Submitted:
03 October 2023
Posted:
04 October 2023
Read the latest preprint version here
Abstract
Keywords:
Introduction
Materials and methods
Study Design
Data Collection and Management
Serology
Vaccine Effectiveness Analysis
Further Analyses and Sensitivity Analyses
Statistical Model
Ethics and Study Registration
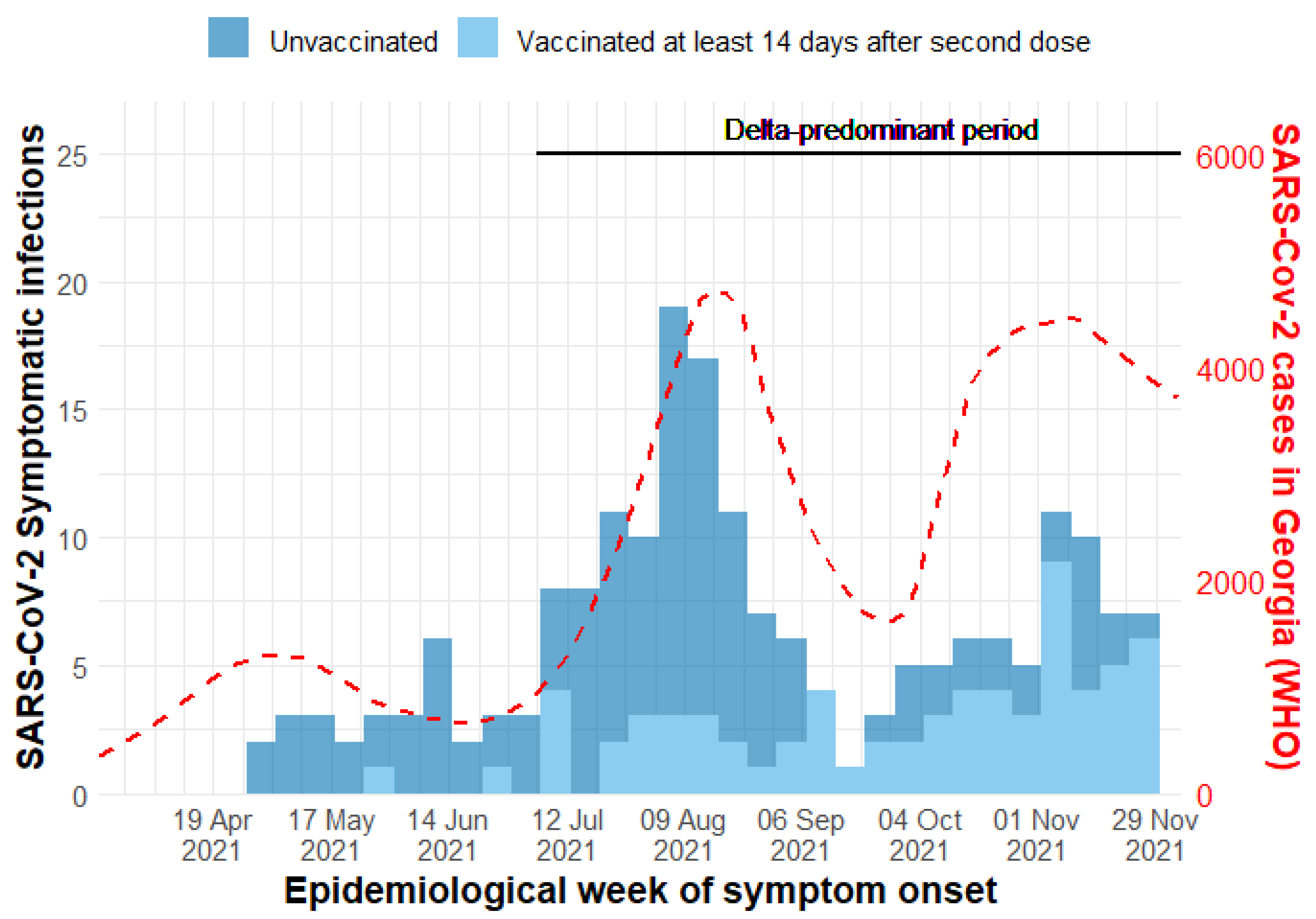
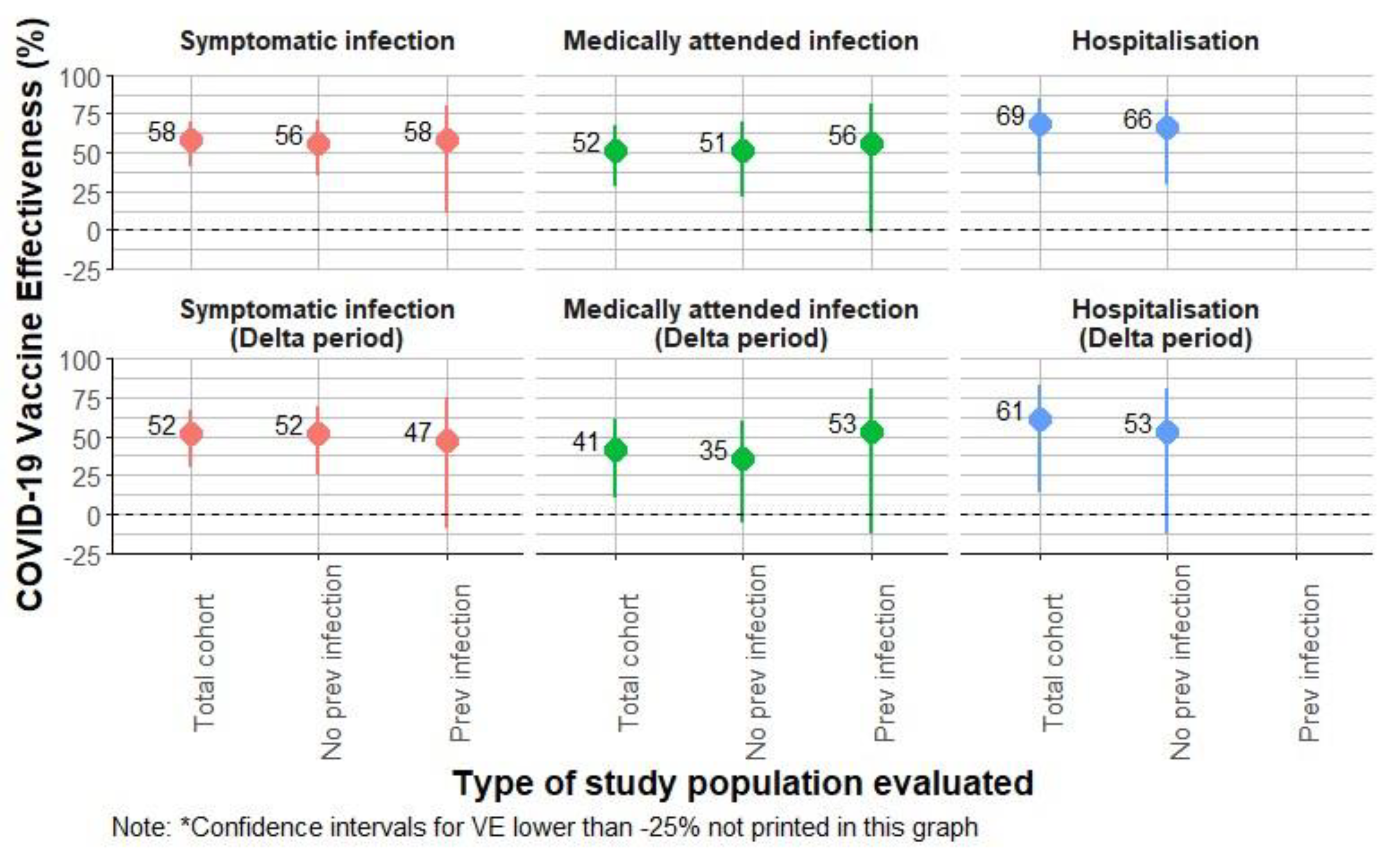
Results
Discussion
Conclusions
Author Contributions
Funding
Acknowledgements
Conflict of Interest
References
- Bandyopadhyay S, Baticulon RE, Kadhum M, et al. Infection and mortality of healthcare workers worldwide from COVID-19: a systematic review. BMJ Glob Health. 2020;5(12). [CrossRef]
- Hamid H, Abid Z, Amir A, Rehman TU, Akram W, Mehboob T. Current burden on healthcare systems in low- and middle-income countries: recommendations for emergency care of COVID-19. Drugs & Therapy Perspectives. 2020;36(10):466. [CrossRef]
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard. https://covid19.who.int/. Accessed June 30, 2022. https://covid19.who.int/. 30 June.
- Watson OJ, Barnsley G, Toor J, Hogan AB, Winskill P, Ghani AC. Global impact of the first year of COVID-19 vaccination: a mathematical modelling study. Lancet Infect Dis. 2022;22(9):1293-1302. [CrossRef]
- Meslé MMI, Brown J, Mook P, et al. Estimated number of deaths directly averted in people 60 years and older as a result of COVID-19 vaccination in the WHO European Region, December 2020 to November 2021. Eurosurveillance. 2021;26(47):2101021. 20 December. [CrossRef]
- Effectiveness Studies | ViewHub. Accessed September 12, 2022. https://view-hub.org/covid-19/effectiveness-studies. 12 September.
- WHO/Europe Covid-19 vaccine programme monitor. Accessed October 17, 2022. https://worldhealthorg.shinyapps.io/EURO_COVID-19_vaccine_monitor/. 17 October.
- WHO Europe. WHO/Europe COVID-19 Vaccine Programme Monitor. Accessed July 11, 2022. https://worldhealthorg.shinyapps.io/EURO_COVID-19_vaccine_monitor/. 11 July.
- Lucaccioni H, Chakhunashvili G, McKnight CJ, et al. Sociodemographic and Occupational Factors Associated with Low Early Uptake of COVID-19 Vaccine in Hospital-Based Healthcare Workers, Georgia, March–July 2021. Vaccines (Basel). 2022;10(8):1197. 20 July. [CrossRef]
- World Health Organization. Regional Office for Europe. Cohort Study to Measure COVID-19 Vaccine Effectiveness among Healthcare Workers in the WHO European Region: Guidance Document.; 2021.
- World Health Organization. Coronavirus disease (COVID-19) technical guidance: The Unity Studies: Early Investigation Protocols. Accessed November 8, 2021. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/early-investigations. 8 November.
- Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2). [CrossRef]
- Riester E, Findeisen P, Hegel JK, et al. Performance evaluation of the Roche Elecsys Anti-SARS-CoV-2 S immunoassay. J Virol Methods. 2021;297:114271. [CrossRef]
- Khare S, Gurry C, Freitas L, et al. GISAID’s Role in Pandemic Response. China CDC Wkly. 2021;3(49):1049-1051. [CrossRef]
- Wajnberg A, Mansour M, Leven E, et al. Humoral response and PCR positivity in patients with COVID-19 in the New York City region, USA: an observational study. Lancet Microbe. 2020;1(7):e283-e289. [CrossRef]
- Higdon MM, Wahl B, Jones CB, et al. A Systematic Review of Coronavirus Disease 2019 Vaccine Efficacy and Effectiveness Against Severe Acute Respiratory Syndrome Coronavirus 2 Infection and Disease. Open Forum Infect Dis. 2022;9(6). [CrossRef]
- Mallapaty S. China’s COVID vaccines have been crucial - now immunity is waning. Nature. 2021;598(7881):398-399. [CrossRef]
- Wu D, Zhang Y, Tang L, et al. Effectiveness of Inactivated COVID-19 Vaccines Against Symptomatic, Pneumonia, and Severe Disease Caused by the Delta Variant: Real World Study and Evidence — China, 2021. China CDC Weekly, 2022, Vol 4, Issue 4, Pages: 57-65. 2022;4(4):57-65. [CrossRef]
- Ma C, Sun W, Tang T, et al. Effectiveness of adenovirus type 5 vectored and inactivated COVID-19 vaccines against symptomatic COVID-19, COVID-19 pneumonia, and severe COVID-19 caused by the B.1.617.2 (Delta) variant: Evidence from an outbreak in Yunnan, China, 2021. Vaccine. 2022;40(20):2869-2874. [CrossRef]
- Fahmy Esmail O, Drug Authority E, Samar Kabeel E. Effectiveness and safety of inactivated SARS-CoV2 vaccine (BBIBP-CorV) among healthcare workers: A seven-month follow-up study at fifteen hospitals. Published online ,March 11, 2022. [CrossRef]
- Vokó Z, Kiss Z, Surján G, et al. Effectiveness and Waning of Protection With Different SARS-CoV-2 Primary and Booster Vaccines During the Delta Pandemic Wave in 2021 in Hungary (HUN-VE 3 Study). Front Immunol. 2022;13:3609. [CrossRef]
- Heidarzadeh A, Amini Moridani M, Khoshmanesh S, Kazemi S, Hajiaghabozorgi M, Karami M. Effectiveness of COVID-19 vaccines on hospitalization and death in Guilan, Iran: a test-negative case-control study. International Journal of Infectious Diseases. 2023;128:212-222. [CrossRef]
- Nittayasoot N, Suphanchaimat R, Thammawijaya P, et al. Real-World Effectiveness of COVID-19 Vaccines against Severe Outcomes during the Period of Omicron Predominance in Thailand: A Test-Negative Nationwide Case–Control Study. Vaccines 2022, Vol 10, Page 2123. 2022;10(12):2123. [CrossRef]
- The Lancet Infectious Diseases. Why hybrid immunity is so triggering. Lancet Infect Dis. 2022;22(12):1649. [CrossRef]
- Hall V, Foulkes S, Insalata F, et al. Protection against SARS-CoV-2 after Covid-19 Vaccination and Previous Infection. New England Journal of Medicine. 2022;386(13):1207-1220. [CrossRef]
- Rubin-Smith J, Rojas Castro MY, Preza I, et al. Primary Series COVID-19 Vaccine Effectiveness Among Health Care Workers in Albania, February–December 2021. SSRN Electronic Journal. Published online 2022. 20 December. [CrossRef]
- Interim statement on hybrid immunity and increasing population seroprevalence rates. Accessed January 2, 2023. https://www.who.int/news/item/01-06-2022-interim-statement-on-hybrid-immunity-and-increasing-population-seroprevalence-rates. 2 January.
- Dhakal S, Yu T, Yin A, et al. Reconsideration of anti-nucleocapsid IgG antibody as a marker of SARS-CoV-2 infection post-vaccination for mild COVID-19 patients. Open Forum Infect Dis. Published online December 15, 2022. 15 December. [CrossRef]
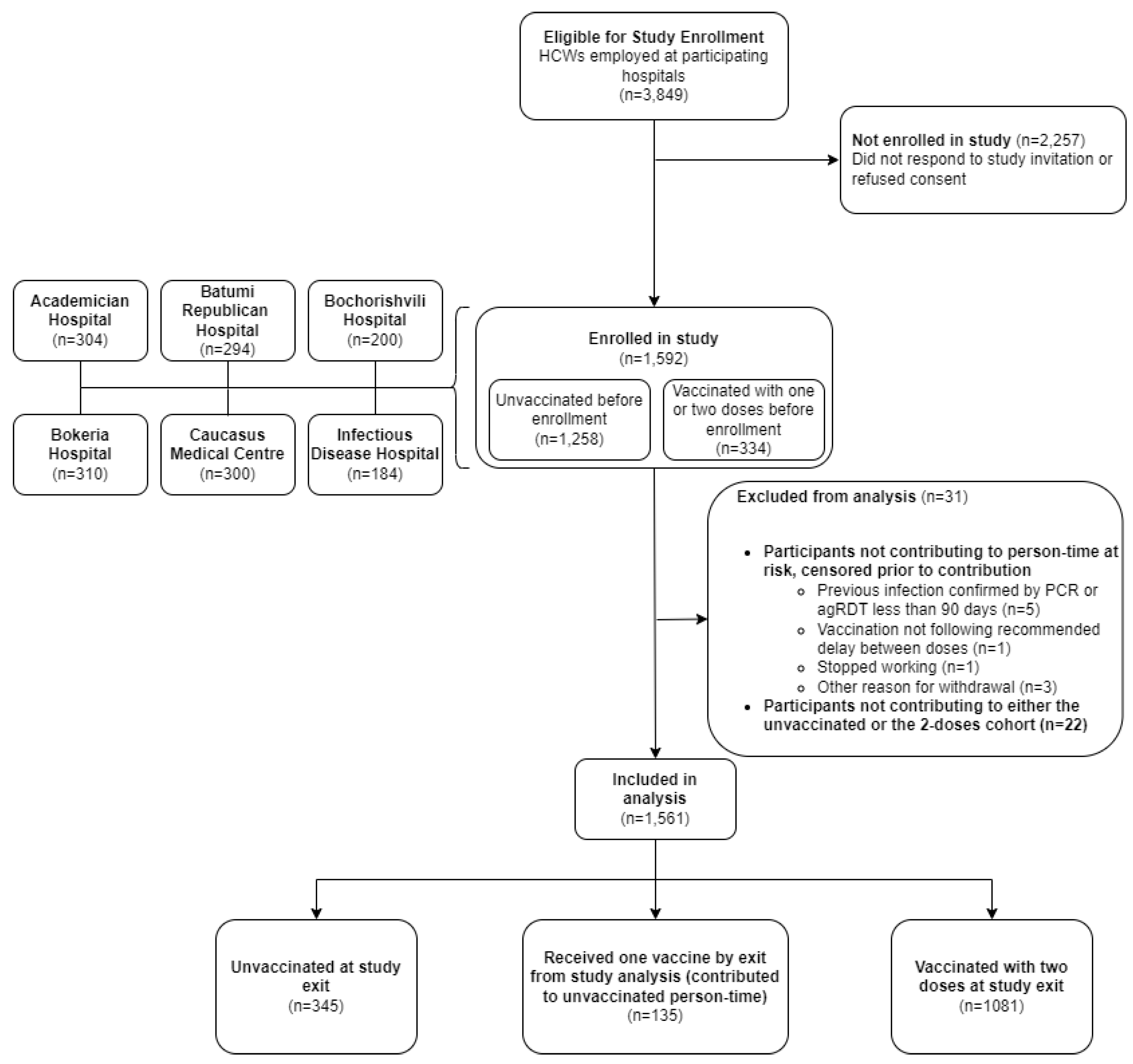
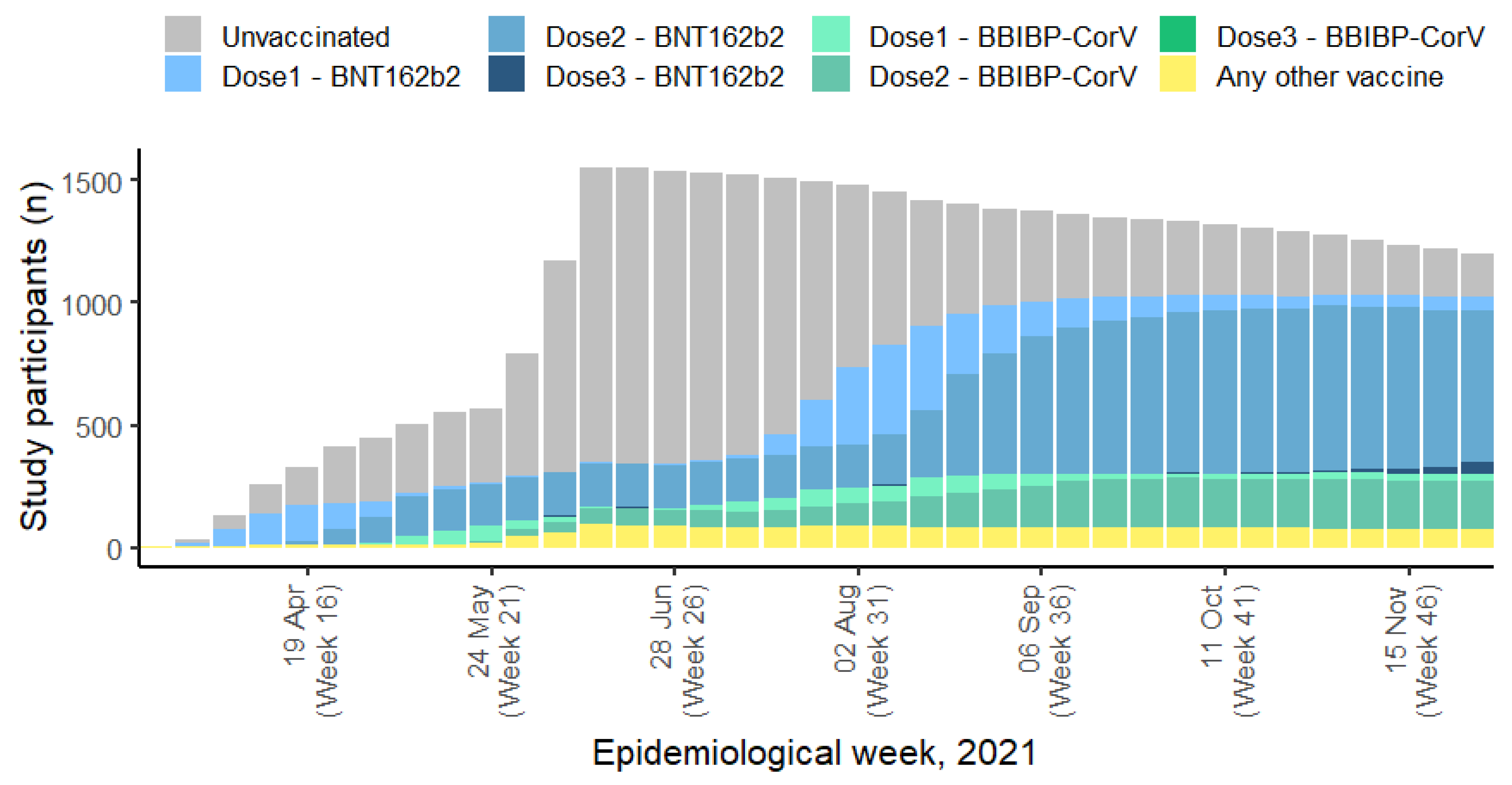
| Characteristic/Category | All Participants, n=1561 | Unvaccinated, n=1300 | Partially vaccinated (1 dose), n=224 | Vaccinated with primary series (2 doses), n=37 |
|---|---|---|---|---|
| Age, n=1561 | ||||
| Median (IQR) | 40 (30-53) | 40 (28.8-52) | 47 (35-56.2) | 40 (31-52) |
| Age group, n=1561 | ||||
| <20, n (%) | 16 (1) | 15 (1) | 1 (<1) | 0 (0) |
| 20-29, n (%) | 373 (24) | 335 (26) | 31 (14) | 7 (19) |
| 30-39, n (%) | 343 (22) | 291 (22) | 42 (19) | 10 (27) |
| 40-49, n (%) | 330 (21) | 270 (21) | 51 (23) | 9 (24) |
| 50-59, n (%) | 314 (20) | 248 (19) | 60 (27) | 6 (16) |
| 60+, n (%) | 185 (12) | 141 (11) | 39 (17) | 5 (14) |
| Sex, n=1561 | ||||
| female, n (%) | 1318 (84) | 1105 (85) | 189 (84) | 24 (65) |
| male, n (%) | 243 (16) | 195 (15) | 35 (16) | 13 (35) |
| Hospital, n=1561 | ||||
| Acad. K Central University Hosp., n (%) | 300 (19) | 240 (18) | 49 (22) | 11 (30) |
| Batumi Republican Hospital, n (%) | 276 (18) | 216 (17) | 52 (23) | 8 (22) |
| Bochorishvili Clinic, n (%) | 194 (12) | 178 (14) | 14 (6) | 2 (5) |
| Bokeria Tbilisi Referral Hospital, n (%) | 309 (20) | 266 (20) | 37 (17) | 6 (16) |
| Caucasus Medical Centre, n (%) | 299 (19) | 251 (19) | 40 (18) | 8 (22) |
| Infectious Disease Hospital, n (%) | 183 (12) | 149 (11) | 32 (14) | 2 (5) |
| Occupation/Role in hospital, n=1561 | ||||
| Nurse or Midwife, n (%) | 604 (39) | 537 (41) | 58 (26) | 9 (24) |
| Medical Doctor, n (%) | 306 (20) | 181 (14) | 108 (48) | 17 (46) |
| Other, n (%) | 651 (42) | 582 (45) | 58 (26) | 11 (30) |
| Household size, n=1561 | ||||
| 1-3, n (%) | 703 (45) | 579 (45) | 107 (48) | 17 (46) |
| 4-5, n (%) | 622 (40) | 526 (40) | 83 (37) | 13 (35) |
| 6+, n (%) | 236 (15) | 195 (15) | 34 (15) | 7 (19) |
| Any chronic condition, n=1561 | ||||
| No, n (%) | 1171 (75) | 988 (76) | 155 (69) | 28 (76) |
| Yes, n (%) | 390 (25) | 312 (24) | 69 (31) | 9 (24) |
| Number of chronic conditions, n=1561 | ||||
| 0, n (%) | 1171 (75) | 988 (76) | 155 (69) | 28 (76) |
| 1, n (%) | 307 (20) | 252 (19) | 48 (21) | 7 (19) |
| ≥2, n (%) | 83 (5) | 60 (5) | 21 (9) | 2 (5) |
| Body mass index, n=1561 | ||||
| Underweight or normal, n (%) | 721 (46) | 607 (47) | 101 (45) | 13 (35) |
| Overweight, n (%) | 481 (31) | 394 (30) | 74 (33) | 13 (35) |
| Obese, n (%) | 359 (23) | 299 (23) | 49 (22) | 11 (30) |
| Smoking, n=1560 | ||||
| Currently smokes, n (%) | 388 (25) | 323 (25) | 48 (21) | 17 (46) |
| Never smokes, n (%) | 1030 (66) | 865 (67) | 148 (66) | 17 (46) |
| Previously smokes, n (%) | 142 (9) | 111 (9) | 28 (12) | 3 (8) |
| Self-assessed health status, n=1561 | ||||
| Excellent, n (%) | 127 (8) | 104 (8) | 17 (8) | 6 (16) |
| Very good, n (%) | 252 (16) | 206 (16) | 37 (17) | 9 (24) |
| Good, n (%) | 521 (33) | 438 (34) | 68 (30) | 15 (41) |
| Fair, n (%) | 641 (41) | 533 (41) | 101 (45) | 7 (19) |
| Poor, n (%) | 20 (1) | 19 (1) | 1 (<1) | 0 (0) |
| Hands on care, n=1561 | ||||
| No, n (%) | 745 (48) | 639 (49) | 91 (41) | 15 (41) |
| Yes, n (%) | 816 (52) | 661 (51) | 133 (59) | 22 (59) |
| Received influenza vaccine during 2020-2021 influenza season, n=1561 | ||||
| No, n (%) | 1068 (68) | 944 (73) | 105 (47) | 19 (51) |
| Yes, n (%) | 492 (32) | 355 (27) | 119 (53) | 18 (49) |
| Face-to-face patient contact, n=1561 | ||||
| No, n (%) | 336 (22) | 292(22) | 37 (17) | 7 (19) |
| Yes, n (%) | 1125 (78) | 1008 (78) | 187 (81) | 30 (81) |
| Previous SARS-CoV-2 infection (before enrollment) confirmed by PCR or RAT, n=1561 | ||||
| 0, n (%) | 814 (52) | 645 (50) | 136 (61) | 33 (89) |
| 1, n (%) | 747 (48) | 655 (50) | 88 (39) | 4 (11) |
| Previous SARS-CoV-2 infection (before enrollment) confirmed by any test: PCR, RAT or serology, n=1561 | ||||
| 0, n (%) | 558 (36) | 437 (34) | 120 (54) | 1 (3) |
| 1, n (%) | 1003 (64) | 863 (66) | 104 (46) | 36 (97) |
| Seropositive at enrolment (AntiS+ or AntiN+), n=1555 | ||||
| 0, n (%) | 569 (37) | 446 (34) | 122 (55) | 1 (3) |
| 1, n (%) | 986 (63) | 850 (66) | 100 (45) | 36 (97) |
| Anti-S+, n=1558 | ||||
| 0, n (%) | 589 (38) | 460 (35) | 124 (56) | 5 (14) |
| 1, n (%) | 969 (62) | 838 (65) | 99 (44) | 32 (86) |
| Anti-N+, n=1552 | ||||
| 0, n (%) | 639 (41) | 502 (39) | 130 (59) | 7 (19) |
| 1, n (%) | 913 (59) | 791 (61) | 92 (41) | 30 (81) |
| Delay between first dose and start of person-time contribution, in days (n=261) | ||||
| Median (IQR) | 2 (1-4) | _ | 2 (1-3) | 30 (29-31) |
| Delay between second dose and start of person-time contribution, in days (n=37) | ||||
| )Median (IQR) | 7 (3-9) | _ | _ | 7 (3-9) |
| COVID-19 Vaccine product received prior to start of person-time contribution, n= 1561 | ||||
| Unvaccinated, n (%) | 1300 (83) | 1300 (100) | 0 (0) | 0 (0) |
| ChAdOx1-S - 2 doses, n (%) | 3 (<1) | 0 (0) | 0 (0) | 3 (8) |
| BNT162b2 - 1 dose, n (%) | 145 (9) | 0 (0) | 145 (65) | 0 (0) |
| BNT162b2 - 2 dose, n (%) | 26 (2) | 0 (0) | 0 (0) | 26 (70) |
| BBIBP-CorV - 1 dose, n (%) | 33 (2) | 0 (0) | 33 (15) | 0 (0) |
| BBIBP-CorV - 2 dose, n (%) | 4 (<1) | 0 (0) | 0 (0) | 4 (11) |
| CoronaVac - 1 dose, n (%) | 28 (2) | 0 (0) | 28 (12) | 0 (0) |
| CoronaVac - 2 doses, n (%) | 4 (<1) | 0 (0) | 0 (0) | 4 (11) |
| N participants | Total person-time (days) | PCR-confirmed symptomatic SARS-CoV-2 infection | RAT- confirmed symptomatic SARS-CoV-2 infection | All symptomatic SARS-CoV-2 infections | Unadjusted HR | (95% CI) | Unadjusted VE | (95%CI) | Adjusted VE | (95%CI) | ||
| Overall study period | Two doses - any vaccine | |||||||||||
| Total cohort | 1561 | |||||||||||
| Unvaccinated | 1300 | 112050 | 90 | 34 | 124 | |||||||
| ≥14d from 2nd dose | 1054 | 105080 | 52 | 15 | 67 | 0.56 | (0.40; 0.80) | 44 | (20; 60) | 58 | (41; 70) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 437 | 36109 | 63 | 25 | 88 | |||||||
| ≥14d from 2nd dose | 357 | 37683 | 40 | 14 | 54 | 0.48 | (0.32; 0.70) | 52 | (30; 68) | 56 | (35; 70) | |
| With Prior Infection | ||||||||||||
| Unvaccinated | 863 | 75941 | 27 | 9 | 36 | |||||||
| ≥14d from 2nd dose | 697 | 67397 | 12 | 1 | 13 | 0.41 | (0.20; 0.84) | 59 | (16; 80) | 58 | (11; 80) | |
| Two doses - BNT162b2 vaccine | ||||||||||||
| Total cohort | 1470 | |||||||||||
| Unvaccinated | 1300 | 112050 | 90 | 34 | 124 | |||||||
| ≥14d from 2nd dose | 732 | 72695 | 25 | 6 | 31 | 0.40 | (0.26; 0.63) | 60 | (37; 74) | 68 | (51; 79) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 437 | 36109 | 63 | 25 | 88 | |||||||
| ≥14d from 2nd dose | 239 | 25763 | 22 | 6 | 28 | 0.37 | (0.24; 0.60) | 63 | (40; 76) | 63 | (43; 77) | |
| Two doses - BBIBP-CorV vaccine | ||||||||||||
| Total cohort | 1337 | |||||||||||
| Unvaccinated | 1300 | 112050 | 90 | 34 | 124 | |||||||
| ≥14d from 2nd dose | 227 | 21136 | 18 | 6 | 24 | 0.87 | (0.52; 1.45) | 13 | (-45; 48) | 40 | (1; 64) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 437 | 36109 | 63 | 25 | 88 | |||||||
| ≥14d from 2nd dose | 91 | 8977 | 15 | 5 | 20 | 0.74 | (0.41; 1.33) | 26 | (-33; 59) | 31 | (-26; 62) | |
| Delta period | Two doses - any vaccine | |||||||||||
| Total cohort | 1556 | |||||||||||
| Unvaccinated | 1162 | 72917 | 67 | 25 | 92 | |||||||
| ≥14d from 2nd dose | 1068 | 96751 | 49 | 15 | 64 | 0.60 | (0.41; 0.86) | 40 | (14; 59) | 52 | (30; 66) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 378 | 21896 | 45 | 19 | 64 | |||||||
| ≥14d from 2nd dose | 339 | 30932 | 36 | 12 | 48 | 0.53 | (0.35; 0.80) | 47 | (20; 65) | 52 | (26; 69) | |
| With Prior Infection | ||||||||||||
| Unvaccinated | 784 | 51021 | 22 | 6 | 28 | |||||||
| ≥14d from 2nd dose | 729 | 65819 | 13 | 3 | 16 | 0.49 | (0.25; 0.98) | 51 | (2; 75) | 47 | (-10; 74) | |
| Two doses - BNT162b2 vaccine | ||||||||||||
| Total cohort | 1371 | |||||||||||
| Unvaccinated | 1162 | 72917 | 67 | 25 | 92 | |||||||
| ≥14d from 2nd dose | 733 | 64109 | 23 | 6 | 29 | 0.46 | (0.28; 0.73) | 54 | (27; 72) | 61 | (38; 75) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 378 | 21896 | 45 | 19 | 64 | |||||||
| ≥14d from 2nd dose | 222 | 19615 | 19 | 5 | 24 | 0.46 | (0.28; 0.77) | 54 | (23; 72) | 55 | (26; 73) | |
| Two doses - BBIBP-CorV vaccine | ||||||||||||
| Total cohort | 1250 | |||||||||||
| Unvaccinated | 1161 | 72906 | 67 | 25 | 92 | |||||||
| ≥14d from 2nd dose | 227 | 20221 | 18 | 6 | 24 | 0.88 | (0.52; 1.5) | 12 | (-50; 48) | 37 | (-9; 63) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 378 | 21896 | 45 | 19 | 64 | |||||||
| ≥14d from 2nd dose | 91 | 8474 | 15 | 5 | 20 | 0.72 | (0.39; 1.31) | 28 | (-31; 61) | 34 | (-23; 64) | |
| N participants | Total person-time (days) | PCR-confirmed symptomatic SARS-CoV-2 infection | RAT-confirmed symptomatic SARS-CoV-2 infection | All symptomatic SARS-CoV-2 infections | Unadjusted HR | (95% CI) | Unadjusted VE | (95%CI) | Adjusted VE | (95%CI) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Overall period | Medically attended SARS-CoV-2 infection | |||||||||||
| Two doses - any vaccine | ||||||||||||
| Total cohort | 1561 | |||||||||||
| Unvaccinated | 1300 | 112050 | 64 | 22 | 86 | |||||||
| ≥14d from 2nd dose | 1054 | 105080 | 37 | 9 | 46 | 0.64 | (0.43; 0.95) | 36 | (5; 57) | 52 | (28; 67) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 437 | 36109 | 47 | 15 | 62 | |||||||
| ≥14d from 2nd dose | 357 | 37683 | 30 | 8 | 38 | 0.55 | (0.35; 0.85) | 45 | (15; 65) | 51 | (21; 70) | |
| With Prior Infection | ||||||||||||
| Unvaccinated | 863 | 75941 | 17 | 7 | 24 | |||||||
| ≥14d from 2nd dose | 697 | 67397 | 7 | 1 | 8 | 0.42 | (0.18; 0.98) | 58 | (2; 82) | 56 | (-2; 81) | |
| Two doses - BNT162b2 | ||||||||||||
| Total cohort | 1470 | |||||||||||
| Unvaccinated | 1300 | 112050 | 64 | 22 | 86 | |||||||
| ≥14d from 2nd dose | 732 | 72695 | 18 | 5 | 23 | 0.49 | (0.30; 0.81) | 51 | (19; 70) | 63 | (39; 78) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 437 | 36109 | 47 | 15 | 62 | |||||||
| ≥14d from 2nd dose | 239 | 25763 | 16 | 5 | 21 | 0.44 | (0.26; 0.75) | 56 | (25; 74) | 54 | (24; 72) | |
| Delta period | Two doses - any vaccine | |||||||||||
| Total cohort | 1556 | |||||||||||
| Unvaccinated | 1162 | 72917 | 45 | 13 | 58 | |||||||
| ≥14d from 2nd dose | 1068 | 96751 | 35 | 9 | 44 | 0.69 | (0.45; 1.08) | 31 | (-8; 55) | 41 | (10; 61) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 378 | 21896 | 31 | 9 | 40 | |||||||
| ≥14d from 2nd dose | 339 | 30932 | 28 | 7 | 35 | 0.68 | (0.42; 1.11) | 32 | (-11; 58) | 35 | (-6; 60) | |
| With Prior Infection | ||||||||||||
| Unvaccinated | 784 | 51021 | 14 | 4 | 18 | |||||||
| ≥14d from 2nd dose | 729 | 65819 | 7 | 2 | 9 | 0.44 | (0.19; 1.02) | 56 | (-2; 81) | 53 | (-13; 80) | |
| Overall period | Hospitalization due to SARS-CoV-2 infection | |||||||||||
| Two doses - any vaccine | ||||||||||||
| Total cohort | 1561 | |||||||||||
| Unvaccinated | 1300 | 112050 | 25 | 8 | 33 | |||||||
| ≥14d from 2nd dose | 1054 | 105080 | 9 | 2 | 11 | 0.46 | (0.22; 0.96) | 54 | (4; 78) | 69 | (36; 85) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 437 | 36109 | 21 | 6 | 27 | |||||||
| ≥14d from 2nd dose | 357 | 37683 | 8 | 2 | 10 | 0.37 | (0.18; 0.78) | 63 | (22; 82) | 66 | (29; 84) | |
| Delta period | Two doses - any vaccine | |||||||||||
| Total cohort | 1556 | |||||||||||
| Unvaccinated | 1162 | 72917 | 13 | 3 | 16 | |||||||
| ≥14d from 2nd dose | 1068 | 96751 | 8 | 2 | 10 | 0.5 | (0.21; 1.17) | 50 | (-17; 79) | 61 | (13; 81) | |
| Without Prior Infection | ||||||||||||
| Unvaccinated | 378 | 21896 | 10 | 2 | 12 | |||||||
| ≥14d from 2nd dose | 339 | 30932 | 7 | 2 | 9 | 0.46 | (0.19; 1.11) | 54 | (-11; 81) | 53 | (-13; 81) | |
| N participants | Total person-time (days) | PCR-confirmed symptomatic SARS-CoV-2 infection | RAT-confirmed symptomatic SARS-CoV-2 infection | All symptomatic SARS-CoV-2 infections | Unadjusted HR | (95% CI) | Unadjusted VE | (95%CI) | Adjusted VE | (95%CI) | ||
| Overall period | Two doses (all vaccines) | 1561 | ||||||||||
| Unvaccinated without previous infection [ref] | 437 | 36109 | 63 | 25 | 88 | |||||||
| Unvaccinated with previous infection | 863 | 75941 | 27 | 9 | 36 | 0.17 | (0.11; 0.25) | 83 | (75; 89) | 85 | (77; 90) | |
| ≥14d from 2nd dose without previous infection | 357 | 37683 | 40 | 14 | 54 | 0.48 | (0.32; 0.70) | 52 | (30; 68) | 56 | (35; 70) | |
| ≥14d from 2nd dose with previous infection | 697 | 67397 | 12 | 1 | 13 | 0.06 | (0.03; 0.13) | 94 | (87; 97) | 95 | (90; 98) | |
| Two doses (BNT162b2) | 1470 | |||||||||||
| Unvaccinated without previous infection [ref] | 437 | 36109 | 63 | 25 | 88 | |||||||
| Unvaccinated with previous infection | 863 | 75941 | 27 | 9 | 36 | 0.17 | (0.11; 0.25) | 83 | (75; 89) | 85 | (77; 90) | |
| ≥14d from 2nd dose without previous infection | 239 | 25763 | 22 | 6 | 28 | 0.37 | (0.24; 0.60) | 63 | (40; 76) | 64 | (43; 77) | |
| Two doses (BBIBP-CorV) | 1337 | |||||||||||
| Unvaccinated without previous infection [ref] | 437 | 36109 | 63 | 25 | 88 | |||||||
| Unvaccinated with previous infection | 863 | 75941 | 27 | 9 | 36 | 0.17 | (0.11; 0.25) | 83 | (75; 89) | 85 | (77; 90) | |
| ≥14d from 2nd dose without previous infection | 91 | 8977 | 15 | 5 | 20 | 0.74 | (0.41; 1.33) | 26 | (-33; 59) | 31 | (-26; 62) | |
| Delta period | Two doses (all vaccines) | 1556 | ||||||||||
| Unvaccinated without previous infection [ref] | 378 | 21896 | 45 | 19 | 64 | |||||||
| Unvaccinated with previous infection | 784 | 51021 | 22 | 6 | 28 | 0.17 | (0.11; 0.27) | 83 | (73; 89) | 86 | (77; 91) | |
| ≥14d from 2nd dose without previous infection | 339 | 30932 | 36 | 12 | 48 | 0.53 | (0.35; 0.80) | 47 | (20; 65) | 52 | (26; 69) | |
| ≥14d from 2nd dose with previous infection | 729 | 65819 | 13 | 3 | 16 | 0.08 | (0.04; 0.16) | 92 | (84; 96) | 93 | (86; 96) | |
| Two doses (BNT162b2)/Previous infection status | 1371 | |||||||||||
| Unvaccinated without previous infection [ref] | 378 | 21896 | 45 | 19 | 64 | |||||||
| Unvaccinated with previous infection | 784 | 51021 | 22 | 6 | 28 | 0.17 | (0.11; 0.27) | 83 | (73; 89) | 86 | (77; 91) | |
| ≥14d from 2nd dose without previous infection | 222 | 19615 | 19 | 5 | 24 | 0.46 | (0.28; 0.77) | 54 | (23; 72) | 55 | (26; 73) | |
| Two doses (BBIBP-CorV) | 1250 | |||||||||||
| Unvaccinated without previous infection [ref] | 378 | 21896 | 45 | 19 | 64 | |||||||
| Unvaccinated with previous infection | 783 | 51010 | 22 | 6 | 28 | 0.17 | (0.11; 0.27) | 83 | (73; 89) | 86 | (77; 91) | |
| ≥14d from 2nd dose without previous infection | 91 | 8474 | 15 | 5 | 20 | 0.72 | (0.39; 1.31) | 28 | (-31; 61) | 34 | (-23; 64) |
| N participants | Total person-time (days) | PCR-confirmed symptomatic SARS-CoV-2 infection | RAT-confirmed symptomatic SARS-CoV-2 infection | All symptomatic SARS-CoV-2 infections | Unadjusted HR | (95% CI) | Unadjusted VE | (95%CI) | Adjusted VE | (95%CI) | |||
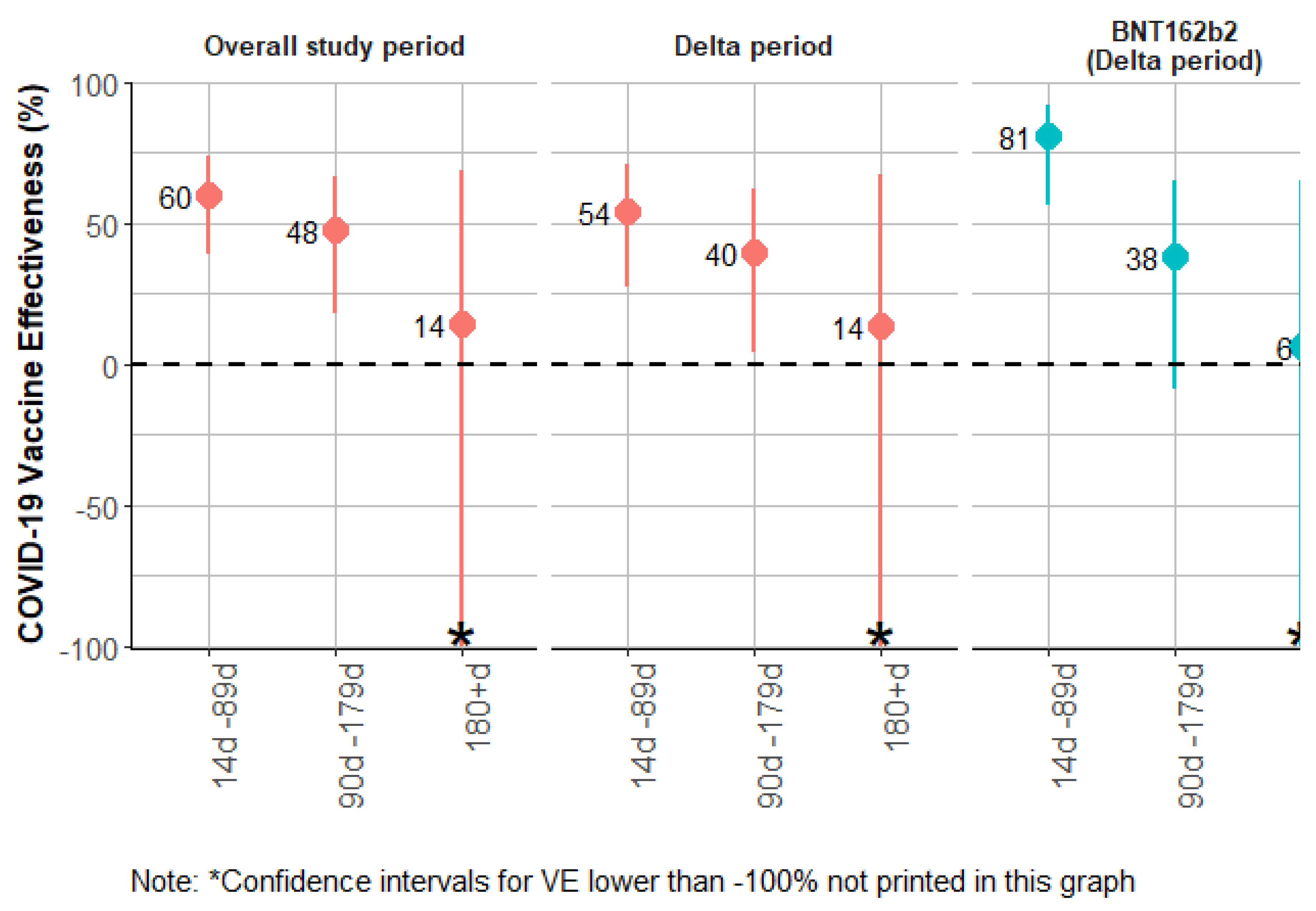
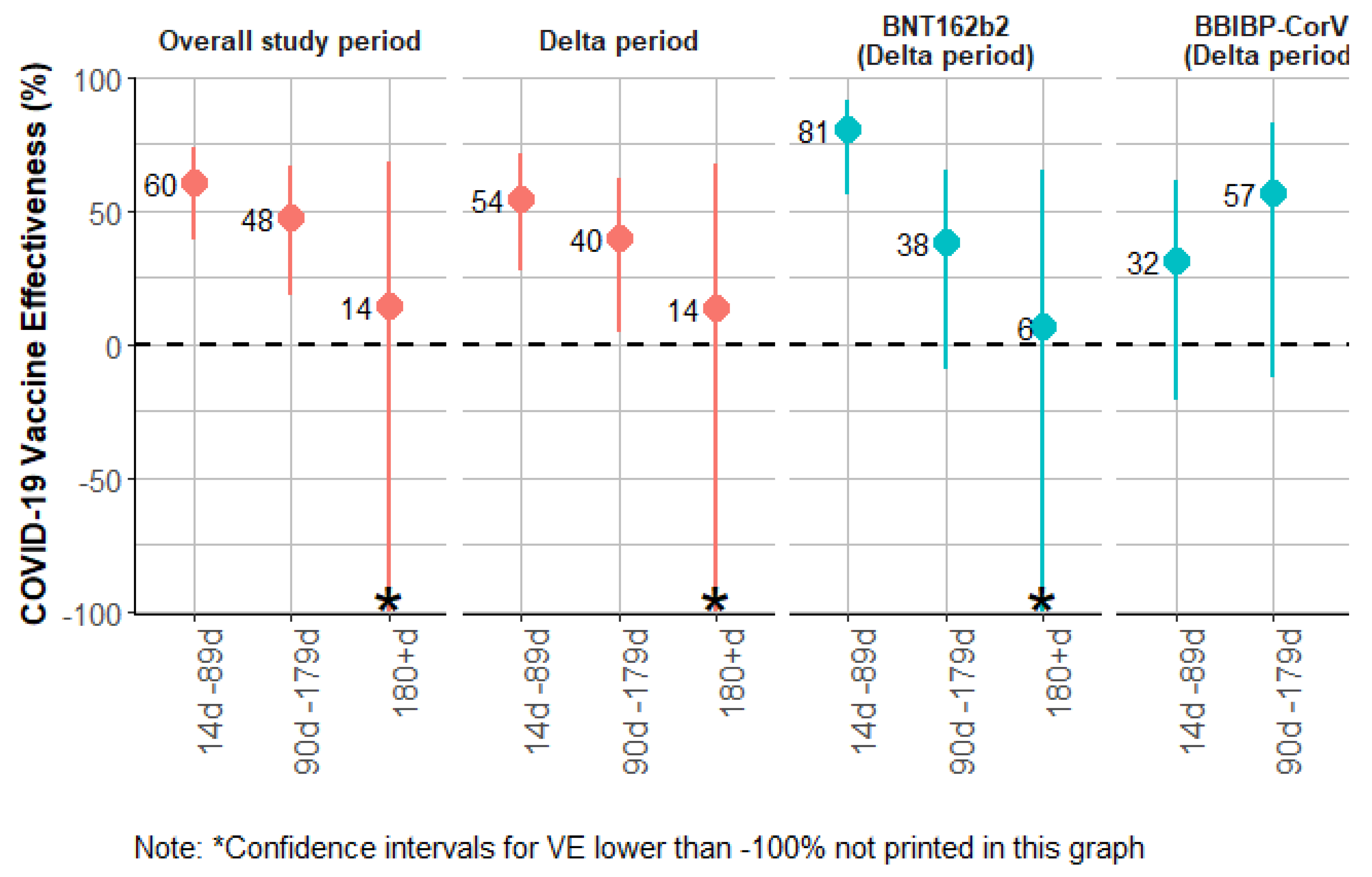
| Overall period | Time since two doses (all vaccines) | 1561 | |||||||||||
| Unvaccinated [ref] | 1300 | 112050 | 90 | 34 | 124 | ||||||||
| 14d - 89d from 2nd dose | 1054 | 71373 | 28 | 7 | 35 | 0.47 | (0.30; 0.72) | 53 | (28; 70) | 60 | (39; 74) | ||
| 90d-179d from 2nd dose | 697 | 29150 | 19 | 7 | 26 | 0.73 | (0.46; 1.16) | 27 | (-16; 54) | 48 | (18; 66) | ||
| ≥180d from 2nd dose | 186 | 4557 | 5 | 1 | 6 | 0.92 | (0.36; 2.38) | 8 | (-138; 64) | 14 | (-134; 68) | ||
| Time since two doses (BNT162b2) | 1470 | ||||||||||||
| Unvaccinated [ref] | 1300 | 112050 | 90 | 34 | 124 | ||||||||
| 14d - 89d from 2nd dose | 732 | 49837 | 9 | 1 | 10 | 0.21 | (0.1; 0.44) | 79 | (56; 90) | 82 | (63; 91) | ||
| 90d-179d from 2nd dose | 485 | 18572 | 11 | 4 | 15 | 0.71 | (0.40; 1.24) | 29 | (-24; 60) | 52 | (13; 73) | ||
| ≥180d from 2nd dose | 151 | 4286 | 5 | 1 | 6 | 0.95 | (0.37; 2.48) | 5 | (-148; 63) | 40 | (-55; 77) | ||
| Time since two doses (BBIBP-CorV) | 1337 | ||||||||||||
| Unvaccinated [ref] | 1300 | 112050 | 90 | 34 | 124 | ||||||||
| 14d - 89d from 2nd dose | 227 | 14894 | 13 | 5 | 18 | 0.85 | (0.49; 1.50) | 15 | (-50; 51) | 36 | (-10; 63) | ||
| 90d-179d from 2nd dose | 132 | 6108 | 5 | 1 | 6 | 0.76 | (0.30; 1.91) | 24 | (-91; 70) | 57 | (-9; 83) | ||
| Delta period | Time since two doses (all vaccines) | 1556 | |||||||||||
| Unvaccinated [ref] | 1162 | 72917 | 67 | 25 | 92 | ||||||||
| 14d - 89d from 2nd dose | 1043 | 61860 | 25 | 7 | 32 | 0.47 | (0.30; 0.75) | 53 | (25; 70) | 54 | (28; 71) | ||
| 90d-179d from 2nd dose | 715 | 30230 | 19 | 7 | 26 | 0.74 | (0.46; 1.21) | 26 | (-21; 54) | 40 | (4; 62) | ||
| ≥180d from 2nd dose | 189 | 4661 | 5 | 1 | 6 | 1 | (0.38; 2.60) | -0 | (-160; 62) | 14 | (-130; 68) | ||
| Time since two doses (BNT162b2) | 1371 | ||||||||||||
| Unvaccinated [ref] | 1162 | 72917 | 67 | 25 | 92 | ||||||||
| 14d - 89d from 2nd dose | 708 | 41088 | 7 | 1 | 8 | 0.21 | (0.09; 0.49) | 79 | (51; 91) | 81 | (56; 91) | ||
| 90d-179d from 2nd dose | 489 | 18631 | 11 | 4 | 15 | 0.77 | (0.44; 1.37) | 23 | (-37; 56) | 38 | (-9; 65) | ||
| ≥180d from 2nd dose | 154 | 4390 | 5 | 1 | 6 | 1.03 | (0.39; 2.72) | -3 | (-172; 61) | 6 | (-151;65) | ||
| Time since two doses (BBIBP-CorV) | 1250 | ||||||||||||
| Unvaccinated [ref] | 1161 | 72906 | 67 | 25 | 92 | ||||||||
| 14d - 89d from 2nd dose | 227 | 13979 | 13 | 5 | 18 | 0.91 | (0.52; 1.59) | 9 | (-59; 48) | 32 | (-21; 61) | ||
| 90d-179d from 2nd dose | 132 | 6108 | 5 | 1 | 6 | 0.75 | (0.30; 1.91) | 25 | (-91; 70) | 57 | (-12; 83) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




