Submitted:
21 August 2023
Posted:
25 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
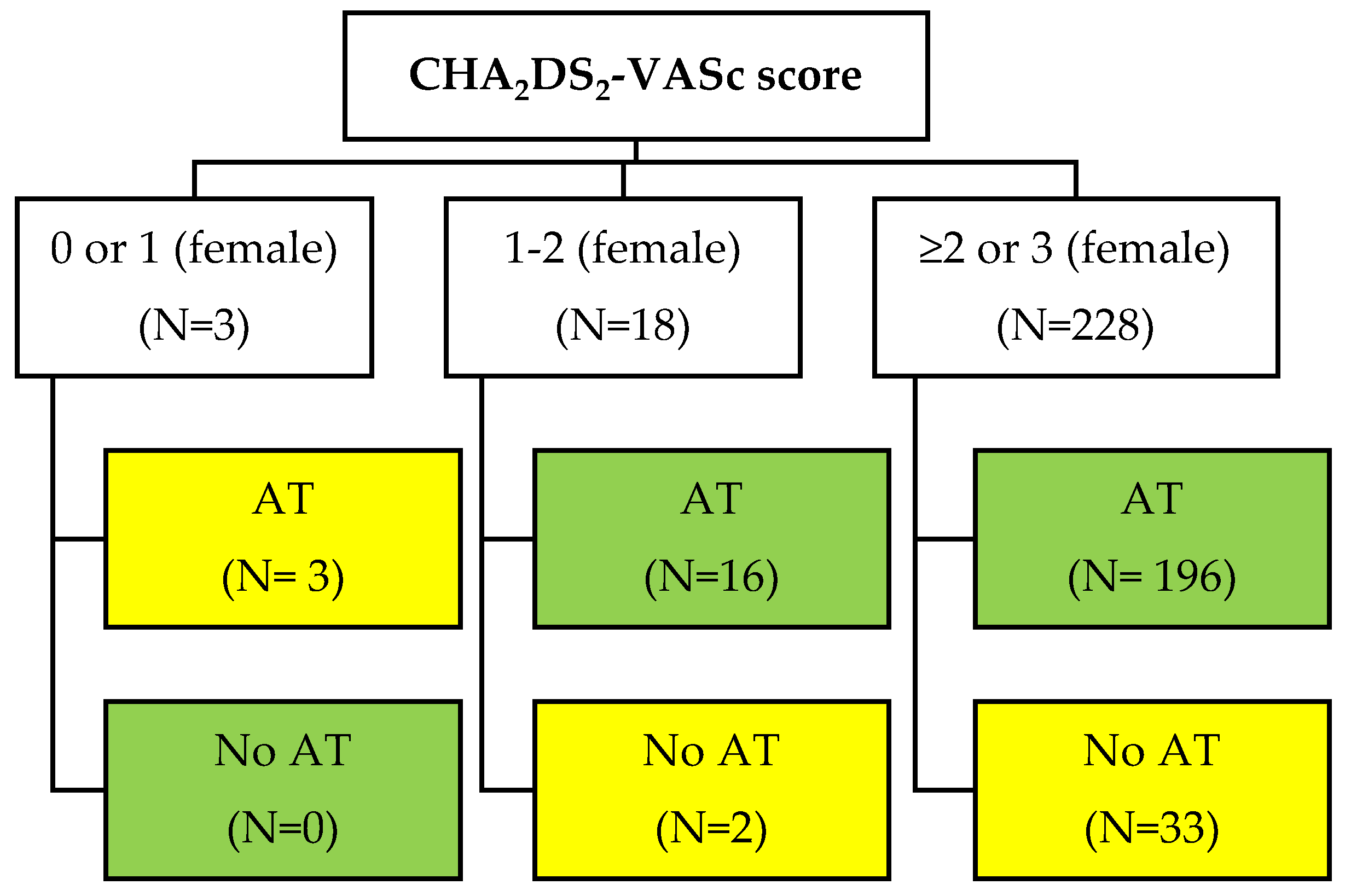
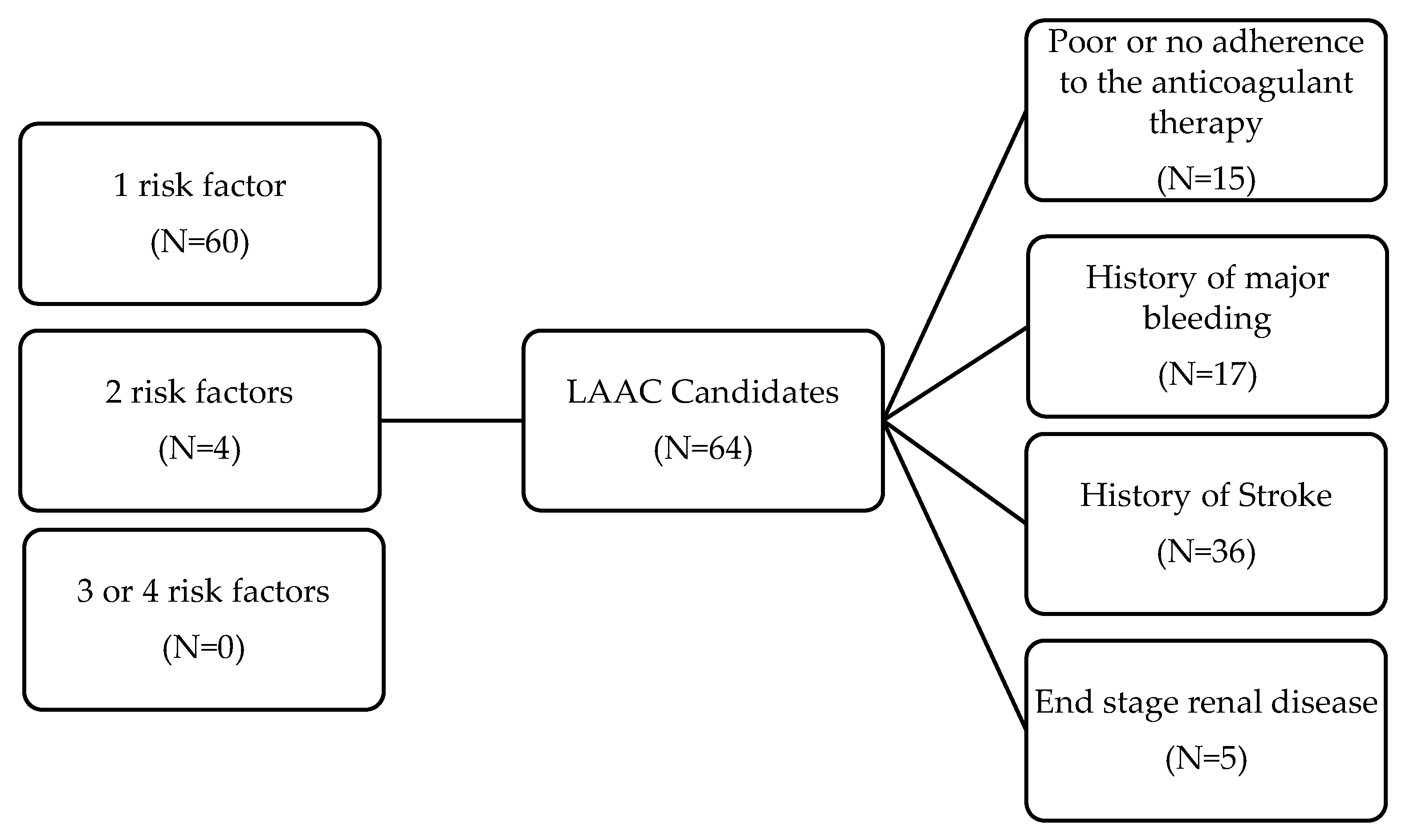
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. European heart journal 2021, 42, 373–498. [Google Scholar] [CrossRef]
- January, C.T.; Wann, L.S.; Calkins, H.; Chen, L.Y.; Cigarroa, J.E.; Cleveland, J.C., Jr.; Ellinor, P.T.; Ezekowitz, M.D.; Field, M.E.; Furie, K.L.; et al. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Journal of the American College of Cardiology 2019, 74, 104–132. [Google Scholar] [CrossRef]
- Kartas, A.; Samaras, A.; Vasdeki, D.; Dividis, G.; Fotos, G.; Paschou, E.; Forozidou, E.; Tsoukra, P.; Kotsi, E.; Goulas, I.; et al. Flaws in Anticoagulation Strategies in Patients With Atrial Fibrillation at Hospital Discharge. Journal of cardiovascular pharmacology and therapeutics 2019, 24, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Papakonstantinou, P.E.; Tsioufis, C.; Konstantinidis, D.; Iliakis, P.; Leontsinis, I.; Tousoulis, D. Anticoagulation in Deep Venous Thrombosis: Current Trends in the Era of Non- Vitamin K Antagonists Oral Anticoagulants. Current pharmaceutical design 2020, 26, 2692–2702. [Google Scholar] [CrossRef] [PubMed]
- Holmes, D.R., Jr.; Alkhouli, M.; Reddy, V. Left Atrial Appendage Occlusion for The Unmet Clinical Needs of Stroke Prevention in Nonvalvular Atrial Fibrillation. Mayo Clinic proceedings 2019, 94, 864–874. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, M.; Vidal, X.; Ballarin, E.; Rottenkolber, M.; Schmiedl, S.; Grave, B.; Huerta, C.; Martin-Merino, E.; Montero, D.; Leon-Muñoz, L.M.; et al. Adherence to Direct Oral Anticoagulants in Patients With Non-Valvular Atrial Fibrillation: A Cross-National Comparison in Six European Countries (2008-2015). Frontiers in pharmacology 2021, 12, 682890. [Google Scholar] [CrossRef]
- Papakonstantinou, P.E.; Asimakopoulou, N.I.; Papadakis, J.A.; Leventis, D.; Panousieris, M.; Mentzantonakis, G.; Hoda, E.; Panagiotakis, S.; Gikas, A. Frailty Status Affects the Decision for Long-Term Anticoagulation Therapy in Elderly Patients with Atrial Fibrillation. Drugs & aging 2018, 35, 897–905. [Google Scholar] [CrossRef]
- Alkhouli, M.; Ellis, C.R.; Daniels, M.; Coylewright, M.; Nielsen-Kudsk, J.E.; Holmes, D.R. Left Atrial Appendage Occlusion. JACC: Advances 2022, 1, 100136. [Google Scholar] [CrossRef]
- Holmes, D.R., Jr.; Alkhouli, M. The History of the Left Atrial Appendage Occlusion. Cardiac electrophysiology clinics 2020, 12, 1–11. [Google Scholar] [CrossRef]
- Vrana, E.; Kartas, A.; Samaras, A.; Vasdeki, D.; Forozidou, E.; Liampas, E.; Karvounis, H.; Giannakoulas, G.; Tzikas, A. Indications for percutaneous left atrial appendage occlusion in hospitalized patients with atrial fibrillation. Journal of cardiovascular medicine (Hagerstown, Md.) 2022, 23, 176–182. [Google Scholar] [CrossRef]
- Leontsinis, I.; Farmakis, D.; Avramidis, D.; Andrikou, E.; Valatsou, A.; Gartzonikas, E.; Doundoulakis, I.; Zarifis, I.; Karpouzis, I.; Kafkala, K.; et al. Cardiorenal multimorbidity in hospitalized cardiology patients: The Hellenic Cardiorenal Morbidity Snapshot (HECMOS) study. Hellenic journal of cardiology : HJC = Hellenike kardiologike epitheorese 2023. [Google Scholar] [CrossRef]
- Hsu, J.C.; Maddox, T.M.; Kennedy, K.F.; Katz, D.F.; Marzec, L.N.; Lubitz, S.A.; Gehi, A.K.; Turakhia, M.P.; Marcus, G.M. Oral Anticoagulant Therapy Prescription in Patients With Atrial Fibrillation Across the Spectrum of Stroke Risk: Insights From the NCDR PINNACLE Registry. JAMA cardiology 2016, 1, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.Y.X.; Malo, S.; Svenson, L.W.; Wilton, S.B.; Hill, M.D. Temporal Trends in the Use and Comparative Effectiveness of Direct Oral Anticoagulant Agents Versus Warfarin for Nonvalvular Atrial Fibrillation: A Canadian Population-Based Study. Journal of the American Heart Association 2017, 6. [Google Scholar] [CrossRef]
- Marzec, L.N.; Wang, J.; Shah, N.D.; Chan, P.S.; Ting, H.H.; Gosch, K.L.; Hsu, J.C.; Maddox, T.M. Influence of Direct Oral Anticoagulants on Rates of Oral Anticoagulation for Atrial Fibrillation. Journal of the American College of Cardiology 2017, 69, 2475–2484. [Google Scholar] [CrossRef]
- Admassie, E.; Chalmers, L.; Bereznicki, L.R. Changes in Oral Anticoagulant Prescribing for Stroke Prevention in Patients With Atrial Fibrillation. The American journal of cardiology 2017, 120, 1133–1138. [Google Scholar] [CrossRef]
- Proietti, M.; Laroche, C.; Opolski, G.; Maggioni, A.P.; Boriani, G.; Lip, G.Y.H. 'Real-world' atrial fibrillation management in Europe: Observations from the 2-year follow-up of the EURObservational Research Programme-Atrial Fibrillation General Registry Pilot Phase. Europace : European pacing, arrhythmias, and cardiac electrophysiology : Journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology 2017, 19, 722–733. [Google Scholar] [CrossRef] [PubMed]
- Cullen, M.W.; Kim, S.; Piccini, J.P., Sr.; Ansell, J.E.; Fonarow, G.C.; Hylek, E.M.; Singer, D.E.; Mahaffey, K.W.; Kowey, P.R.; Thomas, L.; et al. Risks and benefits of anticoagulation in atrial fibrillation: Insights from the Outcomes Registry for Better Informed Treatment of Atrial Fibrillation (ORBIT-AF) registry. Circulation. Cardiovascular quality and outcomes 2013, 6, 461–469. [Google Scholar] [CrossRef]
- Kourlaba, G.; Stefanou, G.; Tsalamandris, S.; Oikonomou, E.; Papageorgiou, G.; Nikas, N.; Tousoulis, D.; Maniadakis, N. Incidence and cost of bleeding events requiring hospitalization in patients with atrial fibrillation treated with acenocoumarol in Greece. Hellenic journal of cardiology : HJC = Hellenike kardiologike epitheorese 2021, 62, 234–240. [Google Scholar] [CrossRef]
- Yang, E. A clinician's perspective: Novel oral anticoagulants to reduce the risk of stroke in nonvalvular atrial fibrillation--full speed ahead or proceed with caution? Vascular health and risk management 2014, 10, 507–522. [Google Scholar] [CrossRef] [PubMed]
- Labori, F.; Persson, J.; Bonander, C.; Jood, K.; Svensson, M. Cost-effectiveness analysis of left atrial appendage occlusion in patients with atrial fibrillation and contraindication to oral anticoagulation. European heart journal 2022, 43, 1348–1356. [Google Scholar] [CrossRef] [PubMed]
- Glikson, M.; Wolff, R.; Hindricks, G.; Mandrola, J.; Camm, A.J.; Lip, G.Y.H.; Fauchier, L.; Betts, T.R.; Lewalter, T.; Saw, J.; et al. EHRA/EAPCI expert consensus statement on catheter-based left atrial appendage occlusion - an update. Europace : European pacing, arrhythmias, and cardiac electrophysiology : Journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology 2020, 22, 184. [Google Scholar] [CrossRef] [PubMed]
- Tzikas, A.; Freixa, X.; Llull, L.; Gafoor, S.; Shakir, S.; Omran, H.; Giannakoulas, G.; Berti, S.; Santoro, G.; Kefer, J.; et al. Patients with intracranial bleeding and atrial fibrillation treated with left atrial appendage occlusion: Results from the Amplatzer Cardiac Plug registry. International journal of cardiology 2017, 236, 232–236. [Google Scholar] [CrossRef]
- Cruz-González, I.; González-Ferreiro, R.; Freixa, X.; Gafoor, S.; Shakir, S.; Omran, H.; Berti, S.; Santoro, G.; Kefer, J.; Landmesser, U.; et al. Left atrial appendage occlusion for stroke despite oral anticoagulation (resistant stroke). Results from the Amplatzer Cardiac Plug registry. Revista espanola de cardiologia (English ed.) 2020, 73, 28–34. [Google Scholar] [CrossRef]
- Potpara, T.S.; Ferro, C.J.; Lip, G.Y.H. Use of oral anticoagulants in patients with atrial fibrillation and renal dysfunction. Nature reviews. Nephrology 2018, 14, 337–351. [Google Scholar] [CrossRef]
- Kefer, J.; Tzikas, A.; Freixa, X.; Shakir, S.; Gafoor, S.; Nielsen-Kudsk, J.E.; Berti, S.; Santoro, G.; Aminian, A.; Landmesser, U.; et al. Impact of chronic kidney disease on left atrial appendage occlusion for stroke prevention in patients with atrial fibrillation. International journal of cardiology 2016, 207, 335–340. [Google Scholar] [CrossRef]
- Banerjee, A.; Benedetto, V.; Gichuru, P.; Burnell, J.; Antoniou, S.; Schilling, R.J.; Strain, W.D.; Ryan, R.; Watkins, C.; Marshall, T.; et al. Adherence and persistence to direct oral anticoagulants in atrial fibrillation: A population-based study. Heart (British Cardiac Society) 2020, 106, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, J.; Vasdeki, D.; Tzikas, A.; Meier, B.; Saw, J. Incidence, Prevention, and Management of Periprocedural Complications of Left Atrial Appendage Occlusion. Interventional cardiology clinics 2018, 7, 243–252. [Google Scholar] [CrossRef]
- Badimon, J.J.; Escolar, G.; Zafar, M.U. Factor XI/XIa Inhibition: The Arsenal in Development for a New Therapeutic Target in Cardio- and Cerebrovascular Disease. Journal of cardiovascular development and disease 2022, 9. [Google Scholar] [CrossRef] [PubMed]
| History of Atrial Fibrillation | |
|---|---|
| Classification (N=256) | |
| Paroxysmal | 103 (40.2%) |
| Permanent | 153 (59.8%) |
| Patients’ needs (N=256) | |
| Anticoagulation treatment and takes it consistently | 223 (89.2%) |
| Anticoagulation treatment, but they receive it with poor compliance | 11 (4.4%) |
| Anticoagulation treatment and do not take it because do not want it | 4 (1.6%) |
| Anticoagulation treatment and do not take it because no one recommended it | 12 (4.8%) |
| Anticoagulation treatment (N=253) | |
| VKA- good control of INR | 25 (11.2%) |
| VKA – poor control of INR | 7 (3.1%) |
| Dabigatran | 27 (12.1%) |
| Rivaroxaban | 53 (23.8%) |
| Apixaban | 111 (49.8%) |
| Beyond anticoagulants, are on any other medical treatment which increases the bleeding risk? (Yes) (N=276) | 43 (16.8%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




