Introduction
The chronic myeloid leukemia (CML) is characterized by BCR-ABL1 fusion gene in Philadelphia chromosome [
1]. BCR-ABL1 fusion gene produces the BCR-ABL tyrosine kinase, which leads to leukemia cell proliferation [
2,
3,
4]. Three tyrosine kinase inhibitors (TKIs) were used to frontline treatment of CML-CP including imatinib, nilotinib and dasatinib [
5]. Imatinib was first approved for therapy of ND-CML-CP with a superior efficacy of imatinib compared to interferon-α plus cytarabine [
6]. Nilotinib and dasatinib as second-generation TKIs were also approved for therapies [
7,
8]. In vitro, nilotinib was deteted to be of greater selectivity for ABL kinase and more inhibitory compared to imatinib [
9,
10]. The dose of nilotinib recommended is 300 mg twice daily (BID) for patients with ND-CML-CP [
11]. Frontline nilotinib (300 mg BID) was reported a higher rate of deep molecular response (DMR) compared with imatinib (MR
4.5 by 2 years: nilotinib 26% versus imatinib 10%; P < 0.0001) in the ENESTnd study [
12]. Besides, 54% and 61.0% of patients in nilotinib arm (300 mg BID) achieved MR
4.5 compared with 31% and 39.2% of patients in imatinib arm by 5 years and 10 years follow-up, respectively [
12,
13].
Recently, treatment-free remission (TFR) was the new goal for CML-CP patients. DMR was served as a milestone in TFR, and described in European LeukemiaNet (ELN) and LALNET recommendations and NCCN guideline [
14,
15,
16]. Patients with achievement of early stable DMR can acquire a chance to discontinue medication for TFR. Therefore, it is significant to early distinguish patients who can obtain DMR and fit for TFR. However, the cumulative MR
4.5 rate of nilotinib was not yet detected in the ENESTchina study with a 12-month follow-up, and the early cumulative incidence of MR
4.5 rate of nilotinib by 18 months was also not investigated in previous studies. Besides, early molecular response (EMR) may be as the first milestone for treatment of CML-CP, and a new marker for long-term outcome in progression-free survival (PFS) or overall survival (OS) [
16,
17,
18]. Masahiro’s study showed that patients achieved EMR at 3 or 6 months might lead to higher rate of DMR by 36 months and better PFS than those without EMR at 3 or 6 months with second-generation TKIs treatments [
19]. Therefore, we explore the relation betweeen ultra-early molecular response (u-EMR) including international scale (IS) decreased more than 10% from baseline at 6 weeks and the cumulative incidence of MR
4.5 rate in this study.
We designed a phase IV, prospective, multicenter study to detect the early cumulative incidence of MR4.5 rate by 18 months with nilotinib baesd on ELTS score in patients with ND-CML-CP in China. Because the cumulative MR4.5 rate of nilotinib was not yet detected in the ENESTchina study with a 12-month follow-up, this clinical data will be important supplement for the efficacy of MR4.5 in real world ND-CML-CP patients in China. The cumulative incidence of MR4.0 and MMR, u-EMR, PFS and OS were also evaluated in this trial.
Materials and Methods
Trial design and patients
This phase IV, multi-centre, single-arm, prospective study was performed in China. The key eligibility criteria, including age ≥18 years, a confirmed ND-CML-CP (postive BCR-ABL mRNA or positive Philadelphia chromosome) within 6 months of study registration, no accelerated phase (AP) or blast phase (BP) criteria, and an Eastern Cooperative Oncology Group performance-status (ECOG) score less than 2 were eligible for inclusion. Patients with T315I mutations of BCR-ABL or previous receipt of any TKI treatment were excluded. Patient with history of severe heart or lung disease was excluded from this trial.
This research was approved by the institutional review boards at Shenzhen Second People's Hospital and other participating institutions. This trial was adhered to the ethical principles of the Declaration of Helsinki. Written informed consents for all patients were obtained before study procedures. This study was registered in Clinical Trial prs. gov Registry (NCT03942094).
Intervention and End Points
Each patient in this trial received nilotinib 300 mg BID until disease progression. Primary endpoint was the cumulative MR4.5 rate by 18 months. Secondary endpoints were as follows: cumulative MR4.5, molecular response 4.0 (MR4.0), and major molecular response (MMR) at 3,6,9,12 and 24 months; ultra-early molecular response (u-EMR); PFS and OS.
MMR was defined as BCR-ABL1 IS (BCR-ABL1/ABL1 ratio on the International Scale [IS]) ≤ 0.1%. MR
4.0 was defined as BCR-ABL1 IS ≤ 0.01%. MR
4.5 was defined as BCR-ABL1IS ≤ 0.0032% [
5]. We defined ultra-early molecular response (u-EMR) of nilotinib including international scale (IS) decreased more than 10% from baseline at 6 weeks, considering previous papers defined an early molecular response (EMR) as BCR-ABL1 transcript level <10% according to the International Scale after 3 months of therapy [
17,
20,
21]. PFS was defined as disease progression to AP/BP or loss of response. OS was defined as duration from day 0 to last follow-up or death. Adverse events due to TKIs were decided according to Common Terminology Criteria for Adverse Events (CTCAE) version 5.0.
Statistical Analysis
This clinical data cutoff date was September 30, 2022. Population was performed with the intention-to-treat method. Data were described as median (range). The cumulative MMR, MR4.0, MR4.5, PFS and OS were assessed using the Kaplan–Meier method, patients’ background or the achievement of u-EMR, etc., using log-rank test. All statistical analyses were detected using stata software version 14.1.
Results
Characteristics of patients
Between July 2019 and September 2022, 106 newly diagnosed CML-CP patients were enrolled from 29 institutes in China. The median age was 36 years (range, 18–76 years), and 68 (64.2%) patients were men. Median time from diagnosis to nilotinib treatment was 11 days (range, 0–84 days). The number of patients with low, intermediate and high EUTOS long-term survival (ELTS) score were 72 (67.9%), 21(19.8%) and 11(10.4%), respectively. 5 (4.7%) patients were of chromosomal abnormalities in addition to the philadelphia chromosome, and 31 (29.2%) patients were of large spleen size ≥ 10cm below costal margin. The median hemoglobin (Hb), platelet count (PLT) and white-cell count (WBC) in the peripheral blood at the time of CML diagnosis were 108 g/L (range, 56-384 g/L), 451 x 10^9/L (range, 73-3444 x 10^9/L) and 122.4 x 10^9/L (range, 8.0-525.1 x 10^9/L), respectively. Clinical characteristics and the key baseline are summarized in
Table 1.
Primary and Secondary endpoints
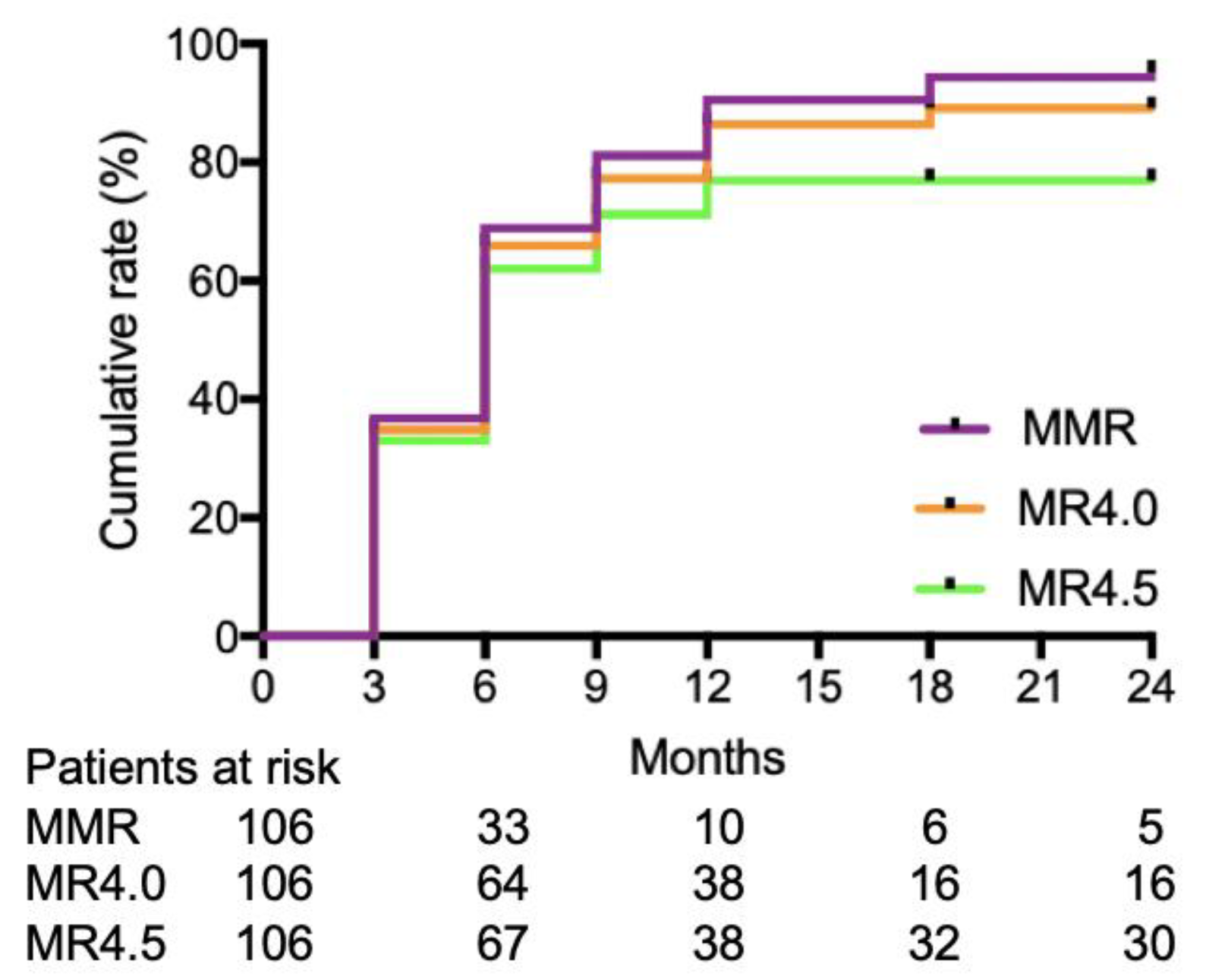
The cumulative MR
4.5 rate of nilotinib by 18 months was 69.8% (74/106). The median time for patients obatined MR
4.5 was 6 (range, 3-24) months. The cumulative MMR and MR
4.0 rates of nilotinib by 18 months was 94.3% (100/106) and 84.9% (90/106), respectively (
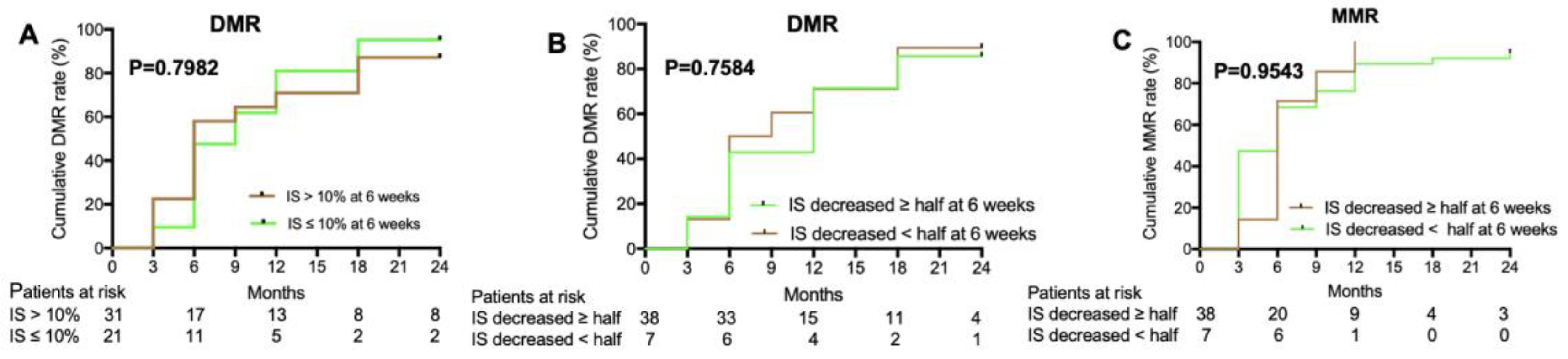
Figure 1). Additionally, u-EMR at 6 weeks (BCR-ABL IS≤ 10% or IS decreased ≥ half of IS at diagnose) was achieved 40.4% (21/54) and 84.4% (38/45), respectively. However, patients with u-EMR at 6 weeks were not significantly different to achieve DMR or MMR by 24 months compared to those without u-EMR (P=0.7584, P=0.9543,
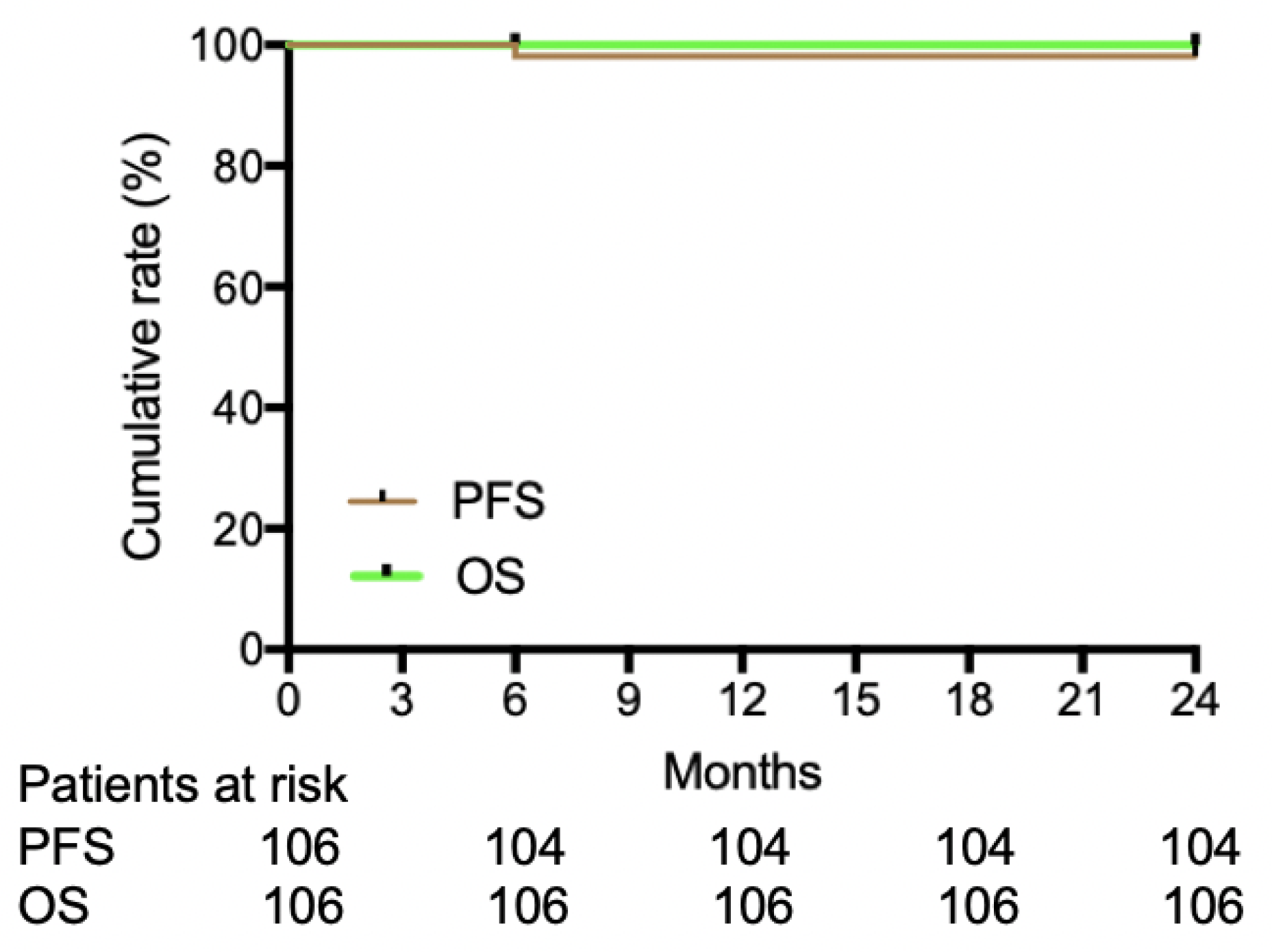
Figure 2). The cumulative incidence of PFS and OS were 98.1% (104/106) and 100% (106/106), respectively. The median PFS and OS in this trial both were not reached at 24 months (
Figure 3).
For the univariate analysis of subgroups, patients with intermediate or high ELTS score tended to achieve lower cumulative response of MR
4.5 by 18 months (HR=0.462 P=0.020; HR=0.387 P=0.042, respectively). Similarly, patients with spleen size below costal margin < 10cm tended to achieve higher cumulative response of MR
4.5 by 18 months than those with spleen size below costal margin ≥ 10cm (HR=1.927, P=0.021). However, there were no significant difference using multivariate analysis for nilotinib with cumulative response of MR
4.5 before 18 months treatment (
Table 2).
Safety analysis
The most common hematologic adverse events (AEs) were thrombocytopenia [46% (grade 3–4, 2%)], anemia [30% (grade 3–4, 4%)], leukopenia [14% (grade 3–4, 4%)] and neutropenia [13% (grade 3–4, 11%)]. The most common non-hematologic AEs were rash (70%), myalgia (48%), fatigue (41%), dry eyes (37.6%) and itching (36.8%). Most patients with non-hematologic AEs were at grade 1-2 and could recover lately. Besides, one patient gave up the treatment of nilotinib because of coronary heart disease, and one patient gave up the treatment of nilotinib due to high blood glucose and lipid (
Table 3).
Discussion
Early stable DMR of nilotinib is associated with the goal of TFR for patients with CML-CP. Therefore, it is significant to early distinguish patients who can obtain DMR and fit for TFR. Our prospective and multicenter study investigated the early cumulative MR4.5 rate by 18 months with nilotinib in patients with ND-CML-CP. The results of this trail showed that treatment of nilotinib was contributed to obtain high early MR4.5 for ND-CML-CP patients in real world.
From 29 institutes in China, the cumulative MR
4.5 rate by 18 months was 69.8% for patients with ND-CML-CP in this study, which was higher than the MR
4.5 achieved in 24-month follow-up of ENEST1st trial (50%) [
22] and 24-month follow-up of N-Road study (45.7%) [
23] and in the Michihide’s trial with a median observation period of 3.4 years (50%) [
24].Besides, the overall MR
4.5 rates in treatments of imatinib 400 mg daily, imatinib 800 mg daily, dasatinib 100 mg daily, and nilotinib 800 mg daily after a long follow-up time were 57%, 74%, 71%, and 71%, respectively [
25]. However, the cumulative MR
4.5 rate was no yet reported in the ENESTchina study with a 12-month follow-up. Frontline nilotinib 800mg daily with a median follow-up of 78.3 months and frontline dasatinib 100mg daily with a median follow-up of 6.5 year achieved MR
4.5 in ND-CML-CP were 75% and 88.2%, respectively [
26,
27]. In the future, it is significant to perform study of frontline nilotnib 300mg daily in ND-CML-CP with long time follow-up in China.
Similarly, for patients with ND-CML-CP, the cumulative MMR and MR
4.0 rates of nilotinib by 18 months in this trial were higher than that of ENEST1st study and N-Road study both with a 24-month follow-up (94.3% versus 80.4% versus 82.2%, and 84.9% versus 55.2% versus 58.3%, respectively) [
22,
23]. Besides, the cumulative MMR rates of nilotinib was 52.2% in the ENESTchina study with a 12-month follow-up [
28]. The highest MR
4.5, MR
4.0, MMR rate in our study may be relative to a high proportion of low ELTS score (67.9%, 72/106) and small body surface area (BSA) in Asian patients. Our findings suggest that nilotinib could achieve high early DMR for ND-CML-CP patients during TKI treatment.
DMR served as a milestone in TFR, and was described in ELN and LALNET recommendations and NCCN guideline [
14,
15,
16]. As TFR will be the new goal of CML-CP treatment in future [
29], u-EMR at 6 weeks might predict early achievement of DMR for ND-CML-CP treatment. In our study, 84.4% ND-CML-CP patients achieved u-EMR at 6 weeks, which indicated nilotinib could quickly reduce tumor load. The reasons may be that nilotinib has a high binding affinity and great selectivity for ABL kinase and high inhibitory activity [
9,
10]. Besides, Masahiro et. al’s study reported that 87.0% (328/377) ND-CML-CP patients with treatment of nilotinib achieved EMR at 3 months (BCR-ABL IS < 10%), which indicated a significantly superior PFS than those without EMR after 5-year follow-up (P < 0.0001) [
19]. In our study, patients with u-EMR at 6 weeks were not significantly different to achieve DMR or MMR by 24 months compared to those without u-EMR. Long-term follow-up of nilotinib need to be carried out to identify the relationship between the u-EMR and DMR or PFS.
No patient showed exacerbation to AP/BC, and the reasons maybe that nilotinib conquers the poor prognosis of intermediate or high-risk based on Euro score as presented in ENESTnd trial and DASISION trail [
13,
30]. The cumulative rate of PFS and OS in our study were 98.1% and 100%, which is similarly to the TARGET system’s study with 94.1% PFS and 97.1% OS after 5-year follow-up [
19]. Univariate analysis of subgroups, patients with intermediate or high ELTS score tended to achieve lower cumulative response of MR
4.5 by 18 months. Similarly, patients with spleen size below costal margin < 10cm tended to achieve higher cumulative response of MR
4.5 by 18 months than those with spleen size below costal margin ≥ 10cm. The reason maybe that intermediate or high ELTS score and the bigger size of spleen size are relative high tumor load for ND-CML-CP patients.
Some limitations were found in this trial. It was hard to analyzed subgroups with small number of participants. Besides, this study was designed as a single-arm clinical trial, which lacked of a comparator with other TKI treatment. Therefore, it is limited for the generalizability of results. Long-term follow-up of nilotinib and compared with other TKI treatment are required to explore in the future study in China.
Conclusions
This prospetive and multicentre study demonstrated that treatment with nilotinib in patients with newly diagnosed CML-CP contributed to obtain high early molecular response 4.5 (MR4.5).
Author Contributions
XD contributed to conception and design of the study. YZ, HL, JL, CT, YJ, XL, YC, HH, ZL, XX, WH and LP contributed to carry out this study and acquisition of data. BW performed analysis of data and drafted the manuscript. All authors contributed to interpretation of the results and revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by Shenzhen Key Medical Discipline (No. SZXK008).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments
The authors thank the patients who participated in this study and their families, as well as investigators and staffs.
Conflicts of Interest
The authors declare that they have no competing interests.
References
- Soverini S, de Benedittis C, Mancini M, et al. Mutations in the BCR-ABL1 Kinase Domain and Elsewhere in Chronic Myeloid Leukemia. Clinical lymphoma, myeloma & leukemia. 2015;15 Suppl: S120-128. [CrossRef]
- Garcia-Manero G, Faderl S, O'Brien S, et al. Chronic myelogenous leukemia: a review and update of therapeutic strategies. Cancer. 2003;98(3):437-457. [CrossRef]
- Jemal A, Tiwari RC, Murray T, et al. Cancer statistics, 2004. CA Cancer J Clin. 2004;54(1):8-29.
- Redaelli A, Bell C, Casagrande J, et al. Clinical and epidemiologic burden of chronic myelogenous leukemia. Expert review of anticancer therapy. 2004;4(1):85-96. [CrossRef]
- Baccarani M, Deininger MW, Rosti G, et al. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood. 2013;122(6):872-884. [CrossRef]
- Druker BJ, Guilhot F, O'Brien SG, et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. The New England journal of medicine. 2006;355(23):2408-2417. [CrossRef]
- Kantarjian H, Shah NP, Hochhaus A, et al. Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. The New England journal of medicine. 2010;362(24):2260-2270. [CrossRef]
- Saglio G, Kim DW, Issaragrisil S, et al. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. The New England journal of medicine. 2010;362(24):2251-2259. [CrossRef]
- Golemovic M, Verstovsek S, Giles F, et al. AMN107, a novel aminopyrimidine inhibitor of Bcr-Abl, has in vitro activity against imatinib-resistant chronic myeloid leukemia. Clinical cancer research: an official journal of the American Association for Cancer Research. 2005;11(13):4941-4947.
- O'Hare T, Walters DK, Stoffregen EP, et al. In vitro activity of Bcr-Abl inhibitors AMN107 and BMS-354825 against clinically relevant imatinib-resistant Abl kinase domain mutants. Cancer Res. 2005;65(11):4500-4505. [CrossRef]
- Jabbour E, Kantarjian H. Chronic myeloid leukemia: 2022 update on diagnosis, therapy, and monitoring. American journal of hematology. 2022;97(9):1236-1256. [CrossRef]
- Hochhaus A, Saglio G, Hughes TP, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30(5):1044-1054. [CrossRef]
- Kantarjian HM, Hughes TP, Larson RA, et al. Long-term outcomes with frontline nilotinib versus imatinib in newly diagnosed chronic myeloid leukemia in chronic phase: ENESTnd 10-year analysis. Leukemia. 2021;35(2):440-453. [CrossRef]
- Pavlovsky C, Abello Polo V, Pagnano K, et al. Treatment-free remission in patients with chronic myeloid leukemia: recommendations of the LALNET expert panel. Blood advances. 2021;5(23):4855-4863. [CrossRef]
- Chronic Myeloid Leukemia. NCCN Clinical Practice Guidelines in Oncology. 2022; Version 1.2023.
- Jain P, Kantarjian H, Nazha A, et al. Early responses predict better outcomes in patients with newly diagnosed chronic myeloid leukemia: results with four tyrosine kinase inhibitor modalities. Blood. 2013;121(24):4867-4874. [CrossRef]
- Hanfstein B, Müller MC, Hehlmann R, et al. Early molecular and cytogenetic response is predictive for long-term progression-free and overall survival in chronic myeloid leukemia (CML). Leukemia. 2012;26(9):2096-2102. [CrossRef]
- Jabbour E, Kantarjian HM, Saglio G, et al. Early response with dasatinib or imatinib in chronic myeloid leukemia: 3-year follow-up from a randomized phase 3 trial (DASISION). Blood. 2014;123(4):494-500. [CrossRef]
- Kizaki M, Takahashi N, Iriyama N, et al. Efficacy and safety of tyrosine kinase inhibitors for newly diagnosed chronic-phase chronic myeloid leukemia over a 5-year period: results from the Japanese registry obtained by the New TARGET system. International journal of hematology. 2019;109(4):426-439. [CrossRef]
- Branford S, Yeung DT, Ross DM, et al. Early molecular response and female sex strongly predict stable undetectable BCR-ABL1, the criteria for imatinib discontinuation in patients with CML. Blood. 2013;121(19):3818-3824. [CrossRef]
- Marin D, Ibrahim AR, Lucas C, et al. Assessment of BCR-ABL1 transcript levels at 3 months is the only requirement for predicting outcome for patients with chronic myeloid leukemia treated with tyrosine kinase inhibitors. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2012;30(3):232-238. [CrossRef]
- Hochhaus A, Mahon FX, le Coutre P, et al. Nilotinib first-line therapy in patients with Philadelphia chromosome-negative/BCR-ABL-positive chronic myeloid leukemia in chronic phase: ENEST1st sub-analysis. Journal of cancer research and clinical oncology. 2017;143(7):1225-1233. [CrossRef]
- Nishiwaki K, Sugimoto KJ, Tamaki S, et al. Optimal treatment strategy with nilotinib for patients with newly diagnosed chronic-phase chronic myeloid leukemia based on early achievement of deep molecular response (MR(4.5) ): The phase 2, multicenter N-Road study. Cancer medicine. 2020;9(11):3742-3751.
- Tokuhira M, Kimura Y, Sugimoto K, et al. Efficacy and safety of nilotinib therapy in patients with newly diagnosed chronic myeloid leukemia in the chronic phase. Medical oncology (Northwood, London, England). 2018;35(3):38. [CrossRef]
- Jabbour E, Kantarjian H. Chronic myeloid leukemia: 2020 update on diagnosis, therapy and monitoring. American journal of hematology. 2020;95(6):691-709. [CrossRef]
- Masarova L, Cortes JE, Patel KP, et al. Long-term results of a phase 2 trial of nilotinib 400 mg twice daily in newly diagnosed patients with chronic-phase chronic myeloid leukemia. Cancer. 2020;126(7):1448-1459. [CrossRef]
- Maiti A, Cortes JE, Patel KP, et al. Long-term results of frontline dasatinib in chronic myeloid leukemia. Cancer. 2020;126(7):1502-1511. [CrossRef]
- Wang J, Shen ZX, Saglio G, et al. Phase 3 study of nilotinib vs imatinib in Chinese patients with newly diagnosed chronic myeloid leukemia in chronic phase: ENESTchina. Blood. 2015;125(18):2771-2778. [CrossRef]
- Hughes TP, Ross DM. Moving treatment-free remission into mainstream clinical practice in CML. Blood. 2016;128(1):17-23. [CrossRef]
- Cortes JE, Saglio G, Kantarjian HM, et al. Final 5-Year Study Results of DASISION: The Dasatinib Versus Imatinib Study in Treatment-Naïve Chronic Myeloid Leukemia Patients Trial. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2016;34(20):2333-2340. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).