3. Results
In 2016, we received a case of meningitis in the Pathogenic Protozoa and Endosymbiont Laboratory in Lima, Peru. At first, it was assumed that it could be another classic free-living amoeba.
A series of CSF samples arrived, mainly from pediatric patients between four months old and sixteen years of age over a two-year period (2016-2018). The clinical presentation was meningitis or encephalitis of unknown origin. Viral and bacterial infections were discarded and no pathogen was found; therefore, an infection caused by free-living amoebas was suspected.
We observed 7/8 pediatric patients with similar clinical characteristics in 2016; 6/12 patients in 2017; and 11/14 patients until March 2018.
All samples were CSF and were analyzed through microscopy based on morphology. We used non-nutritive agar plates, but the amoeba did not grow. No cysts were observed. The PCR to Balamuthia mandrillaris and others amoeba was negative in all samples.
The EUK A/B primers used amplified human DNA. They were not in one case, they were in all cases. There is no clear explanation. The most plausible thing is that the primers are not specific enough, the variable regions do not amplify, and the parasite load is too low to be detected by these primers.
Protist case 1:
The sample was CFS from an 8-year-old boy, found of swimming. He swam in a pool in the south of Lima. His symptoms started with an intense headache and vomits, and was preliminarily diagnosed in a Pediatric Hospital in Lima.
In the CSF, a microorganism of approximately 10-12µm in length with fast but erratic movements and capable of moving through very short pseudopods was observed. These pseudo-pseudopods, were short pseudopods, at some point in the movement, dichotomous pseudopods were observed with slightly granular cytoplasm and extremely light hyaline layer. It was not possible to visualize the nucleus or to grow it in a conventional medium for free-living amoebas. At that time, we did not have standardized molecular methods; that is why it was not possible to establish and confirm the real identity of this protist for molecular biology. We believe that the child acquired it in the pool, since there is no other event that is directly related to the patient’s history. Finally, based on morphology characteristics, we believe this protist is a species related to the Valhkamphiae family (
Naegleria sp.) [
4].
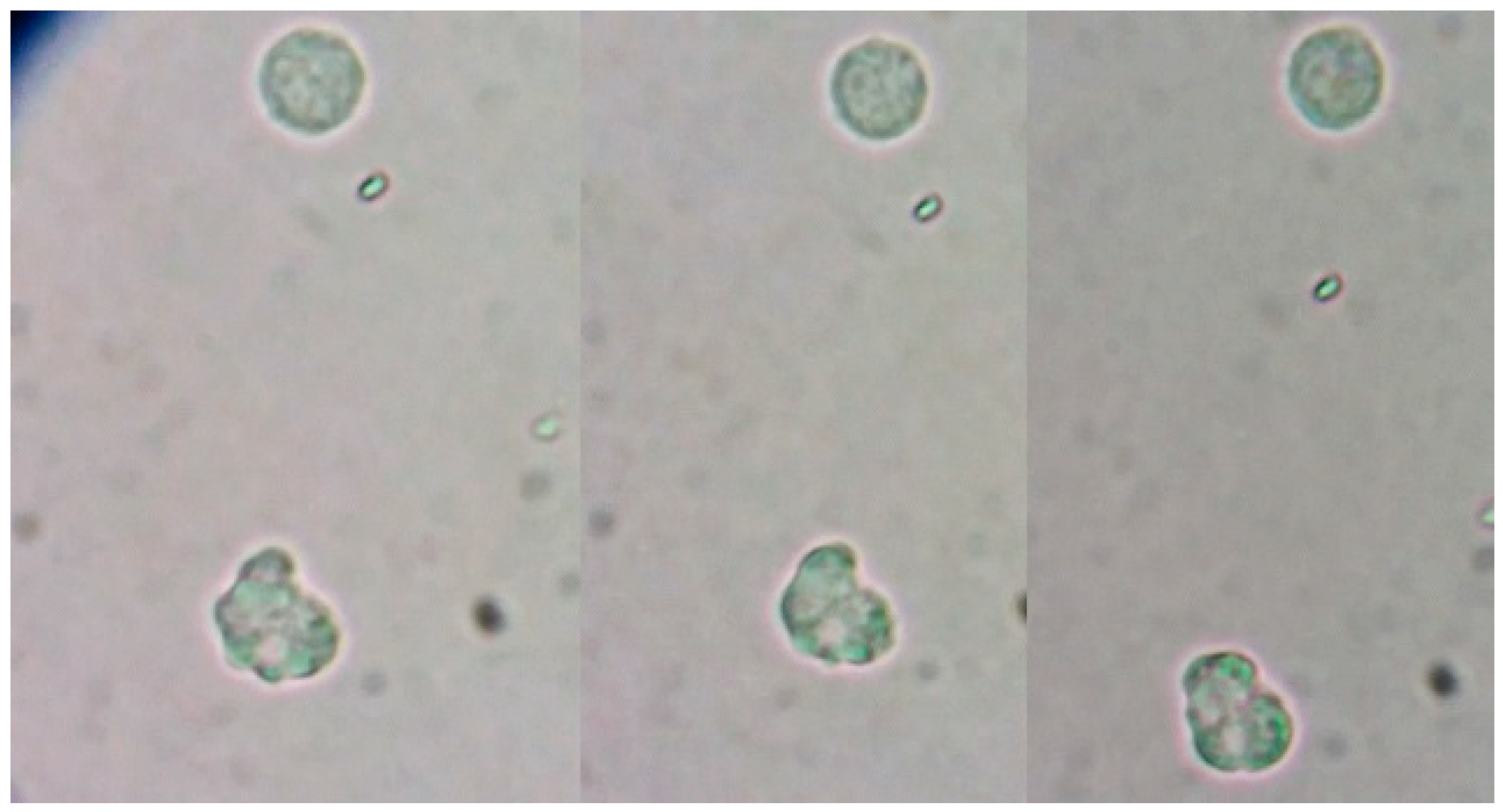
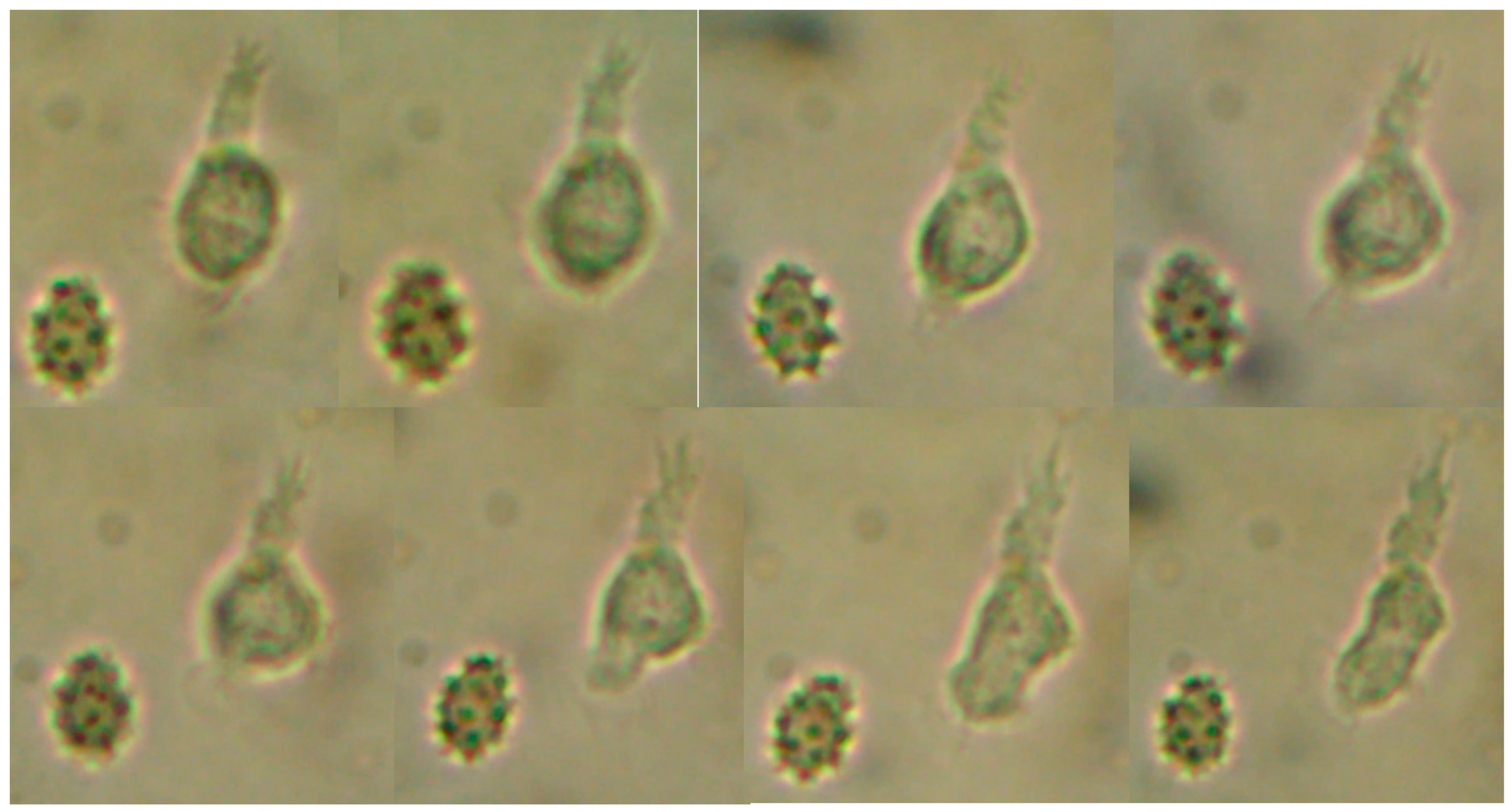
Figure 1.
Trophozoite of freshly extracted CSF observed under the microscope to 40x. It is possible to see in the sequence of images the morphological change of the protist.
Figure 1.
Trophozoite of freshly extracted CSF observed under the microscope to 40x. It is possible to see in the sequence of images the morphological change of the protist.
We could observe erratic movement, although it was sometimes dichotomous with two pseudopods and, at times, an erratic amoeboid movement was seen. Locomotion of the protist and changes in shape can be observed. Through a more detailed analysis of the image of the protist, we could noticed that it produced very well defined short pseudopods and the hyaline layer was not clearly observed. Uroid is not observed.
Host type: Human LCR
Locality: Lima, Peru.
Host collection: Hospital of Lima
Description: 11 μm (9–12 μm, n=30) in length, and 5-6μm (5-8μm) in width. Ovoid cell body shape, with pseudopodos shorts and eruptive.
There was not enough sample to perform the molecular test.
Protist 2
A healthy 12-year-old girl without important risk factors, swimmer of the Peruvian Swimming Federation, had a clinical picture of meningitis. In the CSF, two morphotypes of amoebas were observed: a) Large amoeba of around 20 -25µm with thick and long pseudopods and b) Long amoeba of 25-30 microns with a filamentous uroid which is somewhat longer than other species described. In both, the locomotion was fast and changed in form. The nucleus was not observed in amoeba [amoeba-1], but in amoeba [amoeba-2]. We do not know if they are the same or if they were really two types of amoebas. However, they did not grow in any culture medium. These protists were kept active for several days in axenic medium at 37°C. Then they disappeared. None of these forms were observed in the pool sample.
In this sample, we tried to elucidate the identity of this amoeba by performing the PCR using the FLA primers, but we only managed to amplify human DNA.
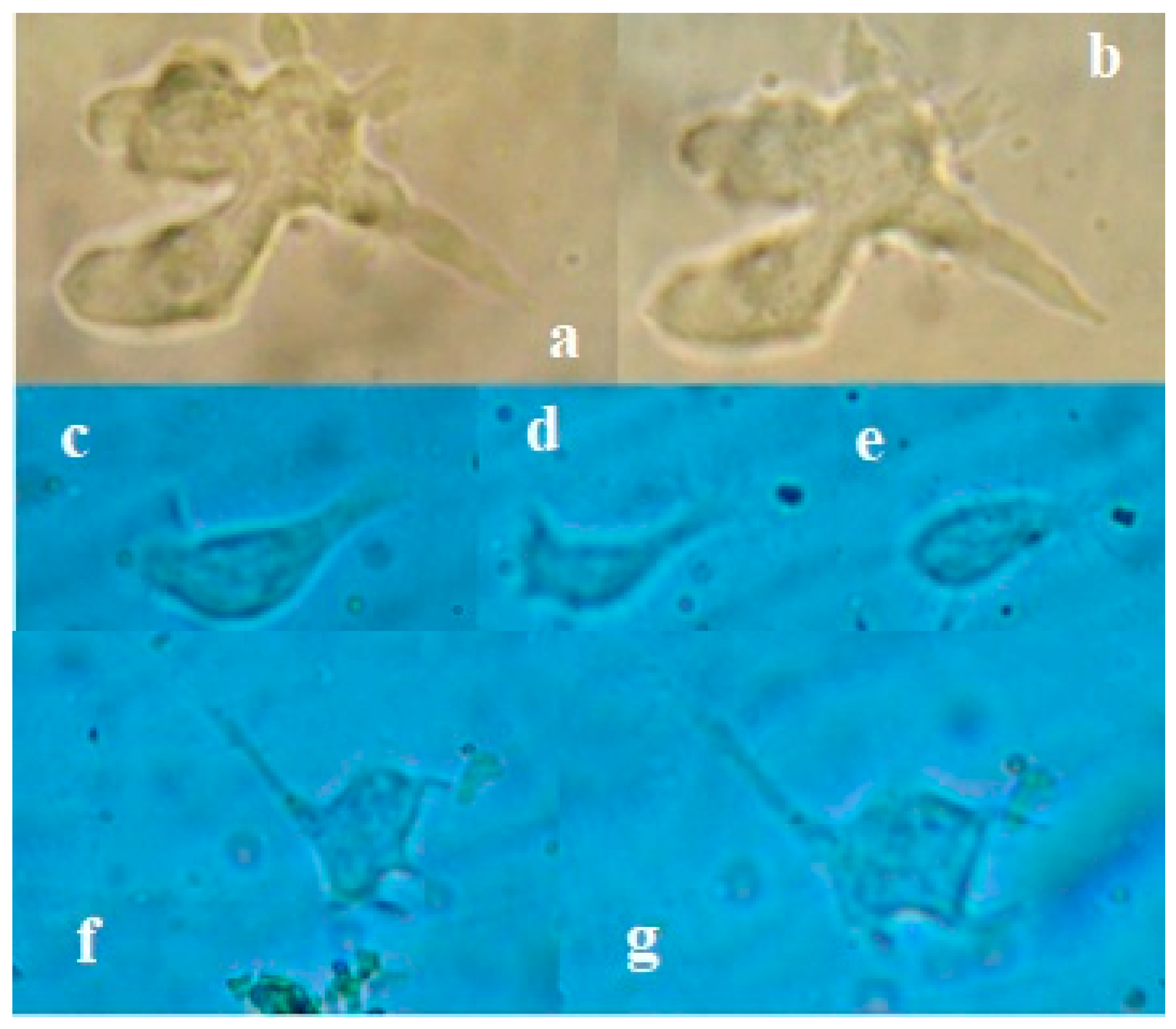
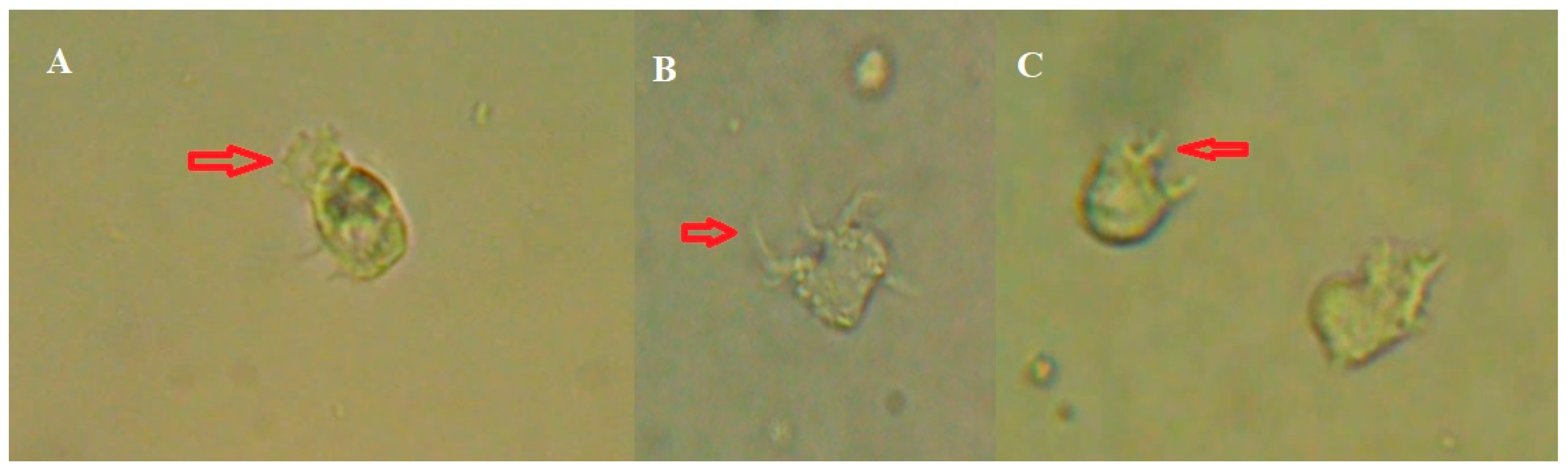
Figure 2.
Amoeba-1: a, b) Pseudopods observed in the patient's CSF. Amoeba-2: c, d, e, f, g) movement sequence of the amoeba observed in the CSF.
Figure 2.
Amoeba-1: a, b) Pseudopods observed in the patient's CSF. Amoeba-2: c, d, e, f, g) movement sequence of the amoeba observed in the CSF.
In the image below, we can see one trophozoite with a giant vacuole in axenic culture. The cysts that we see belong to axenic cultivation and the cultivation of the soil with which the girl played in her house. Figure 3
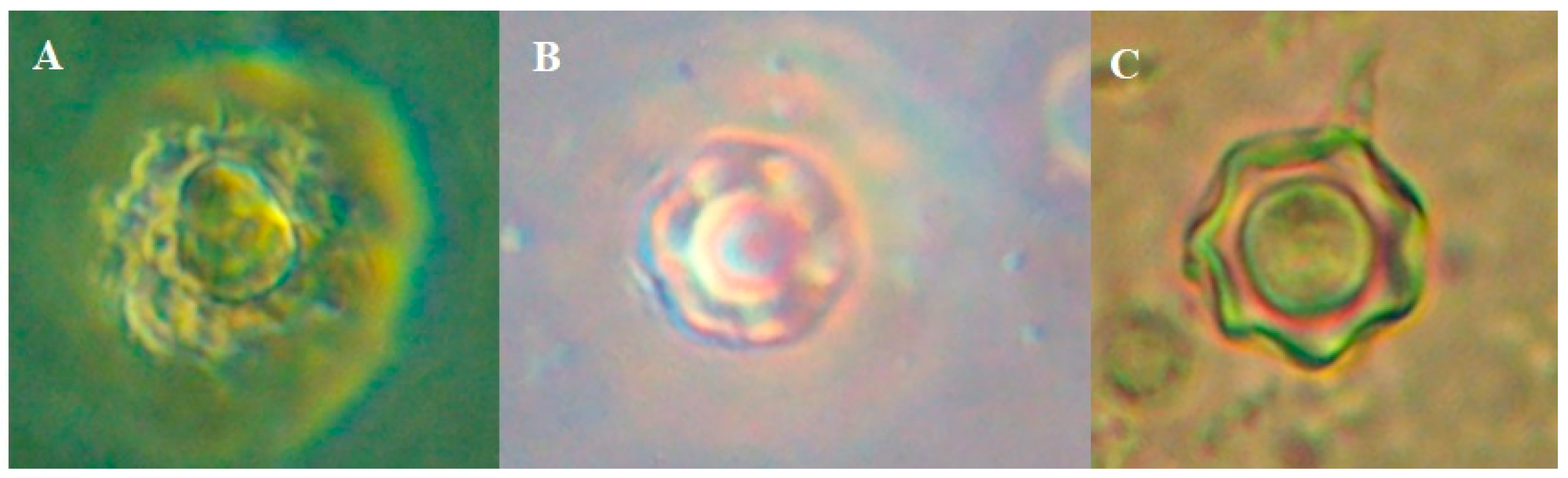
Figure 3.
A; trophozoite with a giant vacuole in axenic culture, B; Cyst in axenic from CSF (face contrast 60x) and C; Cyst in monoxenic culture (soil from home of the girl).
Figure 3.
A; trophozoite with a giant vacuole in axenic culture, B; Cyst in axenic from CSF (face contrast 60x) and C; Cyst in monoxenic culture (soil from home of the girl).
We believe that the cyst and the trophozoite observed in the images belong to the same microorganism. However, we do not have molecular evidence.
Host type: Human CSF
Locality: Lima, Peru.
Host collection: Hospital of Lima
Description: Amoeba -1 size: 50 μm in length (40–60 μm, n=10) and 50 μm (30-40 μm) in width. Ovoid cell body shape, with pseudopods long, branched, and cap hyaline.
The EUK A/B primers used amplified human DNA.
Protist 3
A 4-month-old girl with congenital hydrocephalus, with no clear history of being in contact with any water source, was preliminarily diagnosed in a pediatric hospital in Lima. CFS culture was positive and klebsiella sp was isolated. Subsequently, the presence of an amoeba in the CSF was confirmed. The amoeba did not grow in any culture medium. This protist showed several outstanding features: it did not form a cyst, did not adhere to the slide, but lysed after 2 hours, disappearing from the CSF. We believe lysis occurs because there are anaerobes microorganisms. The movement is active in a floating state, and also could change shape while floating. Cytoplasmic extensions compatible with short pseudopods in the form of plant roots are seen. With all these characteristics, we named it with the candidate name of Rhadiculamoeba anaerobica.
Figure 4.
The images have been taken under a Primus microscope at 40 X. Trophozoites are observed in a floating state; they do not form cysts nor adhere to the coverslip. A) The extension of small pseudopods and a faint hyaline layer can be seen. B) Pseudopods extension, C) Pseudopods with mini lateral extension.
Figure 4.
The images have been taken under a Primus microscope at 40 X. Trophozoites are observed in a floating state; they do not form cysts nor adhere to the coverslip. A) The extension of small pseudopods and a faint hyaline layer can be seen. B) Pseudopods extension, C) Pseudopods with mini lateral extension.
Host type: Human CSF
Locality: Lima, Peru.
Host collection: Hospital of Lima
Description: 30 μm in lenght (25–35 μm, n=30) and 20 μm in width (15–20 μm). Ovoid cell body shape. With pseudopodos long and others short and branched.
Etymology: This species is named Rhadiculamoeba anaerobica due to its morphology and its physiological condition. The protist lysed on contact with oxygen, hence, we assume that it is anaerobic. Rhadicula = Root; Anaeobica = Anaerobic
Name: Candidate Rhadiculamoeba anaerobica
The EUK A/B primers used amplified human DNA
Protist 4
A girl aged 20 months, with no history of contact with water sources, presented with fever, motor deficit, and spoke incoherently. She was taken to a pediatric hospital in Lima. To physicians, their first presumptive diagnosis was tuberculosis. However, acid fast bacilli were not observed in the CSF although an amoeba sp. could be observed. One month later, CSF cultured was positive for Mycobacterium tuberculosis in the MODS and PCR test. The amoeba looked like a leukocyte at first; after four minutes it changed its shape and increased its length. What we see under the microscope is a microorganism that changes shape. We believe that there may be two parts, although we are not very sure: the anterior part and the posterior part. In the anterior part, the structure is circular or oval. In the posterior part, an extension of the cytoplasm is observed that seems to be moving on its axis, implying that it twists forming a kind of screw with terminal filaments. It changes shape on its axis and in the same place, lengthening the posterior part and elongating the spherical part corresponding to the anterior part of the protist's structure. This anterior part seems to have a kind of cilia or cover and, apparently, direction is modified as the anterior part changes from a spherical to an oval.
Figure 5 shows the protist morphological change, before (left) and after (right) in a few minutes and a crenated erythrocyte.
Figure 5.
video sequence, where the trophozoites is seen transforming together with the erythrocyte.
Figure 5.
video sequence, where the trophozoites is seen transforming together with the erythrocyte.
Unfortunately, the identity of these protists has not been elucidated yet because they do not grow in conventional culture media for free-living protists. Cell culture was attempted only once. It is worth mentioning that they keep moving for a longer time but exponential growth was not observed.
Monoxenic medium (with an overlay of Escherichia coli on the agar) and axenic medium (liquid medium based on Proteose-peptone, glucose and yeast extract) do not seem to be optimal for the growth of these amoebic forms or they may grow under conditions devoid of oxygen.
The most commonly found morphotypes are Monotactic, Palmado, and Acanthopodia. The Vahlkampfia family appears to be a morphologically dominant group in certain areas. However, the forms observed escape the 19 morphotypes described and do not fit in any of them [
10,
11,
12]
In this document, the clinical cases are not described and, therefore, we will not delve into the treatment; however we can mention that the use of anti-amoebic and antibiotics agents usually gave good results.
We emphasize the morphological description of these protists, since they possibly belong to other classes or kingdoms and, therefore, the primers did not allow DNA amplification.
Host type: Human CSF
Locality: Lima, Peru.
Host collection: Hospital of Lima
Description: 16 μm in length (15–24 μm, n=10) and 10 μm in width (8–12 μm). Ovoid cell elongated body. With filamentous structures on its back or cytoplasmic extension.
Etymology: nameless
The EUK A/B primers used amplified human DNA