Submitted:
17 July 2023
Posted:
19 July 2023
You are already at the latest version
Abstract
Keywords:
Introduction
1. History of Biguanide: From the First Animal Experiments to the Use of Synthetic Compounds for Oral Use in Overweight Type 2 Diabetics
United Kingdom Prospective Diabetes Study (UKPDS) and Metformin
UKPDS: Some Details About the Metformin Response (46,47)
Pharmakocinetic and Pharmacodynamic of Metformin
Short- and Long-Term Side Effects and Adherence to Metformin Therapy
Mechanisms of Action of Metformin Therapy: Direct or Indirect?
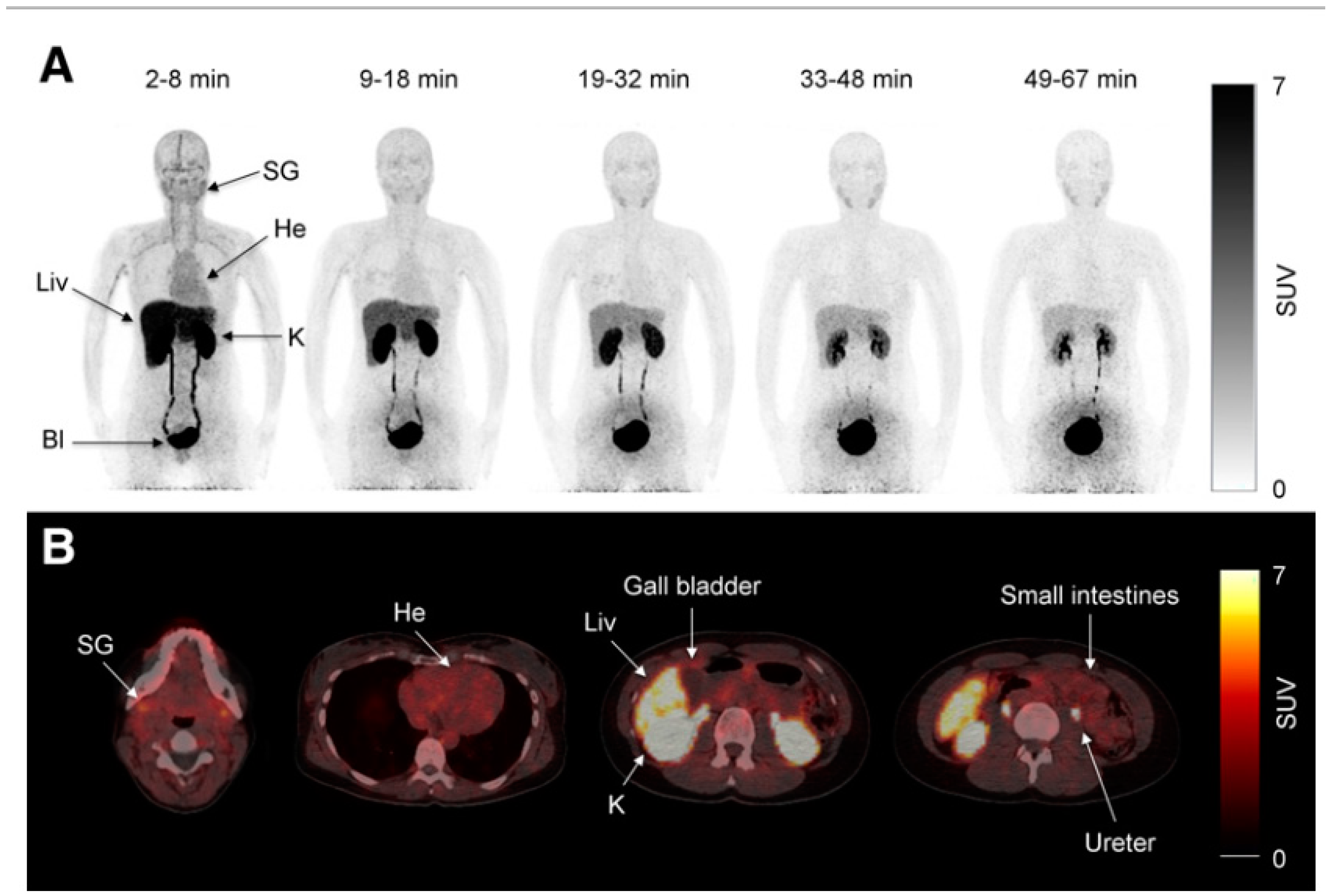
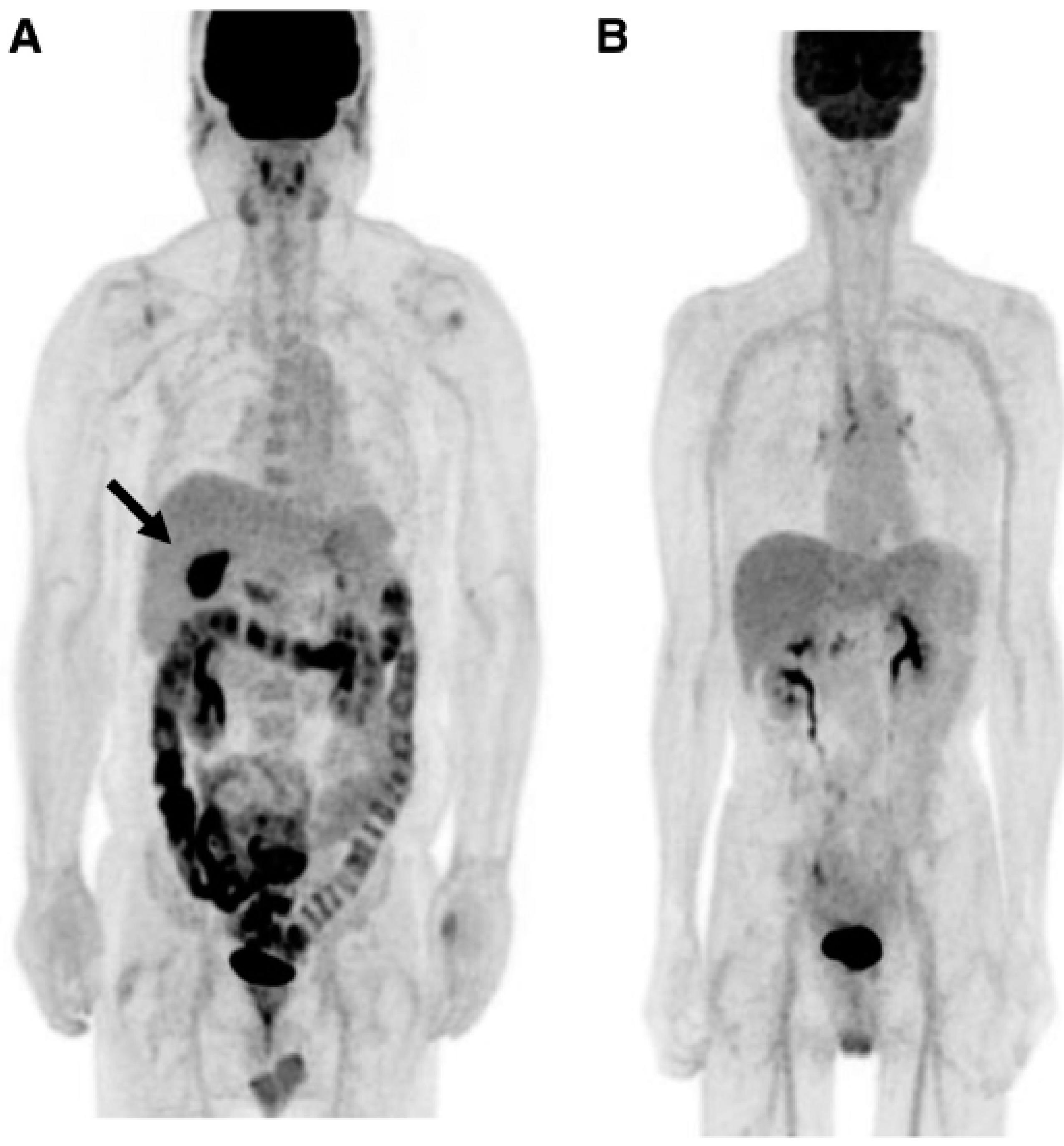
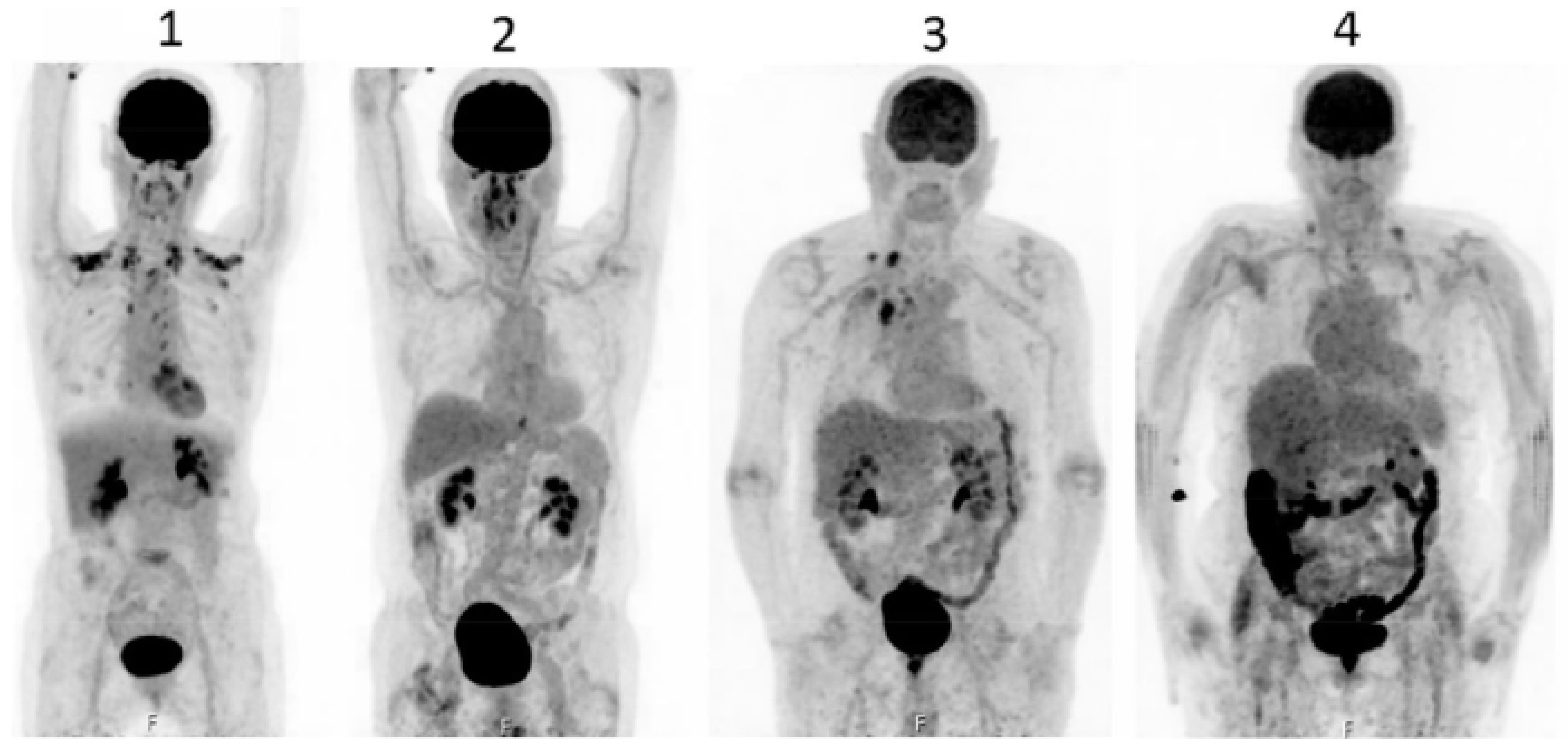
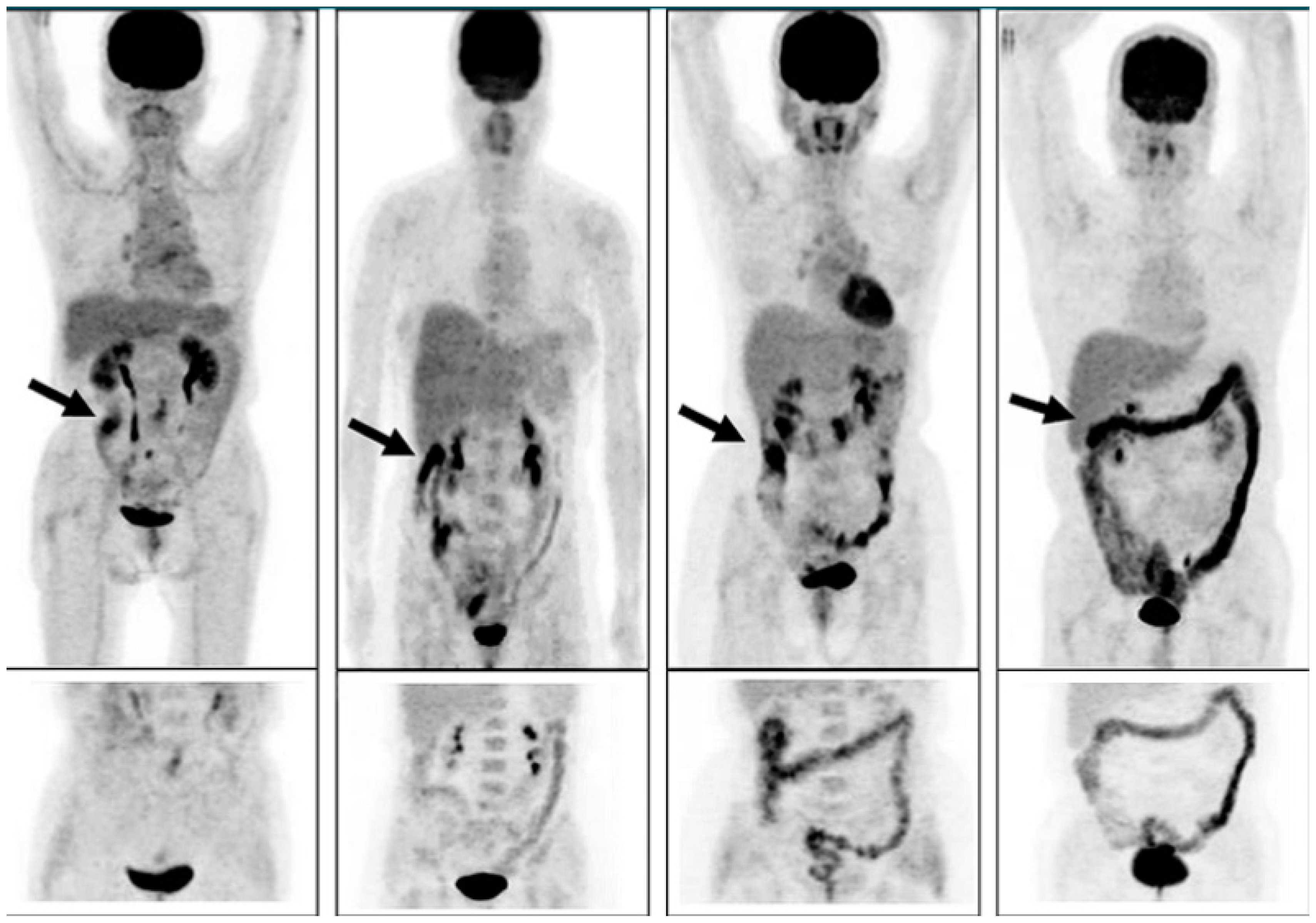
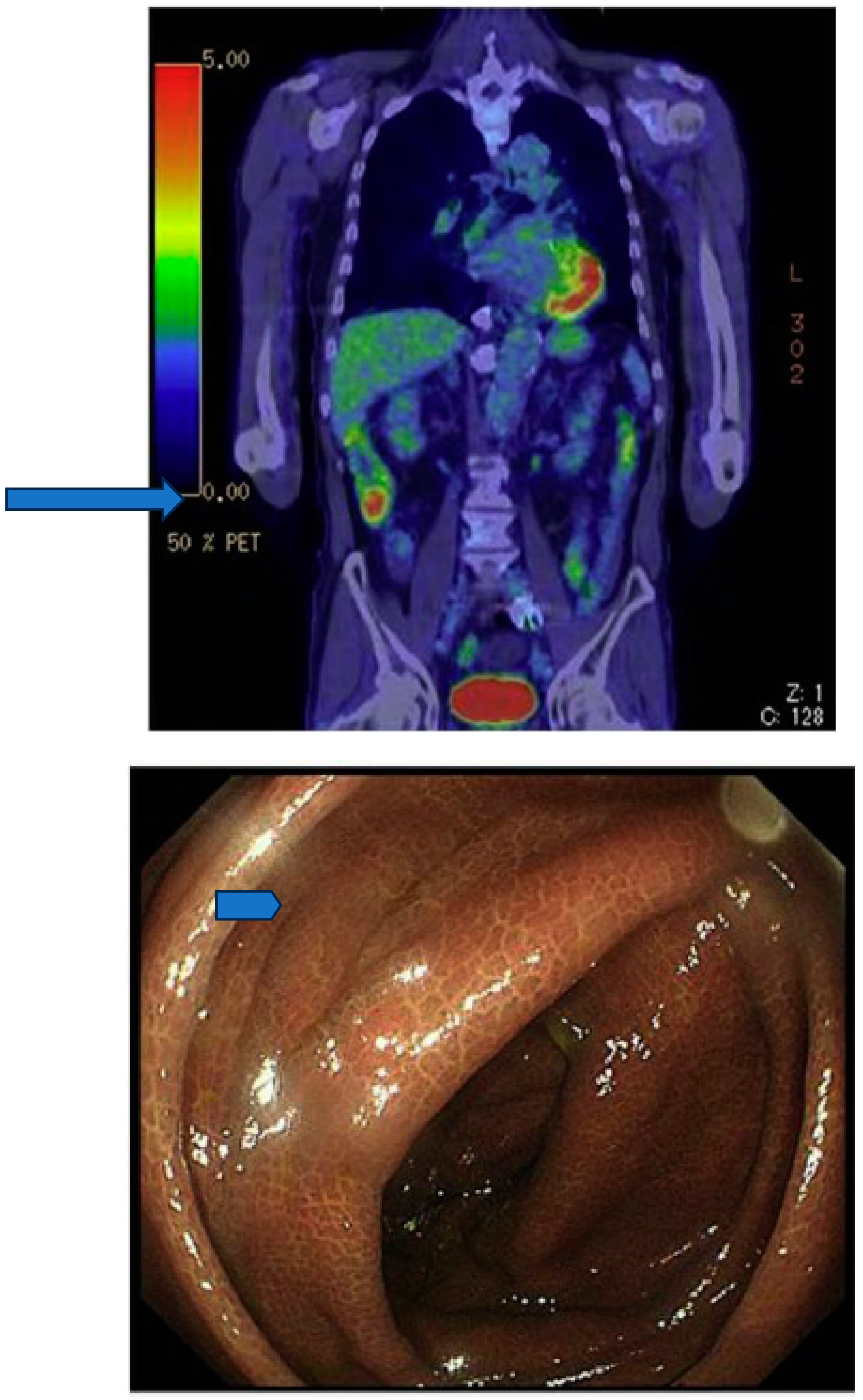
Concluions
References
- 1. Foretz M,Viollet B.:Le nouvelles promesses de la metformine.Vers une meilleure comprehension de ses mechanismes d‘action.Medicine/sciences 2014;30 (1):82-92.DOI10.1051/medsci/20143001010. [CrossRef]
- Sterne J.:Du nouveau dans les antidiabetiques.La NNdimethylamino guanyl guanidine(NNDG).Maroc Med 1957;36:1295-1296.
- Bailey CJ.The origin of type 2 diabetes medications.Br J Diabetes.2022;22:112-120.
- Staub H:Übersichten.40 Jahre insulin und intermediärer Stoffwechsel.Kli Wschr 1965;42(2): 61-69.
- Minkowski O,Verhandlungen des I.internationalen Physiologenkongresses.Basel 1889.Zentbl.Physiol.1889;312-313.
- Falkenberg M .Verhandlungen des X.Congresses für Innere Medizin.Wiesbaden 1891 S.502.
- Hirsch R.:Glykosurie nach Schilddrüsenextirpation by Hunden.Z.exp.Path.u.Therap.1906;3:393-400.
- Fühner H Über den Angriffsort der peripheren Guanidinwirkung.Zugleich eine Erwiederung.Archiv f.experiment.Pathol.u.Pharmakol.1909;65:402-427.
- Koch WF.On the occurrence of methyl Guanidine in the urine of parathyroidectomized animals.J Biol Chem 1912;12(3):313-315. [CrossRef]
- Koch WF.Toxic bases in the urine of parathyroidectomized dogs. J Biol Chem 1913;15:43-64. [CrossRef]
- Underhill FP,Blatherwick NR.Studies in carbohydrate metabolism.VI.The influence of Thyreoparathyroidectomy upon the sugar content of the blood and the gycogen content oft he liver.J Biol Chem 1914;118 (1):87-90.
- Underhill FP,Blatherwick NR.Studies in Carbohydrate Metabolism.VII.The influence of subcutaneous injections of dextrose and of calcium lactate upon the blood sugar content and upon tetany after thyreoparathyroidectomy.J Biol Chem 1914;119(1):119-126. [CrossRef]
- Underhill FP,Saiki T.The influence of complete thyroidectomy and thyroid feeding upon certain phases of intermediary metabolism. BiolChem.1908;5:225—241. [CrossRef]
- Burns D,Sharpe JS.THE PARATHYROIDS-TETANIA PARATHYREOPRIVA,IST NATURE,CAUSE,AND RELATIONS TO IDIOPATHIC TETANY.PART V: GUANIDIN AND METHYL-GUANIDINE IN THE BLOOD AND URINE IN TETANIA PARATHYREOPRIVA AND IN THE URINE IN IDIOPATHIC TETANY.Quart.J Exp Physiol 1916;10:345-354. [CrossRef]
- Watanabe CK.Studies in the metabolic changes induced by administration of guanidine bases.I.Influence of injected guanidine hydrochloride upon blood sugar content.J Biol Chem 1918;33:253-265.
- Watanabe CK.:Studies in the metabolic changes induced by administration of guanidine bases.II.The influence of guanidine upon urinary ammonia and acid excretion.J Biol Chem 1918;34(1)51-63.
- Watanabe CK.Studies in the metabolic changes induced by administration of guanidine bases.IV.Th influence of the administration of calcium upon blood sugar content in rabbits with guanidine hypoglycemia.J Biol Chem 1918;34(19:73-76.
- Watanabe CK.:Studies in the metabolic changes induced by the administration of guanidine bases.V.The change of phosphate and calcium content in serum in guanidine tetany and the relation between the calcium content and sugar in the blood.J Biol Chem 1918; 34(1):531-546. [CrossRef]
- Frank E,Northmann M,Wagner A.:Über Synthetisch Dargestellte Körper mit insulinartiger Wirkung auf den Normalen und diabetischen Organismus.Kli Wochenschr. 1926;45:2100-2107.
- Banting FC,Best CH.The internal secretion oft he pancreas.J Lab clin.Med.1922;7(5):251-266.
- Minkowski O.:Synthetische Insulinähnlich Wirkende Substanzen.Bemerkungen zu den vorstehenden Mitteilungen von Frank,Nothmann und Wagner.Klin Wochenschr.1926;45:2107.
- Calvert EGB.:Observations on the treatment of Diabetes By Synthalin.Lancet 1927;5430(2):649-651.DOI:10.1016/S0140-6736(01)31246-1. [CrossRef]
- Anonimus.:Synthalin in the treatment of Diabetes.Preliminary reports to the medical research council.Lancet 1927;5427 (2)517-521.DOI:10.1016/S0140-6736(01)31155-8. [CrossRef]
- Bodo R Marks A .:The relation of SYNTHALIN TO CARBOHYDRATE METABOLISM.J Physiol 1928;65:83-99. [CrossRef]
- Ringer AI,Bilon S,Harris MM,Landy A.:SYNTHALIN.its use in the treatment of Diabetes.Arch Int Med.1928;41(4):453-471.
- Staub H,Küng O.:Zum SANTHALINMECHANISMUS.Klin Woch.1928;7(29):1365-1366.
- Frank E,Nothmann M,Wagner A.:Über die Experimentelle und Klinische Wirkung des Dodekamethylendiguanids (Synthalin B).Klin Woch.1928;42:1996-2000. [CrossRef]
- Bischoff F,Sahyun M,Long L.:GUANIDINE STRUCTURE AND HYPOGLYCEMIA.J Biol Chem 1929;81:325-349.
- Anonymus.Insulin and Synthalin.Nature 1928;3039(121):151-153.
- Bloom A,Richards JG.:PHENFORMIN AS ADJUVANT ORAL THERAPY IN DIABETES.Br Med J 1961; 1796-1799. [CrossRef]
- Goldner M.:Oral Hypoglycemic Agents Past and Present.Archiv Int Med.1958;102:830-840.
- Williams RH,Tanner DC,Odell WD.Hypoglycemic Actions of Phenethyl-Amyl-,and Isoamyl-Diguanide.Diabetes 1958;7(2):87-92. [CrossRef]
- Beaser S.:THERAPY OF DIABETES MELLITUS WITH COMBINATIONS OF DRUGS GIVEN ORALLY.N Engl J Med 1958;259 (25):1207-1210. [CrossRef]
- Clarke BF,Duncan UP:Comparison of chlorpropamide and metformin tretment on weight and blood glucose response and uncontrolled obese diabetics.Lancet 1968;1:123-126.
- Beaser SB.:Oral Treatment of Diabetes Mellitus.JAMA 1964;187(12):887-893. [CrossRef]
- DeFronzo R,Goodmann A.:Efficacy of metformin in patients with non-insulin-dependent diabetes mellitus.NEngl J Med 1995;333(9):541-549. [CrossRef]
- Stumvoll M,Nurjahan N,Perriello G,Dailey G,Gerich JE.:Metabolic effects of metformin in non-insulin-dependent diabetes Mellitus.N Engl J Med 1995;333:550-554. [CrossRef]
- Guriguian J,Green L,Misbin RJ,Stadel B,Fleming GA.:Efficacy of Metformn in non-insulin-dependent diabetes mellitus.N Engl J Med 1996;334(4):269.
- Misbin RI,Green L,Stadel BV,Gueriguian JL,Gubbi A,Fleming GA.N Engl J Med 1998;338:265-266.
- Campbell RK,White JR,Saulie BA.:Metformin: A new Oral Biguanide.CLINICAL THERAPEUTICS 1996;18(3):360-370. [CrossRef]
- Ungar G,Freedman L,Shapiro S.:Pharmacological Studies of a new Oral Hypoglicemic Drug.Proc.Soc.Exp.Biol.Med 1957;100:190-192. [CrossRef]
- Dandona P,Fonseca V,Mier A,Beckett A.:Diarrhea and Metformin in Diabetic Clinic.Diabetes Care 1983;6(5):472-474. [CrossRef]
- Bailey CJ,Turner RC.:Metformin.N Engl J Med 1996;334(9):574-579.
- Inzucchi SE,Lipska KJ,Mayo H,Bailey CJ,McGuire DK.Metformin in patients with type 2 diabetes and kidney disease: a systematic review. JAMA 2014;312:2668-2675. [CrossRef]
- .46.UKPDS (33).Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33).Lancet 1998;352:837-853. [CrossRef]
- UKPDS(34).Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes(UKPDS 34) Lancet 1998;352:854-865. [CrossRef]
- Beaser SB.The Clinical Characteristics of Early Diabetes Mellitus.New Engl J Med 1948;239 (21):765-769. [CrossRef]
- Keys A,Menotti A,Karvonen MJ,Arvanis C,Blackburn H,Buzina R,Djorjevic BS,Dontas AS,Fidanza F,Keys MH,Kromhout D,Nedeljkovic S,Punsar S,Seccareccia F,Thoshima H.:The diet and 15-year death rate in the seven countries study.Am J Epidemiol.1986;124(6):903-915. [CrossRef]
- Dawber TR,Moore E,Mann GV:II.Corornary heart Disease in the Framingham study.Am J Publ Health.1957;47:4-24. [CrossRef]
- Huijbregets P,Feskens E,Raesanen L,Fidanza F,Nissinen A, Menotti A, Kromhout D.:Dietary pattern and 20 year Mortality in elderly men in Finland,Italy,and the Nederlands:longitudinal cohort study.BMJ 1997;315:13-17. [CrossRef]
- Sheps MC.:SHALL WE COUNT THE LIVING OR THE DEAD?N Engl J Med 1958;259 (25):1210-1214.
- Doyle JT,Dawber TR,Kannel WB,Heslin SA,Kahn HA:Cigarette smoking and coronary hearth disease.Combined experience oft he Albany and Framingham Studies.N Engl J Med 1962;266(16):796-801. [CrossRef]
- Doll R,Hill AB.:Lung cancer and other causes of death in relation to smoking.A second report on mortality of british doctors.BMJ 1956; 2(5001) :1070-1081. DOI: 10.1136/bmj.2.5001.1071. [CrossRef]
- Garcia MJ, McNamara PM, Gordon T, Kannell WB. Morbidity and mortality in diabetics in the Framingham population. Sixteen year follow-up study. Diabetes. 1974;23:105–111. [CrossRef]
- Freund K,Belanger AJ,DÀgostino RB,Kanner WB.:The health Risk of Smoking.The framingham Study:34 years of follow-up.AEP 1992;3(4):417-424.
- Doll R,Peto R.:Mortality in relation to smoking:20 years observations on male british doctors BMJ 1976;2:1525-1536. [CrossRef]
- Doll RD,Peto R,Hall E,Wheatley K,Gray R::Mortality in relation to consumption of alcohol:13 years observation of male british doctors.BMJ 1994;309:911-918. [CrossRef]
- Farchi G,Fidanza F,Menotti S,Menotti A.:Alcohol and Mortality in the italian rural cohorts of the seven countries study.Int J Epidemiol 1992;21(1):74-81. [CrossRef]
- Gronbaek M, Dies A,Sorensen TIA,Becker U,Borch-Johnsen K,Müller C,Schnohr P,Jensen G.:Influence of sex,age,body mass index,and smoking on alcohol intake and mortality.BMJ 1994,308:302-305. [CrossRef]
- Chyou P-H,Burchfiel CM,Yano K,Sharp DS,Rodriguez BL,Curb D,Nomura AMY.:Obesity,Alcohol Consumption,Smoking,and Mortality.AEP 1997;7(4):311-317. [CrossRef]
- Grunberg NE.Cigarette smoking and body weight:information may be hazardous to your health.Tabacco Control 1997;6:80. [CrossRef]
- Hermann LS.:Combination THERAPY WITH INSULIN AND METFORMIN.ENDOCRINE PRACTICE 1998;4(6):404-412. [CrossRef]
- Bailey CJ,Grant PJ,Evans M,Fine Olivarius N,Andreasen AH,Fowler PSB,Good CB,Turner RC,Holman R,Stratton I,Kerner W.:CORRESPONDENCE:The UK Prospective Diabetes Study. Lancet 1998;352:1932-1934.
- Ewart RM.:The UKPDS:what was the question?The Lancet 1999;353:1882. [CrossRef]
- Amiel SA.:Hypoglycaemia avoidance-technology and knowledge.The Lancet 1998;352:502-503. [CrossRef]
- Nathan DM.:Some answers,more controversy,from UKPDS.COMMENTARY.The Lancet 1998;352:832-833.
- Holman RR,Paul SK,Bethel MA,Matthews DR,Neil AW..10-Year Follow-up of intensive Glucose Control in Type 2 Diabetes.N Engl J Med 2008;359:1577-1589. [CrossRef]
- Sambol NC,Chiang J,O´Conner M,Liu CY,Lin ET,Goodman AM,Benet LZ,Karam JH.Pharmacokinetics and Pharmacodynamics of Metformin in Healthy Subjects and Patients with Non-Insulin-Dependent Diabetes Mellitus.J clin Pharmacol. 1996;36:1012-1021. [CrossRef]
- Pentikäinen PJ,Neuvonen PJ,Penttilä A.Pharmacokinetics of Metformin After intravenous and Oral Administration to Man.Eur J Clin Pharmacol 1979;16:185-202. [CrossRef]
- Gormensen LC,Sundelin EI,Jensen JB,Vendelbo MH,Jakobsen S,Munk OL,Christensen MMH,Broesen K,Froekiaer J, Jessen N.In vivo Imaging of Human C11-Metformin in Peripheral Organs:Dosimetry,Biodistribution,and Kinetic Analyses.J Nucl Med 2016;57(12):1920-1926.
- Garg,PK,Lokitz SJ,Nazih R,Garg S.Biodistribtution and Radiation Dosimetry of C11-Nicotine from Whole-Body PET Imaging in Humans.J Nucl Med 2017;58:473-478.
- Gierloeff T,Jakobsen S,Nahimi A,Munk OL, Bender D,Alstrup AKO,Vase KH,Hansen SB,Brooks DJ,Berghammer P.In Vivo Imaging of Human Acetylcholinesterase Density in Peripheral Organs using C11-Donezepezil:Dosimetry,Biodistribution,and Kinetic Analyses.J Nucl Med 2014;55:1818-1824. [CrossRef]
- Gierloeff T,Fedorova T,Knudsen K,Munk OL,Nahimi A,Jacobsen S,Danielsen EH,Terkelsen AJ,Hansen J,Pavese N,Brooks DJ,Borghammer P.Imaging acetylcholinesterase density in peripheral organs in Parkinson‘s diesease with C11-donepezil PET.BRAIN 2015;138:653-663. [CrossRef]
- Wisse E,De Zanger RB,Van Der Smissen CP,McCuskey RS.The Liver Sieve:Considerations Concerning the Structure and Function of Endothelial Fenestrae,the Sinusoidal Wall and the Space of Disse.Hepatology 1985;5(4):683-692. [CrossRef]
- Le Couteur DG,Fraser R,Hilmer S,Rivory LP,McLean AJ.The Hepatic Sinusoid in Aging and Cirrhosis.Effects on Hepatic Substrate Disposition and Drug Clearance.Clin Pharmacokinet.2005;44(2):187:200. [CrossRef]
- Chou C-H.Uptake and Dispersion of Metformin in the Isolated Perfused Rat Liver.J Pharm Pharmacol 2000;52:1011-1016. [CrossRef]
- Bonora E,Cigolini M,Bosello O,Zancanaro C,Capretti L,Zavaroni I,Coscelli CButturini U.:Lack of effect of intravenous metformin on plasma concentrations of glucose,insulin,C-peptide,glucagon and growth hormone in non-diabetic subjects.Curr.Med.Res.Opin.1984;9:47-51. [CrossRef]
- Sum CF,Webster JM,Johnson AB,Catalano C,Cooper BG,Taylor R.:The effect of intravenous Metformin on Glucose Metabolism During Hyperglicemia in Type 2 Diabetes.DIABETIC MEDICINE 1992;9:61-65. [CrossRef]
- Ong CR,Molyneaux LM,Binfotech MIC,Twigg SM, Yue DK.:Long-Term Efficacy of Metformin Therapy in Nonobese Individuals With Type 2 Diabetes.Diabetes Care.2006;29(11):2361-2364. [CrossRef]
- Goldberg RB,Orchard TJ,Crandall JP,Boyko EJ, Budoff M, Dabelea D,Gadde KM, Knowler WC, Lee CG, Nathan DM.:Diabetes prevention Program Research Group.Effects of long-term metformin and life-style interventions on cardiovascular events in the Diabetes Prevention Program and ist outcome study.Circulation 2022;145:1632-1641.DOI:10.1161/CIRCULATIONAHA.121.056756. [CrossRef]
- Vidon N,Chaussade S,Noel M,Franchisseur C,Huchet B,Bernier JJ..Metformin in the digestive tract.Diabetes Research and Clinical Practice.1988;4:223-229. [CrossRef]
- McCreight LJ,Bailey CJ,Pearson ER.:Metformin and the gastrointestinal tract.Diabetologia 2016;59:426-435. [CrossRef]
- Fatima M, Sadeeqa S,Nazir SUR.:Metformin and ist gastrointestinal problems:A review.Biomedical Research 2018;29(11):2285-2289.aw.
- Ghosal S,Ghosal S.:The Side Effects of Metformin-A Review.J Diabetes Metab.Disorders 2019.DOI:10.24966/DMD-201X/100030. [CrossRef]
- Caspary WF,Creutzfeldt W.:Inhibition of Bile salt absorption by blood sugar lowering biguanides.Diabetologia 1975;11:113-117. [CrossRef]
- Scarpello JHB,Hodgson E,Howlett HCS..Effect of Metformin on Bile salt Circulation and Intestinal Motility in Type 2 Diabetes Mellitus.Diabetic Medicine 1998;15:651-656.
- Sansome DJ,Xie C,Veedfald S, Horowitz M, Rayner CK,Wu T.:Mechanisms of glucose-lowering by metformin in type 2 diabetes:Role of bile acids.Diabetes Obes Metab.2019;.DOI:10.1111/dom.13869. [CrossRef]
- Metry M,Krug SA,Karra VK,Kane MA,Fink JC,Shu Y,Wang H,Polii JE.:Differential effects of metformin-mediated BSEP repression on pravastatin and bile acid pharmacokinetics in humans: A randomized controlled trial.Clin Transl Sci.2022;15:2468-2478. [CrossRef]
- Nakajima A,Ishizaki S, Matsuda K,Kurosu S,Taniguchi S,Gilberg PG,Mattsson JP,Hasumuma T,Camillen M.:Impact of elobixibat on serum and fecal bile acid levels and constipation symptoms in patients with chronic constipation.Journal of Gastroenterology and Hepatology 2022;37:883-890. [CrossRef]
- Spiller R.Inhibiting glucoe absorption to treat constipation.Lancet Gastroenterol Hepatol 2018.DOI:10.1016/S2468-1253 (18)30214-0. [CrossRef]
- Unger RH,Madison LL,Carter NW.:Tolbutamide-Phenformin in Ketoacidosis-Resistant Patients.JAMA 1960;174(17):2132-2136. [CrossRef]
- Bloom A,RicharsJG.Phenformin as adjuvant oral therapy in Diabetes.Br Med J 1961;1(5242): 1796-1799. [CrossRef]
- Cornfield J.:The university Group Diabetes Program.A Further Statistical Analysis of the Mortality Findings.JAMA 1971;217(12):1676-1687. [CrossRef]
- Palmer JP.Farewell to Phenformin for treating diabetes Mellitus.Ann Int Med 1975;83(4):567-568. [CrossRef]
- Fitzgerald E,Mathieu S,Ball A.:Metformin associated lactic acidosis.BMJ 2009;339:1254-1257.
- Blough B,Moreland A,Mora A.:Metformin-induced lactic acidosis with emphasis on the anoin gap.Proc(Bayl Univ Med Cent) 2015;28(1):31-33. [CrossRef]
- DeFronzo R,Fleming A,Chen K, Bicsak TA.:Metformin-associated lactic acidosis:current perspectives on causes and risk.Metabolism Clinical and Experimental 2016;65:20-29. [CrossRef]
- Gong O,Zhang P,Wang J,Ma J,An Y,Chen Y,Zhang B,Feng X,Li H,Chen X.:Da Qing Diabetes Prevention Study Group.Morbidity and Mortality after lifestyle intervention for people with impaired glucose tolerance:30-year results of the Da Oing Diabetes Prevention Outcome Study.Lancet Diabetes Endocrinol.2019;7:452-461.DOI:10.1016/S2213-8587(19)30093-2. [CrossRef]
- Faggian G,Cesaro A,Faggian R,Del Piano C,Vitagliano A,Del Piano D,Salzano M,Diglio A,Faggian A.Insufficienza renale acuta,acidosi lattica e metforrmina:due casi clinici e review della letteratura.Giornale Italiano di Nefrologia. 2023;3(3): ISSN 1724-5990.
- Cheng X,Liu MY-M,Li H, Zhang X,Lei F, Quin JJ,Chen Z,Deng KQ,Lin L,Chen M-MSong X, Xia M et al.Metformin is associated with higher incidence of acidosis,but not mortality,in individuals with COVID-19 and pre-existing type 2 diabetes.Cell Metabolism 2020;32:537-547.
- Vordoni A,Theofilis P,Vlachopanos G,Koukoulaki M,Kalaitzidis RG.Metfromin-associated lactic acidosis and acute kidney injury in the era of COVID-19.Frontiers in Bioscience Scholar 2021;13 (2):202-207.DOI:10.52586/S563. [CrossRef]
- Takayama K,Obata Y,Maruo Y,Yamaguchi H,Kosugi M,Irie Y,Hazama Y,Yasuda T.Metformin-associated Lactic Acidosis with Hypoglycemia during Covid-19 Pandemic.Intern Med 2022;61:2333-23337.DOI:10.2169/internalmedicine.9179-21. [CrossRef]
- Baley CJ,Gwilt M.Diabetes,Metformin and Clinical Course of COVID-19:Outcomes, Mechanisms and Suggestions on the Therapeutic Use of Metformin.Front Pharmacol 2022;13:78459.DOI:10.3369/iphar.2022.784459.
- Donelly LA,Morris AD,Pearson ER.:Adherence in patients transferred from immediate release metformin to a sustained release formulation:a population based study.Diabetes,Obesity and Metabolism.2009;11:338-342. [CrossRef]
- Rashid M,Warraich NY,Laique T,Shujaat K,Zawar S,Munir A.:Reasons of NON-Compliance to metformin among type 2 dibetics attending diabetic clinic in Lahore.Journal of Akhtar Saeed Medical and Dental College 2019;01(3):111-116.
- Christofides EA.:Practical Insight Into Improving Adherence to Metformin Therapy in Patients With Type 2 Diabetes.CLINICAL.DIABETES JOURNALS.ORG.2019;37 (3):234-241.DOI:10.2337/cd18-0063. [CrossRef]
- Walker EA,Gonzalez JS,Tripputi MT,Dagogo-Jack S,Matulik MJ,Montez MG,Tadros S,Edelstein SL.:Long-term metformin adherence in the Diabetes Prevention Program Outcomes Study.BMJ Open Diab Res Care 2020;8:e001537.DOI:10.1136/bmjdrc-2020-001537. [CrossRef]
- Syafhan NF,Donnelly R,Harper R,Harding J,Mulligan C,Hogg A,Scott M,Fleming G,Scullin C,Hawwa AF,Chen G,Pearson C,McEnlay JC.:Adherence to Metformin in adults with type 2 diabetes:a combined method approach.Journal of Pharmaceutical Policy and Practice 2022;15:61.DOI:10.1186/s40545-022-00457-5. [CrossRef]
- McGovern A,Tippu Z,Hinton W,Munro N, Whyte M,de Lusignan S.:A comparison of adherence and persistence by medication class in type 2 diabetes:A systematic review and meta-analysis.Diabetes Obes Metab.2018;20(4):1040-1043.
- Foret M,Guigas B,Viollet B.Metfrormin:update on mechanisms of action and repurposing potential.Nature rev endocrinol 2023.DOI:10.1038/s41574-023-00833-4. [CrossRef]
- Berchtold P,Bolli P,Arbenz U,Keiser G.:Intestinale Absorptionsstörung infolge Metforminbehandlung(Zur Frage der Wirkungsweise der Biguanide).Diabetologia 1969;5:405-412.
- Tomkin GH,Hadden DR,Weaver JA,Montgomery DAD..Vitamin-B12 Status of Patients on long-term Metformin Therapy.BMJ 1971;2:685-687.
- Kim J,Ahn CW,Fang S,Lee HS,Park JS.Association between metformin dose and vitamin B12 deficiency in patients with type 2 diabetes.Medicine 2019;98:46(e17918).
- Jager J,Kooy A, Lehret Ph, Wulfele‘MG, Kolk J,Bets D, Verbung J, Donker AJM,Stehouver CDA.:Long term treatment with metformin in patients with type 2 diabetes and risk of vitamin B-12 deficiency:randomized placebo controlled trial.BMJ 2010;340:c2181.DOI:10.1136/bmj.c2181. [CrossRef]
- Farland MZ,McPheeters CM, Renwick RC,Barlow PB,Williams JD,Keeble DS,Franks AS.:Comparison of patients receiving Long-Term Metformin Therapy and Vitamin B12 Monitoring.Journal of Pharmacy Technology.2015;31(4):184-189.
- Donnelly L,Dennis JM,Coleman RI,Sattar N,Hattersley AT,Holman RR,Pearson ER.:Risk of anemia with metformin Use in Type 2 diabetes:A MASTERMIND Study. Diabetes Care 2020;43:2493-2499.
- Serra MC,Kancherla V,Khakharia A,Allen LL,Phillips LS,Rhee MK,Wilson PWF,Vaughan CP.:Long-term metformin treatment and risk of peripheral neuropathy in older Veterans.Diabetes Research and Clinical Practice 2020;170:108486..DOI:10.1016/j.diabres.2020.108486. [CrossRef]
- Ahmed HH,Fadl NN,Kotob SE.Impact of long term Metformin therapy on hepcidin and Iron status in type II Diabetic Patients.IJPCR 2015;7 (3):185-193.
- 119. Bischoff F,Long L.STUDIES IN CARBOHYDRATE METABOLISM FOLLOWING GUANIDINE DEGLYCOGENATION.J Nutr 1930;3(2):201-216. doi.org/10.1093/jn/3.2.201. [CrossRef]
- Wilcock C,Bailey CJ.:Reconsideration of inhibitory effect of metformin on intestinal glucose absorption.J.Pharm.Pharmacol.1991;43:120-121. [CrossRef]
- Bailey CJ.Metformin and intestinal glucose handling.Diabetes Metab Rev 1995;11 Suppl 1:S23-32.
- Wu T, Xie C,Wu H, Jones KI,Horowitz M,Rayner CK.:Metformin reduces the rate of the small intestinal glucose absorption in type 2 diabetes.Diabetes,Obesity and Metab 2017;119(2):290-293.DOI.10.1111/dom.12812. [CrossRef]
- Horakova O,Kroupova P,Bardova K,Buresova J,Janowska P,Kopecky J,Rossmeisl M.:Metformin acutely lowers blood glucose levels by inhibition of intestinal glucose transport.Scientific Reports 2019;9:6156.DOI:10.1038/s41598-019-42531-0. [CrossRef]
- Penicaud L,Hitier Y,Ferre P,Girard J.Hypoglycemic effect of metformin in genetically obese(fa/fa) rats results from an increased utilization of blood glucose by intestine.Biochem J.1989;262:881-885. [CrossRef]
- Bronden A,Alber A,Rohde U,Rehfeld JF,Holst JJ,Vilsboell T,Knop FK.:Single-dose Metformin enhances Bile-Acid-induced Glucagon-like Peptide-1 Secretion in Patients with type-2 Diabetes.J Clin Endocrinol Metab 2017;102(11):4153-4162. [CrossRef]
- Ritzel U,Fromme A,Otteleben M,Leonhardt U,Ramadori G.Release of glucagon-like peptide-1(GLP-1) by carbohydrates in the perfused rat ileum.Acta Diabetol 1997;34:18-21. [CrossRef]
- Raddatz D,Nolte W,Roßbach C,Leonhardt U,Buchwald A,Scholz KH,Ramadori G.Measuring the effect of a Study Meal on Portal Concentrations of Glucagon-Like Peptide-1 (GLP-1) in Non Diabetic and Diabetic Patients with Liver Cirrhosis:Transjugular Intrahepatic Portosystemic Stent Shunt(TIPSS) as a New Method for Metabolic Measurement.Exp Clin Endocrinol Diabetes 2008;116:461-467. [CrossRef]
- Li B, Hu Y,Wang G, liu L.The effect of exenatide on fasting bile acids in newly diagnosed type 2 diabetes mellitus patients, a pilot study.BMC Pharmacology and toxicology 2020;21:44.DOI:10.1186/s40360-020-00422-5. [CrossRef]
- 129.Kim S,Chung JK,Kim BT, Relationship between gastrointestinal F-18-fluorodoxyglucose accumulation and gastrointestinal symptoms in whole-body PET.Clin Positron Imaging 1999;2:273-279. [CrossRef]
- Gontier E,Fourme E,Wartski M,Blondet C Bonardel G,Le Stanc E,Mantzarides M,Foehrenbach H,Pecking A-P,Alberini J-L.High and typical F18-FDG bowel uptake in patients treated with metformin.Eur J Med Mol Imaging 2008;35:95-99. [CrossRef]
- Morita Y,Nogami M,Sakaguchi K,Okada Y,Hirota Y,Sugawara K,Tamori Y,Zeng F,Murokami T,Ogawa W.:Enhanced Release of Glucose Into Intraluminal Space of the Intestine Associated With Metformin Treatment as Revealed by [18F]Fluorodeoxyglucose PET-MRI.Diabetes Care 2020;43:1796-1802.DOI:10.2337/dc20-0093. [CrossRef]
- Özülkur T,Özülkur F,Mert M,Özpacaci T.Clearance of high intestinal F18-FDG uptake associated with metformin after stopping the drug.Eur J Nucl Med Mol Imaging 2010;37:1011-1017.
- Schreuder N,Klarenbeek H,Vendel BN,Jager PL,Kosterink JGW,van Puijenbroek EP.Discontinuation of metformin to prevent metformin-induced high colonic FDG uptake:is 48 h sufficient?.Annals of Neclear Medicine 2020;34:833-839. [CrossRef]
- Vangu MDT,Momodu JI.F-18 FDG PET/CT Imaging in Normal Variants,Pitfalls and Artifacts in the Abdomen and Pelvis.Front. Nucl. Med.1:826109.DOI:10.3389/fnume.2021.826109. [CrossRef]
- Soyka JD,Strobel K,Veit-Haibach P,Schaefer NG,Schmid DT,Tschopp A,Hany TF.Influence of Bowel Preparation Before F18-FDG PET/CT on Physiologic F-18-FDG Activity in the Intestine.J Nucl Med 2010;51:507-510. [CrossRef]
- Tu DG,Chen C-R,Wang Y-W,Tu C-W,Huang YC.Bowel-cleansing methods affecting PET-CT Image interpretation.Nucl Med Commun 2011;32:570-574. [CrossRef]
- Chen Y-K,Chen J-H,Tsui C-C,Chou H-H,Cheng R-H,Chiu J-S.Use of Laxative-augmented Contrast Medium in the Evaluation of Colorectal Foci at FDG PET.Radiology 2011;259 (2):525-533. [CrossRef]
- Katsumata R,Manabe N,Ayaki M,Tanikawa T,Fujita M,Ono Y, Fujiwara H,Suehiro M,Monobe Y,Kato K,Kawamoto H,Haruma K.Increased Colonic Fluorodeoxyglucose Uptake in Melanosis Coli-A Case Series of Three Patients.Gastro Hep Advances 2022;1:125-128. [CrossRef]
- Soskin S,Allweiss MD,Cohn DJ.INFLUENCE OF THE PANCREAS AND THE LIVER UPON THE DEXTROSE TOLERANCE CURVE.Am J Physiol-Legacycontent 1934;109(1):155-165.DOI:10.1152/ajphysiog.1934.109.1.155.
- Madison LL.Role of insulin in the hepatic Handling of glucose.Arch Int Med 1969;123:284-292. [CrossRef]
- Balks H-J,Jungermann K.Regulation of peripheral insulin/glucagon levels by rat liver.Eur J Biochem 1984;141:645-650. [CrossRef]
- Guzelian Ph,Boyer JL.Glucose Reabsorption from Bile.J Clin Invest 1974;53:526-535.
- Ikeda T,Iwata K,Murakami H.Inhibition Effect of Metformin on Intestinal Glucose Absorption in the perfused Rat Intestine.Biochem Pharmacology 2000;59:887-890. [CrossRef]
- Albai O,Timar B,Paun DL,Sima A,Roma D,Timar R.:Metformin Treatment:A potential Cause of Megaloblastic Anemia in Patients with Type 2 Diabetes Mellitus.Diabetes,Metabolic Syndrome and Obesity:Targets and Therapy 2020;13:3873-3878.
- Infante M,Leoni M,Caprio M,Fabbri A.:Long-term Metformin therapy and Vitamin B12 deficiency:An association to bear in mind.World Journal Diabetes 2021;12(7):916-931.
- Wijnen JCF,Van De Riet IR,Lijfering WM,Van Der Meer FJM.Metformin use decreases the anticoagulant effect of phenprocoumon.J Thromb Haemost 2014;12:887-890. [CrossRef]
- Baker EH,Sandle GI.Complications of Laxative Abuse.Ann Rev Med 1996;47:127-134. [CrossRef]
- Hart SL,McColl I.The effect of the laxative oxyphenisatin on the intestinal absorption of glucose in rat and man.Br J Pharmac Chemother 1968;32:683-686. [CrossRef]
- Nilsson SE,Takkinen S,Johansson B,Dotevall G,Melander a,Berg S,McClearn G.Laxatie treatment elevates plasma homocysteine:a study on a population-based Swedish sample of old people.Eur J Clin Pharmacol 2004;60:45-49. [CrossRef]
- Sturtzel B,DIETRICH A,Wagner KH,Gisinger C,Elmafda I.The status of vitamins B6,B12,Folate,and of HOmocysteine in Geriatric home residents receiving laxatives or dietary fiber.The Journal of Nutrition Health.2010;14(3):219-223. [CrossRef]
- Sugimoto H,Yamada U.Iron deficiency anemia induced by magnesium overuse:a case report.BioPsychoSocial Medicine 2019;13:18.DOI:10.1186/s13030-019-0159-y. [CrossRef]
- Naz N,Malik IA,Sheikh N,Ahmad S,Kahn S,Blaschke M,Schultze F,Ramadori G.Ferroportin-1 is a nuclear negative acute-phase protein in rat liver:a comparison with other iro-transport proteins.Lab Invest 2012;92:842-856. [CrossRef]
- Ahmad S,Moriconi F,Naz N,Sultan S,Sheikh N,Ramadori G,Malik IA.Ferritin L and Ferritin H are differentially located within hepatic and extra hepatic organs under physiological and acute-phase conditions.Int J Clin Exp Pathol 2013;6(4):622-629.
- 83.Tan J,Wang Y,Liu S,Shi Q,Zhou X,Zhou Y, Yang X, Chen P,Li s.:Long-Acting Metformin Vs Metformin Immediate Release in Patients With Type 2 Diabetes:A Systematic Review.Frontiers in Pharmacology 2021;12.DOI:10.3389/fphar.2021.669814. [CrossRef]
- 67.Chalmers J,Cooper ME.:UKPDS and the Legacy Effect.N Engl J Med.2008;359:1618-1620. [CrossRef]
- 100.Tang Y,Weiss T,Liu J,Rajpathak S,Khunti K.:Metformin adherence and discontinuation among patients with type 2 diabetes:A retrospective cohort study.Journal of Clinical and Translational Endocrinology 2020;20:100235.DOI:10.1016/j.jcte.2020.100225. [CrossRef]
- 108.Wamil M,Coleman RL,Adler AI,McMurray JJV,Holman RR.Increased risk of Incident heart Failure and death is asociated with insulin Resistance in people with newly diagnosed Type 2 Diabetes:UKPDS 89.Diabetes Care 2021;44:1877-1884. [CrossRef]
- 118.Love KM,Barrett EJ,Horton WB.Metforrmin‘s impact on the Microvascular Response to Insulin.Endocrinology 2022;;162(11).DOI:10.1210/endocr/bqac162. [CrossRef]
- 80.Spiller R:Inhibiting glucose absorption to treat constipation.Lancet Gastroenterol Hepatol 2018;.DOI:10.1016/S2468-1253(18)30214-0. [CrossRef]
- Kuriyama S,Ohmori K,Miura Ch,Suzuki Y,Nakaya N,Fujita K,Sato Y, Tsubono Y,Tsuji I,Fukao A, Hisamichi S.:Body Mass Index and Mortality in Japan:The Miyagi Cohort Study.J Epidemiol 2004;14 (Supl1):S-33-38. [CrossRef]
- Gerstein HC.:Shouln‘t Preventing Type 2 Diabetes Also Prevent Ist long-term Consequences?.Circulation 22022;145:1642-1644.DOI:10.1161/CIRCULATIONAHA.122.060026. [CrossRef]
- Rehee E-J.:Extra-Glycemic Effects of Anti-Diabetic Medications:Two Birds with One Stone?.Endocrinol Metab 2022;37:415-429.DOI:10.3803/EnM.2022.304. [CrossRef]
- Campbell JE,Drucker DJ.Pharmacology,Physiology,and Mechanisms of Incretin Hormon Action.Cell Metabolism.2013;17:819-836.
- 35.Palmer JP.:Farewell to Phenformin for treating Diabetes Mellitus.Ann Int Med 1975;83(4):567-568. [CrossRef]
- 34.Clarke BF,Duncan LJP.Comparison of chlorpropamide and metformin treatment on weight and blood-glucose response and uncontrolled obese diabetics.Lancet 1968;1:123-126. [CrossRef]
- 40.Hermann LS,Schersten B,Bitzen P-O,Kjellström T,Lindgärde F,Melander A.:Therapeutic Comparison of Metformin and Sulfonylurea,Alone and in Various Combinations.A double-blind controlled study. Diabetes Care 1994;17(10):1100-1109. [CrossRef]
- .Heyden S,Cassel JC,Bartel A,Tyroler HA,Hames CG,Ga C,Cornoni JC.Body Weight and Cigarette Smoking As Risk Factors.Arch Intern Med 1971;128:915-919. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



