Submitted:
11 October 2023
Posted:
12 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
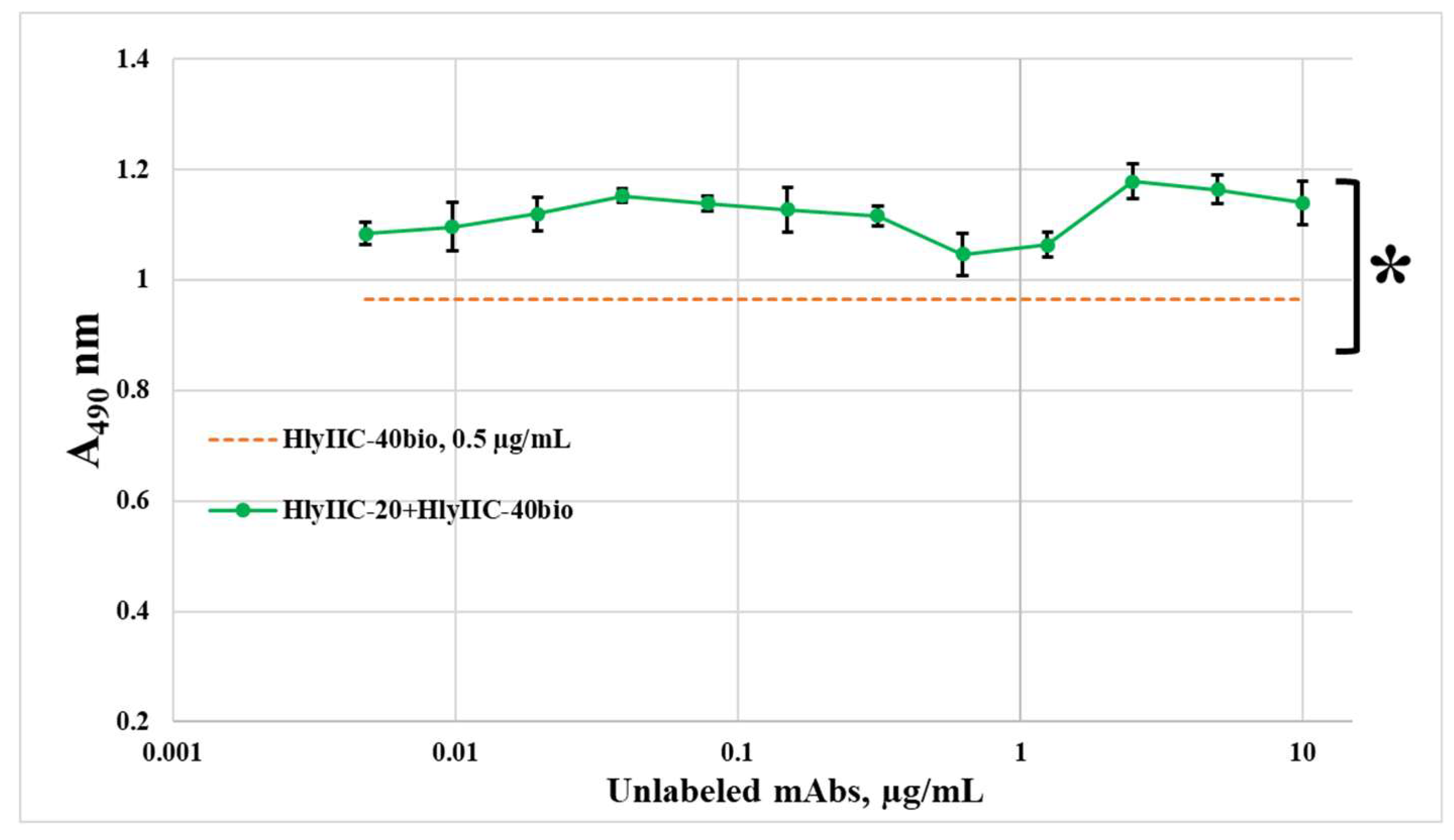
2.1. Monoclonal Antibodies HlyIIC-20 and HlyIIC-40 Recognize Non-Overlapping Regions on the Surface of the Spatial Structure of HlyIICTD
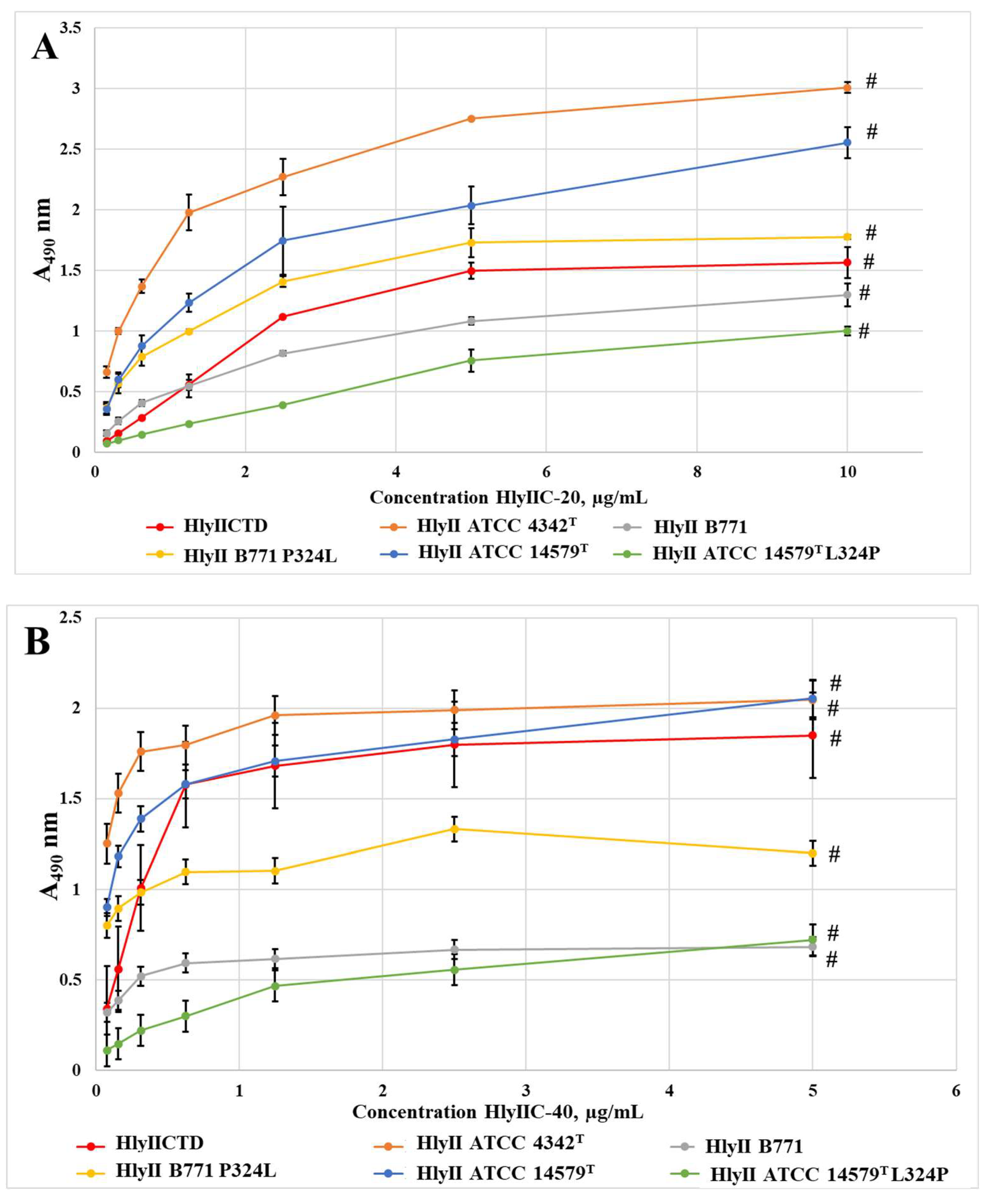
2.2. Efficiency of HlyIIC-20 Binding to Full-Length Hemolysins of B. cereus Strains ATCC 14579T, B771, ATCC 4342T
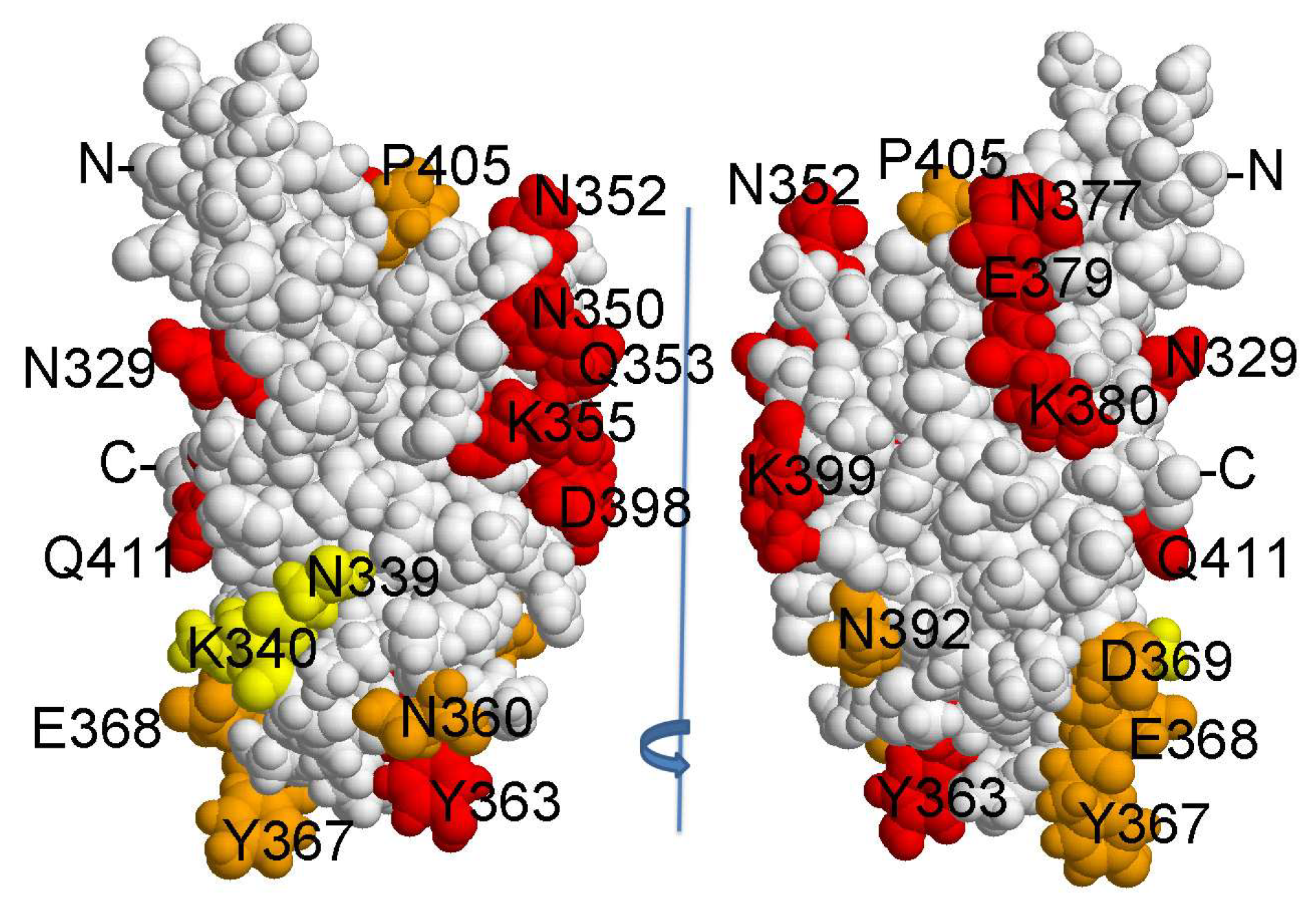
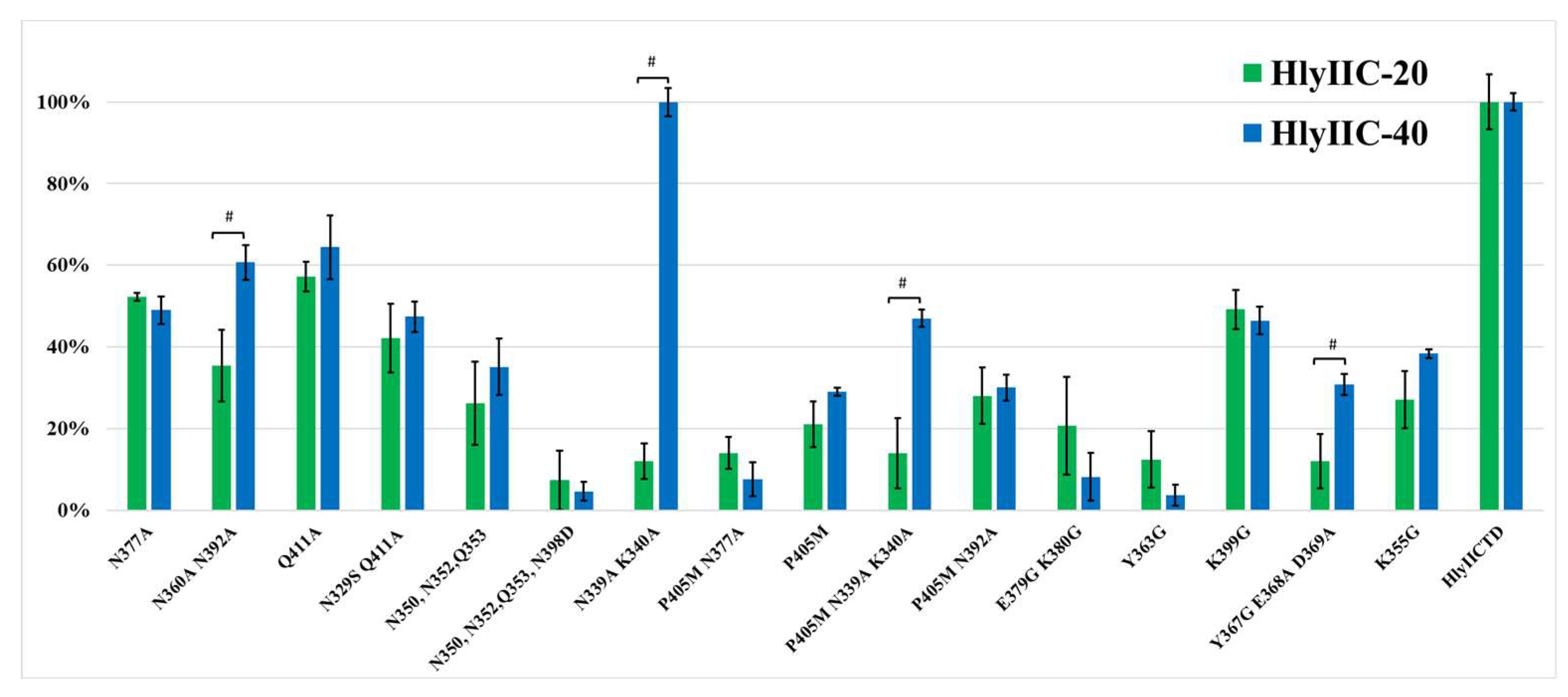
2.3. Determination of HlyIIC-20 Epitope
3. Discussion
4. Materials and Methods
4.1. Strains, Plasmids and Enzymes
4.2. Molecular Cloning and Site-Directed Mutagenesis
4.3. Expression and Purification of HlyIICTD His6 and Its Mutant Forms
4.4. Enzyme Immunosorbent Assay
4.5. Conjugation of Antibodies with Biotin
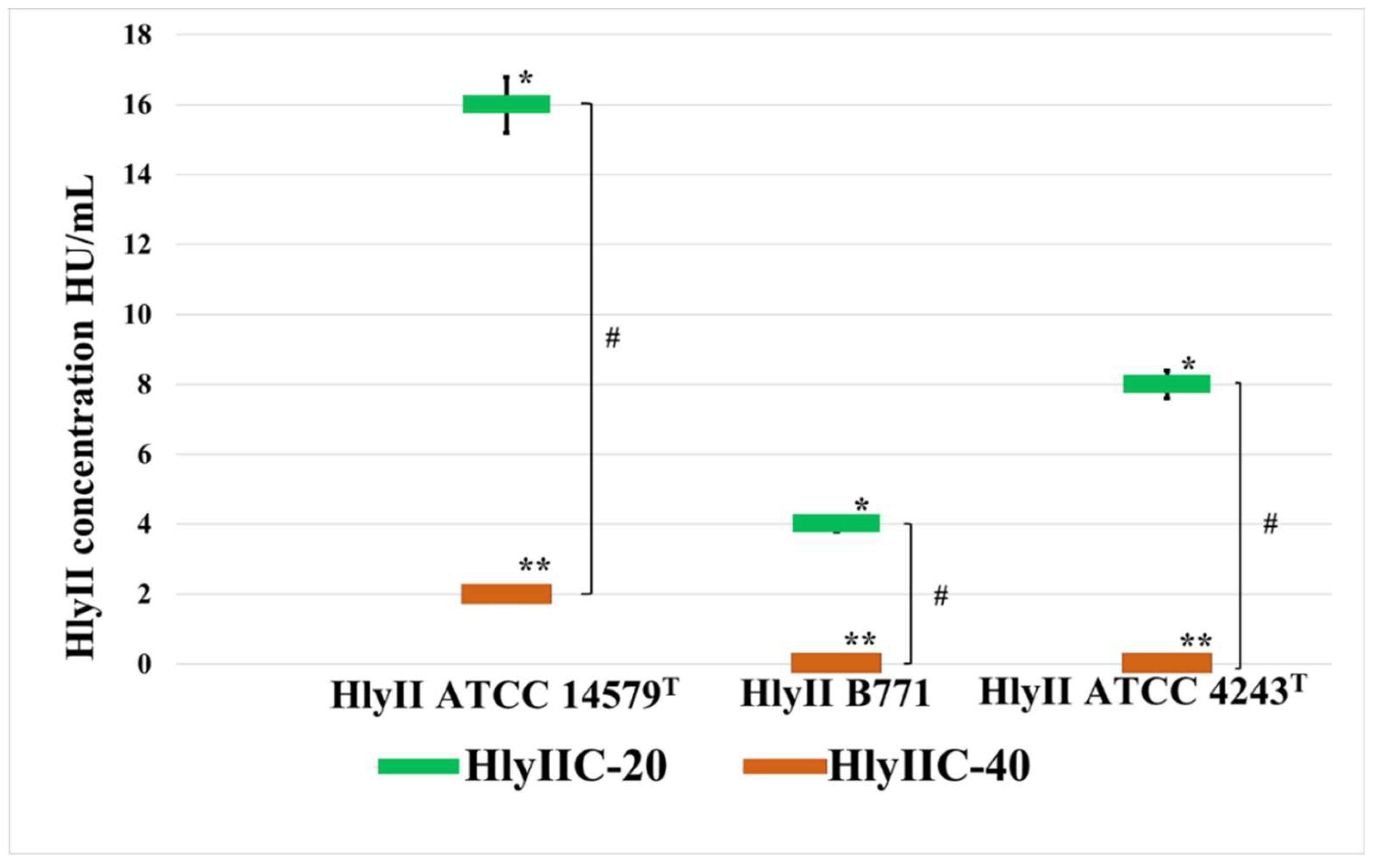
4.6. Measurement of Hemolytic Activity of Hemolysin II in the Presence of mAbs
Author Contributions
Funding
Conflicts of Interest
References
- Logan, N.A. Bacillus and relatives in foodborne illness. J Appl Microbiol. 2012, 3, 417–29. [Google Scholar] [CrossRef] [PubMed]
- Ramarao, N.; Sanchis, V. The pore-forming haemolysins of Bacillus cereus: a review. Toxins (Basel) 2013, 6, 1119–39. [Google Scholar] [CrossRef] [PubMed]
- Miles, G.; Bayley, H.; Cheley, S. Properties of Bacillus cereus hemolysin II: a heptameric transmembrane pore. Protein Sci. 2002, 7, 1813–24. [Google Scholar] [CrossRef] [PubMed]
- Andreeva, Z.I.; Nesterenko, V.F.; Fomkina, M.G.; Ternovsky, V.I.; Suzina, N.E.; Bakulina, A.Y.; Solonin, A.S.; Sineva, E.V. The properties of Bacillus cereus hemolysin II pores depend on environmental conditions. Biochim Biophys Acta 2007, 2, 253–63. [Google Scholar] [CrossRef] [PubMed]
- Andreeva, Z.I.; Nesterenko, V.F.; Yurkov, I.S.; Budarina, Z.I.; Sineva, E.V.; Solonin, A.S. Purification and cytotoxic properties of Bacillus cereus hemolysin II. Protein Expr Purif. 2006, 1, 186–93. [Google Scholar] [CrossRef] [PubMed]
- Kholodkov, O.A.; Budarina, Zh.; Kovalevskaya, J.I.; Siunov, A.V.; Solonin, A. A. Effect of Bacillus cereus hemolysin II on hepatocyte cells. Prikl Biokhim Mikrobiol. 2015, 2, 258–67. [Google Scholar] [CrossRef]
- Kataev, A.A.; Andreeva-Kovalevskaya, Z.I.; Solonin, A.S.; Ternovsky, V.I. Bacillus cereus can attack the cell membranes of the alga Chara corallina by means of HlyII. Biochim Biophys Acta 2012, 5, 1235–1241. [Google Scholar] [CrossRef] [PubMed]
- Teplova, V.V.; Andreeva-Kovalevskaya, Z.I.; Sineva, E.V.; Solonin, A.S. Quick assessment of cytotoxins effect on Daphnia magna using in vivo fluorescence microscopy. Environ Toxicol Chem. 2010, 6, 1345–1348. [Google Scholar] [CrossRef] [PubMed]
- Baida, G.; Budarina, Z.I.; Kuzmin, N.P.; Solonin, A.S. Complete nucleotide sequence and molecular characterization of hemolysin II gene from Bacillus cereus. FEMS Microbiol Lett. 1999, 1, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, N.V.; Karatovskaya, A.P.; Zamyatina, A.V.; Siunov, A.V.; Andreeva-Kovalevskaya, Z.I.; Nagel, A.S.; Brovko, F.A.; Solonin, A.S. C-terminal domain of Bacillus cereus hemolysin II is able to interact with erythrocytes. Russ J Bioorg Chem. 2020, 46, 321–326. [Google Scholar] [CrossRef]
- Rudenko, N.; Siunov, A.; Zamyatina, A.; Melnik, B.; Nagel, A.; Karatovskaya, A.; Borisova, M.; Shepelyakovskaya, A.; Andreeva-Kovalevskaya, Zh.; Kolesnikov, A.; Surin, A.; Brovko, F.; Solonin, A. The C-terminal domain of Bacillus cereus hemolysin II oligomerizes by itself in the presence of cell membranes to form ion channels. Int J Biol Macromol. 2022, 200, 416–427. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.R.; Kaus, K.; De, S.; Olson, R.; Alexandrescu, A.T. NMR structure of the Bacillus cereus hemolysin II C-terminal domain reveals a novel fold. Sci Rep. 2017, 1, 3277. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.R.; Maciejewski, M.W.; Olson, R.; Alexandrescu, A.T. NMR assignments for the cis and trans forms of the hemolysin II C-terminal domain. Biomol NMR Assign. 2014, 2, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.R.; Olson, R.; Alexandrescu, A.T. Protein yoga: conformational versatility of the hemolysin II C-terminal domain detailed by NMR structures for multiple states. Protein Sci. 2021, 5, 990–1005. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, N.; Nagel, A.; Zamyatina, A.; Karatovskaya, A.; Salyamov, V.; Andreeva-Kovalevskaya, Zh.; Siunov, A.; Kolesnikov, A.; Shepelyakovskaya, A.; Boziev, Kh.; Melnik, B.; Brovko, F.; Solonin, A. A monoclonal antibody against the C-terminal domain of Bacillus cereus hemolysin II inhibits HlyII cytolytic activity. Toxins 2020, 12, 806. [Google Scholar] [CrossRef] [PubMed]
- Janeway's immunobiology / Kenneth Murphy, Casey Weaver; with contributions by Allan Mowat, Leslie Berg, David Chaplin; with acknowledgment to Charles A. Janeway Jr., Paul Travers, Mark Walport. Murphy, Kenneth (Kenneth M.), New York, NY Garland Science/Taylor & Francis Group, LLC, 9th edition, 2017.
- Gouaux, E. alpha-Hemolysin from Staphylococcus aureus: an archetype of beta-barrel, channel-forming toxins. J Struct Biol. 1998, 121, 110–22. [Google Scholar] [CrossRef] [PubMed]
- Li, Y., Mengist, H.M., Shi, C., Zhang, C., Wang, B., Li, T., Huang, Y., Xu, Y., Jin, T. Structural basis of the pore-forming toxin/membrane interaction. Toxins (Basel) 2021, 13, 128.
- Sambrook, J.; Russell, D.W. Site-specific mutagenesis by overlap extension. CSH Protoc. 2006, 1, pdb.prot3468. [CrossRef]
| Mutation name | Oligonucleotide name | Sequence 5’->3’ |
|---|---|---|
| N377A | F_N377A | CTTTGTAGCTGGTGAAAAGGTCTATAC |
| R_N377A | TTCACCAGCTACAAAGATACCCCAAT | |
| N360A N392A | F_N360A R_N360A F_N392A R_N392A |
TCTAGCGCAGCTGGTTATGGTATC ACCAGCTGCGCTAGATGTAGCTTTAAG GTAGGCGCTATCTCTAATGATATTAACAAAT AGAGATAGCGCCTACAGTTGATTTTTC |
| Q411A | R_Q411A | TAACTCGAGGGTACCGATAGCTTTAATCTCGATATAAGGTCC |
| N329S Q411A | R_Q411A | TAACTCGAGGGTACCGATAGCTTTAATCTCGATATAAGGTCC |
| F_N329S | GAACAAATGAGTAGTATCAACAGCGTG | |
| R_N329S | GCTGTTGATACTACTCATTTGTTCTTCAAG | |
| N350A N352A Q353A N398D | F-NNQ | ATGGCTGGAGCTGCTCTTAAAGCTACATCTAGC |
| R-NNQ | AAGAGCAGCTCCAGCCATTGAAAGAGATAATTTC | |
| F_N398D | CTCTAATGATATTGATAAATTAAACATTAAAGG | |
| R_N398D | CTTTAATGTTTAATTTATCAATATCATTAGAGATATTG | |
| N339A K340A | F_NK339 | AACTTGCTGCTGGAAAAGGGAAATTATC |
| R_NK339 | CTTTTCCAGCAGCAAGTTTATCATTCACGC | |
| P405M N377A | F_P405M | AAAGGAATGTATATCGAGATTAAACAGATC |
| R_P405M | GATATACATTCCTTTAATGTTTAATTTG | |
| F_N377A | CTTTGTAGCTGGTGAAAAGGTCTATAC | |
| R_N377A | TTCACCAGCTACAAAGATACCCCAAT | |
| P405M | F_P405M | AAAGGAATGTATATCGAGATTAAACAGATC |
| R_P405M | GATATACATTCCTTTAATGTTTAATTTG | |
| P405M N339A K340A | F_P405M | AAAGGAATGTATATCGAGATTAAACAGATC |
| R_P405M | GATATACATTCCTTTAATGTTTAATTTG | |
| F_NK339 | AACTTGCTGCTGGAAAAGGGAAATTATC | |
| R_NK339 | CTTTTCCAGCAGCAAGTTTATCATTCACGC | |
| P405M N392A | F_P405M | AAAGGAATGTATATCGAGATTAAACAGATC |
| R_P405M | GATATACATTCCTTTAATGTTTAATTTG | |
| F_N392A | GTAGGCGCTATCTCTAATGATATTAACAAAT | |
| R_N392A | AGAGATAGCGCCTACAGTTGATTTTTC | |
| E379G K380G | F_EK379G | GGTGGTGGAGTCTATACTTTTAATGAAAAATCAAC |
| R_EK379G | GTATAGACTCCACCACCATTTACAAAGATACCC | |
| Y363G | F_Y363G | GCTGGTGGAGGTATCAGTTACGAAG |
| R_Y363G | GATACCTCCACCAGCATTGCTAGATG | |
| K399G | F_K399G | GATATTAACGGATTAAACATTAAAGGACCTTATATC |
| R_K399G | AATGTTTAATCCGTTAATATCATTAGAGATATTGC | |
| Y367G E368A D369A | F_YED367GAA | CAGTGGAGCAGCAAAAAATTGGGGTATCTTTG |
| R_YED367GAA | TTTTGCTGCTCCACTGATACCATAACCAG | |
| K355G | F_K355G | CAACTTGGAGCTACATCTAGCAATGC |
| R_K355G | GATGTAGCTCCAAGTTGATTTCCATTC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




