Submitted:
22 August 2023
Posted:
22 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
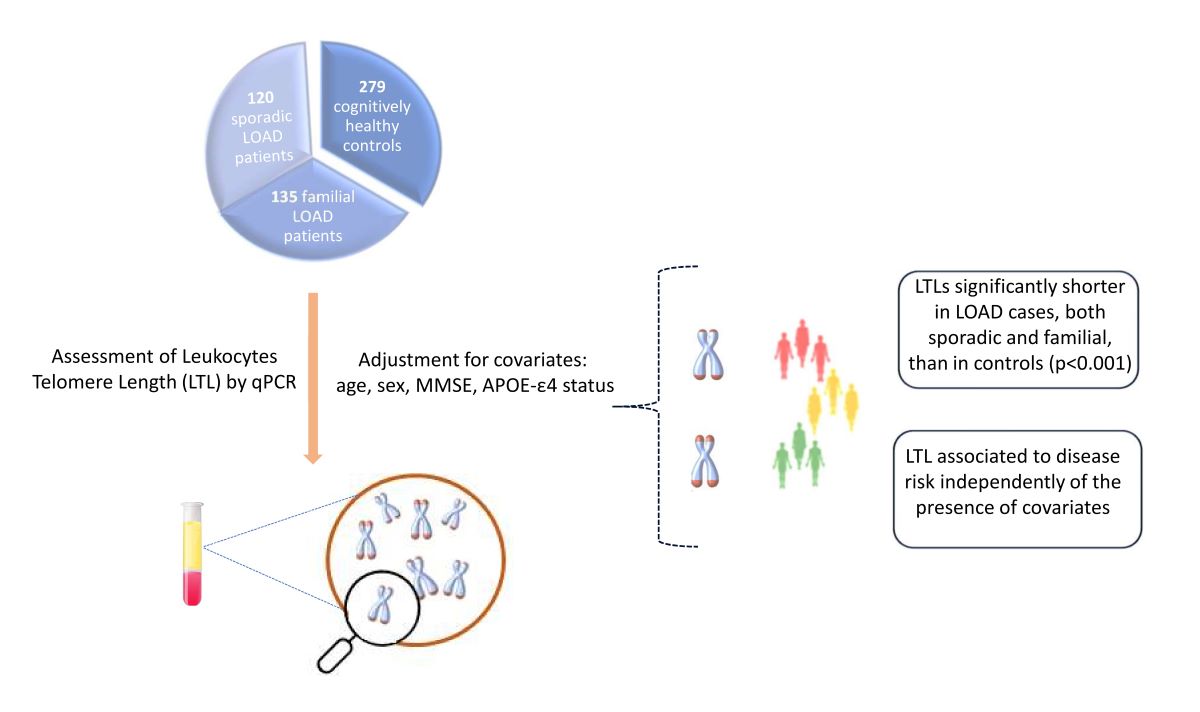
2.1. Subjects
2.2. Ethics statement.
2.3. Leukocyte telomere length (LTL)
2.4. APOE genotyping
2.5. Statistical analyses
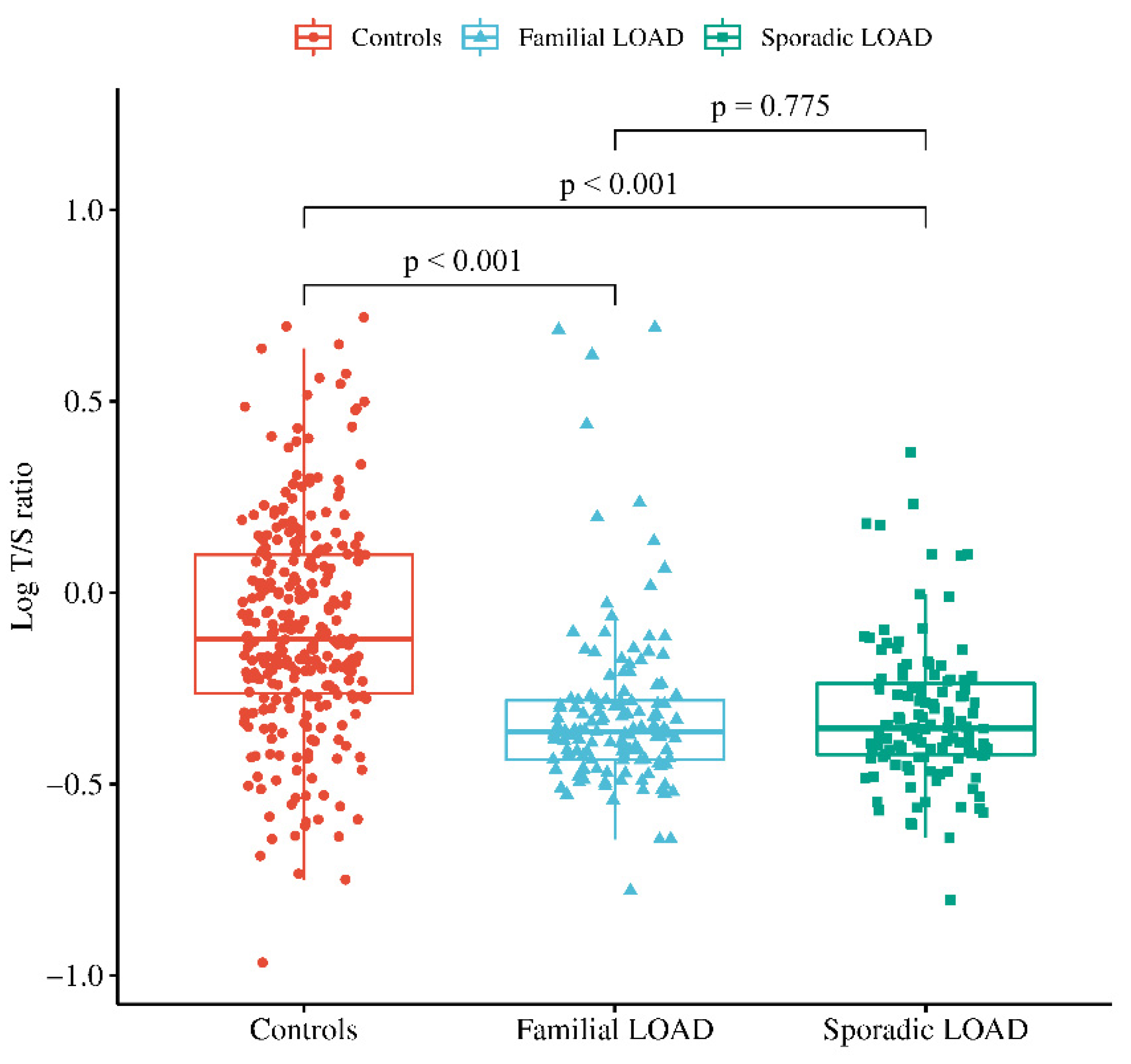
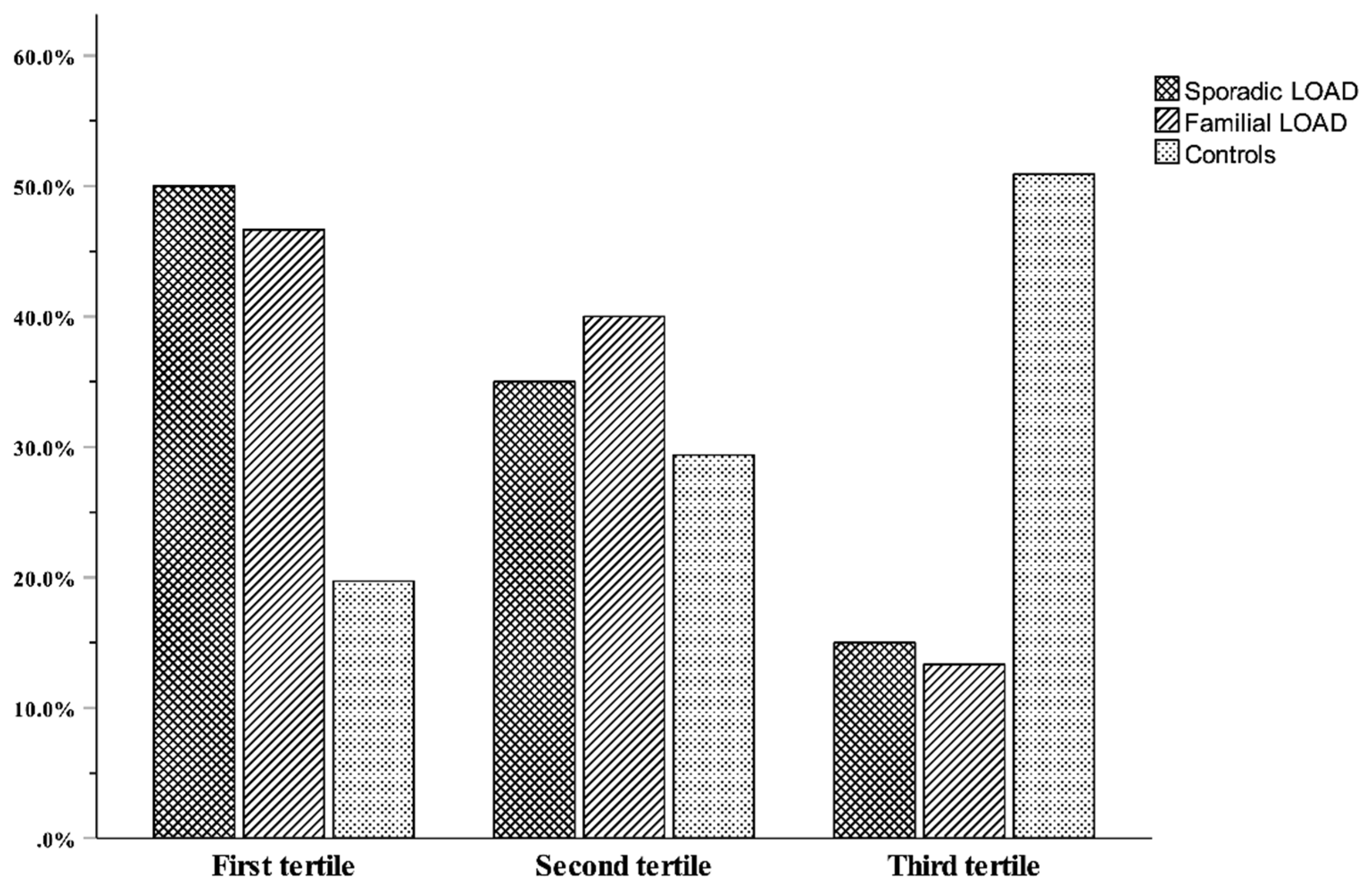
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, X.X.; Tian, Y.; Wang, Z.T.; Ma, Y.H.; Tan, L.; Yu, J.T. The Epidemiology of Alzheimer’s Disease Modifiable Risk Factors and Prevention. J Prev Alzheimers Dis 2021, 8, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Rossiello, F.; Jurk, D.; Passos, J.F.; d’Adda di Fagagna, F. Telomere dysfunction in ageing and age-related diseases. Na Cell Biol 2022, 24, 135–147. [Google Scholar] [CrossRef]
- Cawthon, R.M.; Smith, K.R.; O’Brien, E.; Sivatchenko, A.; Kerber, R.A. Association between telomere length in blood and mortality in people aged 60 years or older. Lancet 2003, 361, 393–395. [Google Scholar] [CrossRef] [PubMed]
- Aviv, A.; Shay, J.W. Reflections on telomere dynamics and ageing-related diseases in humans. Philos Trans R Soc Lond B Biol Sci 2018, 373, 20160436. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Ezquerro, J.D.; Rodríguez-Castañeda, A.; Ortiz-Ramírez, M.; Sánchez-García, S.; Rosas-Vargas, H.; Sánchez-Arenas, R.; García-De la Torre, P. Oxidative Stress, Telomere Length, and Frailty in an Old Age Population. Rev Invest Clin 2020, 71, 393–401. [Google Scholar] [CrossRef]
- Tian, Y.; Wang, S.; Jiao, F.; Kong, Q.; Liu, C.; Wu, Y. Telomere Length: A Potential Biomarker for the Risk and Prognosis of Stroke. Front Neurol 2019, 10. [Google Scholar] [CrossRef]
- Fragkiadaki, P.; Nikitovic, D.; Kalliantasi, K.; Sarandi, E.; Thanasoula, M.; Stivaktakis, P.; Nepka, C.; Spandidos, D.; Theodoros, T.; Tsatsakis, A. Telomere length and telomerase activity in osteoporosis and osteoarthritis (Review). Exp Ther Med 2019, 19, 1626–1632. [Google Scholar] [CrossRef]
- Okamoto, K.; Seimiya, H. Revisiting Telomere Shortening in Cancer. Cells 2019, 8, 107. [Google Scholar] [CrossRef]
- Rodríguez-Fernández, B.; Gispert, J.D.; Guigo, R.; Navarro, A.; Vilor-Tejedor, N.; Crous-Bou, M. Genetically predicted telomere length and its relationship with neurodegenerative diseases and life expectancy. Comput Struct Biotechnol J 2022, 20, 4251–4256. [Google Scholar] [CrossRef]
- Chen, X.; Guo, C.; Kong, J. Oxidative stress in neurodegenerative diseases. Neural Regen Res 2012, 7, 376–385. [Google Scholar]
- Boccardi, V.; Arosio, B.; Cari, L.; Bastiani, P.; Scamosci, M.; Casati, M.; Ferri, E.; Bertagnoli, L.; Ciccone, S.; Rossi, P.D.; Nocentini, G.; Mecocci, P. Beta-carotene, telomerase activity and Alzheimer’s disease in old age subjects. Eur J Nutr 2020, 59, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Forero, D.A.; González-Giraldo, Y.; López-Quintero, C.; Castro-Vega, L.J.; Barreto, G.E.; Perry, G. Meta-analysis of Telomere Length in Alzheimer’s Disease. J Gerontol A Biol Sci Med Sci 2016, 71, 1069–1073. [Google Scholar] [CrossRef] [PubMed]
- Honig, L.S.; Schupf, N.; Lee, J.H.; Tang, M.X.; Mayeux, R. Shorter telomeres are associated with mortality in those with APOE ϵ4 and dementia. Ann Neurol 2006, 60, 181–187. [Google Scholar] [CrossRef]
- Panossian, L.; Porter, V.R.; Valenzuela, H.F.; Zhu, X.; Reback, E.; Masterman, D.; Cummings, J.L.; Effros, R.B. Telomere shortening in T cells correlates with Alzheimer’s disease status. Neurobiol Aging 2003, 24, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Scarabino, D.; Broggio, E.; Gambina, G.; Corbo, R.M. Leukocyte telomere length in mild cognitive impairment and Alzheimer’s disease patients. Exp Gerontol 2017, 98, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Wikgren, M.; Karlsson, T.; Nilbrink, T.; Nordfjall, K.; Hultdin, J.; Sleegers, K.; Van Broeckhoven, C.; Nyberg, L. , Roos, G.; Nilsson, L.G.; Adolfsson, R.; Norrback, K.F. APOE epsilon4 is associated with longer telomeres, and longer telomeres among epsilon4 carriers predicts worse episodic memory. Neurobiol Aging 2012, 33, 335–344. [Google Scholar] [CrossRef]
- Franco, S.; Blasco, M.A.; Siedlak, S.L.; Harris, P.L. R.; Moreira, P.I.; Perry, G.; Smith, M.A. Telomeres and telomerase in Alzheimer’s disease: Epiphenomena or a new focus for therapeutic strategy? Alzheimers Dement 2006, 2, 164–168. [Google Scholar] [CrossRef]
- Zekry, D.; Herrmann, F.R.; Irminger-Finger, I.; Ortolan, L.; Genet, C.; Vitale, A.M.; Michel, J.P.; Gold, G.; Krause, K.H. Telomere length is not predictive of dementia or MCI conversion in the oldest old. Neurobiol Aging 2010, 31, 719–720. [Google Scholar] [CrossRef]
- Zekry, D.; Herrmann, F.R.; Irminger-Finger, I.; Graf, C.; Genet, C.; Vitale, A.M.; Michel, J.P.; Gold, G.; Krause, K.H. Telomere length and ApoE polymorphism in mild cognitive impairment, degenerative and vascular dementia. J Neurol Sci 2010, 299, 108–111. [Google Scholar] [CrossRef]
- Takata, Y.; Kikukawa, M.; Hanyu, H.; Koyama, S.; Shimizu, S.; Umahara, T.; Sakurai, H.; Iwamoto, T.; Ohyashiki, K.; Ohyashiki, J.H. Association Between ApoE Phenotypes and Telomere Erosion in Alzheimer’s Disease. J Gerontol A Biol Sci Med Sci 2012, 330–335. [Google Scholar] [CrossRef]
- Movérare-Skrtic, S.; Johansson, P.; Mattsson, N.; Hansson, O.; Wallin, A.; Johansson, J.O.; Zetterberg, H.; Blennow, K.; Svensson, J. Leukocyte Telomere Length (LTL) is reduced in stable mild cognitive impairment but low LTL is not associated with conversion to Alzheimer’s Disease: A pilot study. Exp Gerontol 2012, 47, 179–182. [Google Scholar] [CrossRef] [PubMed]
- Fani, L.; Hilal, S.; Sedaghat, S.; Broer, L.; Licher, S.; Arp, P.P.; van Meurs, J.B. J.; Ikram, M.K.; Ikram, M.A. Telomere Length and the Risk of Alzheimer’s Disease: The Rotterdam Study. J Alzheimers Dis 2020, 73, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.O.; Boardman, L.A.; Cha, R.H.; Pankratz, V.S.; Johnson, R.A.; Druliner, B.R.; Christianson, T.J. H.; Roberts, L.R.; Petersen, R.C. Short and long telomeres increase risk of amnestic mild cognitive impairment. Mech Ageing Dev 2014, 141–142, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Crocco, P.; Barale, R.; Rose, G.; Rizzato, C.; Santoro, A.; de Rango, F.; Carrai, M.; Fogar, P.; Monti, D.; Biondi, F.; Bucci, L.; Ostan, R.; Tallaro, F.; Montesanto, A.; Zambon, C.-F.; Franceschi, C.; Canzian, F.; Passarino, G.; Campa, D. Population-specific association of genes for telomere-.
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. Mini-mental state. J Psychiatric Res 1975, 12, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Magni, E.; Binetti, G.; Bianchetti, A.; Rozzini, R.; Trabucchi, M. Mini-Mental State Examination: a normative study in Italian elderly population. Eur J Neurol 1996, 3, 198–202. [Google Scholar] [CrossRef] [PubMed]
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R.; Mohs, R.C.; Morris, J.C.; Rossor, M.N.; Scheltens, P.; Carrillo, M.C.; Thies, B.; Weintraub, S.; Phelps, C.H. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011, 7, 263–269. [Google Scholar] [CrossRef]
- McKeith, I.G.; Galasko, D.; Kosaka, K.; Perry, E.K.; Dickson, D.W.; Hansen, L.A.; Salmon, D.P.; Lowe, J.; Mirra, S.S.; Byrne, E.J.; Lennox, G.; Quinn, N.P.; Edwardson, J.A.; Ince, P.G.; Bergeron, C.; Burns, A.; Miller, B.L.; Lovestone, S.; Collerton, D.; Jansen, E.N.; Ballard, C.; de Vos, R.A.; Wilcock, G.K.; Jellinger, K.A.; Perry, R.H. Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the consortium on DLB international workshop. Neurology 1996, 47, 1113–1124. [Google Scholar] [CrossRef]
- Englund, B.; Brun, A.; Gustafson, L.; Passant, U.; Mann, D.; Neary, D.; Snowden, J.S. Clinical and neuropathological criteria for frontotemporal dementia. J Neurol Neurosurg Psychiatry 1994, 57, 416–418. [Google Scholar]
- Roman, G.C.; Tatemichi, T.K.; Erkinjuntti, T.; Cummings, J.L.; Masdeu, J.C.; Garcia, J.H.; Amaducci, L.; Orgogozo, J.M.; Brun, A.; Hofman, A.; Moody, D.M.; O’Brien, M.D.; Yamaguchi, T.; Grafman, J.; Drayer, B.P.; Bennett, D.A.; Fisher, M.; Ogata, J.; Kokmen, E.; Bermejo, F.; Wolf, P.A.; Gorelick, P.B.; Bick, K.L.; Pajeau, A.K.; Bell, M.A.; DeCarli, C.; Culebras, A.; Korczyn, A.D.; Bogousslavsky, J.; Hartman, A.; Scheinberg, P. Vascular dementia: Diagnostic criteria for research studies. Report of the NINDS-AIREN InternationalWorkshop. Neurology 1993, 43, 250–260. [Google Scholar]
- Cawthon, R.M. Telomere measurement by quantitative PCR. Nucleic Acids Res 2002, 30, 47e–447. [Google Scholar] [CrossRef]
- Testa, R.; Olivieri, F.; Sirolla, C.; Spazzafumo, L.; Rippo, M.R.; Marra, M.; Bonfigli, A.R.; Ceriello, A.; Antonicelli, R.; Franceschi, C.; Castellucci, C.; Testa, I.; Procopio, A.D. Leukocyte telomere length is associated with complications of Type 2 diabetes mellitus. Diabet Med 2011, 28, 1388–1394. [Google Scholar] [CrossRef] [PubMed]
- Carrieri, G.; Bonafè, M.; de Luca, M.; Rose, G.; Varcasia, O.; Bruni, A.; Maletta, R.; Nacmias, B.; Sorbi, S.; Corsonello, F.; Feraco, E.; Andreev, K.F.; Yashin, A.I.; Franceschi, C.; de Benedictis, G. Mitochondrial DNA haplogroups and APOE4 allele are non-independent variables in sporadic Alzheimer’s disease. Hum Genet 2001, 108, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, C.; Zhao, A.; Li, M.; Ren, J.; Qu, X. New Insights in Amyloid Beta Interactions with Human Telomerase. J Am Chem Soc 2015, 137, 1213–1219. [Google Scholar] [CrossRef]
- Spilsbury, A.; Miwa, S.; Attems, J.; Saretzki, G. The Role of Telomerase Protein TERT in Alzheimer’s Disease and in Tau-Related Pathology In Vitro. J Neurosci 2015, 35, 1659–1674. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, V.S.; Deo, P.; Chua, A. , Thomas, P.; Fenech, M. Shorter Telomere Length in Carriers of APOE-ε4 and High Plasma Concentration of Glucose, Glyoxal and Other Advanced Glycation End Products (AGEs). J Gerontol A Biol Sci Med Sci 2020, 75, 1894–1898. [Google Scholar] [CrossRef]
- Hackenhaar, F.S.; Josefsson, M.; Adolfsson, A.N.; Landfors, M.; Kauppi, K.; Hultdin, M.; Adolfsson, R.; Degerman, S.; Pudas, S. Short leukocyte telomeres predict 25-year Alzheimer’s disease incidence in non-APOE ε4-carriers. Alzheimers Res Ther 2021, 13, 130. [Google Scholar] [CrossRef]
- Valdes, A.M.; Deary, I.J.; Gardner, J.; Kimura, M.; Lu, X.; Spector, T.D.; Aviv, A.; Cherkas, L.F. Leukocyte telomere length is associated with cognitive performance in healthy women. Neurobiol Aging 2010, 31, 986–992. [Google Scholar] [CrossRef]
- Hochstrasser, T.; Marksteiner, J.; Humpel, C. Telomere length is age-dependent and reduced in monocytes of Alzheimer patients. Exp Gerontol 2012, 47, 160–163. [Google Scholar] [CrossRef]
- Liu, M.; Huo, Y.R.; Wang, J.; Wang, C.; Liu, S.; Liu, S.; Wang, J.; Ji, Y. Telomere Shortening in Alzheimer’s Disease Patients. Ann Clin Lab Sci 2016, 46, 260–265. [Google Scholar]
- Mahoney, E.R.; Dumitrescu, L.; Seto, M.; Nudelman, K.N. H.; Buckley, R.F.; Gifford, K.A.; Saykin, A.J.; Jefferson, A.J.; Hohman, T.J. Telomere length associations with cognition depend on Alzheimer’s disease biomarkers. Alzheimers Dement 2019, 5, 883–890. [Google Scholar] [CrossRef]
- Koh, S.-H.; Choi, S.H.; Jeong, J.H.; Jang, J.W.; Park, K.W.; Kim, E.J.; Kim, H.J.; Hong, J.Y.; Yoon, S.J.; Yoon, B.; Kang, J.H.; Lee, J.M.; Park, H.H.; Ha, J.; Suh, Y.J.; Kang, S. Telomere shortening reflecting physical aging is associated with cognitive decline and dementia conversion in mild cognitive impairment due to Alzheimer’s disease. Aging 2020, 12, 4407–4423. [Google Scholar] [CrossRef] [PubMed]
- Lukens, J.N.; van Deerlin, V.; Clark, C.M.; Xie, S.X.; Johnson, F.B. Comparisons of telomere lengths in peripheral blood and cerebellum in Alzheimer’s disease. Alzheimes Dement 2009, 5, 463–469. [Google Scholar] [CrossRef] [PubMed]
| LOAD | Controls (N=279) | |||
|---|---|---|---|---|
| Sporadic (N=120) | Familial (N=135) | P-value* | ||
| Age (mean ± SD) | 76.9± 2.75 | 77.86 ± 2.77 | 73.67 ± 5.49 | <0.001 |
| Males (%) | 37.5 | 37.0 | 52.7 | <0.001 |
| Age onset (mean ± SD) | 73.9 ± 5.40 | 73.72 ± 4.44 | - | |
| MMSE** (mean ± SD) | 14.34 ± 5.72 | 14.48 ± 5.01 | 24.75 ± 3.73 | <0.001 |
| APOE-ε4 carriers (%) | 38.3 | 43.7 | 8.6 | <0.001 |
| Variables | OR (95% CI) | P value | Nagelkerke R2 |
|---|---|---|---|
| Sporadic LOAD | |||
| MMSE scores | 0.70 (0.65-0.76) | <0.001 | 0.712 |
| LogTS ratio | 0.03 (0.006-0.15) | <0.001 | |
| APOE-ε4 status | 5.89 (2.47-13.99) | <0.001 | |
| Age | 1.11 (1.03-1.19) | 0.008 | |
| Familial LOAD | |||
| MMSE scores | 0.69 (0.64-0.75) | <0.001 | 0.734 |
| LogTS ratio | 0.09 (0.02-0.40) | <0.001 | |
| APOE-ε4 status | 4.33 (1.88-9.96) | <0.001 | |
| Age | 1.13 (1.04-1.22) | 0.002 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




