Submitted:
03 November 2023
Posted:
07 November 2023
You are already at the latest version
Abstract
Keywords:
Background
Methods
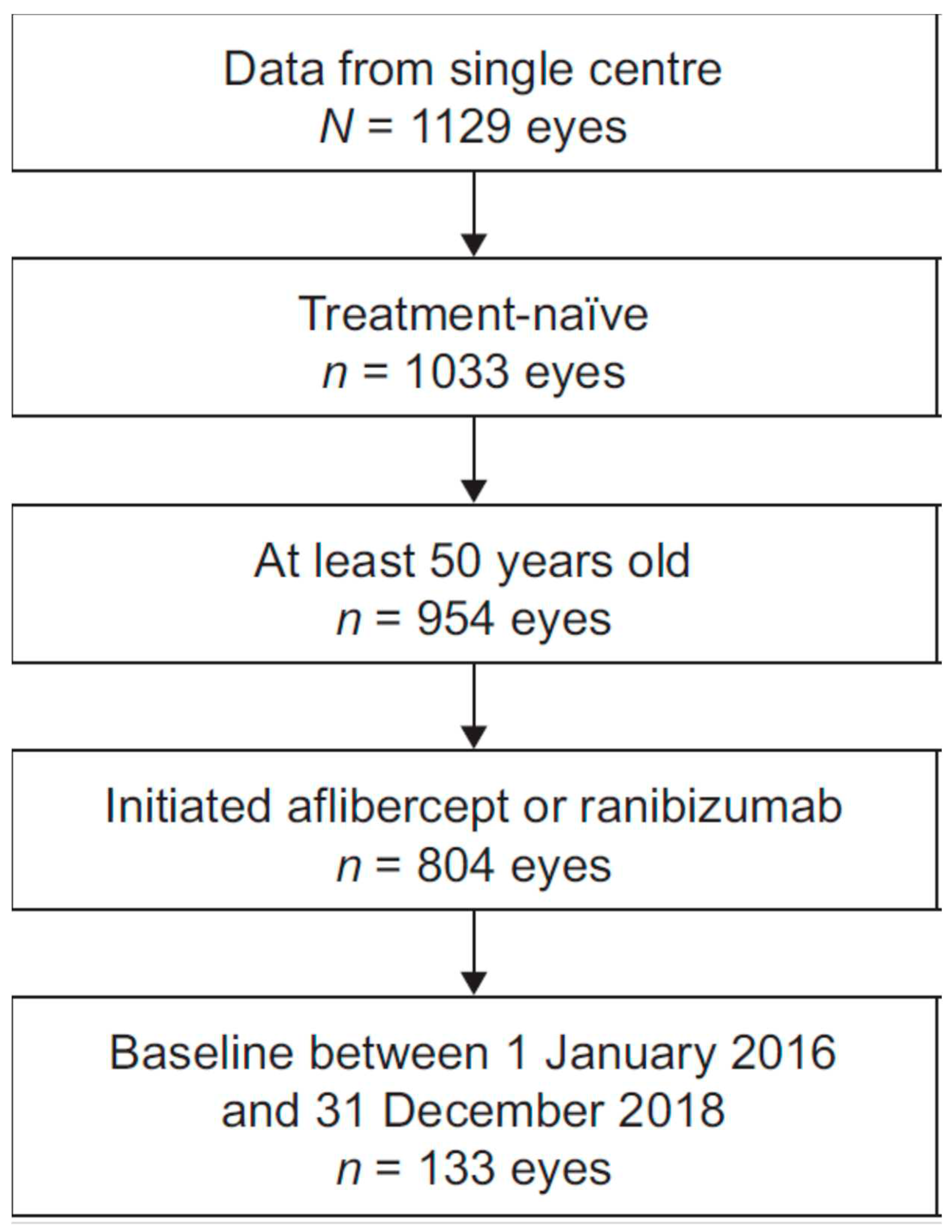
Patients and procedures
Study objectives and endpoints
Statistical analyses
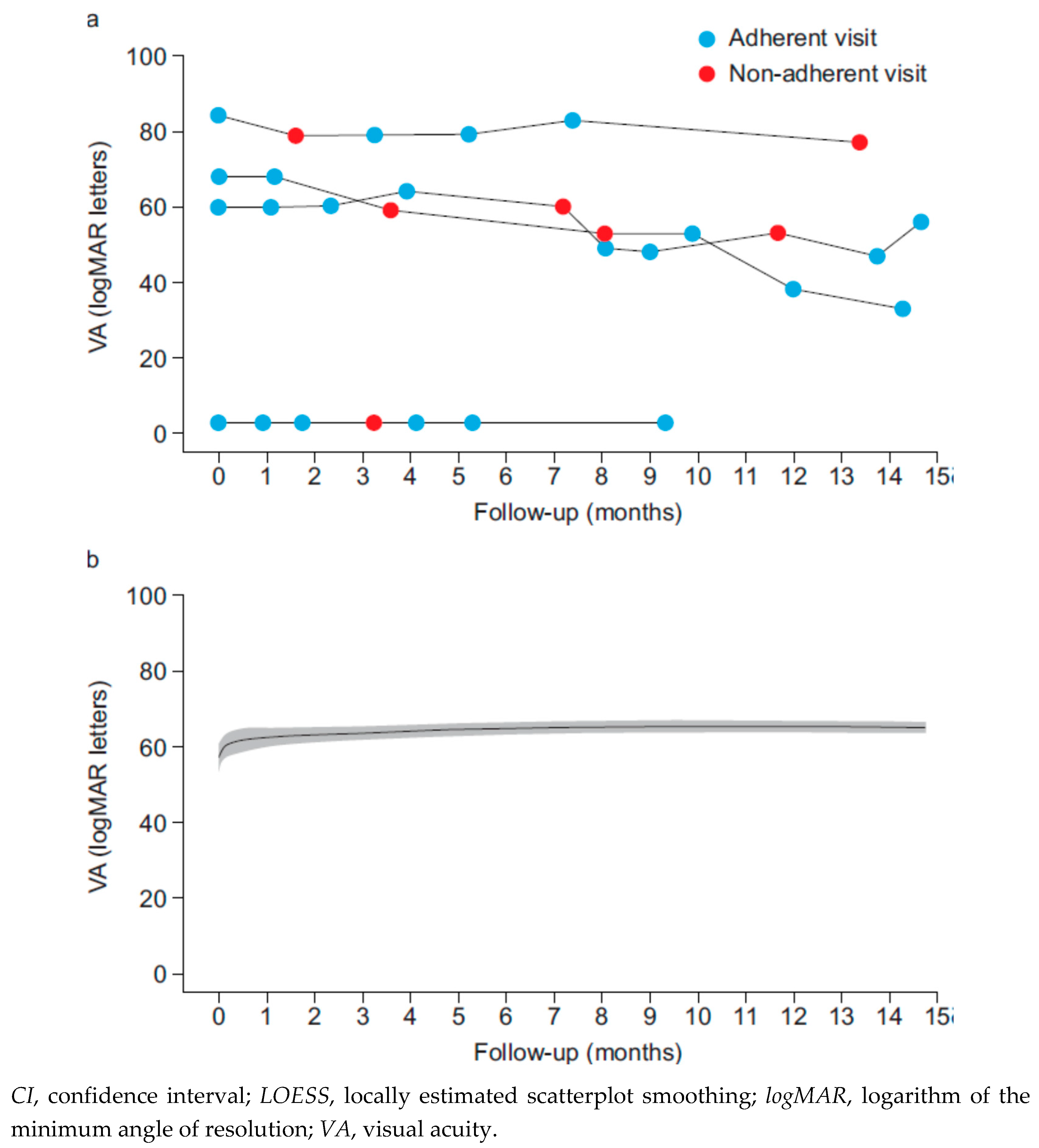
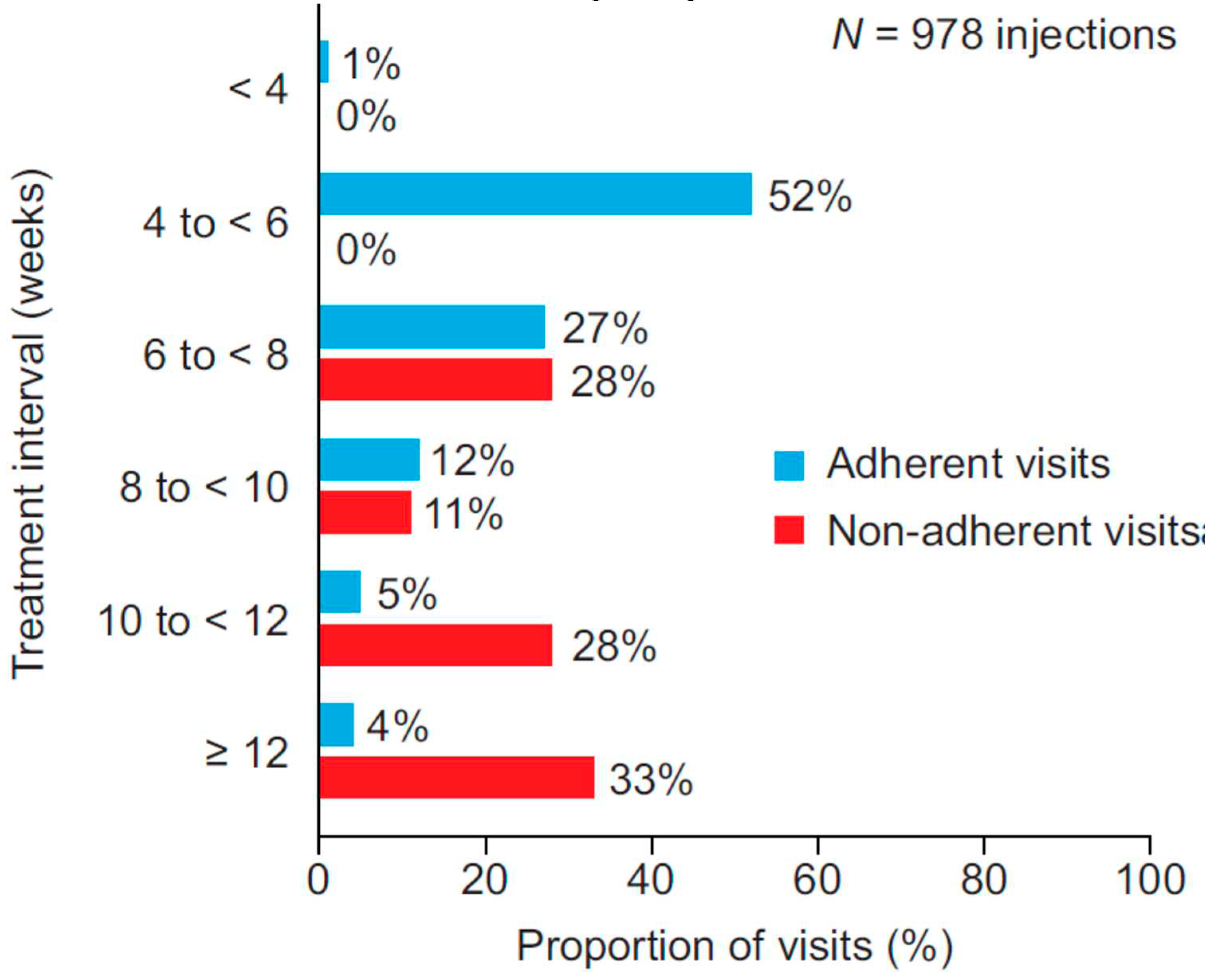
Results
Discussion
Conclusions
Author Contributions
Funding
Ethics approval and consent to participate
Consent for publication
Availability of data and materials
Acknowledgments
Competing interests
References
- Birch, D.G.; Liang, F.Q. Age-related macular degeneration: a target for nanotechnology derived medicines. Int J Nanomedicine 2007, 2, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Gass, J.D.; Agarwal, A.; Lavina, A.M.; Tawansy, K.A. Focal inner retinal hemorrhages in patients with drusen: an early sign of occult choroidal neovascularization and chorioretinal anastomosis. Retina 2003, 23, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Bourne, R.; Price, H.; Taylor, H.; Leasher, J.; Keeffe, J.; Glanville, J.; Sieving, P.C.; Khairallah, M.; Wong, T.Y.; Zheng, Y.; et al. New systematic review methodology for visual impairment and blindness for the 2010 Global Burden of Disease study. Ophthalmic Epidemiol 2013, 20, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Jager, R.D.; Mieler, W.F.; Miller, J.W. Age-related macular degeneration. N Engl J Med 2008, 358, 2606–2617. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Erfurth, U.; Chong, V.; Loewenstein, A.; Larsen, M.; Souied, E.; Schlingemann, R.; Eldem, B.; Mones, J.; Richard, G.; Bandello, F.; et al. Guidelines for the management of neovascular age-related macular degeneration by the European Society of Retina Specialists (EURETINA). Br J Ophthalmol 2014, 98, 1144–1167. [Google Scholar] [CrossRef] [PubMed]
- Kovach, J.L.; Schwartz, S.G.; Flynn, H.W., Jr.; Scott, I.U. Anti-VEGF treatment strategies for wet AMD. J Ophthalmol 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- Brown, D.M.; Kaiser, P.K.; Michels, M.; Soubrane, G.; Heier, J.S.; Kim, R.Y.; Sy, J.P.; Schneider, S.; ANCHOR Study Group. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 2006, 355, 1432–1444. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, P.J.; Brown, D.M.; Heier, J.S.; Boyer, D.S.; Kaiser, P.K.; Chung, C.Y.; Kim, R.Y.; MARINA Study Group. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 2006, 355, 1419–1431. [Google Scholar] [CrossRef]
- Prenner, J.L.; Halperin, L.S.; Rycroft, C.; Hogue, S.; Williams Liu, Z.; Seibert, R. Disease burden in the treatment of age-related macular degeneration: findings from a time-and-motion study. Am J Ophthalmol 2015, 160, 725–731. [Google Scholar] [CrossRef]
- Rofagha, S.; Bhisitkul, R.B.; Boyer, D.S.; Sadda, S.R.; Zhang, K.; SEVEN-UP Study Group. Seven-year outcomes in ranibizumab-treated patients in ANCHOR, MARINA, and HORIZON: a multicenter cohort study (SEVEN-UP). Ophthalmology 2013, 120, 2292–2299. [Google Scholar] [CrossRef]
- Mosen, D.M.; Schmittdiel, J.; Hibbard, J.; Sobel, D.; Remmers, C.; Bellows, J. Is patient activation associated with outcomes of care for adults with chronic conditions? J Ambul Care Manage 2007, 30, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Graffigna, G.; Barello, S.; Bonanomi, A. The role of Patient Health Engagement Model (PHE-model) in affecting patient activation and medication adherence: a structural equation model. PLoS One 2017, 12, e0179865. [Google Scholar] [CrossRef] [PubMed]
- Chong Teo, K.Y.; Saxena, N.; Gan, A.; Wong, T.Y.; Gillies, M.C.; Chakravarthy, U.; Gemmy Cheung, C.M. Detrimental Effect of Delayed Re-treatment of Active Disease on Outcomes in Neovascular Age-Related Macular Degeneration: The RAMPS Study. Ophthalmol Retina 2020, 4, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Ehlken, C.; Ziemssen, F.; Eter, N.; Lanzl, I.; Kaymak, H.; Lommatzsch, A.; Schuster, A.K. Systematic review: non-adherence and non-persistence in intravitreal treatment. Graefes Arch Clin Exp Ophthalmol 2020, 258, 2077–2090. [Google Scholar] [CrossRef] [PubMed]
- Holz, F.G.; Tadayoni, R.; Beatty, S.; Berger, A.R.; Cereda, M.G.; Hykin, P.; Staurenghi, G.; Wittrup-Jensen, K.; Nilsson, J.; Kim, K.; et al. Determinants of visual acuity outcomes in eyes with neovascular AMD treated with anti-VEGF agents: an instrumental variable analysis of the AURA study. Eye (Lond) 2016, 30, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, M.S.; Yu, Y.; VanderBeek, B.L. Association of Visit Adherence and Visual Acuity in Patients With Neovascular Age-Related Macular Degeneration: Secondary Analysis of the Comparison of Age-Related Macular Degeneration Treatment Trial. JAMA Ophthalmol 2020, 138, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Yalamanchili, S.P.; Maatouk, C.M.; Enwere, D.U.; Conti, T.F.; Hom, G.L.; Briskin, I.N.; Greenlee, T.E.; Babiuch, A.S.; Singh, R.P. The Short-term Effect of a Single Lapse in Anti-Vascular Endothelial Growth Factor Treatment for Diabetic Macular Edema Within Routine Clinical Practice. Am J Ophthalmol 2020, 219, 215–221. [Google Scholar] [CrossRef]
- Gillies, M.C.; Walton, R.; Liong, J.; Arnold, J.J.; McAllister, I.; Morlet, N.; Hunyor, A.; Guymer, R.; Keeffe, J.; Essex, R.; et al. Efficient capture of high-quality data on outcomes of treatment for macular diseases: the Fight Retinal Blindness! project. Retina 2014, 34, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Ehlken, C.; Helms, M.; Bohringer, D.; Agostini, H.T.; Stahl, A. Association of treatment adherence with real-life VA outcomes in AMD, DME, and BRVO patients. Clin Ophthalmol 2018, 12, 13–20. [Google Scholar] [CrossRef]
- Gillies, M.C.; Walton, R.J.; Arnold, J.J.; McAllister, I.L.; Simpson, J.M.; Hunyor, A.P.; Guymer, R.; Essex, R.W.; Morlet, N.; Barthelmes, D. Comparison of outcomes from a phase 3 study of age-related macular degeneration with a matched, observational cohort. Ophthalmology 2014, 121, 676–681. [Google Scholar] [CrossRef]
- Clemens, A.; Sagkriotis, A.; Griner, R.; Durus, A.; Doyle, O.; Wintermantel, T.; Chakravarthy, U. Key confounders for translating results from non-interventional trials (NIS) to those observed in randomized controlled trials (RCTs): applying predictive analytics in neovascular age-related macular degeneration (nAMD). Proceedings of 19th EURETINA Congress, Paris, France., 5–8 September 2019. [Google Scholar]
- Holz, F.G.; Tadayoni, R.; Beatty, S.; Berger, A.; Cereda, M.G.; Cortez, R.; Hoyng, C.B.; Hykin, P.; Staurenghi, G.; Heldner, S.; et al. Multi-country real-life experience of anti-vascular endothelial growth factor therapy for wet age-related macular degeneration. Br J Ophthalmol 2015, 99, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Kim, L.N.; Mehta, H.; Barthelmes, D.; Nguyen, V.; Gillies, M.C. Metaanalysis of real-world outcomes of intravitreal ranibizumab for the treatment of neovascular age-related macular degeneration. Retina 2016, 36, 1418–1431. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.Y.; Dubois, L.; Tadayoni, R.; Fajnkuchen, F.; Nghiem-Buffet, S.; Delahaye-Mazza, C.; Guiberteau, B.; Quentel, G. Results of one-year's treatment with ranibizumab for exudative age-related macular degeneration in a clinical setting. Am J Ophthalmol 2009, 148, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Gillies, M.C.; Nguyen, V.; Daien, V.; Arnold, J.J.; Morlet, N.; Barthelmes, D. Twelve-month outcomes of ranibizumab vs. aflibercept for neovascular age-related macular degeneration: data from an observational study. Ophthalmology 2016, 123, 2545–2553. [Google Scholar] [CrossRef]
- Teo, K.; Saxena, N.; Gan, A.; Wong, T.Y.; Gillies, M.C.; Chakravarthy, U.; Cheung, C. Detrimental effect of delayed retreatment of active disease on outcomes in neovascular age related macular degeneration – RAMPS study. Ophthalmol Retina 2020. [Google Scholar] [CrossRef]
- Parvin, P.; Zola, M.; Dirani, A.; Ambresin, A.; Mantel, I. Two-year outcome of an observe-and-plan regimen for neovascular age-related macular degeneration treated with Aflibercept. Graefes Arch Clin Exp Ophthalmol 2017, 255, 2127–2134. [Google Scholar] [CrossRef]
| Parameter | Overall | Adherent | Non-adherent |
|---|---|---|---|
| Patients | 109 | 105 | 4 |
| Eyes | 133 | 129 | 4 |
| Sex (% women) | 63.9 | 63.8 | 50 |
| Age (mean ± SD) | 80.1 ± 8.1 | 79.9 ± 8.1 | 86.8 ± 5.7 |
| LogMAR VA (mean ± SD) | 56.9 ± 23.8 | 57.0 ± 23.6 | 53.8 ± 35.3 |
| ≤ 35 letters (%) | 25 (18.8) | 24 (18.6) | 1 (25.0) |
| 36–69 letters (%) | 59 (44.4) | 57 (44.2) | 2 (50.0) |
| ≥ 70 letters (%) | 49 (36.8) | 48 (37.2) | 1 (25.0) |
| Angiographic lesion type (%) | |||
| Type 1 | 77 (57.9) | 74 (57.4) | 3 (75.0) |
| Type 2 | 32 (24.1) | 31 (24.0) | 1 (25.0) |
| Type 3 | 4 (3.0) | 4 (3.1) | 0 (0.0) |
| Other | 12 (9.0) | 12 (9.3) | 0 (0.0) |
| Unknown | 8 (6.0) | 8 (6.2) | 0 (0.0) |
| Parameter | Adherent eyes (n = 129) |
Non-adherent eyes (n = 4) |
|---|---|---|
| Baseline logMAR VA (mean ± SD) | 57.0 ± 23.6 | 53.8 ± 35.3 |
| Final logMAR VA (mean ± SD) | 63.3 ± 23.3 | 42.8 ± 31.0 |
| ≤ 35 letters (%) | 18 (14) | 1 (25) |
| 36–69 letters (%) | 40 (31) | 2 (50) |
| ≥ 70 letters (%) | 71 (55) | 1 (25) |
| Mean VA change from baseline (95% CI) | 6.3 (3.7, 8.9) | −11 (−31.8, 9.8) |
| Injections, median (Q1, Q3) | 8 (7, 10) | 6 (5.8, 6.2) |
| Visits, median (Q1, Q3) | 10 (9, 12) | 7.5 (7, 8) |
| Non-injection visits, median (Q1, Q3) | 1 (0, 2) | 1.5 (1, 2) |
| Time period | Outcome | Adherent visits | Non-adherent visits |
|---|---|---|---|
| Overall | Number of eyes | 133 | 19 |
| Number of visits | 1403 | 23 | |
| Mean VA change (95% CI) | 0.67 (0.32, 1.02) | −2.30 (−5.01, 0.40) | |
| 0–3 months | Number of eyes | 132 | 2 |
| Number of visits | 435 | 2 | |
| Mean VA change (95% CI) | 2.43 (1.73, 3.14) | 0.00 (−9.04, 9.04) | |
| > 3 to 6 months | Number of eyes | 132 | 2 |
| Number of visits | 304 | 4 | |
| Mean VA change (95% CI) | 0.44 (−0.26, 1.14) | −1.25 (−7.64, 5.14) | |
| > 6 to 12 months | Number of eyes | 130 | 15 |
| Number of visits | 664 | 17 | |
| Mean VA change (95% CI) | −0.02 (−0.50, 0.45) | −2.82 (−5.92, 0.28) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




