Submitted:
11 December 2025
Posted:
12 December 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
HeLa Cell Culture and Transfection
PC12 Cell Culture and Transfection
Bovine Adrenal Chromaffin Cell Culture
Measurement of [Ca²⁺]ER Changes with Aequorin
On-Line Measurement of Catecholamine Release
Single-Cell [Ca²⁺]c Measurements
Chemicals
Statistics
Results
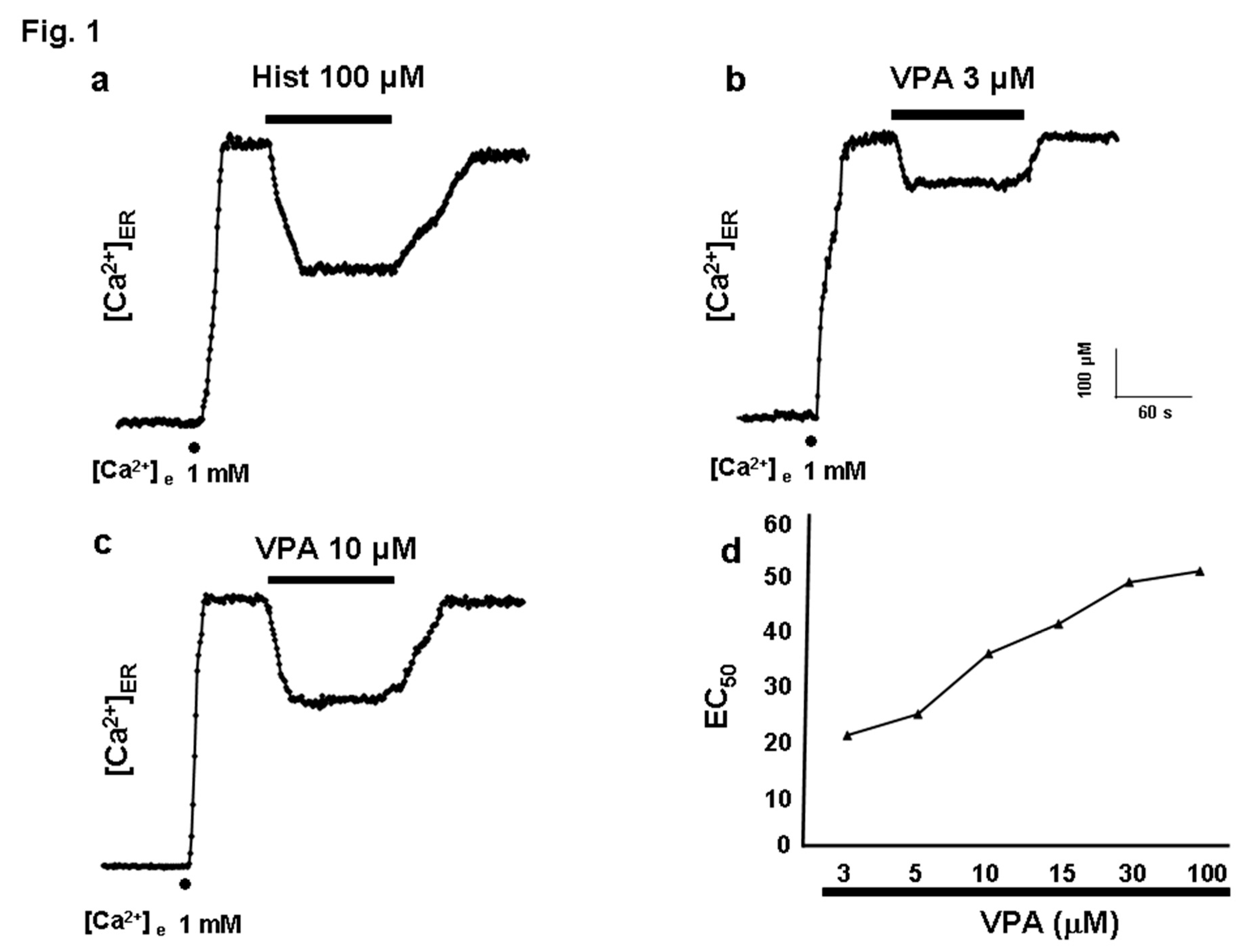
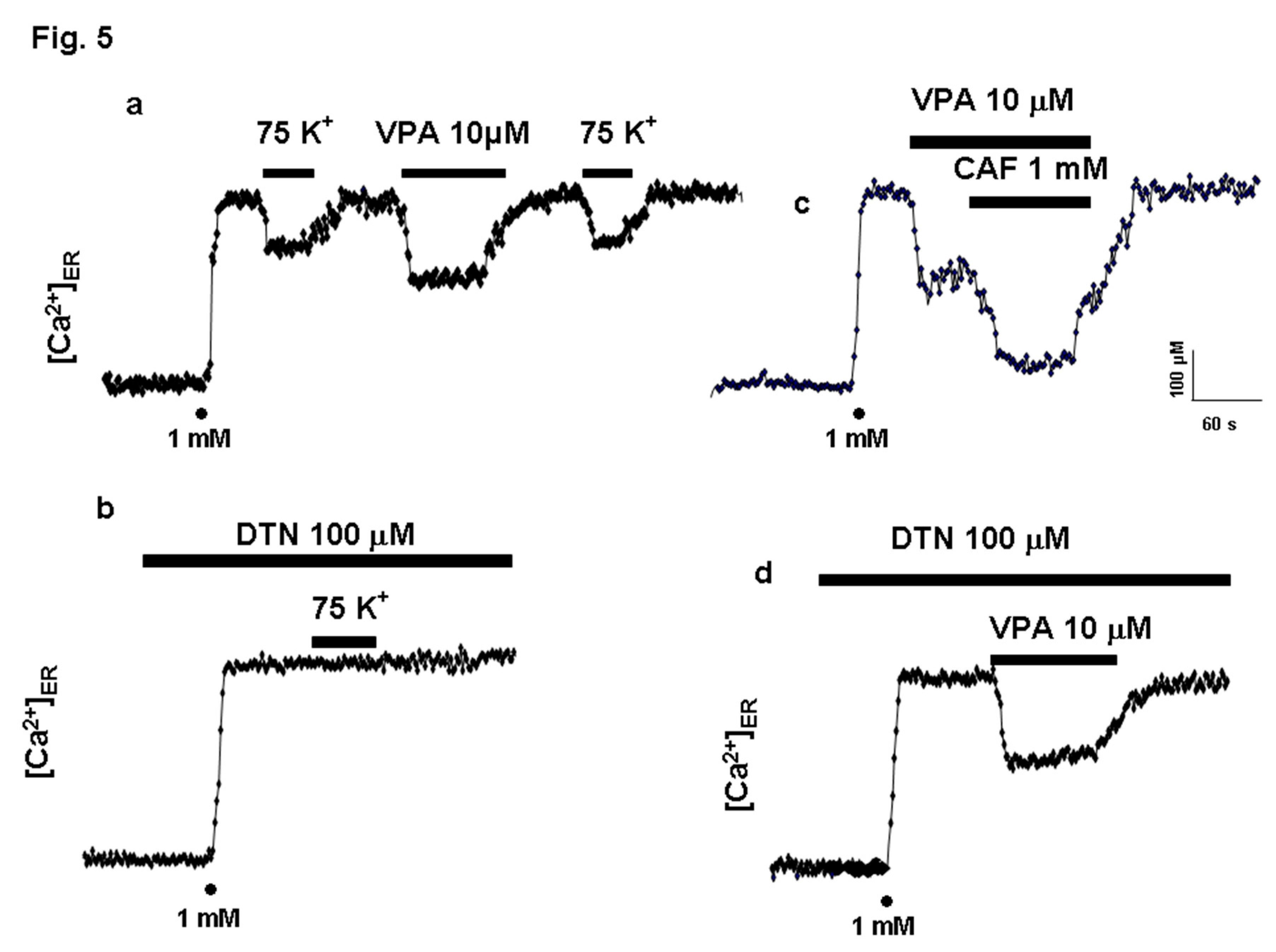
Valproic Acid Induces Ca²⁺ Release from the Endoplasmic Reticulum
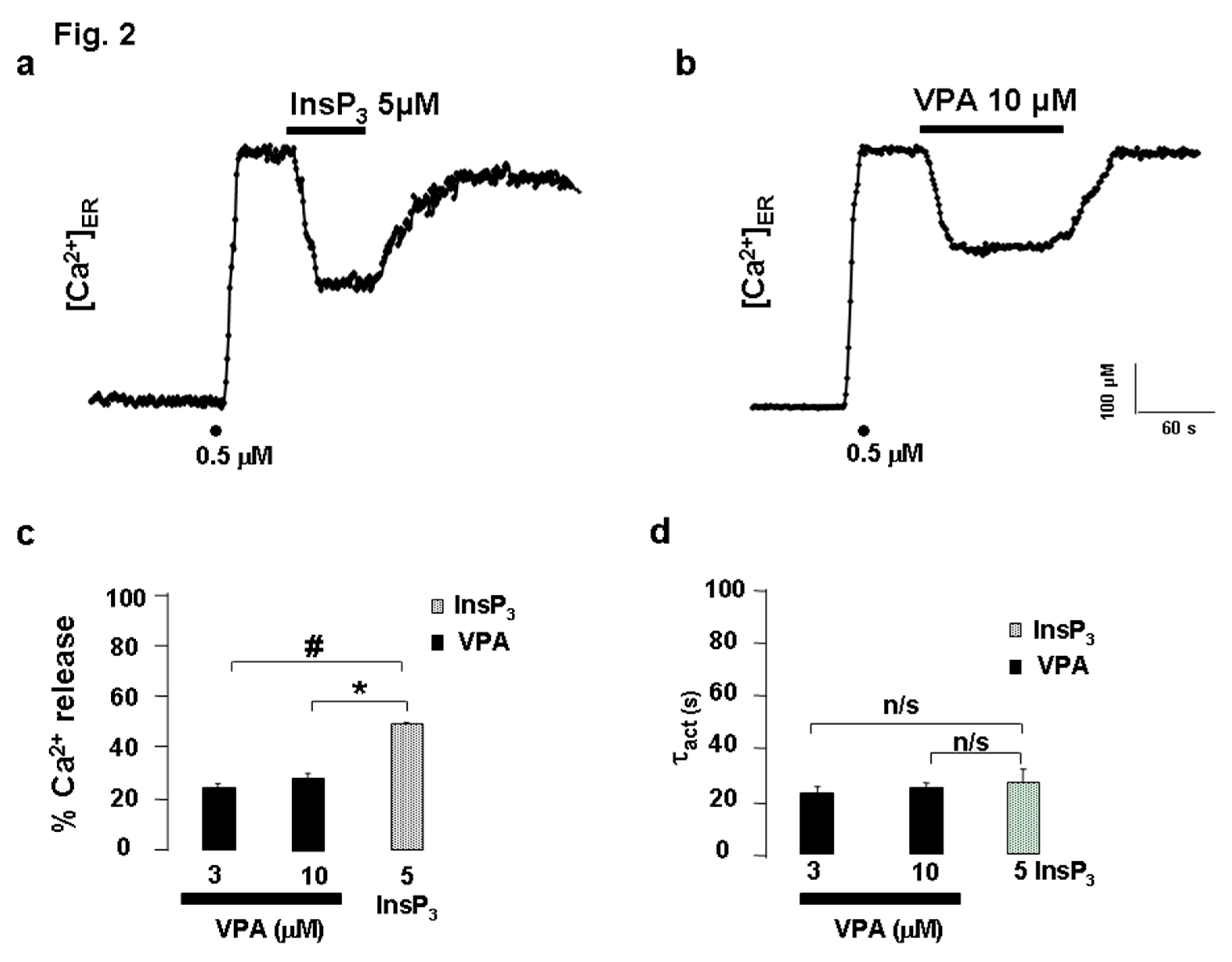
ER Ca²⁺ Release Elicited by VPA in Permeabilized HeLa Cells
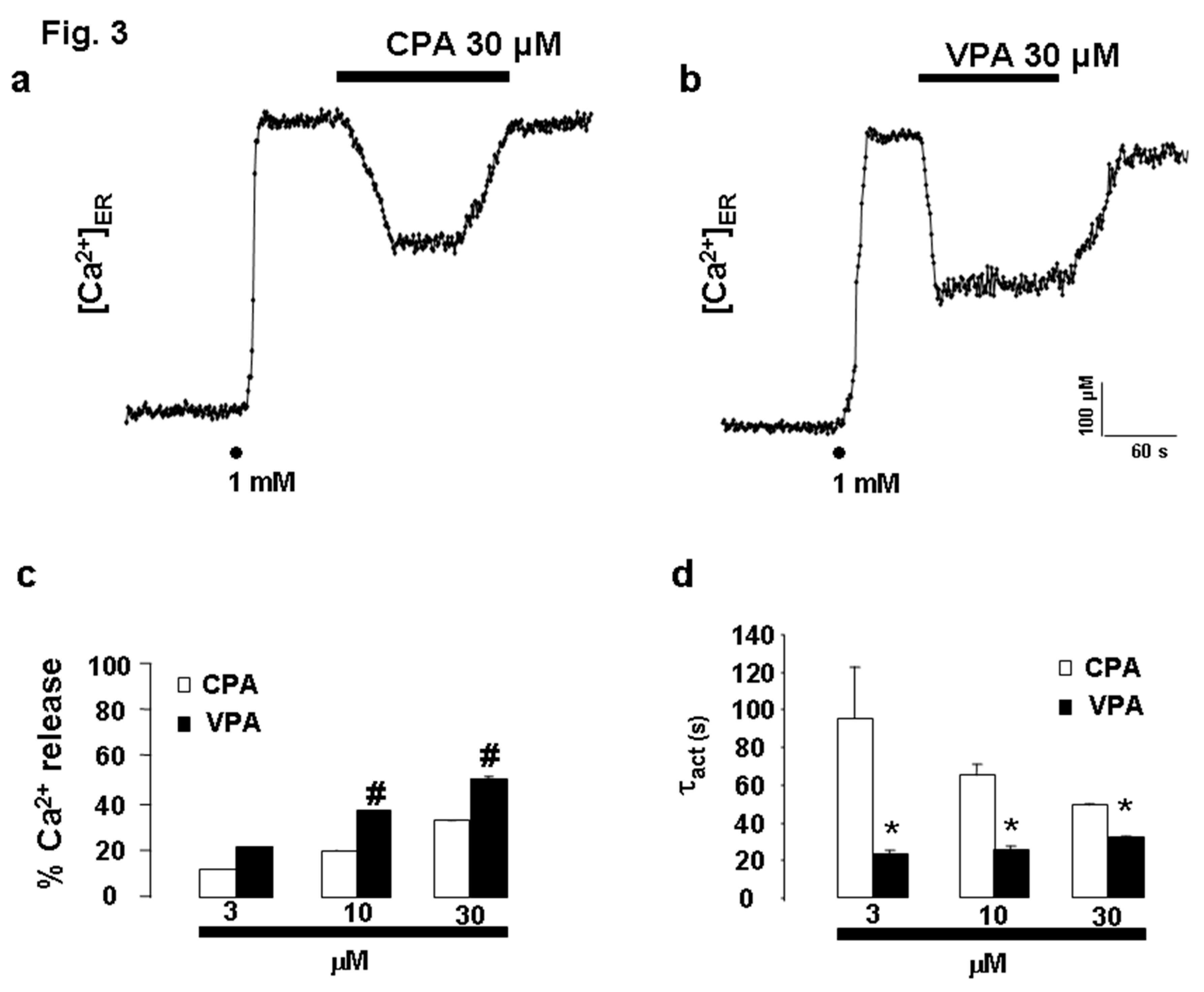
Comparison of the Kinetics of ER Ca²⁺ Release Induced by CPA and VPA
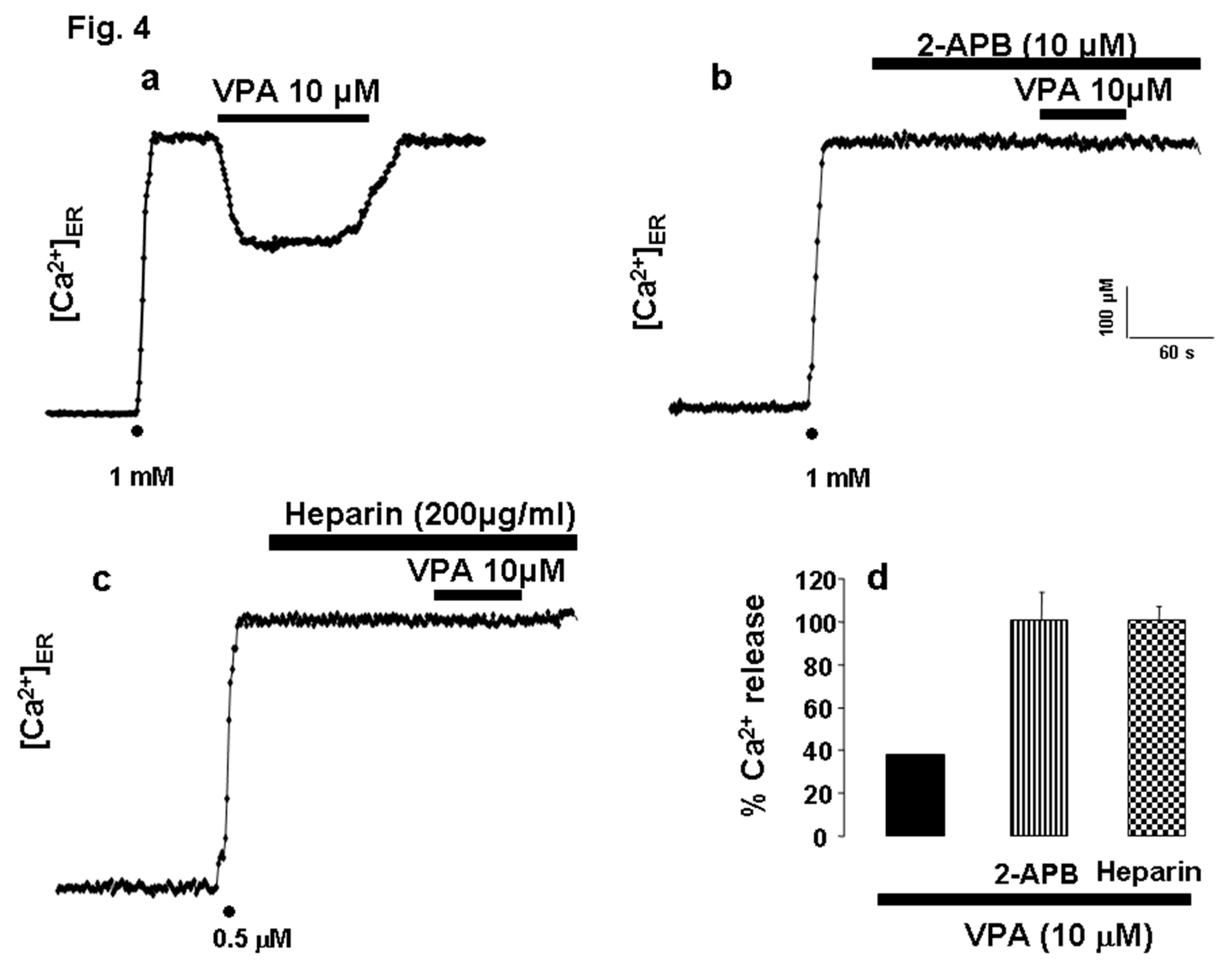
[Ca²⁺]ER Is Unaffected by 2-APB and Heparin
VPA Releases Ca²⁺ via InsP₃R in PC12 Cells
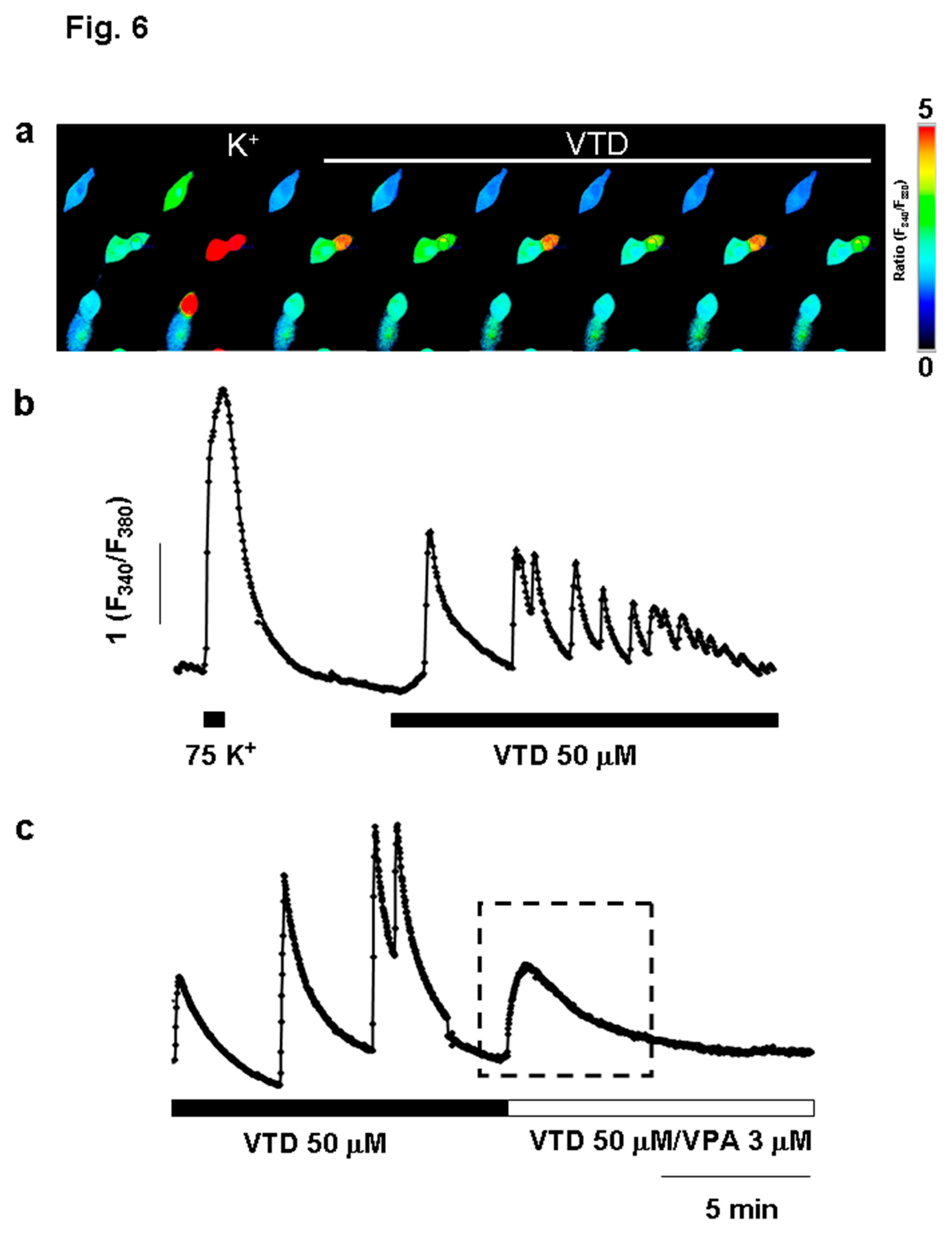
VPA Mitigates Intracellular Ca²⁺ Oscillations Induced by Veratridine
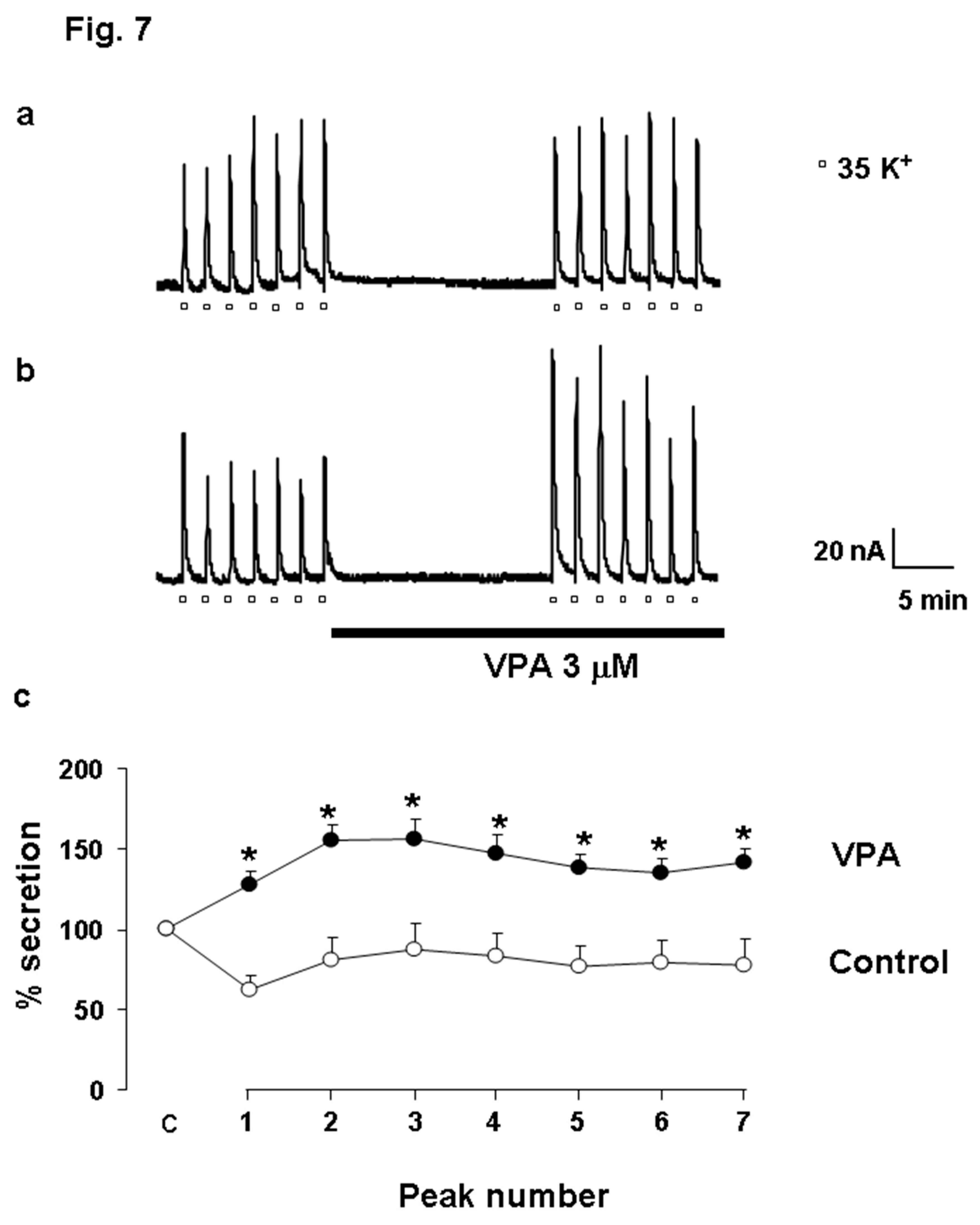
VPA Facilitates Catecholamine Release in Bovine Chromaffin Cells
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Malenka, R.C.; Nicoll, R.A. Long-term potentiation a decade of progress? Science 1999, 285, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Akimoto, T.; Kusumi, I.; Suzuki, K.; Masui, T.; Koyama, T. Effects of valproate on serotonin-induced intracellular calcium mobilization in human platelets. J Psychiatry Neurosci. 2007, 32, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Limbrick, D.D., Jr.; Rafiq, A.; DeLorenzo, R.J. Induction of spontaneous recurrent epileptiform discharges causes long term changes in intracellular calcium homeostatic mechanisms. Cell Calcium 2000, 28, 181–193. [Google Scholar] [CrossRef]
- Pal, S.; Sun, D.; Limbrick, D.; Rafiq, A.; DeLorenzo, R.J. Epileptogenesis induces long-term alterations in intracellular calcium release and sequestration mechanisms in the hippocampal neuronal culture model of epilepsy. Cell Calcium 2000, 30, 285–296. [Google Scholar] [CrossRef]
- DeLorenzo, R.J.; Sun, D.A.; Deshpande, L.S. Cellular mechanisms underlying acquired epilepsy: The calcium hypothesis of the induction and maintainance of epilepsy. Pharmacol. Therap 2005, 105, 229–266. [Google Scholar] [CrossRef]
- Ehrlich, B.E.; Kaftan, E.; Bezprozvannaya, S.; Bezprozvanny, I. The pharmacology of intracellular Ca2+ release channels. Trends Pharmacol. 1994, 15, 145–149. [Google Scholar] [CrossRef]
- Bennett, D.L.; Cheek, T.R.; Berridge, M.J.; De Smedti, H.; Parysi, J.B.; Missiaeni, L.; Bootman, M.D. Expression and function of ryanodine receptors in nonexcitable cells. J Biol. Chem. 1996, 271, 6356–6362. [Google Scholar] [CrossRef]
- Berridge, M.J. Neuronal calcium signaling. Neuron 1998, 21, 13–26. [Google Scholar] [CrossRef]
- Royero, PX; Higa, GSV; Kostecki, DS; Dos Santos, BA; Almeida, C; Andrade, KA; Kinjo, ER; Kihara, AH. Ryanodine receptors drive neuronal loss and regulate synaptic proteins during epileptogenesis. Exp Neurol. 2020, 327, 113213. [Google Scholar] [CrossRef] [PubMed]
- Capocchi, G.; Balducci, A.; Cecconi, M.; Pelli, MA.; Picchiarelli, A.; Silvestrelli, G.; Zampolini, M. Valproate-induced epileptic tonic status. Seizure 1998, 7, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Ekwuru, MO.; Cunningham, JR. Phaclofen increases GABA release from valproate treated rats. Br. J. Pharmacol. 1990, 99, 251. [Google Scholar]
- Gram, L.; Larsson, O.M.; Johnsen, A.H.; Schousboe, A. Effects of valproate, vigabatrin and aminooxyacetic acid on release of endogenous and exogenous GABA from cultured neurons. Epilepsy Res. 1988, 2, 87–95. [Google Scholar] [CrossRef]
- Kelly, K.M.; Gross, R.A.; Macdonald, R.L. Valproic acid selectively reduces the low-threshold (T) calcium current in rat nodose neurons. Neurosci. Lett. 1990, 116, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.J.; Shi, Y.; Austin, R.C.; Werstuck, G.H. Valproate protects cells from ER stress-induced lipid accumulation and apoptosis by inhibiting glycogen synthase kinase-3. J. Cell Sci. 2005, 118, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Kurita, M.; Nishino, S.; Ohtomo, K.; Rai, M.; Shirakawa, H.; Mashiko, H.; Niwa, S.; Nakahata, N. Sodium valproate at therapeutic concentrations changes Ca2+ response accompanied with its weak inhibition of protein kinase C in human astrocytoma cells. Prog Neuropsychopharmacol. Biol. Psychiatry. 2007, 31, 600–604. [Google Scholar] [CrossRef]
- Löscher, W. Basic pharmacology of valproate: A review after 35 years of clinical use for the treatment of epilepsy. CNS drugs 2002, 16, 669–694. [Google Scholar] [CrossRef]
- Luef, G.J.; Lechleitner, M.; Bauer, G.; Trinka, E.; Hengster, P. Valproic acid modulates islet cell insulin secretion: A possible mechanism of weight gain in epilepsy patients. Epilepsy Res. 2003, 55, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Vreugdenhil, M.; van Veelen, C.W.; van Rijen, P.C.; Lopes da Silva, F.H.; Wadman, W.J. Effect of valproic acid on sodium currents in cortical neurons from patients with pharmaco-resistant temporal lobe epilepsy. Epilepsy Res. 1998, 32, 309–320. [Google Scholar] [CrossRef]
- Suzuki, K.; Kusumi, I.; Akimoto, T.; Sasaki, Y.; Koyama, T. Effects of lithium and valproate on agonist-induced platelet intracellular calcium movilization: Relevance to myosin light chain kinase. Prog Neuropsychopharmacol Biol Psychiatry 2004, 28, 67–72. [Google Scholar] [CrossRef]
- Pizzo, P.; Fasolato, C.; Pozzan, T. Dynamic properties of an inositol 1,4,5-trisphosphate- and thapsigargine-insensitive calcium pool in mammalian cell lines. J Cell Biol. 1997, 136, 355–366. [Google Scholar] [CrossRef]
- Clementi, E.; Scheer, H.; Zacchetti, D.; Fasolato, C.; Pozzan, T.; Meldolesi, J. Receptor-activated Ca2+ influx. Two independently regulated mechanisms of influx stimulation coexist in neurosecretory PC12 cells. J Biol Chem. 1992, 267, 2164–2172. [Google Scholar] [CrossRef]
- Ehrlich, B.E.; Kaftan, E.; Bezprozvannaya, S.; Bezprozvanny, I. The pharmacology of intracellular Ca2+ release channels. Trends Pharmacol. 1994, 15, 145–149. [Google Scholar] [CrossRef]
- Fasolato, C.; Zottini, M.; Clementi, E.; Zacchetti, D.; Meldolesi, J.; Pozzan, T. Intracellular Ca2+ pools in PC12 cells. J. Biol. Chem. 1991, 266, 20159–20167. [Google Scholar] [CrossRef]
- García-Sancho, J.; Montero, M.; Alvarez, J. Ca2+-induced Ca2+ release in chromaffin cells seen from inside the ER with targeted aequorin. J Cell Biol. 1999, 144, 241–254. [Google Scholar]
- Montero, M.; Alvarez, J.; Scheenen, W.J.J.; Rizzuto, R.; Meldolesi, J.; Pozzan, T. Ca2+ Homeostasis in the endoplasmic reticulum: Coexistence of high and low [Ca2+] subcompartments in intact HeLa cells. J. Cell. Biol. 1997, 139, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Moreno ALobatón, C.D.; SantoDomingo, J.; Vay, L.; Hernandez-SanMiguel, E.; Rizzuto, R.; Montero, M.; Alvarez, J. Calcium dynamics in catecholamine containing secretory vesicles. Cell Calcium 2005, 37, 555–564. [Google Scholar] [CrossRef]
- Mori, F.; Okada, M.; Tomiyama, M.; Kaneko, S.; Wakabayashi, K. Effects of ryanodine receptor activation on neurotransmitter release and neuronal cell death following kainic acid-induced status epilepticus. Epilepsy Res. 2005, 65, 59–70. [Google Scholar] [CrossRef]
- Zima, A.V.; Bare, D.J.; Mignery, G.A.; Blatter, L.A. IP3-dependent nuclear Ca2+ signalling in the mammalian heart. J Physiol. 2007, 584, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Franco, J.; Fill, M.; Mignery, G.A. Isoform-specific function of single inositol 1,4,5-trisphosphate receptor channels. Biophysic. J. 1998, 75, 834–839. [Google Scholar] [CrossRef]
- Rizzuto, R. Intracellular Ca2+ pools in neuronal signalling. Curr Opin Neurobiol. 2001, 11, 306–311. [Google Scholar] [CrossRef]
- Yamamoto, R.; Yanagita, T.; Kobayashi, H.; Yokoo, H.; Wada, A. Up-regulation of sodium channel subunit mRNAs and their cell surface expression by antiepileptic valproic acid: Activation of calcium channel and catecholamine secretion in adrenal chromaffin cells. J. Neurochem. 1997, 68, 1655–1662. [Google Scholar] [CrossRef]
- Zona, C.; Avoli, M. Effects induced by the antiepileptic drug valproic acid upon the ionic currents recorded in rat neocortical neurons in cell culture. Exp. Brain. Res. 1990, 81, 313–317. [Google Scholar] [CrossRef]
- Otoom, S.A.; Alkadhi, K.A. Valproic acid intensifies epileptiform activity in the hippocampal pyramidal neurons. Neurosci Res. 1999, 35, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, J.; Montero, M. Measuring Ca2+ in the endoplasmic reticulum with aequorin. Cell Calcium 1999, 32, 251–260. [Google Scholar] [CrossRef]
- Chen, C.A.; Okayama, H. Calcium phosphate-mediated gene transfer: A highly efficient transfection system for stably transforming cells with plasmid DNA. Biotechniques 1988, 6, 632–638. [Google Scholar] [PubMed]
- Díaz-Prieto, N.; Herrera-Peco, I.; de Diego, A.M.; Ruiz-Nuño, A.; Gallego-Sandín, S.; López, M.G.; García, A.G.; Cano-Abad, M.F. Bcl2 mitigates Ca2+ entry and mitochondrial Ca2+ overload through downregulation of L-type Ca2+ channels in PC12 cells. Cell Calcium 2008, 44, 339–352. [Google Scholar] [CrossRef]
- Moro, M.A.; Lopez, M.G.; Gandia, L.; Michelena, P.; Garcia, A.G. Separation and culture of living adrenal- and noradrenalina-containing cells from bovine adrenal medullae. Anal. Biochem. 1990, 185, 243–248. [Google Scholar] [CrossRef]
- Borges, R.; Sala, F.; Garcia, A.G. Continuous monitoring of catecholamine release from perfused cat adrenals. J. Neurosci. Methods 1986, 16, 289–300. [Google Scholar] [CrossRef]
- Cano-Abad, M.F.; Lopez, M.G.; Hernandez-Guijo, J.M.; Zapater, P.; Gandia, L.; Sanchez-Garcia, P.; Garcia, A.G. Effects of the neuroprotectant lubeluzole on the cytotoxic actions of veratridine, barium, ouabain and 6-hydroxydopamine in chromaffin cells. Br. J. Pharmacol. 1998, 124, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Otoom, S.; Tian, L.M.; Alkadhi, K.A. Veratridine-treated brain slices: A cellular model for epileptiform activity. Brain Res. 1998, 789, 150–156. [Google Scholar] [CrossRef]
- Otoom, S.A.; Alkadhi, K.A. Epileptiform activity of veratridine model in rat brain slices: Effects of antiepileptic drugs. Epilepsy Res. 2000, 38, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Conceiçao, I.M.; Lebrun, I.; Cano-Abad, M.F.; Gandía, L.; Hernández-Guijo, J.M.; López, M.G.; Villarroya, M.; Jurkiewicz, A.; García, A.G. Synergism between toxin gamma from the brazilian scorpion Tityus serrulatus and veratridine in chromaffin cells. Am. J. Physiol. 1998, 274, 1745–1754. [Google Scholar] [CrossRef] [PubMed]
- Cano-Abad, M.F.; Villarroya, M.; Garcia, A.G.; Gabilan, N.H.; Lopez, M.G. Calcium entry through L-type calcium channels causes mitochondrial disruption and chromaffin cell death. J. Biol. Chem. 2001, 276, 39695–39704. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



