Submitted:
24 December 2023
Posted:
27 December 2023
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and methods
Granulocyte isolation
Extraction, purification and characterization of mycobacterial antigens
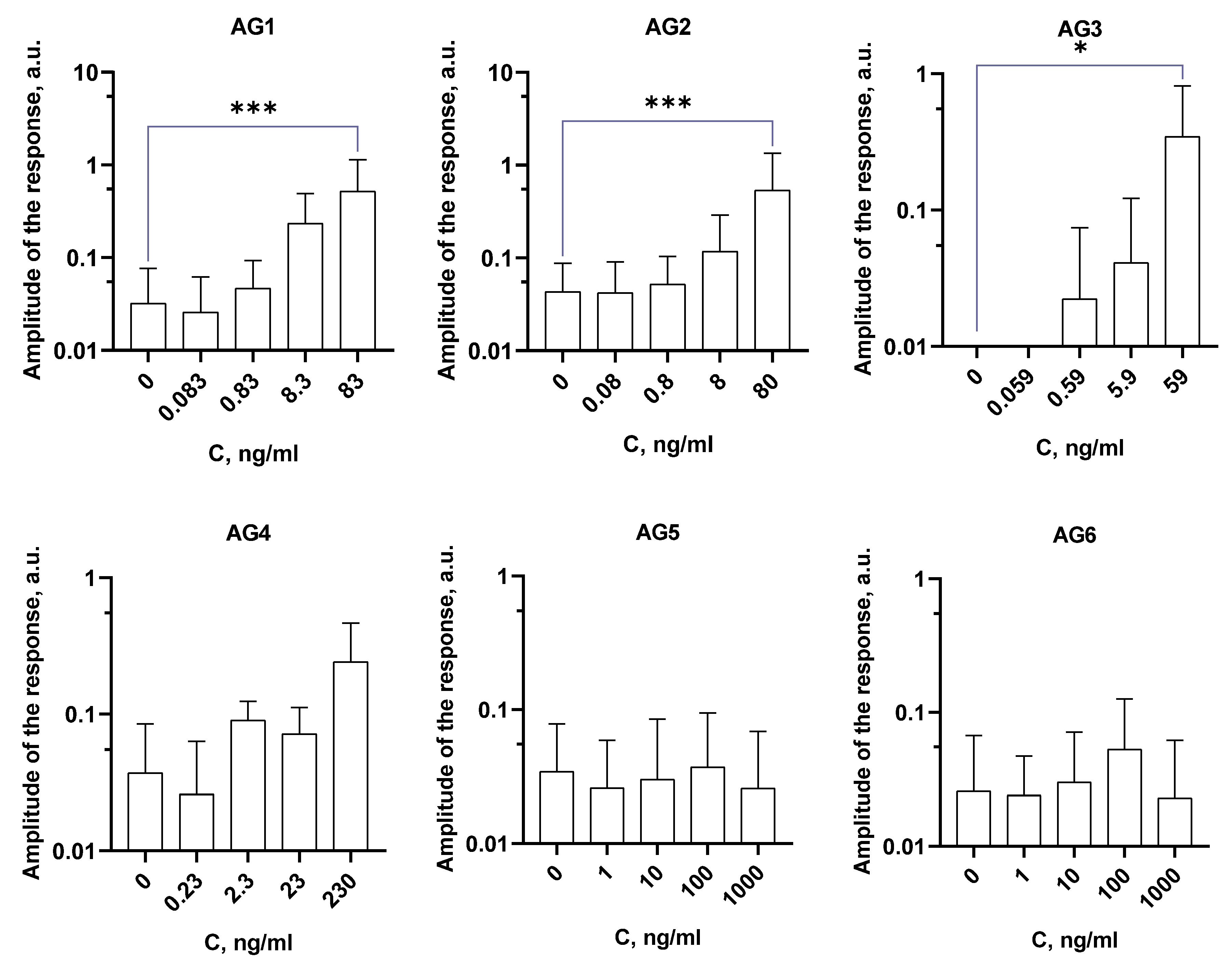
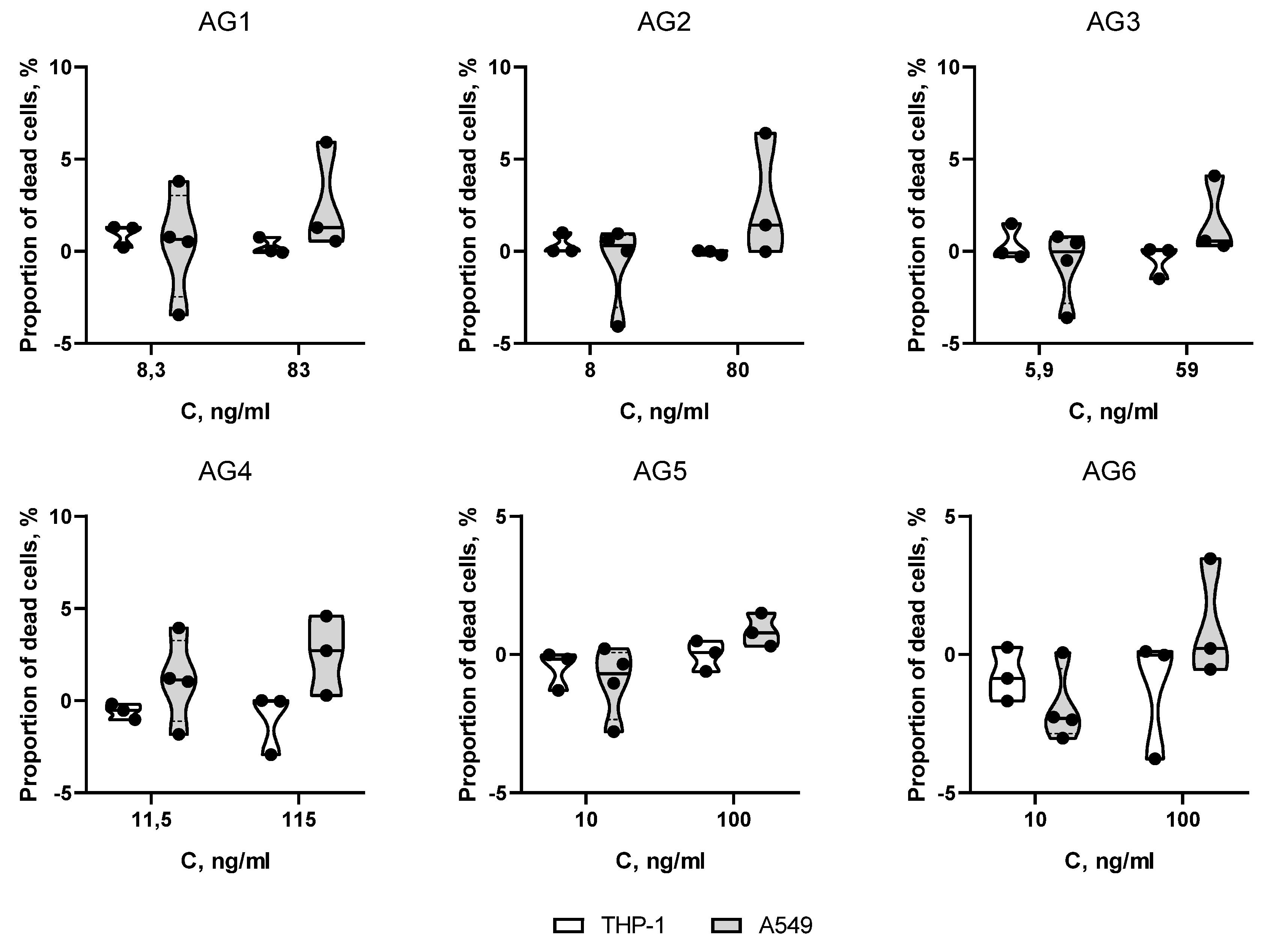
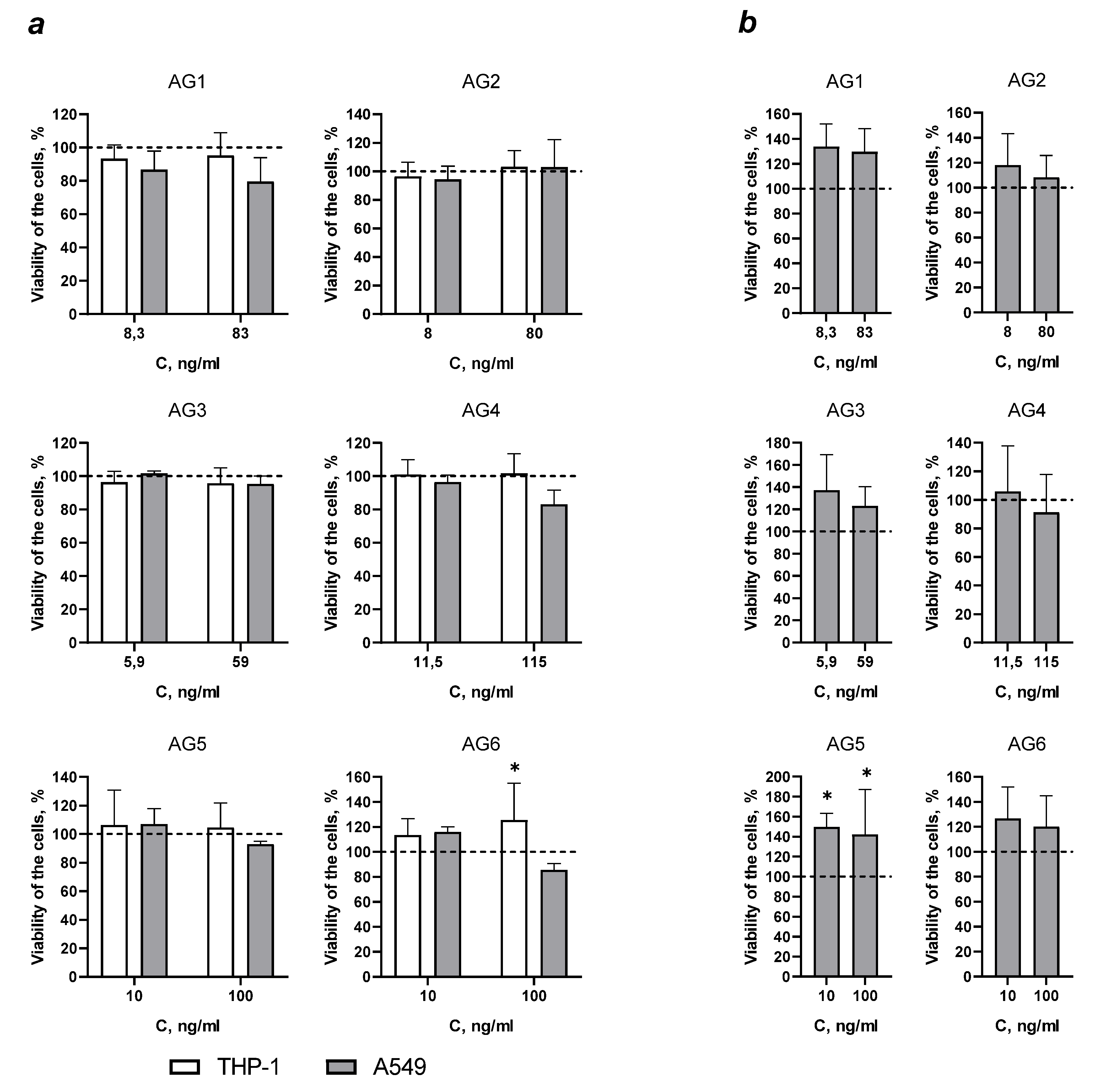
Evaluation of cytotoxicity of mycobacterial antigens
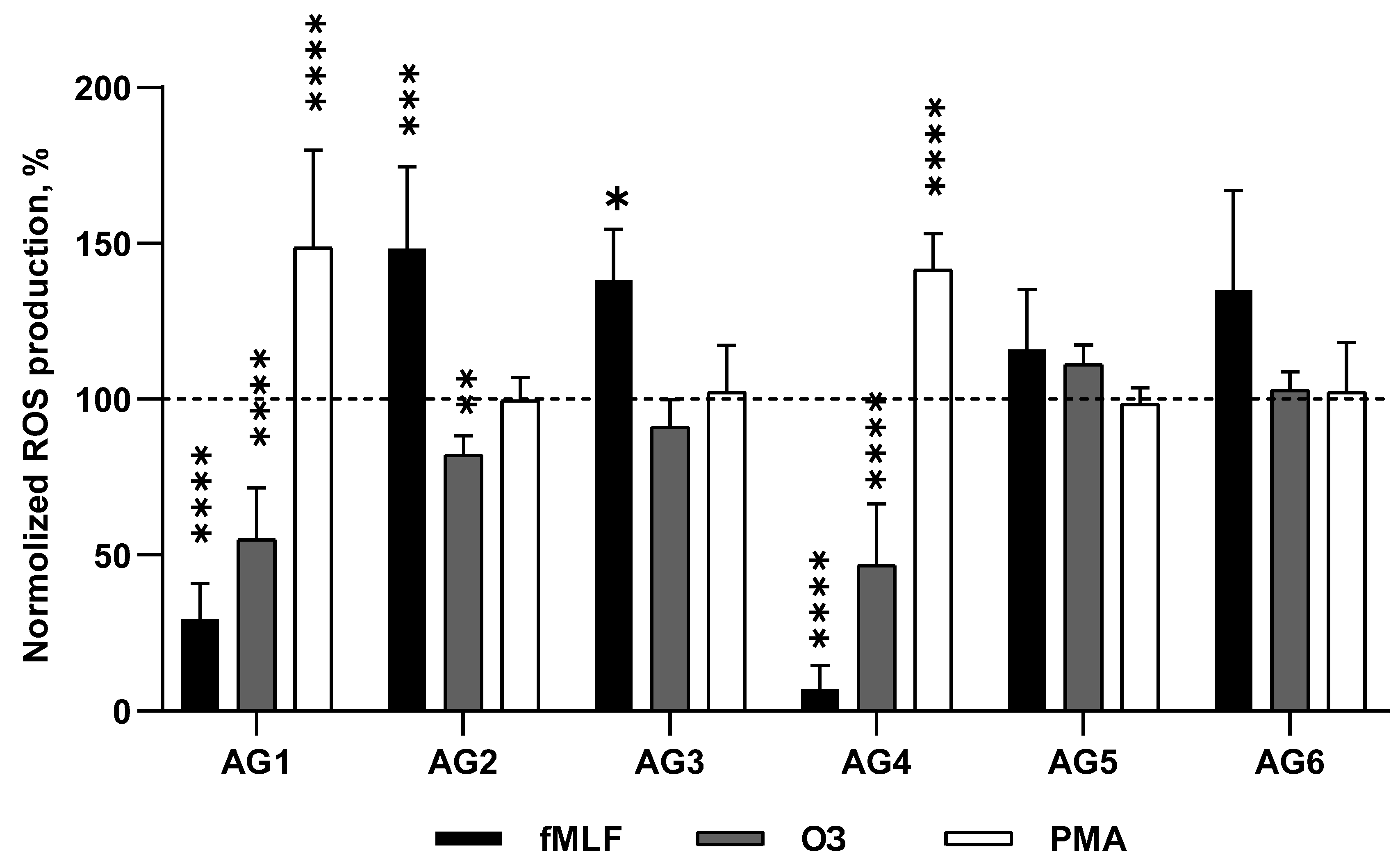
Assessment of ROS production
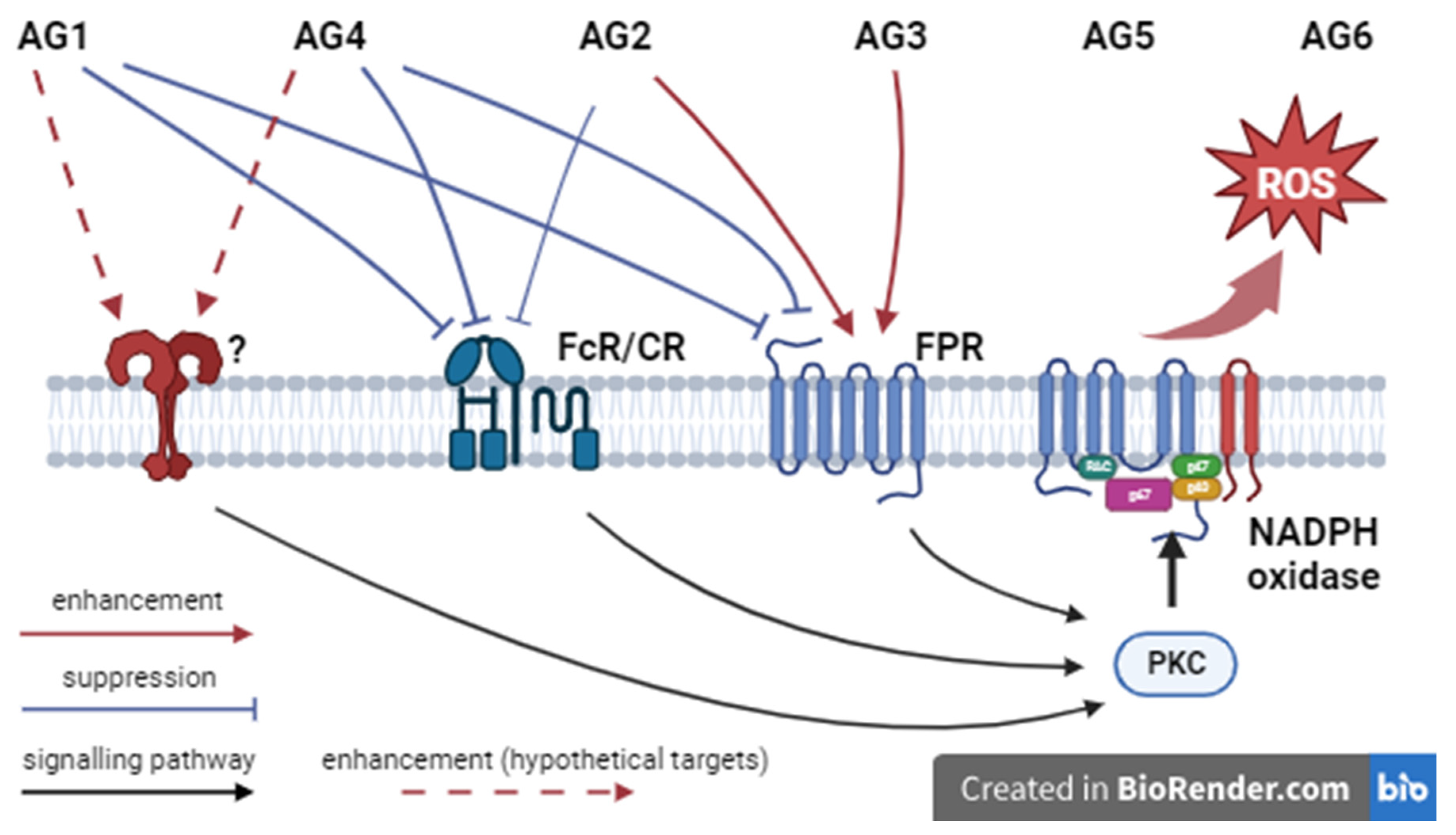
Results
Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Compliance with ethical norms
Abbreviations
References
- Grange, J.M. Mycobacterium bovis infection in human beings. Tuberculosis 2001, 81, 71–77. [Google Scholar] [CrossRef]
- Hlavsa, M.C.; Moonan, P.K.; Cowan, L.S.; Navin, T.R.; Kammerer, J.S.; Morlock, G.P.; Crawford, J.T.; Lobue, P.A. Human tuberculosis due to Mycobacterium bovis in the United States, 1995-2005. Clin Infect Dis 2008, 47, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Torres-Gonzalez, P.; Cervera-Hernandez, M.E.; Martinez-Gamboa, A.; Garcia-Garcia, L.; Cruz-Hervert, L.P.; Bobadilla-Del Valle, M.; Ponce-de Leon, A.; Sifuentes-Osornio, J. Human tuberculosis caused by Mycobacterium bovis: a retrospective comparison with Mycobacterium tuberculosis in a Mexican tertiary care centre, 2000-2015. BMC Infect Dis 2016, 16, 657. [Google Scholar] [CrossRef] [PubMed]
- Alcantara, C.A.; Glassman, I.; Nguyen, K.H.; Parthasarathy, A.; Venketaraman, V. Neutrophils in Mycobacterium tuberculosis. Vaccines 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Borkute, R.R.; Woelke, S.; Pei, G.; Dorhoi, A. Neutrophils in Tuberculosis: Cell Biology, Cellular Networking and Multitasking in Host Defense. International journal of molecular sciences 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Simmons, J.D.; Stein, C.M.; Seshadri, C.; Campo, M.; Alter, G.; Fortune, S.; Schurr, E.; Wallis, R.S.; Churchyard, G.; Mayanja-Kizza, H.; Boom, W.H.; Hawn, T.R. Immunological mechanisms of human resistance to persistent Mycobacterium tuberculosis infection. Nature reviews. Immunology 2018, 18, 575–589. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Newton, S.M.; Wilkinson, K.A.; Kampmann, B.; Hall, B.M.; Nawroly, N.; Packe, G.E.; Davidson, R.N.; Griffiths, C.J.; Wilkinson, R.J. Neutrophil-mediated innate immune resistance to mycobacteria. The Journal of clinical investigation 2007, 117, 1988–1994. [Google Scholar] [CrossRef]
- Ufimtseva, E.; Eremeeva, N.; Bayborodin, S.; Umpeleva, T.; Vakhrusheva, D.; Skornyakov, S. Mycobacterium tuberculosis with different virulence reside within intact phagosomes and inhibit phagolysosomal biogenesis in alveolar macrophages of patients with pulmonary tuberculosis. Tuberculosis 2019, 114, 77–90. [Google Scholar] [CrossRef]
- Hilda, J.N.; Das, S.; Tripathy, S.P.; Hanna, L.E. Role of neutrophils in tuberculosis: A bird's eye view. Innate immunity 2020, 26, 240–247. [Google Scholar] [CrossRef]
- MacMicking, J.D. Cell-autonomous effector mechanisms against mycobacterium tuberculosis. Cold Spring Harbor perspectives in medicine 2014, 4. [Google Scholar] [CrossRef]
- Koeken, V.; Verrall, A.J.; Netea, M.G.; Hill, P.C.; van Crevel, R. Trained innate immunity and resistance to Mycobacterium tuberculosis infection. Clinical microbiology and infection: the official publication of the European Society of Clinical Microbiology and Infectious Diseases 2019, 25, 1468–1472. [Google Scholar] [CrossRef] [PubMed]
- Verrall, A.J.; Netea, M.G.; Alisjahbana, B.; Hill, P.C.; van Crevel, R. Early clearance of Mycobacterium tuberculosis: a new frontier in prevention. Immunology 2014, 141, 506–513. [Google Scholar] [CrossRef]
- Coulter, F.; Parrish, A.; Manning, D.; Kampmann, B.; Mendy, J.; Garand, M.; Lewinsohn, D.M.; Riley, E.M.; Sutherland, J.S. IL-17 Production from T Helper 17, Mucosal-Associated Invariant T, and gammadelta Cells in Tuberculosis Infection and Disease. Frontiers in immunology 2017, 8, 1252. [Google Scholar] [CrossRef] [PubMed]
- Petry, A.; Weitnauer, M.; Gorlach, A. Receptor activation of NADPH oxidases. Antioxid Redox Signal 2010, 13, 467–487. [Google Scholar] [CrossRef]
- Fu, H.; Bylund, J.; Karlsson, A.; Pellme, S.; Dahlgren, C. The mechanism for activation of the neutrophil NADPH-oxidase by the peptides formyl-Met-Leu-Phe and Trp-Lys-Tyr-Met-Val-Met differs from that for interleukin-8. Immunology 2004, 112, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Makni-Maalej, K.; Chiandotto, M.; Hurtado-Nedelec, M.; Bedouhene, S.; Gougerot-Pocidalo, M.A.; Dang, P.M.; El-Benna, J. Zymosan induces NADPH oxidase activation in human neutrophils by inducing the phosphorylation of p47phox and the activation of Rac2: involvement of protein tyrosine kinases, PI3Kinase, PKC, ERK1/2 and p38MAPkinase. Biochemical pharmacology 2013, 85, 92–100. [Google Scholar] [CrossRef]
- Yang, Z.; Zeng, X.; Tsui, S.K. Investigating function roles of hypothetical proteins encoded by the Mycobacterium tuberculosis H37Rv genome. BMC genomics 2019, 20, 394. [Google Scholar] [CrossRef]
- Filina, J.V.; Gabdoulkhakova, A.G.; Safronova, V.G. RhoA/ROCK downregulates FPR2-mediated NADPH oxidase activation in mouse bone marrow granulocytes. Cellular signalling 2014, 26, 2138–2146. [Google Scholar] [CrossRef]
- Filina, Y.; Gabdoulkhakova, A.; Rizvanov, A.; Safronova, V. MAP kinases in regulation of NOX activity stimulated through two types of formyl peptide receptors in murine bone marrow granulocytes. Cellular signalling 2022, 90, 110205. [Google Scholar] [CrossRef] [PubMed]
- Khaertynov, K.S.; Valeeva, A.R.; Ivanov, A.V. Extraction and Serological Properties of Mycobacterium Cell Surface and Excreted Proteins. BioNanoScience 2018, 8, 8. [Google Scholar] [CrossRef]
- Tsuchiya, S.; Yamabe, M.; Yamaguchi, Y.; Kobayashi, Y.; Konno, T.; Tada, K. Establishment and characterization of a human acute monocytic leukemia cell line (THP-1). International journal of cancer 1980, 26, 171–176. [Google Scholar] [CrossRef]
- Giard, D.J.; Aaronson, S.A.; Todaro, G.J.; Arnstein, P.; Kersey, J.H.; Dosik, H.; Parks, W.P. In vitro cultivation of human tumors: establishment of cell lines derived from a series of solid tumors. Journal of the National Cancer Institute 1973, 51, 1417–1423. [Google Scholar] [CrossRef]
- Akhunzianov, A.A.; Nesterova, A.I.; Wanrooij, S.; Filina, Y.V.; Rizvanov, A.A.; Miftakhova, R.R. Unravelling the Therapeutic Potential of Antibiotics in Hypoxia in a Breast Cancer MCF-7 Cell Line Model. International journal of molecular sciences 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Safronova, V.G.; Gabdoulkhakova, A.G.; Santalov, B.F. Immunomodulating action of low intensity millimeter waves on primed neutrophils. Bioelectromagnetics 2002, 23, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Dekker, L.V.; Leitges, M.; Altschuler, G.; Mistry, N.; McDermott, A.; Roes, J.; Segal, A.W. Protein kinase C-beta contributes to NADPH oxidase activation in neutrophils. The Biochemical journal 2000, 347 Pt 1, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.; Thorsen, N.W.; Hallberg, L.A.E.; Hagglund, P.; Hawkins, C.L. New insight into the composition of extracellular traps released by macrophages exposed to different types of inducers. Free radical biology & medicine 2023, 202, 97–109. [Google Scholar] [CrossRef]
- Weng, W.; Hu, Z.; Pan, Y. Macrophage Extracellular Traps: Current Opinions and the State of Research regarding Various Diseases. Journal of immunology research 2022, 2022, 7050807. [Google Scholar] [CrossRef]
- Riazanski, V.; Sui, Z.; Nelson, D.J. Kinetic Separation of Oxidative and Non-oxidative Metabolism in Single Phagosomes from Alveolar Macrophages: Impact on Bacterial Killing. iScience 2020, 23, 101759. [Google Scholar] [CrossRef]
- Lee, P.P.; Chan, K.W.; Jiang, L.; Chen, T.; Li, C.; Lee, T.L.; Mak, P.H.; Fok, S.F.; Yang, X.; Lau, Y.L. Susceptibility to mycobacterial infections in children with X-linked chronic granulomatous disease: a review of 17 patients living in a region endemic for tuberculosis. The Pediatric infectious disease journal 2008, 27, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Khan, T.A.; Cabral-Marques, O.; Schimke, L.F.; de Oliveira, E.B., Jr.; Amaral, E.P.; D'Imperio Lima, M.R.; Scancetti Tavares, F.; Costa Carvalho, B.T.; Condino-Neto, A. Tuberculosis in an autosomal recessive case of chronic granulomatous disease due to mutation of the NCF1 gene. Allergologia et immunopathologia 2016, 44, 276–279. [Google Scholar] [CrossRef]
- Garcia, B.; Leon-Lara, X.; Espinosa, S.; Blancas-Galicia, L. [Mycobacterial disease in patients with chronic granulomatous disease]. Revista alergia Mexico 2021, 68, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Vignesh, P.; Sil, A.; Aggarwal, R.; Laha, W.; Mondal, S.; Dhaliwal, M.; Sharma, S.; Pilania, R.K.; Jindal, A.K.; Suri, D.; Sethi, S.; Rawat, A.; Singh, S. Tuberculosis and Bacillus Calmette-Guerin Disease in Patients with Chronic Granulomatous Disease: an Experience from a Tertiary Care Center in North India. Journal of clinical immunology 2023. [Google Scholar] [CrossRef] [PubMed]
- Deffert, C.; Cachat, J.; Krause, K.H. Phagocyte NADPH oxidase, chronic granulomatous disease and mycobacterial infections. Cellular microbiology 2014, 16, 1168–1178. [Google Scholar] [CrossRef] [PubMed]
- Yaseen, K.H.B. a. I. Mycobacterium tuberculosis: Macrophage Takeover and Modulation of Innate Effector Responses. 2018. [CrossRef]
- Schafer, G.; Jacobs, M.; Wilkinson, R.J.; Brown, G.D. Non-opsonic recognition of Mycobacterium tuberculosis by phagocytes. J Innate Immun 2009, 1, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Faldt, J.; Dahlgren, C.; Karlsson, A.; Ahmed, A.M.; Minnikin, D.E.; Ridell, M. Activation of human neutrophils by mycobacterial phenolic glycolipids. Clin Exp Immunol 1999, 118, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Eum, S.Y.; Kong, J.H.; Hong, M.S.; Lee, Y.J.; Kim, J.H.; Hwang, S.H.; Cho, S.N.; Via, L.E.; Barry, C.E.; 3rd. Neutrophils are the predominant infected phagocytic cells in the airways of patients with active pulmonary TB. Chest 2010, 137, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, A.; Villa, S.; Castelli, V.; Bandera, A.; Gori, A. T-Cell Exhaustion in Mycobacterium tuberculosis and Nontuberculous Mycobacteria Infection: Pathophysiology and Therapeutic Perspectives. Microorganisms 2021, 9. [Google Scholar] [CrossRef]
- Rankin, A.N.; Hendrix, S.V.; Naik, S.K.; Stallings, C.L. Exploring the Role of Low-Density Neutrophils During Mycobacterium tuberculosis Infection. Frontiers in cellular and infection microbiology 2022, 12, 901590. [Google Scholar] [CrossRef]
- Kang, P.B.; Azad, A.K.; Torrelles, J.B.; Kaufman, T.M.; Beharka, A.; Tibesar, E.; DesJardin, L.E.; Schlesinger, L.S. The human macrophage mannose receptor directs Mycobacterium tuberculosis lipoarabinomannan-mediated phagosome biogenesis. J Exp Med 2005, 202, 987–999. [Google Scholar] [CrossRef]
- Neufert, C.; Pai, R.K.; Noss, E.H.; Berger, M.; Boom, W.H.; Harding, C.V. Mycobacterium tuberculosis 19-kDa lipoprotein promotes neutrophil activation. J Immunol 2001, 167, 1542–1549. [Google Scholar] [CrossRef] [PubMed]
- Futosi, K.; Fodor, S.; Mocsai, A. Neutrophil cell surface receptors and their intracellular signal transduction pathways. Int Immunopharmacol 2013, 17, 638–650. [Google Scholar] [CrossRef] [PubMed]
- Mayadas, T.N.; Cullere, X.; Lowell, C.A. The multifaceted functions of neutrophils. Annu Rev Pathol 2014, 9, 181–218. [Google Scholar] [CrossRef] [PubMed]
- Killick, K.E.; Ni Cheallaigh, C.; O'Farrelly, C.; Hokamp, K.; MacHugh, D.E.; Harris, J. Receptor-mediated recognition of mycobacterial pathogens. Cell Microbiol 2013, 15, 1484–1495. [Google Scholar] [CrossRef] [PubMed]
- Master, S.S.; Springer, B.; Sander, P.; Boettger, E.C.; Deretic, V.; Timmins, G.S. Oxidative stress response genes in Mycobacterium tuberculosis: role of ahpC in resistance to peroxynitrite and stage-specific survival in macrophages. Microbiology (Reading) 2002, 148, 3139–3144. [Google Scholar] [CrossRef] [PubMed]
- Manca, C.; Paul, S.; Barry, C.E., 3rd; Freedman, V.H.; Kaplan, G. Mycobacterium tuberculosis catalase and peroxidase activities and resistance to oxidative killing in human monocytes in vitro. Infect Immun 1999, 67, 74–79. [Google Scholar] [CrossRef]
- Su, R.; Peng, Y.P.; Deng, Z.; Deng, Y.T.; Ye, J.Q.; Guo, Y.; Huang, Z.K.; Luo, Q.; Jiang, H.; Li, J.M. Mycobacterium tuberculosis Infection Induces Low-Density Granulocyte Generation by Promoting Neutrophil Extracellular Trap Formation via ROS Pathway. Front Microbiol 2019, 10, 1468. [Google Scholar] [CrossRef]
- Corleis, B.; Korbel, D.; Wilson, R.; Bylund, J.; Chee, R.; Schaible, U.E. Escape of Mycobacterium tuberculosis from oxidative killing by neutrophils. Cell Microbiol 2012, 14, 1109–1121. [Google Scholar] [CrossRef]
- Garcia-Bengoa, M.; Meurer, M.; Goethe, R.; Singh, M.; Reljic, R.; von Kockritz-Blickwede, M. Role of phagocyte extracellular traps during Mycobacterium tuberculosis infections and tuberculosis disease processes. Front Microbiol 2023, 14, 983299. [Google Scholar] [CrossRef]
- Kalsum, S.; Braian, C.; Koeken, V.; Raffetseder, J.; Lindroth, M.; van Crevel, R.; Lerm, M. The Cording Phenotype of Mycobacterium tuberculosis Induces the Formation of Extracellular Traps in Human Macrophages. Front Cell Infect Microbiol 2017, 7, 278. [Google Scholar] [CrossRef]
- Hmama, Z.; Pena-Diaz, S.; Joseph, S.; Av-Gay, Y. Immunoevasion and immunosuppression of the macrophage by Mycobacterium tuberculosis. Immunol Rev 2015, 264, 220–232. [Google Scholar] [CrossRef]
- Sun, J.; Singh, V.; Lau, A.; Stokes, R.W.; Obregon-Henao, A.; Orme, I.M.; Wong, D.; Av-Gay, Y.; Hmama, Z. Mycobacterium tuberculosis nucleoside diphosphate kinase inactivates small GTPases leading to evasion of innate immunity. PLoS Pathog 2013, 9, e1003499. [Google Scholar] [CrossRef] [PubMed]
- Koster, S.; Upadhyay, S.; Chandra, P.; Papavinasasundaram, K.; Yang, G.; Hassan, A.; Grigsby, S.J.; Mittal, E.; Park, H.S.; Jones, V.; Hsu, F.F.; Jackson, M.; Sassetti, C.M.; Philips, J.A. Mycobacterium tuberculosis is protected from NADPH oxidase and LC3-associated phagocytosis by the LCP protein CpsA. Proc Natl Acad Sci U S A 2017, 114, E8711–E8720. [Google Scholar] [CrossRef] [PubMed]
- Pal, R.; Bisht, M.K.; Mukhopadhyay, S. Secretory proteins of Mycobacterium tuberculosis and their roles in modulation of host immune responses: focus on therapeutic targets. FEBS J 2022, 289, 4146–4171. [Google Scholar] [CrossRef] [PubMed]
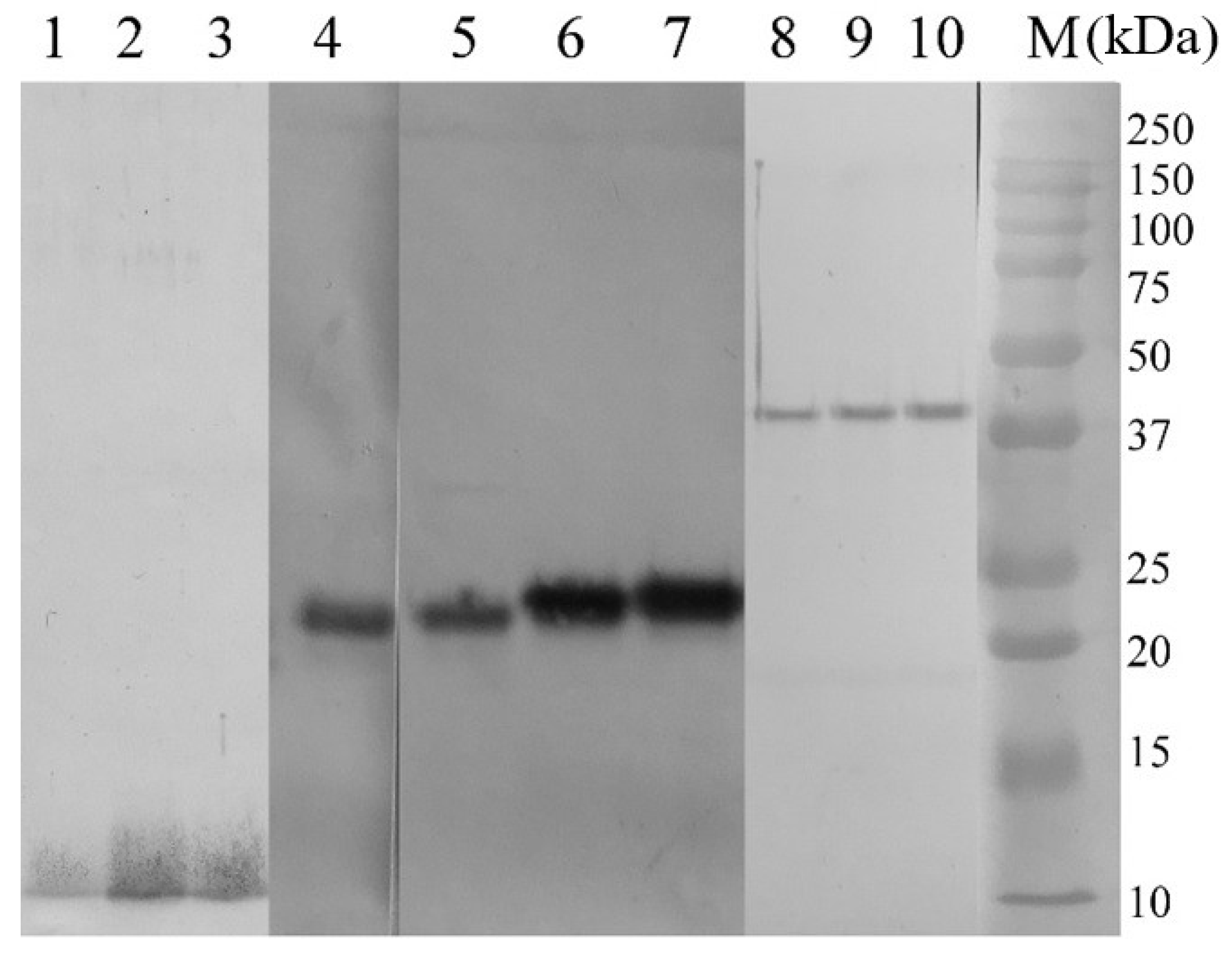
| Antigens | AG1 | AG2 | AG3 | AG4 | AG5 | AG6 |
| Molecular mass (kDa) | 10 | 22 | 24 | 40 | 3 | 3 |
| Availability of functional groups: | ||||||
| Oligosaccharide groups | + | - | - | + | - | - |
| Lipid groups | + | - | - | + | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




