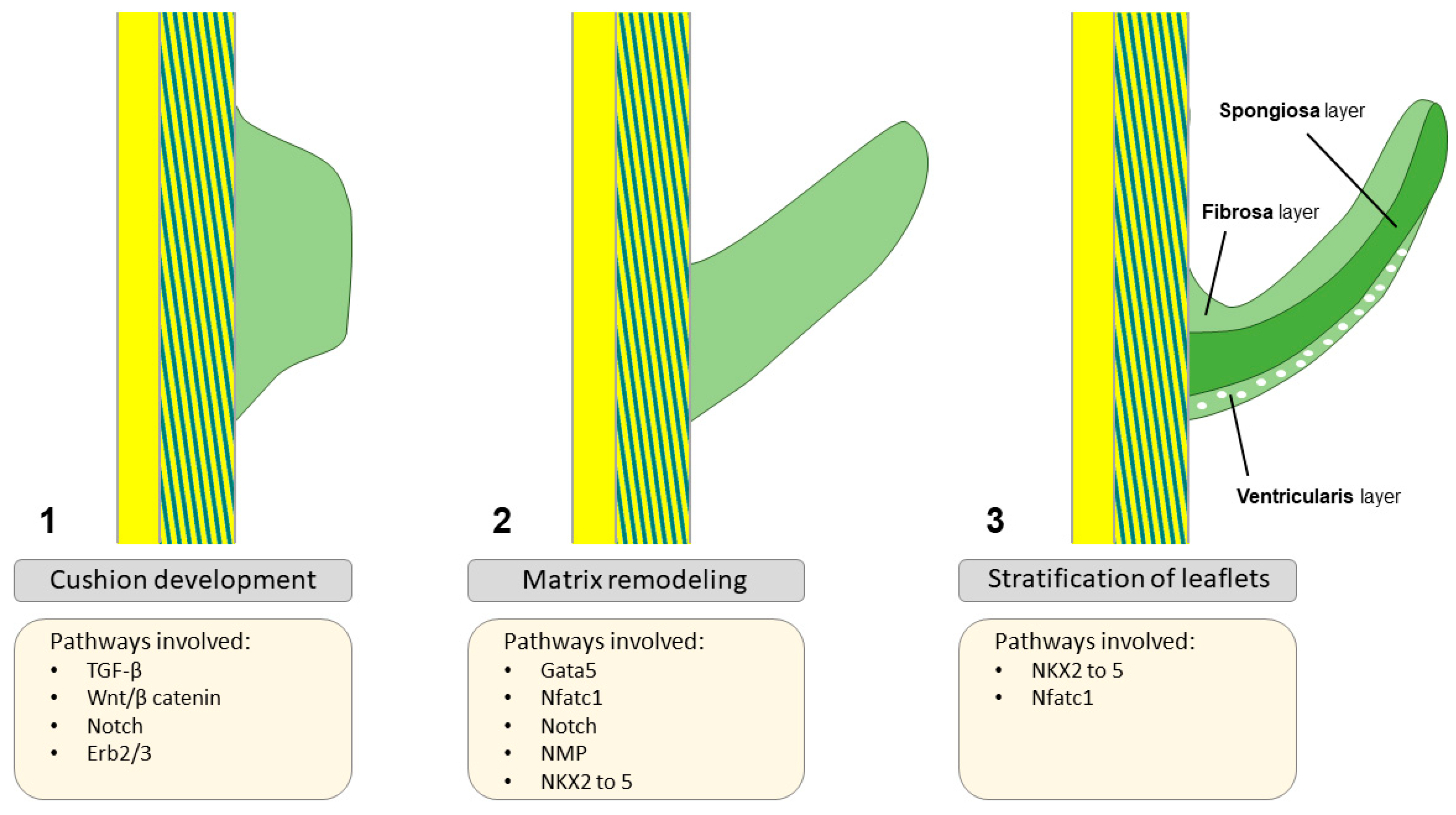
Timing/expression of transcription factors and deficient aortic valve formation
4. Anatomy and Hemodynamics
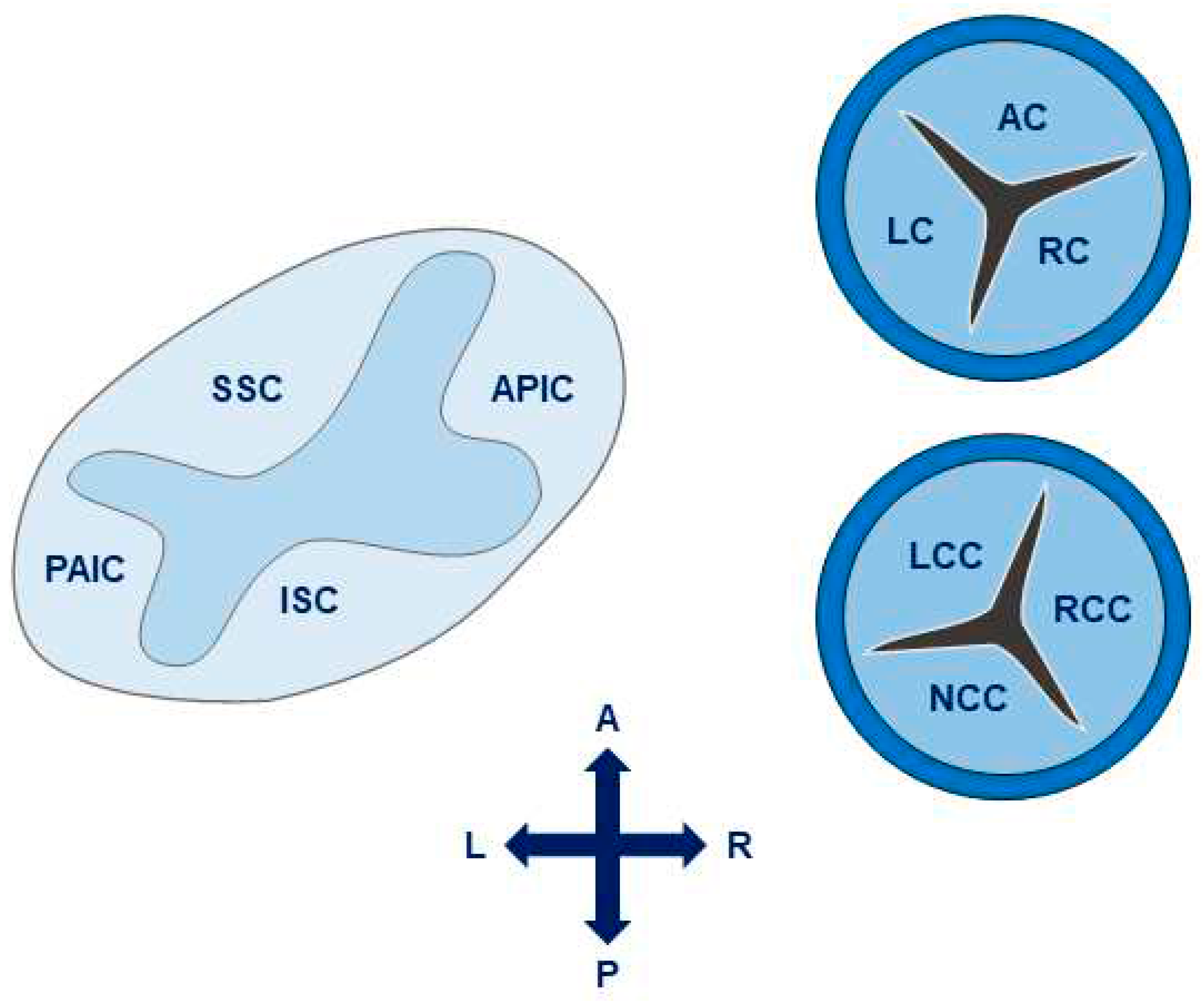
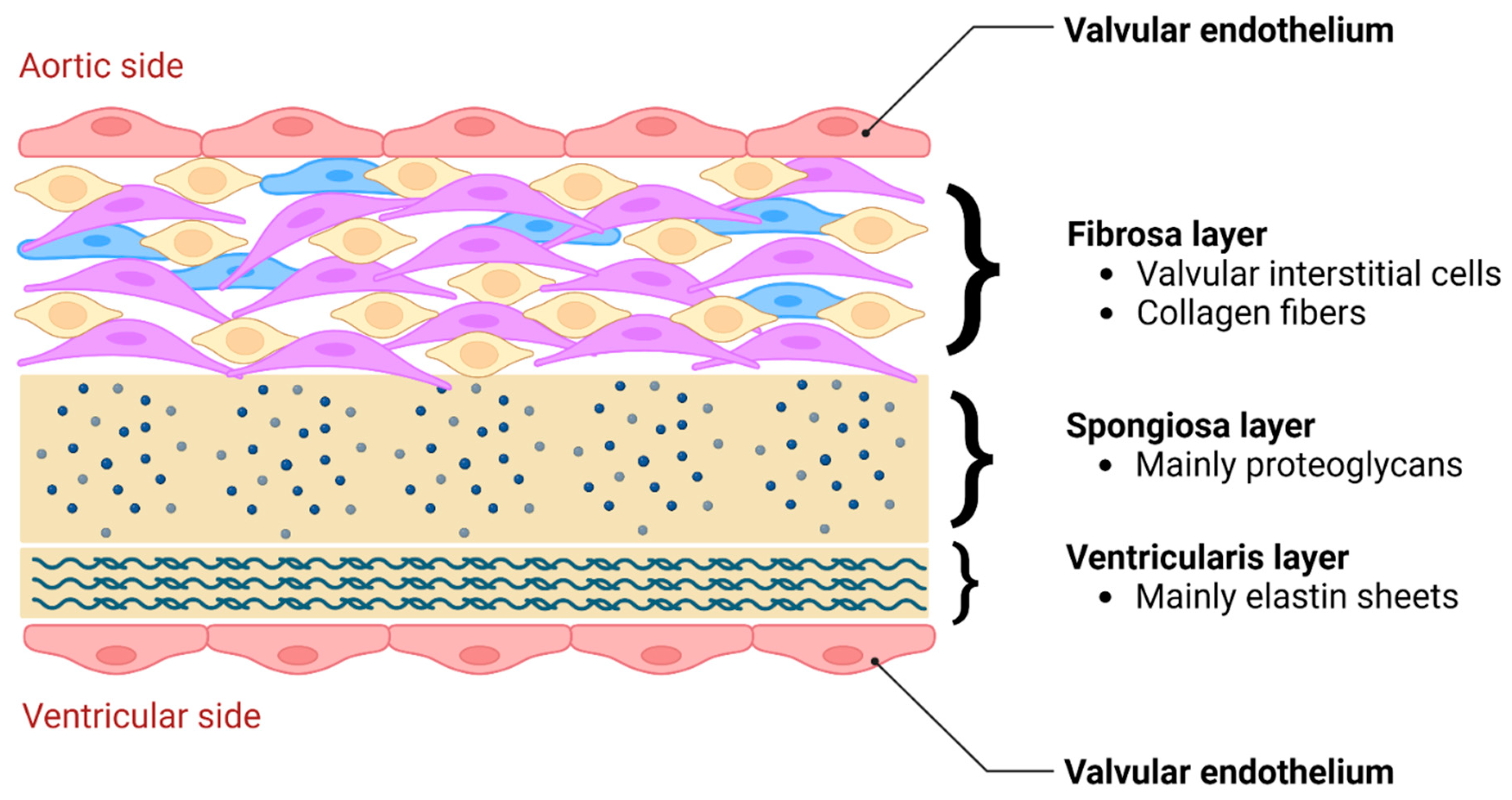
4.1. The aortic valve and root
4.2. Valvular fluid dynamics
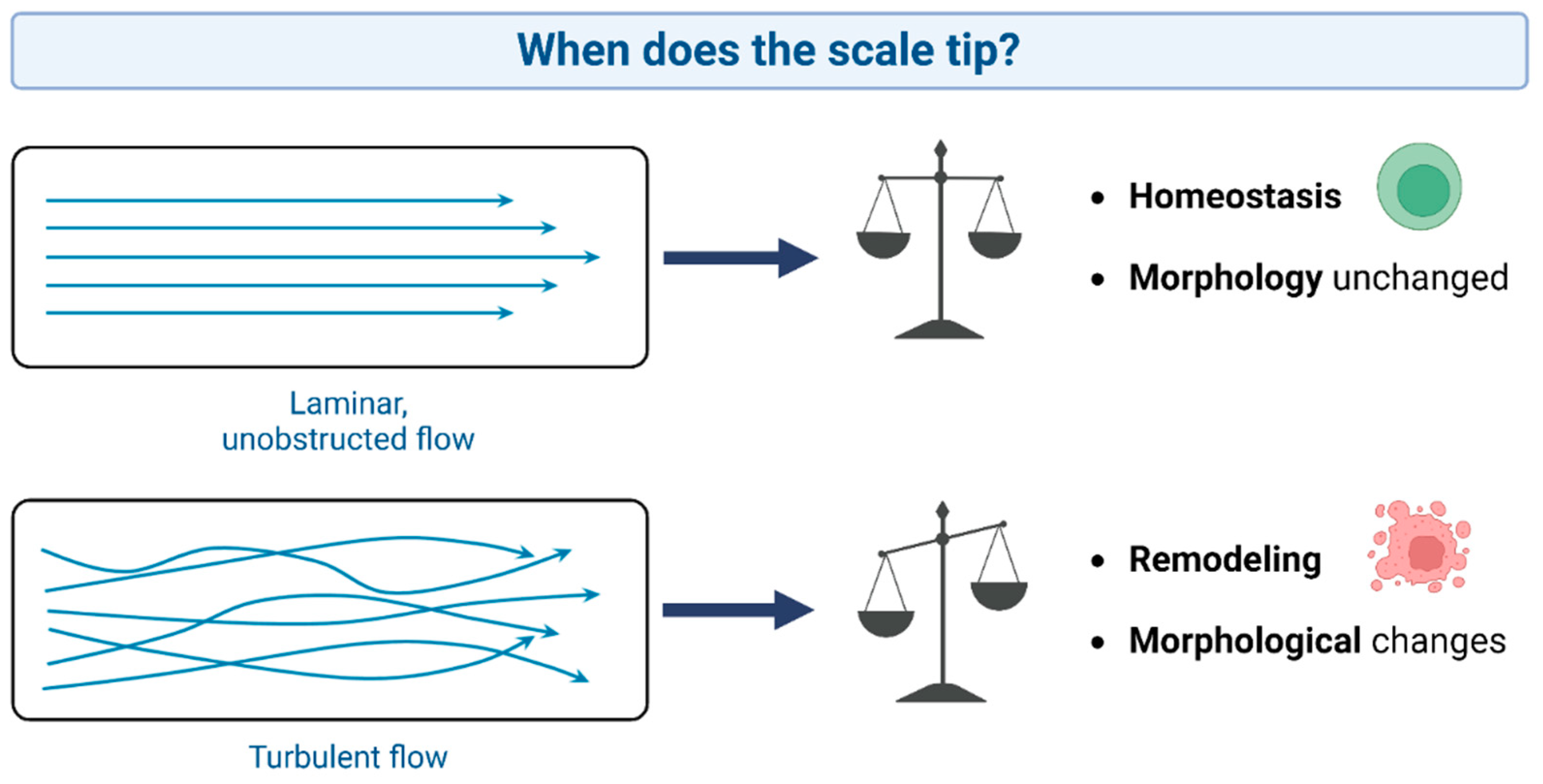
5. Biomechanics and Cellular Responses
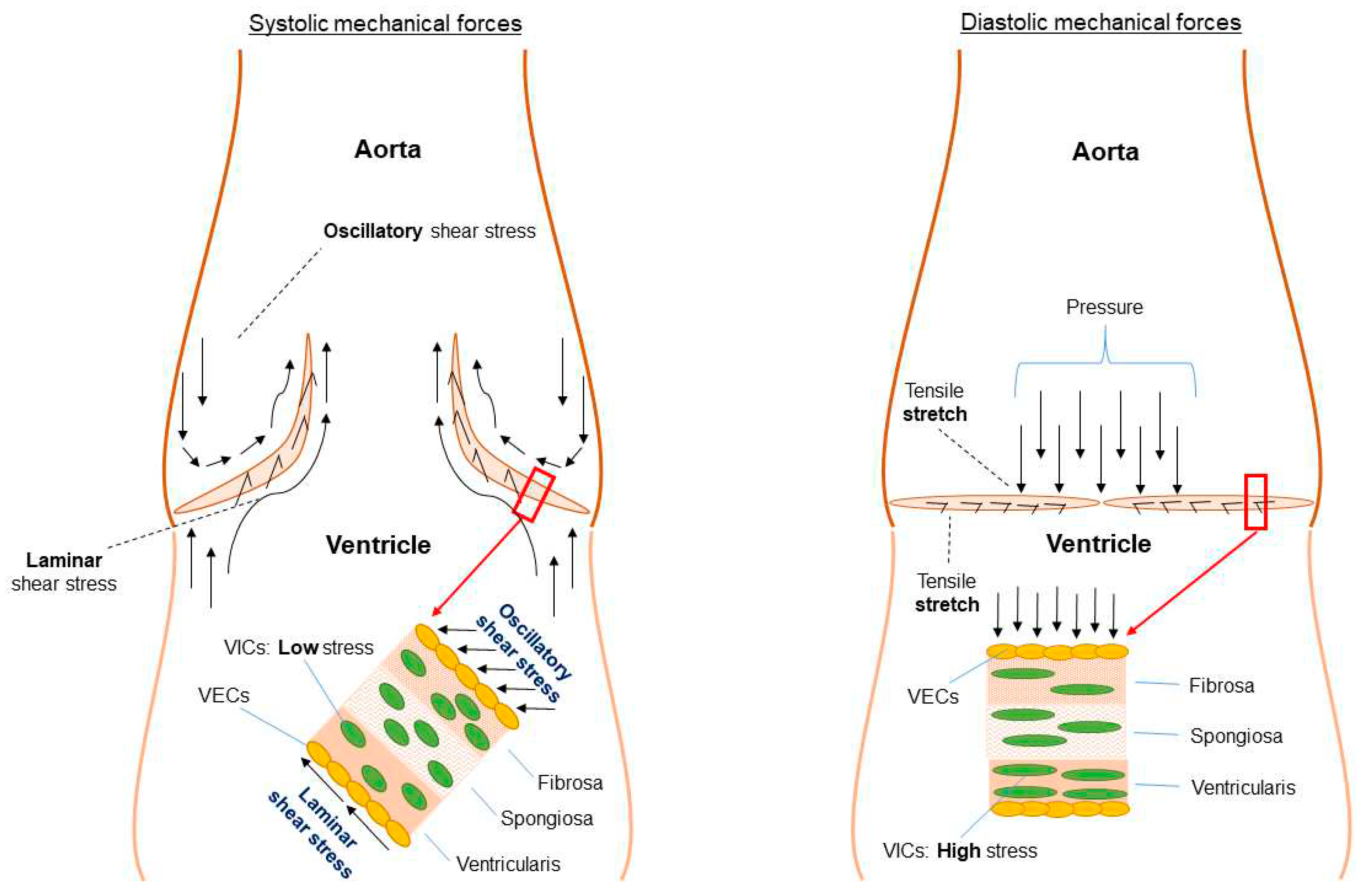
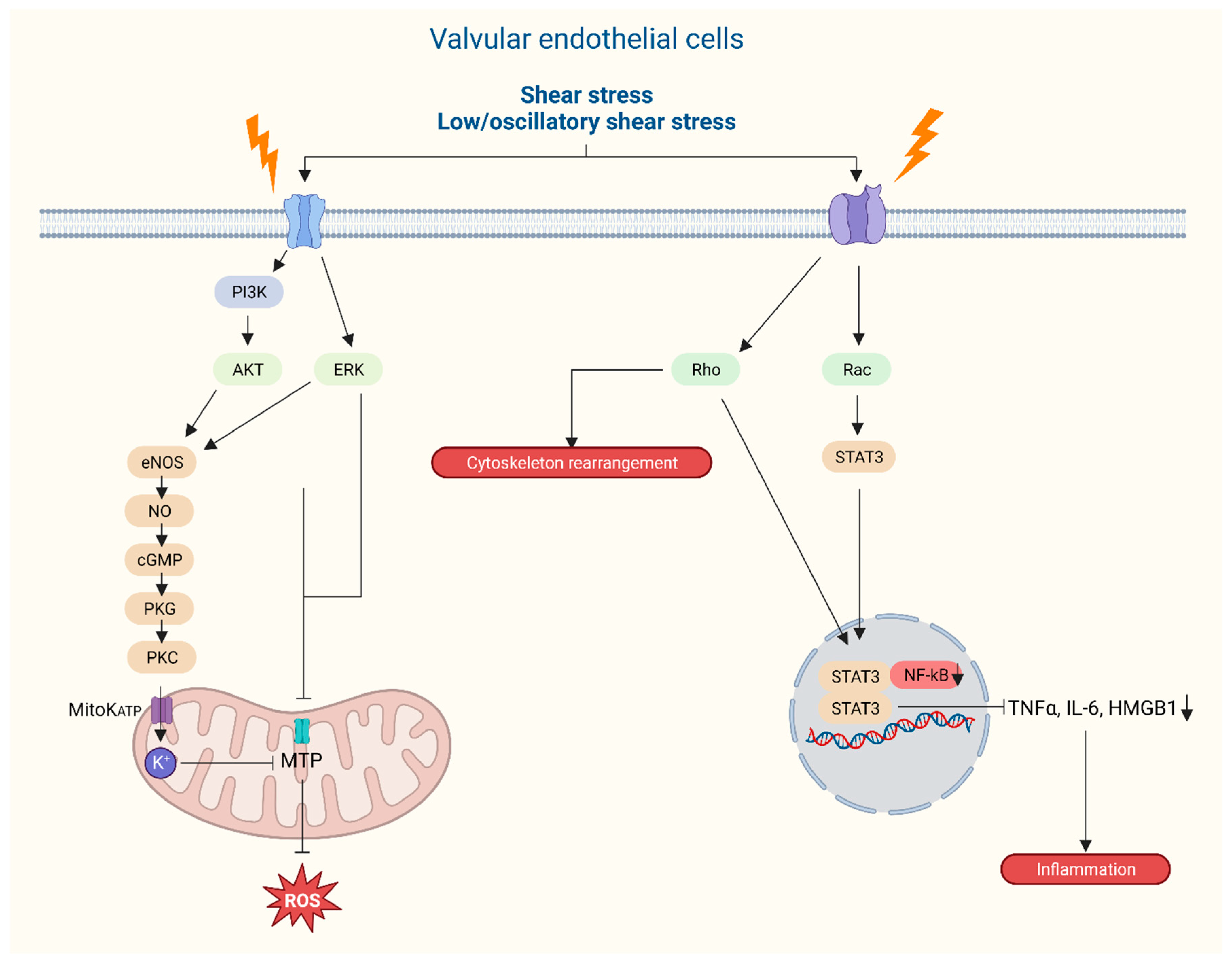
5.1. Functional morphology and mechanical stimuli
5.2. Adaptive remodeling
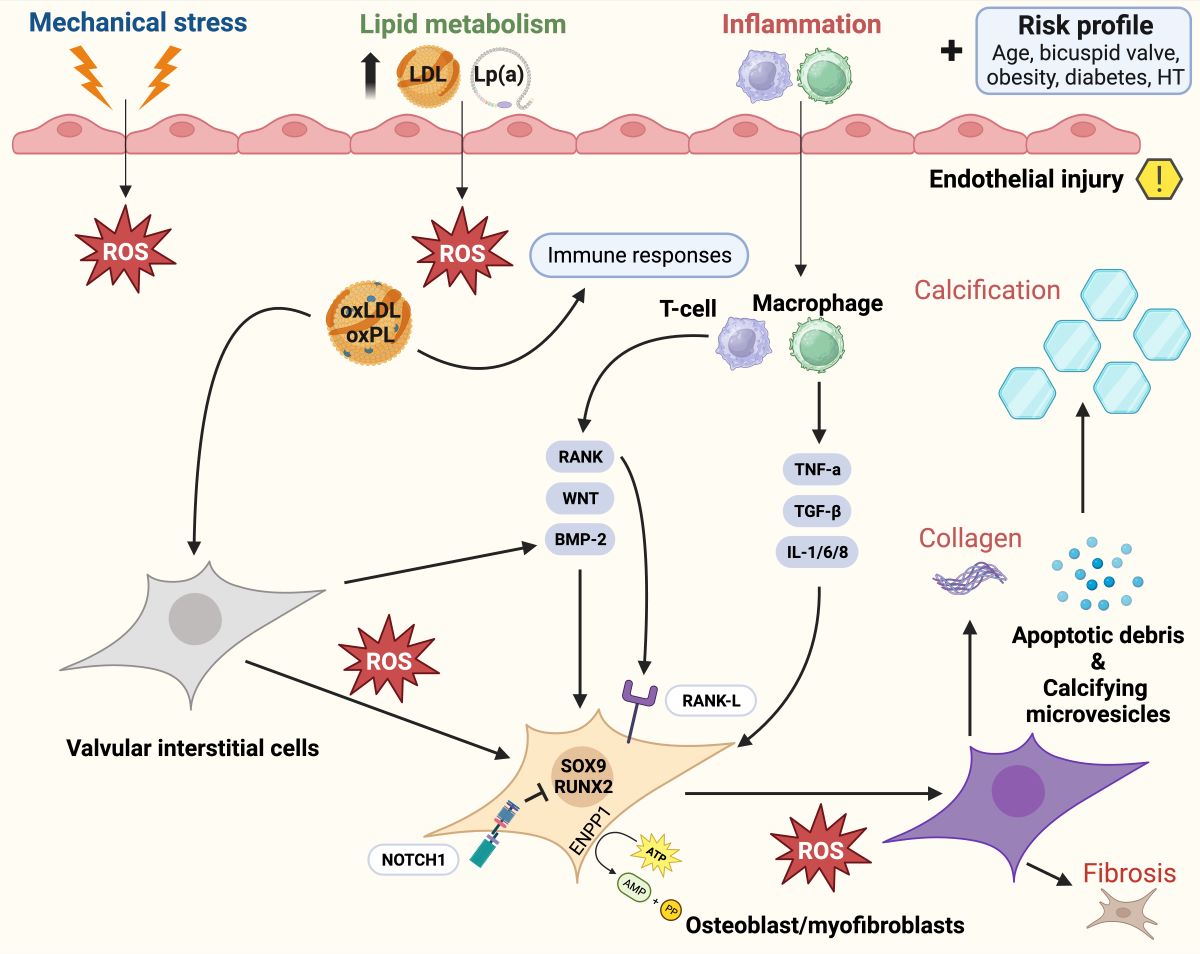
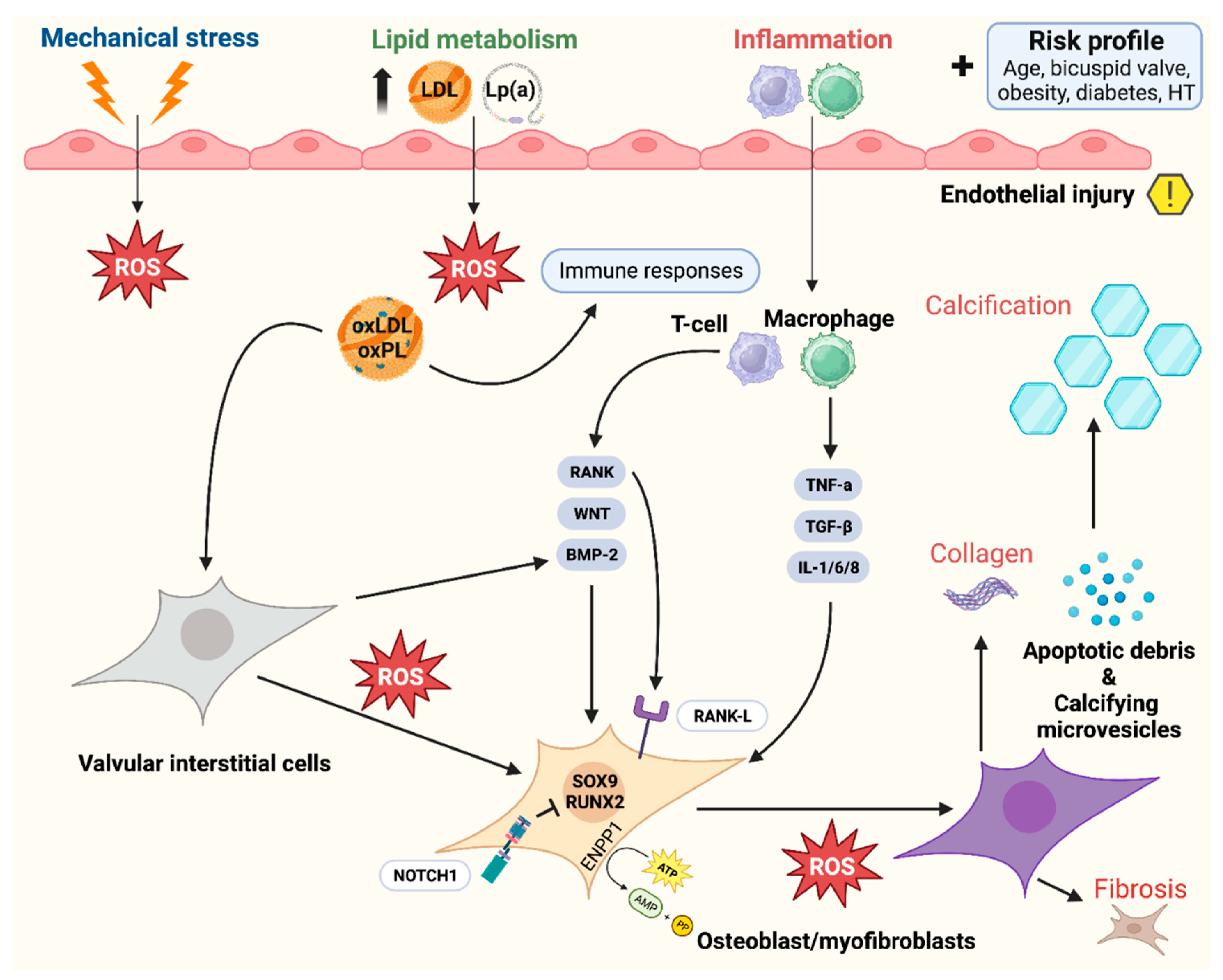
5.3. Calcific aortic valve disease
6.(. Surgical) Treatment
6.1. Evidence-based medicine and clinical decision-making
7. Conclusions and Directions
Author Contributions
Funding
Disclosures
References
- d'Arcy JL, Prendergast BD, Chambers JB, Ray SG, Bridgewater B. Valvular heart disease: the next cardiac epidemic. Heart. 2011;97(2):91-3. [CrossRef]
- Coffey S, Cox B, Williams MJ. Lack of progress in valvular heart disease in the pre-transcatheter aortic valve replacement era: increasing deaths and minimal change in mortality rate over the past three decades. Am Heart J. 2014;167(4):562-7 e2. [CrossRef]
- Rosenhek R, Zilberszac R, Schemper M, Czerny M, Mundigler G, Graf S, et al. Natural history of very severe aortic stenosis. Circulation. 2010;121(1):151-6. [CrossRef]
- Ross J, Jr., Braunwald E. Aortic stenosis. Circulation. 1968;38(1 Suppl):61-7.
- Philippe G, Rahul PS, Robert JC, Lucy A, Omar MA, Konstantinos PK, et al. The Mortality Burden of Untreated Aortic Stenosis. Journal of the American College of Cardiology. 2023;82(22):2101-9.
- Goldbarg SH, Elmariah S, Miller MA, Fuster V. Insights Into Degenerative Aortic Valve Disease. Journal of the American College of Cardiology. 2007;50(13):1205-13. [CrossRef]
- El-Hamamsy I, Chester AH, Yacoub MH. Cellular regulation of the structure and function of aortic valves. Journal of Advanced Research. 2010;1(1):5-12. [CrossRef]
- El-Hamamsy I, Yacoub MH, Chester AH. Neuronal regulation of aortic valve cusps. Curr Vasc Pharmacol. 2009;7(1):40-6. [CrossRef]
- El-Hamamsy I, Balachandran K, Yacoub MH, Stevens LM, Sarathchandra P, Taylor PM, et al. Endothelium-dependent regulation of the mechanical properties of aortic valve cusps. J Am Coll Cardiol. 2009;53(16):1448-55. [CrossRef]
- Boodhwani M, de Kerchove L, Glineur D, Poncelet A, Rubay J, Astarci P, et al. Repair-oriented classification of aortic insufficiency: impact on surgical techniques and clinical outcomes. J Thorac Cardiovasc Surg. 2009;137(2):286-94. [CrossRef]
- Misfeld M, Chester AH, Sievers HH, Yacoub MH. Biological mechanisms influencing the function of the aortic root. J Card Surg. 2002;17(4):363-8. [CrossRef]
- Ross, DN. Replacement of aortic and mitral valves with a pulmonary autograft. Lancet. 1967;2(7523):956-8. [CrossRef]
- Lansac E, de Kerchove L. Aortic valve repair techniques: state of the art. Eur J Cardiothorac Surg. 2018;53(6):1101-7.
- Combs MD, Yutzey KE. Heart valve development: regulatory networks in development and disease. Circ Res. 2009;105(5):408-21.
- Gittenberger-de Groot AC, Bartelings MM, Deruiter MC, Poelmann RE. Basics of cardiac development for the understanding of congenital heart malformations. Pediatr Res. 2005;57(2):169-76.
- Moorman AF, Christoffels VM. Cardiac chamber formation: development, genes, and evolution. Physiol Rev. 2003;83(4):1223-67. [CrossRef]
- Lamers WH, Moorman AF. Cardiac septation: a late contribution of the embryonic primary myocardium to heart morphogenesis. Circ Res. 2002;91(2):93-103.
- Soufan AT, van den Hoff MJ, Ruijter JM, de Boer PA, Hagoort J, Webb S, et al. Reconstruction of the patterns of gene expression in the developing mouse heart reveals an architectural arrangement that facilitates the understanding of atrial malformations and arrhythmias. Circ Res. 2004;95(12):1207-15. [CrossRef]
- Schroeder JA, Jackson LF, Lee DC, Camenisch TD. Form and function of developing heart valves: coordination by extracellular matrix and growth factor signaling. J Mol Med (Berl). 2003;81(7):392-403. [CrossRef]
- Wirrig EE, Yutzey KE. Conserved transcriptional regulatory mechanisms in aortic valve development and disease. Arterioscler Thromb Vasc Biol. 2014;34(4):737-41.
- Hinton RB, Jr., Lincoln J, Deutsch GH, Osinska H, Manning PB, Benson DW, et al. Extracellular matrix remodeling and organization in developing and diseased aortic valves. Circ Res. 2006;98(11):1431-8.
- Martin PS, Kloesel B, Norris RA, Lindsay M, Milan D, Body SC. Embryonic Development of the Bicuspid Aortic Valve. Journal of Cardiovascular Development and Disease. 2015;2(4):248-72. [CrossRef]
- Rabkin-Aikawa E, Farber M, Aikawa M, Schoen FJ. Dynamic and reversible changes of interstitial cell phenotype during remodeling of cardiac valves. J Heart Valve Dis. 2004;13(5):841-7.
- de Lange FJ, Moorman AF, Anderson RH, Männer J, Soufan AT, de Gier-de Vries C, et al. Lineage and morphogenetic analysis of the cardiac valves. Circ Res. 2004;95(6):645-54. [CrossRef]
- Butcher JT, Markwald RR. Valvulogenesis: the moving target. Philos Trans R Soc Lond B Biol Sci. 2007;362(1484):1489-503.
- Gobergs R, Salputra E, Lubaua I. Hypoplastic left heart syndrome: a review. Acta Med Litu. 2016;23(2):86-98. [CrossRef]
- Rahman A, DeYoung T, Cahill LS, Yee Y, Debebe SK, Botelho O, et al. A mouse model of hypoplastic left heart syndrome demonstrating left heart hypoplasia and retrograde aortic arch flow. Dis Model Mech. 2021;14(11). [CrossRef]
- Greenberg HZE, Zhao G, Shah AM, Zhang M. Role of oxidative stress in calcific aortic valve disease and its therapeutic implications. Cardiovascular Research. 2021;118(6):1433-51. [CrossRef]
- Dayawansa NH, Baratchi S, Peter K. Uncoupling the Vicious Cycle of Mechanical Stress and Inflammation in Calcific Aortic Valve Disease. Front Cardiovasc Med. 2022;9:783543. [CrossRef]
- Peng Q, Shan D, Cui K, Li K, Zhu B, Wu H, et al. The Role of Endothelial-to-Mesenchymal Transition in Cardiovascular Disease. Cells. 2022;11(11). [CrossRef]
- Henderson DJ, Eley L, Turner JE, Chaudhry B. Development of the Human Arterial Valves: Understanding Bicuspid Aortic Valve. Front Cardiovasc Med. 2021;8:802930. [CrossRef]
- Hutson MR, Kirby ML. Model systems for the study of heart development and disease: Cardiac neural crest and conotruncal malformations. Seminars in Cell & Developmental Biology. 2007;18(1):101-10. [CrossRef]
- George RM, Maldonado-Velez G, Firulli AB. The heart of the neural crest: cardiac neural crest cells in development and regeneration. Development. 2020;147(20). [CrossRef]
- Poelmann RE, Mikawa T, Gittenberger-de Groot AC. Neural crest cells in outflow tract septation of the embryonic chicken heart: differentiation and apoptosis. Dev Dyn. 1998;212(3):373-84.
- Chakraborty S, Combs MD, Yutzey KE. Transcriptional regulation of heart valve progenitor cells. Pediatr Cardiol. 2010;31(3):414-21. [CrossRef]
- Maleki S, Poujade FA, Bergman O, Gådin JR, Simon N, Lång K, et al. Endothelial/Epithelial Mesenchymal Transition in Ascending Aortas of Patients With Bicuspid Aortic Valve. Front Cardiovasc Med. 2019;6:182. [CrossRef]
- Armstrong EJ, Bischoff J. Heart valve development: endothelial cell signaling and differentiation. Circ Res. 2004;95(5):459-70.
- Camenisch TD, Molin DG, Person A, Runyan RB, Gittenberger-de Groot AC, McDonald JA, et al. Temporal and distinct TGFbeta ligand requirements during mouse and avian endocardial cushion morphogenesis. Dev Biol. 2002;248(1):170-81.
- Ayoub S, Ferrari G, Gorman RC, Gorman JH, Schoen FJ, Sacks MS. Heart Valve Biomechanics and Underlying Mechanobiology. Compr Physiol. 2016;6(4):1743-80.
- Rochais F, Mesbah K, Kelly RG. Signaling pathways controlling second heart field development. Circ Res. 2009;104(8):933-42. [CrossRef]
- Grewal N, Gittenberger-de Groot AC, Lindeman JH, Klautz A, Driessen A, Klautz RJM, et al. Normal and abnormal development of the aortic valve and ascending aortic wall: a comprehensive overview of the embryology and pathology of the bicuspid aortic valve. Ann Cardiothorac Surg. 2022;11(4):380-8. [CrossRef]
- Harmon AW, Nakano A. Nkx2-5 lineage tracing visualizes the distribution of second heart field-derived aortic smooth muscle. Genesis. 2013;51(12):862-9. [CrossRef]
- Sawada H, Rateri DL, Moorleghen JJ, Majesky MW, Daugherty A. Smooth Muscle Cells Derived From Second Heart Field and Cardiac Neural Crest Reside in Spatially Distinct Domains in the Media of the Ascending Aorta-Brief Report. Arterioscler Thromb Vasc Biol. 2017;37(9):1722-6. [CrossRef]
- George V, Colombo S, Targoff KL. An early requirement for nkx2.5 ensures the first and second heart field ventricular identity and cardiac function into adulthood. Dev Biol. 2015;400(1):10-22. [CrossRef]
- Garside VC, Chang AC, Karsan A, Hoodless PA. Co-ordinating Notch, BMP, and TGF-β signaling during heart valve development. Cell Mol Life Sci. 2013;70(16):2899-917.
- Walker GA, Masters KS, Shah DN, Anseth KS, Leinwand LA. Valvular myofibroblast activation by transforming growth factor-beta: implications for pathological extracellular matrix remodeling in heart valve disease. Circ Res. 2004;95(3):253-60.
- Niessen K, Fu Y, Chang L, Hoodless PA, McFadden D, Karsan A. Slug is a direct Notch target required for initiation of cardiac cushion cellularization. J Cell Biol. 2008;182(2):315-25. [CrossRef]
- Loomes KM, Taichman DB, Glover CL, Williams PT, Markowitz JE, Piccoli DA, et al. Characterization of Notch receptor expression in the developing mammalian heart and liver. Am J Med Genet. 2002;112(2):181-9. [CrossRef]
- Varadkar P, Kraman M, Despres D, Ma G, Lozier J, McCright B. Notch2 is required for the proliferation of cardiac neural crest-derived smooth muscle cells. Dev Dyn. 2008;237(4):1144-52. [CrossRef]
- Timmerman LA, Grego-Bessa J, Raya A, Bertrán E, Pérez-Pomares JM, Díez J, et al. Notch promotes epithelial-mesenchymal transition during cardiac development and oncogenic transformation. Genes Dev. 2004;18(1):99-115. [CrossRef]
- Garg V, Muth AN, Ransom JF, Schluterman MK, Barnes R, King IN, et al. Mutations in NOTCH1 cause aortic valve disease. Nature. 2005;437(7056):270-4. [CrossRef]
- Kamath BM, Bauer RC, Loomes KM, Chao G, Gerfen J, Hutchinson A, et al. NOTCH2 mutations in Alagille syndrome. J Med Genet. 2012;49(2):138-44.
- Fu Y, Chang A, Chang L, Niessen K, Eapen S, Setiadi A, et al. Differential regulation of transforming growth factor beta signaling pathways by Notch in human endothelial cells. J Biol Chem. 2009;284(29):19452-62.
- Kokubo H, Miyagawa-Tomita S, Tomimatsu H, Nakashima Y, Nakazawa M, Saga Y, et al. Targeted disruption of hesr2 results in atrioventricular valve anomalies that lead to heart dysfunction. Circ Res. 2004;95(5):540-7.
- Cripe L, Andelfinger G, Martin LJ, Shooner K, Benson DW. Bicuspid aortic valve is heritable. J Am Coll Cardiol. 2004;44(1):138-43. [CrossRef]
- Tessler I, Albuisson J, Piñeiro-Sabarís R, Verstraeten A, Kamber Kaya HE, Siguero-Álvarez M, et al. Novel Association of the NOTCH Pathway Regulator MIB1 Gene With the Development of Bicuspid Aortic Valve. JAMA Cardiology. 2023;8(8):721-31. [CrossRef]
- MacGrogan D, D'Amato G, Travisano S, Martinez-Poveda B, Luxán G, Del Monte-Nieto G, et al. Sequential Ligand-Dependent Notch Signaling Activation Regulates Valve Primordium Formation and Morphogenesis. Circ Res. 2016;118(10):1480-97. [CrossRef]
- Small AM, Peloso GM, Linefsky J, Aragam J, Galloway A, Tanukonda V, et al. Multiancestry Genome-Wide Association Study of Aortic Stenosis Identifies Multiple Novel Loci in the Million Veteran Program. Circulation. 2023;147(12):942-55. [CrossRef]
- Gehlen J, Stundl A, Debiec R, Fontana F, Krane M, Sharipova D, et al. Elucidation of the genetic causes of bicuspid aortic valve disease. Cardiovascular Research. 2022;119(3):857-66. [CrossRef]
- Anderson, RH. Clinical anatomy of the aortic root. Heart. 2000;84(6):670-3.
- Anderson, RH. The surgical anatomy of the aortic root. Multimed Man Cardiothorac Surg. 2007;2007(102):mmcts 2006 002527. [CrossRef]
- Yacoub MH, Kilner PJ, Birks EJ, Misfeld M. The aortic outflow and root: a tale of dynamism and crosstalk. Ann Thorac Surg. 1999;68(3 Suppl):S37-43. [CrossRef]
- Sarsam MA, Yacoub M. Remodeling of the aortic valve anulus. J Thorac Cardiovasc Surg. 1993;105(3):435-8. [CrossRef]
- David TE, Feindel CM. An aortic valve-sparing operation for patients with aortic incompetence and aneurysm of the ascending aorta. J Thorac Cardiovasc Surg. 1992;103(4):617-21; discussion 22.
- Chester AH, El-Hamamsy I, Butcher JT, Latif N, Bertazzo S, Yacoub MH. The living aortic valve: From molecules to function. Glob Cardiol Sci Pract. 2014;2014(1):52-77. [CrossRef]
- Rajamannan, NM. Bicuspid aortic valve disease: the role of oxidative stress in Lrp5 bone formation. Cardiovasc Pathol. 2011;20(3):168-76. [CrossRef]
- Fries R, Graeter T, Aicher D, Reul H, Schmitz C, Böhm M, et al. In vitro comparison of aortic valve movement after valve-preserving aortic replacement. The Journal of Thoracic and Cardiovascular Surgery. 2006;132(1):32-7. [CrossRef]
- Arjunon S, Rathan S, Jo H, Yoganathan AP. Aortic valve: mechanical environment and mechanobiology. Ann Biomed Eng. 2013;41(7):1331-46. [CrossRef]
- Bellhouse BJ, Talbot L. The fluid mechanics of the aortic valve. Journal of Fluid Mechanics. 1969;35:721 - 35. [CrossRef]
- Campinho P, Vilfan A, Vermot J. Blood Flow Forces in Shaping the Vascular System: A Focus on Endothelial Cell Behavior. Front Physiol. 2020;11:552. [CrossRef]
- Yacoub MH, Aguib H, Gamrah MA, Shehata N, Nagy M, Donia M, et al. Aortic root dynamism, geometry, and function after the remodeling operation: Clinical relevance. J Thorac Cardiovasc Surg. 2018;156(3):951-62 e2. [CrossRef]
- Bellhouse B, Bellhouse F. Fluid mechanics of model normal and stenosed aortic valves. Circ Res. 1969;25(6):693-704.
- Cheng A, Dagum P, Miller DC. Aortic root dynamics and surgery: from craft to science. Philos Trans R Soc Lond B Biol Sci. 2007;362(1484):1407-19. [CrossRef]
- Bruno RM, Climie R, Gallo A. Aortic pulsatility drives microvascular organ damage in essential hypertension: New evidence from choroidal thickness assessment. J Clin Hypertens (Greenwich). 2021;23(5):1039-40. [CrossRef]
- Um KJ, McClure GR, Belley-Cote EP, Gupta S, Bouhout I, Lortie H, et al. Hemodynamic outcomes of the Ross procedure versus other aortic valve replacement: a systematic review and meta-analysis. J Cardiovasc Surg (Torino). 2018;59(3):462-70. [CrossRef]
- Stewart S, Afoakwah C, Chan Y-K, Strom JB, Playford D, Strange GA. Counting the cost of premature mortality with progressively worse aortic stenosis in Australia: a clinical cohort study. The Lancet Healthy Longevity. 2022;3(9):e599-e606. [CrossRef]
- Harris P, Kuppurao L. Quantitative Doppler echocardiography. BJA Education. 2015;16(2):46-52. [CrossRef]
- Chan V, Rubens F, Boodhwani M, Mesana T, Ruel M. Determinants of persistent or recurrent congestive heart failure after contemporary surgical aortic valve replacement. J Heart Valve Dis. 2014;23(6):665-70.
- Duebener LF, Stierle U, Erasmi A, Bechtel MF, Zurakowski D, Böhm JO, et al. Ross procedure and left ventricular mass regression. Circulation. 2005;112(9 Suppl):I415-22.
- Hauser M, Bengel FM, Kühn A, Sauer U, Zylla S, Braun SL, et al. Myocardial blood flow and flow reserve after coronary reimplantation in patients after arterial switch and ross operation. Circulation. 2001;103(14):1875-80. [CrossRef]
- Gaudino M, Piatti F, Lau C, Sturla F, Weinsaft JW, Weltert L, et al. Aortic flow after valve sparing root replacement with or without neosinuses reconstruction. J Thorac Cardiovasc Surg. 2019;157(2):455-65. [CrossRef]
- Balachandran K, Sucosky P, Yoganathan AP. Hemodynamics and mechanobiology of aortic valve inflammation and calcification. Int J Inflam. 2011;2011:263870. [CrossRef]
- Rego BV, Sacks MS. A functionally graded material model for the transmural stress distribution of the aortic valve leaflet. J Biomech. 2017;54:88-95. [CrossRef]
- Kodigepalli KM, Thatcher K, West T, Howsmon DP, Schoen FJ, Sacks MS, et al. Biology and Biomechanics of the Heart Valve Extracellular Matrix. J Cardiovasc Dev Dis. 2020;7(4). [CrossRef]
- Butcher JT, Penrod AM, García AJ, Nerem RM. Unique morphology and focal adhesion development of valvular endothelial cells in static and fluid flow environments. Arterioscler Thromb Vasc Biol. 2004;24(8):1429-34. [CrossRef]
- Bäck M, Gasser TC, Michel J-B, Caligiuri G. Biomechanical factors in the biology of aortic wall and aortic valve diseases. Cardiovascular Research. 2013;99(2):232-41. [CrossRef]
- Farivar RS, Cohn LH, Soltesz EG, Mihaljevic T, Rawn JD, Byrne JG. Transcriptional profiling and growth kinetics of endothelium reveals differences between cells derived from porcine aorta versus aortic valve. Eur J Cardiothorac Surg. 2003;24(4):527-34. [CrossRef]
- Simmons CA, Grant GR, Manduchi E, Davies PF. Spatial heterogeneity of endothelial phenotypes correlates with side-specific vulnerability to calcification in normal porcine aortic valves. Circ Res. 2005;96(7):792-9. [CrossRef]
- Butcher JT, Tressel S, Johnson T, Turner D, Sorescu G, Jo H, et al. Transcriptional profiles of valvular and vascular endothelial cells reveal phenotypic differences: influence of shear stress. Arterioscler Thromb Vasc Biol. 2006;26(1):69-77.
- Yap CH, Saikrishnan N, Yoganathan AP. Experimental measurement of dynamic fluid shear stress on the ventricular surface of the aortic valve leaflet. Biomech Model Mechanobiol. 2012;11(1-2):231-44. [CrossRef]
- Yu Chen H, Dina C, Small AM, Shaffer CM, Levinson RT, Helgadóttir A, et al. Dyslipidemia, inflammation, calcification, and adiposity in aortic stenosis: a genome-wide study. European Heart Journal. 2023;44(21):1927-39. [CrossRef]
- Scott AJ, Simon LR, Hutson HN, Porras AM, Masters KS. Engineering the aortic valve extracellular matrix through stages of development, aging, and disease. J Mol Cell Cardiol. 2021;161:1-8. [CrossRef]
- El-Hamamsy I, Yacoub MH. Cellular and molecular mechanisms of thoracic aortic aneurysms. Nat Rev Cardiol. 2009;6(12):771-86.
- Leopold, JA. Cellular mechanisms of aortic valve calcification. Circ Cardiovasc Interv. 2012;5(4):605-14. [CrossRef]
- Chen JH, Simmons CA. Cell-matrix interactions in the pathobiology of calcific aortic valve disease: critical roles for matricellular, matricrine, and matrix mechanics cues. Circ Res. 2011;108(12):1510-24.
- Yip CY, Chen JH, Zhao R, Simmons CA. Calcification by valve interstitial cells is regulated by the stiffness of the extracellular matrix. Arterioscler Thromb Vasc Biol. 2009;29(6):936-42. [CrossRef]
- Notenboom ML, Schuermans A, Etnel JRG, Veen KM, van de Woestijne PC, Rega FR, et al. Paediatric aortic valve replacement: a meta-analysis and microsimulation study. Eur Heart J. 2023. [CrossRef]
- Schoof PH, Cromme-Dijkhuis AH, Bogers JJ, Thijssen EJ, Witsenburg M, Hess J, et al. Aortic root replacement with pulmonary autograft in children. J Thorac Cardiovasc Surg. 1994;107(2):367-73. [CrossRef]
- Van Hoof L, Verbrugghe P, Jones EAV, Humphrey JD, Janssens S, Famaey N, et al. Understanding Pulmonary Autograft Remodeling After the Ross Procedure: Stick to the Facts. Front Cardiovasc Med. 2022;9:829120. [CrossRef]
- Rabkin-Aikawa E, Aikawa M, Farber M, Kratz JR, Garcia-Cardena G, Kouchoukos NT, et al. Clinical pulmonary autograft valves: pathologic evidence of adaptive remodeling in the aortic site. J Thorac Cardiovasc Surg. 2004;128(4):552-61. [CrossRef]
- Mazine A, El-Hamamsy I, Verma S, Peterson MD, Bonow RO, Yacoub MH, et al. Ross Procedure in Adults for Cardiologists and Cardiac Surgeons: JACC State-of-the-Art Review. J Am Coll Cardiol. 2018;72(22):2761-77.
- Gorczynski A, Trenkner M, Anisimowicz L, Gutkowski R, Drapella A, Kwiatkowska E, et al. Biomechanics of the pulmonary autograft valve in the aortic position. Thorax. 1982;37(7):535-9. [CrossRef]
- Mazine A, El-Hamamsy I. The Ross procedure is an excellent operation in non-repairable aortic regurgitation: insights and techniques. Ann Cardiothorac Surg. 2021;10(4):463-75. [CrossRef]
- Tanaka D, Mazine A, Ouzounian M, El-Hamamsy I. Supporting the Ross procedure: preserving root physiology while mitigating autograft dilatation. Curr Opin Cardiol. 2022;37(2):180-90. [CrossRef]
- Ismail E-H, Patrick TOG, David HA. The Ross Procedure. Journal of the American College of Cardiology. 2022;79(10):1006-9.
- Notenboom ML, Melina G, Veen K, De Robertis F, Coppola G, De Siena P, et al. Long-Term Clinical and Echocardiographic Outcomes Following the Ross Procedure: A Post Hoc Analysis of a Randomized Controlled Trial. JAMA Cardiol. 2023;In press.
- Latif N, Mahgoub A, Nagy M, Sarathchandra P, Yacoub MH. Severe degeneration of a sub-coronary pulmonary autograft in a young adult. Glob Cardiol Sci Pract. 2021;2021(2):e202114. [CrossRef]
- Thanassoulis G, Campbell CY, Owens DS, Smith JG, Smith AV, Peloso GM, et al. Genetic Associations with Valvular Calcification and Aortic Stenosis. New England Journal of Medicine. 2013;368(6):503-12. [CrossRef]
- Torzewski M, Ravandi A, Yeang C, Edel A, Bhindi R, Kath S, et al. Lipoprotein(a) Associated Molecules are Prominent Components in Plasma and Valve Leaflets in Calcific Aortic Valve Stenosis. JACC Basic Transl Sci. 2017;2(3):229-40. [CrossRef]
- Capoulade R, Chan KL, Yeang C, Mathieu P, Bossé Y, Dumesnil JG, et al. Oxidized Phospholipids, Lipoprotein(a), and Progression of Calcific Aortic Valve Stenosis. Journal of the American College of Cardiology. 2015;66(11):1236-46. [CrossRef]
- Moncla LM, Briend M, Bossé Y, Mathieu P. Calcific aortic valve disease: mechanisms, prevention and treatment. Nat Rev Cardiol. 2023;20(8):546-59. [CrossRef]
- Khosla S, Eghbali-Fatourechi GZ. Circulating cells with osteogenic potential. Ann N Y Acad Sci. 2006;1068:489-97. [CrossRef]
- Egan KP, Kim JH, Mohler ER, 3rd, Pignolo RJ. Role for circulating osteogenic precursor cells in aortic valvular disease. Arterioscler Thromb Vasc Biol. 2011;31(12):2965-71.
- Eghbali-Fatourechi GZ, Lamsam J, Fraser D, Nagel D, Riggs BL, Khosla S. Circulating osteoblast-lineage cells in humans. N Engl J Med. 2005;352(19):1959-66. [CrossRef]
- Goel SS, Kleiman NS, Zoghbi WA, Reardon MJ, Kapadia SR. Renin-Angiotensin System Blockade in Aortic Stenosis: Implications Before and After Aortic Valve Replacement. J Am Heart Assoc. 2020;9(18):e016911. [CrossRef]
- Gollmann-Tepeköylü C, Graber M, Hirsch J, Mair S, Naschberger A, Pölzl L, et al. Toll-Like Receptor 3 Mediates Aortic Stenosis Through a Conserved Mechanism of Calcification. Circulation. 2023. [CrossRef]
- Mokhles S, Takkenberg JJM, Treasure T. Evidence-Based and Personalized Medicine. It’s [AND] not [OR]. The Annals of Thoracic Surgery. 2017;103(1):351-60.
- David LS, William MCR, Gray JAM, Haynes RB, Richardson WS. Evidence based medicine: what it is and what it isn't. Bmj. 1996;312(7023):71-2. [CrossRef]
- Etnel JRG, Huygens SA, Grashuis P, Pekbay B, Papageorgiou G, Roos Hesselink JW, et al. Bioprosthetic Aortic Valve Replacement in Nonelderly Adults: A Systematic Review, Meta-Analysis, Microsimulation. Circ Cardiovasc Qual Outcomes. 2019;12(2):e005481.
- Goldstone AB, Chiu P, Baiocchi M, Lingala B, Patrick WL, Fischbein MP, et al. Mechanical or Biologic Prostheses for Aortic-Valve and Mitral-Valve Replacement. N Engl J Med. 2017;377(19):1847-57. [CrossRef]
- Korteland NM, Etnel JRG, Arabkhani B, Mokhles MM, Mohamad A, Roos-Hesselink JW, et al. Mechanical aortic valve replacement in non-elderly adults: meta-analysis and microsimulation. Eur Heart J. 2017;38(45):3370-7. [CrossRef]
- El-Hamamsy I, Eryigit Z, Stevens LM, Sarang Z, George R, Clark L, et al. Long-term outcomes after autograft versus homograft aortic root replacement in adults with aortic valve disease: a randomised controlled trial. Lancet. 2010;376(9740):524-31. [CrossRef]
- Yacoub M, Rasmi NR, Sundt TM, Lund O, Boyland E, Radley-Smith R, et al. Fourteen-year experience with homovital homografts for aortic valve replacement. J Thorac Cardiovasc Surg. 1995;110(1):186-93; discussion 93-4. [CrossRef]
- Klieverik LM, Takkenberg JJ, Bekkers JA, Roos-Hesselink JW, Witsenburg M, Bogers AJ. The Ross operation: a Trojan horse? Eur Heart J. 2007;28(16):1993-2000.
- Reece TB, Welke KF, O'Brien S, Grau-Sepulveda MV, Grover FL, Gammie JS. Rethinking the ross procedure in adults. Ann Thorac Surg. 2014;97(1):175-81. [CrossRef]
- El-Hamamsy I, Toyoda N, Itagaki S, Stelzer P, Varghese R, Williams EE, et al. Propensity-Matched Comparison of the Ross Procedure and Prosthetic Aortic Valve Replacement in Adults. J Am Coll Cardiol. 2022;79(8):805-15. [CrossRef]
- Yacoub MH, Notenboom ML, Melina G, Takkenberg JJM. Surgical Heritage: You Had to be There, Ross: The Comeback Kid. Seminars in Thoracic and Cardiovascular Surgery: Pediatric Cardiac Surgery Annual. 2023.
- Tom T, Asif H, Magdi Y. Is there a risk in avoiding risk for younger patients with aortic valve disease? Bmj. 2011;342:d2466.
- Fioretta ES, Motta SE, Lintas V, Loerakker S, Parker KK, Baaijens FPT, et al. Next-generation tissue-engineered heart valves with repair, remodelling and regeneration capacity. Nat Rev Cardiol. 2021;18(2):92-116. [CrossRef]
- Huygens SA, Rutten-van Mölken M, Noruzi A, Etnel JRG, Corro Ramos I, Bouten CVC, et al. What Is the Potential of Tissue-Engineered Pulmonary Valves in Children? Ann Thorac Surg. 2019;107(6):1845-53.
- van Haaften EE, Bouten CVC, Kurniawan NA. Vascular Mechanobiology: Towards Control of In Situ Regeneration. Cells. 2017;6(3).
- Wissing TB, Bonito V, Bouten CVC, Smits A. Biomaterial-driven in situ cardiovascular tissue engineering-a multi-disciplinary perspective. NPJ Regen Med. 2017;2:18. [CrossRef]
- Yacoub MH, Tseng YT, Kluin J, Vis A, Stock U, Smail H, et al. Valvulogenesis of a living, innervated pulmonary root induced by an acellular scaffold. Commun Biol. 2023;6(1):1017. [CrossRef]
- Meccanici F, Notenboom ML, Meijssen J, Smit V, van de Woestijne PC, van den Bosch AE, et al. Long-term surgical outcomes of congenital supravalvular aortic stenosis: a systematic review, meta-analysis and microsimulation study. Eur J Cardiothorac Surg. 2023. [CrossRef]
- Notenboom ML, Rhellab R, Etnel JRG, van den Bogerd N, Veen KM, Taverne Y, et al. Aortic Valve Repair in Neonates, Infants and Children: A Systematic Review, Meta-Analysis and Microsimulation Study. Eur J Cardiothorac Surg. 2023. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



