Submitted:
05 June 2023
Posted:
05 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Transgenic mice
2.2. Recombinant RLC proteins – cloning, expression and purification
2.3. Mechanical and ATP turnover measurements in skinned left ventricular papillary muscle (LVPM) fibers from transgenic Tg-RLC and Tg-ELC mice
2.3.1. Preparation of skinned LVPM fibers
2.3.2. Depletion of endogenous RLC from LVPM and reconstitution with recombinant RLC proteins
2.3.3. Steady-state force measurements and Ca2+ dependence of force development in Tg skinned muscle strips
2.3.4. Mant-ATP chase experiments
2.4. Treatment of skinned muscle strips with Omecamtiv Mecarbil (OM)
2.5. Statistical Analysis
3. Results
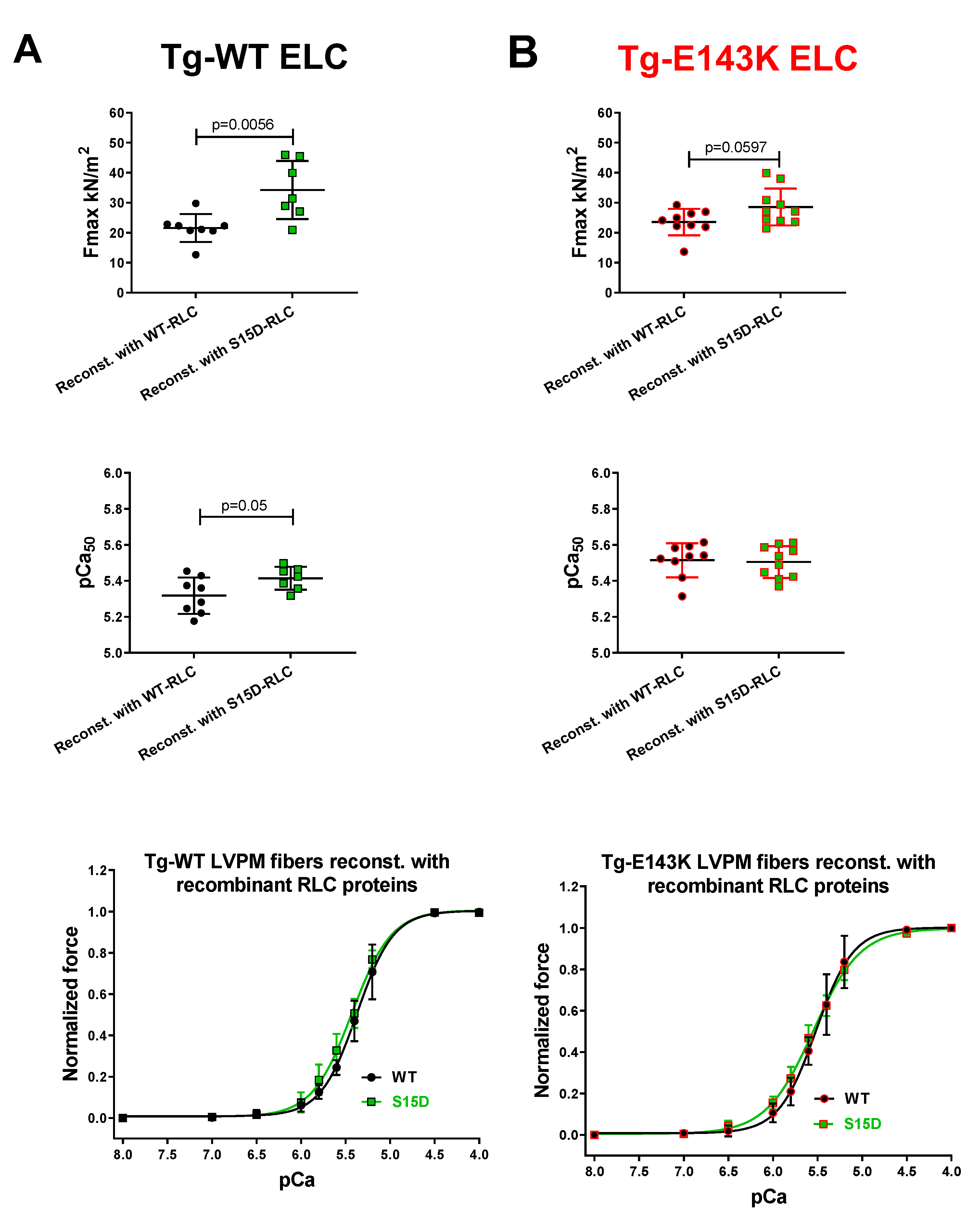
3.1. Effect of phosphomimetic S15D-RLC on force measurements in LVPM fibers from Tg-ELC mouse model of RCM
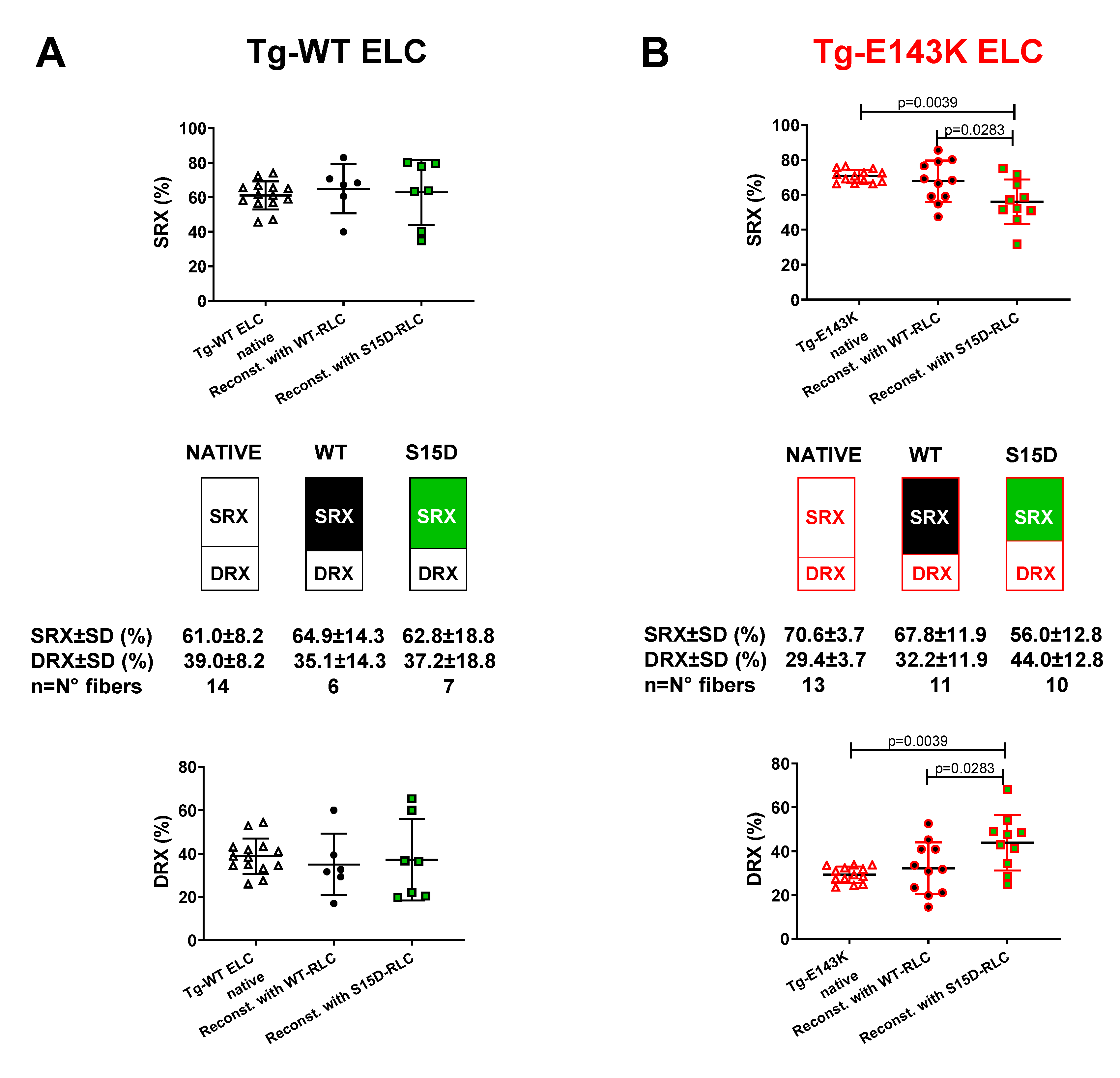
3.2. Effect of phosphomimetic S15D-RLC on super-relaxed state in LVPM fibers from Tg-ELC and Tg-RLC mouse modes of RCM and DCM.
3.2.1. Transgenic ELC mice
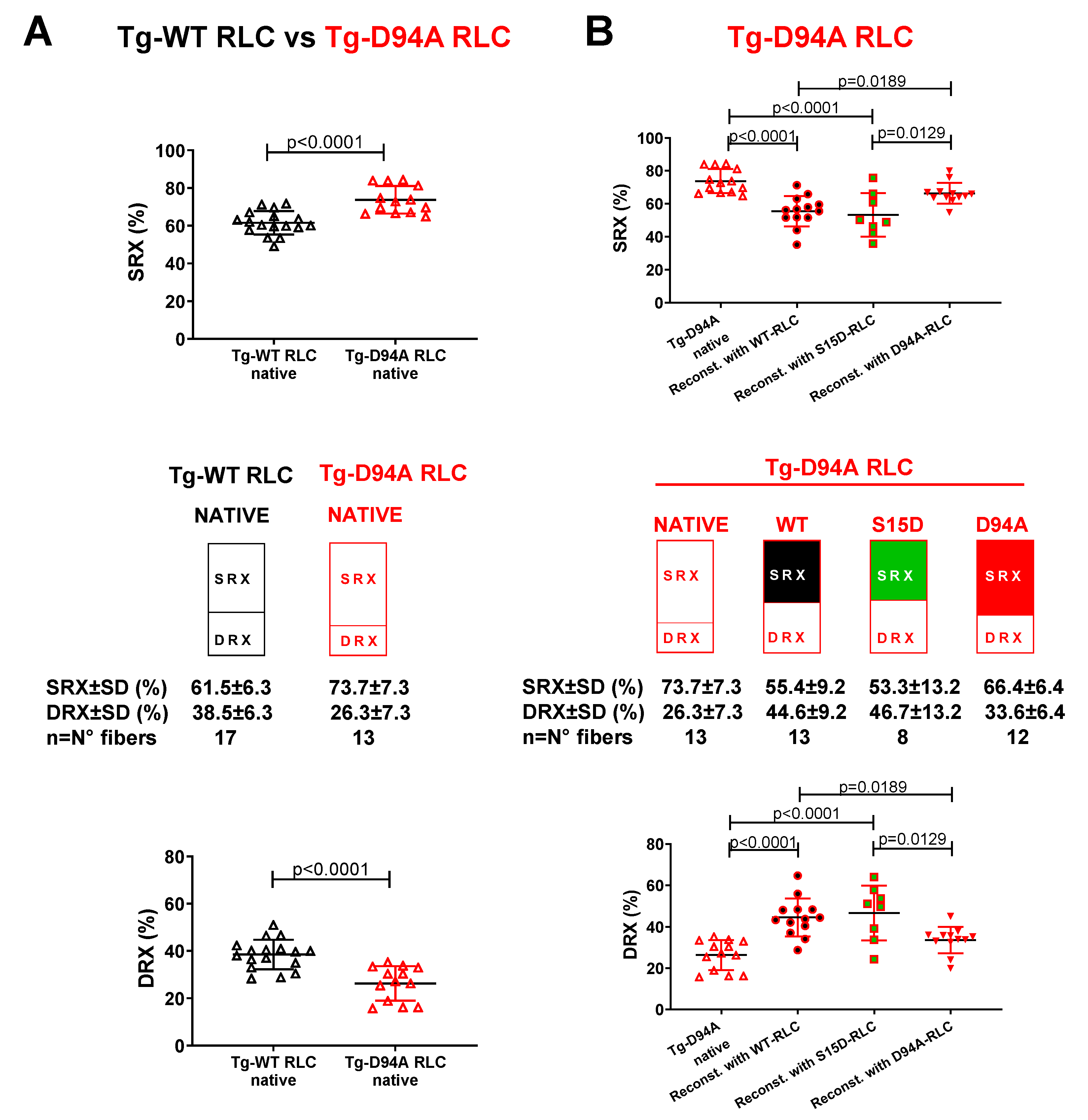
3.2.2. Transgenic RLC mice
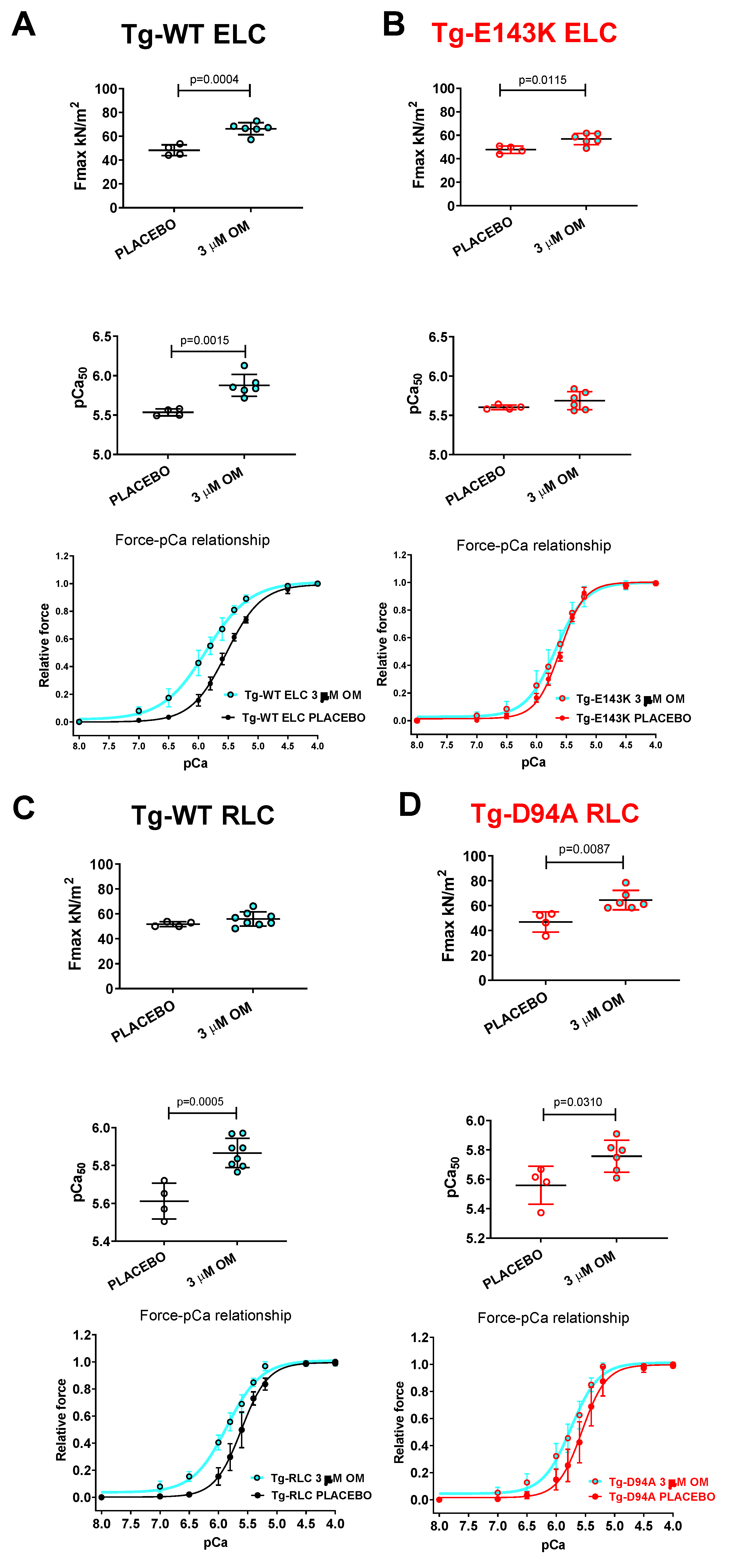
3.3. Treatment of RCM and DCM mice with Omecamtiv Mecarbil (OM).
3.3.1. Steady-state force and force-pCa relationship
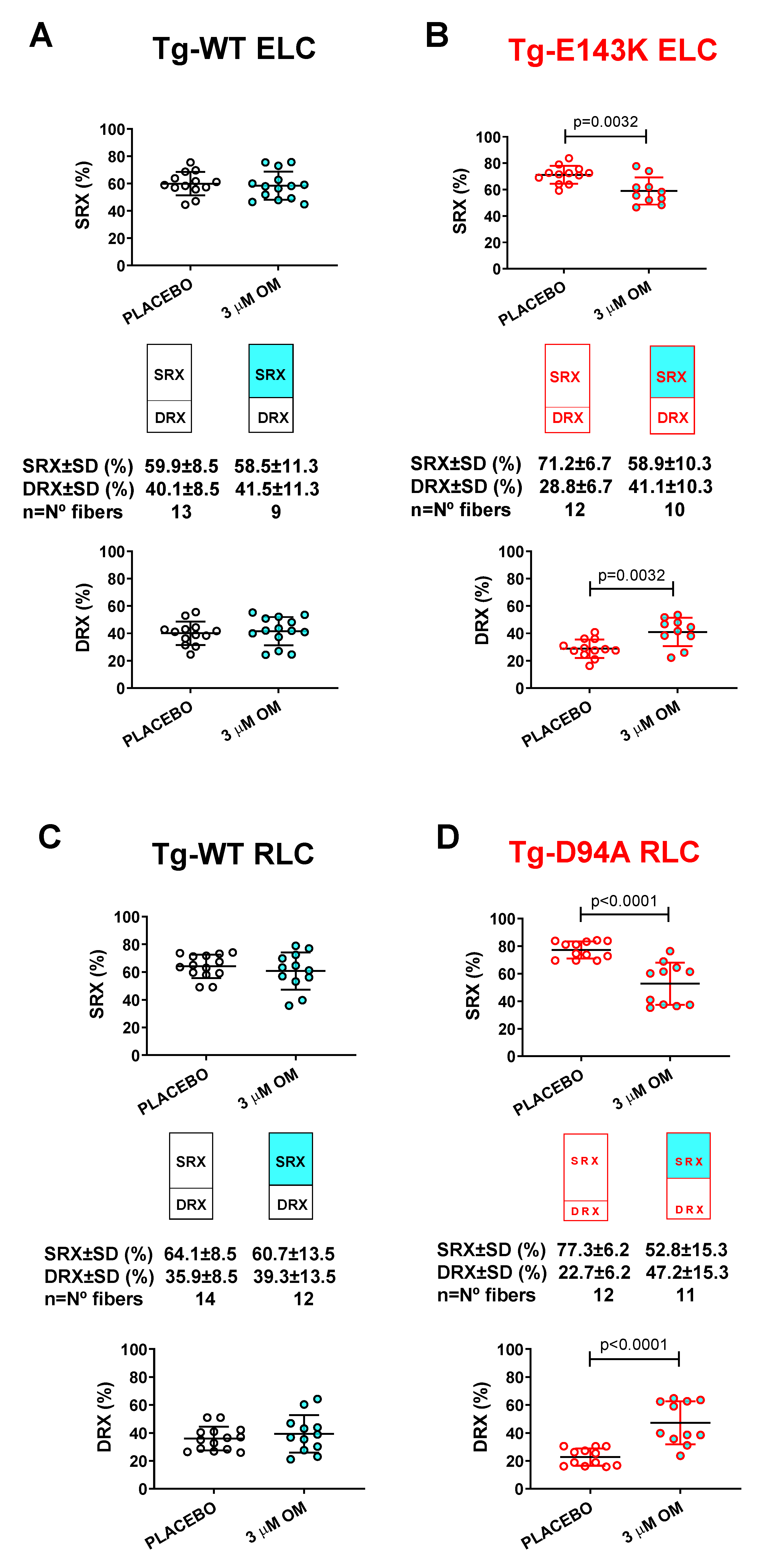
3.3.2. SRX↔DRX equilibrium in LVPM fibers from RCM and DCM mice
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Geeves, M. A.; Holmes, K. C. , The molecular mechanism of muscle contraction. Adv Protein Chem 2005, 71, 161–93. [Google Scholar] [CrossRef]
- McKillop, D.; Geeves, M. Regulation of the interaction between actin and myosin subfragment 1: evidence for three states of the thin filament. Biophys. J. 1993, 65, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Brunello, E.; Fusi, L.; Ghisleni, A.; Park-Holohan, S.-J.; Ovejero, J.G.; Narayanan, T.; Irving, M. Myosin filament-based regulation of the dynamics of contraction in heart muscle. Proc. Natl. Acad. Sci. USA 2020, 117, 8177–8186. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yuan, C.-C.; Kazmierczak, K.; Szczesna-Cordary, D.; Burghardt, T.P. Single cardiac ventricular myosins are autonomous motors. Open Biol. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, J.P.; Debold, E.P.; Ahmad, F.; Armstrong, A.; Frederico, A.; Conner, D.A.; Mende, U.; Lohse, M.J.; Warshaw, D.; Seidman, C.E.; et al. Cardiac myosin missense mutations cause dilated cardiomyopathy in mouse models and depress molecular motor function. Proc. Natl. Acad. Sci. 2006, 103, 14525–14530. [Google Scholar] [CrossRef] [PubMed]
- Alcalai, R.; Seidman, J.G.; Seidman, C.E. Genetic Basis of Hypertrophic Cardiomyopathy: From Bench to the Clinics. J. Cardiovasc. Electrophysiol. 2008, 19, 104–110. [Google Scholar] [CrossRef]
- Burghardt, T.P.; Sikkink, L.A. Regulatory Light Chain Mutants Linked to Heart Disease Modify the Cardiac Myosin Lever Arm. Biochemistry 2013, 52, 1249–1259. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Liang, J.; Yuan, C.-C.; Kazmierczak, K.; Zhou, Z.; Morales, A.; McBride, K.L.; Fitzgerald-Butt, S.M.; Hershberger, R.E.; Szczesna-Cordary, D. Novel familial dilated cardiomyopathy mutation inMYL2affects the structure and function of myosin regulatory light chain. FEBS J. 2015, 282, 2379–2393. [Google Scholar] [CrossRef]
- Huang, W.; Szczesna-Cordary, D. Molecular mechanisms of cardiomyopathy phenotypes associated with myosin light chain mutations. J. Muscle Res. Cell Motil. 2015, 36, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Muthu, P.; Kazmierczak, K.; Jones, M.; Szczesna-Cordary, D. The effect of myosin RLC phosphorylation in normal and cardiomyopathic mouse hearts. J. Cell. Mol. Med. 2012, 16, 911–919. [Google Scholar] [CrossRef]
- Yuan, C.-C.; Muthu, P.; Kazmierczak, K.; Liang, J.; Huang, W.; Irving, T.C.; Kanashiro-Takeuchi, R.M.; Hare, J.M.; Szczesna-Cordary, D. Constitutive phosphorylation of cardiac myosin regulatory light chain prevents development of hypertrophic cardiomyopathy in mice. Proc. Natl. Acad. Sci. USA 2015, 112, E4138–E4146. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Kazmierczak, K.; Liang, J.; Sitbon, Y.H.; Szczesna-Cordary, D. Phosphomimetic-mediated in vitro rescue of hypertrophic cardiomyopathy linked to R58Q mutation in myosin regulatory light chain. FEBS J. 2018, 286, 151–168. [Google Scholar] [CrossRef] [PubMed]
- Sitbon, Y.H.; Kazmierczak, K.; Liang, J.; Yadav, S.; Veerasammy, M.; Kanashiro-Takeuchi, R.M.; Szczesna-Cordary, D. Ablation of the N terminus of cardiac essential light chain promotes the super-relaxed state of myosin and counteracts hypercontractility in hypertrophic cardiomyopathy mutant mice. FEBS J. 2020, 287, 3989–4004. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, H.L.; Bowman, B.F.; Stull, J.T. Myosin light chain phosphorylation in vertebrate striated muscle: regulation and function. Am. J. Physiol. Physiol. 1993, 264, C1085–C1095. [Google Scholar] [CrossRef] [PubMed]
- E Kamm, K.; Stull, J.T. The Function of Myosin and Myosin Light Chain Kinase Phosphorylation in Smooth Muscle. Annu. Rev. Pharmacol. Toxicol. 1985, 25, 593–620. [Google Scholar] [CrossRef]
- Kamm, K.E.; Stull, J.T. Signaling to Myosin Regulatory Light Chain in Sarcomeres. J. Biol. Chem. 2011, 286, 9941–9947. [Google Scholar] [CrossRef]
- Seguchi, O.; Takashima, S.; Yamazaki, S.; Asakura, M.; Asano, Y.; Shintani, Y.; Wakeno, M.; Minamino, T.; Kondo, H.; Furukawa, H.; et al. A cardiac myosin light chain kinase regulates sarcomere assembly in the vertebrate heart. J. Clin. Investig. 2007, 117, 2812–2824. [Google Scholar] [CrossRef]
- Terry, M.; Walker, D.D.; Ferrari, M.B. Protein Phosphatase Activity Is Necessary for Myofibrillogenesis. Cell Biochem. Biophys. 2006, 45, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.N.; Battiprolu, P.K.; Cowley, P.M.; Chen, G.; Gerard, R.D.; Pinto, J.R.; Hill, J.A.; Baker, A.J.; Kamm, K.E.; Stull, J.T. Constitutive Phosphorylation of Cardiac Myosin Regulatory Light Chain in Vivo. J. Biol. Chem. 2015, 290, 10703–10716. [Google Scholar] [CrossRef]
- Kazmierczak, K.; Liang, J.; Gomez-Guevara, M.; Szczesna-Cordary, D. Functional comparison of phosphomimetic S15D and T160D mutants of myosin regulatory light chain exchanged in cardiac muscle preparations of HCM and WT mice. Front. Cardiovasc. Med. 2022, 9, 988066. [Google Scholar] [CrossRef]
- Yadav, S.; Yuan, C.-C.; Kazmierczak, K.; Liang, J.; Huang, W.; Takeuchi, L.M.; Kanashiro-Takeuchi, R.M.; Szczesna-Cordary, D. Therapeutic potential of AAV9-S15D-RLC gene delivery in humanized MYL2 mouse model of HCM. J. Mol. Med. 2019, 97, 1033–1047. [Google Scholar] [CrossRef]
- Mamidi, R.; Li, J.; Gresham Kenneth, S.; Verma, S.; Doh Chang, Y.; Li, A.; Lal, S.; dos Remedios Cristobal, G.; Stelzer Julian, E. , Dose-Dependent Effects of the Myosin Activator Omecamtiv Mecarbil on Cross-Bridge Behavior and Force Generation in Failing Human Myocardium. Circulation: Heart Failure 2017, 10, (10), e004257. [Google Scholar] [CrossRef]
- Teerlink, J. R.; Felker, G. M.; McMurray, J. J.; Ponikowski, P.; Metra, M.; Filippatos, G. S.; Ezekowitz, J. A.; Dickstein, K.; Cleland, J. G.; Kim, J. B.; Lei, L.; Knusel, B.; Wolff, A. A.; Malik, F. I.; Wasserman, S. M.; Investigators, A.-A. , Acute Treatment With Omecamtiv Mecarbil to Increase Contractility in Acute Heart Failure: The ATOMIC-AHF Study. J Am Coll Cardiol 2016, 67, (12), 1444–55. [Google Scholar] [CrossRef]
- Yuan, C.-C.; Kazmierczak, K.; Liang, J.; Zhou, Z.; Yadav, S.; Gomes, A.V.; Irving, T.C.; Szczesna-Cordary, D. Sarcomeric perturbations of myosin motors lead to dilated cardiomyopathy in genetically modified MYL2 mice. Proc. Natl. Acad. Sci. USA 2018, 115, E2338–E2347. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.-C.; Kazmierczak, K.; Liang, J.; Kanashiro-Takeuchi, R.; Irving, T.C.; Gomes, A.V.; Wang, Y.; Burghardt, T.P.; Szczesna-Cordary, D. Hypercontractile mutant of ventricular myosin essential light chain leads to disruption of sarcomeric structure and function and results in restrictive cardiomyopathy in mice. Cardiovasc. Res. 2017, 113, 1124–1136. [Google Scholar] [CrossRef] [PubMed]
- Sitbon, Y.H.; Diaz, F.; Kazmierczak, K.; Liang, J.; Wangpaichitr, M.; Szczesna-Cordary, D. Cardiomyopathic mutations in essential light chain reveal mechanisms regulating the super relaxed state of myosin. J. Gen. Physiol. 2021, 153. [Google Scholar] [CrossRef] [PubMed]
- Szczesna, D.; Ghosh, D.; Li, Q.; Gomes, A.V.; Guzman, G.; Arana, C.; Zhi, G.; Stull, J.T.; Potter, J.D. Familial Hypertrophic Cardiomyopathy Mutations in the Regulatory Light Chains of Myosin Affect Their Structure, Ca2+Binding, and Phosphorylation. J. Biol. Chem. 2001, 276, 7086–7092. [Google Scholar] [CrossRef] [PubMed]
- Muthu, P.; Liang, J.; Schmidt, W.; Moore, J.R.; Szczesna-Cordary, D. In vitro rescue study of a malignant familial hypertrophic cardiomyopathy phenotype by pseudo-phosphorylation of myosin regulatory light chain. Arch. Biochem. Biophys. 2013, 552-553, 29–39. [Google Scholar] [CrossRef]
- Pant, K.; Watt, J.; Greenberg, M.; Jones, M.; Szczesna-Cordary, D.; Moore, J.R. Removal of the cardiac myosin regulatory light chain increases isometric force production. FASEB J. 2009, 23, 3571–3580. [Google Scholar] [CrossRef]
- Yuan, C. C.; Kazmierczak, K.; Liang, J.; Ma, W.; Irving, T. C.; Szczesna-Cordary, D. , Molecular basis of force-pCa relation in MYL2 cardiomyopathy mice: Role of the super-relaxed state of myosin. Proc Natl Acad Sci U S A 2022, 119, (8). [Google Scholar] [CrossRef]
- Hooijman, P.; Stewart, M. A.; Cooke, R. , A new state of cardiac Myosin with very slow ATP turnover: a potential cardioprotective mechanism in the heart. Biophys J 2011, 100, (8), 1969–76. [Google Scholar] [CrossRef]
- Szczesna-Cordary, D.; Guzman, G.; Ng, S.-S.; Zhao, J. Familial Hypertrophic Cardiomyopathy-linked Alterations in Ca2+ Binding of Human Cardiac Myosin Regulatory Light Chain Affect Cardiac Muscle Contraction. J. Biol. Chem. 2004, 279, 3535–3542. [Google Scholar] [CrossRef] [PubMed]
- Muthu, P.; Huang, W.; Kazmierczak, K.; Szczesna-Cordary, D., Functional Consequences of Mutations in the Myosin Regulatory Light Chain Associated with Hypertrophic Cardiomyopathy. In: Veselka J (Ed.) 2012, Cardiomyopathies – From Basic Research to Clinical Management. Ch. 17. InTech, Croatia, pp 383-408.
- Greenberg, M.J.; Kazmierczak, K.; Szczesna-Cordary, D.; Moore, J.R. Cardiomyopathy-linked myosin regulatory light chain mutations disrupt myosin strain-dependent biochemistry. Proc. Natl. Acad. Sci. USA 2010, 107, 17403–17408. [Google Scholar] [CrossRef]
- Pulcastro, H.C.; Awinda, P.O.; Breithaupt, J.J.; Tanner, B.C. Effects of myosin light chain phosphorylation on length-dependent myosin kinetics in skinned rat myocardium. Arch. Biochem. Biophys. 2016, 601, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Alamo, L.; Ware, J. S.; Pinto, A.; Gillilan, R. E.; Seidman, J. G.; Seidman, C. E.; Padron, R. , Effects of myosin variants on interacting-heads motif explain distinct hypertrophic and dilated cardiomyopathy phenotypes. Elife 2017, 6. [Google Scholar] [CrossRef]
- Woodhead, J.L.; Zhao, F.-Q.; Craig, R.; Egelman, E.H.; Alamo, L.; Padrón, R. Atomic model of a myosin filament in the relaxed state. Nature 2005, 436, 1195–1199. [Google Scholar] [CrossRef]
- Malik, F.I.; Hartman, J.J.; Elias, K.A.; Morgan, B.P.; Rodriguez, H.; Brejc, K.; Anderson, R.L.; Sueoka, S.H.; Lee, K.H.; Finer, J.T.; et al. Cardiac Myosin Activation: A Potential Therapeutic Approach for Systolic Heart Failure. Science 2011, 331, 1439–1443. [Google Scholar] [CrossRef]
- Teerlink, J. R.; Diaz, R.; Felker, G. M.; McMurray, J. J. V.; Metra, M.; Solomon, S. D.; Adams, K. F.; Anand, I.; Arias-Mendoza, A.; Biering-Sørensen, T.; Böhm, M.; Bonderman, D.; Cleland, J. G. F.; Corbalan, R.; Crespo-Leiro, M. G.; Dahlström, U.; Echeverria, L. E.; Fang, J. C.; Filippatos, G.; Fonseca, C.; Goncalvesova, E.; Goudev, A. R.; Howlett, J. G.; Lanfear, D. E.; Li, J.; Lund, M.; Macdonald, P.; Mareev, V.; Momomura, S. I.; O'Meara, E.; Parkhomenko, A.; Ponikowski, P.; Ramires, F. J. A.; Serpytis, P.; Sliwa, K.; Spinar, J.; Suter, T. M.; Tomcsanyi, J.; Vandekerckhove, H.; Vinereanu, D.; Voors, A. A.; Yilmaz, M. B.; Zannad, F.; Sharpsten, L.; Legg, J. C.; Varin, C.; Honarpour, N.; Abbasi, S. A.; Malik, F. I.; Kurtz, C. E. , Cardiac Myosin Activation with Omecamtiv Mecarbil in Systolic Heart Failure. N Engl J Med 2021, 384, (2), 105–116. [Google Scholar] [CrossRef]
- Woody, M.S.; Greenberg, M.J.; Barua, B.; Winkelmann, D.A.; Goldman, Y.E.; Ostap, E.M. Positive cardiac inotrope omecamtiv mecarbil activates muscle despite suppressing the myosin working stroke. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef]
- Mamidi, R.; Holmes, J.B.; Doh, C.Y.; Dominic, K.L.; Madugula, N.; Stelzer, J.E. cMyBPC phosphorylation modulates the effect of omecamtiv mecarbil on myocardial force generation. J. Gen. Physiol. 2021, 153. [Google Scholar] [CrossRef] [PubMed]
- Abraham, T.P.; Jones, M.; Kazmierczak, K.; Liang, H.-Y.; Pinheiro, A.C.; Wagg, C.S.; Lopaschuk, G.D.; Szczesna-Cordary, D. Diastolic dysfunction in familial hypertrophic cardiomyopathy transgenic model mice. Cardiovasc. Res. 2009, 82, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Kazmierczak, K.; Xu, Y.; Jones, M.; Guzman, G.; Hernandez, O.M.; Kerrick, W.G.L.; Szczesna-Cordary, D. The Role of the N-Terminus of the Myosin Essential Light Chain in Cardiac Muscle Contraction. J. Mol. Biol. 2009, 387, 706–725. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ajtai, K.; Burghardt, T.P. Ventricular myosin modifies in vitro step-size when phosphorylated. J. Mol. Cell. Cardiol. 2014, 72, 231–237. [Google Scholar] [CrossRef]
- Yu, H.; Chakravorty, S.; Song, W.; Ferenczi, M.A. Phosphorylation of the regulatory light chain of myosin in striated muscle: methodological perspectives. Eur. Biophys. J. 2016, 45, 779–805. [Google Scholar] [CrossRef]
- Toepfer, C.N.; Garfinkel, A.C.; Venturini, G.; Wakimoto, H.; Repetti, G.; Alamo, L.; Sharma, A.; Agarwal, R.; Ewoldt, J.K.; Cloonan, P.; et al. Myosin Sequestration Regulates Sarcomere Function, Cardiomyocyte Energetics, and Metabolism, Informing the Pathogenesis of Hypertrophic Cardiomyopathy. Circ. 2020, 141, 828–842. [Google Scholar] [CrossRef]
| Force parameter /recombinant RLC-reconstituted LVPM |
LVPM from Tg-WT ELC mice | LVPM from Tg-E143K mice | ||
|---|---|---|---|---|
| WT | S15D | WT | S15D | |
| N° fibers | 8 | 7 | 9 | 10 |
| Fmax (kN/m2) ± SD | 21.56±4.64 | 34.25±9.66** | 23.56±4.42 | 28.58±1.95 |
| pCa50 ± SD | 5.32±0.1 | 5.41±0.06* | 5.51±0.09 | 5.51±0.09 |
| nH ± SD | 2.03±0.74 | 1.87±0.41 | 2.2±0.61 | 1.73±0.24* |
| Scheme . | LVPM from Tg-WT ELC mice | LVPM from Tg-E143K mice | ||||
|---|---|---|---|---|---|---|
| Native | WT | S15D | Native | WT | S15D | |
| N° fibers | 14 | 6 | 7 | 13 | 11 | 10 |
| DRX (%) ± SD | 38.9±8.2 | 35.1±14.2 | 37.2±18.8 | 29.4±3.7## | 32.3±11.9 | 44±12.8* |
| SRX (%) ± SD | 61.1±8.2 | 64.9±14.2 | 62.8±18.8 | 70.6±3.7## | 67.8±11.9 | 56±12.8* |
| T1 (s) ± SD | 4.4±2.4 | 6.6±4.9 | 5.3±2.5 | 4.2±2.5 | 4.8±3.6 | 7±5.2 |
| T2 (s) ± SD | 129.8±67 | 125.3±81.3 | 173.4±167.7 | 126.5±81.2 | 92.2±64 | 136.4±116.5 |
| SRX parameter / recombinant RLC-reconstituted LVPM | LVPM from Tg-D94A mice | |||
|---|---|---|---|---|
| Native | WT | S15D | D94A | |
| No. fibers | 13 | 13 | 8 | 12 |
| DRX±SD | 26.3±7.3 | 44.9±10.2****, ^ | 46.7±13.2****, ^ | 33.6±6.4 |
| SRX±SD | 73.7±7.3 | 55.1±10.2****, ^ | 53.3±13.2****, ^ | 66.4±6.4 |
| T1±SD | 6.5±3.3 | 4.9±2.8 | 5.7±5.1 | 2.7±1.7* |
| T2±SD | 161.8±149 | 136.9±103.1 | 117.7±130.8 | 67.4±77.3 |
| Force parameter // OM vs placebo treated fibers | LVPM from Tg-WT ELC mice | LVPM from Tg-E143K mice | LVPM from Tg-WT RLC mice | LVPM from Tg-D94A mice | ||||
|---|---|---|---|---|---|---|---|---|
| OM | placebo | OM | placebo | OM | placebo | OM | placebo | |
| No. fibers | 6 | 4 | 6 | 4 | 8 | 4 | 6 | 4 |
| Fmax (kN/m2) ± SD | 66.4±5.1*** | 48.3±4.5 | 56.8±4.8* | 47.8±3.2 | 55.8±5.7 | 51.7±2 | 64.5±7.8** | 46.9±8.1 |
| pCa50 ± SD | 5.9±0.1** | 5.5±0.04 | 5.7±0.1 | 5.6±0.03 | 5.9±0.1*** | 5.6±0.1 | 5.8±0.1* | 5.6±0.1 |
| nH ± SD | 1.2±0.1* | 1.5±0.2 | 1.8±0.2* | 2.2±0.1 | 1.5±0.2** | 2±0.2 | 1.9±0.3* | 2.2±0.2 |
| SRX parameter // OM vs placebo treated fibers | LVPM from Tg-WT ELC mice | LVPM from Tg-E143K mice | LVPM from Tg-WT RLC mice | LVPM from Tg-D94A mice | ||||
|---|---|---|---|---|---|---|---|---|
| OM | placebo | OM | placebo | OM | placebo | OM | placebo | |
| No. fibers | 14 | 13 | 10 | 12 | 12 | 14 | 11 | 12 |
| DRX±SD | 41.6±10.4 | 40.1±8.5 | 41.1±10.3** | 28.8±6.7 | 39.3±13.4 | 35.9±8.5 | 47.2±15.3**** | 22.7±6.2 |
| SRX±SD | 58.4±10.4 | 59.9±8.5 | 58.9±10.3** | 71.2±6.7 | 60.7±13.4 | 64.1±8.5 | 52.8±15.3**** | 77.3±6.2 |
| T1±SD | 3.7±3.4 | 3.2±1.8 | 3.7±2.4 | 4.2±2.7 | 4.2±3 | 5.6±3.8 | 3.7±1.9* | 6.9±4.1 |
| T2±SD | 101.4±152.5 | 69±38.5 | 89.3±78.9 | 112.9±94.3 | 123.8±86.8 | 119.6±122.4 | 83.1±65.6* | 230.6±213.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




