Submitted:
07 August 2023
Posted:
10 August 2023
You are already at the latest version
Abstract
Keywords:
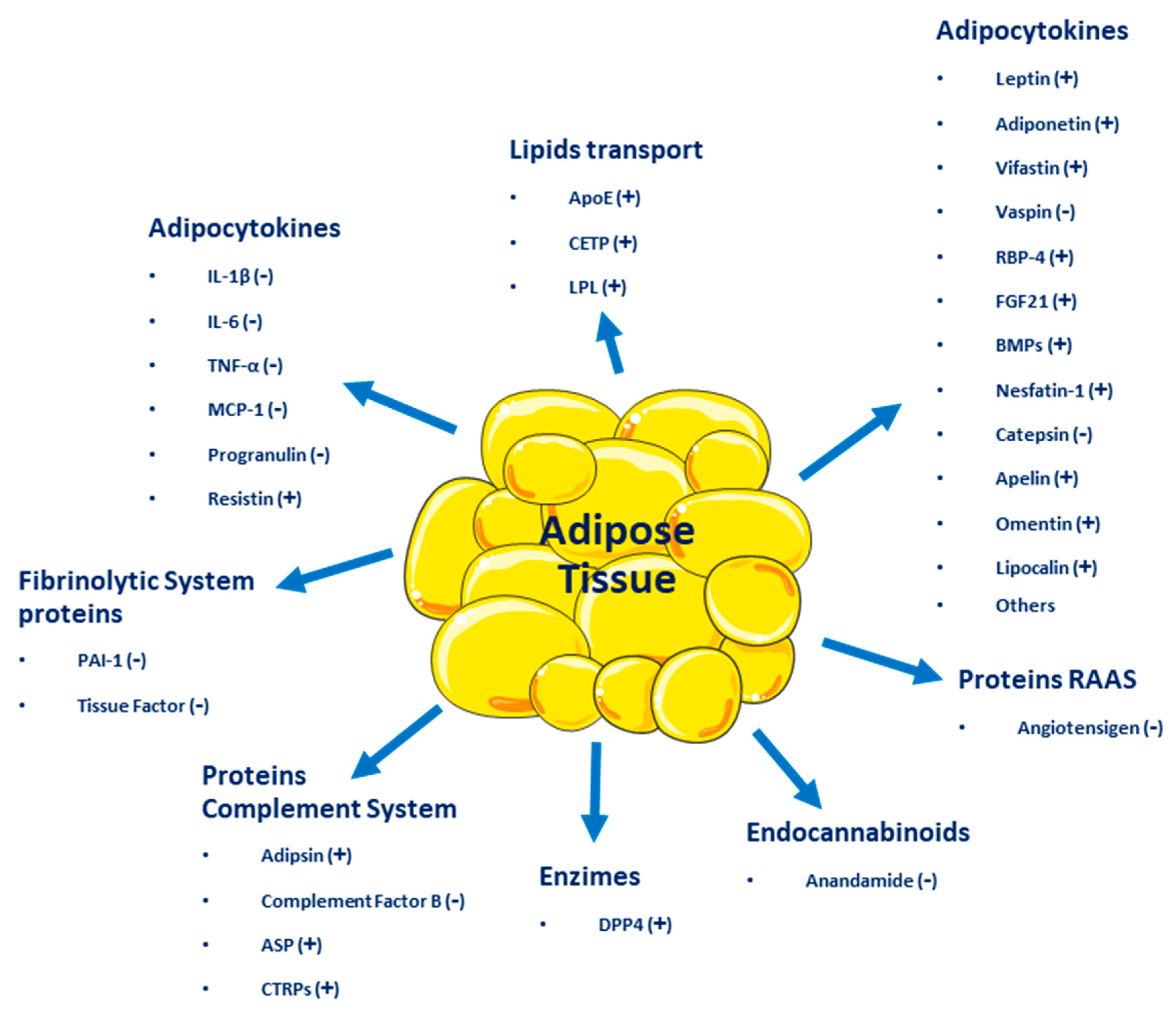
Adipose Tissue
Adipose Tissue as an Endocrine Organ
Definition and Incidence of Overweight and Obesity
Causes or Mechanisms of Obesity
Genetic Factors
Fat Cells
Dysregulation of Energy Balance
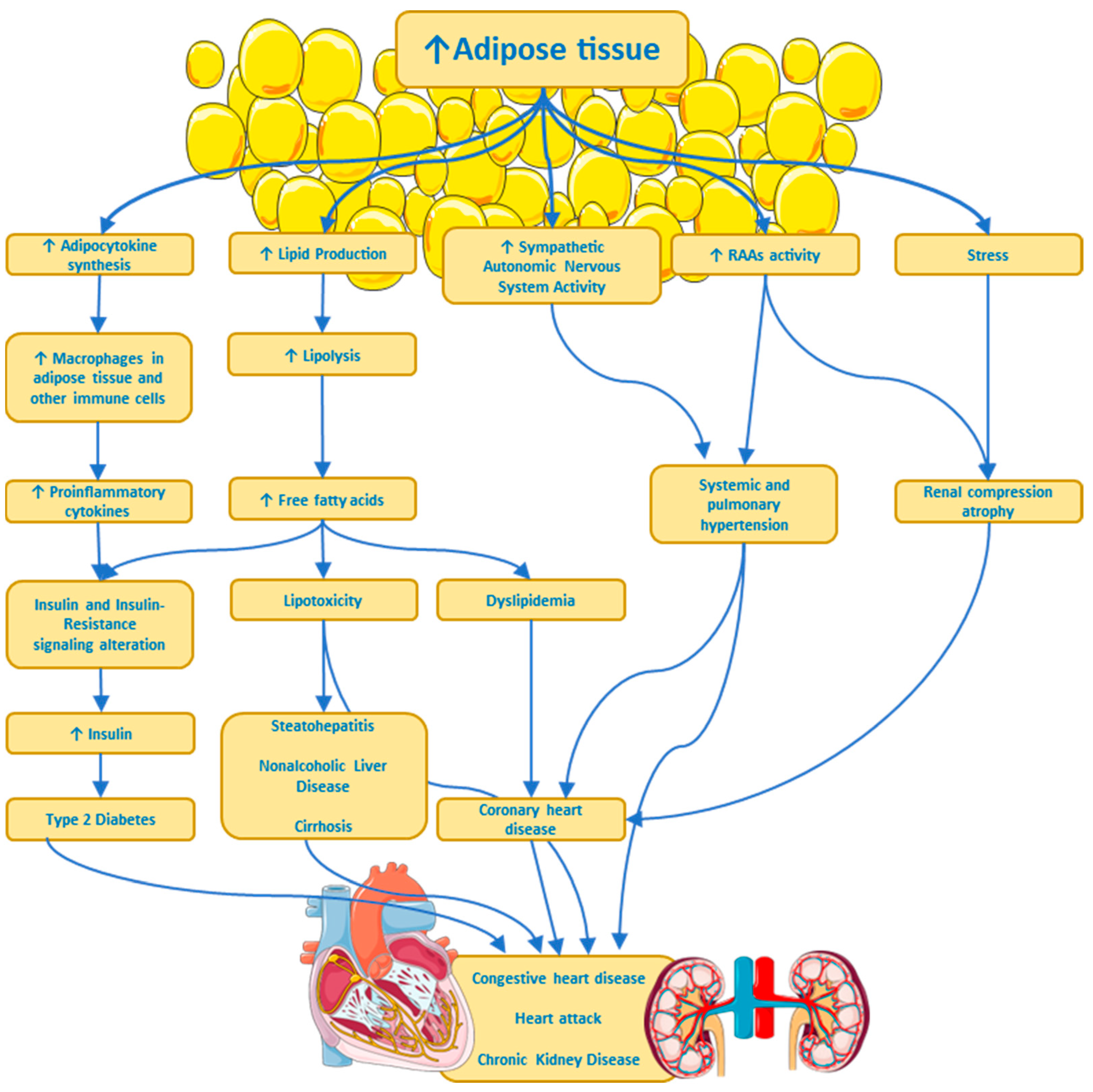
Metabolic and Physiological Effects
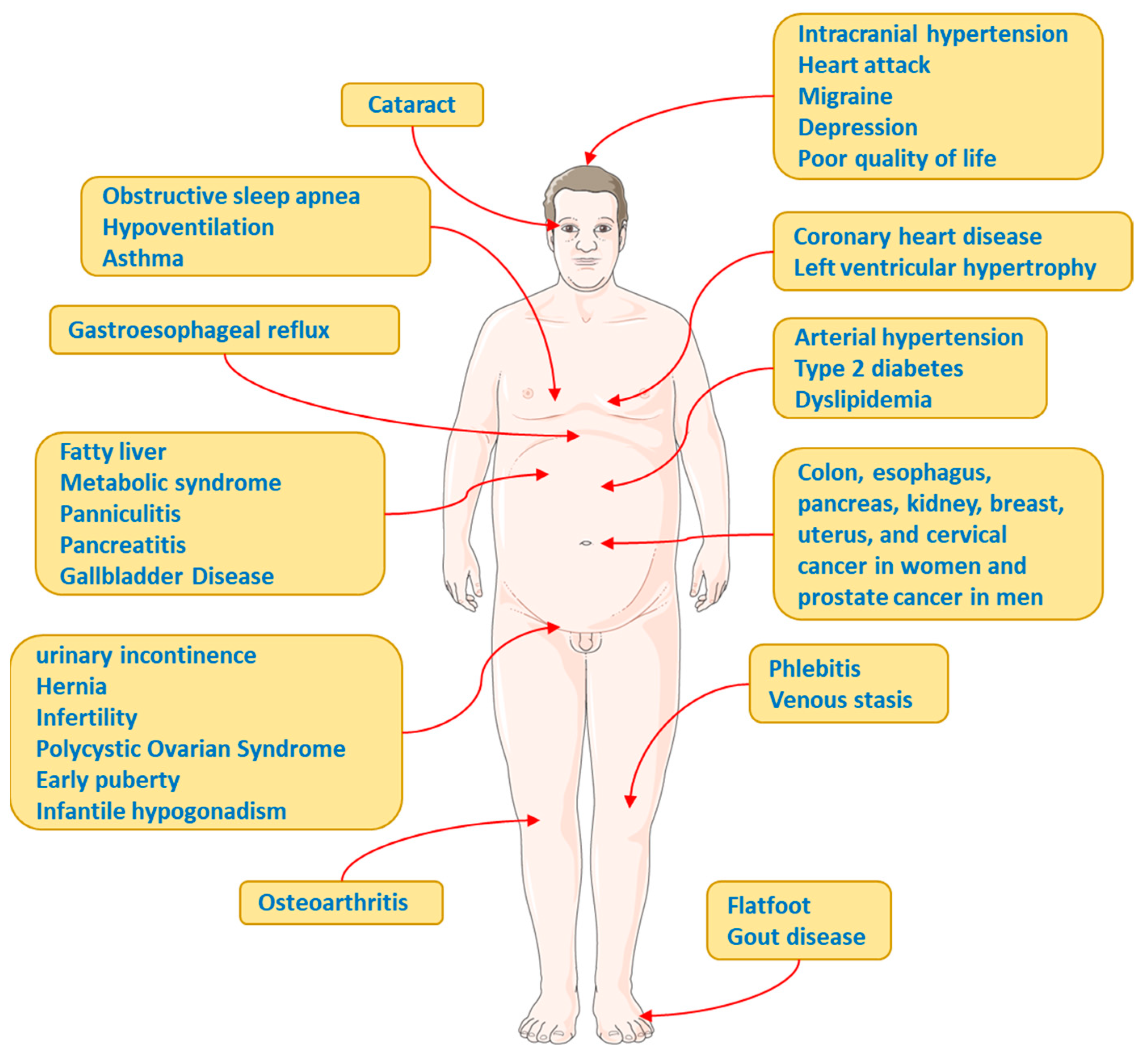
Complications and Comorbidities Associated with Obesity
Insulin Resistance (IR)
Type 2 Diabetes
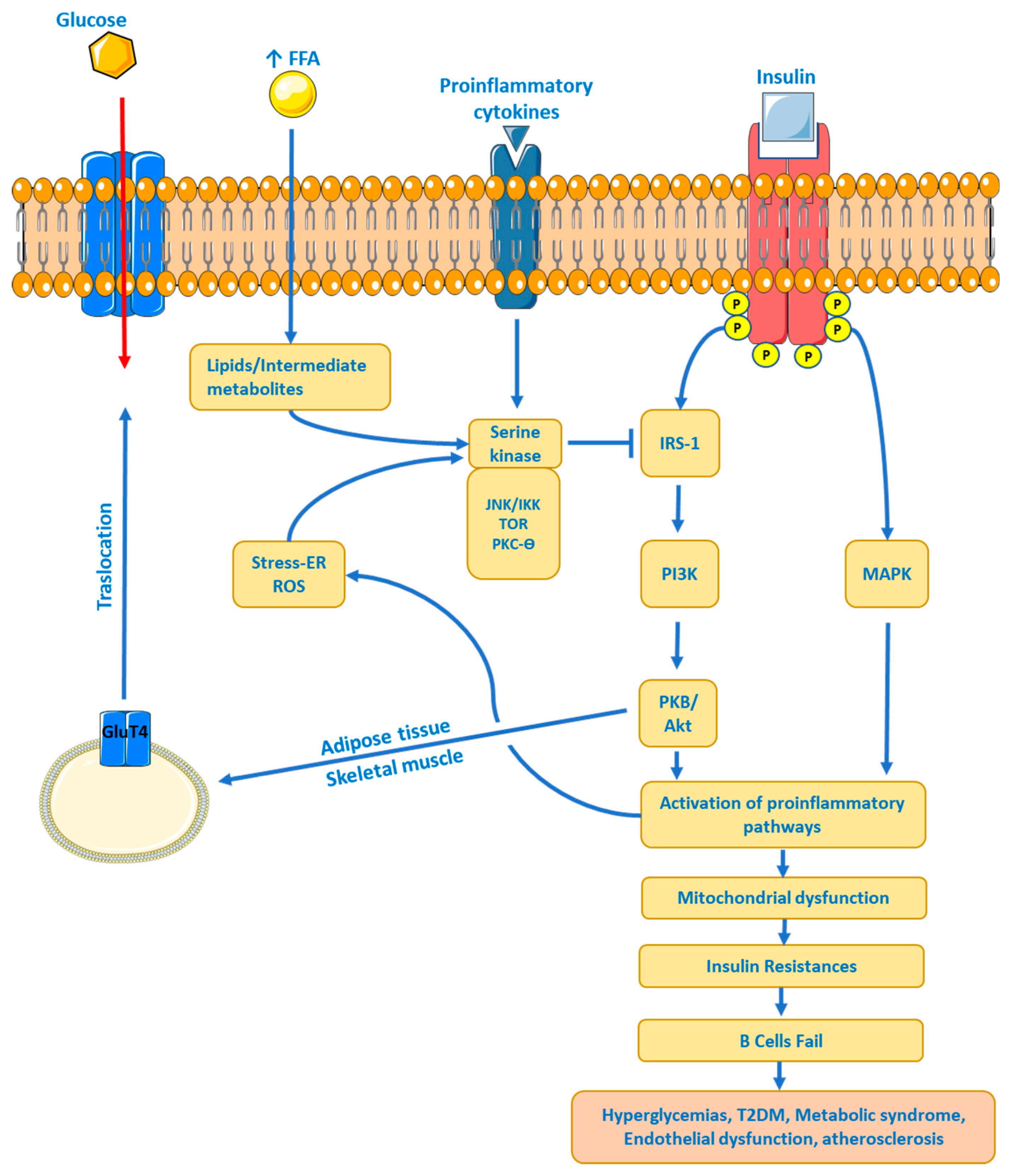
Dyslipidaemia
Hypertension
Other Complications and Comorbidities
Management of the Overweight-Obesity Patient
Lifestyle Modification
Pharmacotherapy: Who Are Candidates to Receive Anti-Obesity Drugs?
Orlistat
Liraglutide
Phentermine
Naltrexone/Bupropion
Phentermine/Topiramate
Diethylpropion
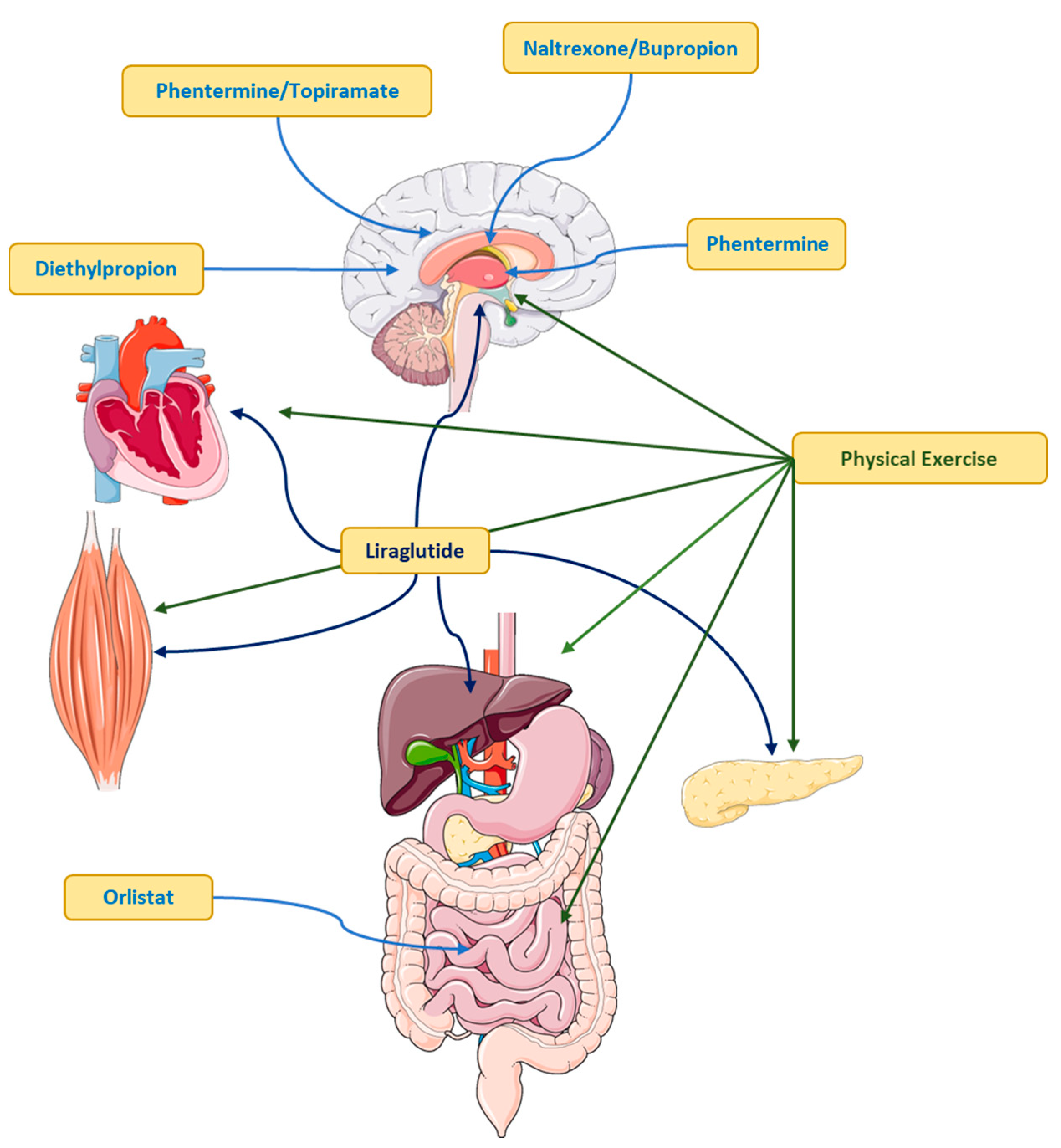
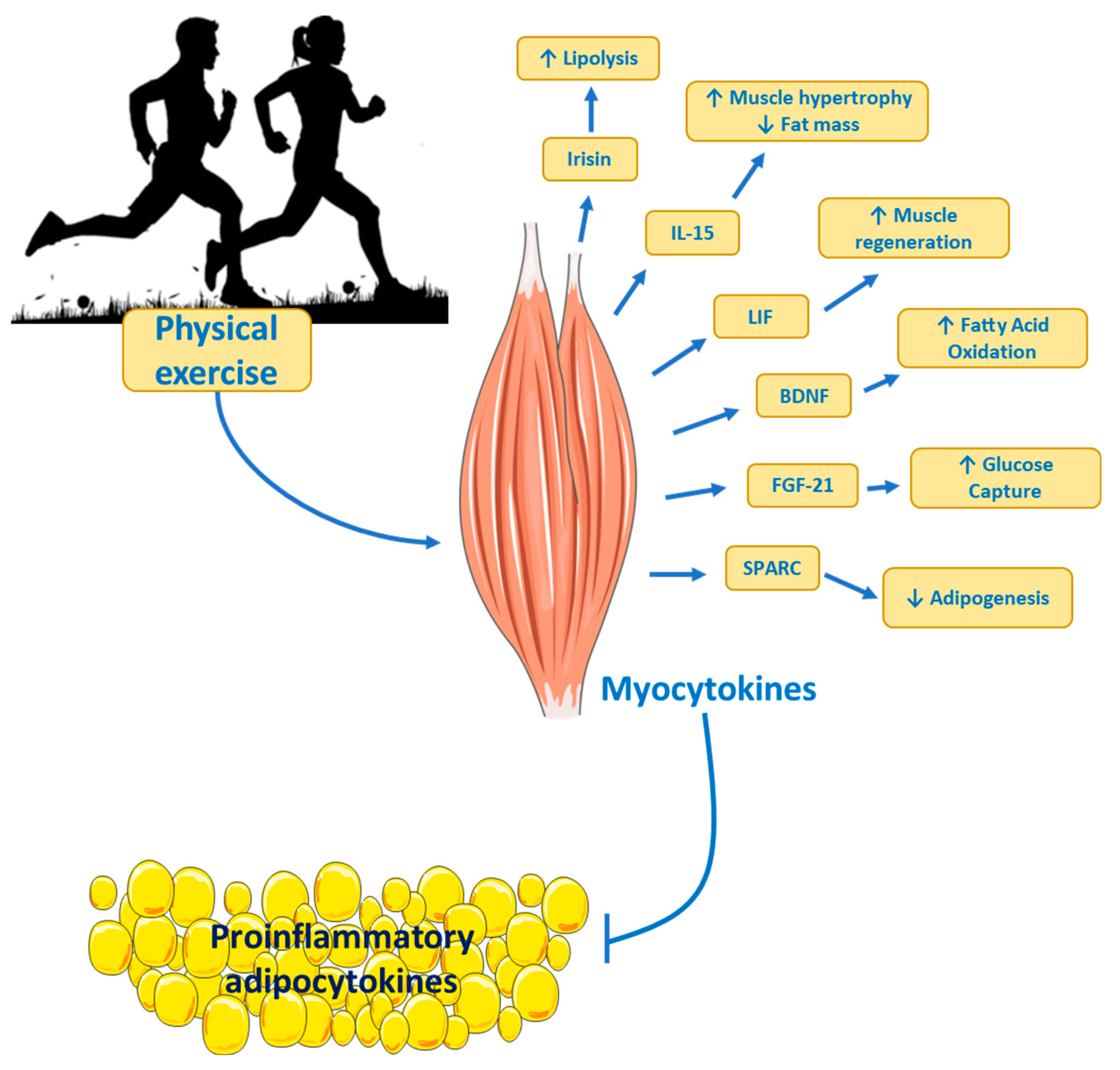
Physical Exercise as Treatment
Conclusions
References
- Booth, A. Magnuson, J. Fouts, M.T. Foster, Adipose tissue: an endocrine organ playing a role in metabolic regulation. Horm Mol Biol Clin Investig 2016, 26, 25–42. [CrossRef] [PubMed]
- D. García-Torres, Castellanos-González, M., Cedeño-Morales, R., Benet-Rodríguez, M., Ramírez-Arteaga, I., Adipose Tissue as an Endocrine Gland. Pathophysiological Implications, Revista Finlay [revista en Internet]. 2011 [citado 2017 Ene 5]; 1(2): [aprox. 20p.]. Disponible en: http://www.revfinlay.sld.cu/index.php/finlay/article/view/39, 2017.
- A.E. Ringel, J.M. Drijvers, G.J. Baker, A. Catozzi, J.C. García-Cañaveras, B.M. Gassaway, B.C. Miller, V.R. Juneja, T.H. Nguyen, S. Joshi, C.H. Yao, H. Yoon, P.T. Sage, M.W. LaFleur, J.D. Trombley, C.A. Jacobson, Z. Maliga, S.P. Gygi, P.K. Sorger, J.D. Rabinowitz, A.H. Sharpe, M.C. Haigis, Obesity Shapes Metabolism in the Tumor Microenvironment to Suppress Anti-Tumor Immunity. Cell 2020, 183, 1848–1866.
- L. Gartner, Hiatt, J., Color Textbook of Histology, McGraw-Hill Interamericana Editores, S.A. de C.V., 2007.
- F. Wang, Vihma, V., Soronen, J., Turpeinen, U., Hämäläinen, E., Savolainen-Peltonen, H., et al., 17β estradiol and estradiol fatty acyl esters and estrogen-converting enzyme expression in adipose tissue in obese men and women, J Clin Endochrinol Metab, 2013, pp. 4923–4931.
- G. Vega-Robledo, Rico-Rosillo, MG., Adipose tissue: immune function and alterations caused by obesity, Rev Alerg Mex, 2016, pp. 340–353.
- K. Svensson, Long, JZ., Jedrychowski, MP., Cohen, P., Lo, JC., Serag, S., et al., A secreted Slit2 fragment regulates adipose tissue thermogenesis and metabolic function, Cell Metab, 2016, pp. 454–466.
- Chernukha, Fedulova, LV., Kotenkova, EA., White, beige and brown adipose tissue: structure, function, specific features and possibility formation and divergence in pigs, Foods and Raw Materials, 2022, pp. 10–18.
- S. O'Neill, L. O'Driscoll, Metabolic syndrome: a closer look at the growing epidemic and its associated pathologies. Obes Rev 2015, 16, 1–12. [CrossRef] [PubMed]
- J.C. Rathmell, Obesity, Immunity, and Cancer. N Engl J Med 2021, 384, 1160–1162. [CrossRef]
- M. Fasshauer, M. Blüher, Adipokines in health and disease. Trends Pharmacol Sci 2015, 36, 461–70. [CrossRef]
- L. Szablewski, Introductory Chapter: Adipose Tissue, IntechOpen, 2019.
- G. Atzmon, X.M. Yang, R. Muzumdar, X.H. Ma, I. Gabriely, N. Barzilai, Differential gene expression between visceral and subcutaneous fat depots, Horm Metab Res 34(11-12) (2002) 622-8.
- J.P. Bastard, M. Maachi, C. Lagathu, M.J. Kim, M. Caron, H. Vidal, J. Capeau, B. Feve, Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw 2006, 17, 4–12.
- J.N. Fain, A.K. Madan, M.L. Hiler, P. Cheema, S.W. Bahouth, Comparison of the release of adipokines by adipose tissue, adipose tissue matrix, and adipocytes from visceral and subcutaneous abdominal adipose tissues of obese humans. Endocrinology 2004, 145, 2273–82. [CrossRef]
- H.M. Choi, H.M. Doss, K.S. Kim, Multifaceted Physiological Roles of Adiponectin in Inflammation and Diseases. Int J Mol Sci 2020, 21.
- A.-R. Aida-Souki, N.J., Prieto-Fuenmayor, C., Cano-Ponce, C., Aspectos básicos en obesidad, Ediciones Universidad Simón Bolívar, Barranquilla, Colombia:, 2018, pp. 1–44.
- D. Ulbricht, J. Pippel, S. Schultz, R. Meier, N. Sträter, J.T. Heiker, A unique serpin P1' glutamate and a conserved β-sheet C arginine are key residues for activity, protease recognition and stability of serpinA12 (vaspin). Biochem J 2015, 470, 357–67. [CrossRef]
- Fukuhara, M. Matsuda, M. Nishizawa, K. Segawa, M. Tanaka, K. Kishimoto, Y. Matsuki, M. Murakami, T. Ichisaka, H. Murakami, E. Watanabe, T. Takagi, M. Akiyoshi, T. Ohtsubo, S. Kihara, S. Yamashita, M. Makishima, T. Funahashi, S. Yamanaka, R. Hiramatsu, Y. Matsuzawa, I. Shimomura, Visfatin: a protein secreted by visceral fat that mimics the effects of insulin. Science 2005, 307, 426–30.
- J. DeFuria, A.C. Belkina, M. Jagannathan-Bogdan, J. Snyder-Cappione, J.D. Carr, Y.R. Nersesova, D. Markham, K.J. Strissel, A.A. Watkins, M. Zhu, J. Allen, J. Bouchard, G. Toraldo, R. Jasuja, M.S. Obin, M.E. McDonnell, C. Apovian, G.V. Denis, B.S. Nikolajczyk, B cells promote inflammation in obesity and type 2 diabetes through regulation of T-cell function and an inflammatory cytokine profile. Proc Natl Acad Sci U S A 2013, 110, 5133–8.
- Y. Ren, H. Y. Ren, H. Zhao, C. Yin, X. Lan, L. Wu, X. Du, H.R. Griffiths, D. Gao, Adipokines, Hepatokines and Myokines: Focus on Their Role and Molecular Mechanisms in Adipose Tissue Inflammation, Front Endocrinol (Lausanne) 13 (2022) 873699.
- C.o.O.G. WHO, Switzerland) & World Health Organization, Obesity: Preventing and Managing the Global Epidemic. Report of a WHO Consultation. Report of a WHO Consultation., World Health Organ Tech Rep Ser, 2000, pp. 1–253.
- P. Poirier, T.D. Giles, G.A. Bray, Y. Hong, J.S. Stern, F.X. Pi-Sunyer, R.H. Eckel, Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss. Arterioscler Thromb Vasc Biol 2006, 26, 968–76. [CrossRef]
- C.M. Apovian, Obesity: definition, comorbidities, causes, and burden, Am J Manag Care 22(7 Suppl) (2016) s176-85.
- X. Lin, H. Li, Obesity: Epidemiology, Pathophysiology, and Therapeutics, Front Endocrinol (Lausanne) 12 (2021) 706978.
- J. Upadhyay, O. Farr, N. Perakakis, W. Ghaly, C. Mantzoros, Obesity as a Disease. Med Clin North Am 2018, 102, 13–33.
- G. Whitlock, S. Lewington, P. Sherliker, R. Clarke, J. Emberson, J. Halsey, N. Qizilbash, R. Collins, R. Peto, P.S. Collaboration, Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 2009, 373, 1083–96. [CrossRef]
- G.A. Bray, K.K. Kim, J.P.H. Wilding, W.O. Federation, Obesity: a chronic relapsing progressive disease process. A position statement of the World Obesity Federation. Obes Rev 2017, 18, 715–723. [CrossRef] [PubMed]
- B.A. Swinburn, G. Sacks, K.D. Hall, K. McPherson, D.T. Finegood, M.L. Moodie, S.L. Gortmaker, The global obesity pandemic: shaped by global drivers and local environments. Lancet 2011, 378, 804–14. [CrossRef] [PubMed]
- K. Sahoo, B. Sahoo, A.K. Choudhury, N.Y. Sofi, R. Kumar, A.S. Bhadoria, Childhood obesity: causes and consequences. J Family Med Prim Care 2015, 4, 187–92.
- WHO, Obesidad y Sobrepeso, 2021.
- D. Gómez-Pérez, M. Cancino, P.I. Moreno, M.S. Ortiz, Weight Stigma, Chronic Stress, Unhealthy Diet, and Obesity in Chilean Adults. Int J Behav Med 2021, 28, 292–298. [CrossRef]
- Instituto de Nutrición y Tecnología de los Alimentos, ESPECIAL: Obesidad, la otra pandemia, INTA, 2021.
- S.B. Heymsfield, T.A. Wadden, Mechanisms, Pathophysiology, and Management of Obesity. N Engl J Med 2017, 376, 1492.
- T.S. Church, D.M. Thomas, C. Tudor-Locke, P.T. Katzmarzyk, C.P. Earnest, R.Q. Rodarte, C.K. Martin, S.N. Blair, C. Bouchard, Trends over 5 decades in U.S. occupation-related physical activity and their associations with obesity. PLoS One 2011, 6, e19657.
- V.Y. Njike, T.M. Smith, O. Shuval, K. Shuval, I. Edshteyn, V. Kalantari, A.L. Yaroch, Snack Food, Satiety, and Weight. Adv Nutr 2016, 7, 866–78.
- V. Medici, S.A. McClave, K.R. Miller, Common Medications Which Lead to Unintended Alterations in Weight Gain or Organ Lipotoxicity. Curr Gastroenterol Rep 2016, 18, 2. [CrossRef] [PubMed]
- Y. Wu, H. Duan, X. Tian, C. Xu, W. Wang, W. Jiang, Z. Pang, D. Zhang, Q. Tan, Genetics of Obesity Traits: A Bivariate Genome-Wide Association Analysis, Front Genet 9 (2018) 179.
- D. Shungin, T.W. Winkler, D.C. Croteau-Chonka, T. Ferreira, A.E. Locke, R. Mägi, R.J. Strawbridge, T.H. Pers, K. Fischer, A.E. Justice, T. Workalemahu, J.M.W. Wu, M.L. Buchkovich, N.L. Heard-Costa, T.S. Roman, A.W. Drong, C. Song, S. Gustafsson, F.R. Day, T. Esko, T. Fall, Z. Kutalik, J. Luan, J.C. Randall, A. Scherag, S. Vedantam, A.R. Wood, J. Chen, R. Fehrmann, J. Karjalainen, B. Kahali, C.T. Liu, E.M. Schmidt, D. Absher, N. Amin, D. Anderson, M. Beekman, J.L. Bragg-Gresham, S. Buyske, A. Demirkan, G.B. Ehret, M.F. Feitosa, A. Goel, A.U. Jackson, T. Johnson, M.E. Kleber, K. Kristiansson, M. Mangino, I.M. Leach, C. Medina-Gomez, C.D. Palmer, D. Pasko, S. Pechlivanis, M.J. Peters, I. Prokopenko, A. Stančáková, Y.J. Sung, T. Tanaka, A. Teumer, J.V. Van Vliet-Ostaptchouk, L. Yengo, W. Zhang, E. Albrecht, J. Ärnlöv, G.M. Arscott, S. Bandinelli, A. Barrett, C. Bellis, A.J. Bennett, C. Berne, M. Blüher, S. Böhringer, F. Bonnet, Y. Böttcher, M. Bruinenberg, D.B. Carba, I.H. Caspersen, R. Clarke, E.W. Daw, J. Deelen, E. Deelman, G. Delgado, A.S. Doney, N. Eklund, M.R. Erdos, K. Estrada, E. Eury, N. Friedrich, M.E. Garcia, V. Giedraitis, B. Gigante, A.S. Go, A. Golay, H. Grallert, T.B. Grammer, J. Gräßler, J. Grewal, C.J. Groves, T. Haller, G. Hallmans, C.A. Hartman, M. Hassinen, C. Hayward, K. Heikkilä, K.H. Herzig, Q. Helmer, H.L. Hillege, O. Holmen, S.C. Hunt, A. Isaacs, T. Ittermann, A.L. James, I. Johansson, T. Juliusdottir, I.P. Kalafati, L. Kinnunen, W. Koenig, I.K. Kooner, W. Kratzer, C. Lamina, K. Leander, N.R. Lee, P. Lichtner, L. Lind, J. Lindström, S. Lobbens, M. Lorentzon, F. Mach, P.K. Magnusson, A. Mahajan, W.L. McArdle, C. Menni, S. Merger, E. Mihailov, L. Milani, R. Mills, A. Moayyeri, K.L. Monda, S.P. Mooijaart, T.W. Mühleisen, A. Mulas, G. Müller, M. Müller-Nurasyid, R. Nagaraja, M.A. Nalls, N. Narisu, N. Glorioso, I.M. Nolte, M. Olden, N.W. Rayner, F. Renstrom, J.S. Ried, N.R. Robertson, L.M. Rose, S. Sanna, H. Scharnagl, S. Scholtens, B. Sennblad, T. Seufferlein, C.M. Sitlani, A.V. Smith, K. Stirrups, H.M. Stringham, J. Sundström, M.A. Swertz, A.J. Swift, A.C. Syvänen, B.O. Tayo, B. Thorand, G. Thorleifsson, A. Tomaschitz, C. Troffa, F.V. van Oort, N. Verweij, J.M. Vonk, L.L. Waite, R. Wennauer, T. Wilsgaard, M.K. Wojczynski, A. Wong, Q. Zhang, J.H. Zhao, E.P. Brennan, M. Choi, P. Eriksson, L. Folkersen, A. Franco-Cereceda, A.G. Gharavi, Å. Hedman, M.F. Hivert, J. Huang, S. Kanoni, F. Karpe, S. Keildson, K. Kiryluk, L. Liang, R.P. Lifton, B. Ma, A.J. McKnight, R. McPherson, A. Metspalu, J.L. Min, M.F. Moffatt, G.W. Montgomery, J.M. Murabito, G. Nicholson, D.R. Nyholt, C. Olsson, J.R. Perry, E. Reinmaa, R.M. Salem, N. Sandholm, E.E. Schadt, R.A. Scott, L. Stolk, E.E. Vallejo, H.J. Westra, K.T. Zondervan, P. Amouyel, D. Arveiler, S.J. Bakker, J. Beilby, R.N. Bergman, J. Blangero, M.J. Brown, M. Burnier, H. Campbell, A. Chakravarti, P.S. Chines, S. Claudi-Boehm, F.S. Collins, D.C. Crawford, J. Danesh, U. de Faire, E.J. de Geus, M. Dörr, R. Erbel, J.G. Eriksson, M. Farrall, E. Ferrannini, J. Ferrières, N.G. Forouhi, T. Forrester, O.H. Franco, R.T. Gansevoort, C. Gieger, V. Gudnason, C.A. Haiman, T.B. Harris, A.T. Hattersley, M. Heliövaara, A.A. Hicks, A.D. Hingorani, W. Hoffmann, A. Hofman, G. Homuth, S.E. Humphries, E. Hyppönen, T. Illig, M.R. Jarvelin, B. Johansen, P. Jousilahti, A.M. Jula, J. Kaprio, F. Kee, S.M. Keinanen-Kiukaanniemi, J.S. Kooner, C. Kooperberg, P. Kovacs, A.T. Kraja, M. Kumari, K. Kuulasmaa, J. Kuusisto, T.A. Lakka, C. Langenberg, L. Le Marchand, T. Lehtimäki, V. Lyssenko, S. Männistö, A. Marette, T.C. Matise, C.A. McKenzie, B. McKnight, A.W. Musk, S. Möhlenkamp, A.D. Morris, M. Nelis, C. Ohlsson, A.J. Oldehinkel, K.K. Ong, L.J. Palmer, B.W. Penninx, A. Peters, P.P. Pramstaller, O.T. Raitakari, T. Rankinen, D.C. Rao, T.K. Rice, P.M. Ridker, M.D. Ritchie, I. Rudan, V. Salomaa, N.J. Samani, J. Saramies, M.A. Sarzynski, P.E. Schwarz, A.R. Shuldiner, J.A. Staessen, V. Steinthorsdottir, R.P. Stolk, K. Strauch, A. Tönjes, A. Tremblay, E. Tremoli, M.C. Vohl, U. Völker, P. Vollenweider, J.F. Wilson, J.C. Witteman, L.S. Adair, M. Bochud, B.O. Boehm, S.R. Bornstein, C. Bouchard, S. Cauchi, M.J. Caulfield, J.C. Chambers, D.I. Chasman, R.S. Cooper, G. Dedoussis, L. Ferrucci, P. Froguel, H.J. Grabe, A. Hamsten, J. Hui, K. Hveem, K.H. Jöckel, M. Kivimaki, D. Kuh, M. Laakso, Y. Liu, W. März, P.B. Munroe, I. Njølstad, B.A. Oostra, C.N. Palmer, N.L. Pedersen, M. Perola, L. Pérusse, U. Peters, C. Power, T. Quertermous, R. Rauramaa, F. Rivadeneira, T.E. Saaristo, D. Saleheen, J. Sinisalo, P.E. Slagboom, H. Snieder, T.D. Spector, K. Stefansson, M. Stumvoll, J. Tuomilehto, A.G. Uitterlinden, M. Uusitupa, P. van der Harst, G. Veronesi, M. Walker, N.J. Wareham, H. Watkins, H.E. Wichmann, G.R. Abecasis, T.L. Assimes, S.I. Berndt, M. Boehnke, I.B. Borecki, P. Deloukas, L. Franke, T.M. Frayling, L.C. Groop, D.J. Hunter, R.C. Kaplan, J.R. O'Connell, L. Qi, D. Schlessinger, D.P. Strachan, U. Thorsteinsdottir, C.M. van Duijn, C.J. Willer, P.M. Visscher, J. Yang, J.N. Hirschhorn, M.C. Zillikens, M.I. McCarthy, E.K. Speliotes, K.E. North, C.S. Fox, I. Barroso, P.W. Franks, E. Ingelsson, I.M. Heid, R.J. Loos, L.A. Cupples, A.P. Morris, C.M. Lindgren, K.L. Mohlke, A. Consortium, C.D. Consortium, C. Consortium, G. Consortium, G. Consortium, GLGC, ICBP, I.E. Consortium, L.C. Study, M. Investigators, M. Consortium, P. Consortium, R. Consortium, New genetic loci link adipose and insulin biology to body fat distribution. Nature 2015, 518, 187–196. [Google Scholar]
- V.V. Thaker, GENETIC AND EPIGENETIC CAUSES OF OBESITY. Adolesc Med State Art Rev 2017, 28, 379–405.
- H. Huvenne, B. Dubern, K. Clément, C. Poitou, Rare Genetic Forms of Obesity: Clinical Approach and Current Treatments in 2016. Obes Facts 2016, 9, 158–73. [CrossRef] [PubMed]
- G. Koochakpour, Z. Esfandiar, F. Hosseini-Esfahani, P. Mirmiran, M.S. Daneshpour, B. Sedaghati-Khayat, F. Azizi, Evaluating the interaction of common FTO genetic variants, added sugar, and trans-fatty acid intakes in altering obesity phenotypes. Nutr Metab Cardiovasc Dis 2019, 29, 474–480. [CrossRef]
- N. Gupta, V. Jain, Prader Willi Syndrome - A Common Epigenetic Cause of Syndromic Obesity. Indian J Pediatr 2017, 84, 809–810. [CrossRef]
- H. Cena, L. Chiovato, R.E. Nappi, Obesity, Polycystic Ovary Syndrome, and Infertility: A New Avenue for GLP-1 Receptor Agonists. J Clin Endocrinol Metab 2020, 105.
- C.S. D'Angelo, C.P. Koiffmann, Copy number variants in obesity-related syndromes: review and perspectives on novel molecular approaches. J Obes 2012, 2012, 845480.
- M. Rosenbaum, R. Knight, R.L. Leibel, The gut microbiota in human energy homeostasis and obesity. Trends Endocrinol Metab 2015, 26, 493–501. [CrossRef]
- Wernstedt Asterholm, C. Tao, T.S. Morley, Q.A. Wang, F. Delgado-Lopez, Z.V. Wang, P.E. Scherer, Adipocyte inflammation is essential for healthy adipose tissue expansion and remodeling. Cell Metab 2014, 20, 103–18. [CrossRef] [PubMed]
- Y. Arita, S. Kihara, N. Ouchi, M. Takahashi, K. Maeda, J. Miyagawa, K. Hotta, I. Shimomura, T. Nakamura, K. Miyaoka, H. Kuriyama, M. Nishida, S. Yamashita, K. Okubo, K. Matsubara, M. Muraguchi, Y. Ohmoto, T. Funahashi, Y. Matsuzawa, Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun 1999, 257, 79–83.
- M. Pigeyre, F.T. Yazdi, Y. Kaur, D. Meyre, Recent progress in genetics, epigenetics and metagenomics unveils the pathophysiology of human obesity. Clin Sci (Lond) 2016, 130, 943–86. [CrossRef] [PubMed]
- A.A. van der Klaauw, I.S. Farooqi, The hunger genes: pathways to obesity. Cell 2015, 161, 119–132. [CrossRef]
- P.S. MacLean, J.A. Higgins, E.D. Giles, V.D. Sherk, M.R. Jackman, The role for adipose tissue in weight regain after weight loss, Obes Rev 16 Suppl 1 (2015) 45-54.
- C.N. Ochner, A.G. Tsai, R.F. Kushner, T.A. Wadden, Treating obesity seriously: when recommendations for lifestyle change confront biological adaptations. Lancet Diabetes Endocrinol 2015, 3, 232–4. [CrossRef]
- T. Tchkonia, T. Thomou, Y. Zhu, I. Karagiannides, C. Pothoulakis, M.D. Jensen, J.L. Kirkland, Mechanisms and metabolic implications of regional differences among fat depots. Cell Metab 2013, 17, 644–656. [CrossRef]
- S.B. Heymsfield, H.H. Hu, W. Shen, O. Carmichael, Emerging Technologies and their Applications in Lipid Compartment Measurement. Trends Endocrinol Metab 2015, 26, 688–698. [CrossRef]
- A.J. McCullough, The clinical features, diagnosis and natural history of nonalcoholic fatty liver disease. Clin Liver Dis 2004, 8, 521–33. [CrossRef]
- J. Kaur, A comprehensive review on metabolic syndrome, Cardiol Res Pract 2014 (2014) 943162.
- J.E. Hall, A.A. da Silva, J.M. do Carmo, J. Dubinion, S. Hamza, S. Munusamy, G. Smith, D.E. Stec, Obesity-induced hypertension: role of sympathetic nervous system, leptin, and melanocortins. J Biol Chem 2010, 285, 17271–6. [CrossRef]
- M. Ortiz-Martínez, M. González-González, A.J. Martagón, V. Hlavinka, R.C. Willson, M. Rito-Palomares, Recent Developments in Biomarkers for Diagnosis and Screening of Type 2 Diabetes Mellitus. Curr Diab Rep 2022, 22, 95–115. [CrossRef]
- M. Zhang, T. Hu, S. Zhang, L. Zhou, Associations of Different Adipose Tissue Depots with Insulin Resistance: A Systematic Review and Meta-analysis of Observational Studies, Sci Rep 5 (2015) 18495.
- S.H. Lee, S.Y. Park, C.S. Choi, Insulin Resistance: From Mechanisms to Therapeutic Strategies. Diabetes Metab J 2022, 46, 15–37. [CrossRef]
- Y.T. Wondmkun, Obesity, Insulin Resistance, and Type 2 Diabetes: Associations and Therapeutic Implications, Diabetes Metab Syndr Obes 13 (2020) 3611-3616.
- M.C. Petersen, G.I. Shulman, Mechanisms of Insulin Action and Insulin Resistance. Physiol Rev 2018, 98, 2133–2223. [CrossRef] [PubMed]
- T. Ota, Obesity-induced inflammation and insulin resistance, Front Endocrinol (Lausanne) 5 (2014) 204.
- U. Ozcan, E. Yilmaz, L. Ozcan, M. Furuhashi, E. Vaillancourt, R.O. Smith, C.Z. Görgün, G.S. Hotamisligil, Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science 2006, 313, 1137–40. [CrossRef] [PubMed]
- M. Krssak, A. Brehm, E. Bernroider, C. Anderwald, P. Nowotny, C. Dalla Man, C. Cobelli, G.W. Cline, G.I. Shulman, W. Waldhäusl, M. Roden, Alterations in postprandial hepatic glycogen metabolism in type 2 diabetes. Diabetes 2004, 53, 3048–56.
- R.A. DeFronzo, D. Tripathy, Skeletal muscle insulin resistance is the primary defect in type 2 diabetes, Diabetes Care 32 Suppl 2(Suppl 2) (2009) S157-63.
- S.E. Kahn, R.L. Hull, K.M. Utzschneider, Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006, 444, 840–6. [CrossRef] [PubMed]
- G.F. Lewis, A.C. Carpentier, S. Pereira, M. Hahn, A. Giacca, Direct and indirect control of hepatic glucose production by insulin. Cell Metab 2021, 33, 709–720. [CrossRef]
- A.E. Butler, J. Janson, S. Bonner-Weir, R. Ritzel, R.A. Rizza, P.C. Butler, Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes 2003, 52, 102–10. [CrossRef] [PubMed]
- G. Vazquez, S. Duval, D.R. Jacobs, K. Silventoinen, Comparison of body mass index, waist circumference, and waist/hip ratio in predicting incident diabetes: a meta-analysis, Epidemiol Rev 29 (2007) 115-28.
- K.S.R. Thapa SD, Gautam S, Gyawali D., Dyslipidemia in Type 2 Diabetes mellitus, Journal of Pathology of Nepal, 2017, pp. 1149 - 1154.
- D.C. Chan, H.P. Barrett, G.F. Watts, Dyslipidemia in visceral obesity: mechanisms, implications, and therapy. Am J Cardiovasc Drugs 2004, 4, 227–46.
- Beers, M.J. Haas, N.C. Wong, A.D. Mooradian, Inhibition of apolipoprotein AI gene expression by tumor necrosis factor alpha: roles for MEK/ERK and JNK signaling. Biochemistry 2006, 45, 2408–13. [CrossRef] [PubMed]
- A.D. Mooradian, M.J. Haas, K.R. Wehmeier, N.C. Wong, Obesity-related changes in high-density lipoprotein metabolism. Obesity (Silver Spring) 2008, 16, 1152–60. [CrossRef]
- M.A. Alpert, J. Omran, B.P. Bostick, Effects of Obesity on Cardiovascular Hemodynamics, Cardiac Morphology, and Ventricular Function. Curr Obes Rep 2016, 5, 424–434. [CrossRef] [PubMed]
- Grillo, L. Salvi, P. Coruzzi, P. Salvi, G. Parati, Sodium Intake and Hypertension, Nutrients 2019, 11, 2019; 11.
- M.E. Hall, J.M. do Carmo, A.A. da Silva, L.A. Juncos, Z. Wang, J.E. Hall, Obesity, hypertension, and chronic kidney disease, Int J Nephrol Renovasc Dis 7 (2014) 75-88.
- D. Susic, J. Varagic, Obesity: A Perspective from Hypertension. Med Clin North Am 2017, 101, 139–157.
- Virdis, S. Taddei, Endothelial Dysfunction in Resistance Arteries of Hypertensive Humans: Old and New Conspirators. J Cardiovasc Pharmacol 2016, 67, 451–7. [Google Scholar] [CrossRef] [PubMed]
- Y. Lu, K. Hajifathalian, M. Ezzati, M. Woodward, E.B. Rimm, G. Danaei, G.B.o.M.R.F.f.C.D.C.B.M. Effects), Metabolic mediators of the effects of body-mass index, overweight, and obesity on coronary heart disease and stroke: a pooled analysis of 97 prospective cohorts with 1·8 million participants. Lancet 2014, 383, 970–83.
- T.M. Barber, S. Franks, Obesity and polycystic ovary syndrome. Clin Endocrinol (Oxf) 2021, 95, 531–541. [CrossRef]
- T.M. Barber, M.I. McCarthy, J.A. Wass, S. Franks, Obesity and polycystic ovary syndrome. Clin Endocrinol (Oxf) 2006, 65, 137–45.
- Purwar, S. Nagpure, Insulin Resistance in Polycystic Ovarian Syndrome. Cureus 2022, 14, e30351. [Google Scholar]
- W. Kositanurit, D. Muntham, S. Udomsawaengsup, N. Chirakalwasan, Prevalence and associated factors of obstructive sleep apnea in morbidly obese patients undergoing bariatric surgery. Sleep Breath 2018, 22, 251–256. [CrossRef]
- A.B. Newman, G. Foster, R. Givelber, F.J. Nieto, S. Redline, T. Young, Progression and regression of sleep-disordered breathing with changes in weight: the Sleep Heart Health Study. Arch Intern Med 2005, 165, 2408–13. [CrossRef]
- G.S. Hamilton, S.A. Joosten, Obstructive sleep apnoea and obesity. Aust Fam Physician 2017, 46, 460–463.
- E.E. Calle, C. Rodriguez, K. Walker-Thurmond, M.J. Thun, Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med 2003, 348, 1625–38. [CrossRef] [PubMed]
- J. Park, D.M. Euhus, P.E. Scherer, Paracrine and endocrine effects of adipose tissue on cancer development and progression. Endocr Rev 2011, 32, 550–70. [CrossRef] [PubMed]
- K.M. Gadde, J.W. Apolzan, H.R. Berthoud, Pharmacotherapy for Patients with Obesity. Clin Chem 2018, 64, 118–129. [CrossRef] [PubMed]
- M.D. Jensen, D.H. Ryan, C.M. Apovian, J.D. Ard, A.G. Comuzzie, K.A. Donato, F.B. Hu, V.S. Hubbard, J.M. Jakicic, R.F. Kushner, C.M. Loria, B.E. Millen, C.A. Nonas, F.X. Pi-Sunyer, J. Stevens, V.J. Stevens, T.A. Wadden, B.M. Wolfe, S.Z. Yanovski, A.C.o.C.A.H.A.T.F.o.P. Guidelines, O. Society, 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society, J Am Coll Cardiol 63(25 Pt B) (2014) 2985-3023.
- Y. Preiss Contreras, X. Ramos Salas, C. Ávila Oliver, M.A. Saquimux Contreras, R. Muñoz Claro, C. Canales Ferrada, C.C.p.e.E.d.l. Obesidad, Obesity in adults: Clinical practice guideline adapted for Chile. Medwave 2022, 22, e2649.
- K.M. Gadde, C.K. Martin, H.R. Berthoud, S.B. Heymsfield, Obesity: Pathophysiology and Management. J Am Coll Cardiol 2018, 71, 69–84.
- R.R. Wing, D.F. Tate, A.A. Gorin, H.A. Raynor, J.L. Fava, A self-regulation program for maintenance of weight loss. N Engl J Med 2006, 355, 1563–71. [CrossRef]
- C.L. Rock, S.W. Flatt, N.E. Sherwood, N. Karanja, B. Pakiz, C.A. Thomson, Effect of a free prepared meal and incentivized weight loss program on weight loss and weight loss maintenance in obese and overweight women: a randomized controlled trial. JAMA 2010, 304, 1803–10. [CrossRef]
- E.Y. Lee, K.H. Yoon, Epidemic obesity in children and adolescents: risk factors and prevention. Front Med 2018, 12, 658–666. [CrossRef]
- Nguyen, J. Clements, Obesity management among patients with type 2 diabetes and prediabetes: a focus on lifestyle modifications and evidence of antiobesity medications. Expert Rev Endocrinol Metab 2017, 12, 303–313. [Google Scholar] [CrossRef]
- M.P. Pedersen SD, Wharton S., Canadian Adult Obesity Clinical Practice Guidelines: Pharmacotherapy in Obesity Management, Obesity Canada, 2020.
- E.S. Leblanc, E. O'Connor, E.P. Whitlock, C.D. Patnode, T. Kapka, Effectiveness of primary care-relevant treatments for obesity in adults: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med 2011, 155, 434–47. [CrossRef]
- Rucker, R. Padwal, S.K. Li, C. Curioni, D.C. Lau, Long term pharmacotherapy for obesity and overweight: updated meta-analysis. BMJ 2007, 335, 1194–9. [CrossRef]
- Secher, J. Jelsing, A.F. Baquero, J. Hecksher-Sørensen, M.A. Cowley, L.S. Dalbøge, G. Hansen, K.L. Grove, C. Pyke, K. Raun, L. Schäffer, M. Tang-Christensen, S. Verma, B.M. Witgen, N. Vrang, L. Bjerre Knudsen, The arcuate nucleus mediates GLP-1 receptor agonist liraglutide-dependent weight loss. J Clin Invest 2014, 124, 4473–88. [Google Scholar]
- S. Sisley, R. Gutierrez-Aguilar, M. Scott, D.A. D'Alessio, D.A. Sandoval, R.J. Seeley, Neuronal GLP1R mediates liraglutide's anorectic but not glucose-lowering effect. J Clin Invest 2014, 124, 2456–63. [CrossRef] [PubMed]
- ISP, Ficha Registro ISP SAXENDA, 2022.
- N.N. Canada, SAXENDA® liraglutide, 2017.
- D.B. Allison, K.M. Gadde, W.T. Garvey, C.A. Peterson, M.L. Schwiers, T. Najarian, P.Y. Tam, B. Troupin, W.W. Day, Controlled-release phentermine/topiramate in severely obese adults: a randomized controlled trial (EQUIP). Obesity (Silver Spring) 2012, 20, 330–42. [CrossRef]
- R.B. Rothman, E.J. Hendricks, Phentermine cardiovascular safety, Am J Emerg Med 2009, 27, 1010–3. [CrossRef]
- K.E. Steidl, W. Darko, L.A. Probst, J.A. Noviasky, S. Nasser, Rhabdomyolysis associated with phentermine. Am J Health Syst Pharm 2010, 67, 1929–32.
- K.M. Gadde, G.L. Xiong, Bupropion for weight reduction. Expert Rev Neurother 2007, 7, 17–24. [CrossRef] [PubMed]
- S.R. Smith, K. Fujioka, A.K. Gupta, S.K. Billes, C. Burns, D. Kim, E. Dunayevich, F.L. Greenway, Combination therapy with naltrexone and bupropion for obesity reduces total and visceral adiposity. Diabetes Obes Metab 2013, 15, 863–6. [CrossRef] [PubMed]
- R.B. Rothman, M.H. Baumann, Appetite suppressants, cardiac valve disease and combination pharmacotherapy. Am J Ther 2009, 16, 354–64. [CrossRef] [PubMed]
- M.A. Scudeler, S. Morreale, L. Doretto-Silva, G. Petri, J.F.R.D. Santos, C. Nassis, O.M.T. Correa, J.M. Veridiano, Effects of topiramate, bupropion and naltrexone isolated or combined on subcutaneous adipose tissue in obese rats, Einstein (Sao Paulo) 20 (2022) eAO5587.
- F. Food and Drug Administration, Advisory Committee Meeting for Phentermine/Topiramate (Qnexa), Division of Metabolism and Endocrinology Products (DMEP), Office of Drug Evaluation II, Center for Drug Evaluation and Research, Silver Spring, MD, 2010.
- H.R. Arias, A. Santamaría, S.F. Ali, Pharmacological and neurotoxicological actions mediated by bupropion and diethylpropion, Int Rev Neurobiol 88 (2009) 223-55.
- H.S. Jones, Diethylpropion dependence. Med J Aust 1968, 1, 267. [CrossRef]
- B. So, H.J. Kim, J. Kim, W. Song, Exercise-induced myokines in health and metabolic diseases. Integr Med Res 2014, 3, 172–179. [CrossRef] [PubMed]
- B.K. Pedersen, M.A. Febbraio, Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol 2012, 8, 457–65. [CrossRef] [PubMed]
- P.E. Scherer, Adipose tissue: from lipid storage compartment to endocrine organ. Diabetes 2006, 55, 1537–45.
- B.K. Pedersen, Muscles and their myokines, J Exp Biol 214(Pt 2) (2011) 337-46.
- L.G. Leal, M.A. Lopes, M.L. Batista, Physical Exercise-Induced Myokines and Muscle-Adipose Tissue Crosstalk: A Review of Current Knowledge and the Implications for Health and Metabolic Diseases, Front Physiol 9 (2018) 1307.
- A.M. Petersen, B.K. Pedersen, The anti-inflammatory effect of exercise. J Appl Physiol (1985) 2005, 98, 1154–62. [CrossRef]
- S.W. Ng, B.M. Popkin, Time use and physical activity: a shift away from movement across the globe. Obes Rev 2012, 13, 659–80. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




