Submitted:
07 November 2023
Posted:
08 November 2023
You are already at the latest version
Abstract
Keywords:
INTRODUCTION
METHODS
Study design and population
Authorizations and information
PET/CT acquisition
Interpretation
Statistical analysis
RESULTS
Population description
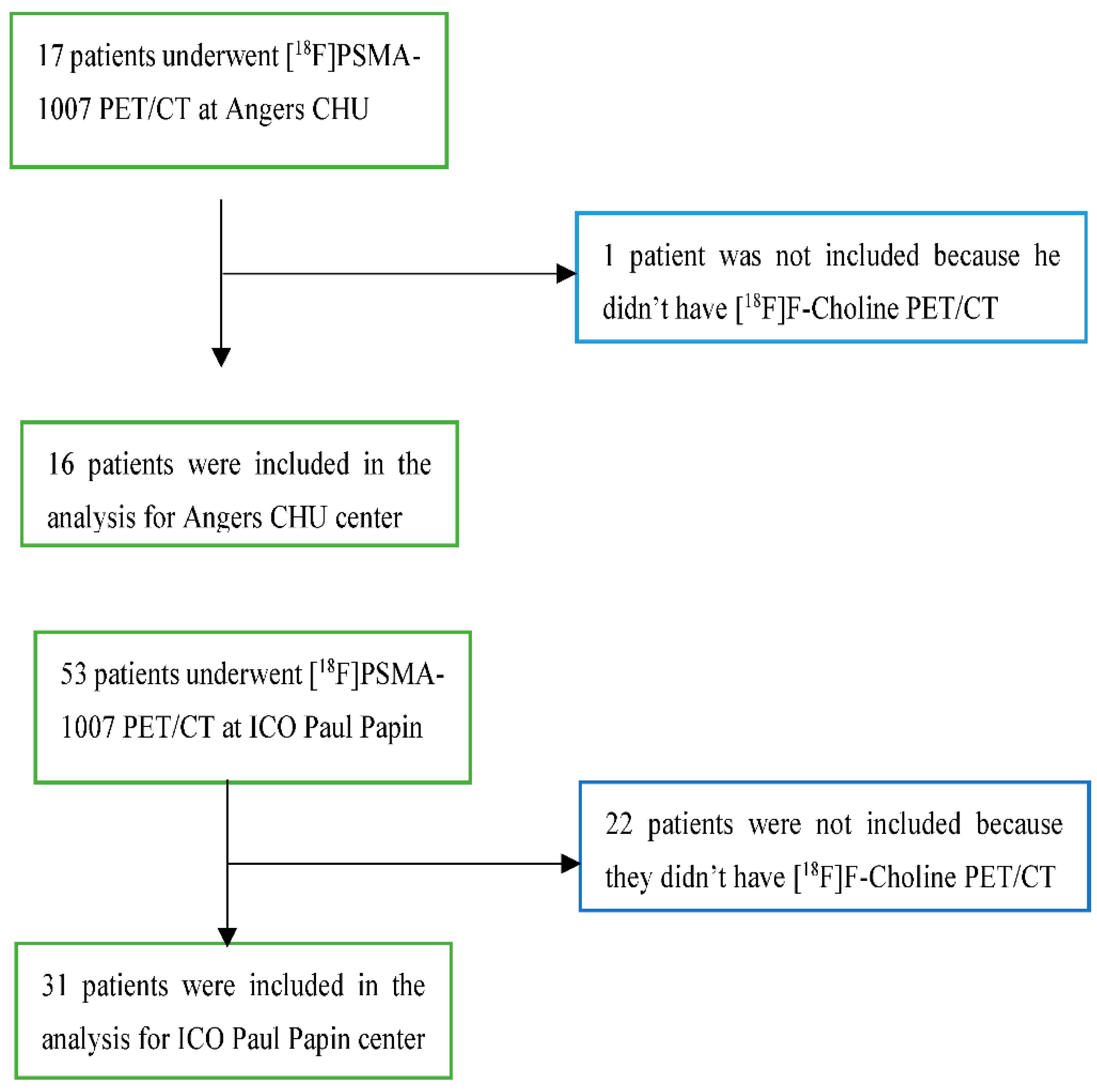
| Total of patients n = 47 | ||
| Age at initial diagnostic – Mean [years] | 62 | |
| Center |
University hospital of Angers ICO Paul Papin |
16 (34%) 31 (66%) |
| Histological type | Adenocarcinoma | 47 (100%) |
| PSA at diagnostic [ng/ml] |
Mean < 10 ng/ml 10-20 ng/ml > 20 ng/ml Missing data |
13.2 24 (51%) 10 (21%) 7 (15%) 6 (13%) |
| ISUP at diagnostic |
1 2 3 4 5 |
5 (11%) 11 (23%) 17 (36%) 10 (21%) 4 (9%) |
| TNM: T |
T1 T2 T3 Doubt between T2 and T3 |
0 (0%) 19 (40%) 24 (51%) 4 (9%) |
| TNM: N |
Nx N0 N1 |
1 (2%) 41 (87%) 5 (11%) |
| TNM: M |
M0 M1 |
46 (98%) 1 (2%) |
| Prostatectomy | 45 (96%) | |
| Scraping |
Total Bilateral ilio-obturator Unilateral ilio-obturator |
31 (66%) 30 (97%) 1 (3%) |
| Initial pelvis radiotherapy | 2 (4%) | |
| Other recurrence before this study | 24 (51%) | |
| Time [years] between initial diagnosis and the recurrence for which [18F]F-Choline was realized |
Mean < 1 year [1-5] years ]5-10] years > 10 years |
7.1 7 (15%) 16 (34%) 14 (30%) 10 (21%) |
| PSA at biological recurrence [ng/ml] |
Mean < 1 ng/ml 1-2 ng/ml > 2 ng/ml |
2.4 14 (30%) 17 (36%) 16 (34%) |
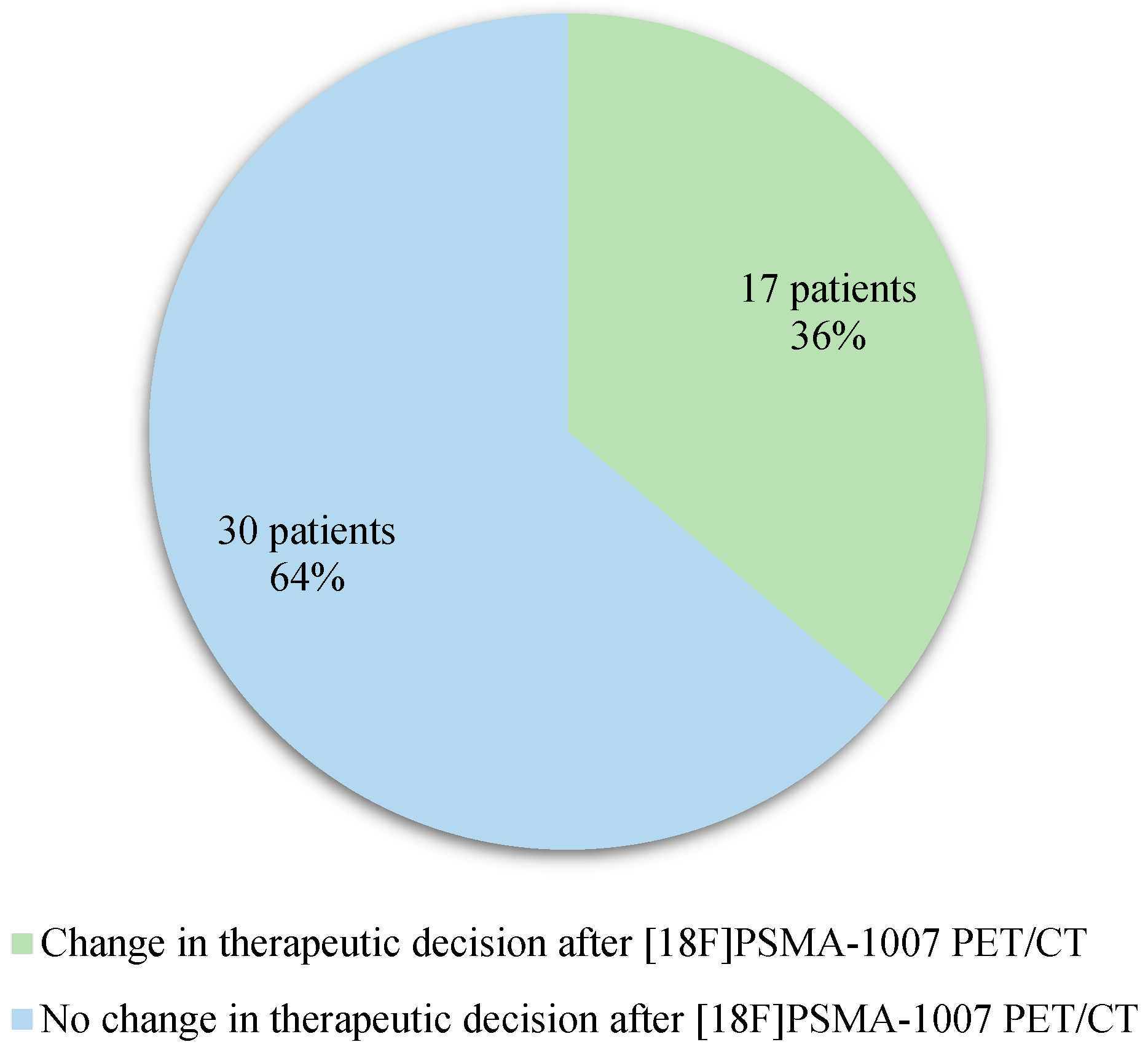
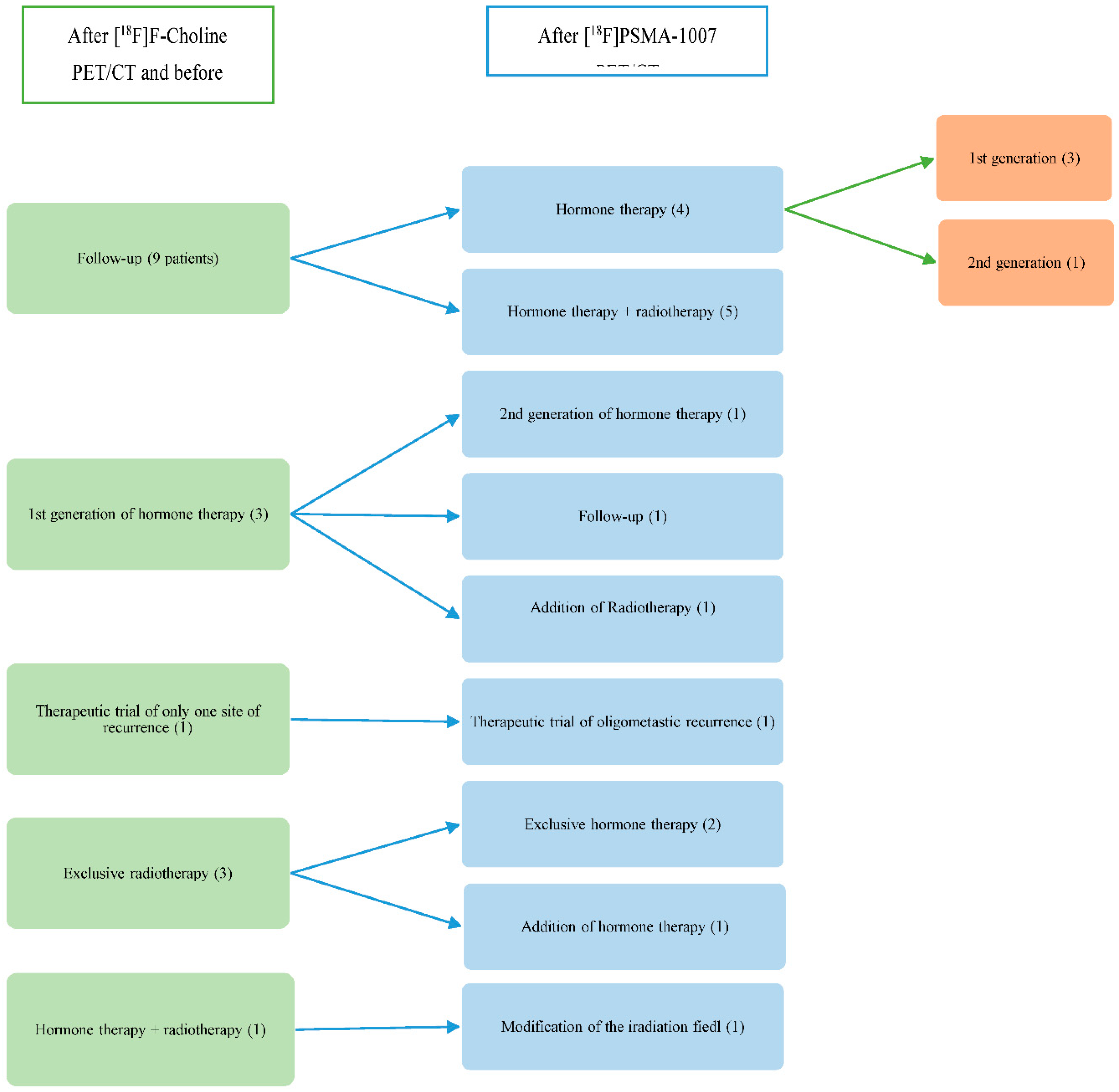
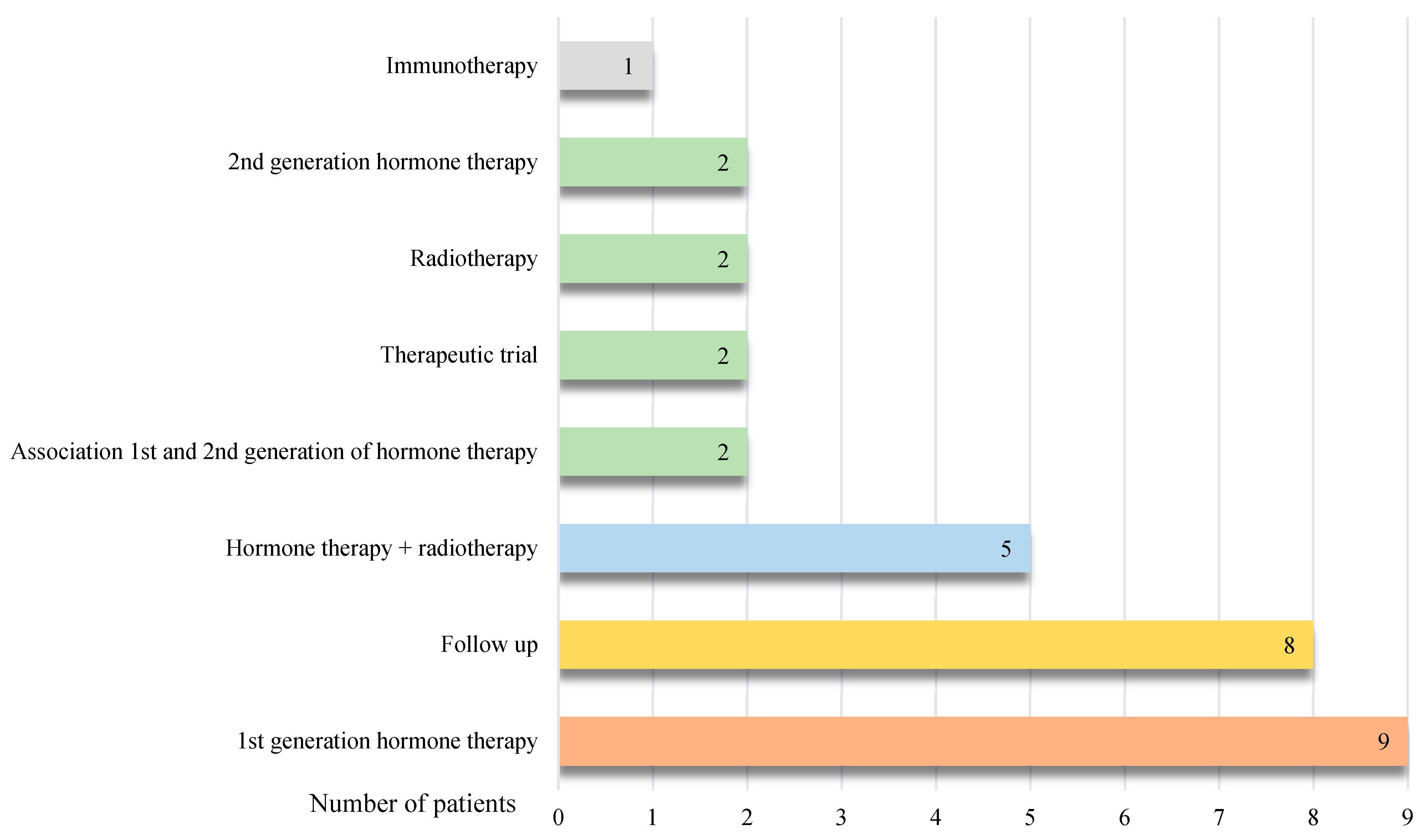
Diagnostic performances of [18F]Fluorocholine PET/CT and [18F]PSMA-1007 PET/CT
| [18F]F-Choline PET/CT | ||||
| Negative | Positive | Doubtful | ||
| [18F]PSMA-1007 PET/CT | Negative | 11 | 0 | 2 |
| Positive | 13 | 13 | 7 | |
| Doubtful | 0 | 0 | 1 | |
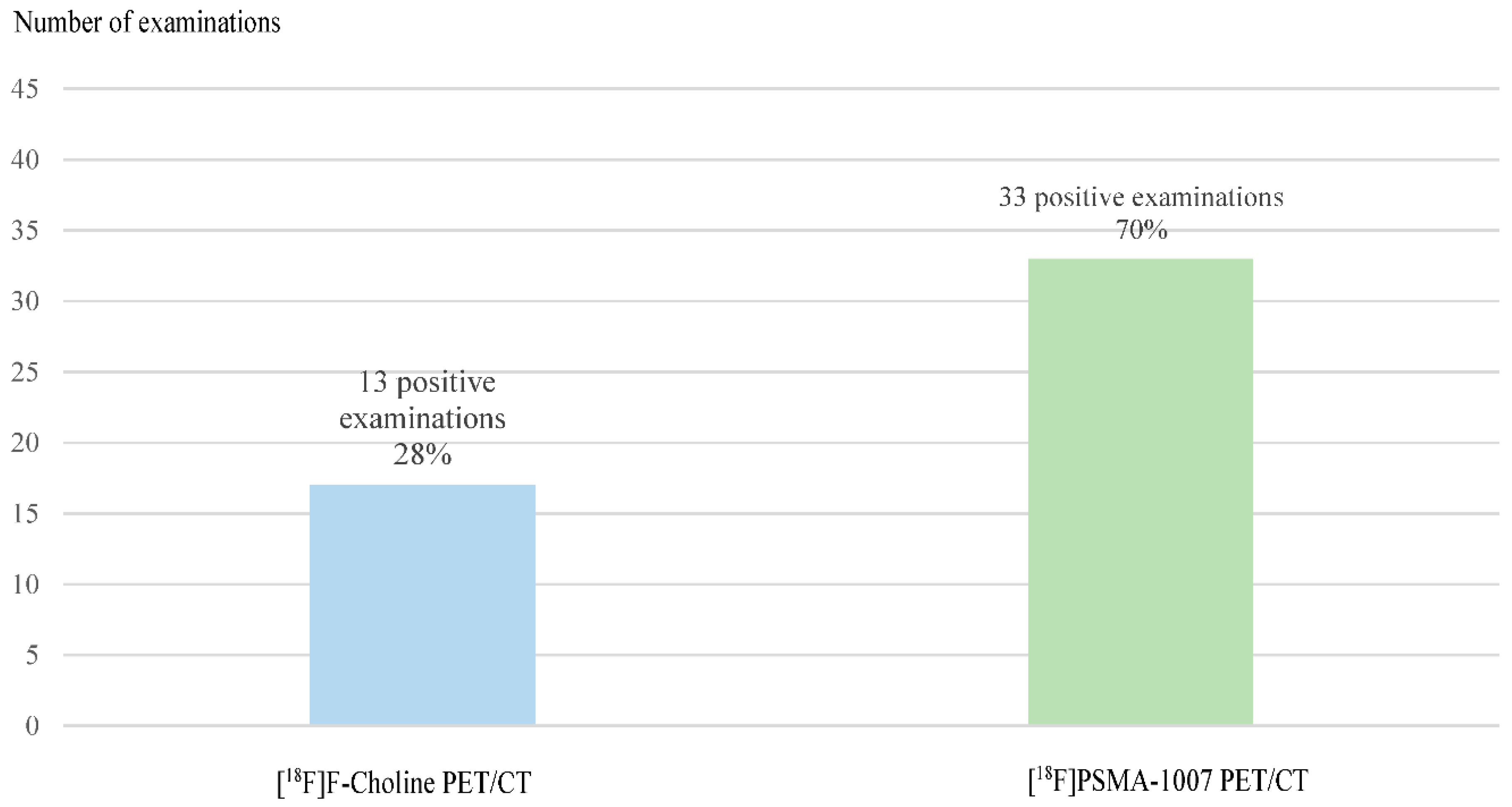
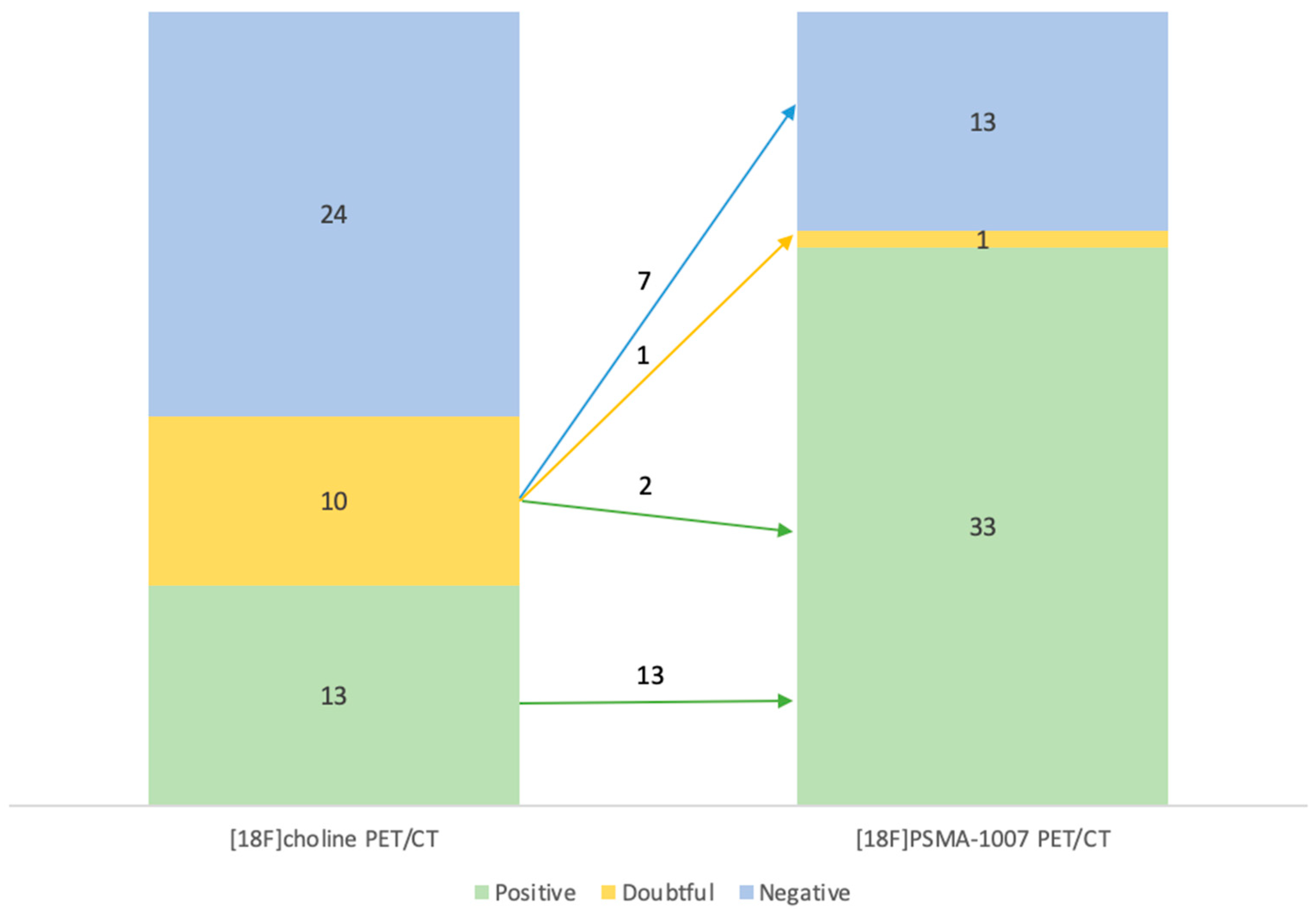
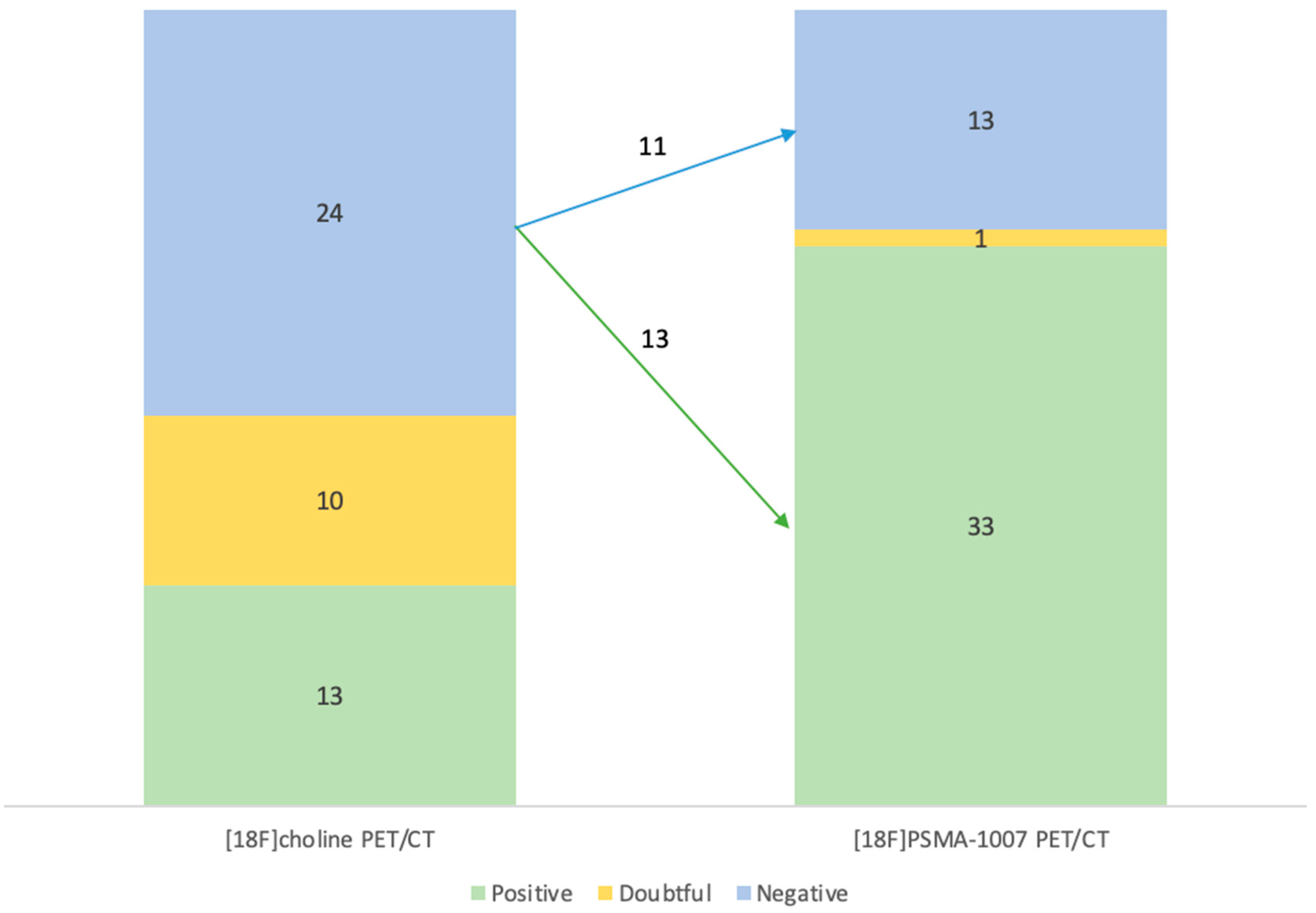
Diagnostic performances by subgroups analysis for PSA levels
| PSA serum levels | ||||
| <1 ng/ml | 1-2 ng/ml | >2 ng/ml | ||
| [18F]F-Choline PET/CT results | Negative | 7 | 11 | 6 |
| Positive | 1 | 4 | 8 | |
| Doubtful | 6 | 2 | 2 | |
| PSA serum levels | ||||
| <1 ng/ml | 1-2 ng/ml | >2 ng/ml | ||
| [18F] PSMA-1007 PET/CT results | Negative | 4 | 7 | 2 |
| Positive | 9 | 10 | 14 | |
| Doubtful | 1 | 0 | 0 | |
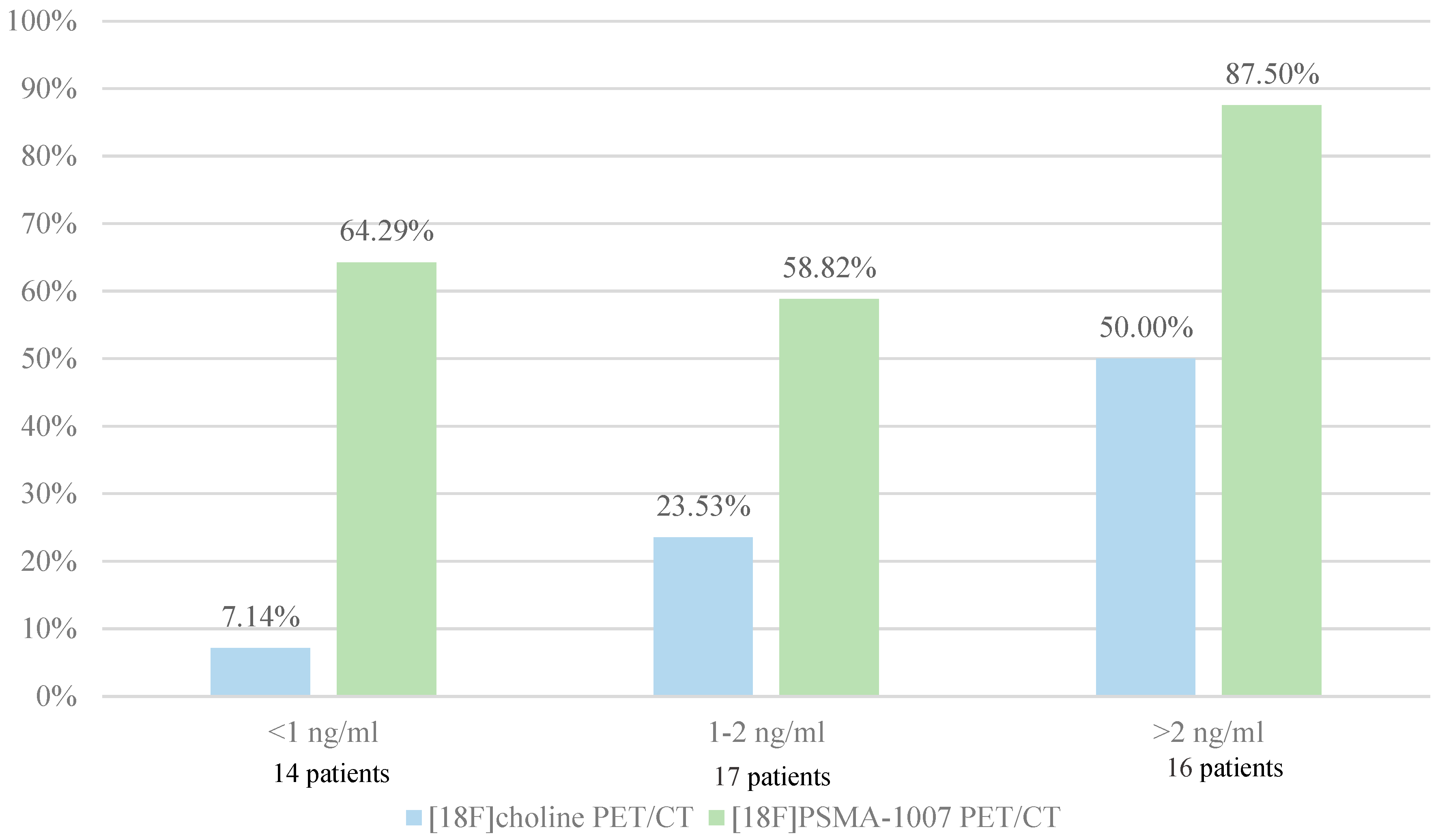
Correlation between PSA levels and SUVmax
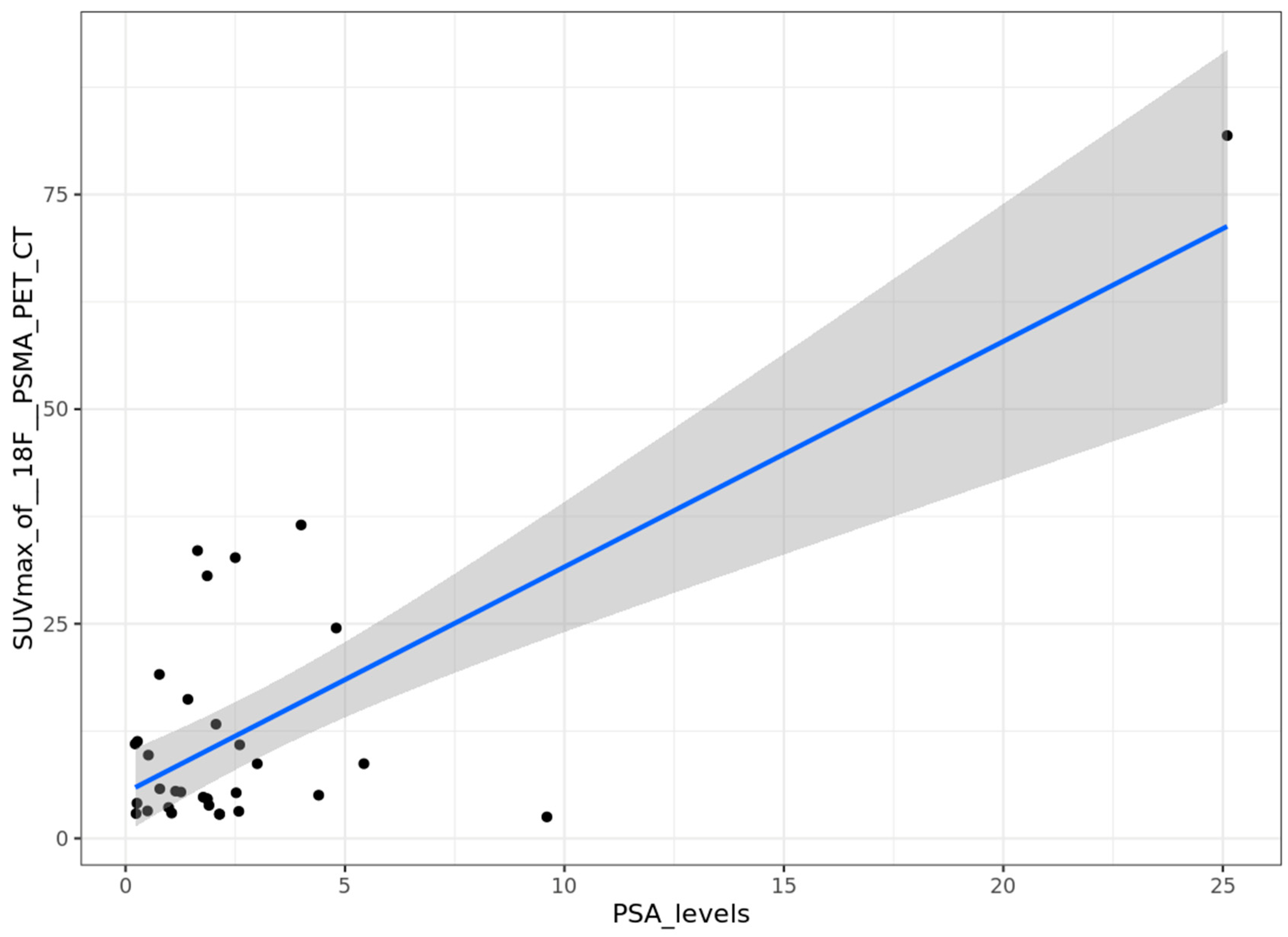
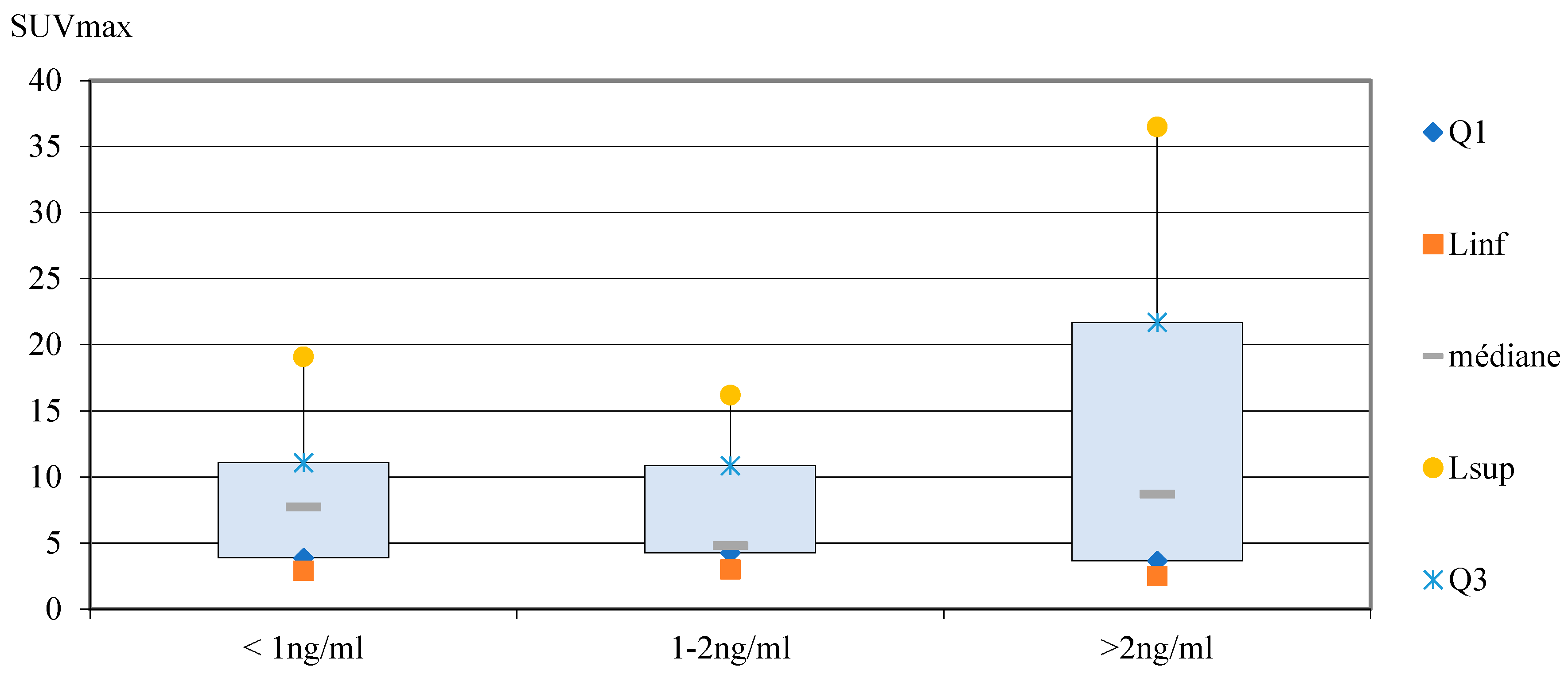
Correlation between ISUP score and SUVmax
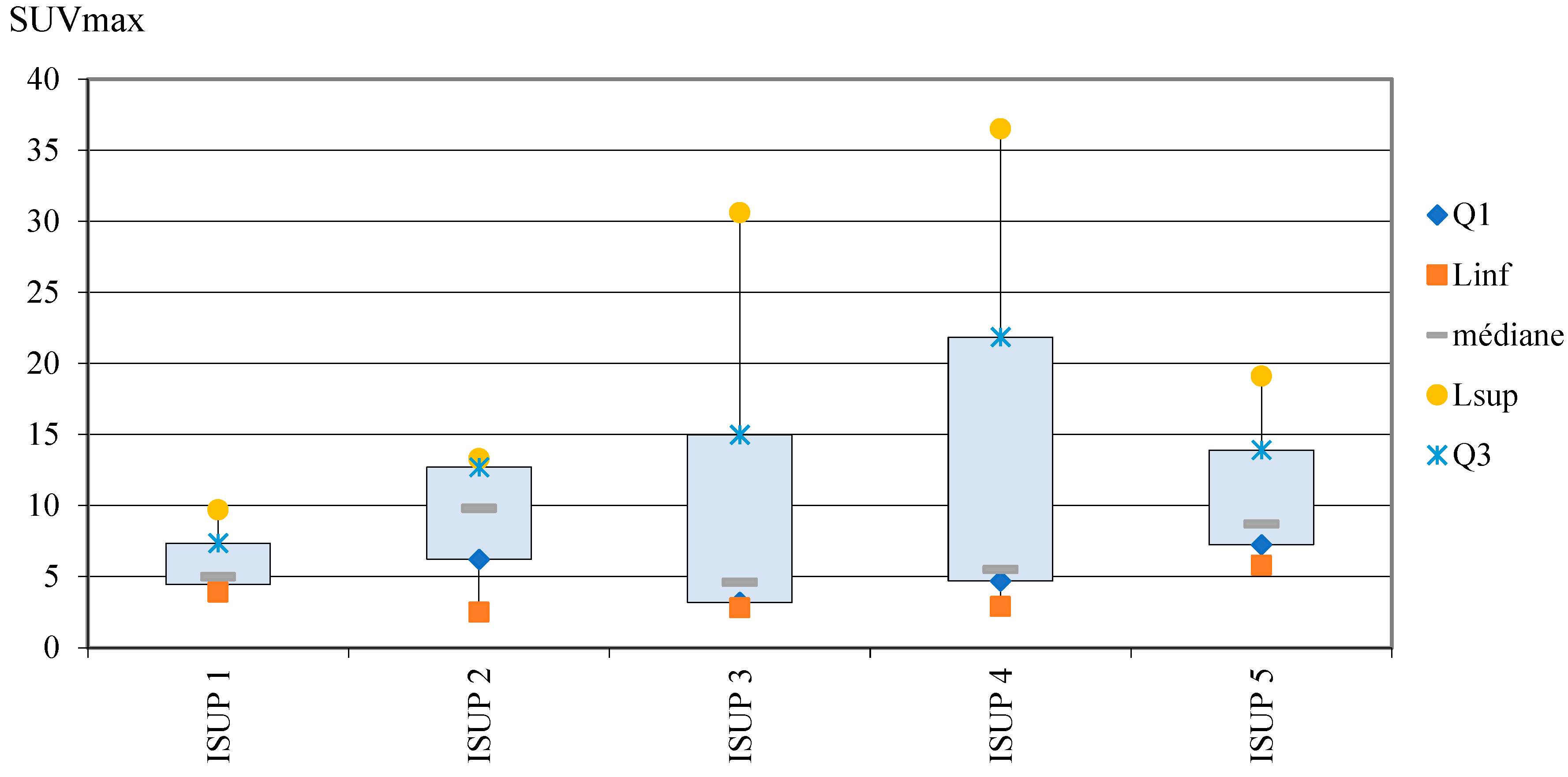
DISCUSSION
CONCLUSION
Funding
Conflicts of Interest
References
- Farolfi A, Calderoni L, Mattana F, Mei R, Telo S, Fanti S, et al. Current and Emerging Clinical Applications of PSMA PET Diagnostic Imaging for Prostate Cancer. J Nucl Med. 2021 May 10;62(5):596–604.
- Beheshti M, Haim S, Zakavi R, Steinmair M, Waldenberger P, Kunit T, et al. Impact of 18F-Choline PET/CT in Prostate Cancer Patients with Biochemical Recurrence: Influence of Androgen Deprivation Therapy and Correlation with PSA Kinetics. J Nucl Med. 2013 Jun 1;54(6):833–40.
- Merriel SWD, Funston G, Hamilton W. Prostate Cancer in Primary Care. Adv Ther. 2018;35(9):1285–94.
- Tsechelidis I, Vrachimis A. PSMA PET in Imaging Prostate Cancer. Front Oncol. 2022 Jan 28;12:831429.
- Wang R, Shen G, Huang M, Tian R. The Diagnostic Role of 18F-Choline, 18F-Fluciclovine and 18F-PSMA PET/CT in the Detection of Prostate Cancer With Biochemical Recurrence: A Meta-Analysis. Front Oncol. 2021 Jun 17;11:684629.
- Rousseau C, Le Thiec M, Ferrer L, Rusu D, Rauscher A, Maucherat B, et al. Preliminary results of a 68Ga-PSMA PET/CT prospective study in prostate cancer patients with occult recurrence: Diagnostic performance and impact on therapeutic decision-making. The Prostate. 2019;79(13):1514–22.
- Lesourd M, Roumiguié M, Beauval JB. Récidive biologique après prostatectomie totale dans le cancer de la prostate : quel bilan et quel traitement en 2019 ? Prog En Urol - FMC. 2019 Mar;29(1):F13–7.
- Ferrari M, Treglia G. 18F-PSMA-1007 PET in Biochemical Recurrent Prostate Cancer: An Updated Meta-Analysis. Contrast Media Mol Imaging. 2021 Dec 18;2021:3502389.
- von Eyben FE, Picchio M, von Eyben R, Rhee H, Bauman G. 68Ga-Labeled Prostate-specific Membrane Antigen Ligand Positron Emission Tomography/Computed Tomography for Prostate Cancer: A Systematic Review and Meta-analysis. Eur Urol Focus. 2018 Sep 1;4(5):686–93.
- Fraum TJ, Ludwig DR, Kim EH, Schroeder P, Hope TA, Ippolito JE. Prostate cancer PET tracers: essentials for the urologist. Can J Urol. 2018;13.
- Ceci F, Oprea-Lager DE, Emmett L, Adam JA, Bomanji J, Czernin J, et al. E-PSMA: the EANM standardized reporting guidelines v1.0 for PSMA-PET. Eur J Nucl Med Mol Imaging. 2021;48(5):1626–38.
- Crocerossa F, Marchioni M, Novara G, Carbonara U, Ferro M, Russo GI, et al. Detection Rate of Prostate Specific Membrane Antigen Tracers for Positron Emission Tomography/Computerized Tomography in Prostate Cancer Biochemical Recurrence: A Systematic Review and Network Meta-Analysis. J Urol. 2021 Feb;205(2):356–69.
- Giesel FL, Hadaschik B, Cardinale J, Radtke J, Vinsensia M, Lehnert W, et al. F-18 labelled PSMA-1007: biodistribution, radiation dosimetry and histopathological validation of tumor lesions in prostate cancer patients. Eur J Nucl Med Mol Imaging. 2017 Apr 1;44(4):678–88.
- Witkowska-Patena E, Giżewska A, Dziuk M, Miśko J, Budzyńska A, Walęcka-Mazur A. Head-to-Head Comparison of 18F-Prostate-Specific Membrane Antigen-1007 and 18F-Fluorocholine PET/CT in Biochemically Relapsed Prostate Cancer. Clin Nucl Med. 2019 Dec;44(12):e629–33.
- Hoberück S, Löck S, Borkowetz A, Sommer U, Winzer R, Zöphel K, et al. Intraindividual comparison of [68 Ga]-Ga-PSMA-11 and [18F]-F-PSMA-1007 in prostate cancer patients: a retrospective single-center analysis. EJNMMI Res. 2021 Oct 19;11(1):109.
- Kesch C, Kratochwil C, Mier W, Kopka K, Giesel FL. 68Ga or 18F for Prostate Cancer Imaging? J Nucl Med. 2017 May 1;58(5):687–8.
- Lawhn-Heath C, Salavati A, Behr SC, Rowe SP, Calais J, Fendler WP, et al. Prostate-specific Membrane Antigen PET in Prostate Cancer. Radiology. 2021 May 1;299(2):248–60.
- Lengana T, Lawal I, Rensburg CJV, Mokoala K, Moshokoa E, Mazibuko S, et al. The Diagnostic Performance of 18F-PSMA-1007 PET/CT in Prostate Cancer Patients with Early Recurrence after Definitive Therapy with a PSA <10 ng/ml. Nukl - Nucl. 2022 Apr;61(02):120–9.
- Virgolini I, Decristoforo C, Haug A, Fanti S, Uprimny C. Current status of theranostics in prostate cancer. Eur J Nucl Med Mol Imaging. 2018;45(3):471–95.
- Advantage of 18F-PSMA-1007 over 68Ga-PSMA-11 PET imaging for differentiation of local recurrence vs. urinary tracer excretion - ProQuest [Internet]. [cited 2021 Nov 24]. Available from: https://www.proquest.com/openview/fc76f63b2f6634fb9a16f3ff8b9fdcb6/1?pq-origsite=gscholar&cbl=42802.
- Krausewitz P, Ritter M. [Clinical aspects in the diagnosis and treatment of prostate cancer]. Radiol. 2021 Sep;61(9):795–801.
- Mena E, Lindenberg L, Choyke P. The Impact of PSMA PET/CT Imaging in Prostate Cancer Radiation Treatment. Semin Nucl Med. 2022 Mar 1;52(2):255–62.
- Udovicich C, Perera M, Hofman MS, Siva S, Del Rio A, Murphy DG, et al. 68Ga-prostate-specific membrane antigen-positron emission tomography/computed tomography in advanced prostate cancer: Current state and future trends. Prostate Int. 2017 Dec;5(4):125–9.
- Eissa A, Elsherbiny A, Coelho RF, Rassweiler J, Davis JW, Porpiglia F, et al. The role of 68Ga-PSMA PET/CT scan in biochemical recurrence after primary treatment for prostate cancer: a systematic review of the literature. Minerva Urol Nefrol [Internet]. 2018 Sep [cited 2021 Nov 25];70(5). Available from: https://www.minervamedica.it/index2.php?show=R19Y2018N05A0462.
- Calais J, Fendler WP, Eiber M, Gartmann J, Chu FI, Nickols NG, et al. Impact of 68Ga-PSMA-11 PET/CT on the Management of Prostate Cancer Patients with Biochemical Recurrence. J Nucl Med. 2018 Mar 1;59(3):434–41.
- Barbaud M, Frindel M, Ferrer L, Thiec ML, Rusu D, Rauscher A, et al. 68Ga-PSMA-11 PET-CT study in prostate cancer patients with biochemical recurrence and non-contributive 18F-Choline PET-CT: Impact on therapeutic decision-making and biomarker changes. The Prostate. 2019 Apr 1;79(5):454–61.
- Young S, Liu W, Zukotynski K, Bauman G. Prostate-specific membrane antigen targeted PET/CT for recurrent prostate cancer: a clinician’s guide. Expert Rev Anticancer Ther. 2021 Jun 3;21(6):641–55.
- Francolini G, Detti B, Bottero M, Zilli T, Lancia A, Bruni A, et al. Detection rate, pattern of relapse and influence on therapeutic decision of PSMA PET/CT in patients affected by biochemical recurrence after radical prostatectomy, a retrospective case series. Clin Transl Oncol. 2021 Feb 1;23(2):364–71.
- Cerci JJ, Fanti S, Lobato EE, Kunikowska J, Alonso O, Medina S, et al. Diagnostic Performance and Clinical Impact of 68Ga-PSMA-11 PET/CT Imaging in Early Relapsed Prostate Cancer After Radical Therapy: A Prospective Multicenter Study (IAEA-PSMA Study). J Nucl Med. 2022 Feb 1;63(2):240–7.
- Lasserre M, Sargos P, Barret E, Beauval JB, Brureau L, Créhange G, et al. Narrative review of PET/CT performances at biochemical recurrence in prostate cancer after radical prostatectomy and impact on patient disease management: Revue narrative à propos des performances de la TEP/TDM en cas de récidive biochimique après prostatectomie radicale dans le cancer de la prostate et impact sur la prise en charge des patients. Prog En Urol. 2022 Jun 1;32(6, Supplement 1):6S33–42.
- Murthy V, Aggarwal R, Koo PJ. The Emerging Role of Next-Generation Imaging in Prostate Cancer. Curr Oncol Rep. 2022 Jan 1;24(1):33–42.
- Karagiannis V, Wichmann V, Saarinen J, Eigeliene N, Andersen H, Jekunen A. Radiotherapy treatment modification for prostate cancer patients based on PSMA-PET/CT. Radiat Oncol Lond Engl. 2022 Jan 29;17:19.
- Artigas C, Diamand R, Shagera QA, Plouznikoff N, Fokoue F, Otte FX, et al. Oligometastatic Disease Detection with 68Ga-PSMA-11 PET/CT in Hormone-Sensitive Prostate Cancer Patients (HSPC) with Biochemical Recurrence after Radical Prostatectomy: Predictive Factors and Clinical Impact. Cancers. 2021 Oct 4;13(19):4982.
- Kaalep A, Sera T, Oyen W, Krause BJ, Chiti A, Liu Y, et al. EANM/EARL FDG-PET/CT accreditation - summary results from the first 200 accredited imaging systems. Eur J Nucl Med Mol Imaging. 2018 Mar 1;45(3):412–22.
- Roberts MJ, Morton A, Donato P, Kyle S, Pattison DA, Thomas P, et al. 68Ga-PSMA PET/CT tumour intensity pre-operatively predicts adverse pathological outcomes and progression-free survival in localised prostate cancer. Eur J Nucl Med Mol Imaging. 2021 Feb 1;48(2):477–82.
- Han S, Woo S, Kim YJ, Suh CH. Impact of 68Ga-PSMA PET on the Management of Patients with Prostate Cancer: A Systematic Review and Meta-analysis. Eur Urol. 2018 Aug 1;74(2):179–90.
- Gauthé M, Belissant O, Girard A, Zhang Yin J, Ohnona J, Cottereau AS, et al. [PET/CT and biochemical recurrence of prostate adenocarcinoma: Added value of 68Ga-PSMA-11 when 18F-fluorocholine is non-contributive]. Progres En Urol J Assoc Francaise Urol Soc Francaise Urol. 2017 Jul;27(8–9):474–81.
- Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG, et al. Comparison of PET imaging with a 68Ga-labelled PSMA ligand and 18F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2014 Jan 1;41(1):11–20.
- Eiber M, Maurer T, Souvatzoglou M, Beer AJ, Ruffani A, Haller B, et al. Evaluation of Hybrid 68 Ga-PSMA Ligand PET/CT in 248 Patients with Biochemical Recurrence After Radical Prostatectomy. J Nucl Med. 2015 May;56(5):668–74.
- Uprimny C, Kroiss AS, Decristoforo C, Fritz J, von Guggenberg E, Kendler D, et al. 68Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur J Nucl Med Mol Imaging. 2017 Jun 1;44(6):941–9.
- Demirci E, Kabasakal L, Şahin OE, Akgün E, Gültekin MH, Doğanca T, et al. Can SUVmax values of Ga-68-PSMA PET/CT scan predict the clinically significant prostate cancer? Nucl Med Commun. 2019 Jan;40(1):86–91.
- Jiao J, Kang F, Zhang J, Quan Z, Wen W, Zhao X, et al. Establishment and prospective validation of an SUVmax cutoff value to discriminate clinically significant prostate cancer from benign prostate diseases in patients with suspected prostate cancer by 68Ga-PSMA PET/CT: a real-world study. Theranostics. 2021 Jul 25;11(17):8396–411.
- Rahbar K, Weckesser M, Huss S, Semjonow A, Breyholz HJ, Schrader AJ, et al. Correlation of Intraprostatic Tumor Extent with 68Ga-PSMA Distribution in Patients with Prostate Cancer. J Nucl Med. 2016 Apr 1;57(4):563–7.
- Hoffmann MA, Wieler HJ, Baues C, Kuntz NJ, Richardsen I, Schreckenberger M. The Impact of 68Ga-PSMA PET/CT and PET/MRI on the Management of Prostate Cancer. Urology. 2019 Aug 1;130:1–12.
- Verburg FA, Pfister D, Drude NI, Mottaghy FM, Behrendt FF. PSA levels, PSA doubling time, Gleason score and prior therapy cannot predict measured uptake of [68Ga]PSMA-HBED-CC lesion uptake in recurrent/metastatic prostate cancer. Nukl - Nucl. 2017 Nov;56(06):225–32.
- Liu X, Wang Q, Zhang B, Jiang T, Zeng W. Diagnostic accuracy of 18F-PSMA-1007 PET/CT for prostate cancer in primary staging and biochemical recurrence with different serum PSA levels: A systematic review and meta-analysis. Hell J Nucl Med. 2022;25(1):88–102.
- Barbosa F de G, Queiroz MA, Nunes RF, Marin JFG, Buchpiguel CA, Cerri GG. Clinical perspectives of PSMA PET/MRI for prostate cancer. Clinics. 2018;73(Suppl 1):e586s.
- Emmett L, Papa N, Buteau J, Ho B, Liu V, Roberts M, et al. The PRIMARY Score: Using Intraprostatic 68Ga-PSMA PET/CT Patterns to Optimize Prostate Cancer Diagnosis. J Nucl Med. 2022 Nov 1;63(11):1644–50.
- Schlemmer HP, Joachim Krause B, Schütz V, Bonekamp D, Marie Schwarzenböck S, Hohenfellner M. Imaging of Prostate Cancer. Dtsch Ärztebl Int. 2021 Oct;118(42):713–9.
- MRI in the Management of Prostate Cancer - ClinicalKey [Internet]. [cited 2023 Feb 25]. Available from: https://www.clinicalkey.fr/#!/content/playContent/1-s2.0-S0887217120300305?scrollTo=%23hl0000264.
- Abecassis JP, Ghazzar N, Peyromaure M, Giraud P. Prostate imaging: Contribution of PET PSMA and MRI. Cancer/Radiothérapie. 2020 Aug 1;24(5):423–8.
- Evangelista L, Zattoni F, Cassarino G, Artioli P, Cecchin D, dal Moro F, et al. PET/MRI in prostate cancer: a systematic review and meta-analysis. Eur J Nucl Med Mol Imaging. 2021 Mar 1;48(3):859–73.
- Eiber M, Weirich G, Holzapfel K, Souvatzoglou M, Haller B, Rauscher I, et al. Simultaneous 68Ga-PSMA HBED-CC PET/MRI Improves the Localization of Primary Prostate Cancer. Eur Urol. 2016 Nov 1;70(5):829–36.
- Afshar-Oromieh A, Haberkorn U, Schlemmer HP, Fenchel M, Eder M, Eisenhut M, et al. Comparison of PET/CT and PET/MRI hybrid systems using a 68Ga-labelled PSMA ligand for the diagnosis of recurrent prostate cancer: initial experience. Eur J Nucl Med Mol Imaging. 2014 May 1;41(5):887–97.
- Zamboglou C, Wieser G, Hennies S, Rempel I, Kirste S, Soschynski M, et al. MRI versus 68Ga-PSMA PET/CT for gross tumour volume delineation in radiation treatment planning of primary prostate cancer. Eur J Nucl Med Mol Imaging. 2016 May 1;43(5):889–97.
- Zamboglou C, Thomann B, Koubar K, Bronsert P, Krauss T, Rischke HC, et al. Focal dose escalation for prostate cancer using 68Ga-HBED-CC PSMA PET/CT and MRI: a planning study based on histology reference. Radiat Oncol. 2018 May 2;13(1):81.
- Evaluation of intensity modulated radiation therapy dose painting for localized prostate cancer using 68 Ga-HBED-CC PSMA-PET/CT: A planning study based on histopathology reference - ClinicalKey [Internet]. [cited 2023 Feb 25]. Available from: https://www.clinicalkey.fr/#!/content/playContent/1-s2.0-S0167814017303481?returnurl=https:%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS0167814017303481%3Fshowall%3Dtrue&referrer=https:%2F%2Fpubmed.ncbi.nlm.nih.gov%2F.
- Eiber M, Herrmann K, Calais J, Hadaschik B, Giesel FL, Hartenbach M, et al. Prostate Cancer Molecular Imaging Standardized Evaluation (PROMISE): Proposed miTNM Classification for the Interpretation of PSMA-Ligand PET/CT. J Nucl Med. 2018 Mar 1;59(3):469–78.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




