Submitted:
01 October 2025
Posted:
02 October 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Patients
Definitions
Statistical Analysis
Results
Patient Demographics and Clinical Characteristics
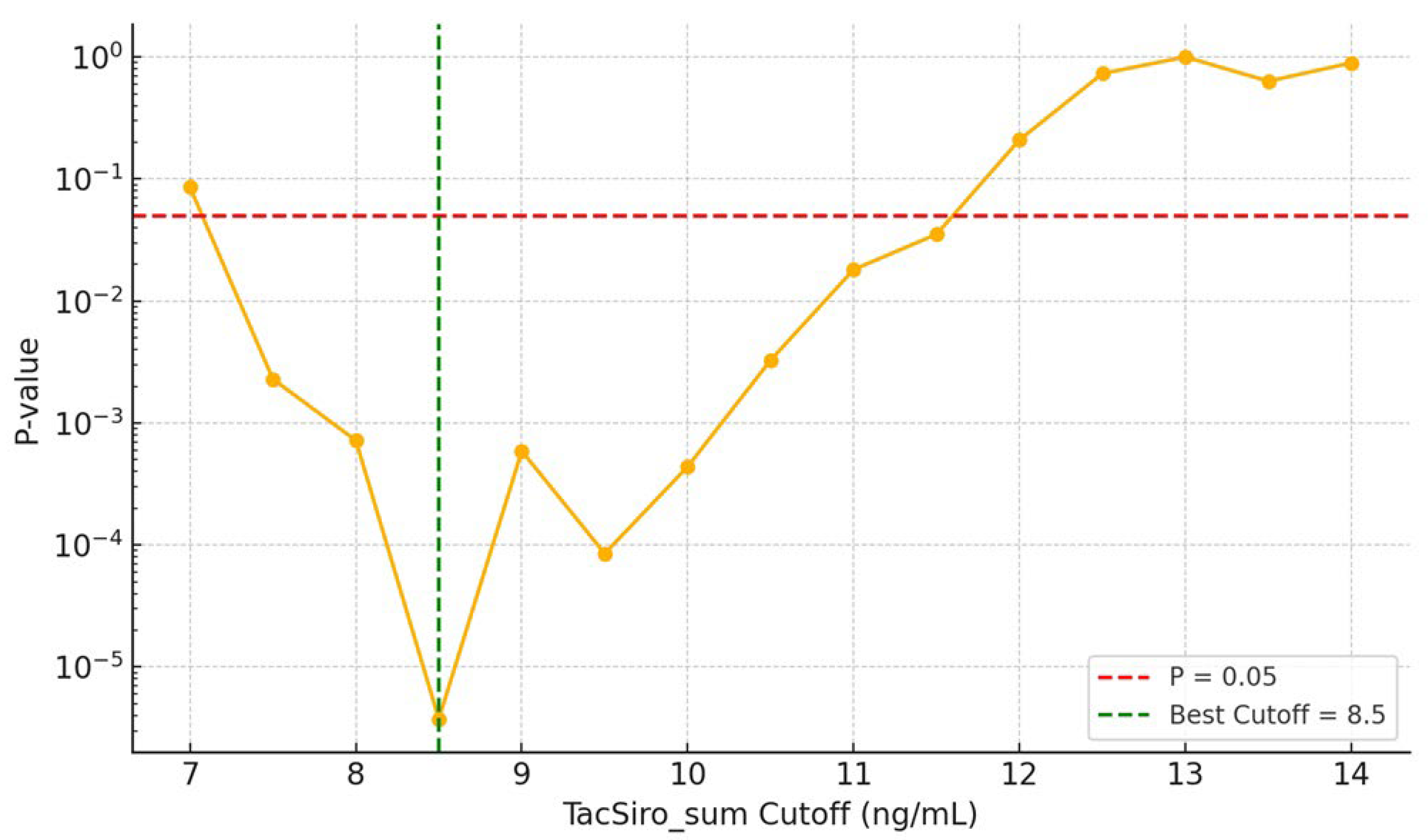
Risk of Acute Rejection by Combined Drug Exposure
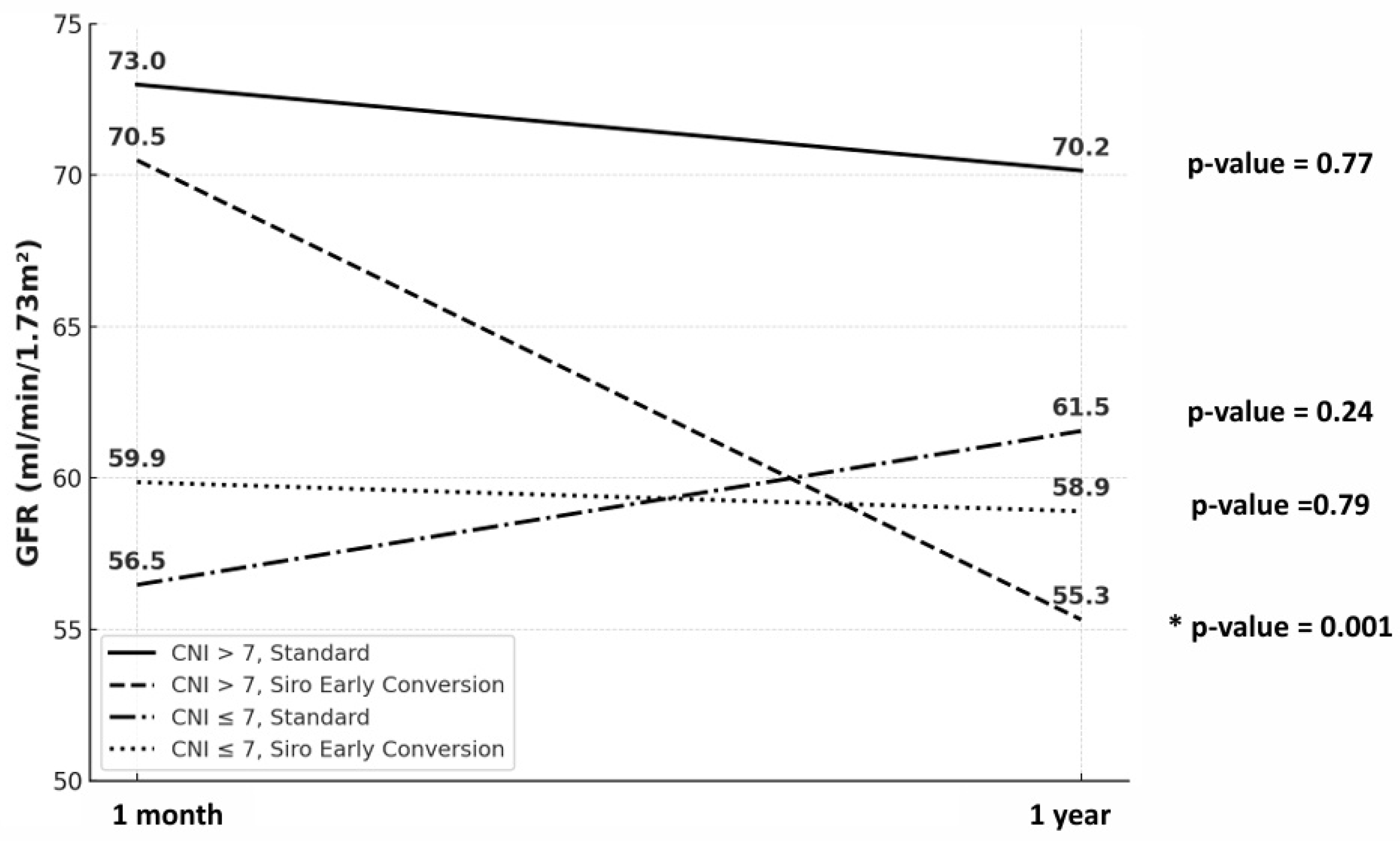
Renal Function and Infection Outcomes by Treatment Strategy
Discussion
Conclusion
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflict of Interest
References
- Vitko, S.; Klinger, M.; Salmela, K.; Wlodarczyk, Z.; Tyden, G.; Senatorski, G.; Ostrowski, M.; Fauchald, P.; Kokot, F.; Stefoni, S.; et al. Two corticosteroid-free regimens-tacrolimus monotherapy after basiliximab administration and tacrolimus/mycophenolate mofetil-in comparison with a standard triple regimen in renal transplantation: results of the Atlas study. Transplantation 2005, 80, 1734-41. [CrossRef]
- Farouk, S.S.; Rein, J.L. The Many Faces of Calcineurin Inhibitor Toxicity-What the FK? Adv Chronic Kidney Dis 2020, 27, 56-66. [CrossRef]
- Budde, K.; Prashar, R.; Haller, H.; Rial, M.C.; Kamar, N.; Agarwal, A.; de Fijter, J.W.; Rostaing, L.; Berger, S.P.; Djamali, A.; et al. Conversion from Calcineurin Inhibitor- to Belatacept-Based Maintenance Immunosuppression in Renal Transplant Recipients: A Randomized Phase 3b Trial. J Am Soc Nephrol 2021, 32, 3252-3264. [CrossRef]
- Karpe, K.M.; Talaulikar, G.S.; Walters, G.D. Calcineurin inhibitor withdrawal or tapering for kidney transplant recipients. Cochrane Database Syst Rev 2017, 7, Cd006750. [CrossRef]
- Cuadrado-Payán, E.; Diekmann, F.; Cucchiari, D. Medical Aspects of mTOR Inhibition in Kidney Transplantation. International Journal of Molecular Sciences 2022, 23, 7707. [CrossRef]
- Shihab, F.; Qazi, Y.; Mulgaonkar, S.; McCague, K.; Patel, D.; Peddi, V.R.; Shaffer, D. Association of Clinical Events With Everolimus Exposure in Kidney Transplant Patients Receiving Low Doses of Tacrolimus. Am J Transplant 2017, 17, 2363-2371. [CrossRef]
- Mjörnstedt, L.; Sørensen, S.S.; von zur Mühlen, B.; Jespersen, B.; Hansen, J.M.; Bistrup, C.; Andersson, H.; Gustafsson, B.; Undset, L.H.; Fagertun, H.; et al. Improved Renal Function After Early Conversion From a Calcineurin Inhibitor to Everolimus: a Randomized Trial in Kidney Transplantation. American Journal of Transplantation 2012, 12, 2744-2753. [CrossRef]
- Cortazar, F.; Molnar, M.Z.; Isakova, T.; Czira, M.E.; Kovesdy, C.P.; Roth, D.; Mucsi, I.; Wolf, M. Clinical outcomes in kidney transplant recipients receiving long-term therapy with inhibitors of the mammalian target of rapamycin. Am J Transplant 2012, 12, 379-87. [CrossRef]
- Manzia, T.M.; Carmellini, M.; Todeschini, P.; Secchi, A.; Sandrini, S.; Minetti, E.; Furian, L.; Spagnoletti, G.; Pisani, F.; Piredda, G.B.; et al. A 3-month, Multicenter, Randomized, Open-label Study to Evaluate the Impact on Wound Healing of the Early (vs Delayed) Introduction of Everolimus in De Novo Kidney Transplant Recipients, With a Follow-up Evaluation at 12 Months After Transplant (NEVERWOUND Study). Transplantation 2020, 104, 374-386. [CrossRef]
- Ueno, P.; Felipe, C.; Ferreira, A.; Cristelli, M.; Viana, L.; Mansur, J.; Basso, G.; Hannun, P.; Aguiar, W.; Tedesco Silva, H., Jr.; et al. Wound Healing Complications in Kidney Transplant Recipients Receiving Everolimus. Transplantation 2017, 101, 844-850. [CrossRef]
- Han, A.; Jo, A.J.; Kwon, H.; Kim, Y.H.; Lee, J.; Huh, K.H.; Lee, K.W.; Park, J.B.; Jang, E.; Park, S.C.; et al. Optimum tacrolimus trough levels for enhanced graft survival and safety in kidney transplantation: a retrospective multicenter real-world evidence study. Int J Surg 2024, 110, 6711-6722. [CrossRef]
- Kim, J.M.; Kwon, H.E.; Han, A.; Ko, Y.; Shin, S.; Kim, Y.H.; Lee, K.W.; Park, J.B.; Kwon, H.; Min, S. Risk Prediction and Management of BKPyV-DNAemia in Kidney Transplant Recipients: A Multicenter Analysis of Immunosuppressive Strategies. Transpl Int 2025, 38, 14738. [CrossRef]
- Berger, S.P.; Sommerer, C.; Witzke, O.; Tedesco, H.; Chadban, S.; Mulgaonkar, S.; Qazi, Y.; de Fijter, J.W.; Oppenheimer, F.; Cruzado, J.M.; et al. Two-year outcomes in de novo renal transplant recipients receiving everolimus-facilitated calcineurin inhibitor reduction regimen from the TRANSFORM study. Am J Transplant 2019, 19, 3018-3034. [CrossRef]
- Huh, K.H.; Lee, J.G.; Ha, J.; Oh, C.K.; Ju, M.K.; Kim, C.D.; Cho, H.R.; Jung, C.W.; Lim, B.J.; Kim, Y.S. De novo low-dose sirolimus versus mycophenolate mofetil in combination with extended-release tacrolimus in kidney transplant recipients: a multicentre, open-label, randomized, controlled, non-inferiority trial. Nephrol Dial Transplant 2017, 32, 1415-1424. [CrossRef]
- Zou, Z.Y.; Dai, L.R.; Hou, Y.B.; Yu, C.Z.; Chen, R.J.; Chen, Y.Y.; Liu, B.; Shi, H.B.; Gong, N.Q.; Chen, Z.S.; et al. Sirolimus in combination with low-dose extended-release tacrolimus in kidney transplant recipients. Front Med (Lausanne) 2023, 10, 1281939. [CrossRef]
- Rangan, G.K. Sirolimus-associated proteinuria and renal dysfunction. Drug Saf 2006, 29, 1153-61. [CrossRef]
- A, M. Immunosuppressive Agents; American College of Clinical Pharmacy: Lenexa, KS, 2011.
- Halloran, P.F. Immunosuppressive drugs for kidney transplantation. N Engl J Med 2004, 351, 2715-29. [CrossRef]
- Hernández, D.; Hernández, D.; Martínez, D.; Martínez, D.; Gutiérrez, E.; Gutiérrez, E.; López, V.; López, V.; Gutiérrez, C.; Gutiérrez, C.; et al. CLINICAL EVIDENCE ON THE USE OF ANTI-mTOR DRUGS IN RENAL TRANSPLANTATION. Nefrología (English Edition) 2011, 31, 27-34. [CrossRef]
- Fantus, D.; Rogers, N.M.; Grahammer, F.; Huber, T.B.; Thomson, A.W. Roles of mTOR complexes in the kidney: implications for renal disease and transplantation. Nat Rev Nephrol 2016, 12, 587-609. [CrossRef]
- Mallat, S.G.; Tanios, B.Y.; Itani, H.S.; Lotfi, T.; McMullan, C.; Gabardi, S.; Akl, E.A.; Azzi, J.R. CMV and BKPyV Infections in Renal Transplant Recipients Receiving an mTOR Inhibitor-Based Regimen Versus a CNI-Based Regimen: A Systematic Review and Meta-Analysis of Randomized, Controlled Trials. Clin J Am Soc Nephrol 2017, 12, 1321-1336. [CrossRef]
- Shen, C.L.; Wu, B.S.; Lien, T.J.; Yang, A.H.; Yang, C.Y. BK Polyomavirus Nephropathy in Kidney Transplantation: Balancing Rejection and Infection. Viruses 2021, 13. [CrossRef]
- Davis, S.; Gralla, J.; Klem, P.; Tong, S.; Wedermyer, G.; Freed, B.; Wiseman, A.; Cooper, J.E. Lower tacrolimus exposure and time in therapeutic range increase the risk of de novo donor-specific antibodies in the first year of kidney transplantation. American Journal of Transplantation 2018, 18, 907-915. [CrossRef]
- Larsson, P.; Englund, B.; Ekberg, J.; Felldin, M.; Broecker, V.; Mjornstedt, L.; Baid-Agrawal, S. Difficult-to-Treat Rejections in Kidney Transplant Recipients: Our Experience with Everolimus-Based Quadruple Maintenance Therapy. J Clin Med 2023, 12. [CrossRef]


| Variables | Total (N = 8,027) |
Standard (N = 7,791) |
Early conversiona (N = 236) |
p-value |
|---|---|---|---|---|
| Mean age, years | 45.5 ± 13.9 | 45.4 ± 13.9 | 49.2 ± 12.2 | <0.001 |
| Male sex | 4,650 (57.9%) | 4,510 (57.9%) | 140 (59.3%) | 0.70 |
| Body mass index, kg/m2 | 23.0 ± 28.8 | 23.0 ± 29.2 | 22.7 ± 3.8 | 0.44 |
| Diabetes | 1,651 (20.6%) | 1,586 (20.4%) | 65 (27.5%) | 0.009 |
| Pre-transplant PRA class I, % | 13.2 ± 25.8 | 13.0 ± 25.7 | 17.5 ± 29.2 | 0.061 |
| Pre-transplant PRA class II, % | 12.2 ± 25.4 | 12.1 ± 25.2 | 15.8 ± 28.1 | 0.1 |
| HLA mismatch | 3.3 ± 1.5 | 3.3 ± 1.5 | 3.4 ± 1.4 | 0.26 |
| Desensitization | 171 (14.6%) | 140 (18.0%) | 31 (13.1%) | 0.54 |
| Donor age, years | 44.5 ± 13.3 | 44.5 ± 13.3 | 45.8 ± 12.1 | 0.11 |
| Preemptive transplantation | 1,454 (18.1%) | 1,403 (18.0%) | 51 (21.6%) | 0.18 |
| Induction | ||||
| No induction | 273 (3.4%) | 258 (3.3%) | 15 (6.4%) | <0.001 |
| Basiliximab | 6,151 (76.6%) | 6,001 (77.0%) | 150 (63.6%) | <0.001 |
| Anti-thymocyte globulin | 1,601 (20.0%) | 1,530 (19.7%) | 71 (30.1%) | <0.001 |
| ABO incompatible | 1,184 (14.8%) | 1,154 (14.8%) | 30 (12.7%) | 0.42 |
| Pre-transplant B flow, positive | 261 (3.3%) | 250 (3.2%) | 11 (4.7%) | 0.33 |
| Pre-transplant T flow, positive | 212 (2.6%) | 207 (2.7%) | 5 (2.1%) | 0.69 |
| Pre-transplant DSA, positive | 916 (11.4%) | 880 (11.3%) | 36 (15.3%) | 0.08 |
| BK virus at 3 months post-transplant, positive | 733 (9.1%) | 693 (8.9%) | 40 (16.9%) | <0.001 |
|
Categorical variables are reported as counts (percentages). Continuous variables are reported as means ± standard deviations. Abbreviations: PRA, panel reactive antibody; HLA, human leukocyte antigen; DSA, donor-specific antibody. aStandard group refers indicates maintenance tacrolimus and MMF; the early conversion group included patients converted from MMF to sirolimus within 3 months post-transplant and maintained on sirolimus for at least 1 year. | ||||
| Variables | Total (N = 1,180) |
Standard group (N = 944) |
Early conversiona (N = 236) |
SMD |
|---|---|---|---|---|
| Mean age, years | 48.53 ± 13.68 | 48.38 ± 14.02 | 49.16 ± 12.21 | 0.39 |
| Male sex | 681 (57.7%) | 541 (57.3%) | 140 (59.3%) | 0.04 |
| Body mass index, kg/m2 | 22.51 ± 3.76 | 22.48 ± 3.76 | 22.66 ± 3.76 | 0.04 |
| Diabetes | 297 (25.2%) | 232 (24.6%) | 65 (27.5%) | 0.06 |
| Pre-transplant PRA class I, % | 17.40 ± 29.3 | 17.38 ± 29.3 | 17.47 ± 29.2 | 0.02 |
| Pre-transplant PRA class II, % | 15.18 ± 28.0 | 15.08 ± 28.0 | 15.77 ± 28.1 | 0.02 |
| HLA mismatch | 3.25 ± 1.51 | 3.21 ± 1.52 | 3.38 ± 1.45 | |
| Desensitization | 171 (14.5%) | 140 (20.2%) | 31 (13.1%) | 0.07 |
| Donor age, years | 45.30 ± 12.87 | 45.19 ± 13.07 | 45.78 ± 12.05 | 0.04 |
| Preemptive transplantation | 242 (20.5%) | 191 (20.2%) | 51 (21.6%) | 0.03 |
| Induction | 0.05 | |||
| No induction | 22 (1.9%) | 7 (0.7%) | 15 (6.4%) | - |
| Basiliximab | 850 (72.0%) | 700 (74.2%) | 150 (63.6%) | - |
| Anti-thymocyte globulin | 279 (23.6%) | 209 (22.1%) | 70 (29.7%) | - |
| ABO incompatible | 170 (14.4%) | 140 (14.8%) | 30 (12.7%) | 0.06 |
| Pre-transplant B flow, positive | 80 (6.8%) | 69 (7.3%) | 11 (4.7%) | 0.01 |
| Pre-transplant T flow, positive | 37 (3.1%) | 32 (3.4%) | 5 (2.1%) | 0.06 |
| Pre-transplant DSA, positive | 137 (11.6%) | 108 (11.4%) | 29 (12.3%) | 0.04 |
| BK virus at 3 months post-transplant, positive | 230 (19.5%) | 190 (20.1%) | 40 (16.9%) | 0.06 |
| Rejection at 3–12 months post-transplant | 45 (7.5%) | 27 (2.9%) | 18 (7.6%) | 0.21/ 0.001b |
| Variables | Total (N = 8,027) |
Standard (N = 7,791) |
Early conversiona (N = 236) | p-value |
|---|---|---|---|---|
| Infection-related admission | 200 (16.9%) | 146 (15.5%) | 54 (22.9%) | 0.009 |
| PCP | 26 (2.2%) | 22 (2.3%) | 4 (1.7%) | 0.729 |
| BK viremia (3–12 months) | 74 (6.3%) | 64 (6.8%) | 10 (4.2%) | 0.155 |
| Abbreviations: PCP, Pneumocystis jirovecii pneumonia. aStandard group indicates maintenance on tacrolimus and MMF; the early conversion group included patients converted from MMF to sirolimus within 3 months post-transplant and maintained on sirolimus for at least 1 year. | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




