Submitted:
01 November 2023
Posted:
02 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Definitions1:
- Timely birth dose coverage: the proportion of live births who receive a HepB-BD within 24 hours after birth. All doses given on day 0 or 1 of life meet this definition (i.e. the date of delivery and the day following delivery). This is the global standard for monitoring HepB-BD coverage.
- Total birth dose coverage: the proportion of live births who receive any HepB-BD, defined as those vaccinated any time up until the first dose is due, or as per country guidance on upper age range.
- Health facility birth dose coverage: the proportion of live births in a health facility who receive HepB-BD. This may be tracked separately if there are different coverage targets for facility births, especially regarding timely birth dose administration.
- Home birth dose coverage: the proportion of live home births who receive HepB-BD. This section outlines the methods for monitoring and evaluating a HepB-BD vaccination
1.2. Rationale:
1.3. Objectives:
- 5.
- Review the uptake of the HepB-BD at the SLBMC, capturing the reasons for refusal
- 6.
- Outline recommendations for quality improvement for the HepB-BD program
2. Materials and Methods
2.1. Methods:
2.2. Data Analysis:
2.3. Ethics:
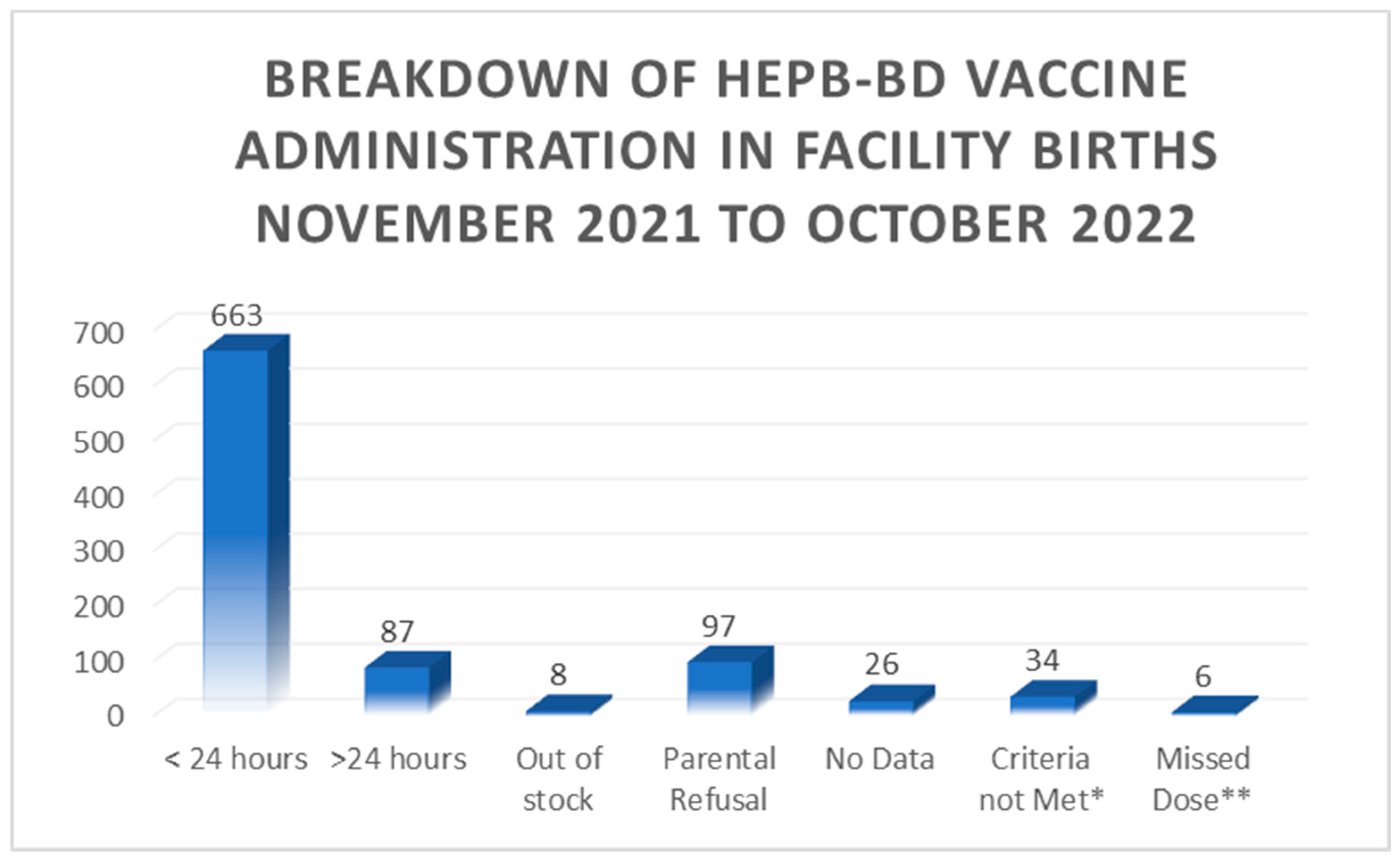
3. Results
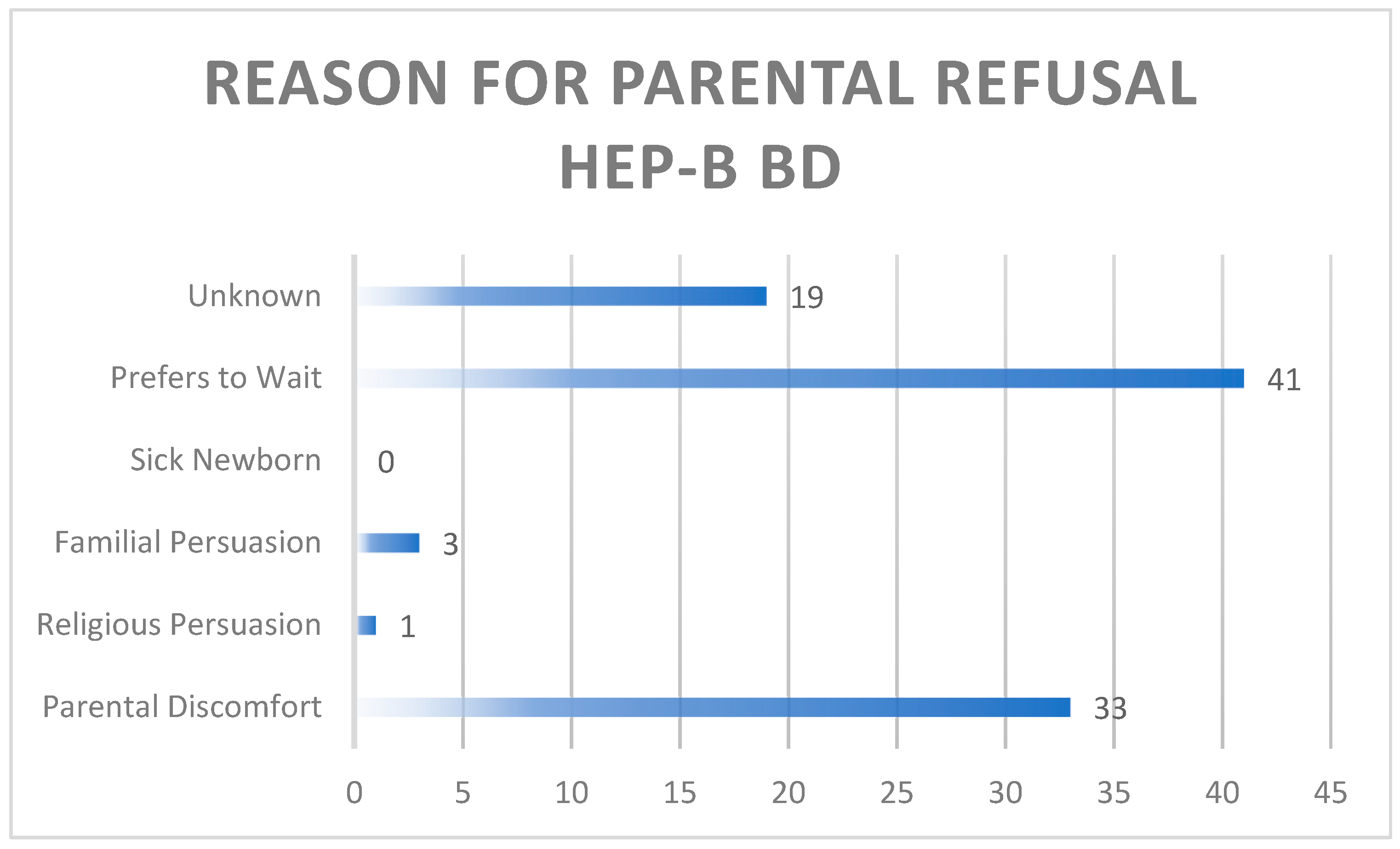
3.1. Parental Refusal
3.2. Maternal Hepatitis B Status
3.3. Factors influencing administration
4. Discussion
4.1. Timeliness
4.2. Barriers/education
4.2.1. Client level
4.2.3. Facility level
- Knowledge of Health Care workers
- Supply chain
- Regulatory Considerations
- Recommendations
5. Conclusion
Author Contributions
Funding
Research in Context
Added Value
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
References
- World Health Organization. Preventing perinatal hepatitis B virus transmission: a guide for introducing and strengthening hepatitis B birth dose vaccination.
- World Health Organization. (2019) Immunization. Available at Immunization (who.int) (Accessed 8 August2023).
- Awuku, Y.A.; Yeboah-Afihene, M. Hepatitis B at-birth dose vaccine: an urgent call for implementation in Ghana. Vaccines. 2018, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- PAHO/WHO | Six Caribbean territories and states eliminate mother-to-child transmission of HIV and syphilis. (Accessed 28 September 2023).
- Plus, P.E. Framework for elimination of mother-to-child transmission of HIV, Syphilis, Hepatitis B, and Chagas; Pan American Health Organisation: Washington, DC, USA, 2017. [Google Scholar]
- Khetsuriani, N.; Lesi, O.; Desai, S.; Armstrong, P.A.; Tohme, R.A. Progress Toward the Elimination of Mother-to-Child Transmission of Hepatitis B Virus — Worldwide, 2016–2021. MMWR Morb Mortal Wkly Rep 2022, 71, 958–963. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Interim guidance for country validation of viral hepatitis elimination.
- WHO. (2022). Introduction of HepB birth dose. Available at Introduction of HepB birth dose (who.int) (Accessed on 7 August 2023).
- Population, total- Antigua and Barbuda, Population, total - Antigua and Barbuda | Data (worldbank.org)(Accessed on December 8 2021).
- Ministry of Health Wellness and the, Environment. Maternal Child and Adolescent Health Manual, Antigua and Barbuda, 3rd ed.; 2020. [Google Scholar]
- Age of Consent in Antigua and Barbuda. (2023) Available at Antigua And Barbuda Age of Consent & Statutory Rape Laws (Accessed 28 September 2023).
- Bada, F.O.; Stafford, K.A.; Osawe, S.; Wilson, E.; Sam-Agudu, N.A.; Chen, H.; Abimiku, A.L.; Campbell, J.D. Factors associated with receipt of a timely infant birth dose of hepatitis B vaccine at a tertiary hospital in North-Central Nigeria. PLOS Global Public Health 2022, 2, e0001052. [Google Scholar] [CrossRef] [PubMed]
- PAHO. (2022) EMTCT Plus Initiative 2010-2021. Available at2023-cde-4-etmi-plus-prev-mtct-childhood-hep-b.pdf (Accessed 7 August 2023).
- Immunization, HepB3 (% of one-year-old children) | Data (worldbank.org) (2023) (Accessed 7 August 2023).
- Introduction of Hepatitis B Birth dose vaccination in Africa: a toolkit for National Immunization Technical Advisory groups 2022. (2022) Available at HepB-BD NITAG toolkit final version_12-16-22_0-FINAL.pdf (globalhep.org) (Accessed 8 August 2023).
- Boisson, A.; Goel, V.; Yotebieng, M.; Parr, J.B.; Fried, B.; Thompson, P. Implementation approaches for introducing and overcoming barriers to hepatitis B birth-dose vaccine in sub-Saharan Africa. Global Health: Science and Practice 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.D.; Roberts, B.; Wong, B.L.; van Kessel, R.; Mossialos, E. The relationship between the COVID-19 pandemic and vaccine hesitancy: a scoping review of literature until August 2021. Frontiers in public health 2021, 9. [Google Scholar]
- World Health Organization. Practices to improve coverage of the hepatitis B birth dose vaccine, World Health Organization, 2012.
- Moturi, E.; Tevi-Benissan, C.; Hagan, J.E.; Shendale, S.; Mayenga, D.; Murokora, D.; Patel, M.; Hennessey, K.; Mihigo, R. Implementing a birth dose of hepatitis B vaccine in Africa: findings from assessments in 5 countries. Journal of immunological sciences 2018, 31. [Google Scholar] [CrossRef]
- Adilo, T.M.; Endale, S.Z.; Demie, T.G.; Dinka, T.G. The impact of COVID-19 on supplies of routine childhood immunization in Oromia regional state, Ethiopia: A mixed method study. Risk management and healthcare policy 2022, 2343–2355. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Maternal and neonatal immunization field guide for Latin America and the Caribbean; World Health Organization: Washington, DC, 2017. [Google Scholar]
| Maternal HBsAg Status | HepB-BD received n (%) |
HepB-BD not received n (%) | No Data n (%) |
Total Live Births n (%) |
|---|---|---|---|---|
| Negative | 677 (73.3) | 129 (14.0) | 23 (2.5) | 829 (89.7) |
| Unknown | 69 (7.5) | 15 (1.6) | 2 (0.2) | 86 (9.3) |
| Positive | 6 (0.6) | 0 | 0 | 6 (0.6) |
| No Data | 1 (0.1) | 1 (0.1) | 1(0.1) | 3 (0.4) |
| Total | 753 (81.5) | 145 (15.7) | 26 (3.0) | 924 |
| Characteristic | All | Timely administration | Untimely administration | OR (95%CI) | p value |
|---|---|---|---|---|---|
| Maternal age < 35 | 742 | 545 | 62 |
1.788 (0.3354 to 0.9328) |
<0.05 |
| Maternal age ≥ 35 | 182 | 118 | 24 | ||
| Weekend | 212 | 193 | 21 | 1.271 (0.7559 – 2.137) |
>0.05 |
| Weekday | 538 | 470 | 65 | ||
| Vaginal | 730 | 574 | 29 | 12.23 (7.403 – 20.21) |
<0.05 |
| C-section | 194 | 89 | 55 |
| WHO Region | HepB-BD Coverage (%) | HepB3 Coverage (%) |
|---|---|---|
| African Region | 17 | 71 |
| Region of the Americas | 59 | 80 |
| Eastern Mediterranean Region | 33 | 82 |
| European Region | 43 | 91 |
| South East Asian Region | 51 | 82 |
| Western Pacific Region | 78 | 90 |
| Global Average | 42 | 80 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




