Submitted:
29 June 2023
Posted:
29 June 2023
You are already at the latest version
Abstract
Keywords:
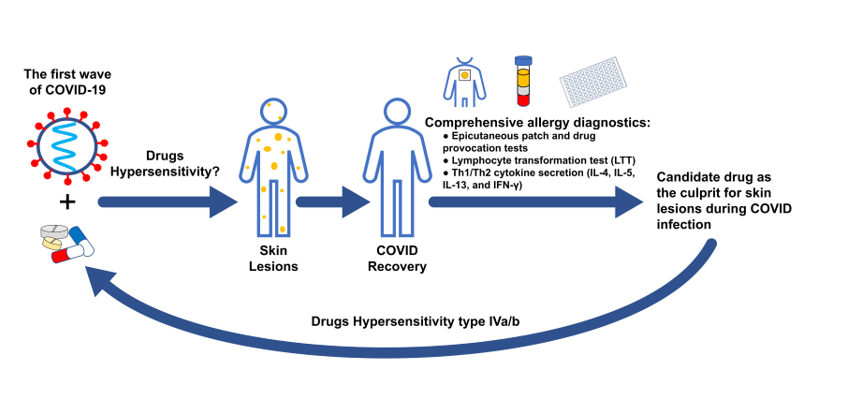
1. Introduction
2. Results
2.1. Design and setting
| Epicutaneous Patch Test | Oral challenges | LTT | |||||||
| Patient | Age (years)/Sex | COVID-19 treatments | Reaction | Positive | Negative | Positive | Negative | Positive | Negative |
| P1 | 60/M | AZT, DOL | MPE(1) | AZT, DOL | AZT, DOL | HCQ | AZT, AMOX, CLA, LOP, RIT | ||
| P2 | 61/V | AZT, KAL, DOL, CEL | UEX (1) | AZT, DOL, KAL, CEL | AZT § | DOL, KAL | HCQ, AZT, AMOX, LOP, RIT | ||
| P3 | 53/V | AZT, KAL, DOL, CEL, AMOX/CLA | MPE(1) | DOL, KAL, CEL, AMOX | AMOX/CLA | KAL, AZT, HCQ | CLA | HCQ, AZT, AMOX, LOP, RIT | |
| P4 | 63/M | AZT, KAL, DOL, CEL | MPE(1), VEX(2) | AZT, DOL, KAL | AZT, HCQ, KAL | LOP, RIT | HCQ, AZT, AMOX | ||
| P5 | 66/V | AZT, KAL, DOL, CEL | MPE(1), VEX(2) | AZT, DOL, KAL, CEL | AZT | KAL, CEL | AZT, HCQ | AMOX, LOP, RIT | |
| P6 | 61/M | AZT, DOL | MPE(1) VEX(2) | AZT, DOL | DOL, AZT | AZT, LOP, RIT | HCQ, AMOX | ||
| P7 | 77/M | KAL, DOL, CEL | MPE(1) | * | * | LOP, RIT | HCQ, AZT, AMOX | ||
| P8 | 84/V | AZT, KAL, DOL, CEL | MPE(1), VEX(2) | AZT, DOL, KAL, CEL | AZT, DOL, KAL, CEL | LOP, RIT | HCQ, AZT, AMOX | ||
| P9 | 76/V | AZT, DOL | VEX(2) | AZT, DOL | AZT, DOL | HCQ, RIT | AZT, AMOX, LOP | ||
| P10 | 74/V | AZT, DOL, CEL | CVAS(3), CLL(4) | AZT, DOL, CEL | * | * | AZT, RIT | AZT, AMOX, LOP | |
| P11 | 64/V | AZT, KAL, DOL | MPE(1) | AZT, KAL, DOL | AZT, KAL, DOL | LOP, RIT | HCQ, AZT, AMOX | ||
| NAP1 | 58/V | AZT, KAL, DOL | HCQ, AZT, AMOX, LOP, RIT | ||||||
| NAP2 | 52/V | AZT, KAL, DOL, CEL | HCQ, AZT, AMOX, LOP, RIT | ||||||
| NAP3 | 59/V | AZT, KAL, DOL, CEL | HCQ, AZT, AMOX, LOP, RIT | ||||||
| NAP4 | 49/V | AZT, DOL | HCQ, AZT, AMOX, LOP, RIT | ||||||
| NAP5 | 70/V | AZT, KAL, DOL, CEL | HCQ, AZT, AMOX, LOP, RIT | ||||||
| (1) Generalized exanthema: MPE, maculopapular exanthem or UEX, urticarial exanthem; (2) VEX, vesicular exanthem; (3) CVAS, cutaneous vasculitis; (4) CLL, chilblain-like lesion. AZT, azitromicin; AMOX, Amoxicillin; CLA, Clavulanic Acid, DOL, Dolquine; HCQ, hydroxychloroquine; KAL, Kaletra; LOP, Lopinavir; RIT, Ritonavir; CEL, Ceftriaxona; § Immediate reaction with 12,5 mg; * Oral challege not posible because generalized severe reaction. | |||||||||
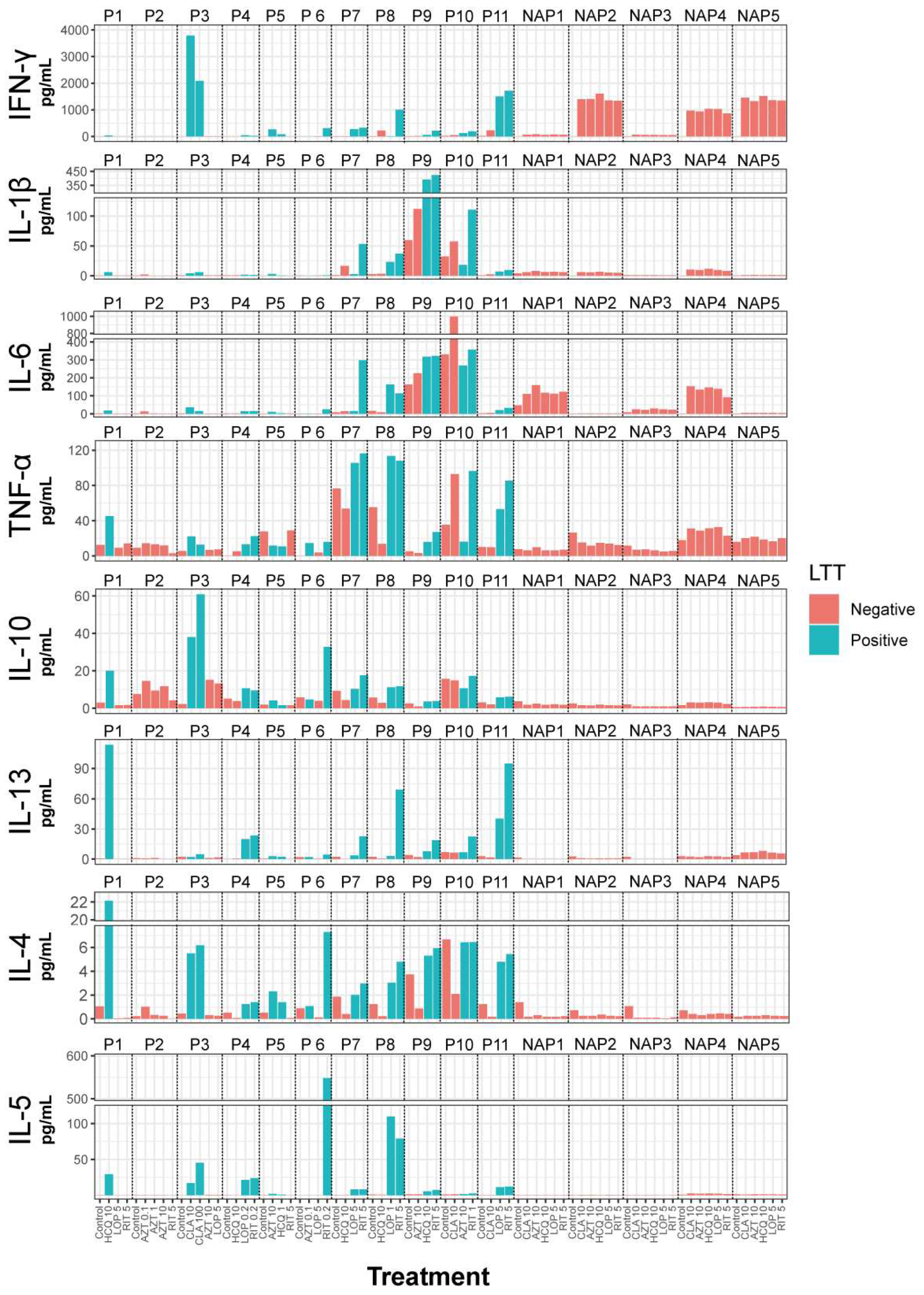
2.2. Lymphocyte transformation test (LTT)
2.3. Cytokine secretion
3. Discussion
4. Materials and Methods
4.1. Lymphocyte transformation test (LTT)
4.2. Secreted cytokine measurement
5. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Riggioni, C.; Comberiati, P.; Giovannini, M.; Agache, I.; Akdis, M.; Alves-Correia, M.; Anto, J. M.; Arcolaci, A.; Azkur, A. K.; Azkur, D.; Beken, B.; Boccabella, C.; Bousquet, J.; Breiteneder, H.; Carvalho, D.; De Las Vecillas, L.; Diamant, Z.; Eguiluz-Gracia, I.; Eiwegger, T.; Eyerich, S.; Fokkens, W.; Gao, Y. D.; Hannachi, F.; Johnston, S. L.; Jutel, M.; Karavelia, A.; Klimek, L.; Moya, B.; Nadeau, K. C.; O'Hehir, R.; O'Mahony, L.; Pfaar, O.; Sanak, M.; Schwarze, J.; Sokolowska, M.; Torres, M. J.; van de Veen, W.; van Zelm, M. C.; Wang, Y.; Zhang, L.; Jimenez-Saiz, R.; Akdis, C. A. , A compendium answering 150 questions on COVID-19 and SARS-CoV-2. Allergy 2020, 75, 2503–2541. [Google Scholar] [CrossRef] [PubMed]
- Galvan Casas, C.; Catala, A.; Carretero Hernandez, G.; Rodriguez-Jimenez, P.; Fernandez-Nieto, D.; Rodriguez-Villa Lario, A.; Navarro Fernandez, I.; Ruiz-Villaverde, R.; Falkenhain-Lopez, D.; Llamas Velasco, M.; Garcia-Gavin, J.; Baniandres, O.; Gonzalez-Cruz, C.; Morillas-Lahuerta, V.; Cubiro, X.; Figueras Nart, I.; Selda-Enriquez, G.; Romani, J.; Fusta-Novell, X.; Melian-Olivera, A.; Roncero Riesco, M.; Burgos-Blasco, P.; Sola Ortigosa, J.; Feito Rodriguez, M.; Garcia-Doval, I. , Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol 2020, 183, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Sameni, F.; Hajikhani, B.; Yaslianifard, S.; Goudarzi, M.; Owlia, P.; Nasiri, M. J.; Shokouhi, S.; Bakhtiyari, M.; Dadashi, M. , COVID-19 and Skin Manifestations: An Overview of Case Reports/Case Series and Meta-Analysis of Prevalence Studies. Frontiers in medicine 2020, 7, 573188. [Google Scholar] [CrossRef] [PubMed]
- Recalcati, S.; Gianotti, R.; Fantini, F. , COVID-19: The experience from Italy. Clinics in dermatology 2021, 39, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Hernandez, R.; Solano-Solares, E.; Chica-Guzman, V.; Fernandez-Guarino, M.; Fernandez-Nieto, D.; Ortega-Quijano, D.; de-Andres-Martin, A.; Moreno, C.; Carretero-Barrio, I.; Garcia-Abellas, P.; Gonzalez-de-Olano, D.; de-la-Hoz-Caballer, B. , SARS-CoV-2, skin lesions and the need of a multidisciplinary approach. Journal of the European Academy of Dermatology and Venereology : JEADV 2020, 34, e659–e662. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, G. A.; Ripa, M.; Burastero, S.; Benanti, G.; Bagnasco, D.; Nannipieri, S.; Monardo, R.; Ponta, G.; Asperti, C.; Cilona, M. B.; Castagna, A.; Dagna, L.; Yacoub, M. R. , Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Focus on the Pathophysiological and Diagnostic Role of Viruses. Microorganisms 2023, 11. [Google Scholar] [CrossRef]
- Karimi, A.; Pourbakhtiaran, E.; Fallahi, M.; Karbasian, F.; Armin, S.; Babaie, D. , Is It Stevens-Johnson Syndrome or MIS-C with Mucocutaneous Involvement? Case reports in pediatrics 2021, 2021, 1812545. [Google Scholar] [CrossRef]
- Lootah, S.; Alshammari, E.; Alqanatish, J. , Complete Remission in a Child With Multisystem Inflammatory Syndrome and Stevens-Johnson Syndrome Treated With Infliximab. Cureus 2023, 15, e37076. [Google Scholar] [CrossRef]
- Pichler, W. J. , Delayed drug hypersensitivity reactions. Annals of internal medicine 2003, 139, 683–693. [Google Scholar] [CrossRef]
- Porebski, G.; Gschwend-Zawodniak, A.; Pichler, W. J. , In vitro diagnosis of T cell-mediated drug allergy. Clin Exp Allergy 2011, 41, 461–470. [Google Scholar] [CrossRef]
- Sachs, B.; Fatangare, A.; Sickmann, A.; Glassner, A. , Lymphocyte transformation test: History and current approaches. J Immunol Methods 2021, 493, 113036. [Google Scholar] [CrossRef] [PubMed]
- Lochmatter, P.; Beeler, A.; Kawabata, T. T.; Gerber, B. O.; Pichler, W. J. , Drug-specific in vitro release of IL-2, IL-5, IL-13 and IFN-gamma in patients with delayed-type drug hypersensitivity. Allergy 2009, 64, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Srinoulprasert, Y. , Lymphocyte transformation test and cytokine detection assays: Determination of read out parameters for delayed-type drug hypersensitivity reactions. J Immunol Methods 2021, 496, 113098. [Google Scholar] [CrossRef] [PubMed]
- Solano-Solares, E.; Chica-Guzman, V.; Perez-Allegue, I.; Cabrera-Hernandez, R.; Fernandez-Guarino, M.; Fernandez-Nieto, D.; Moreno-Garcia-Del-Real, C.; de-Andres-Martin, A.; Garcia-Bermejo, L.; Gonzalez-de-Olano, D.; de-la-Hoz-Caballer, B. , Role of Drug Hypersensitivity in the Cutaneous Manifestations of SARS-CoV-2 Infection. Journal of investigational allergology & clinical immunology 2022, 32, 218–220. [Google Scholar]
- Cabanas, R.; Ramirez, E.; Sendagorta, E.; Alamar, R.; Barranco, R.; Blanca-Lopez, N.; Dona, I.; Fernandez, J.; Garcia-Nunez, I.; Garcia-Samaniego, J.; Lopez-Rico, R.; Marin-Serrano, E.; Merida, C.; Moya, M.; Ortega-Rodriguez, N. R.; Rivas Becerra, B.; Rojas-Perez-Ezquerra, P.; Sanchez-Gonzalez, M. J.; Vega-Cabrera, C.; Vila-Albelda, C.; Bellon, T. , Spanish Guidelines for Diagnosis, Management, Treatment, and Prevention of DRESS Syndrome. Journal of investigational allergology & clinical immunology 2020, 30, 229–253. [Google Scholar]
- Mendez Maestro, I.; Pena Merino, L.; Udondo Gonzalez Del Tanago, B.; Aramburu Gonzalez, A.; Orbea Sopena, A.; Sanchez De Vicente, J.; Raton Nieto, J. A.; Acebo Marinas, E.; Gardeazabal Garcia, J. , Skin manifestations in patients hospitalized with confirmed COVID-19 disease: a cross-sectional study in a tertiary hospital. International journal of dermatology 2020, 59, 1353–1357. [Google Scholar] [CrossRef]
- Nakashima, C.; Kato, M.; Otsuka, A. , Cutaneous manifestations of COVID-19 and COVID-19 vaccination. The Journal of dermatology 2023, 50, 280–289. [Google Scholar] [CrossRef]
- Fernandez-Nieto, D.; Ortega-Quijano, D.; Suarez-Valle, A.; Jimenez-Cauhe, J.; Jaen-Olasolo, P.; Fernandez-Guarino, M. , Lack of skin manifestations in COVID-19 hospitalized patients during the second epidemic wave in Spain: a possible association with a novel SARS-CoV-2 variant - a cross-sectional study. Journal of the European Academy of Dermatology and Venereology : JEADV 2021, 35, e183–e185. [Google Scholar] [CrossRef]
- Suthumchai, N.; Srinoulprasert, Y.; Thantiworasit, P.; Rerknimitr, P.; Tuchinda, P.; Chularojanamontri, L.; Rerkpattanapipat, T.; Chanprapaph, K.; Disphanurat, W.; Chakkavittumrong, P.; Tovanabutra, N.; Srisuttiyakorn, C.; Sukasem, C.; Klaewsongkram, J. , The measurement of drug-induced interferon gamma-releasing cells and lymphocyte proliferation in severe cutaneous adverse reactions. Journal of the European Academy of Dermatology and Venereology : JEADV 2018, 32, 992–998. [Google Scholar] [CrossRef]
- Yawalkar, N.; Shrikhande, M.; Hari, Y.; Nievergelt, H.; Braathen, L. R.; Pichler, W. J. , Evidence for a role for IL-5 and eotaxin in activating and recruiting eosinophils in drug-induced cutaneous eruptions. J Allergy Clin Immunol 2000, 106, 1171–1176. [Google Scholar] [CrossRef]
- Sachs, B.; Erdmann, S.; Malte Baron, J.; Neis, M.; al Masaoudi, T.; Merk, H. F. , Determination of interleukin-5 secretion from drug-specific activated ex vivo peripheral blood mononuclear cells as a test system for the in vitro detection of drug sensitization. Clin Exp Allergy 2002, 32, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Merk, H. F. , Diagnosis of drug hypersensitivity: lymphocyte transformation test and cytokines. Toxicology 2005, 209, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Glassner, A.; Wurpts, G.; Roseler, S.; Yazdi, A. S.; Sachs, B. , In vitro detection of T cell sensitization by interferon-gamma secretion in immediate-type drug allergy. Clin Exp Allergy 2023, 53, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Halevy, S.; Cohen, A.; Livni, E. , Acute generalized exanthematous pustulosis associated with polysensitivity to paracetamol and bromhexine: the diagnostic role of in vitro interferon-gamma release test. Clinical and experimental dermatology 2000, 25, 652–654. [Google Scholar] [CrossRef] [PubMed]
- Gaspard, I.; Guinnepain, M. T.; Laurent, J.; Bachot, N.; Kerdine, S.; Bertoglio, J.; Pallardy, M.; Lebrec, H. , Il-4 and IFN-gamma mRNA induction in human peripheral lymphocytes specific for beta-lactam antibiotics in immediate or delayed hypersensitivity reactions. Journal of clinical immunology 2000, 20, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Cabanas, R.; Calderon, O.; Ramirez, E.; Fiandor, A.; Caballero, T.; Heredia, R.; Herranz, P.; Madero, R.; Quirce, S.; Bellon, T. , Sensitivity and specificity of the lymphocyte transformation test in drug reaction with eosinophilia and systemic symptoms causality assessment. Clin Exp Allergy 2018, 48, 325–333. [Google Scholar] [CrossRef]
- Glassner, A.; Dubrall, D.; Weinhold, L.; Schmid, M.; Sachs, B. , Lymphocyte transformation test for drug allergy detection: When does it work? Ann Allergy Asthma Immunol 2022, 129, 497–506. [Google Scholar] [CrossRef]
- Hayakawa, J.; Takakura, H.; Mizukawa, Y.; Shiohara, T. , COVID-19-related cutaneous manifestations associated with multiple drug sensitization as shown by lymphocyte transformation test. Journal of the European Academy of Dermatology and Venereology : JEADV 2020, 34, e779–e781. [Google Scholar] [CrossRef]
- Giraldo-Tugores, M.; Sanmartin-Fernandez, M.; Fernandez-Lozano, C.; Martinez-Botas, J.; De-la-Hoz-Caballer, B.; Gonzalez-de-Olano, D. , Kounis Syndrome and Vanadium allergy: heed your hunch. Journal of investigational allergology & clinical immunology 2023, 0. [Google Scholar]
| Azitromicin | Amoxicillin | Clavulanic Acid | Hidroxiloroquine | Lopinavir | Ritonavir | ||||||||||||||||
| Patient | ( Dynabeads™ CD3/CD28) |
0.1 µg/µl |
1 µg/µl |
10 µg/µl |
100 µg/µl |
200 µg/µl |
500 µg/µl |
1 µg/µl |
10 µg/µl |
100 µg/µl |
1 µg/µl |
10 µg/µl |
100 µg/µl |
0.2 µg/ml |
1 µg/ml |
5 µg/ml |
0.2 µg/µl |
1 µg/µl |
5 µg/µl |
||
| P1 | 10.2 | 0.8 | 1.5 | 1.5 | 1.4 | 1.4 | 1.2 | 1.4 | 1.6 | 1.6 | 1.4 | 3.2 | 0.3 | 1.7 | 2.1 | 2.2 | 1.4 | 1.2 | 1.2 | ||
| P2 | 12.2 | 0.5 | 1.0 | 0.5 | 0.9 | 1.4 | 1.5 | 1.2 | 1.2 | 1.4 | 1.2 | 1.8 | 1.9 | 1.1 | 0.8 | 0.9 | 1.3 | 1.0 | 0.8 | ||
| P3 | 15.0 | 1.3 | 1.5 | 1.9 | 1.7 | 1.7 | 1.4 | 1.9 | 3.2 | 3.4 | 1.4 | 1.8 | 0.3 | 2.0 | 1.8 | 0.9 | 2.0 | 1.8 | 1.8 | ||
| P4 | 5.8 | 0.8 | 0.7 | 0.9 | 0.7 | 2.1 | 0.7 | 0.7 | 0.7 | 0.8 | 0.6 | 1.1 | 0.1 | 3.9 | 0.6 | 0.9 | 3.5 | 0.3 | 0.5 | ||
| P5 | 9.0 | 1.7 | 1.4 | 3.1 | 1.6 | 1.4 | 2.8 | 1.9 | 1.7 | 1.6 | 3.4 | 0.8 | 0.7 | 1.3 | 1.0 | 0.8 | 1.1 | 1.2 | 0.6 | ||
| P6 | 11.6 | 3.0 | 1.7 | 1.0 | 1.9 | 1.6 | 1.9 | 2.4 | 2.5 | 2.3 | 1.3 | 1.8 | 1.6 | 1.9 | 3.0 | 2.8 | 2.9 | 3.2 | 2.6 | ||
| P7 | 9.4 | 1.4 | 0.9 | 1.4 | 1.8 | 1.1 | 1.5 | 1.8 | 1.5 | 1.4 | 1.7 | 2.0 | 1.8 | 1.9 | 2.3 | 3.4 | 2.3 | 1.0 | 3.2 | ||
| P8 | 7.4 | 1.9 | 1.7 | 1.6 | 1.9 | 1.8 | 1.9 | 1.9 | 1.5 | 1.6 | 1.6 | 1.9 | 1.9 | 1.1 | 3.3 | 1.6 | 1.8 | 1.5 | 3.2 | ||
| P9 | 6.7 | 1.5 | 1.3 | 1.2 | 1.3 | 1.2 | 1.2 | 1.4 | 1.3 | 1.2 | 1.6 | 3.2 | 1.5 | 1.2 | 2.7 | 1.8 | 1.6 | 3.0 | 3.4 | ||
| P10 | 8.2 | 1.1 | 1.8 | 3.1 | 0.7 | 0.9 | 1.2 | 1.0 | 1.3 | 1.1 | 0.9 | 0.8 | 1.0 | 2.4 | 1.5 | 1.3 | 2.6 | 3.1 | 2.1 | ||
| P11 | 8.4 | 1.6 | 1.4 | 1.2 | 1.4 | 1.2 | 1.2 | 1.5 | 1.8 | 1.4 | 1.5 | 1.3 | 1.3 | 1.7 | 2.1 | 3.2 | 1.9 | 2.7 | 3.3 | ||
| NAP1 | 6.3 | 1.4 | 1.1 | 1.0 | 1.1 | 0.9 | 0.9 | 0.9 | 1.0 | 0.9 | 0.4 | 1.4 | 0.9 | 1.1 | 1.2 | 1.2 | 0.9 | 1.2 | 0.9 | ||
| NAP2 | 5.5 | 1.9 | 1.0 | 1.5 | 1.3 | 0.9 | 0.7 | 1.6 | 1.0 | 0.8 | 0.4 | 1.7 | 0.9 | 1.8 | 1.4 | 1.2 | 1.8 | 1.5 | 1.2 | ||
| NAP3 | 5.2 | 1.5 | 1.1 | 1.7 | 1.9 | 2.0 | 1.5 | 1.9 | 1.2 | 1.3 | 1.8 | 0.8 | 1.6 | 1.0 | 0.6 | 0.8 | 0.9 | 0.8 | 0.7 | ||
| NAP4 | 9.9 | 1.8 | 1.6 | 2.0 | 2.0 | 1.9 | 1.8 | 1.9 | 1.4 | 1.8 | 1.9 | 1.3 | 1.1 | 1.5 | 2.0 | 1.8 | 1.9 | 1.9 | 1.8 | ||
| NAP5 | 7.8 | 1.4 | 1.2 | 0.9 | 0.8 | 0.8 | 0.7 | 0.9 | 0.9 | 0.7 | 0.3 | 0.2 | 1.1 | 1.9 | 0.9 | 0.7 | 0.9 | 0.8 | 0.6 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




