Submitted:
11 August 2023
Posted:
11 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials And Methods
2.1. Plant Materials
2.2. Sample Preparation And Extraction
2.3. Determination Of Protein And Starch
2.4. Estimation Of Mineral Concentration
2.5. Determination Of Phenolic Compounds
2.5.1. Determination of Total Flavonoid Content (TFC)
2.5.2. Determination of Total Tannin Content (TTC)
2.6. Determination of Antioxidant Capacity
2.6.1. DPPH Radical Scavenging Assay
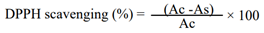
2.6.2. Ferric Reducing Antioxidant Power
2.6.3. Total Antioxidant Capacity With Phosphomolybdenum Assay
2.7. Statistical Data Analysis
3. Results
3.1. Variabilities For Protein And Starch
3.2. Variabilities For Mineral Elements
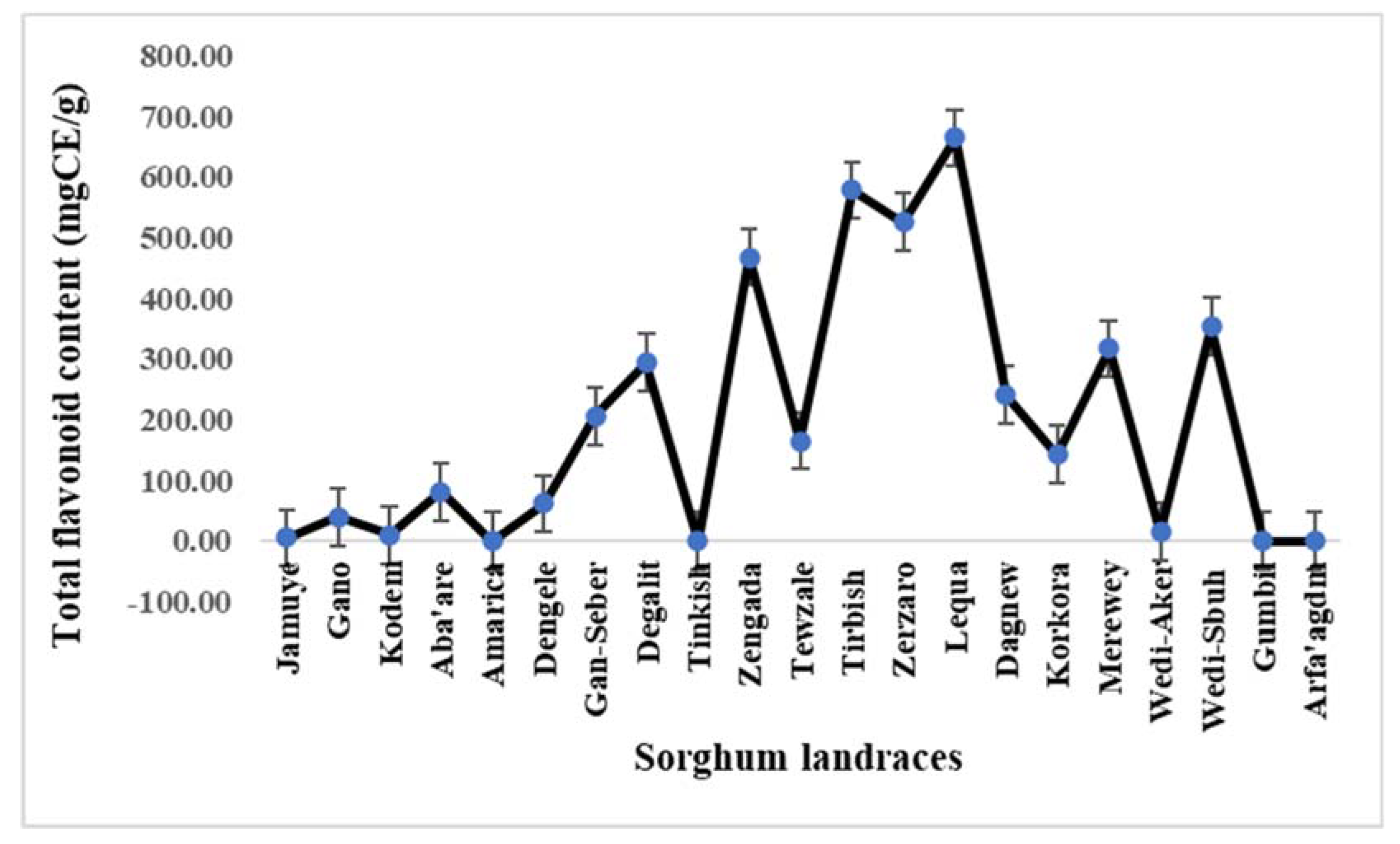
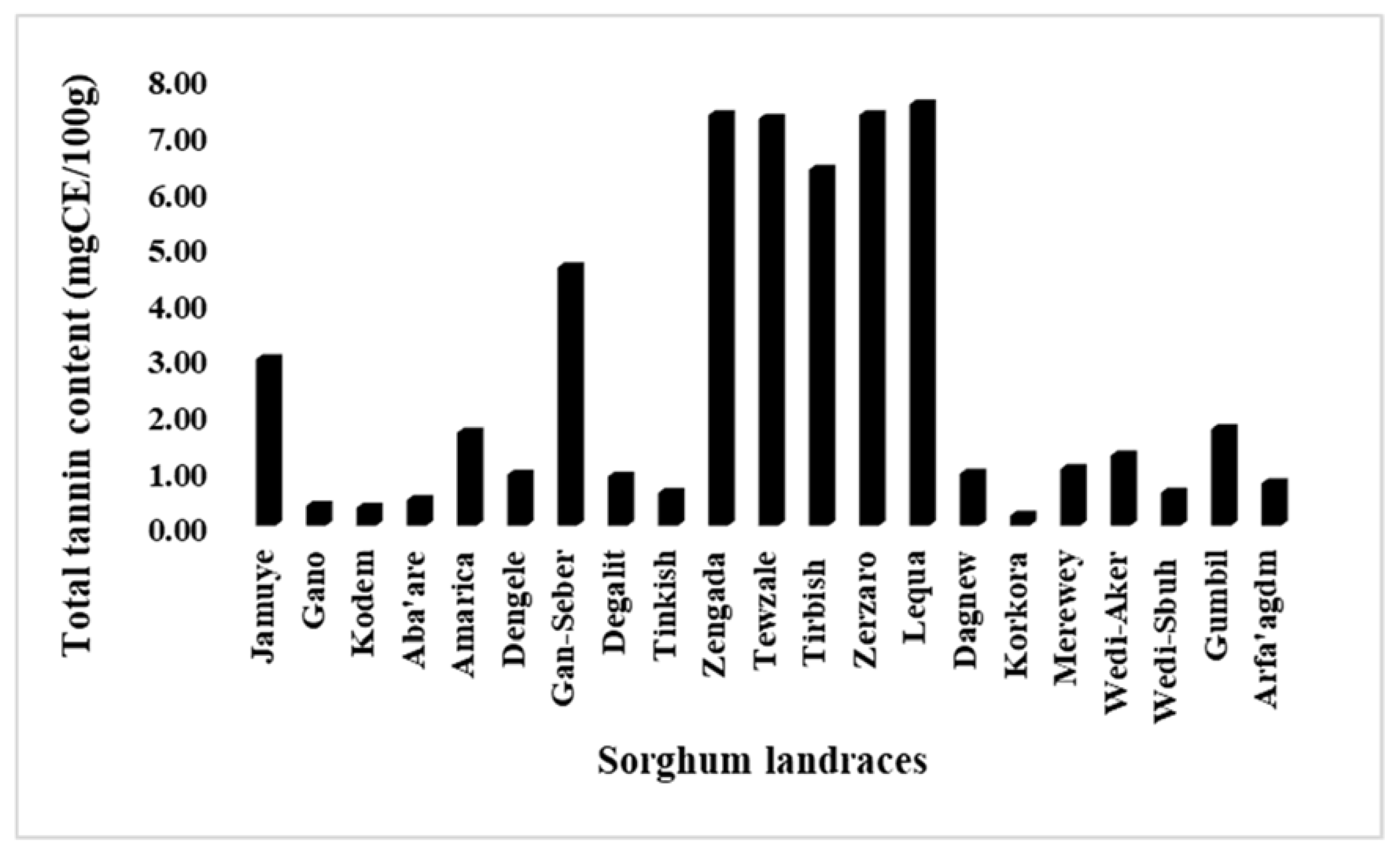
3.3. Variabilities For Total Flavonoid And Tannin Contents
3.4. Variabilities For Antioxidant Activities
3.5. Relationships Of Seed Color With Phenolics And Antioxidant Activities
3.6. Association Between Variables
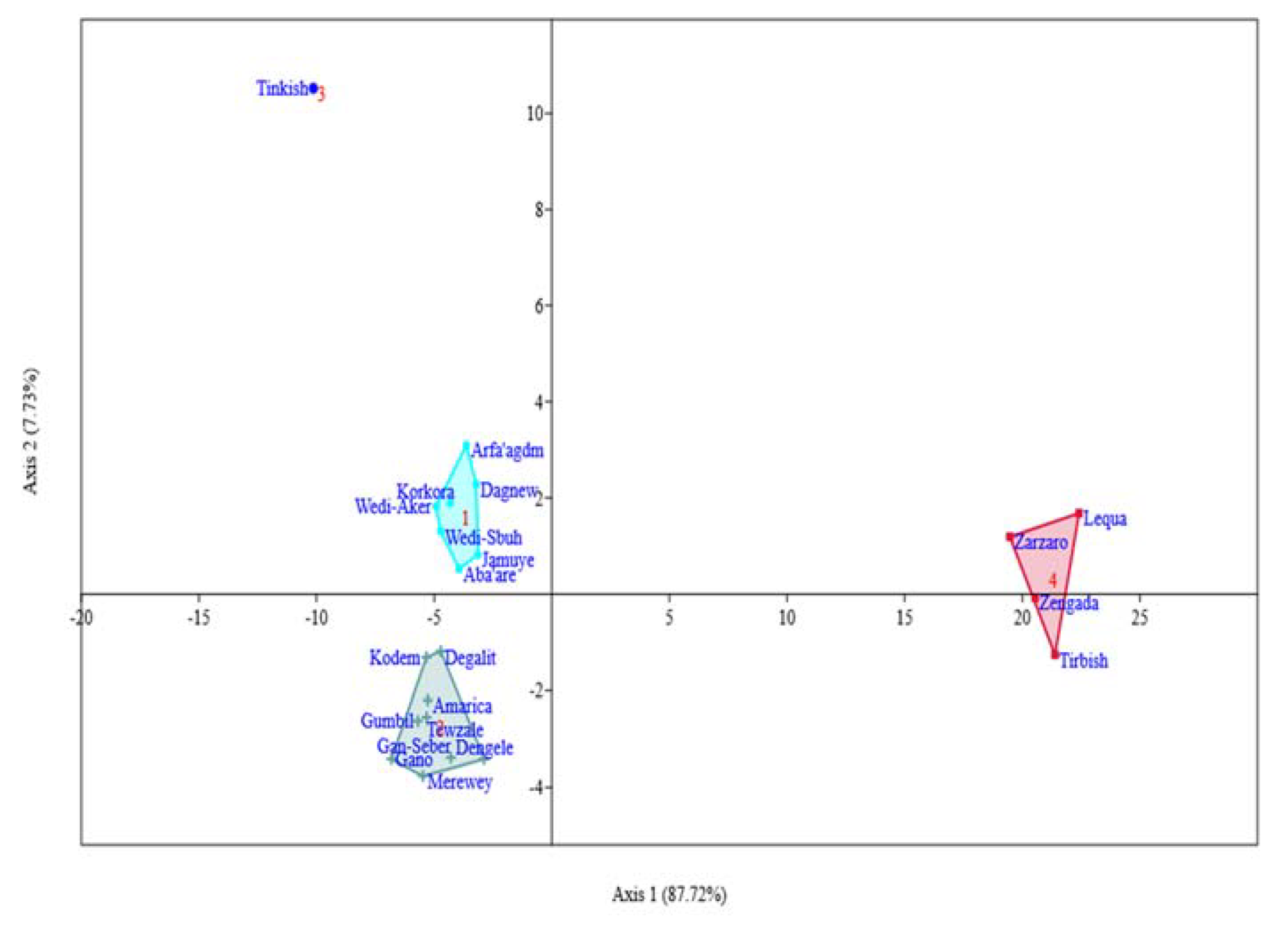
3.7. Grouping And Ordination Of Genotypes
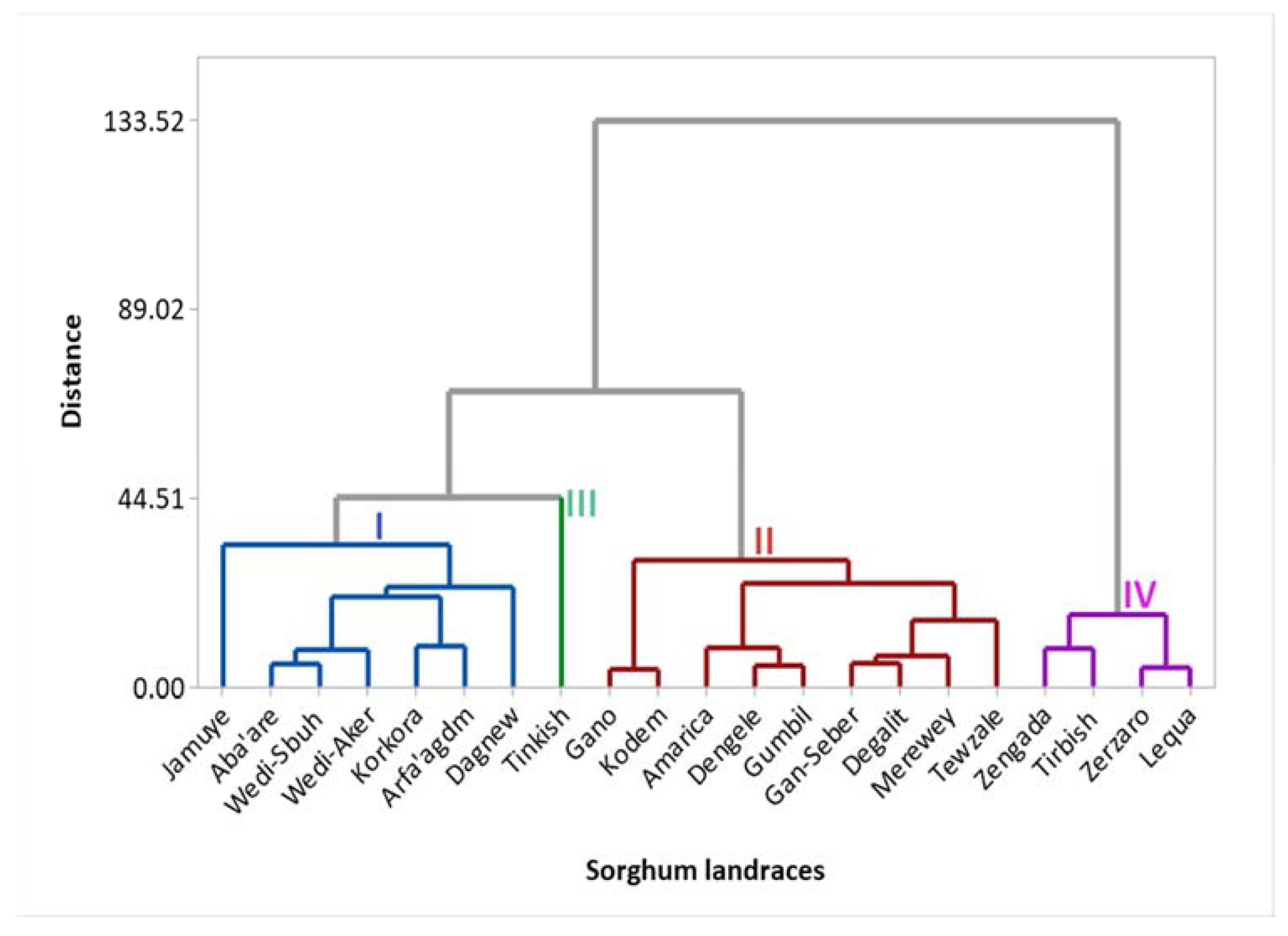
3.7.1. Cluster Analysis
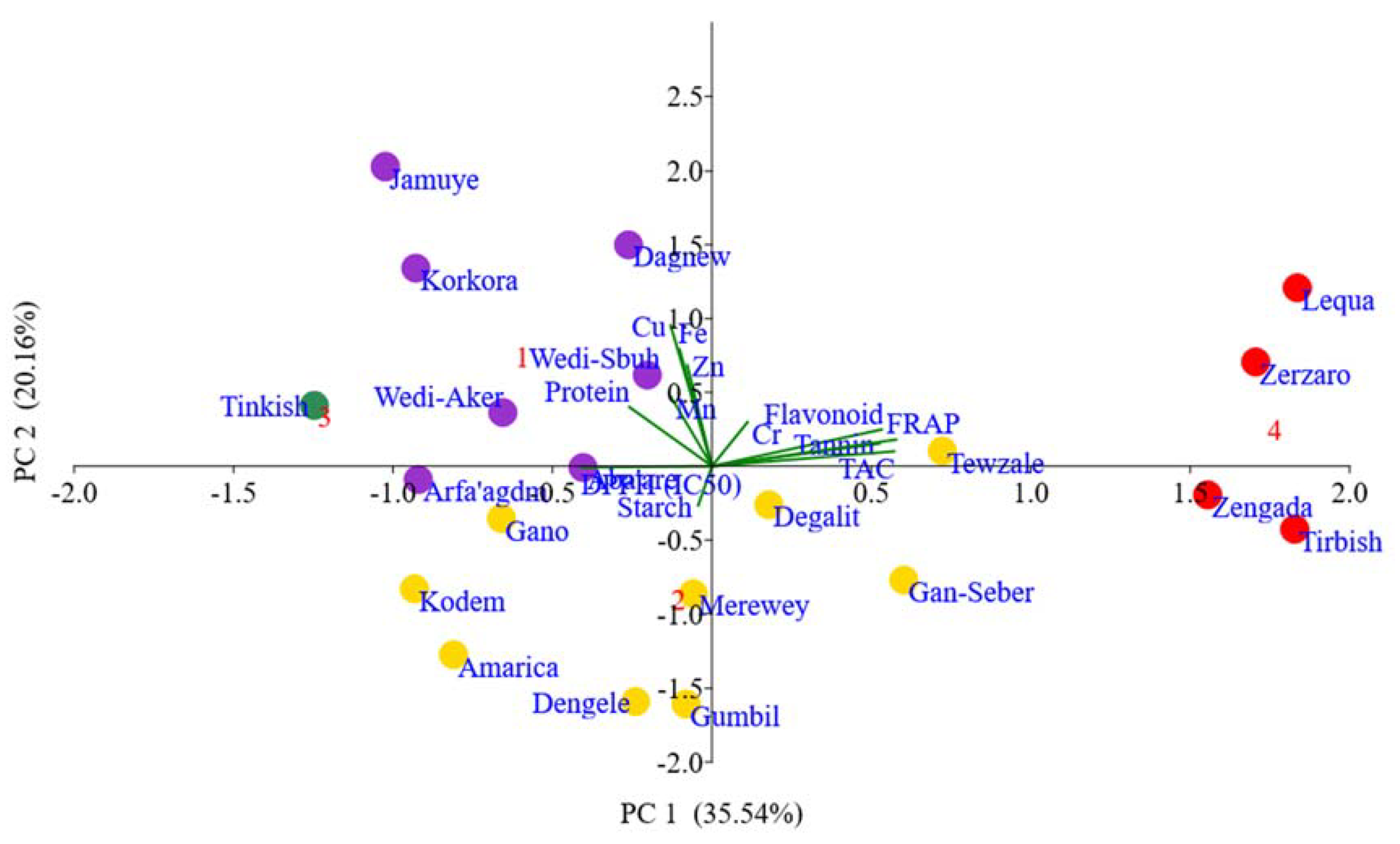
3.7.2. . Principal Component Analysis
4. Discussion
4.1. Proximate Analysis
4.2. Phenolic Compounds And Antioxidant Activities
4.3. Relationship Of Seed Color With Phenolic Compounds And Antioxidant Activities
4.4. Correlation Analysis
4.5. Multivariate Analysis
5. Conclusion
Acknowledgments
References
- Singh, R.; Govindan, V. Zinc-Biofortified Wheat: Harnessing Genetic Diversity for improved Nutritional Quality. Science Brief: Biofortification No. 1. CIMMYT, HarvestPlus, and the Global Crop Trust. Bonn, Germany, 2017.
- Development Initiatives. Global Nutrition Report 2017: Nourishing the SDGs; Development Initiatives: Bristol, UK, 2017. [Google Scholar]
- Maberly, G.F.; Trowbridge, F.L.; Yip, R.; Sullivan, K.M.; E West, C. Programs Against Micronutrient Malnutrition: Ending Hidden Hunger. Annu. Rev. Public Heal. 1994, 15, 277–301. [Google Scholar] [CrossRef] [PubMed]
- Mayer, J.E.; Pfeiffer, W.H.; Beyer, P. Biofortified crops to alleviate micronutrient malnutrition. Curr. Opin. Plant Biol. 2008, 11, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Teshome, A.; Baum, B.R.; Fahrig, L.; Torrance, J.K.; Arnason, T.J.; Lambert, J.D. Sorghum (Sorghum bicolor (L.) Moench) landrace variation and classification in North Shewa and South Welo. Ethiopia. Euphytica 1997, 97, 255–263. [Google Scholar] [CrossRef]
- Villa, T.C.C.; Maxted, N.; Scholten, M.; Ford-Lloyd, B. Defining and Identifying Crop Landraces. Plant Genet. Resour. 2005, 3, 373–384. [Google Scholar] [CrossRef]
- Seboka, B.; Theo van Hintum, T. The dynamics of on-farm management of sorghum in Ethiopia: Implication for the conservation and improvement of plant genetic resources. Genet. Resour. Crop Evol. 2006, 53, 1385–1403. [Google Scholar] [CrossRef]
- Newton, A.C.; Akar, T.; Baresel, J.P.; Bebeli, P.J.; Bettencourt, E.; Bladenopoulos, K.V.; Czembor, J.H.; Fasoula, D.A.; Katsiotis, A.; Koutis, K.; et al. Cereal Landraces for Sustainable Agriculture. 2011, 147–186. [CrossRef]
- Central Statistical Agency (CSA). Agricultural Sample Survey2015/2016 volume 1, a report on area and production of major crops. Statistical Bulletin 2016, 584 Addis Ababa, Ethiopia.
- Teshome, A.; Patterson, D.; Asfew, Z.; Torrance, J.K.; Arnason, J.T. Changes of Sorghum bicolor landrace diversity and farmers’ selection criteria over space and time, Ethiopia. Genet. Resour. Crop. Evol. 2007, 54, 1219–1233. [Google Scholar] [CrossRef]
- Tsehaye, Y.; Abera, Z.; Kebede, A.; Ghebremichael, B. A dynamic sorghum (Sorghum bicolor (L.) Moench) diversity management in situ and livelihood resilience in South and Central Tigray Region, Ethiopia. Momona Ethiopian Journal of Science.
- Duodu, K.; Taylor, J.; Belton, P.; Hamaker, B. Factors affecting sorghum protein digestibility. J. Cereal Sci. 2003, 38, 117–131. [Google Scholar] [CrossRef]
- Saleh, S.M.; Q. Zhang, Q.; Chen, J.; Shen, Q. Millet grains: nutritional quality, processing, and potential health benefits. Compr. Rev. Food Sci. Food Saf. 2013, 12, 281–295. [Google Scholar] [CrossRef]
- Rao, S.; Santhakumar, A.B.; Chinkwo, K.A.; Wu, G.; Johnson, S.K.; Blanchard, C.L. Characterization of phenolic compounds and antioxidant activity in sorghum grains. J. Cereal Sci. 2018, 84, 103–111. [Google Scholar] [CrossRef]
- Shegro, A.; Shargie, N.G.; van Biljon, A.; Labuschagne, M.T. Diversity in starch, protein and mineral composition of sorghum landrace accessions from Ethiopia. J. Crop. Sci. Biotechnol. 2012, 15, 275–280. [Google Scholar] [CrossRef]
- Shegro, A.; Labuschagne, M.T.; Shargie, N.G.; van Biljon, A. Multivariate analysis of nutriotonal diversity in sorghum accesions from wetsern Ethiopia. Journal of Biological Sciences 2013, 13, 67–74. [Google Scholar] [CrossRef]
- Mofokeng, M.A. Diversity analysis of South African sorghum genotypes using agronomic traits, SSR markers and protein content and amino acid composition. PhD Thesis. University of KwaZulu-Natal Pietermaritzburg South Africa, 2015.
- de Morais Cardoso, L.; Pinheiro, S.S.; Martino, H.S.D.; Pinheiro-Sant'Ana, H.M. Sorghum (Sorghum bicolor L.): Nutrients, bioactive compounds, and potential impact on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 372–390. [Google Scholar] [CrossRef] [PubMed]
- Nguni, D.; Geleta, M.; Hofvander, P.; Fatih, M.; Bryngelsson, T. Comparative genetic diversity and nutritional quality variation among some important southern African sorghum accessions [Sorghum bicolor (L.) Moench]. Aust. J. Crop Sci. 2012, 6, 56–64. [Google Scholar]
- Shewayrga, H.; A Sopade, P.; Jordan, D.R.; Godwin, I.D. Characterisation of grain quality in diverse sorghum germplasm using a Rapid Visco-Analyzer and near infrared reflectance spectroscopy. J. Sci. Food Agric. 2011, 92, 1402–1410. [Google Scholar] [CrossRef]
- Jambunathan, R.; Subramanian, V. Grain quality and utilization of sorghum and pearl millet. Biotechnology in tropical crop improvement 1988, 133–139. [Google Scholar]
- Njuguna, V.W.; Cheruiyot, E.K.; Mwonga, S.; Rono, J.K. Effect of genotype and environment on grain quality of sorghum (Sorghum bicolor L. Moench) lines evaluated in Kenya. Afr. J. Plant Sci. 2018, 12, 324–330. [Google Scholar] [CrossRef]
- Pietta, P.G. Flavonoids as antioxidants. Journal of natural products 2000, 63, 1035–1042. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W. Sorghum and millet phenols and antioxidants. J. Cereal Sci. 2006, 44, 236–251. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L. Phenolic compounds in cereal grains and their health benefits. Cereal foods world 2007, 52, 105–111. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, W.L.; Rooney, L.W. Evaluation of phenolics and antioxidant activity of black sorghum hybrids. J. Cereal Sci. 2013, 58, 278–283. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W.; Waniska, R.D.; Rooney, W.L. Phenolic Compounds and Antioxidant Activity of Sorghum Grains of Varying Genotypes. J. Agric. Food Chem. 2005, 53, 6813–6818. [Google Scholar] [CrossRef] [PubMed]
- Awika, J.M.; Rooney, L.W. Sorghum phytochemicals and their potential impact on human health. Phytochemistry 2004, 65, 1199–1221. [Google Scholar] [CrossRef] [PubMed]
- Quettier-Deleu, C.; Gressier, B.; Vasseur, J.; Dine, T.; Brunet, C.; Luyckx, M.; Cazin, M.; Cazin, J.-C.; Bailleul, F.; Trotin, F. Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. J. Ethnopharmacol. 2000, 72, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Chew, K.K.; Khoo, M.Z.; Ng, S.Y.; Thoo, Y.Y.; Aida, W.W.; Ho, C.W. Effect of ethanol concentration, extraction time and extraction temperature on the recovery of phenolic compounds and antioxidant capacity of Orthosiphon stamineus extracts. Int. Food Res. J. 2011, 18, 1427. [Google Scholar]
- Katerere, D.R.; Eloff, J. Antibacterial and antioxidant activity of Sutherlandia frutescens (Fabaceae), a reputed anti-HIV/AIDS. phytomedicine Phytother Res 2005, 19, 779–781. [Google Scholar] [CrossRef]
- Dessalegn, E. In vitro antioxidant and α-amylase inhibition activities of spiced red chili paste (datta) from South Ethiopia. Ethiop. Pharm. J. 2016, 31, 93. [Google Scholar] [CrossRef]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric Quantitation of Antioxidant Capacity through the Formation of a Phosphomolybdenum Complex: Specific Application to the Determination of Vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef]
- Tasie, M.M.; Gebreyes, B.G. Characterization of Nutritional, Antinutritional, and Mineral Contents of Thirty-Five Sorghum Varieties Grown in Ethiopia. Int. J. Food Sci. 2020, 2020, 8243617–11. [Google Scholar] [CrossRef]
- FAO. Sorghum and Pearl Millets in Human Nutrition; Food and Agriculture Organization of the United Nations (FAO) Press: Rome, Italy, 1995. [Google Scholar]
- BSTID-NRC (Board on Science and Technology for International Development- National Research Council). Lost crops of Africa. Academic Press: Washington, DC, USA, 1996; pp. 127–213.
- Singh, R.; Axtell, J.D. High lysine mutant gene (hl) that improves protein quality and biochemical value of grain sorghum. Crop Science 1973, 13, 535–539. [Google Scholar] [CrossRef]
- Kumar, M.S.; Krishna, V.M. Experimental investigation on performance of hybrid PCM’s on addition of nano particles in thermal energy storage. Mater Today Proc. 2019, 17, 271–276. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nation (FAO). Current Worldwide Annual Meat Consumption per capita, Livestock and Fish Primary Equivalent. Food and Agriculture Organization of the United Nations, 2013.
- Yi, B.; Zhou, Y.-F.; Gao, M.-Y.; Zhang, Z.; Han, Y.; Yang, G.-D.; Xu, W.; Huang, R.-D. Effect of Drought Stress During Flowering Stage on Starch Accumulation and Starch Synthesis Enzymes in Sorghum Grains. J. Integr. Agric. 2014, 13, 2399–2406. [Google Scholar] [CrossRef]
- Hariprasanna, K.; Agte, V.; Elangovan, M.; Patil, J.V. Genetic variability for grain iron and zinc content in cultivars, breeding lines and selected germplasm accessions of sorghum [Sorghum bicolor (L.) Moench]. Indian Journal of Genetics 2014, 74, 42–49. [Google Scholar] [CrossRef]
- Mohammed, N.; Ahmed, I.; Babiker, E. Nutritional evaluation of sorghum flour (Sorghum bicolor L. Moench) during processing of injera. Int. J Bio Life Sci. 2010, 6, 35–39. [Google Scholar]
- Welch, R.M. Linkages Between Trace Elements in Food Crops and Human Health. 2008, 287–309. [CrossRef]
- Albrektsson, T.; Johansson, C. Osteoinduction, osteoconduction and osseointegration. Eur. Spine J. 2010, 10 (Suppl. S2), S96–S101. [Google Scholar] [CrossRef]
- Chan, S.S.; Ferguson, E.L.; Bailey, K.; Fahmida, U.; Harper, T.B.; Gibson, R.S. The concentrations of iron, calcium, zinc and phytate in cereals and legumes habitually consumed by infants living in East Lombok, Indonesia. J. Food Compos. Anal. 2007, 20, 609–617. [Google Scholar] [CrossRef]
- Clemens, S. Zn and Fe biofortification: The right chemical environment for human bioavailability. Plant Sci. 2014, 225, 52–57. [Google Scholar] [CrossRef]
- Alfieri, M.; Balconi, C.; Cabassi, G.; Habyarimana, E.; Redaelli, R. Antioxidant activity in a set of sorghum landraces and breeding lines. Maydica 2017, 62, 1–7. [Google Scholar]
- Williams, B.; Kabbage, M.; Kim, H.J.; Britt, R.; Dickman, M.B. Tipping the balance: Sclerotinia sclerotiorum secreted oxalic acid suppresses host defenses by manipulating the host redox environment. PLOS Pathog. 2011, 7, e1002107. [Google Scholar] [CrossRef]
- Punia, H.; Tokas, J.; Malik, A.; Satpal; Sangwan, S. Characterization of phenolic compounds and antioxidant activity in sorghum [Sorghum bicolor (L.) Moench] grains. Cereal Res. Commun. 2021, 49, 343–353. [Google Scholar] [CrossRef]
- Taleon, V.; Dykes, L.; Rooney, W.; Rooney, L. Effect of genotype and environment on flavonoid concentration and profile of black sorghum grains. J. Cereal Sci. 2012, 56, 470–475. [Google Scholar] [CrossRef]
- Awika, J.M. Sorghum: Its Unique Nutritional and Health-Promoting Attributes: In Gluten-Free Ancient Grains: Cereals, Pseudocereals, and Legumes: Sustainable, Nutritious, and Health-Promoting Foods for the 21st Century. 2017.
- Kruger, J.; Taylor, J.R.; Du, X.; De Moura, F.F.; Lönnerdal, B.; Oelofse, A. Effect of phytate reduction of sorghum, through genetic modification, on iron and zinc availability as assessed by an in vitro dialysability bioaccessibility assay, Caco-2 cell uptake assay, and suckling rat pup absorption model. Food Chem. 2013, 141, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Rathore, S.; Karunakar, S.; Vivek, K. Millet Grain Processing, Utilization and its Role in Helath promotion. A review. Int. J. Nutr, Food Sci. 2016, 5, 318–329. [Google Scholar] [CrossRef]
- Elkin, R.G.; Freed, M.B.; Hamaker, B.R.; Zhang, Y.; Parsons, C.M. Condensed Tannins Are Only Partially Responsible for Variations in Nutrient Digestibilities of Sorghum Grain Cultivars. J. Agric. Food Chem. 1996, 44, 848–853. [Google Scholar] [CrossRef]
- Kiprotich, F.K.; Cheruiyot, E.K.; Mwendia, C.M.; Wachira, F.N.; Owuoche, J.O. Biochemical quality indices of sorghum genotypes from East Africa for malting and brewing. African Journal of Biotechnology 2014, 13, 313–321. [Google Scholar]
- Alonso, A.; Aguirre, A.; Marzo, F. Effect of extrusion and traditional processing methods on anti-nutrients and in vitro digestibility of protein and starch in faba bean and kidney beans. Food Chemistry 2000, 68, 159–165. [Google Scholar] [CrossRef]
- Sharma, I.; Mathur, M.; Singh, G.P.; Rishi, A. Quantittaive estimation of phenolic and flavonoid content ns antioxidant activity of various extracts of different parts of Plumbago zeylanica Linn. Int. J. Drug Dev. & Res. 2014.
- Aregay, N.; Belew, D.; Zenebe, A.; Haile, M.; Gebresamuel, G.; Girma, A. Tree Age and Harvesting Season Affected Physico-chemical and Bioactive Compounds of Elite Type of Gunda Gundo Orange (Citrus Spp) in the Northern Ethiopia. Int. J. Fruit Sci. 2021, 21, 26–39. [Google Scholar] [CrossRef]
- Ljiljana, S.; Mihajlo, S.; Vensa, N.; Ljubisa, N.; Dusica, R.; Jasna, C.B.; Vensa, T. Antioxidant Activity and Total Phenolic and Flavonoid Contents of Hieracium pilosella L. Extracts. Sensors 2009, 9, 5702–5714; [Google Scholar]
- Alsayadi, M. Antioxidant activity and nutrient composition of Sorghum bicolor L. and Secale cereale L. in Algeria. Acad. J. Food Res. 2013, 1, 059–065. [Google Scholar]
- Rohman, A.; Riyanto, S.; Yuniarti, N.; Saputra, W.R.; Utami, R.; Mulatsih, W. Antioxidant activity, total phenolic, and total flavaonoid of extracts and fractions of red fruit (Pandanus conoideus Lam). Int. Food Res. J. 2010, 17, 97–106. [Google Scholar]
- Shen, S.; Huang, R.; Li, C.; Wu, W.; Chen, H.; Shi, J.; Chen, S.; Ye, X. Phenolic Compositions and Antioxidant Activities Differ Significantly among Sorghum Grains with Different Applications. Molecules 2018, 23, 1203. [Google Scholar] [CrossRef]
- Awika, J.M. Sorghum phenols as antioxidants. M.Sc. Thesis, Texas A&M University, usa, College Station, TX, 2000. [Google Scholar]
- Semere, T.; Fjellheim, S.; Tsehaye, Y.; Westengen, O.T. Inventory of sorghum landraces diversity on-farm and adaptability responses to changing climate: implications for sorghum breeding and conservation. Genetic Resources and Crop Evolution 2023, 1–18. [Google Scholar]
- Tsehaye, Y.; Berg, T.; Tsegaye, B. & Tanto, T. Farmers’ management of finger millet (Eleusine coracana L.) diversity in Tigray, Ethiopia and implications for on-farm conservation. Biodivers. Conserv. 2006, 15, 4289–4308. [Google Scholar]
- Hikeezi. The Importance of Sorghum Grain Colour and Hardness, and Their Causes and Measurement. International Sorghum and Millet Collaborative Research Support Program (INTSORMIL CRSP) Presentations. 2010. Available online: https://digitalcommons.unl.edu/intsormilpresent/18.
- Harris, H.B.; Burns, R.E. Influence of tannin content on preharvest seed germination in sorghum seed molding. Agronomy Journal 1970, 65, 957–959. [Google Scholar] [CrossRef]
- Asfaw, Z. Variation in the Morphology of the Spike within Ethiopian Barley,Hordeum vulgareL. (Poaceae). Acta Agric. Scand. 1988, 38, 277–288. [Google Scholar] [CrossRef]
- Deepika, K.; Lavuri, K.; Rathod, S.; Yeshala, C.M.; Jukanti, A.K.; Reddy, S.N.; Lv, S.R.; Badri, J. Multivariate analysis of geographically diverse rice germplasm for genetic improvement of yield, dormancy and shattering-related traits. Plant Genet. Resour. Charact. Util. 2021, 19, 144–152. [Google Scholar] [CrossRef]
- Seiler, G.J.; Stafford, R.E. Factor Analysis of Components of Yield in Guar 1. Crop. Sci. 1985, 25, 905–908. [Google Scholar] [CrossRef]
- Yan, W.; Fregeau-Reid, J.A. Breeding line selection based on multiple traits. Crop Sci. 2008, 48, 417–423. [Google Scholar] [CrossRef]
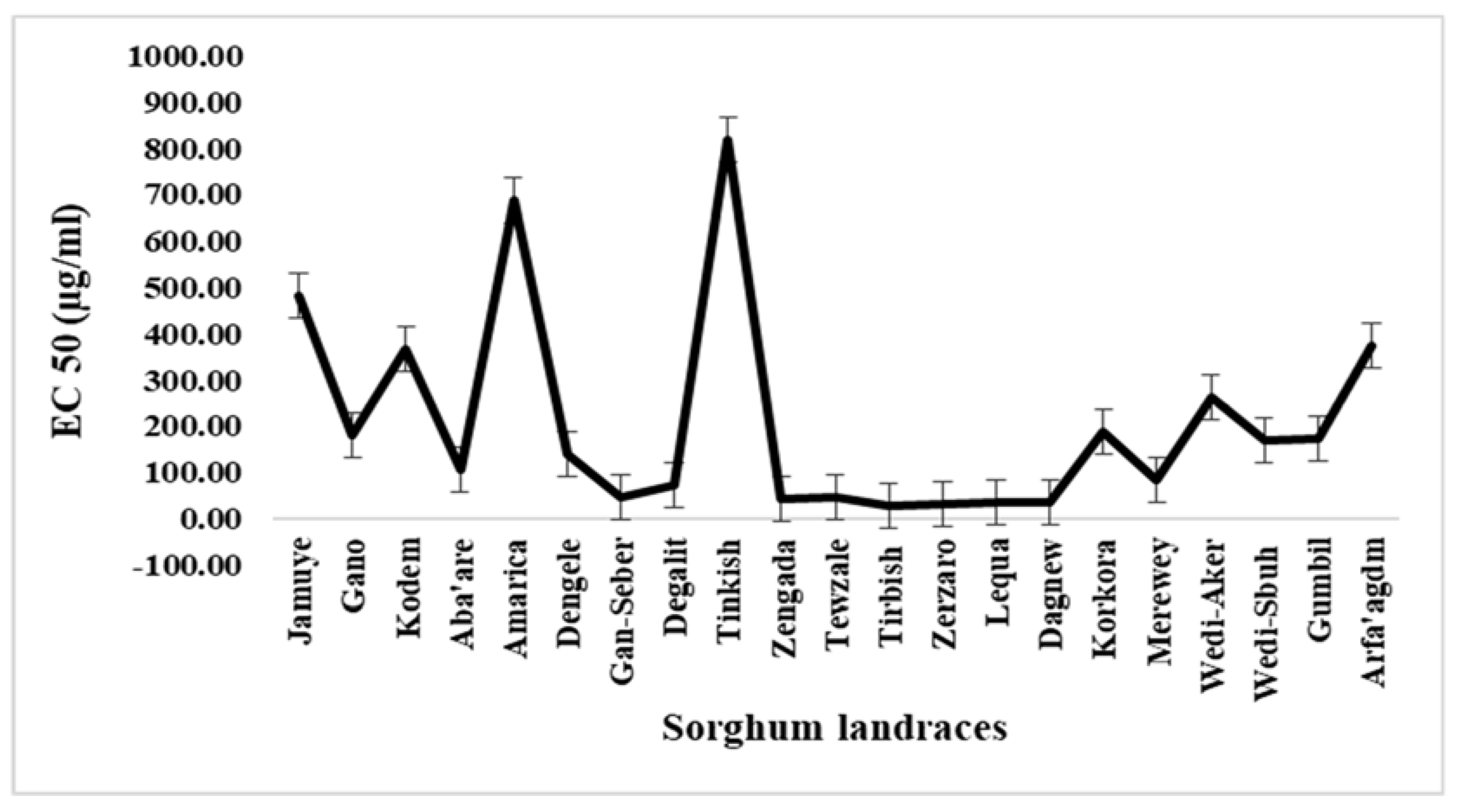


| No. | Landrace name | Zone of origin | Race | Seed color | Preferred end-use |
|---|---|---|---|---|---|
| 1 | Jamuye | Southern Tigray | Durra | Yellow | Swa/Enjera |
| 2 | Gano | Southern Tigray | Durra | Yellow | Enjera |
| 3 | Kodem | Southern Tigray | Durra | Yellow | Enjera/Swa |
| 4 | Aba'are | Southern Tigray | Durra | Yellow | Enjera |
| 5 | Amarica | Southern Tigray | Durra | White | Enjera |
| 6 | Dengele | Southern Tigray | Durra | Yellow | Enjera/Swa |
| 7 | Gan-Seber | Southern Tigray | Durra | White | Swa/Enjera |
| 8 | Degalit | Southern Tigray | Durra | Yellow | Swa/Enjera |
| 9 | Tinkish | Southern Tigray | Durra | Yellow | Enjera/popping |
| 10 | Zengada | Southern Tigray | Bicolor | Red | Enjera/Swa |
| 11 | Tewzale | Central Tigray | Caudatum | Red | Enjera/Swa |
| 12 | Tirbish | Central Tigray | Durra | Yellow | Swa/Enjera |
| 13 | Zerzaro | Central Tigray | Durra | Brown | Swa/Enjera |
| 14 | Lequa | Central Tigray | Bicolor | Brown | Swa |
| 15 | Dagnew | Western Tigray | Bicolor | Yellow | Swa/Enjera |
| 16 | Korkora | Western Tigray | Caudatum | White | Enjera |
| 17 | Merewey | Western Tigray | Durra | Yellow | Enjera |
| 18 | Wedi-Aker | Western Tigray | Caudatum | White | Swa |
| 19 | Wedi-Sbuh | Western Tigray | Durra | White | Enjera |
| 20 | Gumbil | Central Tigray | Durra | Yellow | Enjera |
| 21 | Arfa’agdm | Western Tigray | Caudatum | White | Enjera |
| Variable | Protein | Starch | Fe | Zn | Cu | Mn | Cr | TFC | TTC | EC50 | FRAP | TAC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Max | 18 | 78.3 | 101 | 43 | 5.3 | 20.2 | 1.5 | 665 | 7.5 | 818.4 | 334.8 | 63.9 |
| Min | 6.21 | 33.4 | 32 | 16.9 | 1.5 | 9.21 | 0 | 0 | 0.18 | 29.1 | 17.9 | 1.71 |
| Mean | 11.4 | 69.3 | 51.6 | 27 | 2.9 | 14.7 | 1.01 | 198.4 | 2.61 | 208 | 115 | 23.5 |
| SE | 0.12 | 0.32 | 3.29 | 1.42 | 0.2 | 0.55 | 0.1 | 46.7 | 0.61 | 48.6 | 23.1 | 4.45 |
| SD | 2.2 | 6.12 | 15.1 | 6.5 | 0.9 | 2.51 | 0.35 | 214.2 | 2.78 | 222.6 | 105.9 | 20.4 |
| CV | 19.34 | 8.83 | 29.2 | 24.1 | 32 | 17.1 | 34.3 | 108 | 106.5 | 106.9 | 92.35 | 86.8 |
| T-test | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Protein | Starch | Fe | Zn | Cu | Mn | Cr | TFC | TTC | EC50 | FRAP | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Starch | -0.26 | ||||||||||
| Fe | 0.21 | -0.11 | |||||||||
| Zn | 0.17 | 0.21 | 0.34 | ||||||||
| Cu | 0.40 | -0.17 | 0.73** | 0.53* | |||||||
| Mn | 0.03 | -0.04 | 0.16 | 0.26 | 0.48* | ||||||
| Cr | 0.03 | -0.15 | 0.12 | 0.26 | 0.02 | -0.10 | |||||
| TFC | -0.14 | -0.10 | -0.06 | 0.15 | -0.04 | -0.14 | 0.22 | ||||
| TTC | -0.43 | -0.26 | 0.05 | -0.23 | 0.00 | -0.07 | 0.16 | 0.70** | |||
| EC50 | 0.28 | -0.41 | 0.15 | -0.12 | 0.11 | 0.07 | -0.07 | -0.63** | -0.43 | ||
| FRAP | -0.31 | -0.15 | -0.08 | 0.01 | -0.05 | -0.15 | 0.19 | 0.92** | 0.86** | -0.63** | |
| TAC | -0.31 | -0.18 | -0.06 | -0.18 | -0.10 | -0.15 | 0.10 | 0.87** | 0.90** | -0.57** | 0.95** |
| Mean of nutritional traits | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cluster | Protein | Starch | Fe | Zn | Cu | Mn | Cr | TFC | TTC | EC50 | FRAP | TAC |
| I | 13.17 | 72.38 | 59.99 | 32.48 | 3.7 | 16.24 | 1.12 | 119.6 | 1.02 | 231.6 | 62 | 12.39 |
| II | 10.27 | 72.8 | 45.01 | 23.13 | 2.32 | 14.02 | 0.85 | 121.3 | 2.08 | 199.6 | 82 | 17.47 |
| III | 14.67 | 53 | 55.49 | 25.19 | 2.91 | 14.24 | 1.39 | 0 | 0.59 | 818.37 | 18 | 1.71 |
| IV | 10.34 | 67.88 | 50.74 | 26.28 | 2.83 | 13.7 | 1.07 | 559.3 | 7.12 | 34.8 | 303 | 61.94 |
| Clusters | I | II | III | IV |
|---|---|---|---|---|
| I | - | |||
| II | 40.18** | - | ||
| III | 212.12** | 238.15** | - | |
| IV | 673.98** | 729.15** | 1173.79** | - |
| Trait | PC 1 | PC 2 | PC 3 | PC 4 |
|---|---|---|---|---|
| Protein | -0.43 | 0.38 | -0.27 | 0.32 |
| Starch | -0.07 | -0.25 | 0.89 | -0.02 |
| Fe | -0.17 | 0.75 | -0.06 | -0.07 |
| Zn | -0.13 | 0.65 | 0.53 | 0.27 |
| Cu | -0.22 | 0.91 | 0.01 | -0.21 |
| Mn | -0.22 | 0.45 | 0.16 | -0.59 |
| Cr | 0.19 | 0.28 | -0.05 | 0.72 |
| TFC | 0.89 | 0.24 | 0.05 | 0.15 |
| TTC | 0.88 | 0.15 | -0.24 | -0.21 |
| EC50 | -0.69 | -0.01 | -0.56 | -0.02 |
| FRAP | 0.97 | 0.17 | -0.03 | 0.01 |
| TAC | 0.96 | 0.10 | -0.14 | -0.11 |
| Eigenvalue | 4.26 | 2.42 | 1.57 | 1.17 |
| % Total variance | 35.54 | 20.16 | 13.08 | 9.71 |
| % Cumulative variance | 35.54 | 55.70 | 68.78 | 78.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




