Submitted:
19 September 2023
Posted:
21 September 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Detection of Novel Stx2 subtypes
2.2. Collection of STEC strains
2.3. Illumina and Oxford Nanopore Sequencing and Assembly
2.4. WGS based Characterization
2.5. Stx subtyping
2.6. Cytotoxicity, Ciprofloxacin (Cip) induction, and activation assays
2.7. Polymerase Chain Reaction (PCR) primers to detect all described stx1 and stx2 subtypes
| Primer Name | Primer Sequence | Amplicon Size (bp) | Reference |
|---|---|---|---|
| stx2-PS8-F | 5’-TCACYGGTTTCATCATATCTGG | 400 | This study |
| stx2-PS7-R | 5’-GCCTGTCBCCASTTATCTGACA | ||
| PS19 stx2f-F | 5’-GTACAGGGATGCAGATTGGGCG | 438 | This study |
| PS20 stx2f -R | 5’-CTTTAATGGCCGCCCTGTCTCC | ||
| PS17 eae-F | 5’-CGGCTATTTCCGCATGAGCGG | 223 | This study |
| PS18 eae-R-NEW | 5´AGTTDACACCAAYWGTCRCCGC | ||
| stx1 F3b | 5’-CTGATGATTGATAGTGGCACAGG | 283 | This study |
| stx1 OMNI-R1 | 5’-GCGATTTATCTGCATCCCCGTAC |
2.8. Stx2n and Stx2o pro-phages annotation and discovery
2.9. Data availability
3. Results
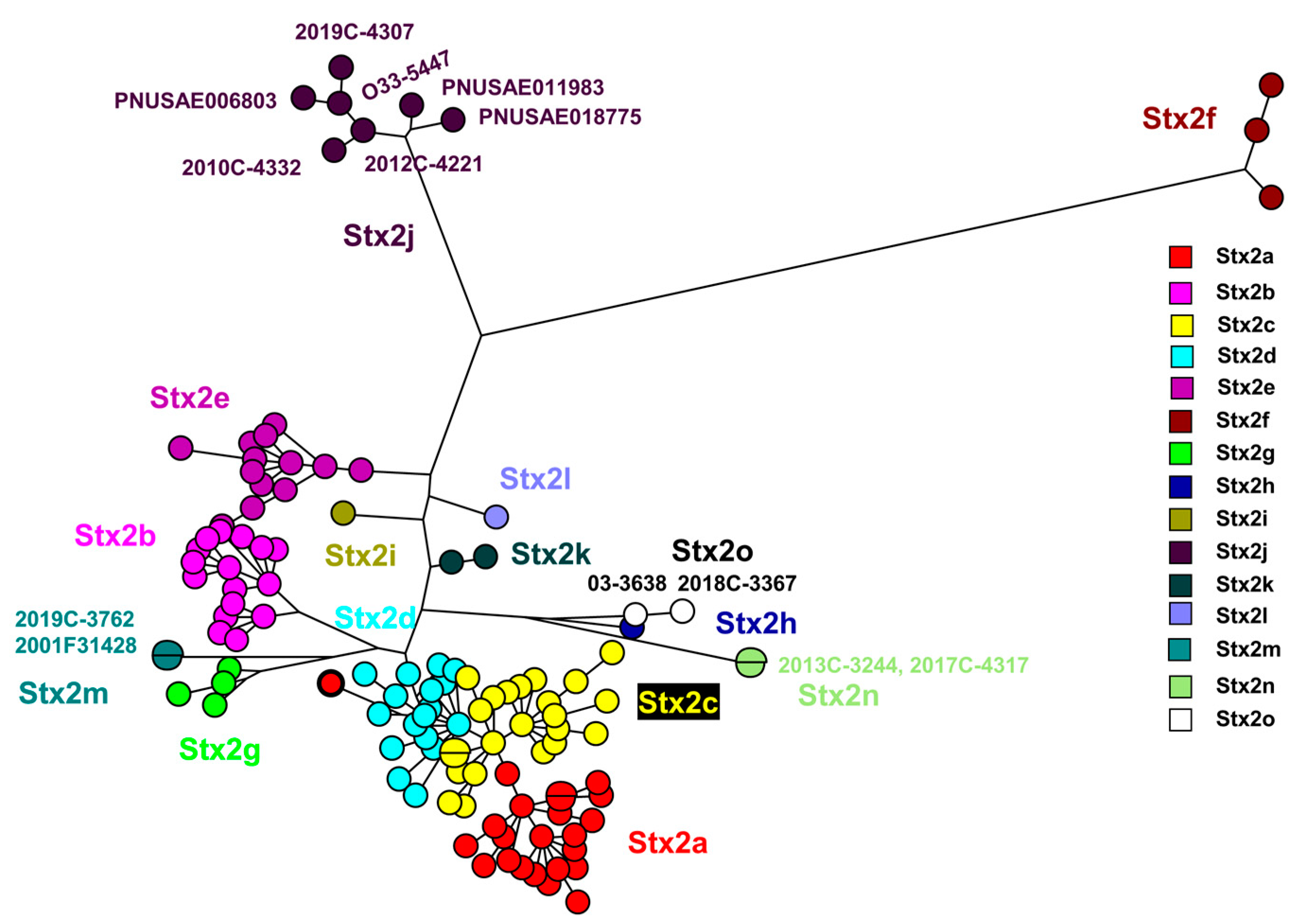
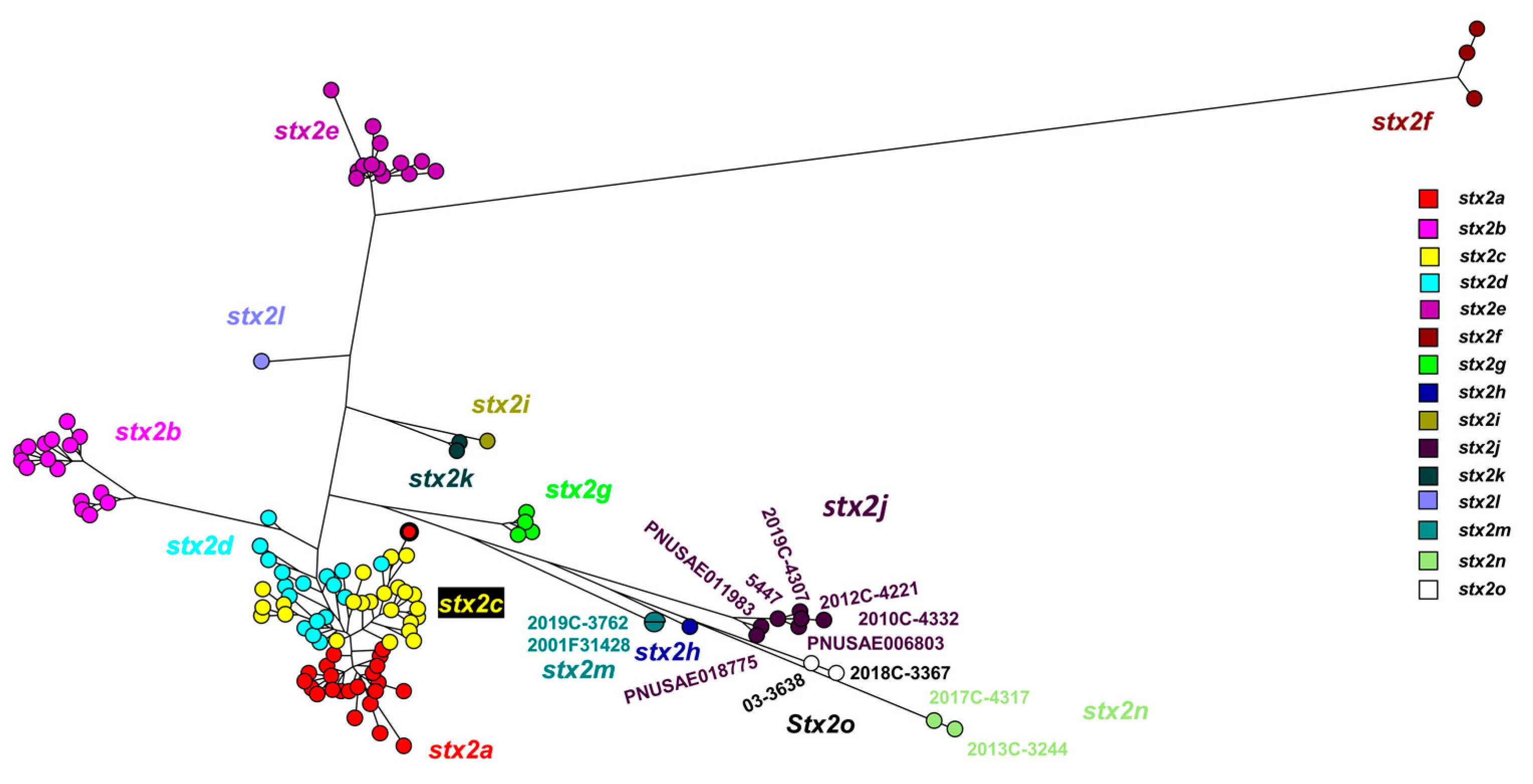
3.1. Identification of two Novel Stx2 Subtypes
3.2. WGS based Characterization of Stx2n and a Stx2o strains
| Stx Subtype | CDC isolate ID | BioSample | ST type | O:(K):H type | PNID | Assembly |
|---|---|---|---|---|---|---|
| stx2j | 2010C-4332 | SAMN04377066 | 5662 | O158:H23 | PNUSAE001889 | GCA_012764415.1 |
| stx2j | 2012C-4221 | SAMN08579578 | 5350 | O162:H6 | None | GCA_003018235.1 |
| stx2j | 2019C-4307 | SAMN12361752 | 5736 | O32:K87:H2 | PNUSAE027323 | GCA_011901845.1 |
| stx2j | See PNUSAE | SAMN10170522 | 491 | ONT:H45 | PNUSAE018775 | GCA_003903075.2 |
| stx2j | See PNUSAE | SAMN07177511 | 5923 | O33:H14 | PNUSAE006803 | GCA_012463025.1 |
| stx2j | See PNUSAE | SAMN08595463 | 657 | O183: H18 | PNUSAE011983 | GCA_012253565.1 |
| stx2m | 2019C-3762 | SAMN11569941 | 9312 | O38:H39 | PNUSAE024072 | GCA_011950125.1 |
| Stx2n* | 2013C-3244 | SAMN04578435 | 1385 | O1:K22:H4 1 | None | GCA_012711215.22 |
| Stx2n* | 2017C-4317 | SAMN07709929 | 70 | O23:H15 | PNUSAE009425 | GCA_013342905.22 |
| Stx2o | 2018C-3367 | SAMN08799860 | 80 | O75:H7 | PNUSAE012694 | GCA_012224845.22 |
3.3. Detection of Shiga Toxin Production
3.4. Design and testing of new OMNI PCR primers
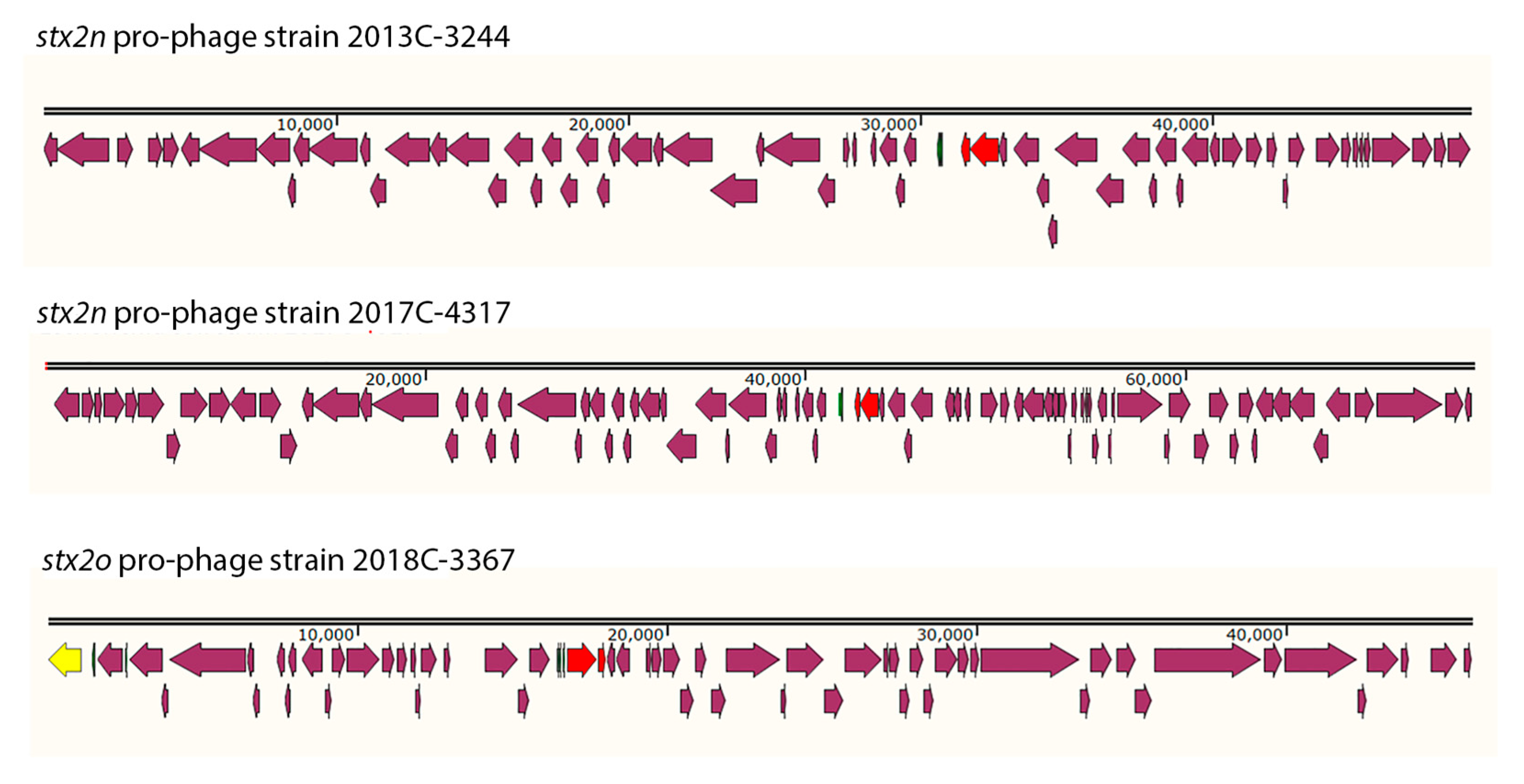
3.5. Identification of the Stx2n and Stx2o pro-phages in the strains from this study
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Gill, A.; Dussault, F.; McMahon, T.; Petronella, N.; Wang, X.; Cebelinski, E.; Scheutz, F.; Weedmark, K.; Blais, B.; Carrillo, C. Characterization of Atypical Shiga Toxin Gene Sequences and Description of Stx2j, a New Subtype. J Clin Microbiol 2022, 60, e0222921. [Google Scholar] [CrossRef]
- Bai, X.; Scheutz, F.; Dahlgren, H.M.; Hedenstrom, I.; Jernberg, C. Characterization of Clinical Escherichia coli Strains Producing a Novel Shiga Toxin 2 Subtype in Sweden and Denmark. Microorganisms 2021, 9. [Google Scholar] [CrossRef]
- European Food Safety, A.; European Centre for Disease, P. ; Control. The European Union One Health 2020 Zoonoses Report. EFSA J 2021, 19, e06971. [Google Scholar] [CrossRef]
- Scheutz, F.; Teel, L.D.; Beutin, L.; Pierard, D.; Buvens, G.; Karch, H.; Mellmann, A.; Caprioli, A.; Tozzoli, R.; Morabito, S.; et al. Multicenter evaluation of a sequence-based protocol for subtyping Shiga toxins and standardizing Stx nomenclature. J Clin Microbiol 2012, 50, 2951–2963. [Google Scholar] [CrossRef]
- Bai, X.; Fu, S.; Zhang, J.; Fan, R.; Xu, Y.; Sun, H.; He, X.; Xu, J.; Xiong, Y. Identification and pathogenomic analysis of an Escherichia coli strain producing a novel Shiga toxin 2 subtype. Sci Rep 2018, 8, 6756. [Google Scholar] [CrossRef] [PubMed]
- Lacher, D.W.; Gangiredla, J.; Patel, I.; Elkins, C.A.; Feng, P.C. Use of the Escherichia coli Identification Microarray for Characterizing the Health Risks of Shiga Toxin-Producing Escherichia coli Isolated from Foods. J Food Prot 2016, 79, 1656–1662. [Google Scholar] [CrossRef]
- Yang, X.; Bai, X.; Zhang, J.; Sun, H.; Fu, S.; Fan, R.; He, X.; Scheutz, F.; Matussek, A.; Xiong, Y. Escherichia coli strains producing a novel Shiga toxin 2 subtype circulate in China. Int J Med Microbiol 2020, 310, 151377. [Google Scholar] [CrossRef] [PubMed]
- Panel, E.B.; Koutsoumanis, K.; Allende, A.; Alvarez-Ordóñez, A.; Bover-Cid, S.; Chemaly, M.; Davies, R.; De Cesare, A.; Herman, L.; Hilbert, F.; et al. Pathogenicity assessment of Shiga toxin-producing Escherichia coli (STEC) and the public health risk posed by contamination of food with STEC. EFSA Journal 2020, 18, e05967. [Google Scholar] [CrossRef]
- Feldgarden, M.; Brover, V.; Gonzalez-Escalona, N.; Frye, J.G.; Haendiges, J.; Haft, D.H.; Hoffmann, M.; Pettengill, J.B.; Prasad, A.B.; Tillman, G.E.; et al. AMRFinderPlus and the Reference Gene Catalog facilitate examination of the genomic links among antimicrobial resistance, stress response, and virulence. Sci Rep 2021, 11, 12728. [Google Scholar] [CrossRef] [PubMed]
- Souvorov, A.; Agarwala, R.; Lipman, D.J. SKESA: strategic k-mer extension for scrupulous assemblies. Genome Biol 2018, 19, 153. [Google Scholar] [CrossRef]
- Souvorov, A.; Agarwala, R. SAUTE: sequence assembly using target enrichment. BMC Bioinformatics 2021, 22, 375. [Google Scholar] [CrossRef] [PubMed]
- Tolar, B.; Joseph, L.A.; Schroeder, M.N.; Stroika, S.; Ribot, E.M.; Hise, K.B.; Gerner-Smidt, P. An Overview of PulseNet USA Databases. Foodborne Pathog Dis 2019, 16, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Stevens, E.L.; Carleton, H.A.; Beal, J.; Tillman, G.E.; Lindsey, R.L.; Lauer, A.C.; Pightling, A.; Jarvis, K.G.; Ottesen, A.; Ramachandran, P.; et al. Use of Whole Genome Sequencing by the Federal Interagency Collaboration for Genomics for Food and Feed Safety in the United States. J Food Prot 2022, 85, 755–772. [Google Scholar] [CrossRef]
- Poates, A.; Truong, J.; Lindsey, R.; Griswold, T.; Williams-Newkirk, A.J.; Carleton, H.; Trees, E. Sequencing of Enteric Bacteria: Library Preparation Procedure Matters for Accurate Identification and Characterization. Foodborne Pathog Dis 2022, 19, 569–578. [Google Scholar] [CrossRef]
- Kolmogorov, M.; Yuan, J.; Lin, Y.; Pevzner, P.A. Assembly of long, error-prone reads using repeat graphs. Nat Biotechnol 2019, 37, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Wick, R.R.; Judd, L.M.; Gorrie, C.L.; Holt, K.E. Unicycler: Resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput Biol 2017, 13, e1005595. [Google Scholar] [CrossRef] [PubMed]
- Darling, A.C.; Mau, B.; Blattner, F.R.; Perna, N.T. Mauve: multiple alignment of conserved genomic sequence with rearrangements. Genome Res 2004, 14, 1394–1403. [Google Scholar] [CrossRef] [PubMed]
- Tatusova, T.; DiCuccio, M.; Badretdin, A.; Chetvernin, V.; Nawrocki, E.P.; Zaslavsky, L.; Lomsadze, A.; Pruitt, K.D.; Borodovsky, M.; Ostell, J. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res 2016, 44, 6614–6624. [Google Scholar] [CrossRef] [PubMed]
- Joensen, K.G.; Tetzschner, A.M.; Iguchi, A.; Aarestrup, F.M.; Scheutz, F. Rapid and Easy In Silico Serotyping of Escherichia coli Isolates by Use of Whole-Genome Sequencing Data. J Clin Microbiol 2015, 53, 2410–2426. [Google Scholar] [CrossRef]
- Joensen, K.G.; Scheutz, F.; Lund, O.; Hasman, H.; Kaas, R.S.; Nielsen, E.M.; Aarestrup, F.M. Real-time whole-genome sequencing for routine typing, surveillance, and outbreak detection of verotoxigenic Escherichia coli. J Clin Microbiol 2014, 52, 1501–1510. [Google Scholar] [CrossRef] [PubMed]
- Sabol, A.; Joung, Y.J.; VanTubbergen, C.; Ale, J.; Ribot, E.M.; Trees, E. Assessment of Genetic Stability During Serial In Vitro Passage and In Vivo Carriage. Foodborne Pathog Dis 2021, 18, 894–901. [Google Scholar] [CrossRef]
- Wirth, T.; Falush, D.; Lan, R.; Colles, F.; Mensa, P.; Wieler, L.H.; Karch, H.; Reeves, P.R.; Maiden, M.C.; Ochman, H.; et al. Sex and virulence in Escherichia coli: an evolutionary perspective. Mol Microbiol 2006, 60, 1136–1151. [Google Scholar] [CrossRef]
- Hauser, J.R.; Atitkar, R.R.; Petro, C.D.; Lindsey, R.L.; Strockbine, N.; O'Brien, A.D.; Melton-Celsa, A.R. The Virulence of Escherichia coli O157:H7 Isolates in Mice Depends on Shiga Toxin Type 2a (Stx2a)-Induction and High Levels of Stx2a in Stool. Front Cell Infect Microbiol 2020, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Melton-Celsa, A.R.; Darnell, S.C.; O'Brien, A.D. Activation of Shiga-like toxins by mouse and human intestinal mucus correlates with virulence of enterohemorrhagic Escherichia coli O91:H21 isolates in orally infected, streptomycin-treated mice. Infect Immun 1996, 64, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Seemann, T. Prokka: rapid prokaryotic genome annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef]
- Meng, Q.; Bai, X.; Zhao, A.; Lan, R.; Du, H.; Wang, T.; Shi, C.; Yuan, X.; Bai, X.; Ji, S.; et al. Characterization of Shiga toxin-producing Escherichia coli isolated from healthy pigs in China. BMC Microbiol 2014, 14, 5. [Google Scholar] [CrossRef] [PubMed]
- Malberg Tetzschner, A.M.; Johnson, J.R.; Johnston, B.D.; Lund, O.; Scheutz, F. In Silico Genotyping of Escherichia coli Isolates for Extraintestinal Virulence Genes by Use of Whole-Genome Sequencing Data. J Clin Microbiol 2020, 58. [Google Scholar] [CrossRef]
- Zhang, Y.; Liao, Y.T.; Salvador, A.; Sun, X.; Wu, V.C.H. Prediction, Diversity, and Genomic Analysis of Temperate Phages Induced From Shiga Toxin-Producing Escherichia coli Strains. Front Microbiol 2019, 10, 3093. [Google Scholar] [CrossRef]
- Scheutz, F.; Nielsen, E.M.; Frimodt-Moller, J.; Boisen, N.; Morabito, S.; Tozzoli, R.; Nataro, J.P.; Caprioli, A. Characteristics of the enteroaggregative Shiga toxin/verotoxin-producing Escherichia coli O104:H4 strain causing the outbreak of haemolytic uraemic syndrome in Germany, May to June 2011. Euro Surveill 2011, 16. [Google Scholar] [CrossRef]
- O'Brien AD, Newland JW, Miller SF, Holmes RK, Smith HW, Formal SB. Shiga-like toxin-converting phages from Escherichia coli strains that cause hemorrhagic colitis or infantile diarrhea. Science. 1984, 226, 694–696. [Google Scholar] [CrossRef]
- Koch C, Hertwig S, Lurz R, Appel B, Beutin L. Isolation of a lysogenic bacteriophage carrying the stx1OX3 gene, which is closely associated with Shiga toxin-producing Escherichia coli strains from sheep and humans. J. Clin. Microbiol. 2001, 39, 3992–3998. [Google Scholar] [CrossRef]
- Paton AW, Beutin L, Paton JC. Heterogeneity of the amino-acid sequences of Escherichia coli Shiga-like toxin type-I operons. Gene. 1995, 153, 71–74. [Google Scholar] [CrossRef]
- Bürk C, Dietrich R, Açar G, Moravek M, Bülte M, Märtlbauer E. Identification and characterization of a new variant of Shiga toxin 1 in Escherichia coli ONT:H19 of bovine origin. J Clin Microbiol. 2003, 41, 2106–2112. [Google Scholar] [CrossRef] [PubMed]
- Piérard D, Muyldermans G, Moriau L, Stevens D, Lauwers S. Identification of new verocytotoxin type 2 variant B-subunit genes in human and animal Escherichia coli isolates. J Clin Microbiol. 1998, 36, 3317–3322. [Google Scholar] [CrossRef] [PubMed]
- Paton AW, Paton JC, Heuzenroeder MW, Goldwater PN, Manning PA. Cloning and nucleotide sequence of a variant Shiga-like toxin II gene from Escherichia coli OX3:H21 isolated from a case of sudden infant death syndrome. Microb Pathog. 1992, 13, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Persson S, Olsen KE, Ethelberg S, Scheutz F. Subtyping method for Escherichia coli shiga toxin (verocytotoxin) 2 variants and correlations to clinical manifestations. J Clin Microbiol. 2007, 45, 2020–2024. [Google Scholar] [CrossRef]
- Weinstein DL, Jackson MP, Samuel JE, Holmes RK, O'Brien AD. Cloning and sequencing of a Shiga-like toxin type II variant from Escherichia coli strain responsible for edema disease of swine. J Bacteriol. 1988, 170, 4223–4230. [Google Scholar] [CrossRef]
- Schmidt H, Scheef J, Morabito S, Caprioli A, Wieler LH, Karch H. A new Shiga toxin 2 variant (Stx2f) from Escherichia coli isolated from pigeons. Appl Environ Microbiol. 2000, 66, 1205–1208. [Google Scholar] [CrossRef]
- Leung PH, Peiris JS, Ng WW, Robins-Browne RM, Bettelheim KA, Yam WC. A newly discovered verotoxin variant, VT2g, produced by bovine verocytotoxigenic Escherichia coli. Appl Environ Microbiol. 2003, 69, 7549–7553. [Google Scholar] [CrossRef]
- Meng Q, Bai X, Zhao A, Lan R, Du H, Wang T, Shi C, Yuan X, Bai X, Ji S, Jin D, Yu B, Wang Y, Sun H, Liu K, Xu J, Xiong Y. Characterization of Shiga toxin-producing Escherichia coli isolated from healthy pigs in China. BMC Microbiol. 2014, 14, 5. [Google Scholar] [CrossRef]
- Arndt, D. ,Grant, J.R.; Marcu, A.,Sajed, T.,Pon, A.,Liang, Y., Wishart, D.S. PHASTER: a better, faster version of the PHAST phage search tool. Nucleic Acids Res 2016, 44, W16–W21. [Google Scholar] [CrossRef] [PubMed]
| Nucleotide\Amino acid | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| stx2a | stx2b | stx2c | stx2d | stx2e | stx2f | stx2g | stx2h | stx2i | stx2j | stx2k | stx2l | stx2m | stx2n | stx2o | |
| 1. Stx2a | 91,9 | 98,4 | 96,9 | 92,2 | 70,8 | 94,2 | 91,7 | 93,2 | 89,5 | 94,4 | 95,5 | 93,2 | 87,9 | 89,7 | |
| 2. Stx2b | 95,3 | 92,2 | 93,4 | 89,4 | 70,6 | 91,4 | 92,2 | 89,2 | 89,2 | 91,3 | 90,0 | 90,6 | 87,8 | 90,2 | |
| 3. Stx2c | 99,2 | 95,4 | 97,4 | 91,9 | 70,5 | 93,1 | 91,7 | 92,4 | 89,6 | 94,7 | 94,8 | 92,2 | 88,1 | 89,7 | |
| 4. Stx2d | 98,4 | 96,0 | 98,5 | 92,1 | 70,5 | 94,0 | 92,2 | 92,9 | 90,2 | 96,1 | 94,8 | 91,5 | 88,2 | 90,2 | |
| 5. Stx2e | 95,2 | 94,2 | 94,9 | 95,7 | 74,8 | 92,2 | 90,2 | 94,7 | 88,3 | 93,8 | 95,0 | 88,9 | 86,9 | 88,8 | |
| 6. Stx2f | 81,9 | 81,2 | 81,4 | 82,0 | 84,1 | 71,4 | 71,2 | 71,3 | 71,1 | 71,0 | 71,5 | 71,1 | 72,2 | 70,4 | |
| 7. Stx2g | 97,0 | 95,1 | 96,5 | 97,1 | 95,9 | 82,2 | 91,9 | 94,5 | 88,2 | 92,9 | 92,8 | 91,2 | 87,0 | 89,1 | |
| 8. Stx2h | 95,2 | 95,3 | 95,0 | 95,6 | 94,4 | 81,8 | 95,5 | 92,0 | 92,1 | 92,8 | 91,1 | 92,1 | 91,4 | 94,1 | |
| 9. Stx2i | 95,9 | 93,7 | 95,4 | 96,1 | 97,3 | 82,3 | 96,5 | 95,4 | 88,6 | 96,5 | 95,1 | 90,0 | 88,1 | 89,4 | |
| 10. Stx2j | 93,5 | 93,2 | 93,7 | 94,2 | 92,6 | 82,7 | 92,7 | 93,5 | 92,5 | 90,6 | 89,8 | 89,2 | 88,1 | 90,9 | |
| 11.Stx2k | 97,2 | 95,4 | 97,3 | 98,2 | 97,2 | 82,2 | 96,8 | 95,9 | 97,9 | 94,1 | 95,5 | 90,9 | 88,1 | 90,3 | |
| 12.Stx2l | 96,8 | 94,1 | 96,4 | 97,1 | 97,3 | 82,3 | 96,2 | 94,7 | 97,3 | 93,9 | 97,8 | 89,9 | 87,4 | 89,9 | |
| 13.Stx2m | 95,7 | 95,1 | 95,3 | 95,5 | 94,3 | 82,0 | 96,1 | 94,8 | 94,5 | 92,1 | 95,0 | 93,8 | 87,7 | 88,9 | |
| 14.Stx2n | 93,2 | 93,1 | 93,4 | 93,6 | 92,6 | 83,9 | 93,5 | 95,0 | 93,1 | 91,8 | 93,9 | 92,4 | 93,2 | 91,4 | |
| 15.Stx2o | 94,3 | 94,0 | 94,5 | 94,7 | 93,6 | 81,8 | 94,5 | 96,9 | 94,3 | 92,8 | 95,2 | 93,8 | 93,8 | 94,6 |
| Virulence gene | Function | 2013C-3244 | 2017C-4317 | 2018C-3367 | |
|---|---|---|---|---|---|
| stx2n | Shiga toxin 2 | + | + | - | |
| stx2o | Shiga toxin 2 | - | - | + | |
| chuA | Outer membrane hemin receptor | + | + | + | |
| focC | S fimbrial/F1C minor subunit | + | - | + | |
| fyuA* | Siderophore receptor | + | - | + | |
| gad | Glutamate decarboxylase | + | + | + | |
| iroN | Enterobactin siderophore receptor protein | + | - | + | |
| irp2 | High molecular weight protein 2 non-ribosomal peptide synthetase | + | - | + | |
| iss | Increased serum survival | + | - | + | |
| kpsE | Capsule polysaccharide export inner-membrane protein | + | + | + | |
| kpsMII_K5 | Polysialic acid transport protein; Group 2 capsule | + | + | + | |
| ompT | Outer membrane protease (protein protease 7) | + | - | + | |
| sfaD | S fimbrial/F1C minor subunit | + | - | + | |
| sfaS | S-fimbriae minor subunit | + | - | - | |
| sitA | Iron transport protein | + | + | + | |
| tcpC | Tir domain-containing protein | + | - | - | |
| terC | Tellurium ion resistance protein | + | + | + | |
| vat* | Vacuolating autotransporter toxin | + | - | + | |
| yfcV* | Fimbrial protein | + | - | + | |
| traT | Outer membrane protein complement resistance | - | + | - | |
| eilA | Salmonella HilA homolog | - | + | - | |
| neuC | Polysialic acid capsule biosynthesis protein | - | - | + | |
| clbB | Hybrid non-ribosomal peptide / polyketide megasynthase | - | - | + | |
| cnf1 | Cytotoxic necrotizing factor | - | - | + | |
| hra | Heat-resistant agglutinin | - | - | + | |
| ibeA | Invasin of brain endothelial cells | - | - | + | |
| mchB | Microcin H47 part of colicin H | - | - | + | |
| mchC | MchC protein | - | - | + | |
| mchF | ABC transporter protein MchF | - | - | + | |
| mcmA | Microcin M part of colicin H | - | - | + | |
| pic | serine protease autotransporters of Enterobacteriaceae (SPATE) | - | - | + | |
| usp | Uropathogenic specific protein | - | - | + | |
| Pathotype | STEC/UPECHM | STEC | STEC/UPECHM | ||
| Stx Subtype | CDC isolate ID | Log CD50/mL supernatant | PCR confirmation | ciprofloxacin induction |
|---|---|---|---|---|
| stx2j | 2010C-4332 | 4.6 | + | No |
| stx2j | 2012C-4221 | 4.5 | + | Yes# |
| stx2j | 2019C-4307 | 3.4 | + | No |
| stx2m | 2019C-3762 | 3.2 | + | No |
| Stx2n | 2013C-3244 | 3.2 | + | No |
| Stx2n | 2017C-4317 | 2.0^ | + | No |
| Stx2o | 2018C-3367 | 3.0 | + | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




