Submitted:
04 January 2024
Posted:
05 January 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Methodology
Materials
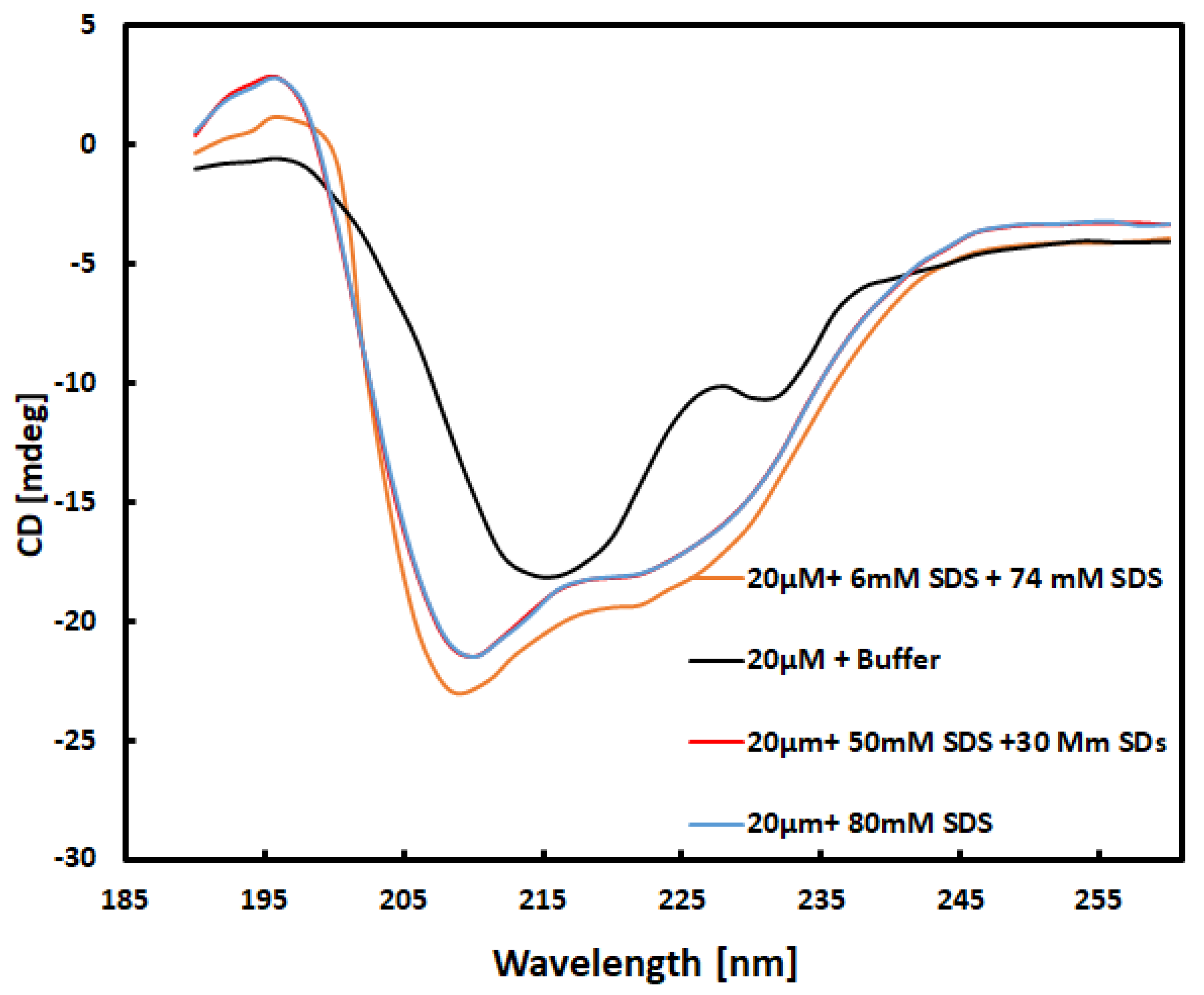
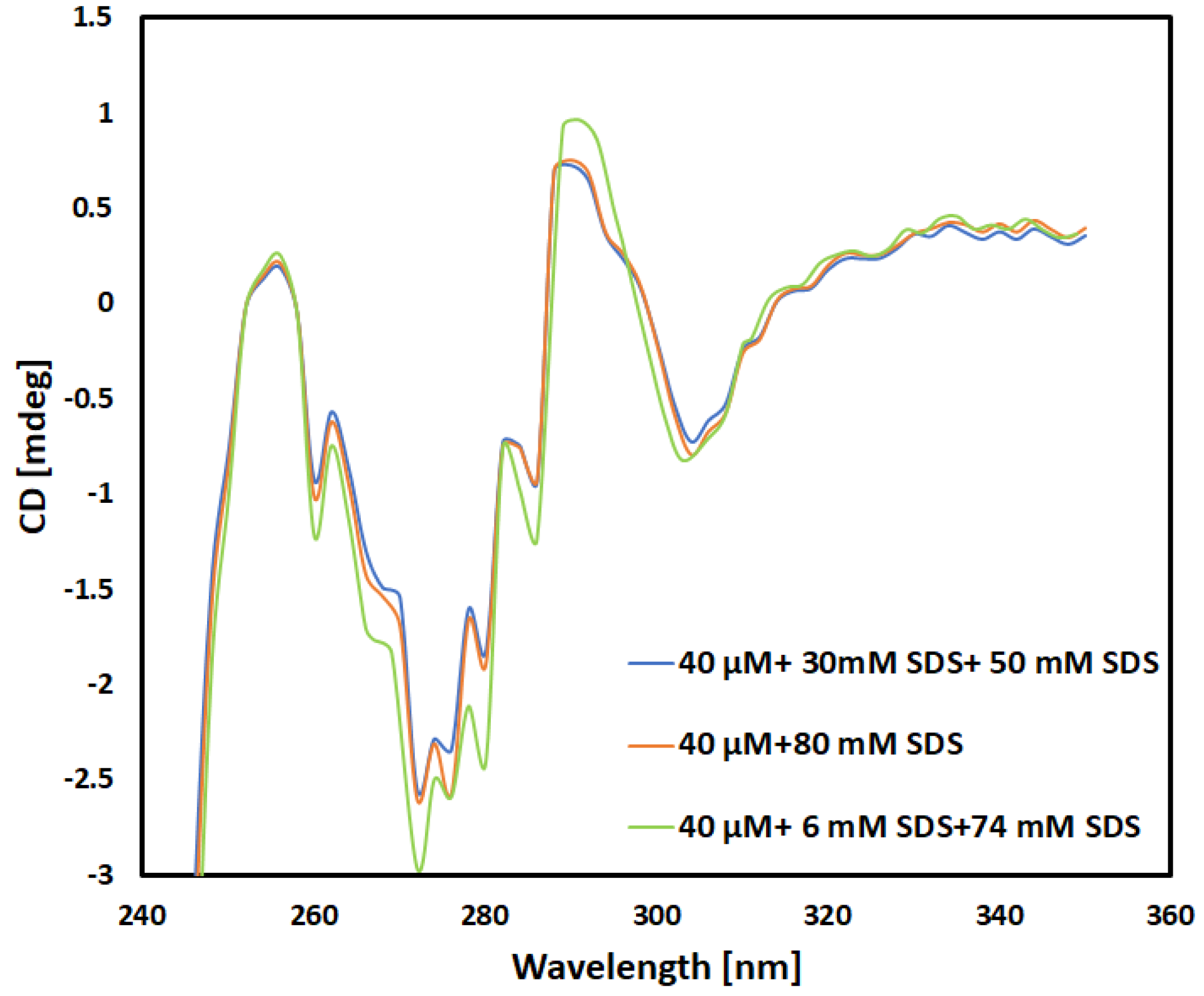
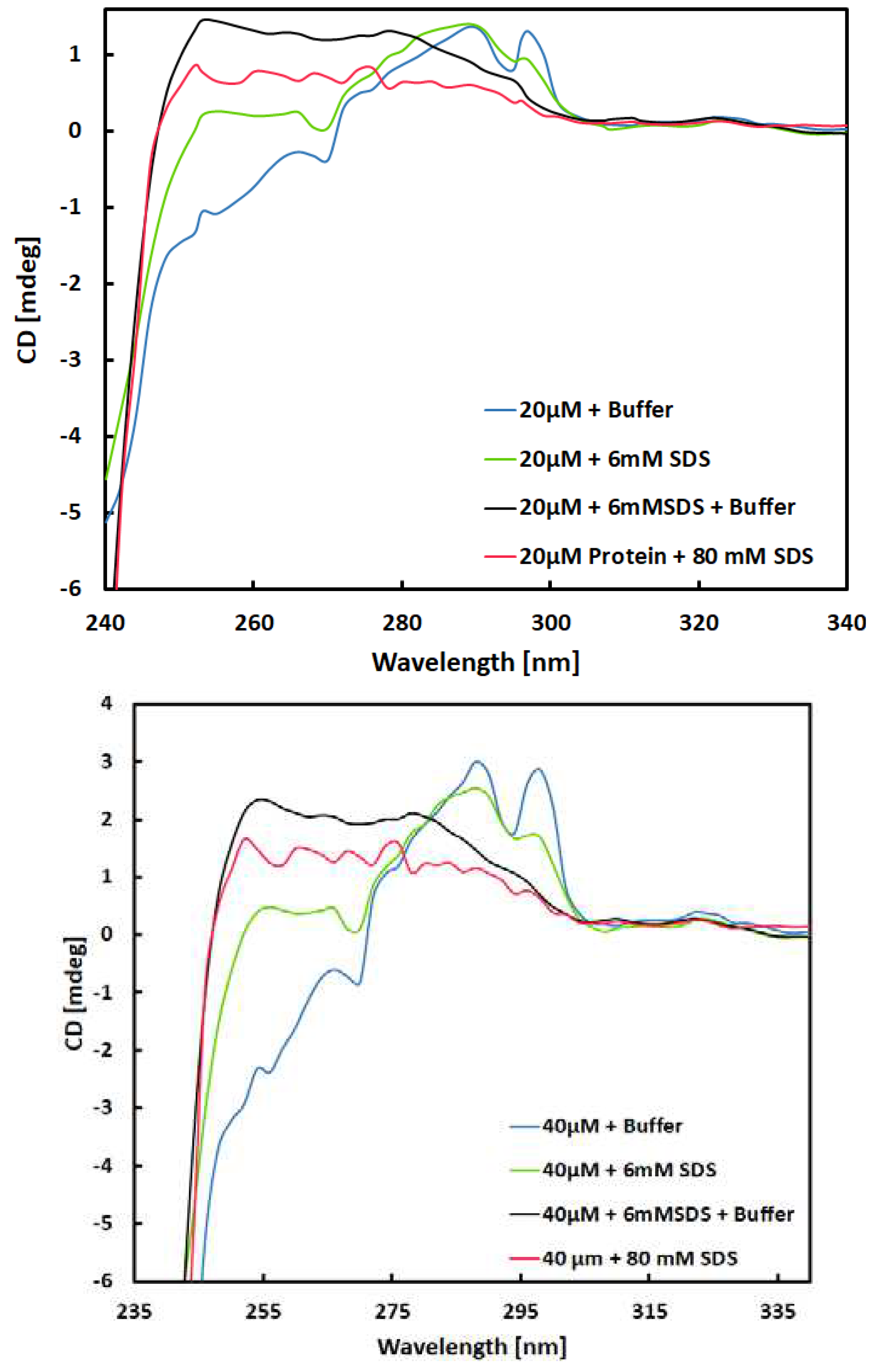
Circular Dichroism (CD) Spectra Collection
Far UV Spectra of Each Sample
Near UV-Spectra
Conclusions
Patents
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Song, J.G.; Lee, S.H.; Han, H.K. The stabilization of biopharmaceuticals: current understanding and future perspectives. Journal of Pharmaceutical Investigation 2017, 47, 475–496. [Google Scholar] [CrossRef]
- Zhu, M.M.; Mollet, M.; Hubert, R.S.; Kyung, Y.S.; Zhang, G.G. Industrial production of therapeutic proteins: cell lines, cell culture, and purification. Handbook of industrial chemistry and biotechnology 2017, 1639–1669. [Google Scholar]
- Agarkhed, M.; O’Dell, C.; Hsieh, M.C.; Zhang, J.; Goldstein, J.; Srivastava, A. Effect of polysorbate 80 concentration on thermal and photostability of a monoclonal antibody. AAPS PharmSciTech 2013, 14, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Komander, D. The emerging complexity of protein ubiquitination. Biochemical Society Transactions 2009, 37(5), 937–953. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.S.; Beauvais, R.M.; Arakawa, T.; Narthi, L.O.; Dong, A.; Aparisio, D.I.; Carpenter, J.F. Formation of an active dimer during storage of interleukin-1 receptor antagonist in aqueous solution. Biophys J 1996, 71, 3399–3406. [Google Scholar] [CrossRef] [PubMed]
- Kelly, S.M.; Jess, T.J.; Price, N.C. How to study proteins by circular dichroism. Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics 2005, 1751(2), 119–139. [Google Scholar] [CrossRef]
- Otzen, D.E.; Pedersen, J.N.; Rasmussen H, Ø.; Pedersen, J.S. How do surfactants unfold and refold proteins? Advances in Colloid and Interface Science 2022, 308, 102754. [Google Scholar] [CrossRef]
- Takeda, K.; Moriyama, Y. Kinetic Aspects of Surfactant-Induced Structural Changes of Proteins - Unsolved Problems of Two-State Model for Protein Denaturation. J. Oleo Sci. 2015, 64, 1143–1158. [Google Scholar] [CrossRef]
- Pedersen, J.N.; Lyngsø, J.; Zinn, T.; Otzen, D.E.; Pedersen, J.S. A Complete Picture of Protein Unfolding and Refolding in Surfactants. Chem. Sci. 2020, 11, 699–712. [Google Scholar] [CrossRef]
- Moosavi-Movahedi, A.A. Thermodynamics of protein denaturation by sodium dodecyl sulfate. Journal of the Iranian Chemical Society 2005, 2, 189–196. [Google Scholar] [CrossRef]
- Anand, U.; Mukherjee, S. Reversibility in protein folding: effect of β-cyclodextrin on bovine serum albumin unfolded by sodium dodecyl sulphate. Physical Chemistry Chemical Physics 2013, 15(23), 9375–9383. [Google Scholar] [CrossRef]
- Bhuyan, A.K. On the mechanism of SDS-induced protein denaturation. Biopolymers: Original Research on Biomolecules 2010, 93(2), 186–199. [Google Scholar] [CrossRef]
- Takeda, K.A. Kinetic Study of the Interaction of Sodium Dodecyl Sulfate with Bovine Serum Albumin by Means of a Pressure-jump Method. Bull. Chem. Soc. Jpn. 1982, 55, 2547–2550. [Google Scholar] [CrossRef]
- Sasa, K.; Takeda, K. Multiple Coexisting Species of Sodium Dodecyl Sulfate-Bovine Serum Albumin Complexes as Detected by Capilary Electrophoresis. J. Colloid Interface Sci. 1993, 157, 516–517. [Google Scholar] [CrossRef]
- Anand, U.; Mukherjee, S. Binding, unfolding and refolding dynamics of serum albumins. Biochimica et Biophysica Acta (BBA)-General Subjects 2013, 1830(12), 5394–5404. [Google Scholar] [CrossRef]
- Whitmore, L.; Woollett, B.; Miles, A.J.; Klose, D.P.; Janes, R.W.; Wallace, B.A. PCDDB: the protein circular dichroism data bank, a repository for circular dichroism spectral and metadata. Nucleic acids research 2011, 39 (Suppl. S1), D480–D486. [Google Scholar] [CrossRef]
- Rinner, U.; Carreira, E.M.; Yamamoto, H. Comprehensive Chirality. 2012. [Google Scholar]
- Stachurska, K.; Marcisz, U.; Długosz, M.; Antosiewicz, J.M. Circular Dichroism Spectra of α-Chymotrypsin–SDS Solutions Depend on the Procedure of Their Preparation. ACS omega 2022, 7(27), 23782–23789. [Google Scholar] [CrossRef]
- Micsonai, A.; Wien, F.; Kernya, L.; Lee, Y.H.; Goto, Y.; Réfrégiers, M.; Kardos, J. Accurate secondary structure prediction and fold recognition for circular dichroism spectroscopy. Proceedings of the National Academy of Sciences 2015, 112(24), E3095–E3103. [Google Scholar] [CrossRef]
- Chalikian, T.V.; Völker, J.; Anafi, D.; Breslauer, K.J. The native and the heat-induced denatured states of α-chymotrypsinogen A: thermodynamic and spectroscopic studies. Journal of molecular biology 1997, 274(2), 237–252. [Google Scholar] [CrossRef]
- Li, Z.; Hirst, J.D. Quantitative first principles calculations of protein circular dichroism in the near-ultraviolet. Chemical science 2017, 8(6), 4318–4333. [Google Scholar] [CrossRef]
- Kaur, H.; Beckman, J.; Zhang, Y.; Li, Z.J.; Szigeti, M.; Guttman, A. Capillary electrophoresis and the biopharmaceutical industry: Therapeutic protein analysis and characterization. TrAC Trends in Analytical Chemistry 2021, 144, 116407. [Google Scholar] [CrossRef]
- Nielsen, M.M.; Andersen, K.K.; Westh, P.; Otzen, D.E. Unfolding of β-sheet proteins in SDS. Biophysical journal 2007, 92(10), 3674–3685. [Google Scholar] [CrossRef]
| Sample No. | Part 1 | Part 2 |
|---|---|---|
| 1 | 40 mM | Buffer |
| 2 | 40 mM | 80 mM SDS |
| 3 | 40 mM | 6 mM SDS |
| 4 | 40 mM+ 6 mM SDS | 74 Mm SDS |
| 5 | 40 mM + 6 mM SDS | Buffer |
| 6 | 20 mM | Buffer |
| 7 | 20 mM | 80 mM SDS |
| 8 | 20 mM | 6 mM SDS |
| 9 | 40 mM + 6 mM SDS | Buffer |
| 10 | 20 mM+ 6 mM SDS | 74 mM SDS |
| 11 | 20 mM+ 30mM SDS | 50 mM SDS |
| 20 μM CG in a 10 mM buffer (%) |
20 μM CG + 6 mM SDS mixed with 74 Mm SDS (%) |
20 μM CG mixed with 80 mM SDS (%) |
20 μM + 30 mM SDS mixed with 50 mM SDS (%) |
|
|---|---|---|---|---|
| Helix | 11.7± | 22.2 | 26.1 | 26 |
| Antiparallel b | 23.5 | 9.2 | 11.8 | 12 |
| Parallel b | 4.1 | 4.8 | 2.5 | 3 |
| Turn | 21.7 | 15.5 | 19 | 19.1 |
| Others | 39 | 48.3 | 41 | 40 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




