Submitted:
31 May 2023
Posted:
01 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Collection, Transportation, and Storage of Brown Alga P. boergesenii
2.2. FTIR Characterization for Functional Groups Analysis
2.3. Characterization of Phytocompounds by GCMS Analysis
2.4. Fatty Acids Characterization by GCMS Analysis
2.5. Characterization of Phytocompounds by HRLCMS Q-TOF
2.6. Measurements of Different Amino Acids by HRLCMS
2.7. Element Analysis by ICP-AES Technique
2.7.1. Digestion and Detection
2.7.2. Digestion and Detection
2.8. Determination of Pigments content
2.8.1. Quantification of Chlorophylls, Carotenoids, Fucoxanthin, Phycoerythrin, and Phycocyanin


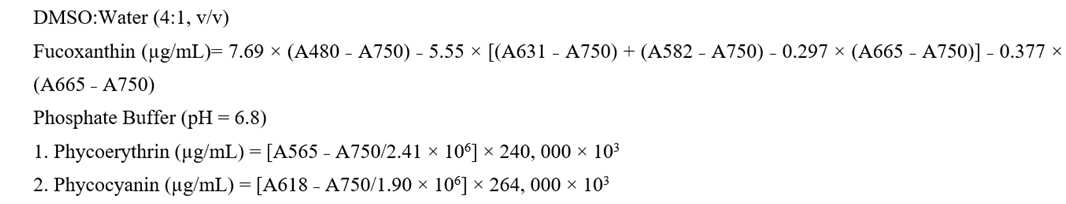
2.8.2. Estimation of Chlorophylls and Lycopene

2.8.3. Estimation of Chlorophylls and Total Carotenoids

2.9. Analysis of Total Polyphenol Content
2.10. Estimation of Total Protein
2.11. In vitro Antioxidant Analysis using the DPPH Method
2.12. Tyrosinase Inhibition Assay
3. Results and Discussion
3.1. Collection of Brown Alga P. boergesenii
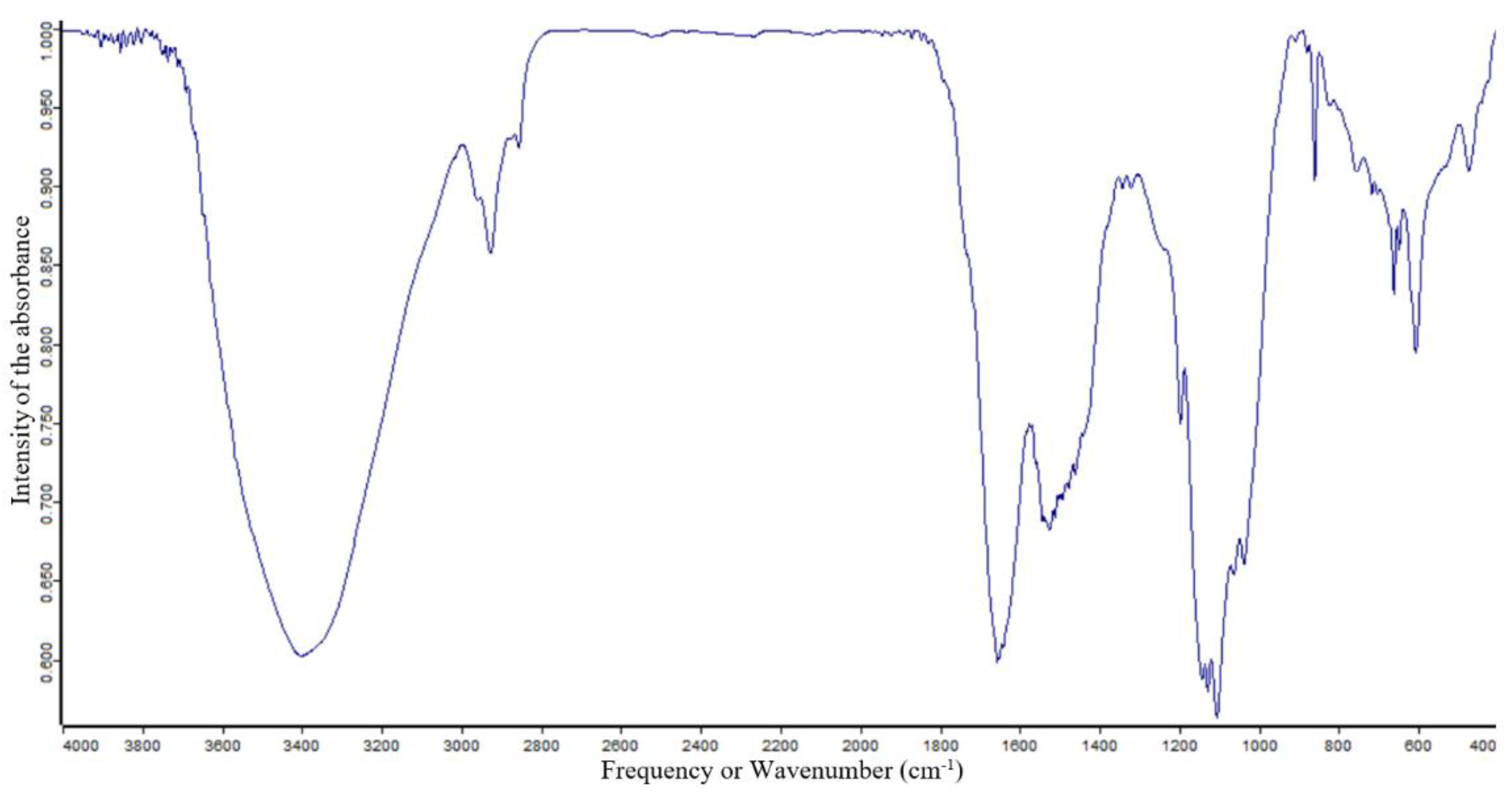
3.2. FTIR Characterization for Functional Groups Analysis
| Frequency (cm−1) | Intensity | Band assignments | Functional group |
|---|---|---|---|
| 604.94 | S | C-Cl stretch | Halo compound |
| S | C-Br stretch | Halo compound | |
| 659.08 | S | C-Br stretch | Halo compound |
| S | C-Cl stretch | Halo compound | |
| 751.88 | S | C-Cl stretch | Halo compound |
| S | C-H bend | 1,2-Disubstituted | |
| S | C-H bend | Monosubstituted | |
| 1038.02 | S | S=O stretch | Sulfoxide |
| S | C-F stretch | Fluoro compound | |
| M | C-N stretch | Amine | |
| 1099.89 | S | C-F starch | Fluoro compound |
| S | C-O starch | Aliphatic ether | |
| S | C-O starch | Secondary alcohol | |
| M | C-N stretch | Amine | |
| 1126.96 | S | C-F stretch | Fluoro compound |
| S | C-O stretch | Tertiary alcohol | |
| S | C-O stretch | Aliphatic ether | |
| M | C-N starch | Amine | |
| 1196.56 | S | C-F stretch | Fluoro compound |
| S | C-O stretch | Tertiary alcohol | |
| S | C-O stretch | Ester | |
| M | C-N stretch | Amine | |
| 1320.30 | S | C-F stretch | Fluoro compound |
| S | C-N stretch | Aromatic amine | |
| S | S=O stretch | Sulfone | |
| M | O-H bend | Phenol | |
| 1343.50 | S | C-F Stretch | Fluoro compound |
| S | S=O Stretch | Sulfonate | |
| S | S=O Stretch | Sulfonamide | |
| S | S=O Stretch | Sulfonic acid | |
| S | S=O Stretch | Sulfone | |
| M | O-H bend | Phenol | |
| 1521.37 | S | N-O stretch | Nitro compound |
| 1648.98 | M | C=N Stretch | Imine/Oxime |
| M | C=C stretch | Alkene (Disubstituted) | |
| M | C=C stretch | Alkene | |
| M | C=C stretch | Conjugated alkene | |
| M | N-H bend | Amine | |
| M | C=C stretch | Cyclic alkene | |
| S | C=C Stretch | Alkene (Mono substituted) | |
| 1656.71 | M | C=C stretch | Alkene (Disubstituted) |
| M | C=N Stretch | Imine/Oxime | |
| M | C=C stretch | Alkene (Vinyldehyd) | |
| W | C-H bend | Aromatic compound | |
| 1873.25 | W | C-H bend | Aromatic compound |
| 2859.29 | SB | O-H stretch | Carboxylic acid |
| WB | O-H stretch | Alcohol | |
| SB | N-H Stretch | Amine salt | |
| M | C-H stretch | Alkane | |
| 2925.03 | SB | O-H stretch | Carboxylic acid |
| WB | O-H stretch | Alcohol | |
| SB | N-H Stretch | Amine salt | |
| M | C-H stretch | Alkane | |
| 2959.83 | SB | O-H stretch | Carboxylic acid |
| WB | O-H stretch | Alcohol | |
| SB | N-H Stretch | Amine salt | |
| M | C-H stretch | Alkane | |
| 3404.51 | SB | O-H stretch | Alcohol |
| 3686.79 | MS | O-H Stretch | Alcohol |
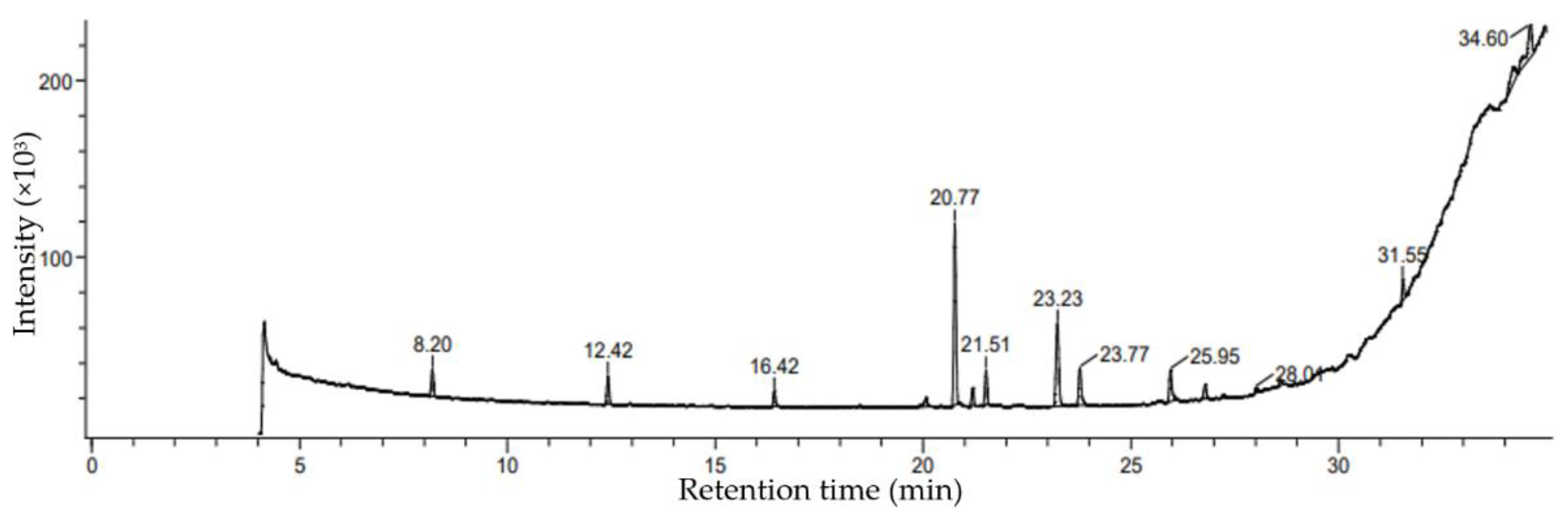
3.2. GCMS Characterization Study in Ethanolic Extracts
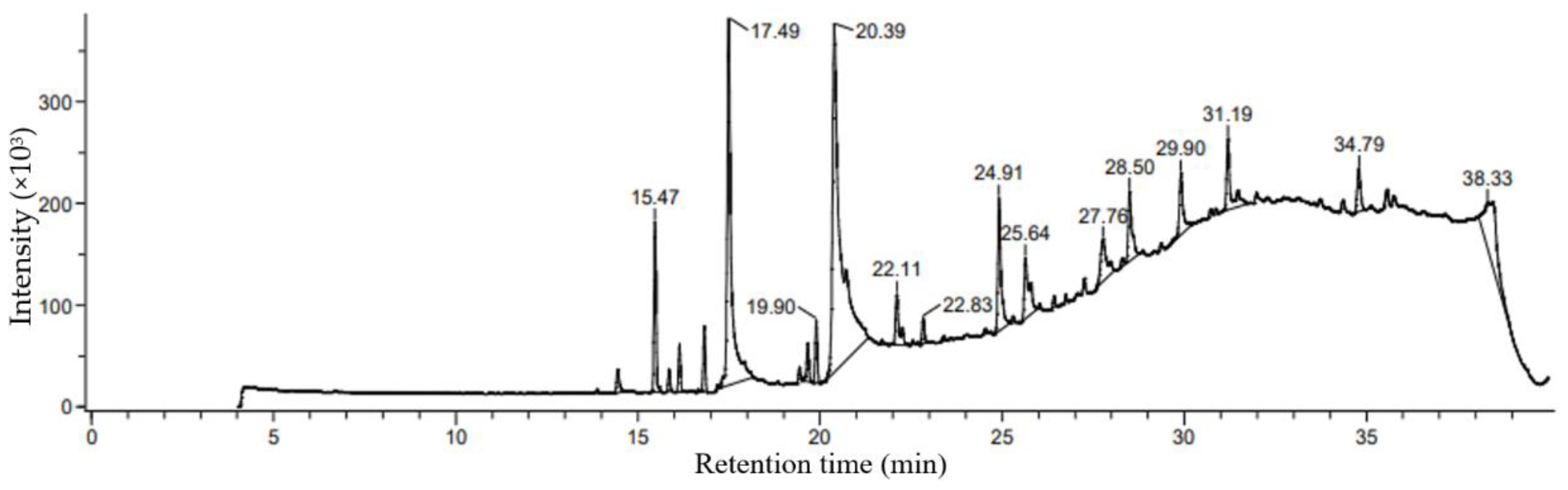
3.3. Fatty Acids Characterization by GC MS Analysis
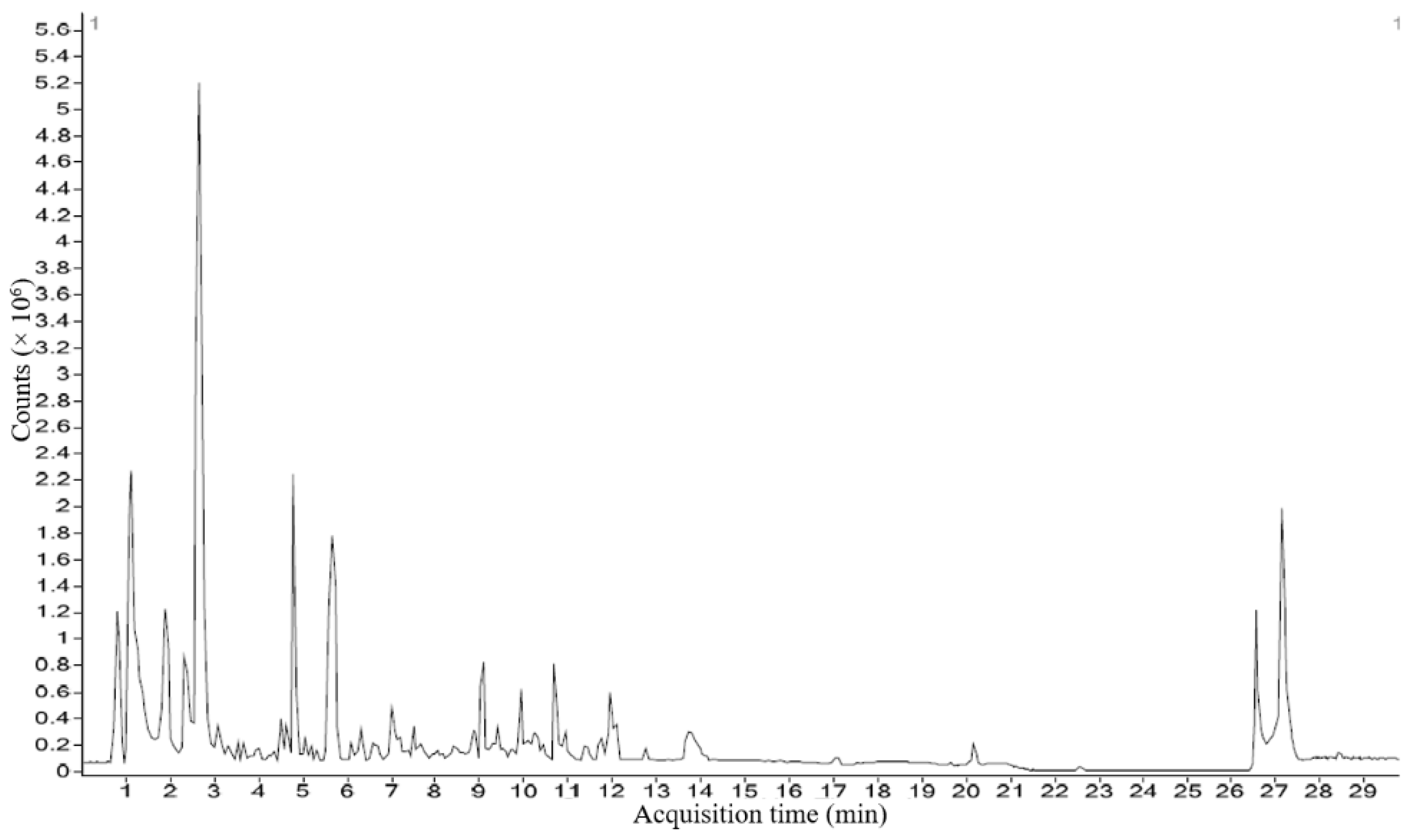
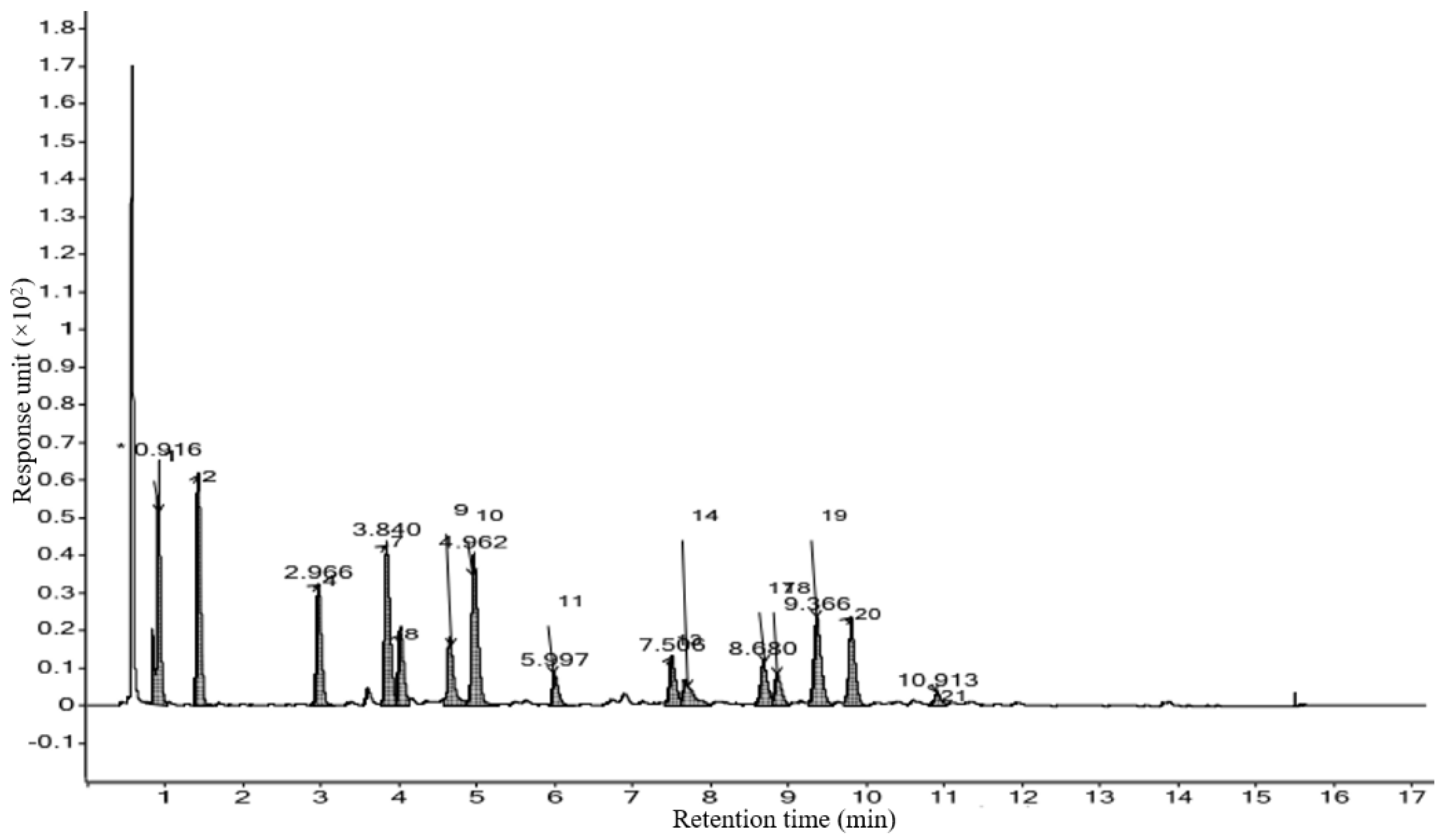
3.4. Characterization of Phytocompounds by HRLCMS Q-TOF
| Phytocompound | Class of compound | Formula | Retention time (min) | Mass (Da) | Abundance | Hits (DB) |
|---|---|---|---|---|---|---|
| 8-Hydroxy-2chlorodibenzofuran | organochlorine compound | C12H7ClO2 | 0.828 | 218.0135 | 828077 | 2 |
| Sulfabenzamide | Sulfur Compounds (sulfonamide) | C13H12N2O3S | 0.829 | 276.0549 | - | 4 |
| 3-((2-Methyl-3-furyl)sulfanyl)-2-butanone | aryl sulfide (organosulfur compound) | C9H12O2S | 0.897 | 184.0573 | 631461 | 7 |
| 8-Amino Caprylic acid | omega-amino fatty acid (Carboxylic Acids) | C8H17NO2 | 1.175 | 159.1249 | 1110887 | 6 |
| L-NIO | Amino Acids, Peptides, and Proteins | C7H15N3O2 | 1.272 | 173.1157 | 449684 | 7 |
| Lys Gly | Dipeptide | C8H17N3O3 | 1.318 | 203.1262 | 189239 | 10 |
| Pirbuterol | Amino Alcohols (Ethanolamines) | C12H20N2O3 | 1.319 | 240.146 | - | 10 |
| N-Guanyl Histamine | Amines | C6H11N5 | 1.329 | 153.1014 | 143807 | 6 |
| Pro Pro His | Pro-Pro-His is an oligopeptide. | C16H23N5O4 | 1.894 | 349.1722 | - | 10 |
| MeIQx | Quinoxalines (aromatic amine) | C11H11N5 | 2.375 | 213.1016 | 208953 | 10 |
| Salsolidine | Isoquinolines (organic heterocyclic compound) | C12H17NO2 | 2.937 | 207.1253 | - | 10 |
| Isopentenyladenine-9-Glucoside | organic heterocyclic compound(aminopurine) | C17H25N5O4 | 3.179 | 363.1882 | - | 10 |
| 2-Amino-1,7,9trimethylimidazo [4,5g]quinoxaline | Heterocyclic Compounds, 2-Ring(Quinoxalines) | C12H13N5 | 3.408 | 227.1171 | 136941 | 7 |
| Niazirinin | carbohydrates and carbohydrate derivatives | C16H19NO6 | 3.831 | 321.1201 | 116378 | 10 |
| Cryptopleurine | Alkaloids | C24H27NO3 | 3.88 | 377.2028 | 92057 | 10 |
| beta-Butoxyethyl nicotinate | an aromatic carboxylic acid and a member of pyridines. | C12H17NO3 | 3.964 | 223.12 | 104913 | 7 |
| LG 100268 | Vitamin B Complex (Nicotinic Acids) | C24H29NO2 | 4.033 | 363.2243 | 177426 | 7 |
| 2-(3′-Methylthio)propyl malic acid | Alcohol | C8H14O5S | 4.169 | 222.0578 | 92654 | 10 |
| Gln Phe Lys | peptide (organic amino compound) | C20H31N5O5 | 4.387 | 421.2295 | 146961 | 10 |
| N-linoleoyl taurine | fatty amide (fatty acid derivative) | C20H37NO4S | 4.6 | 387.2453 | 149019 | 10 |
| Tyr Ile Pro | Peptide | C20H29N3O5 | 4.632 | 391.2192 | 268855 | 10 |
| S-Decyl GSH | Peptide | C20H37N3O6S | 5.096 | 447.245 | 79144 | 5 |
| ORG 20599 | Steroids (Pregnanediones) | C25H40ClNO3 | 5.171 | 437.2696 | 6034 | 10 |
| Nafronyl | Naphthalenes (benzenoid aromatic compound) | C24H33NO3 | 5.27 | 383.2521 | 82233 | 7 |
| Lys Met Lys | Oligopeptide | C17H35N5O4S | 5.533 | 405.2348 | 156109 | 7 |
| Benzenemethanol, 2-(2hydroxypropoxy)-3-methyl- | aromatic ether | C11H16O3 | 5.68 | 196.109 | 719286 | 10 |
| Acetyl Lycopsamine | Pyrrolizines (organic heterocyclic compound) | C17H27NO6 | 5.791 | 341.1862 | 215252 | 10 |
| Benzenemethanol, 2-(2hydroxypropoxy)-3-methyl- | aromatic ether | C11H16O3 | 6.022 | 196.1092 | 130269 | 10 |
| 2,6-Dimethoxy-4-(1propenyl)phenol | phenols | C11H14O3 | 6.247 | 194.0938 | 149475 | 10 |
| Azuleno(5,6-c)furan-1(3H)-one, 4,4a,5,6,7,7a,8,9-octahydro-3,4,8-trihydroxy-6,6,8-trimethyl- | Terpenoids (Sesquiterpenoids) | C15H22O5 | 6.251 | 282.1453 | 113097 | 10 |
| Hericerin | Amides (Lactams) | C27H33NO3 | 6.316 | 419.2504 | 325331 | 5 |
| Di-n-pentyl phthalate | phthalate ester (Phthalic Acids) | C18H26O4 | 6.811 | 306.1819 | 155711 | 7 |
| N-Formyl-norleucyl-leucylphenylalanyl-methylester | peptide | C23H35N3O5 | 7.036 | 433.2662 | 476213 | 2 |
| 2,4,6-Trimethyl-4-phenyl-1,3 dioxane | Dioxanes | C13H18O2 | 7.158 | 206.13 | 240568 | 10 |
3.5. Measurements of Different Amino Acids by HRLCMS
3.6. Element Analysis By ICP-AES Technique
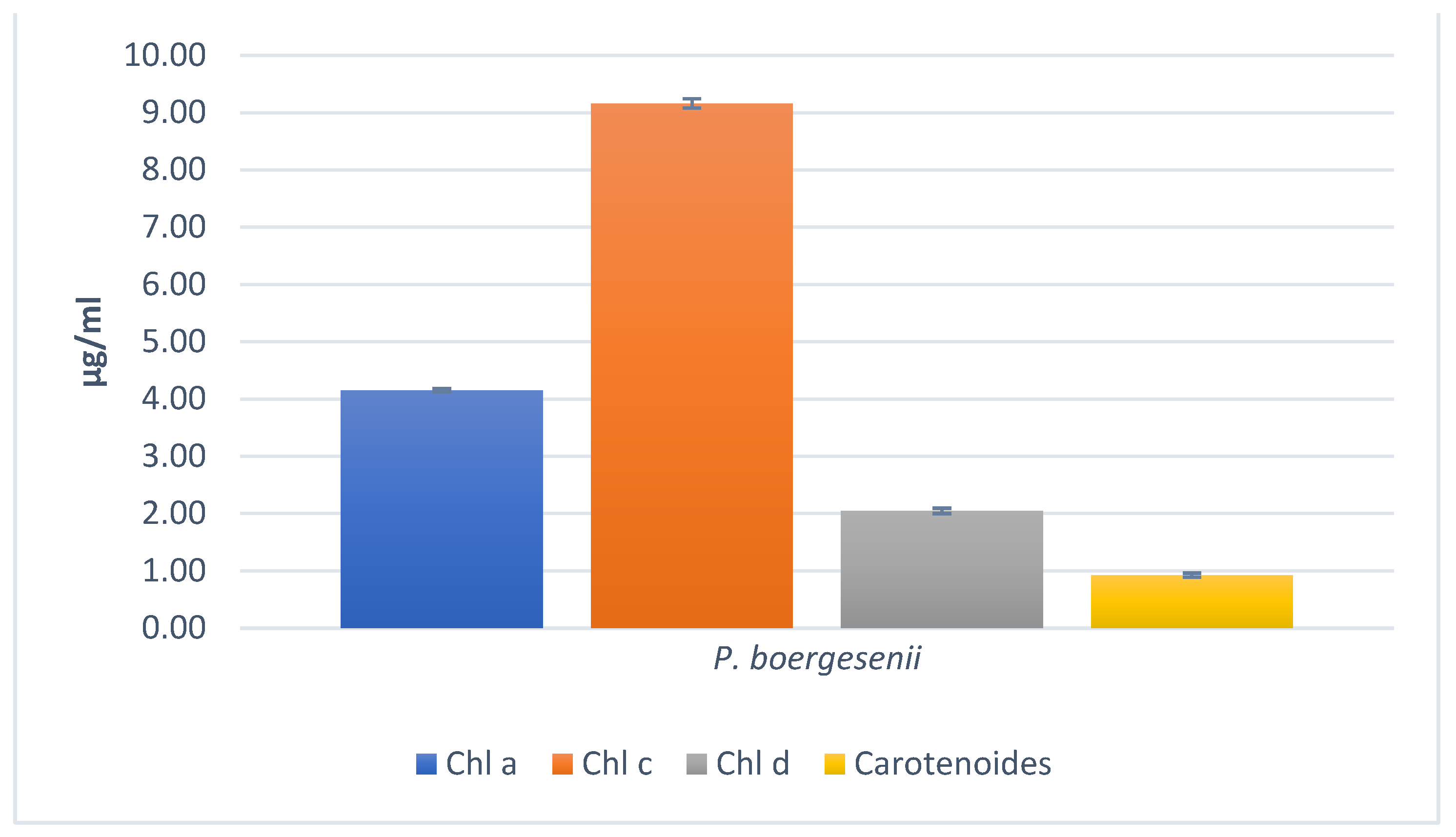
3.7. Determination of Pigments content
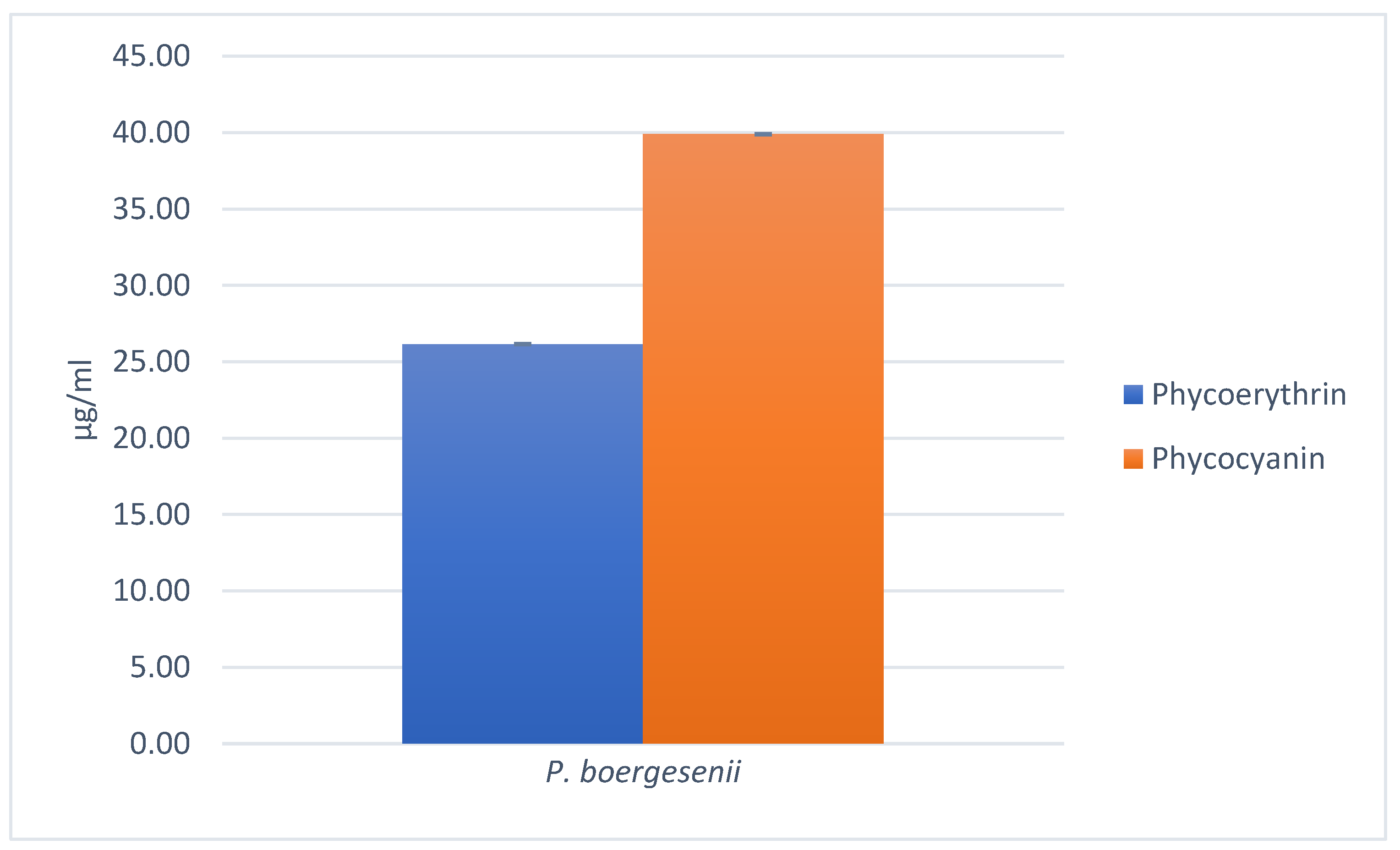
3.7.1. Quantification of Chlorophylls, Carotenoids, Fucoxanthin, Phycoerythrin, and Phycocyanin
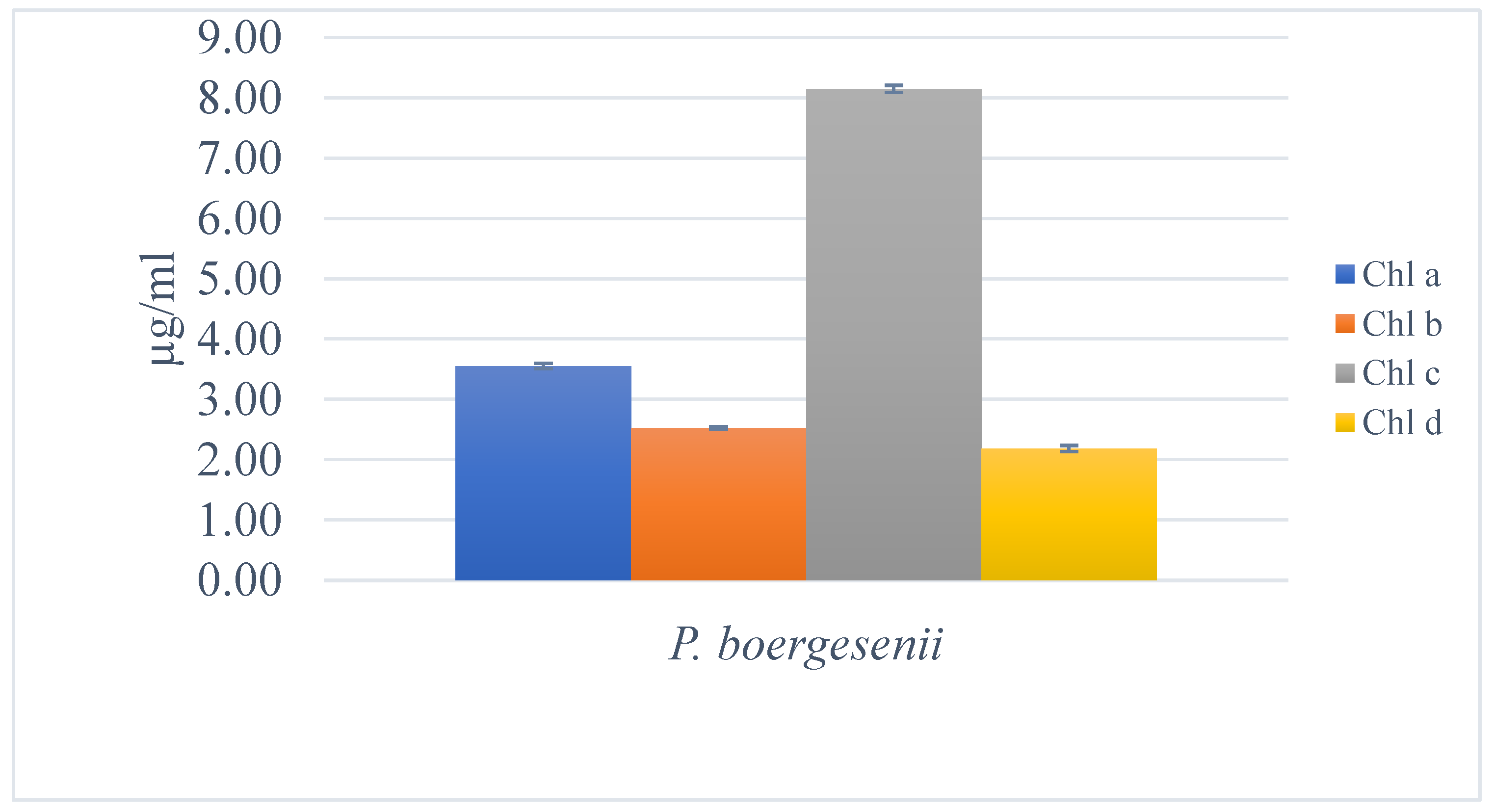
3.7.2. Estimation of Chlorophylls and Lycopene
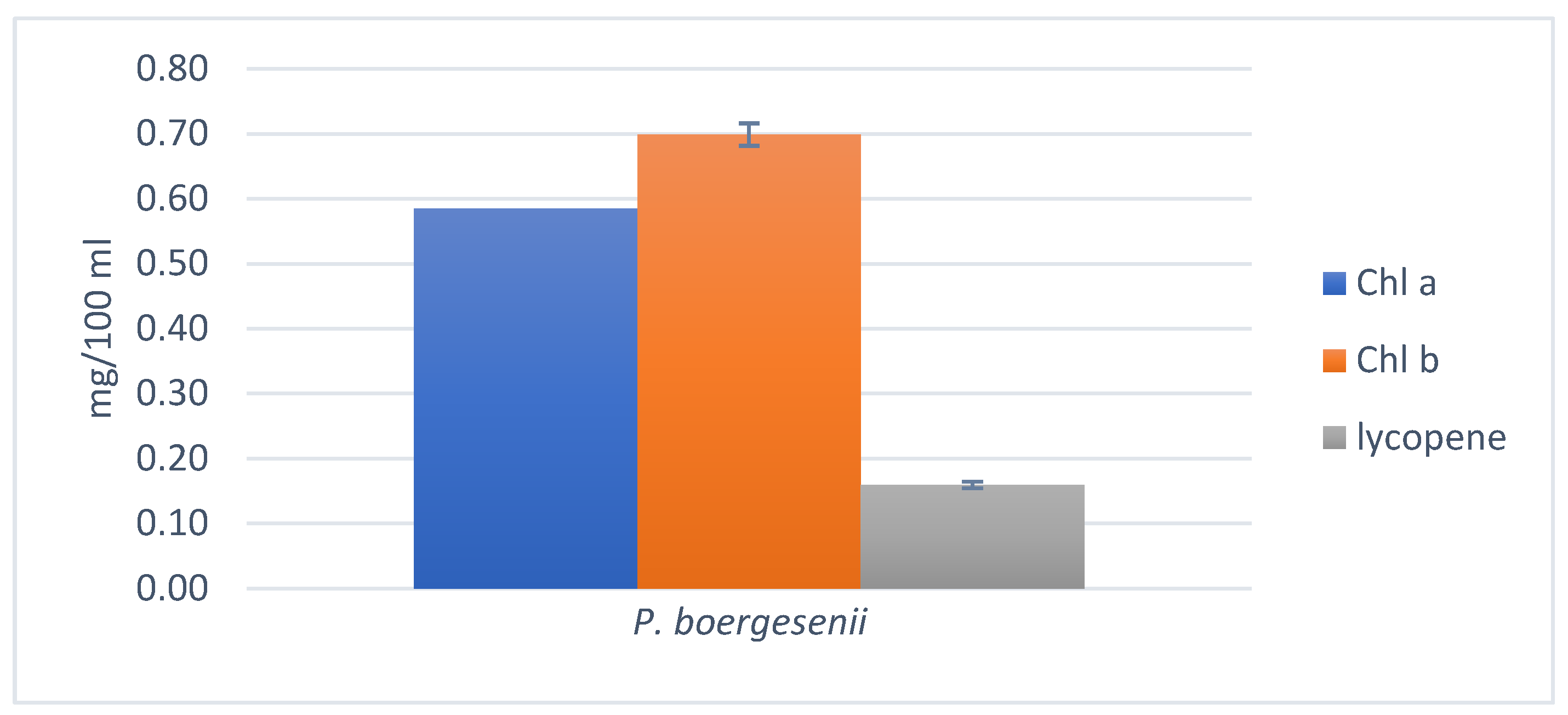
3.7.3. Chlorophylls and Total Carotenoids estimation
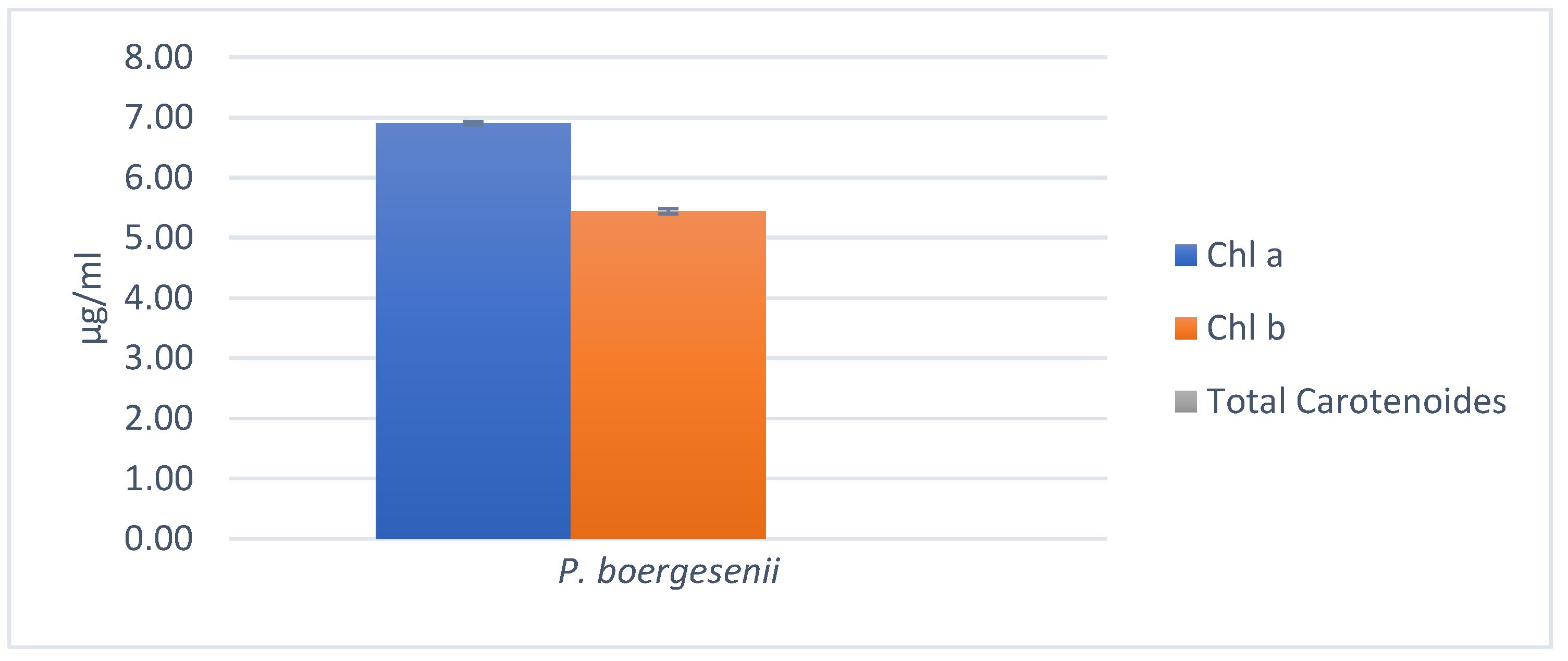
3.8. Analysis of Total Polyphenol Content
3.9. Estimation of Total Protein
3.10. In vitro Antioxidant Analysis using the DPPH Method
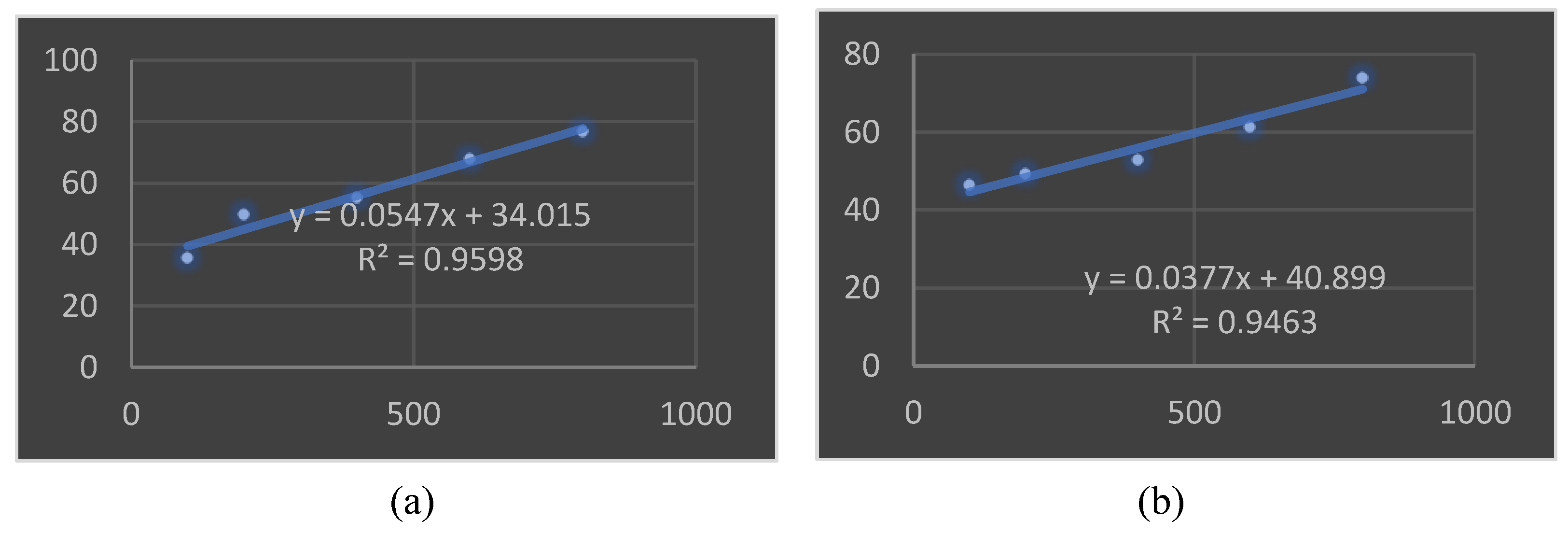
3.13. Tyrosinase Inhibition Assay
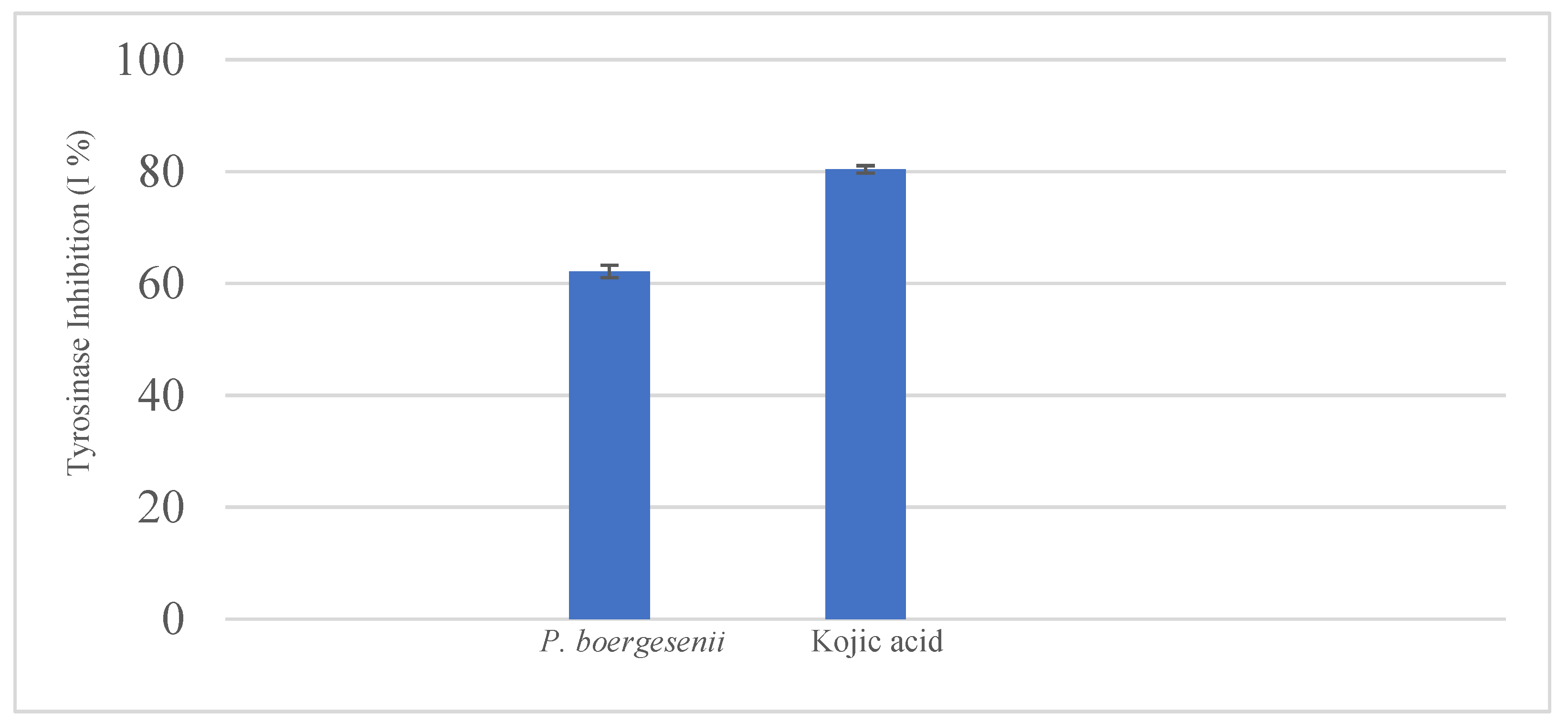
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Kim, S.K. Marine cosmeceuticals. Journal of cosmetic dermatology 2014, 13, 56–67. [Google Scholar] [CrossRef]
- Kerdudo, A.; Burger, P.; Merck, F.; Dingas, A.; Rolland, Y.; Michel, T.; Fernandez, X. Development of a natural ingredient—Natural preservative: A case study. Comptes Rendus Chimie 2016, 19, 1077–1089. [Google Scholar] [CrossRef]
- Hauser, R.; Calafat, A.M. Phthalates and human health. Occupational and environmental medicine 2005, 62, 806–818. [Google Scholar] [CrossRef] [PubMed]
- Garlantézec, R.; Monfort, C.; Rouget, F.; Cordier, S. Maternal occupational exposure to solvents and congenital malformations: a prospective study in the general population. Occupational and environmental medicine 2009, 66, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Kalasariya, H.S.; Patel, N.B.; Yadav, A.; Perveen, K.; Yadav, V.K.; Munshi, F.M.; Yadav, K.K.; Alam, S.; Jung, Y.K.; Jeon, B.H. Characterization of fatty acids, polysaccharides, amino acids, and minerals in marine macroalga Chaetomorpha crassa and evaluation of their potentials in skin cosmetics. Molecules 2021, 26, 7515. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Desai, A.Y.; Mulye, V. Seaweed resources and utilization: an overview. Biotech Express 2015, 2, 22. [Google Scholar]
- Pereira, L.; Neto, J.M. (Eds.) Marine algae: biodiversity, taxonomy, environmental assessment, and biotechnology. CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Jesumani, V.; Du, H.; Aslam, M.; Pei, P.; Huang, N. Potential use of seaweed bioactive compounds in skincare—A review. Marine drugs 2019, 17, 688. [Google Scholar] [CrossRef]
- Pradhan, B.; Bhuyan, P.P.; Patra, S.; Nayak, R.; Behera, P.K.; Behera, C.; Behera, A.K.; Ki, J.S.; Jena, M. Beneficial effects of seaweeds and seaweed-derived bioactive compounds: Current evidence and future prospective. Biocatalysis and Agricultural Biotechnology 2022, 39, 102242. [Google Scholar] [CrossRef]
- Venkatesan, K.; Sivadasan, D.; Alghazwani, Y.; Asiri, Y.I.; Prabahar, K.; Al-Qahtani, A.; Mohamed, J.M.M.; Khan, N.A.; Krishnaraju, K.; Paulsamy, P.; Vasudevan, R. Potential of seaweed biomass: snake venom detoxifying action of brown seaweed Padina boergesenii against Naja naja venom. Biomass Conversion and Biorefinery 2023, 1–14. [Google Scholar] [CrossRef]
- Hakim, M.M.; Patel, I.C. A review on phytoconstituents of marine brown algae. Future Journal of Pharmaceutical Sciences 2020, 6, 1–11. [Google Scholar] [CrossRef]
- John, D.M. Marine algae (seaweeds) associated with coral reefs in the Gulf. Coral Reefs of the Gulf: Adaptation to Climatic Extremes 2012, 309–335. [Google Scholar]
- Moubayed, N.M.; Al Houri, H.J.; Al Khulaifi, M.M.; Al Farraj, D.A. Antimicrobial, antioxidant properties and chemical composition of seaweeds collected from Saudi Arabia (Red Sea and Arabian Gulf). Saudi journal of biological sciences 2017, 24, 162–169. [Google Scholar] [CrossRef]
- Suman, T.Y.; Rajasree, S.R.; Kirubagaran, R. Evaluation of zinc oxide nanoparticles toxicity on marine algae Chlorella vulgaris through flow cytometric, cytotoxicity and oxidative stress analysis. Ecotoxicology and environmental safety 2015, 113, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Kavitha, J.; Palani, S. Phytochemical screening, GC-MS analysis and antioxidant activity of marine algae Chlorococcum humicola. World Journal of Pharmacy and Pharmaceutical Sciences 2016, 5, 1154–1167. [Google Scholar]
- Tatipamula, V.B.; Killari, K.N.; Prasad, K.; Rao, G.S.N.K.; Talluri, M.R.; Vantaku, S.; Bilakanti, D.; Srilakshmi, N. Cytotoxicity studies of the chemical constituents from marine algae Chara baltica. Indian Journal of Pharmaceutical Sciences 2019, 81, 815–823. [Google Scholar] [CrossRef]
- Mustapa, A.N.; Martin, Á.; Mato, R.B.; Cocero, M.J. Extraction of phytocompounds from the medicinal plant Clinacanthus nutans Lindau by microwave-assisted extraction and supercritical carbon dioxide extraction. Industrial Crops and Products 2015, 74, 83–94. [Google Scholar] [CrossRef]
- Ragunathan, V.; Pandurangan, J.; Ramakrishnan, T. Gas chromatography-mass spectrometry analysis of methanol extracts from marine red seaweed Gracilaria corticata. Pharmacognosy Journal 2019, 11. [Google Scholar] [CrossRef]
- Cyriac, B.; Eswaran, K. GC-MS determination of bioactive components of Gracilaria dura (C. agardh) J. Agardh. Science Research Reporter 2015, 5, 100–105. [Google Scholar]
- Salunke, M.; Wakure, B.; Wakte, P. HR-LCMS assisted phytochemical screening and an assessment of anticancer activity of Sargassum squarrossum and Dictyota dichotoma using in vitro and molecular docking approaches. Journal of Molecular Structure 2022, 1270, 133833. [Google Scholar] [CrossRef]
- Salunke, M.A.; Wakure, B.S.; Wakte, P.S. High-resolution liquid chromatography and mass spectrometry (HR-LCMS) assisted phytochemical profiling and an assessment of anticancer activities of Gracilaria foliifera and Turbinaria conoides using in vitro and molecular docking analysis. Journal of Biomolecular Structure and Dynamics 2022, 1–16. [Google Scholar] [CrossRef]
- Rosic, N.N.; Braun, C.; Kvaskoff, D. Extraction and analysis of mycosporine-like amino acids in marine algae. Natural products from marine algae: methods and protocols 2015, 119–129. [Google Scholar]
- Javith, M.A.; Balange, A.K.; Xavier, M.; Hassan, M.A.; Sanath Kumar, H.; Nayak, B.B.; Krishna, G. Comparative studies on the chemical composition of inland saline reared Litopenaeus vannamei. Journal of Culinary Science & Technology 2022, 20, 336–349. [Google Scholar]
- Carreto, J.I.; Carignan, M.O.; Montoya, N.G. A high-resolution reverse-phase liquid chromatography method for the analysis of mycosporine-like amino acids (MAAs) in marine organisms. Marine Biology 2005, 146, 237–252. [Google Scholar] [CrossRef]
- Falandysz, J.; Szymczyk, K.; Ichihashi, H.; Bielawski, L.; Gucia, M.; Frankowska, A.; Yamasaki, S.I. ICP/MS and ICP/AES elemental analysis (38 elements) of edible wild mushrooms growing in Poland. Food Additives & Contaminants 2001, 18, 503–513. [Google Scholar]
- Murugaiyan, K.; Narasimman, S. Elemental composition of Sargassum longifolium and Turbinaria conoides from Pamban Coast, Tamilnadu. International Journal of Research in Biological Sciences 2012, 2, 137–140. [Google Scholar]
- Osório, C.; Machado, S.; Peixoto, J.; Bessada, S.; Pimentel, F.B.; CAlves, R.; Oliveira, M.B.P. Pigments content (chlorophylls, fucoxanthin and phycobiliproteins) of different commercial dried algae. Separations 2020, 7, 33. [Google Scholar] [CrossRef]
- Nagata, M.; Yamashita, I. Simple method for simultaneous determination of chlorophyll and carotenoids in tomato fruit. Nippon Shokuhin Kogyo Gakkaishi 1992, 39, 925–928. [Google Scholar] [CrossRef]
- Yang, C.M.; Chang, K.W.; Yin, M.H.; Huang, H.M. Methods for the determination of the chlorophylls and their derivatives. Taiwania 1998, 43, 116–122. [Google Scholar]
- Wijaya, C.; Elya, B.; Yanuar, A. Study of tyrosinase inhibitory activity and phytochemical screening of Cassia fistula L. Leaves. International Journal of Applied Pharmaceutics 2018, 10, 384–387. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical biochemistry 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Maheswari, M.U.; Reena, A.; Sivaraj, C. GC-MS analysis, antioxidant and antibacterial activity of the brown algae, Padina tetrastromatica. International Journal of Pharmaceutical Sciences and Research 2017, 8, 4014–4020. [Google Scholar]
- Salleh, W.M.N.H.W.; Ahmad, F.; Yen, K.H. Antioxidant and anti-tyrosinase activities from Piper officinarum C. DC (Piperaceae). Journal of Applied Pharmaceutical Science 2014, 4, 087–091. [Google Scholar]
- Soleimani, S.; Yousefzadi, M.; Babaei Mahani Nezhad, S.; Pozharitskaya, O.N.; Shikov, A.N. Potential of the Ethyl Acetate Fraction of Padina boergesenii as a Natural UV Filter in Sunscreen Cream Formulation. Life 2023, 13, 239. [Google Scholar] [CrossRef]
- Kalasariya, H.S.; Pereira, L.; Patel, N.B. Pioneering role of marine macroalgae in cosmeceuticals. Phycology 2022, 2, 172–203. [Google Scholar] [CrossRef]
- Fonseca, S.; Amaral, M.N.; Reis, C.P.; Custódio, L. Marine Natural Products as Innovative Cosmetic Ingredients. Marine Drugs 2023, 21, 170. [Google Scholar] [CrossRef] [PubMed]
- Muthukumar, J.; Chidambaram, R.; Sukumaran, S. Sulfated polysaccharides and its commercial applications in food industries—A review. Journal of Food Science and Technology 2021, 58, 2453–2466. [Google Scholar] [CrossRef]
- Theophilus, T.; Vieira, C.; Culioli, G.; Thomas, O.P.; Antoine De Ramon, N.Y.; Andréfouët, S.; Mattio, L.; Payri, C.E.; Zubia, M. Dictyotaceae (Dictyotales, Phaeophyceae) species from French Polynesia: current knowledge and future research. Advances in Botanical Research 2020, 95, 163–211. [Google Scholar]
- Oliveira, N.M.; Meira, C.L.; Aguiar, R.M.; De Oliveira, D.M.; Moura, C.; Vilho, S. Biological activities of extracts from Padina boergesenii and Sargassum stenophyllum, Seaweeds naturally found in baia de todos os santos, Brazil. Academic Sciences 2015, 7, 351–353. [Google Scholar]
- Namjooyan, F.; Farasat, M.; Alishahi, M.; Jahangiri, A.; Mousavi, H. The anti-melanogenesis activities of some selected brown macroalgae from northern coasts of the Persian Gulf. Brazilian Archives of Biology and Technology 2019, 62. [Google Scholar] [CrossRef]
- Leandro, A.; Pereira, L.; Gonçalves, A. Diverse Applications of Marine Macroalgae. Mar. Drugs 2019, 18, 17. [Google Scholar] [CrossRef]
- Corsetti, G.; D’Antona, G.; Dioguardi, F.S.; Rezzani, R. Topical application of dressing with amino acids improves cutaneous wound healing in aged rats. Acta Histochem. 2010, 112, 497–507. [Google Scholar] [CrossRef]
- Veis, A.; Anesey, J. Modes of intermolecular cross-linking in mature insoluble collagen. J. Biol. Chem. 1965, 240, 3899–3908. [Google Scholar] [CrossRef]
- Choi, H.-R.; Kang, Y.-A.; Ryoo, S.-J.; Shin, J.-W.; Na, J.-I.; Huh, C.-H.; Park, K.-C. Stem cell recovering effect of copper-free GHK in skin. J. Pept. Sci. 2012, 18, 685–690. [Google Scholar] [CrossRef]
- Murakami, H.; Shimbo, K.; Inoue, Y.; Takino, Y.; Kobayashi, H. Importance of amino acid composition to improve skin collagen protein synthesis rates in UV-irradiated mice. Amino Acids 2011, 42, 2481–2489. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, M.; Yokose, U.; Hachiya, A.; Fujimura, T.; Tsukahara, K.; Kawada, H.; Kitahara, T.; Takema, Y.; Terui, T.; Nakagawa, H. Improvement of crow’s feet lines by topical application of 1-carbamimidoyl-L-proline (CLP). Eur. J. Dermatol. 2013, 23, 195–201. [Google Scholar] [CrossRef]
- Yamane, T.; Morioka, Y.; Kitaura, Y.; Iwatsuki, K.; Shimomura, Y.; Oishi, Y. Branched-chain amino acids regulate type I tropocollagen and type III tropocollagen syntheses via modulation of mTOR in the skin. Biosci. Biotechnol. Biochem. 2018, 82, 611–615. [Google Scholar] [CrossRef]
- Puviani, M.; Agostinis, F.; Milani, M. Barrier repair therapy for facial atopic eczema with a non-steroidal emollient cream containing rhamnosoft, ceramides and iso-leucine. A six-case report series. Minerva Pediatr. 2014, 66, 307–311. [Google Scholar]
- Brenner, M.; Hearing, V.J. The Protective Role of Melanin Against UV Damage in Human Skin. Photochem. Photobiol. 2008, 84, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Serre, C.; Busuttil, V.; Botto, J.-M. Intrinsic and extrinsic regulation of human skin melanogenesis and pigmentation. Int. J. Cosmet. Sci. 2018, 40, 328–347. [Google Scholar] [CrossRef]
- Sardana, K.; Garg, V.K. An observational study of methionine-bound zinc with antioxidants for mild to moderate acne vulgaris. Dermatol. Ther. 2010, 23, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Bazer, F.W.; Burghardt, R.C.; Johnson, G.A.; Kim, S.W.; Knabe, D.A.; Li, P.; Li, X.; McKnight, J.R.; Satterfield, M.C.; et al. Proline and hydroxyproline metabolism: Implications for animal and human nutrition. Amino Acids 2010, 40, 1053–1063. [Google Scholar] [CrossRef]
- Elias, P.M.; Ahn, S.K.; Denda, M.; Brown, B.E.; Crumrine, D.; Kimutai, L.K.; Kömüves, L.; Lee, S.H.; Feingold, K.R. Modulations in Epidermal Calcium Regulate the Expression of Differentiation-Specific Markers. J. Investig. Dermatol. 2002, 119, 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- Matz, H.; Orion, E.; Wolf, R. Balneotherapy in dermatology. Dermatol. Ther. 2003, 16, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Denda, M.; Katagiri, C.; Hirao, T.; Maruyama, N.; Takahashi, M. Some magnesium salts and a mixture of magnesium and calcium salts accelerate skin barrier recovery. Arch. Dermatol. Res. 1999, 291, 560–563. [Google Scholar] [CrossRef]
- Schempp, C.M.; Dittmar, H.C.; Hummler, D.; Simon-Haarhaus, B.; Schöpf, E.; Simon, J.C.; Schulte-Mönting, J. Magnesium ions inhibit the antigen-presenting function of human epidermal Langerhans cells in vivo and in vitro. Involvement of ATPase, HLA-DR, B7 molecules, and cytokines. J. Investig. Dermatol. 2000, 115, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration, HHS. Skin protectant drug products for over-the-counter human use; final monograph. Final rule. Fed. Regist. 2003, 68, 33362–33381. [Google Scholar]
- Food and Drug Administration. Sunscreen Drug Products for Over-the-Counter Human Use. Amendment to the Tentative Final Monograph; Enforcement Policy. Fed. Regist. 2019, 63, 6204–6275. [Google Scholar]
- Higdon, J.; Drake, V.J. An Evidenced-Based Approach to Vitamins and Minerals, 2nd ed.; Georg Thieme Verlag: Stuttgart, Germany, 2012; pp. 157–168. [Google Scholar]
- Antoniou, C.; Stefanaki, C. Cosmetic camouflage. J. Cosmet. Dermatol. 2006, 5, 297–301. [Google Scholar] [CrossRef]
- Pickart, L. The human tri-peptide GHK and tissue remodeling. J. Biomater. Sci. Polym. Ed. 2008, 19, 969–988. [Google Scholar] [CrossRef]
- Fowler, J.F., Jr.; Woolery-Lloyd, H.; Waldorf, H.; Saini, R. Innovations in natural ingredients and their use in skin care. Journal of drugs in dermatology: JDD 2010, 9 (Suppl. 6), S72–S81. [Google Scholar]
- Leenutaphong, V. Relationship between skin color and cutaneous response to ultraviolet radiation in Thai. Photodermatology, photoimmunology & photomedicine 1995, 11, 198–203. [Google Scholar]
- O’Connor, I.; O’Brien, N. Modulation of UVA light-induced oxidative stress by β-carotene, lutein and astaxanthin in cultured fibroblasts. Journal of dermatological science 1998, 16, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.D.; Chen, C.C.; Huynh, P.; Chang, J.S. Exploring the potential of using algae in cosmetics. Bioresource technology 2015, 184, 355–362. [Google Scholar] [CrossRef]
- Alves, A.L.; Marques, A.L.; Martins, E.; Silva, T.H.; Reis, R.L. Cosmetic potential of marine fish skin collagen. Cosmetics 2017, 4, 39. [Google Scholar] [CrossRef]
- Zhang, H.; Tang, Y.; Zhang, Y.; Zhang, S.; Qu, J.; Wang, X.; Kong, R.; Han, C.; Liu, Z. Fucoxanthin: A promising medicinal and nutritional ingredient. Evidence-based complementary and alternative medicine 2015. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Bai, L.; Zhu, L.; Yang, L.; Zhang, X. Marine algae-derived bioactive peptides for human nutrition and health. Journal of Agricultural and Food Chemistry 2014, 62, 9211–9222. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.; Parra, R.; MN Iqbal, H. Phycobiliproteins: a novel green tool from marine origin blue-green algae and red algae. Protein and peptide letters 2017, 24, 118–125. [Google Scholar] [CrossRef]
- Kadam, S.U.; Tiwari, B.K.; O’Donnell, C.P. Application of novel extraction technologies for bioactives from marine algae. Journal of agricultural and food chemistry 2013, 61, 4667–4675. [Google Scholar] [CrossRef]
- Khoo, H.E.; Prasad, K.N.; Kong, K.W.; Jiang, Y.; Ismail, A. Carotenoids and their isomers: color pigments in fruits and vegetables. Molecules 2011, 16, 1710–1738. [Google Scholar] [CrossRef]
- Souza, B.W.; Cerqueira, M.A.; Bourbon, A.I.; Pinheiro, A.C.; Martins, J.T.; Teixeira, J.A.; Coimbra, M.A.; Vicente, A.A. Chemical characterization and antioxidant activity of sulfated polysaccharide from the red seaweed Gracilaria birdiae. Food Hydrocolloids 2012, 27, 287–292. [Google Scholar] [CrossRef]
- Kim, K.N.; Heo, S.J.; Kang, S.M.; Ahn, G.; Jeon, Y.J. Fucoxanthin induces apoptosis in human leukemia HL-60 cells through a ROS-mediated Bcl-xL pathway. Toxicology in vitro 2010, 24, 1648–1654. [Google Scholar] [CrossRef]
- Farabegoli, F.; Santaclara, F.J.; Costas, D.; Alonso, M.; Abril, A.G.; Espiñeira, M.; Ortea, I.; Costas, C. Exploring the Anti-Inflammatory Effect of Inulin by Integrating Transcriptomic and Proteomic Analyses in a Murine Macrophage Cell Model. Nutrients 2023, 15, 859. [Google Scholar] [CrossRef]
- Dixit, D.C.; Reddy, C.R.K.; Balar, N.; Suthar, P.; Gajaria, T.; Gadhavi, D.K. Assessment of the nutritive, biochemical, antioxidant and antibacterial potential of eight tropical macro algae along Kachchh coast, India as human food supplements. Journal of aquatic food product technology 2018, 27, 61–79. [Google Scholar] [CrossRef]
- Wang, T.; Jonsdottir, R.; Ólafsdóttir, G. Total phenolic compounds, radical scavenging and metal chelation of extracts from Icelandic seaweeds. Food chemistry 2009, 116, 240–248. [Google Scholar] [CrossRef]
- El-Shafei, R.; Hegazy, H.; Acharya, B. A review of antiviral and antioxidant activity of bioactive metabolite of macroalgae within an optimized extraction method. Energies 2021, 14, 3092. [Google Scholar] [CrossRef]
- Monteiro, M.; Santos, R.A.; Iglesias, P.; Couto, A.; Serra, C.R.; Gouvinhas, I.; Barros, A.; Oliva-Teles, A.; Enes, P.; Díaz-Rosales, P. Effect of extraction method and solvent system on the phenolic content and antioxidant activity of selected macro-and microalgae extracts. Journal of Applied Phycology 2020, 32, 349–362. [Google Scholar] [CrossRef]
- Im, D.Y. Antioxidative activity and tyrosinase inhibitory activity of the extract and fractions from Arctium lappa roots and analysis of phenolic compounds. Korean Journal of Pharmacognosy 2014, 45, 141–146. [Google Scholar]
- Jo, J.H.; Kim, D.; Lee, S.; Lee, T.K. Total phenolic contents and biological activities of Korean seaweed extracts. Food Sci. Biotechnol. 2005, 14, 000–000. [Google Scholar]
- Fitton, J.H.; Dell’Acqua, G.; Gardiner, V.A.; Karpiniec, S.S.; Stringer, D.N.; Davis, E. Topical benefits of two fucoidan-rich extracts from marine macroalgae. Cosmetics 2015, 2, 66–81. [Google Scholar] [CrossRef]
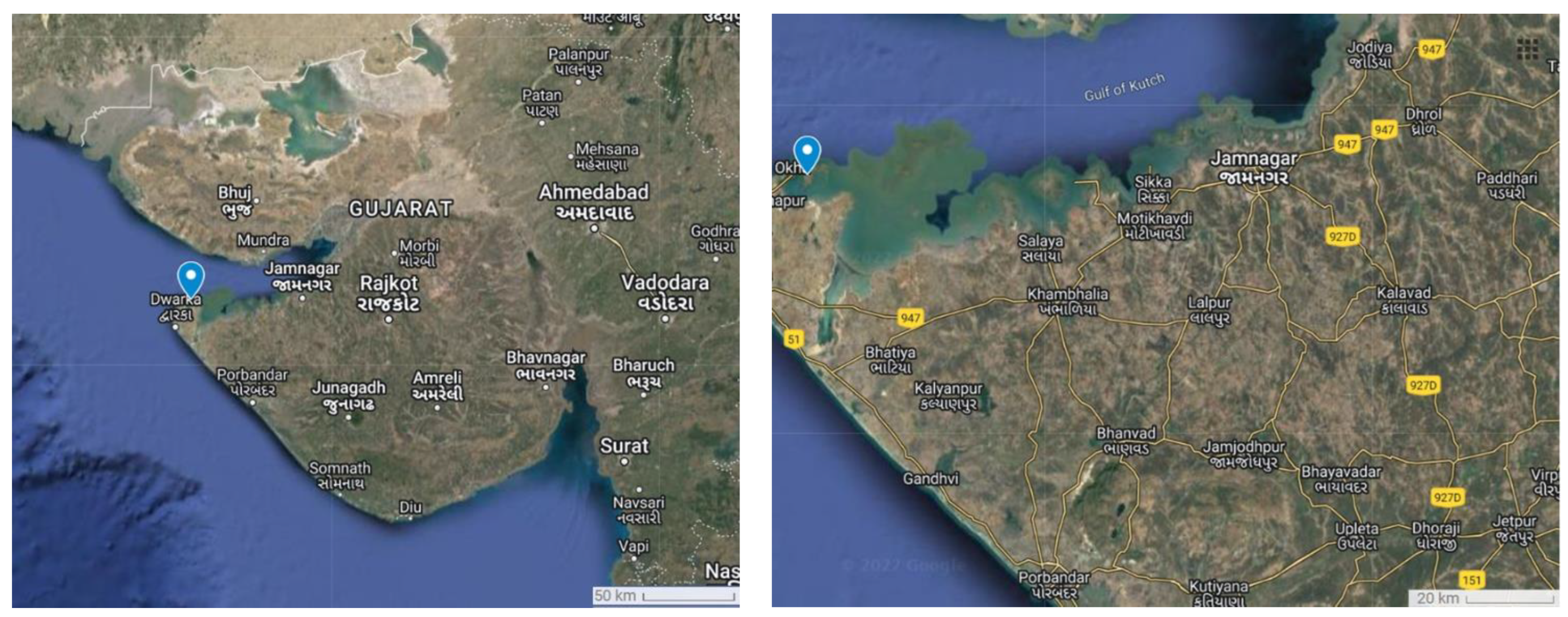


| Name of marine algae | Padina boergesenii |
|---|---|
| Phylum | Ochrophyta |
| Class | Phaeophyceae |
| Order | Dictyotales |
| Family | Dictyotaceae |
| Genus | Padina |
| Species | P. boergesenii |
| No. | Name of compound | PubChem ID | Mol. Formula | Mol. Weight (g/mol) | Retention time (min) | Kovats index (iu) | % Peak area |
|---|---|---|---|---|---|---|---|
| 1. | Decane,6-ethyl-2-methyl- | 43923 | C13H28 | 184.36 | 12.42 | 1185 | 4.32 |
| 2. | Cyclopentanol, 2-methyl-, trans- | 6432271 | C6H12O | 100.16 | 16.42 | 849 | 2.31 |
| 3. | Pentadecanoic acid, tripropylsilyl ester | 632007 | C24H50O2Si | 398.7 | 20.07 | 2484 | 1.68 |
| 4. | 3,7,11,15-Tetramethylhexadec-2-en-1-ol | 145386 | C20H40O | 296.5 | 20.77 | 2045 | 28.64 |
| 5. | Cyclodecanol | 15166 | C10H20O | 156.26 | 21.19 | 1387 | 2.77 |
| 6. | 9-(2-Oxiranyl)-1-nonanol | 565484 | C11H22O2 | 186.29 | 21.51 | 1448 | 5.33 |
| 7. | Phthalic acid,6-ethyl-3-octyl butyl ester | 6423866 | C22H34O4 | 362.5 | 23.23 | 2505 | 16.65 |
| 8. | Hexadecanoic acid, ethyl ester | 12366 | C18H36O2 | 284.5 | 23.77 | 1978 | 6.61 |
| 9. | 17-Octadecen-1-ol acetate | 249903005 | C20H38O2 | 310.5 | 25.95 | 2167 | 5.50 |
| 10. | Cyclooctaneacetic acid, 2-oxo- | 536995 | C10H16O3 | 184.23 | 26.79 | 1647 | 2.35 |
| 11. | Bispalmitic acid 3-methyl-1,2-butanediyl | 549812 | C37H72O4 | 581 | 28.01 | 3905 | 0.87 |
| 12. | 2-Monolinolenin, 2TMS derivative | 5362857 | C27H52O4Si2 | 496.9 | 31.55 | 2804 | 3.25 |
| 13. | Octadecane, 3-ethyl-5-(2-ethylbutyl)- | 292285 | C26H54 | 366.7 | 34.20 | 2413 | 6.97 |
| 14. | Benzyl (6Z,9Z,12Z)-6,9,12-octadecatrienoate | 5368209 | C25H36O2 | 368.6 | 34.60 | 2774 | 8.54 |
| 15. | Oxalic acid, allyl nonyl ester | 6420231 | C14H24O4 | 256.34 | 8.20 | 1738 | 4.19 |
| No. | Name of compound | PubChem ID | Mol. Formula | Mol. Weight (g/mol) | Retention time (min) | Kovats index (iu) | % Peak area |
|---|---|---|---|---|---|---|---|
| 1. | 1,2,4-Trioxolane, 3,5-dipropyl- | 536099 | C8H16O3 | 160.21 | 1.05 | 1086 | 14.45 |
| 2 | Phytol | 5366244 | C20H40O | 296.5 | 6.01 | 2045 | 15.47 |
| 3 | 1,4-Eicosadiene | 5365774 | C20H38 | 278.5 | 1.63 | 2007 | 16.14 |
| 4 | Hexadecanoic acid, methyl ester | 8181 | C17H34O2 | 270.5 | 2.31 | 1878 | 16.82 |
| 5 | n-Hexadecanoic acid | 985 | C16H32O2 | 256.42 | 17.77 | 1968 | 17.49 |
| 6 | 9-Dodecenoic acid, methyl ester, [E]- | 5362755 | C13H24O2 | 212.33 | 1.44 | 1489 | 19.66 |
| 7 | hexahydrofarnesol | 138824 | C15H32O | 228.41 | 2.28 | 1563 | 19.90 |
| 8 | Oleic Acid | 445639 | C18H34O2 | 282.5 | 27.85 | - | 20.39 |
| 38.33 | - | 11.44 | |||||
| 9 | Palmitic acid vinyl ester | 69658 | C18H34O2 | 282.5 | 2.18 | 1968 | 22.11 |
| 10 | 1,1′-Bicyclopentyl, 2-hexadecyl- | 291314 | C26H50 | 362.7 | 1 | 2653 | 22.83 |
| 11 | 9,17-Octadecadienal, [Z]- | 5365667 | C18H32O | 264.4 | 5.92 | 1997 | 24.91 |
| 3.13 | 1997 | 29.90 | |||||
| 12 | 17-Octadecynoic acid | 1449 | C18H32O2 | 280.4 | 3.34 | 2165 | 25.64 |
| 13 | 29-Methylisofucosterol | 6443745 | C30H50O | 426.7 | 3.71 | 2880 | 27.76 |
| 14 | Patchouli alcohol | 10955174 | C15H26O | 222.37 | 3.14 | 1420 | 31.19 |
| 15 | 9-Octadecenal | 5283381 | C18H34O | 266.5 | 2.36 | 2007 | 34.79 |
| Sr.no | Amino Acids | H2 (nmol/mL) |
|---|---|---|
| 1 | Aspartic acid (Asp) | 2034.99 |
| 2 | Glutamic Acid (Glu) | 1332.61 |
| 3 | Asparagine (Asn) | ND |
| 4 | Serine (Ser) | 768.22 |
| 5 | Glutamine (Gln) | ND |
| 6 | Histidine (His) | ND |
| 7 | Glycine (Gly) | 1215.22 |
| 8 | Threonine (Thr) | 527.17 |
| 9 | Arginine (Arg) | 498.77 |
| 10 | Alanine (Ala) | 1268.27 |
| 11 | Tyrosine (Tyr) | 233.26 |
| 12 | Cystine (Cys) | ND |
| 13 | Valine (Val) | 389.20 |
| 14 | Methionine (Met) | 293.46 |
| 15 | Norvaline (Nva) | ND |
| 16 | Tryptophan (Trp) | ND |
| 17 | Phenylalanine (Phe) | 392.66 |
| 18 | Isoleucine (Ile) | 244.84 |
| 19 | Leucine (Leu) | 808.49 |
| 20 | Lysine (Lys) | 354.69 |
| 21 | Hydroxyproline (Hyp) | 806.49 |
| Element | (Amount in %) |
|---|---|
| B | 0.013712 |
| Ca | 4.099749 |
| Cu | 0.000386 |
| Fe | 0.244448 |
| K | 2.709106 |
| Mg | 2.055089 |
| Zn | 0.004538 |
| Na | 0.073146 |
| Si | 20.17574 |
| Se | ND |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




