Submitted:
28 December 2023
Posted:
29 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. System features
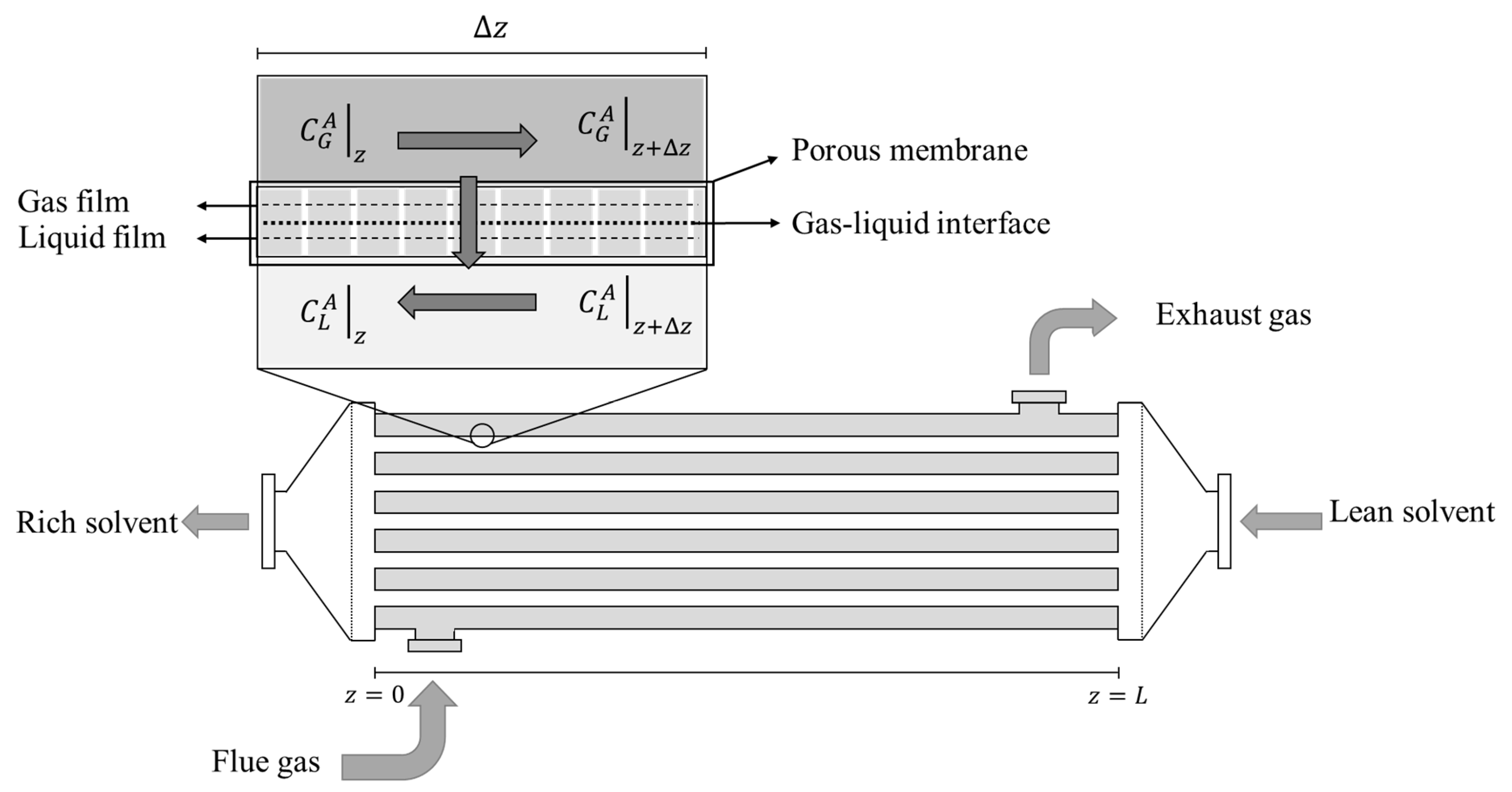
2.1. Geometry and flow
2.2. Main model hypothesis
3. Model development
3.1. Component mas balance equations
3.2. Component mas balance equations
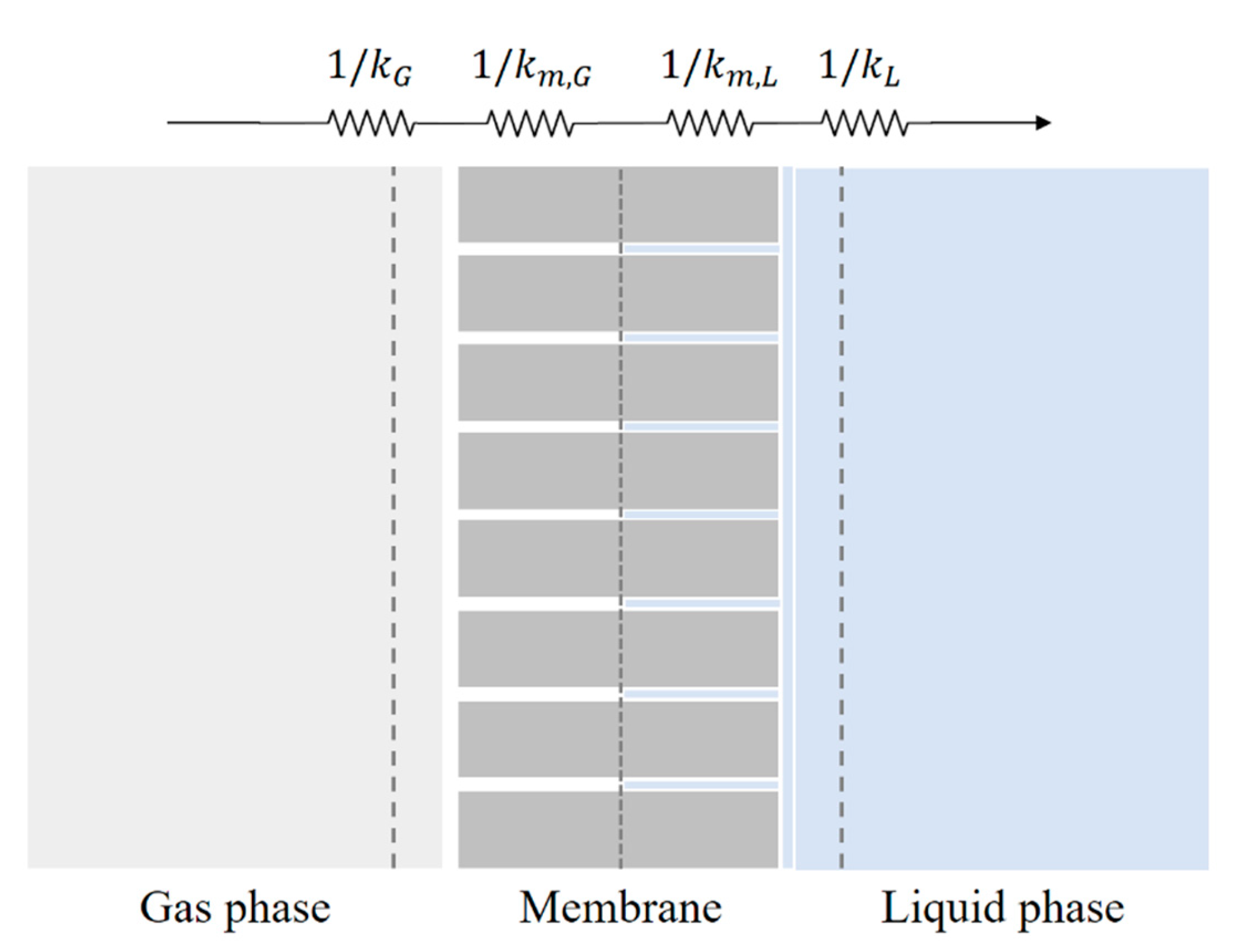
3.3. Kinetics
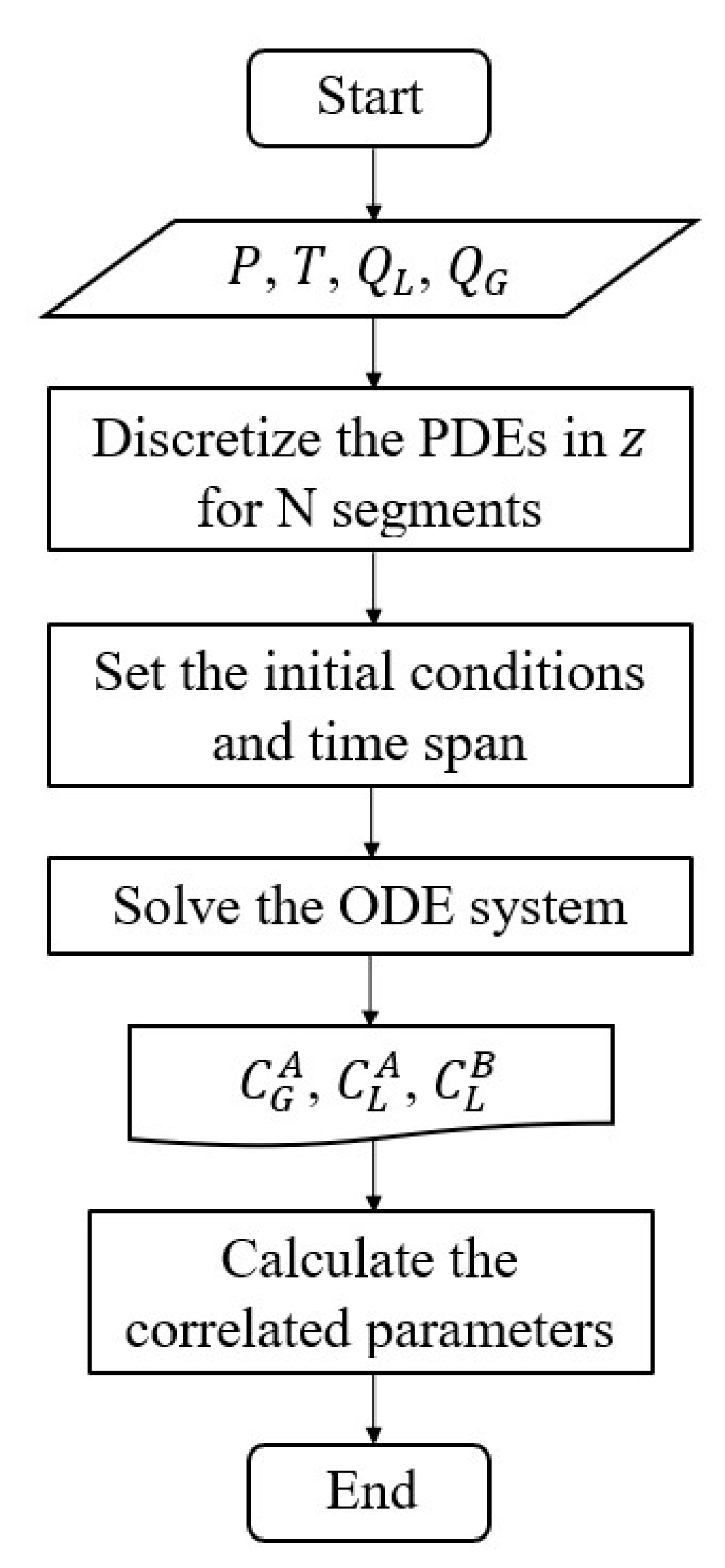
4. Numerical solution and validation parameters
5. Simulation results
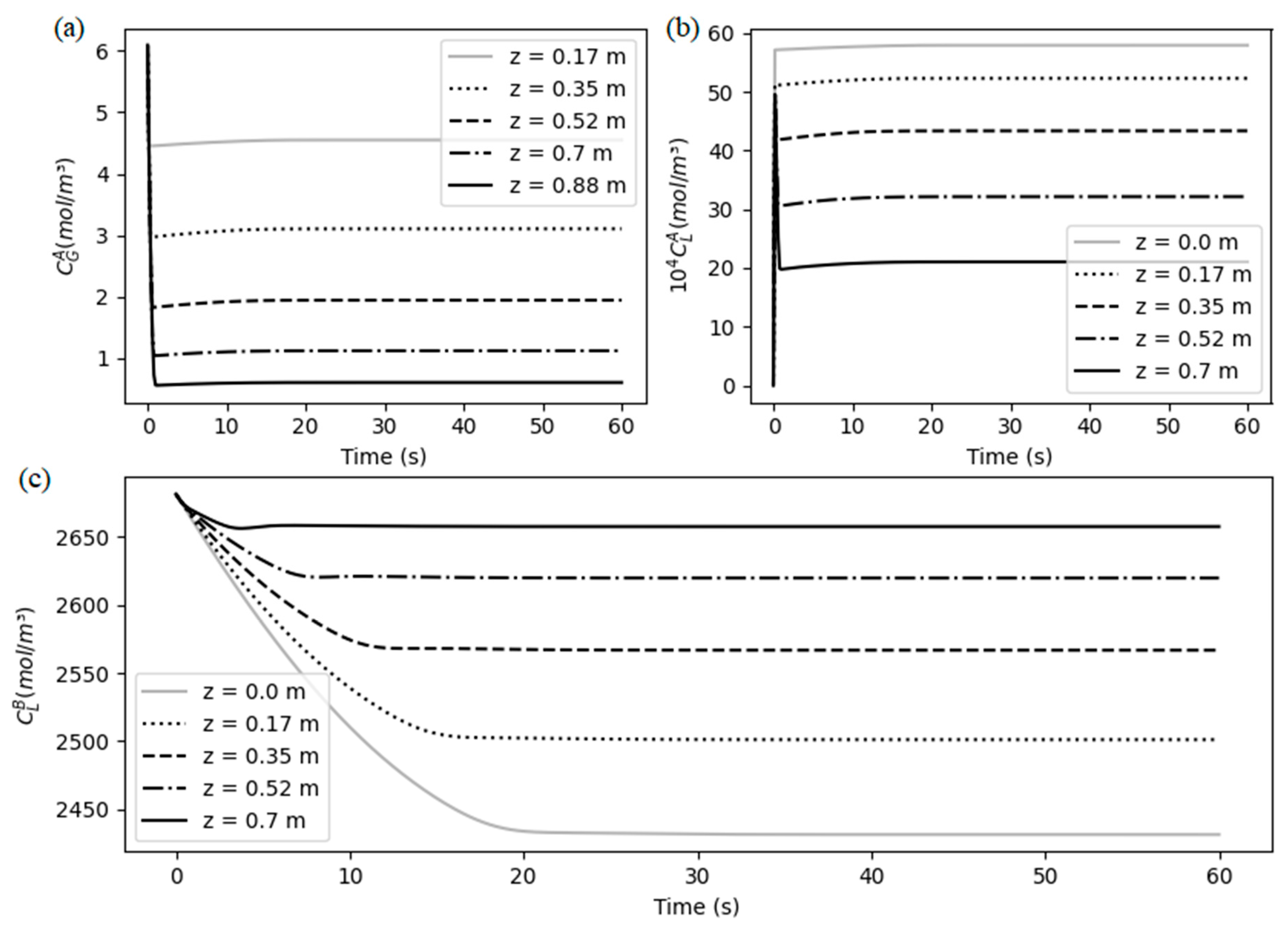
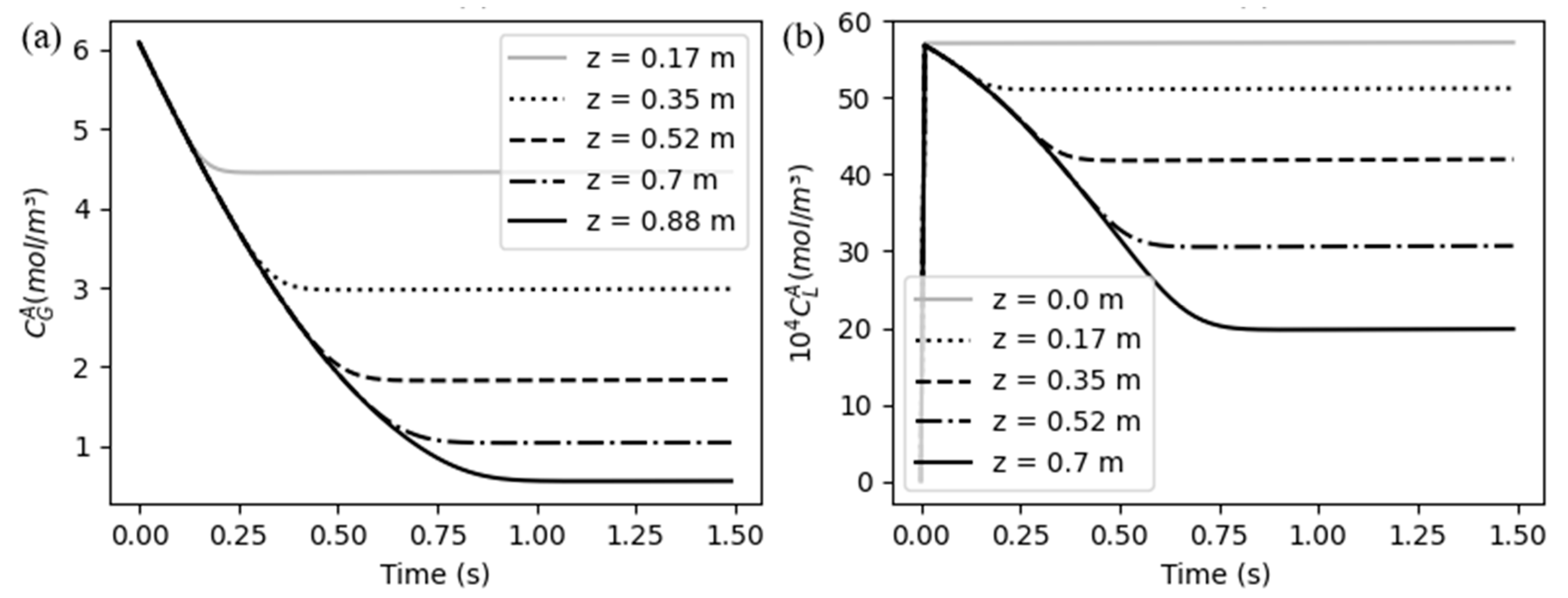
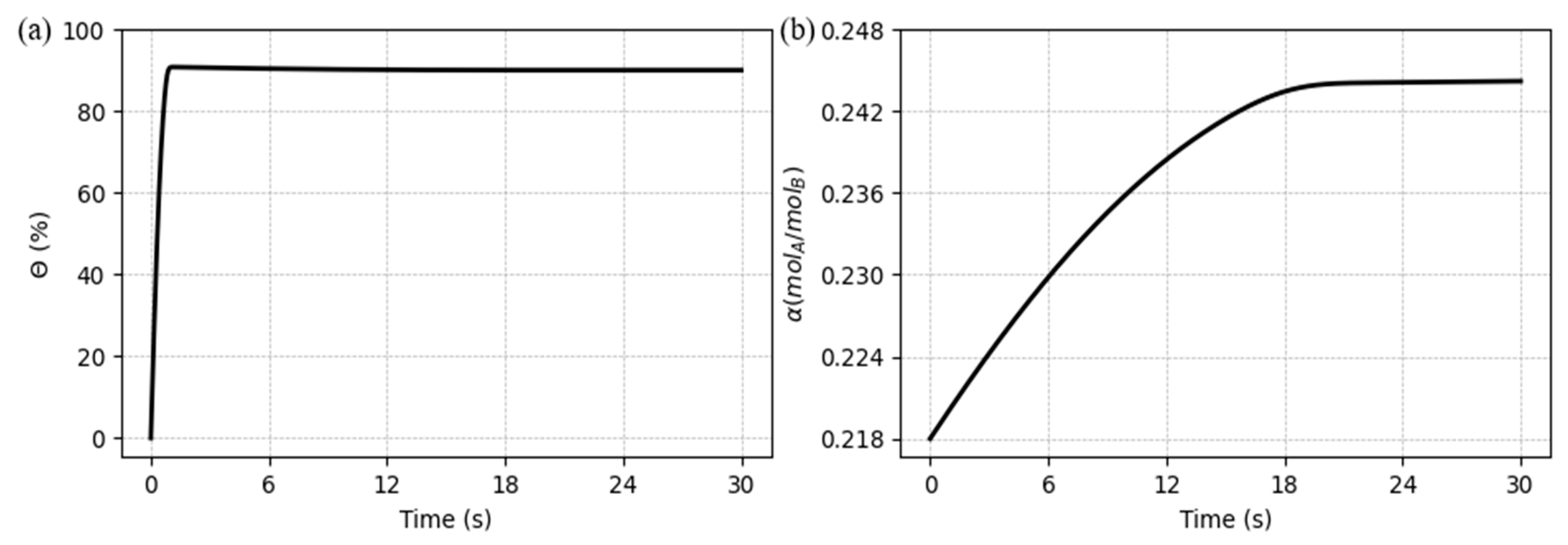
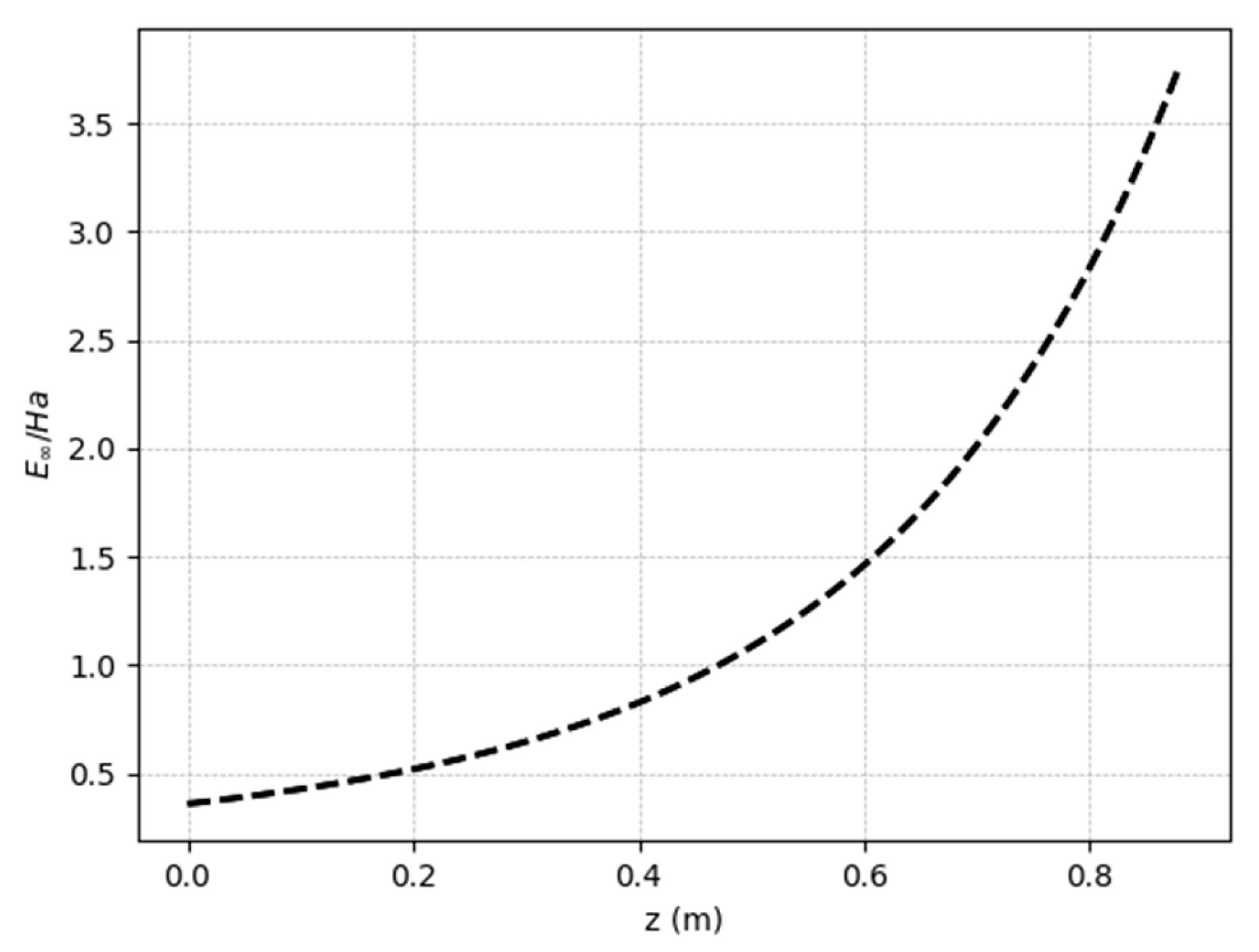
5.1. System behavior
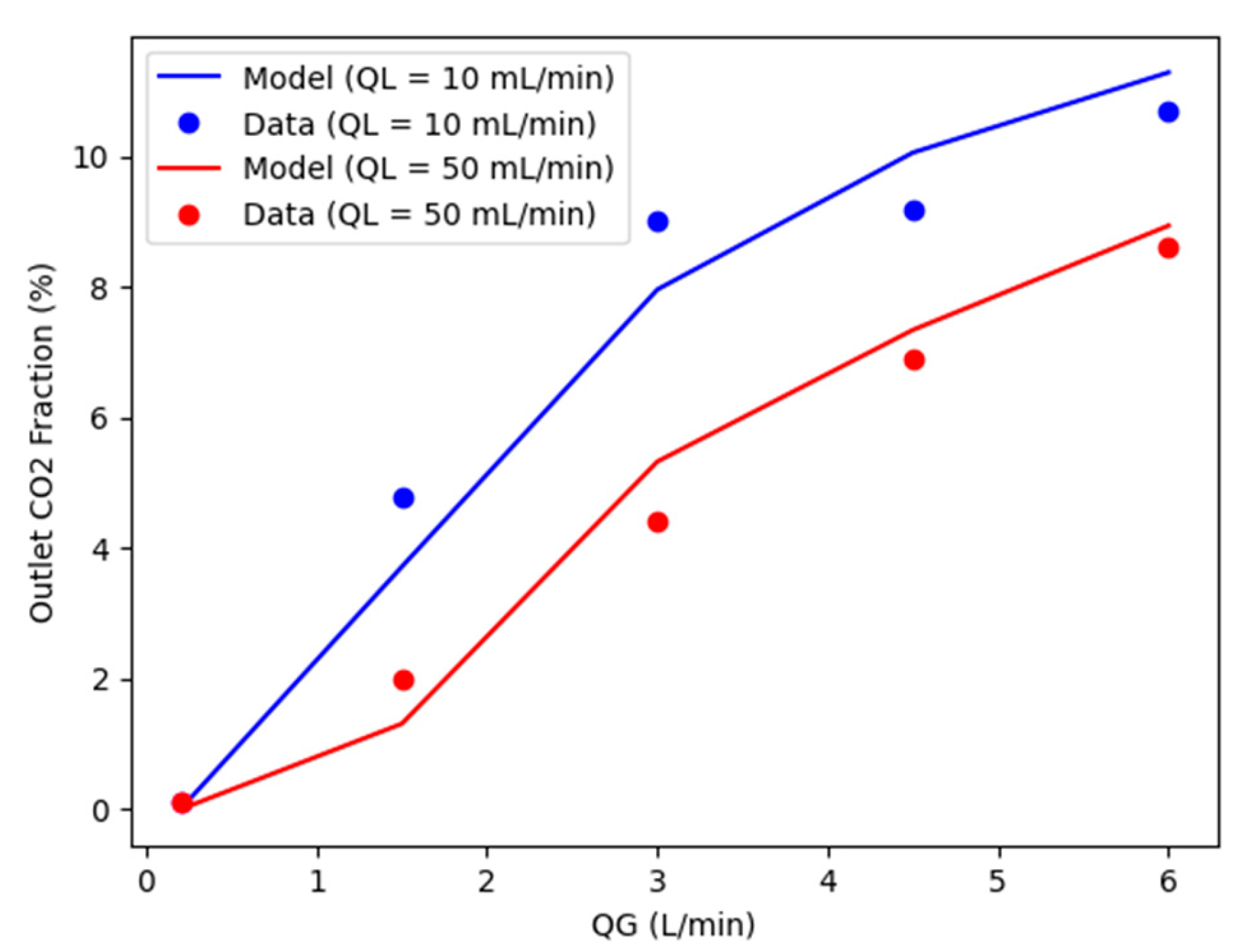
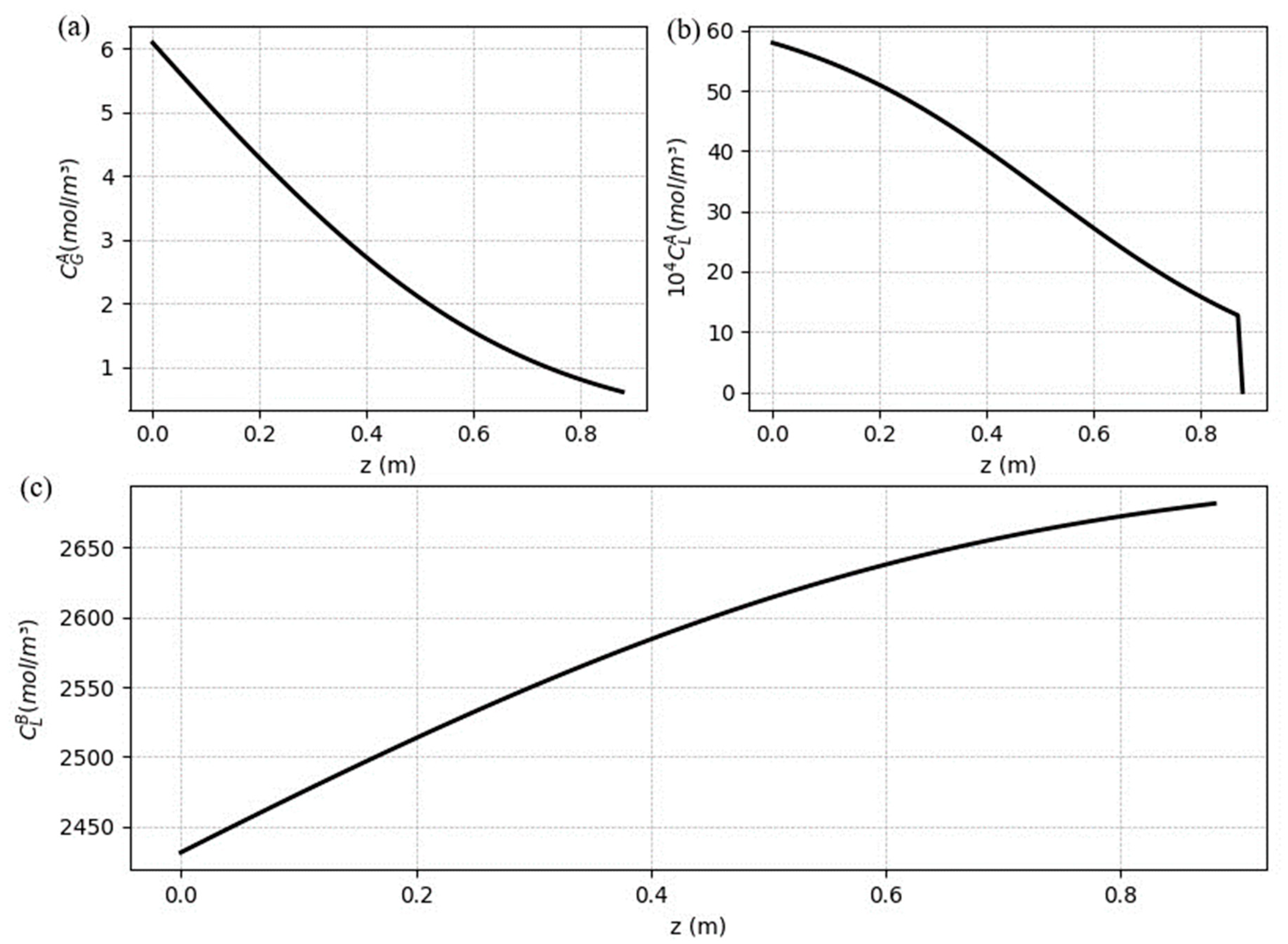
5.2. Steady-state results
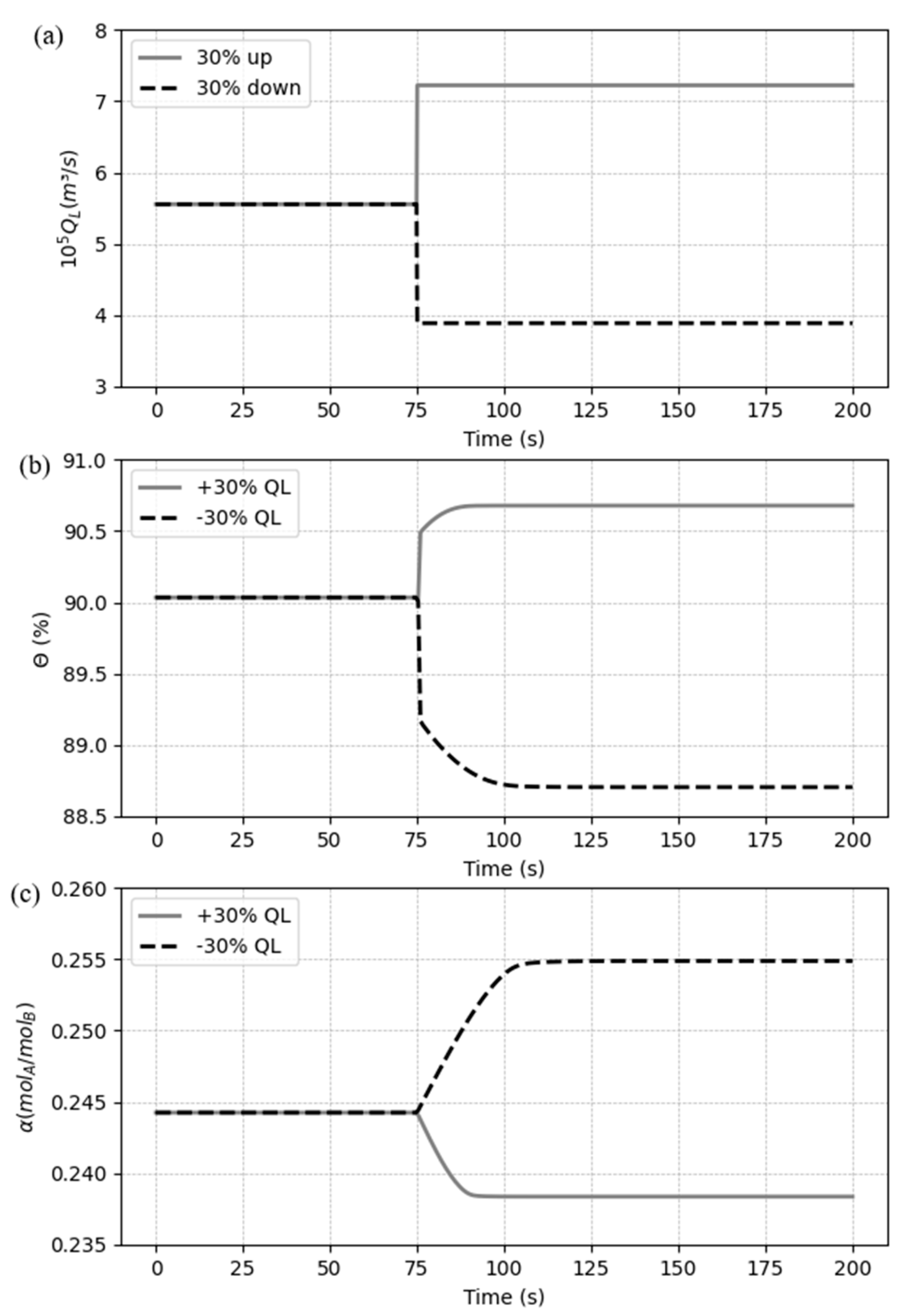
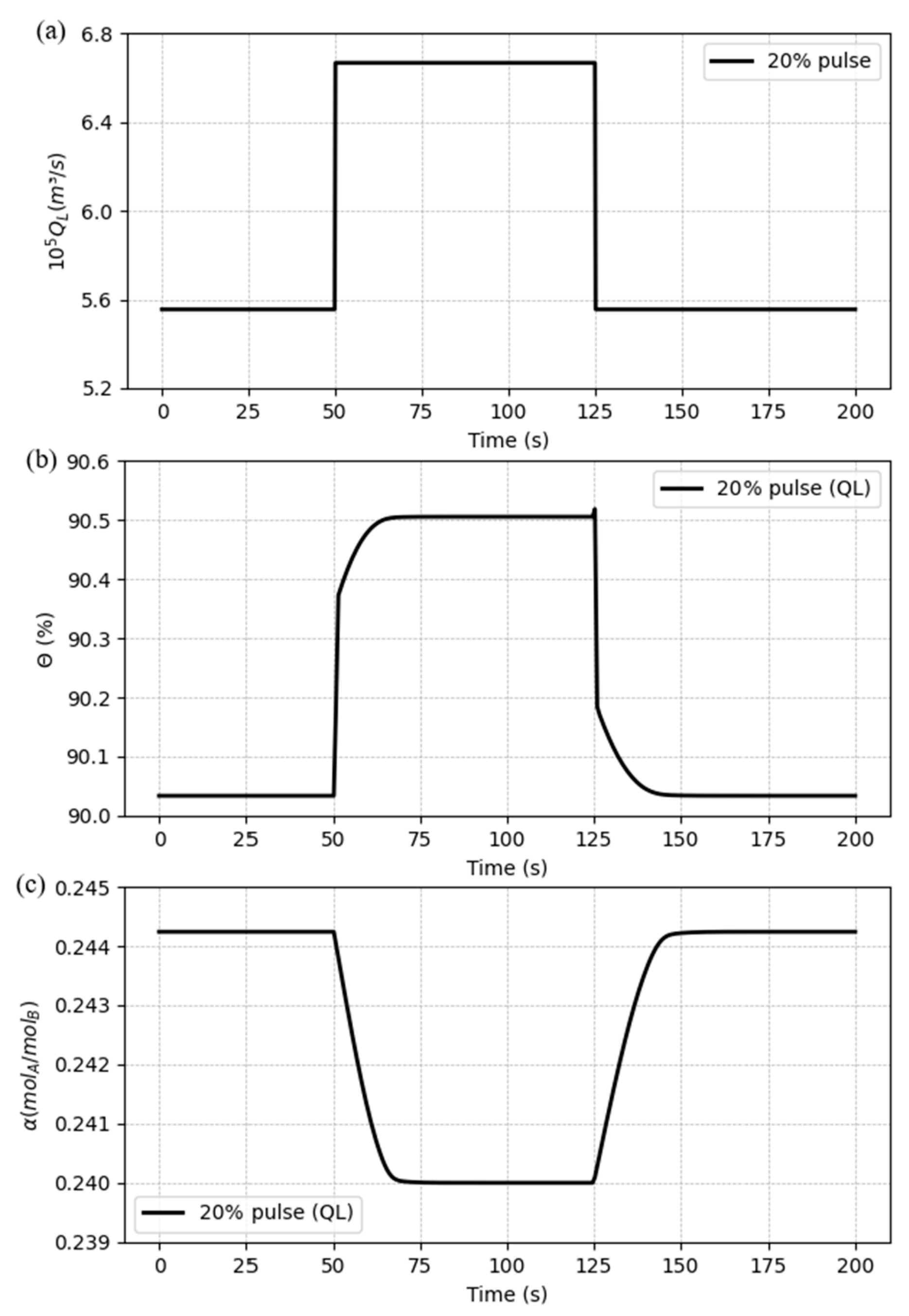
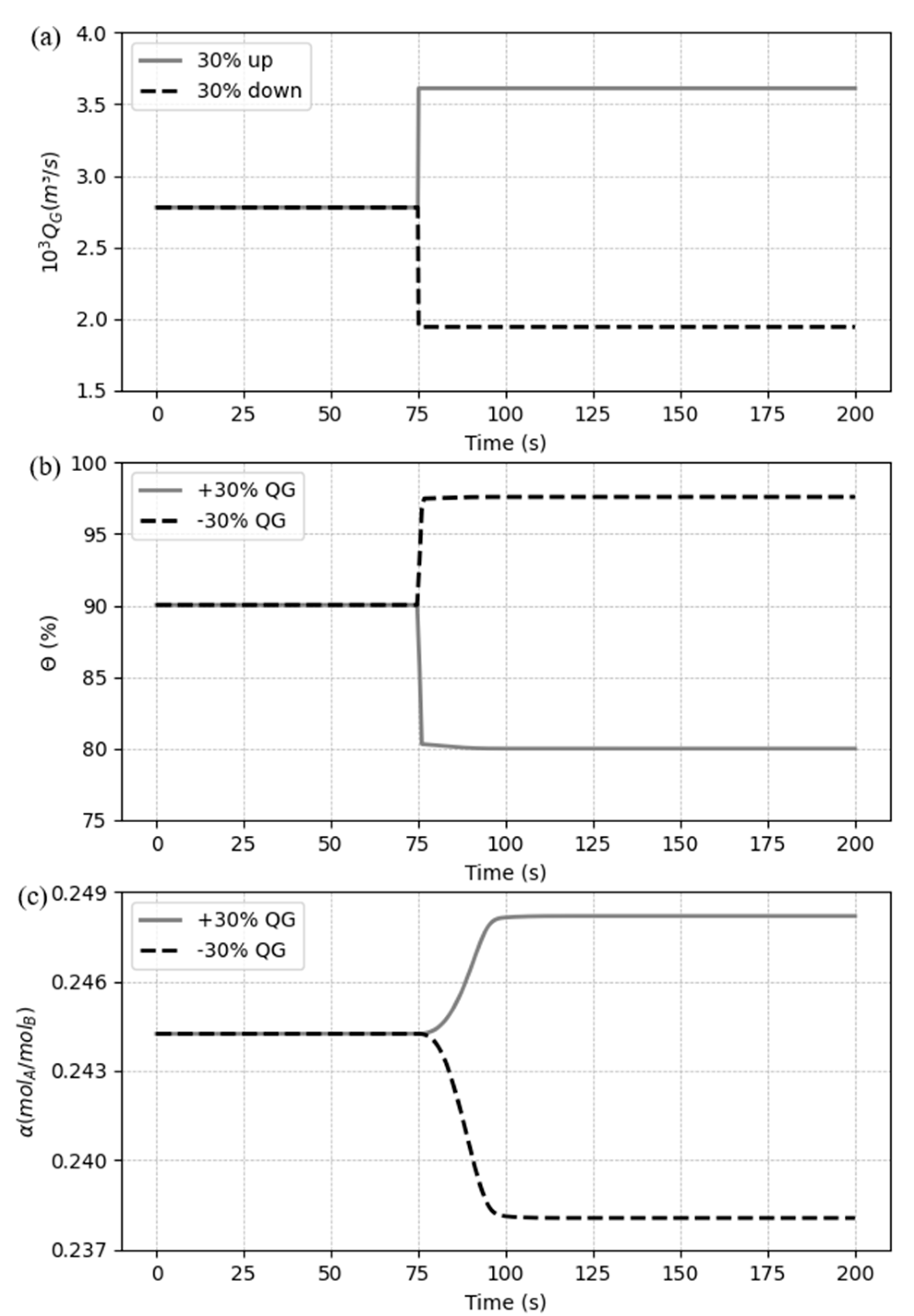
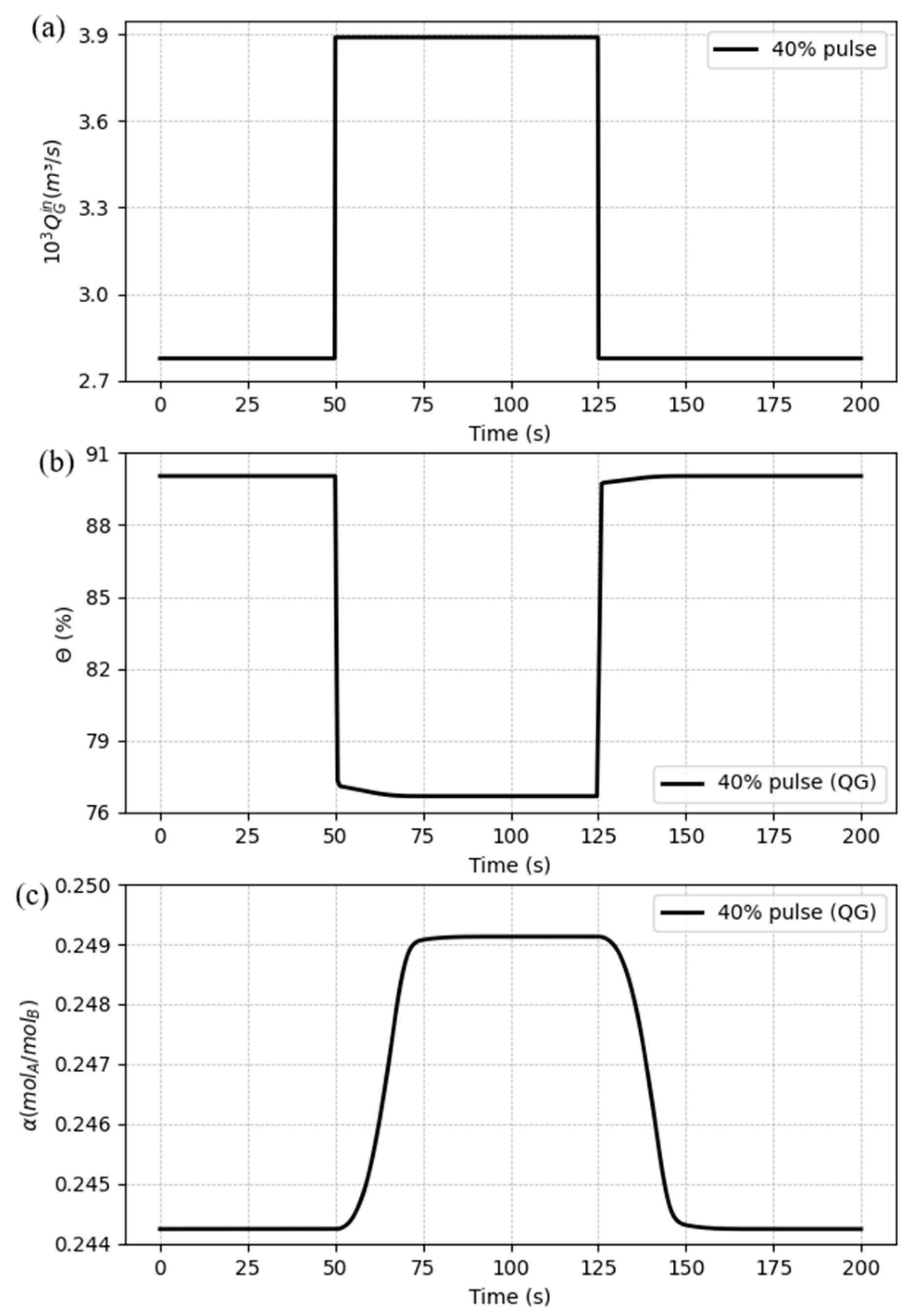
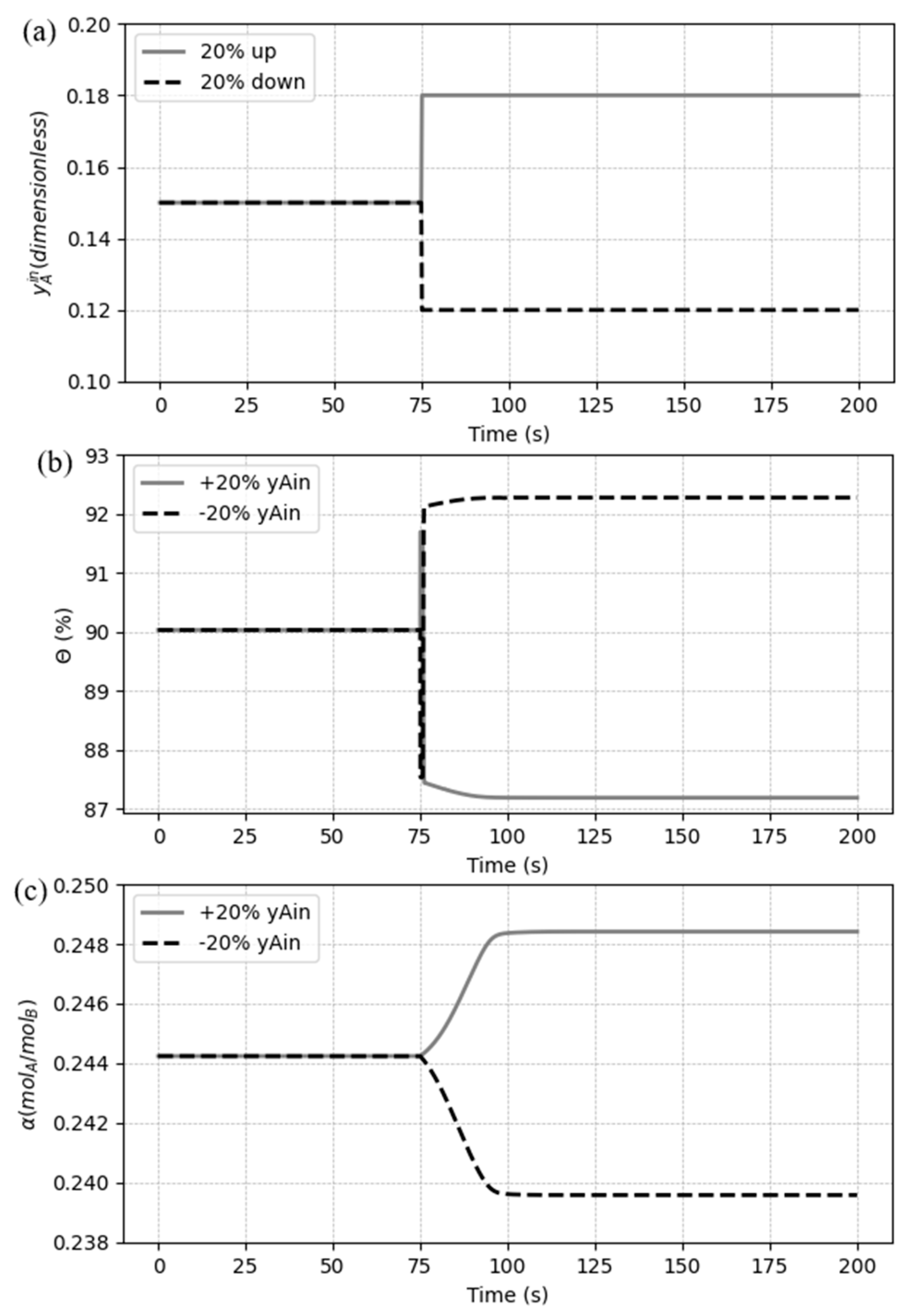
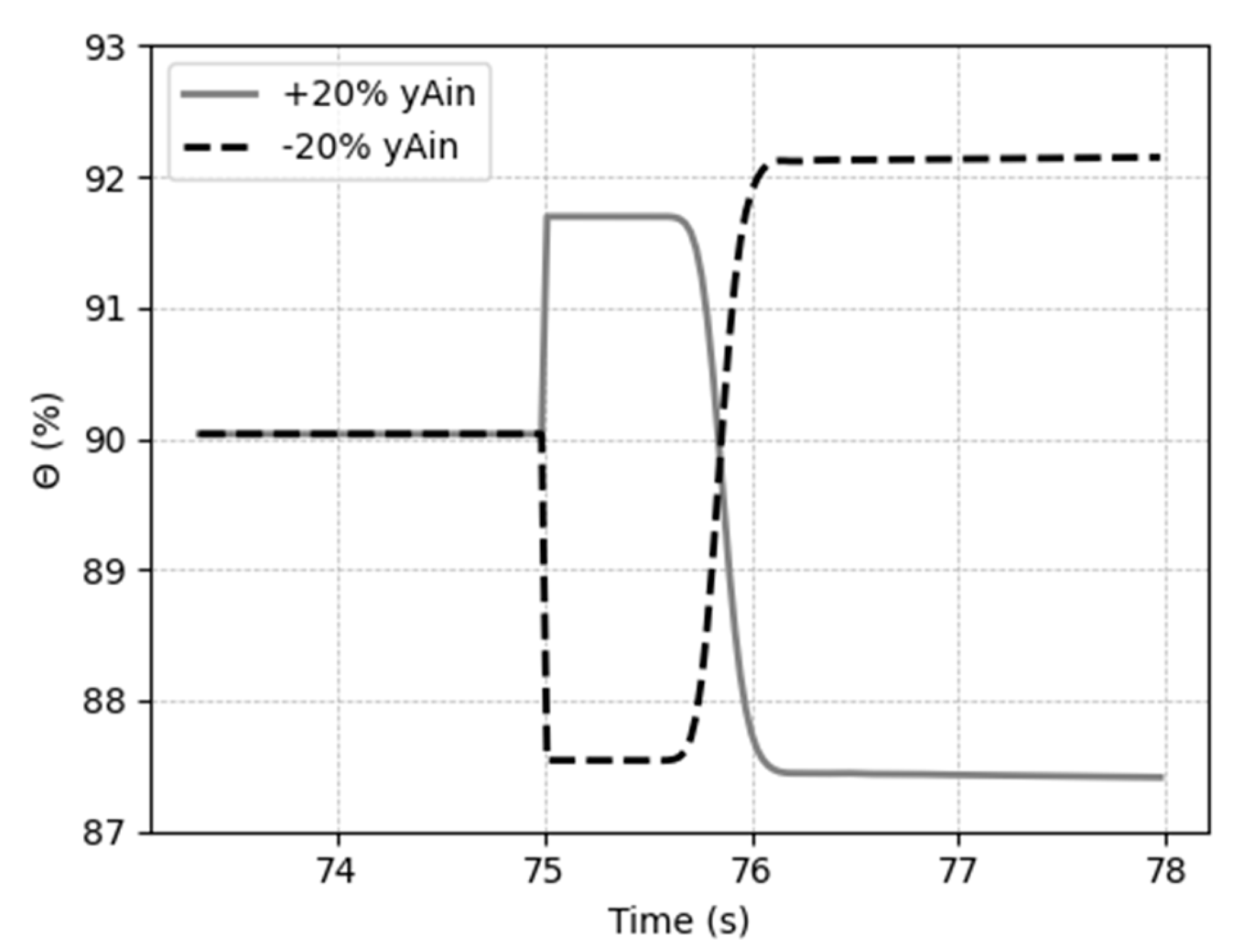
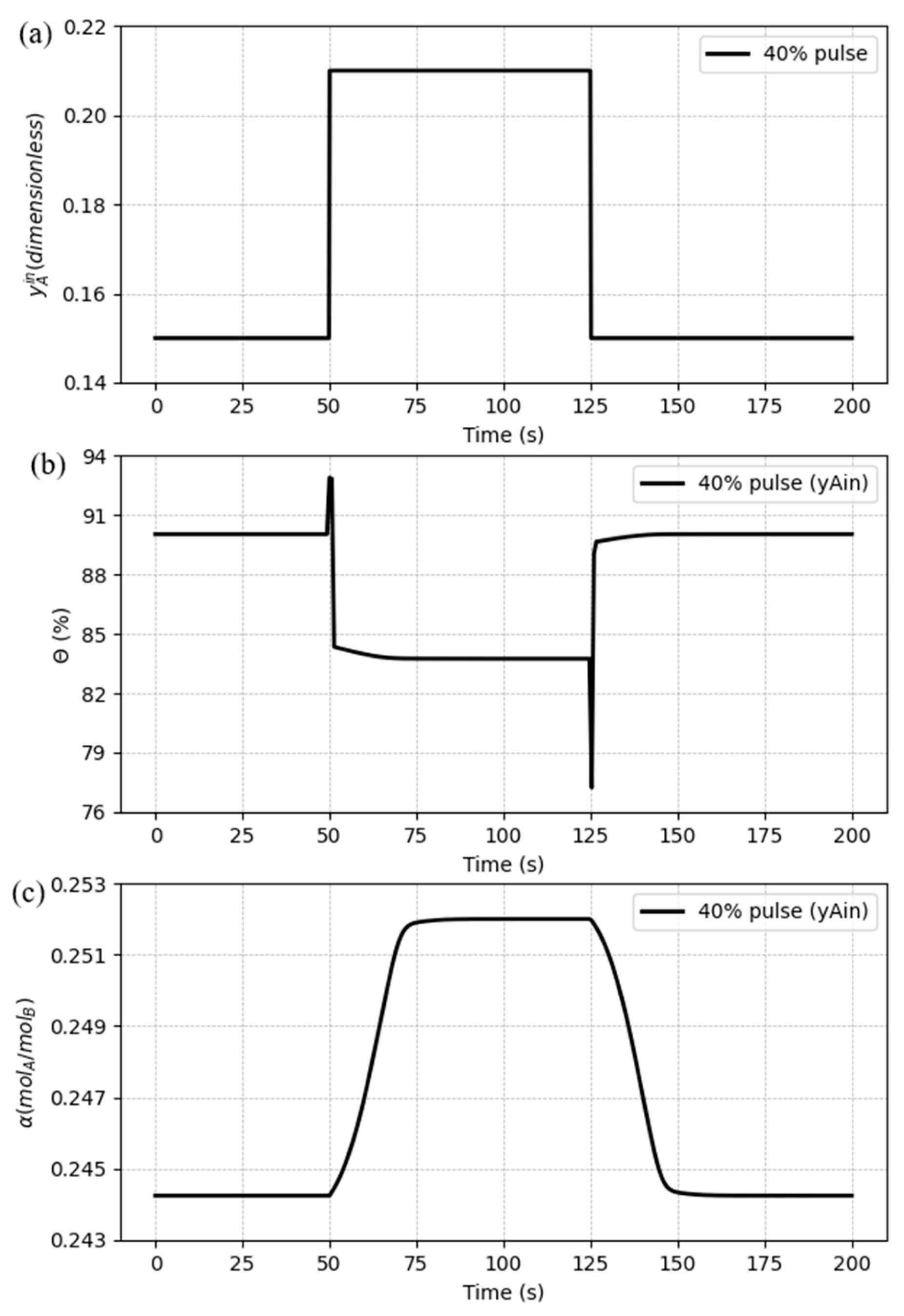
5.3. Dynamic response to process disturbances
5.3.1. Case 1: Disturbances in the liquid flow rate
5.3.2. Case 2: Disturbances in the gas flow rate
5.3.3. Case 3: Disturbances in the inlet CO2 composition
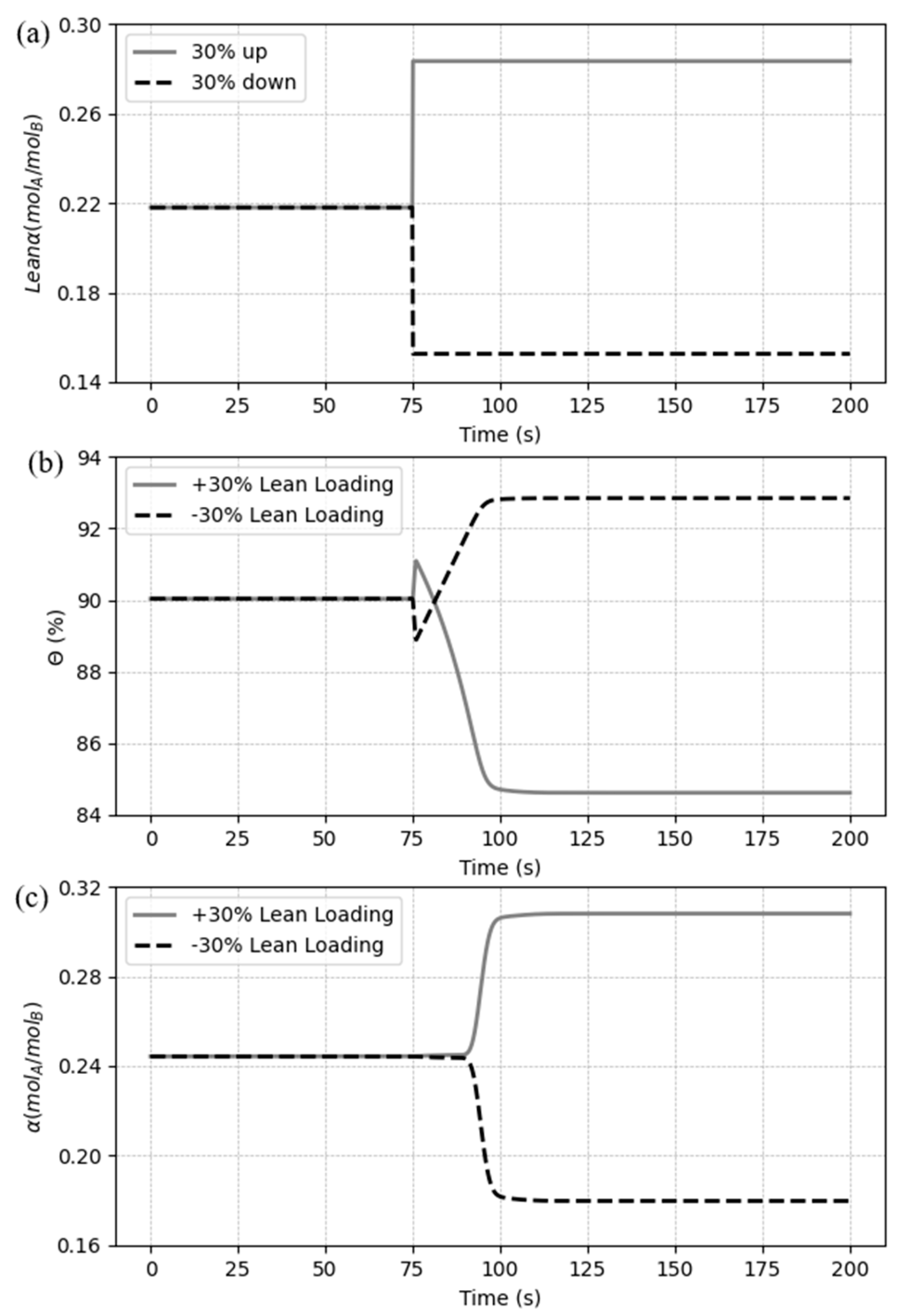
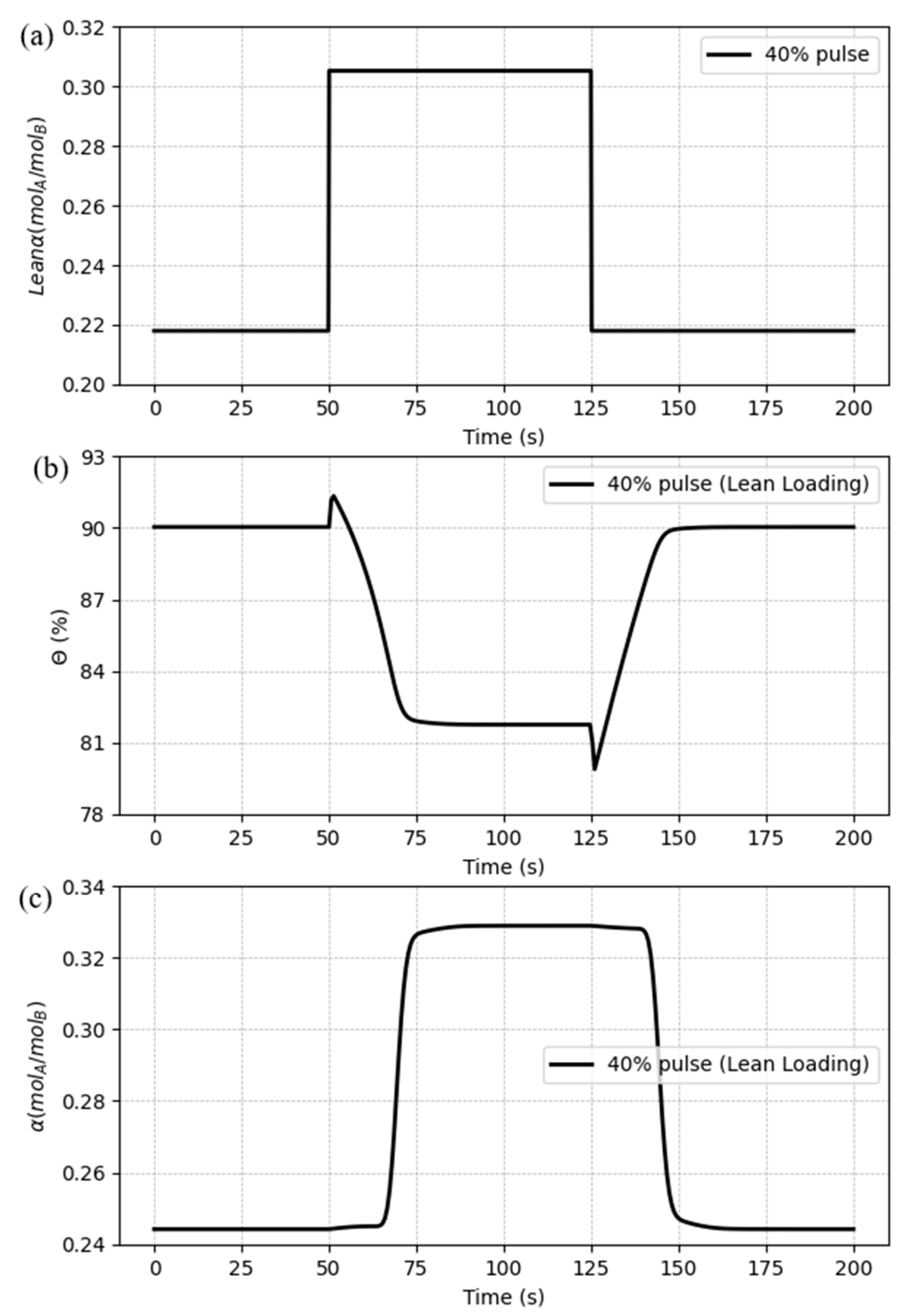
5.3.4. Case 4: Disturbances in the CO2 lean loading
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ghobadi, J.; Ramirez, D.; Khoramfar, S.; Kabir, M.M.; Jerman, R.; Saeed, M. Mathematical modeling of CO2 separation using different diameter hollow fiber membranes. Int. J. Greenh. Gas Control 2021, 104, 103204. [Google Scholar] [CrossRef]
- Leung, D.Y.; Caramanna, G.; Maroto-Valer, M.M. An overview of current status of carbon dioxide capture and storage technologies. Ren. Sus. Energy Reviews 2014, 39, 426–443. [Google Scholar] [CrossRef]
- Chao, C.; Deng, Y.; Dewil., R.; Baeyens, J.; Fan, X. Post-combustion carbon capture. Renew. Sus. Energy Reviews, 1104. [Google Scholar]
- Wang, M.; Lawal, A.; Stephenson, P.; Sidders, J.; Ramshaw, C. Post-combustion CO2 capture with chemical absorption: A state-of-the-art review. Chem. Eng. Res. Des. 2011, 89, 1609–1624. [Google Scholar] [CrossRef]
- Yan, S.P.; Fang, M.X.; Zhang, W.F.; Wang, S.Y.; Xu, Z.K.; Luo, Z.Y.; Cen, K.F. Experimental study on the separation of CO2 from flue gas using hollow fiber membrane contactors without wetting. Fuel Process. Tech. 2007, 88, 501–511. [Google Scholar] [CrossRef]
- Younas, M.; Druon-Bocquet, S.; Sanchez, J. Experimental and theoretical mass transfer transient analysis of copper extraction using hollow fiber membrane contactors. J. Mem. Sci. 2011, 382, 70–81. [Google Scholar] [CrossRef]
- Zhao, S.; Feron, P.H.; Deng, L.; Favre, E.; Chabanon, E.; Yan, S.; Hou, J.; Chen, V.; Qi, H. Status and progress of membrane contactors in post-combustion carbon capture: A state-of-the-art review of new developments. J. Mem. Sci. 2016, 511, 180–206. [Google Scholar] [CrossRef]
- Feron, P.H.M.; Jansen, A.E. Capture of carbon dioxide using membrane gas absorption and reuse in the horticultural industry. E. Conv. Man. 1995, 36, 411–414. [Google Scholar] [CrossRef]
- deMontigny, D.; Tontiwachwuthikul, P.; Chakma, A. Using polypropylene and polytetrafluoroethylene membranes in a membrane contactor for CO2 absorption. J. Mem. Sci. 2006, 277, 99–107. [Google Scholar] [CrossRef]
- Ma'mun, S.; Dindore, V.Y.; Svendsen, H.F. Kinetics of the reaction of carbon dioxide with aqueous solutions of 2-((2-aminoethyl) amino) ethanol. Ind. Eng. Chem. Res. 2007, 46, 385–394. [Google Scholar] [CrossRef]
- Atchariyawut, S.; Jiraratananon, R.; Wang, R. Mass transfer study and modeling of gas–liquid membrane contacting process by multistage cascade model for CO2 absorption. Sep. Purif. Tech. 2008, 63, 15–22. [Google Scholar] [CrossRef]
- Theo, W.L.; Lim, J.S.; Hashim, H.; Mustaffa, A.A.; Ho, W.S. Review of pre-combustion capture and ionic liquid in carbon capture and storage. App. Energy 2016, 183, 1633–1663. [Google Scholar] [CrossRef]
- Gabelman, A.; Hwang, S.T. Hollow fiber membrane contactors. J. Mem. Sci 1999, 159, 61–106. [Google Scholar] [CrossRef]
- Li, J.L.; Chen, B.H. Review of CO2 absorption using chemical solvents in hollow fiber membrane contactors. Sep. Purif. Tech. 2005, 41, 109–122. [Google Scholar] [CrossRef]
- Simons, K.; Nijmeijer, K.; Wessling, M. Gas–liquid membrane contactors for CO2 removal. J. Mem. Sci.
- Cui, Z.; deMontigny, D. Part 7: A Review of CO2 Capture Using Hollow Fiber Membrane Contactors. Carbon Manag. 2013, 4, 69–89. [Google Scholar] [CrossRef]
- Rivero, J.R.; Panagakos, G.; Lieber, A.; Hornbostel, K. Hollow fiber membrane contactors for post-combustion carbon capture: A review of modeling approaches. Mem. 2020, 10, 382. [Google Scholar] [CrossRef]
- Mansourizadeh, A. Experimental study of CO2 absorption/stripping via PVDF hollow fiber membrane contactor. Chem. Eng. Res. Des. 2012, 90, 555–562. [Google Scholar] [CrossRef]
- Rode, S.; Nguyen, P.T.; Roizard, D.; Bounaceur, R.; Castel, C.; Favre, E. Evaluating the intensification potential of membrane contactors for gas absorption in a chemical solvent: A generic one-dimensional methodology and its application to CO2 absorption in monoethanolamine. J. Mem. Sci. 2012, 389, 1–16. [Google Scholar] [CrossRef]
- Zaidiza, D.A.; Billaud, J.; Belaissaoui, B.; Rode, S.; Roizard, D.; Favre, E. Modeling of CO2 post-combustion capture using membrane contactors, comparison between one-and two-dimensional approaches. J. Mem. Sci. 2014, 455, 64–74. [Google Scholar] [CrossRef]
- Khaisri, S.; deMontigny, D.; Tontiwachwuthikul, P.; Jiraratananon, R. A mathematical model for gas absorption membrane contactors that studies the effect of partially wetted membranes. J. Mem. Sci. 2010, 347, 228–239. [Google Scholar] [CrossRef]
- Zaidiza, D.A.; Belaissaoui, B.; Rode, S.; Neveux, T.; Makhloufi, C.; Castel, C.; Roizard, D.; Favre, E. Adiabatic modelling of CO2 capture by amine solvents using membrane contactors. J. Mem. Sci. 2015, 493, 106–119. [Google Scholar] [CrossRef]
- Porcheron, F.; Drozdz, S. Hollow fiber membrane contactor transient experiments for the characterization of gas/liquid thermodynamics and mass transfer properties. Chem. Eng. Sci. 2009, 64, 265–275. [Google Scholar] [CrossRef]
- Pakšiová, D.; Fikar, M.; Skogestad, S. Modeling of carbon dioxide removal using membrane contactors. In 2016 Cybernetics & Informatics (K&I), Levoca, Slovakia, 2016, (pp. 1-6).
- Rosli, A.; Shoparwe, N.F.; Ahmad, A.L.; Low, S.C.; Lim, J.K. Dynamic modelling and experimental validation of CO2 removal using hydrophobic membrane contactor with different types of absorbent. Sep. Purif. Tech. 2019, 219, 230–240. [Google Scholar] [CrossRef]
- Asadi, J.; Kazempoor, P. Dynamic response and flexibility analyses of a membrane-based CO2 separation module. International J. Greenh. Gas Control 2022, 116, 103634. [Google Scholar] [CrossRef]
- Bozonc, A.C.; Cormos, A.M.; Dragan, S.; Dinca, C.; Cormos, C.C. Dynamic Modeling of CO2 Absorption Process Using Hollow-Fiber Membrane Contactor in MEA Solution. Energies 2022, 15, 7241. [Google Scholar] [CrossRef]
- Wattanaphan, P.; Sema, T.; Idem, R.; Liang, Z.; Tontiwachwuthikul, P. Effects of flue gas composition on carbon steel (1020) corrosion in MEA-based CO2 capture process. Int. J. Greenh. Gas Control 2013, 19, 340–349. [Google Scholar] [CrossRef]
- D'Alessandro, D.M.; Smit, B.; Long, J.R. Carbon dioxide capture: prospects for new materials. Angewandte Chemie International Edition 2010, 49, 6058–6082. [Google Scholar] [CrossRef] [PubMed]
- Scholes, C.A.; Ho, M.T.; Wiley, D.E. Membrane-cryogenic post-combustion carbon capture of flue gases from NGCC. Tech. 2016, 4, 14. [Google Scholar] [CrossRef]
- Chabanon, E.; Kimball, E.; Favre, E.; Lorain, O.; Goetheer, E.; Ferre, D.; Gomez, A.; Broutin, P. Hollow fiber membrane contactors for post-combustion CO2 capture: A scale-up study from laboratory to pilot plant. Oil & Gas Sci. Tech.–Revue d’IFP Energies nouvelles 2014, 69, 1035–1045. [Google Scholar]
- Kimball, E.; Al-Azki, A.; Gomez, A.; Goetheer, E.; Booth, N.; Adams, D.; Ferre, D. Hollow fiber membrane contactors for CO2 capture: modeling and up-scaling to CO2 capture for an 800 MWe coal power station. Oil & Gas Sci. Tech.–Revue d’IFP Energies nouvelles 2014, 69, 1047–1058. [Google Scholar]
- Zaidiza, D.A.; Wilson, S.G.; Belaissaoui, B.; Rode, S.; Castel, C.; Roizard, D.; Favre, E. Rigorous modelling of adiabatic multicomponent CO2 post-combustion capture using hollow fiber membrane contactors. Chem. Eng. Sci. 2016, 145, 45–58. [Google Scholar] [CrossRef]
- Zaidiza, D.A.; Belaissaoui, B.; Rode, S.; Favre, E. Intensification potential of hollow fiber membrane contactors for CO2 chemical absorption and stripping using monoethanolamine solutions. Sep. Purif. Tech. 2017, 188, 38–51. [Google Scholar] [CrossRef]
- Happel, J. Viscous flow relative to arrays of cylinders. AIChE Journal 1959, 5, 174–177. [Google Scholar] [CrossRef]
- Boucif, N.; Corriou, J.P.; Roizard, D.; Favre, E. Carbon dioxide absorption by monoethanolamine in hollow fiber membrane contactors: A parametric investigation. AIChE journal 2011, 58, 2843–2855. [Google Scholar] [CrossRef]
- Farjami, M.; Moghadassi, A.; Vatanpour, V. Modeling and simulation of CO2 removal in a polyvinylidene fluoride hollow fiber membrane contactor with computational fluid dynamics. Chem. Eng. Processing: Process Intens. 2015, 98, 41–51. [Google Scholar] [CrossRef]
- Nakhjiri, A.T.; Heydarinasab, A.; Bakhtiari, O.; Mohammadi, T. Modeling and simulation of CO2 separation from CO2/CH4 gaseous mixture using potassium glycinate, potassium argininate and sodium hydroxide liquid absorbents in the hollow fiber membrane contactor. J. Env. Chem. Eng. 2018, 6, 1500–1511. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, H.Y.; Feron, P.H.M.; Liang, D.T. Influence of membrane wetting on CO2 capture in microporous hollow fiber membrane contactors. Sep. Purif. Tech. 2005, 46, 33–40. [Google Scholar] [CrossRef]
- Keshavarz, P.; Fathikalajahi, J.; Ayatollahi, S. Analysis of CO2 separation and simulation of a partially wetted hollow fiber membrane contactor. J. Haz. Mat. 2008, 152, 1237–1247. [Google Scholar] [CrossRef] [PubMed]
- Mavroudi, M.; Kaldis, S.P.; Sakellaropoulos, G.P. A study of mass transfer resistance in membrane gas–liquid contacting processes. J. Mem. Sci. 2006, 272, 103–115. [Google Scholar] [CrossRef]
- Skelland, A.H.P. Diffusional Mass Transfer 1974.
- Miyatake, O.; Iwashita, H. Laminar-flow heat transfer to a fluid flowing axially between cylinders with a uniform wall heat flux. Int. J. Heat Mass Transf. 1991, 34, 322–327. [Google Scholar] [CrossRef]
- Bao, L.; Lipscomb, G.G. Mass transfer in axial flows through randomly packed fiber bundles with constant wall concentration. J. Mem. Sci. 2002, 204, 207–220. [Google Scholar] [CrossRef]
- Bao, L.; Lipscomb, G.G. Well-developed mass transfer in axial flows through randomly packed fiber bundles with constant wall flux. Chem. Eng. Sci. 2002, 57, 125–132. [Google Scholar] [CrossRef]
- Kreulen, H.; Smolders, C.A.; Versteeg, G. F.; Van Swaaij, W.P.M. Determination of mass transfer rates in wetted and non-wetted microporous membranes. Chem. Eng. Sci. 1993, 48, 2093–2102. [Google Scholar] [CrossRef]
- Hikita, H.; Asai, S.; Ishikawa, H.; Honda, M. The kinetics of reactions of carbon dioxide with monoethanolamine, diethanolamine and triethanolamine by a rapid mixing method. The Chem. Eng. J. 1977, 13, 7–12. [Google Scholar] [CrossRef]
- Danckwerts, P.V. The reaction of CO2 with ethanolamines. Chem. Eng. Sci. 1979, 34, 443–446. [Google Scholar] [CrossRef]
- Beek, W.J.; Muttzall, K.M.K.; van Heuven, J.M. Transport Phenomena, 2nd ed.; John Wiley & Sons: London, England, 1999. [Google Scholar]
- Levenspiel, O. Chemical Engineering Reaction, 3rd ed.; John Wiley & Sons: New York, USA, 1999. [Google Scholar]
- Zhang, H.Y.; Wang, R.; Liang, D.T.; Tay, J.H. Modeling and experimental study of CO2 absorption in a hollow fiber membrane contactor. J. Mem. Sci. 2006, 279, 301–310. [Google Scholar] [CrossRef]
- Sohaib, Q.; Muhammad, A.; Younas, M.; Rezakazemi, M. Modeling pre-combustion CO2 capture with tubular membrane contactor using ionic liquids at elevated temperatures. Sep. Purif. Tech. 2020, 241, 116677. [Google Scholar] [CrossRef]
- Ma’mun, S.; Svendsen, H.F. Solubility of N2O in aqueous monoethanolamine and 2-(2-Aminoethyl-amino)ethanol solutions from 298 to 343 K. Energy Procedia 2009, 1, 837–843. [Google Scholar] [CrossRef]
- Poling, B.E.; Prausnitz, J.M.; O’Connell, J.P. The Properties of Gases and Liquids, 5th ed.; McGraw-Hill: New York, USA, 2001. [Google Scholar]
- Weiland, R.H.; Dingman, J.C.; Cronin, D.B.; Browning, G.J. Density and viscosity of some partially carbonated aqueous alkanolamine solutions and their blends. J. Chem. Eng. Data 1998, 43, 378–382. [Google Scholar] [CrossRef]
- Maceiras, R.; Álvarez, E.; Cancela, M.A. Effect of temperature on carbon dioxide absorption in monoethanolamine solutions. Chem. Eng. J. 2008, 138, 295–300. [Google Scholar] [CrossRef]
- Versteeg, G.F.; Van Swaaij, W.P. Solubility and diffusivity of acid gases (carbon dioxide, nitrous oxide) in aqueous alkanolamine solutions. J. Chem. Eng. Data 1988, 33, 29–34. [Google Scholar] [CrossRef]
- Snijder, E.D.; te Riele, M.J.; Versteeg, G.F.; Van Swaaij, W.P.M. Diffusion coefficients of several aqueous alkanolamine solutions. J. Chem. Eng. Data 1993, 38, 475–480. [Google Scholar] [CrossRef]
- Versteeg, G.F.; Van Dijck, L.A.J.; van Swaaij, W.P.M. On the kinetics between CO2 and alkanolamines both in aqueous and non-aqueous solutions. An overview. Chem. Eng. Communi. 1996, 144, 113–158. [Google Scholar] [CrossRef]
| Parameter | Expression | Units | References |
|---|---|---|---|
| Shell radius | n | [36] | |
| Gas flow sectional area | m² | - | |
| Total liquid flow sectional area | m² | - | |
| External hydraulic diameter | m | [20] | |
| Internal hydraulic diameter | m | [20] |
| Condition | Description |
|---|---|
| Flow |
|
| |
| System constraints |
|
| |
| |
| |
| Membranes |
|
| Thermodynamics |
|
| |
| Mass transfer |
|
| Chemical reaction |
|
| Reaction regime | Relationship |
|---|---|
| Pseudo first-order reaction by the gas-liquid interface (reaction is limiting the process) | |
| Fast second order reaction (mass transfer is partially limiting the process) | |
| Instantaneous reaction at the film (mass transfer is limiting the process) |
| Parameter | Expression | Units |
|---|---|---|
| Parameter | Lab-scale | Pilot-scale | |
|---|---|---|---|
| Module | Inner radius (m) | ||
| Effective length (m) | |||
| Number of fibers (-) | |||
| Packing ration (-) | |||
| Specific interfacial area (m²/m³) | |||
| Fiber | Inner radius (m) | ||
| Outer radius (m) | |||
| Porosity (-) | |||
| Material (-) | |||
| Parameter | Expression | Units | References |
|---|---|---|---|
| Henry constant for CO2 in MEA solution |
: CO2, N2 : water, MEA 30% (mass) |
Pa.m3mol-1 | [53] |
| Diffusion coefficient of CO2 on gas mixture |
: N2, O2, H2O |
m2s-1 | [54] |
| Density of MEA solution |
: MEA, H2O, CO2 |
10-3 kg.m-3 | [55] |
| Initial concentration of MEA | mol.m-3 | - | |
| Viscosity of MEA solution | Pa.s | [56] | |
| Diffusion coefficient of CO2 in MEA solution | m2s-1 | [57] | |
| Diffusion coefficient of MEA in MEA solution | m2s-1 | [58] | |
| Kinetic constant | m3mol-1s-1 | [59] |
| Parameter | Value or expression | Units |
|---|---|---|
| Operation pressure | Pa | |
| Operation temperature | K | |
| Inlet volumetric rate of gas | m3s-1 | |
| Overall volumetric rate of liquid | m3s-1 | |
| Inlet molar fraction of CO2 on gas | - | |
| Inlet molar fraction of N2 on gas | ||
| Inlet concentration of CO2 on gas | mol.m-3 | |
| Inlet concentration of free CO2 on liquid | mol.m-3 | |
| Mass fraction of MEA | - | |
| Inlet concentration of free MEA on liquid | mol.m-3 | |
| Lean loading of CO2 | molCO2.molMEA-1 | |
| Number of segments on discretized space | - | |
| Membrane tortuosity | - | |
| Wetting fraction | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




