Submitted:
09 June 2025
Posted:
09 June 2025
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Milestones in Yellow Fever Vaccine Development
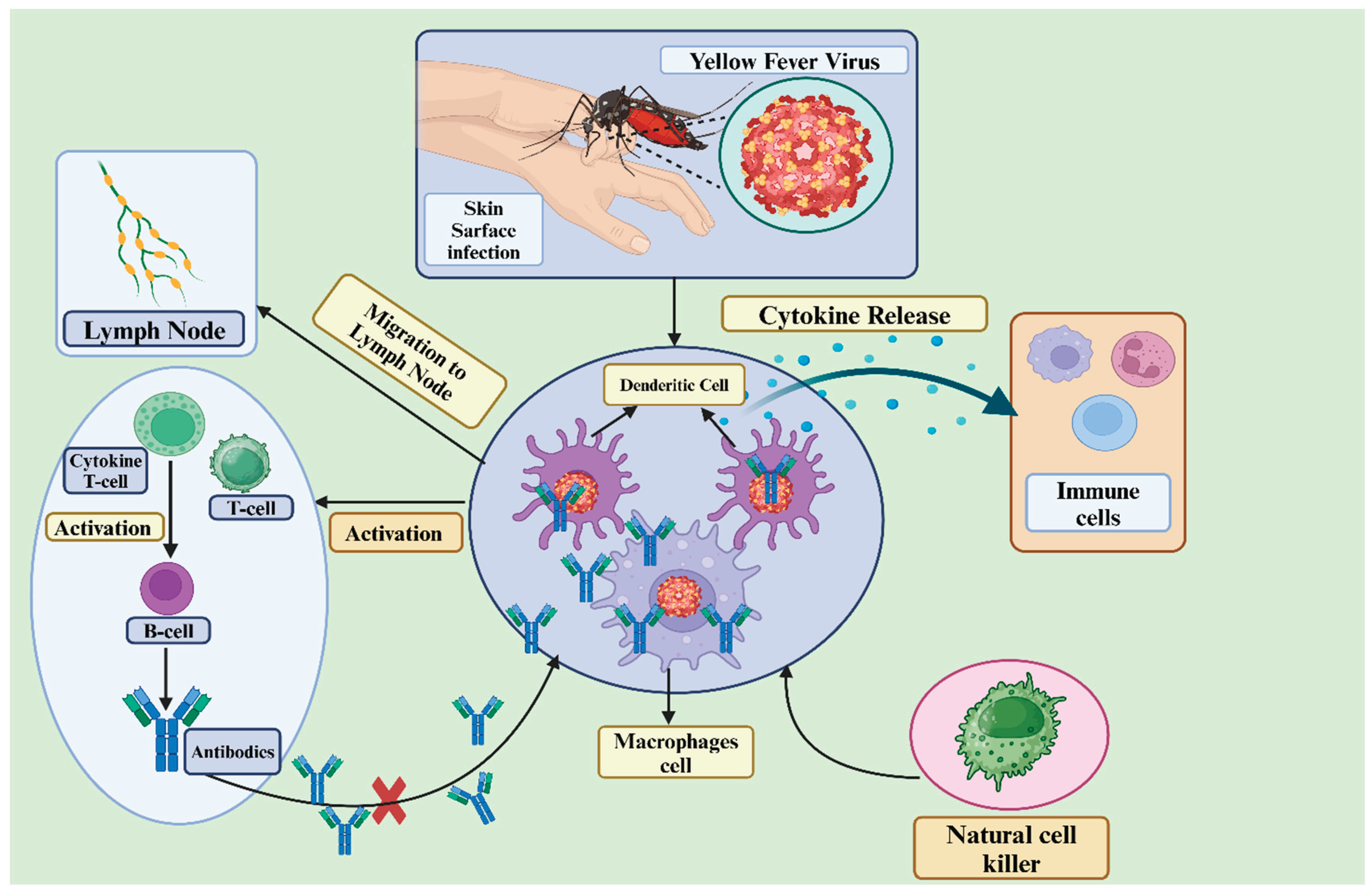
3. Yellow Fever Vaccine Immunology
3.1. Mechanisms of Immune Response to Yellow Fever Virus
3.1.1. Innate Immune Responses to Vaccination with 17D:
3.1.2. Adaptive Immune Response
3.2. Immune Memory and Duration of Protection
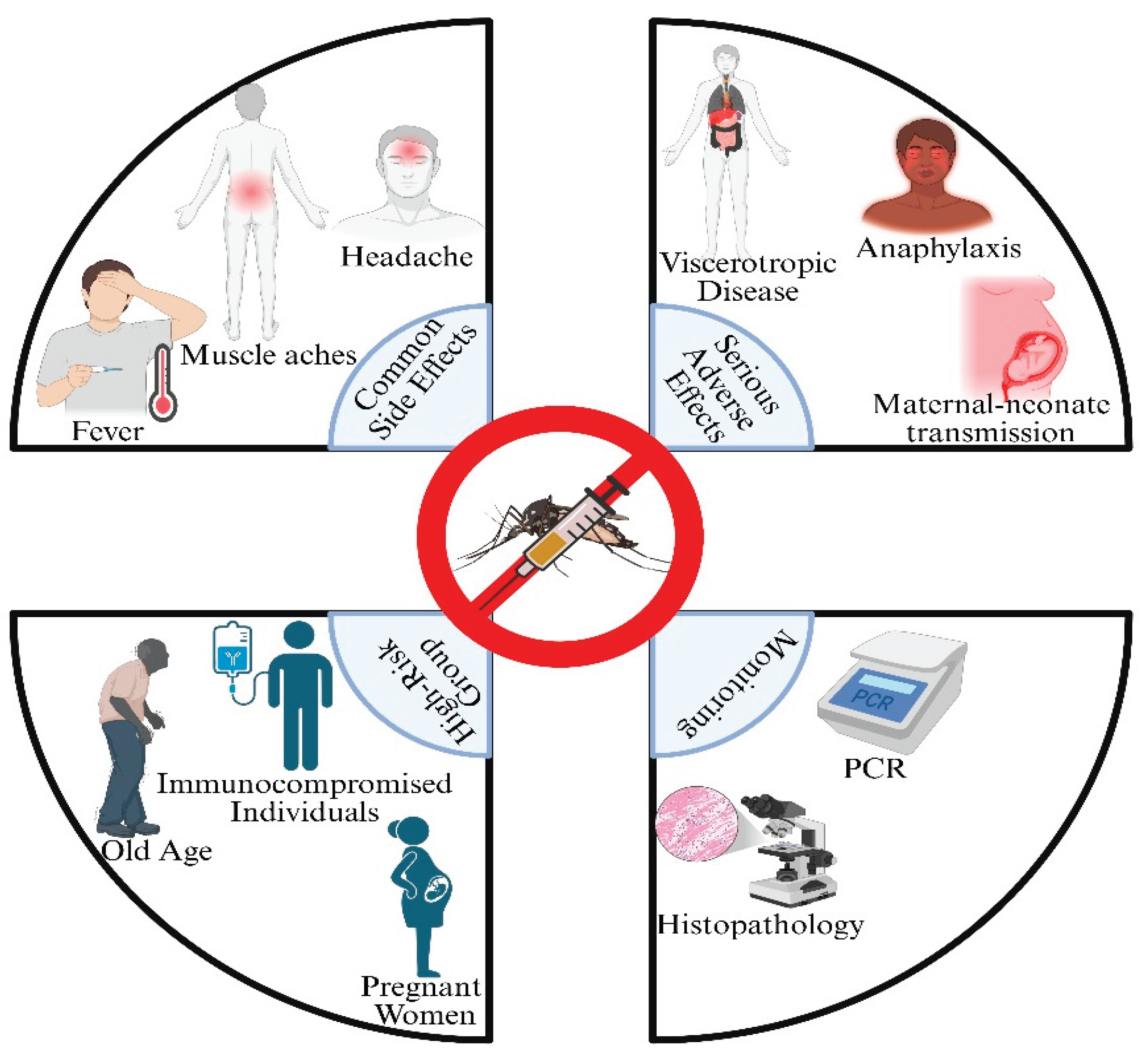
4. Vaccine Safety and Adverse Events
| # | Category | Description | Frequency | Management | Reference |
|---|---|---|---|---|---|
| 1 | Severe and rare adverse events, | YEL-AVD or YEL AND | Rare | Inactivated YF 17D virus | [61] |
| 2 | Serious adverse events | Hypersensitivity events, anaphylactic shock, Viscerotropic disease and neurologic syndrome | 25 in 35 people | 17D and 17DD yellow fever Vaccine | [62] |
| 3 | Allergic Reactions | Anaphylactic reaction | 40 in 5,236,820 | Yellow fever vaccine | [63] |
| 4 | Severe adverse reactions | YEL-AVD, YEL-AEs and YEL-AND | 6 patients | 17D-derived yellow fever vaccine | [64] |
| 5 | adverse events | Fever, myalgia, and headache | 43 in 68 Adult | yellow fever live-attenuated vaccine | [65] |
4.1. Common Adverse Events: Insights from Post-Marketing Surveillance
4.2. Managing Vaccine-Associated Complications
4.3. Balancing Risk and Benefit: The Yellow Fever Vaccination Dilemma
5. Yellow Fever Outbreaks and Control Measures
5.1. Recent Yellow Fever Outbreaks: Lessons Learned and Challenges Faced
5.2. Role of Vaccination in Controlling Epidemics
| # | Outbreak and location | Year | Result | Reference |
|---|---|---|---|---|
| 1. | Angola and Brazil | 1970-2016 | Yellow fever risk zones still have 393–472.9 million individuals who need to be vaccinated to fulfill the World Health Organization’s 80% coverage goal, despite substantial growth in vaccine coverage since 1970. | [14] |
| 2. | Angola and Brazil | 2015-2016 | The 2016 YF outbreak in Luanda, Angola, was analysed using a vector-host epidemic model, revealing that timely vaccination and behavioural changes can reduce deaths and prevent future outbreaks. | [83] |
| 3. | Uganda (East Africa) | 2019-2022 | The proposal suggests the establishment of a YF elimination task force to coordinate surveillance, vaccination campaigns, mosquito management strategies, and risk communication to reduce YF incidence and outbreaks. | [84] |
| 4. | Brazil | 2016-2017 | Due to the presence of animal reservoirs, human susceptibility, and the presence of vectors, unvaccinated travelers in the affected states of Brazil are at risk of contracting the virus. A potential pandemic could be triggered by ecological conditions and enzootics, potentially leading to spillover. | [85] |
| 5. | Brazil | 2017–2018 | In 2016, Brazil experienced the largest yellow fever outbreak in the Americas, primarily in densely populated areas like São Paulo, originating from three South American genotype variants. | [86] |
| 6. | West African and South American | 2001-2003 | Yellow fever, a tropical ailment responsible for 200,000 cases and 30,000 fatalities each year, is spread by humans, mosquitoes, and monkeys, with the possibility of preventative and chimeric vaccines. | [87] |
| 7. | Brazil | 2016-2017 | The YFV outbreak in Brazil necessitates prompt discovery and control via epidemiological and genetic surveillance, accompanied by a global plan aimed at eradicating epidemics by 2026. | [88] |
| 8 | Brazil | 2016-2018 | UYF prevention relies on insect control measures, resistance to insecticides, behavioral measures, and health surveillance; however, recent outbreaks in Brazil have shown the ineffectiveness of these measures. | [89] |
| 9. | Angola | 2015-2016 | Despite multiple vaccination campaigns, the Angola YFV outbreak reached its peak in February 2016, with 4,347 suspected cases and 377 deaths, leading to an emergency campaign in August 2016. | [90] |
| 10. | Angola | 2015 | Yellow fever rapidly spreads from Luanda, Angola, with 49 districts reporting cases within three months. Prioritizing vaccination is recommended; however, constraints such as vaccine supply and delivery logistics must also be considered. | [91] |
| 11. | Brazil and Venezuela | 1990 to 2022 | Nine patients with YF-compatible symptoms in French Guiana, Venezuela, Suriname, and Brazil died within 8 days, requiring stronger vaccination coverage due to the likely persisting sylvatic cycle. | [92] |
| 12 | South American countries | 2024-2025 | Current epidemics with more than 200 cases and more than 100 deaths are associated with a lack of vaccinations in certain age groups in Colombia and Brazil, which have concentrated most of the cases. | https://shiny.paho-phe.org/yellowfever/ |
5.3. Integrating Vaccination Strategies with Vector Control
6. Future Directions in Yellow Fever Vaccination
6.1. Advancements in Vaccine Technology: Novel Approaches and Platforms
6.2. Targeting Vulnerable Populations: Vaccination Equity and Accessibility
6.3. Strengthening Surveillance and Monitoring for Vaccine-Preventable Diseases
7. Yellow Fever in the Context of Emerging Infectious Diseases
7.1. Yellow Fever as a Model for Preparedness and Response
7.2. Potential Cross-Protection with Other Flaviviruses
7.3. One Health Approach: Integrating Animal and Human Health
8. Limitations
9. Conclusions
Funding
Declarations
Declaration of Competing Interest
Acknowledgements
References
- Angerami RN, Socorro Souza Chaves TD, Rodríguez-Morales AJ. Yellow fever outbreaks in South America: Current epidemiology, legacies of the recent past and perspectives for the near future. New Microbes New Infect 2025;65:101580. [CrossRef]
- Srivastava S, Dhoundiyal S, Kumar S, Kaur A, Khatib MN, Gaidhane S, et al. Yellow Fever: Global Impact, Epidemiology, Pathogenesis, and Integrated Prevention Approaches. Infez Med 2024;32:434-50.
- Reno E, Quan NG, Franco-Paredes C, Chastain DB, Chauhan L, Rodriguez-Morales AJ, et al. Prevention of yellow fever in travellers: an update. Lancet Infect Dis 2020;20:e129-e37. [CrossRef]
- Bassey BE, Braka F, Onyibe R, Kolude OO, Oluwadare M, Oluwabukola A, et al. Changing epidemiology of yellow fever virus in Oyo State, Nigeria. BMC Public Health 2022;22:467. [CrossRef]
- Kallas EG, D'Elia Zanella L, Moreira CHV, Buccheri R, Diniz GBF, Castiñeiras ACP, et al. Predictors of mortality in patients with yellow fever: an observational cohort study. Lancet Infect Dis 2019;19:750-8.
- Blake JB. Yellow fever in eighteenth century America. Bulletin of the New York Academy of Medicine 1968;44:673.
- Garske T, Van Kerkhove MD, Yactayo S, Ronveaux O, Lewis RF, Staples JE, et al. Yellow fever in Africa: estimating the burden of disease and impact of mass vaccination from outbreak and serological data. PLoS medicine 2014;11:e1001638. [CrossRef]
- Thomas RE. Yellow fever vaccine-associated viscerotropic disease: current perspectives. Drug design, development and therapy 2016;3345-53. [CrossRef]
- Barrett AD, Higgs S. Yellow fever: a disease that has yet to be conquered. Annu. Rev. Entomol. 2007;52:209-29. [CrossRef]
- PETERSEN JL. BEHAVIORAL DIFFERENCES IN TWO SUBSPECIES OF AEDES AEGYPTI (L.)(DIPTERA: CULICIDAE) IN EAST AFRICA. University of Notre Dame, 1977.
- Sanchez-Rojas IC, Solarte-Jimenez CL, Chamorro-Velazco EC, Diaz-Llerena GE, Arevalo CD, Cuasquer-Posos OL, et al. Yellow fever in Putumayo, Colombia, 2024. New Microbes New Infect 2025;64:101572.
- Tuboi SH, Costa ZGA, da Costa Vasconcelos PF, Hatch D. Clinical and epidemiological characteristics of yellow fever in Brazil: analysis of reported cases 1998–2002. Transactions of the Royal Society of Tropical Medicine and Hygiene 2007;101:169-75. [CrossRef]
- Monath TP, Nichols R, Archambault WT, Moore L, Marchesani R, Tian J, et al. Comparative safety and immunogenicity of two yellow fever 17D vaccines (ARILVAX and YF-VAX) in a phase III multicenter, double-blind clinical trial. The American journal of tropical medicine and hygiene 2002;66:533-41. [CrossRef]
- Shearer FM, Moyes CL, Pigott DM, Brady OJ, Marinho F, Deshpande A, et al. Global yellow fever vaccination coverage from 1970 to 2016: an adjusted retrospective analysis. The Lancet infectious diseases 2017;17:1209-17. [CrossRef]
- Frierson JG. The yellow fever vaccine: a history. The Yale journal of biology and medicine 2010;83:77.
- Lindsey NP, Schroeder BA, Miller ER, Braun MM, Hinckley AF, Marano N, et al. Adverse event reports following yellow fever vaccination. Vaccine 2008;26:6077-82. [CrossRef]
- Monath TP. Dengue and yellow fever—challenges for the development and use of vaccines. New England Journal of Medicine 2007;357:2222-5. [CrossRef]
- Barrett AD, Teuwen DE. Yellow fever vaccine—how does it work and why do rare cases of serious adverse events take place? Current opinion in immunology 2009;21:308-13.
- Bendiner E. Max Theiler: Yellow jack and the jackpot. Hospital Practice 1988;23:211-44. [CrossRef]
- Miller JD, van der Most RG, Akondy RS, Glidewell JT, Albott S, Masopust D, et al. Human effector and memory CD8+ T cell responses to smallpox and yellow fever vaccines. Immunity 2008;28:710-22. [CrossRef]
- Davis EH, Beck AS, Strother AE, Thompson JK, Widen SG, Higgs S, et al. Attenuation of live-attenuated yellow fever 17D vaccine virus is localized to a high-fidelity replication complex. Mbio 2019;10:10.1128/mbio. 02294-19. [CrossRef]
- Pugachev KV, Ocran SW, Guirakhoo F, Furby D, Monath TP. Heterogeneous nature of the genome of the ARILVAX yellow fever 17D vaccine revealed by consensus sequencing. Vaccine 2002;20:996-9. [CrossRef]
- Lam LM, Watson AM, Ryman KD, Klimstra WB. Gamma-interferon exerts a critical early restriction on replication and dissemination of yellow fever virus vaccine strain 17D-204. Npj Vaccines 2018;3:5. [CrossRef]
- Pato TP, Souza MCO, Silva AN, Pereira RC, Silva MV, Caride E, et al. Development of a membrane adsorber based capture step for the purification of yellow fever virus. Vaccine 2014;32:2789-93. [CrossRef]
- Cox MM. Recombinant protein vaccines produced in insect cells. Vaccine 2012;30:1759-66. [CrossRef]
- Ulmer JB, Valley U, Rappuoli R. Vaccine manufacturing: challenges and solutions. Nature biotechnology 2006;24:1377-83. [CrossRef]
- Jean K, Hamlet A, Benzler J, Cibrelus L, Gaythorpe KA, Sall A, et al. Eliminating yellow fever epidemics in Africa: vaccine demand forecast and impact modelling. PLoS neglected tropical diseases 2020;14:e0008304. [CrossRef]
- Okwo-Bele J-M, Cherian T. The expanded programme on immunization: a lasting legacy of smallpox eradication. Vaccine 2011;29:D74-D9. [CrossRef]
- Cetron MS, Marfin AA, Julian KG, Gubler DJ, Sharp DJ, Barwick RS, et al. Yellow fever vaccine recommendations of the Advisory Committee on Immunization Practices (ACIP), 2002. MORBIDITY AND MORTALITY WEEKLY REPORT RECOMMENDATIONS AND REPORTS RR 2002;51.
- Mokaya J, Kimathi D, Lambe T, Warimwe GM. What Constitutes Protective Immunity Following Yellow Fever Vaccination? Vaccines (Basel) 2021;9.
- Gotuzzo E, Yactayo S, Cordova E. Efficacy and duration of immunity after yellow fever vaccination: systematic review on the need for a booster every 10 years. Am J Trop Med Hyg 2013;89:434-44. [CrossRef]
- Wieten RW, Jonker EF, van Leeuwen EM, Remmerswaal EB, Ten Berge IJ, de Visser AW, et al. A Single 17D Yellow Fever Vaccination Provides Lifelong Immunity; Characterization of Yellow-Fever-Specific Neutralizing Antibody and T-Cell Responses after Vaccination. PLoS One 2016;11:e0149871. [CrossRef]
- Mishra N, Boudewijns R, Schmid MA, Marques RE, Sharma S, Neyts J, et al. A Chimeric Japanese Encephalitis Vaccine Protects against Lethal Yellow Fever Virus Infection without Inducing Neutralizing Antibodies. mBio 2020;11. [CrossRef]
- Mateus J, Grifoni A, Voic H, Angelo MA, Phillips E, Mallal S, et al. Identification of Novel Yellow Fever Class II Epitopes in YF-17D Vaccinees. Viruses 2020;12. [CrossRef]
- Lim HX, Lim J, Poh CL. Identification and selection of immunodominant B and T cell epitopes for dengue multi-epitope-based vaccine. Med Microbiol Immunol 2021;210:1-11. [CrossRef]
- da Silva OLT, da Silva MK, Rodrigues-Neto JF, Santos Lima JPM, Manzoni V, Akash S, et al. Advancing molecular modeling and reverse vaccinology in broad-spectrum yellow fever virus vaccine development. Sci Rep 2024;14:10842. [CrossRef]
- Silva ML, Martins MA, Espírito-Santo LR, Campi-Azevedo AC, Silveira-Lemos D, Ribeiro JGL, et al. Characterization of main cytokine sources from the innate and adaptive immune responses following primary 17DD yellow fever vaccination in adults. Vaccine 2011;29:583-92. [CrossRef]
- Campi-Azevedo AC, de Araujo-Porto LP, Luiza-Silva M, Batista MA, Martins MA, Sathler-Avelar R, et al. 17DD and 17D-213/77 yellow fever substrains trigger a balanced cytokine profile in primary vaccinated children. PLoS One 2012;7:e49828.
- Kohler S, Bethke N, Böthe M, Sommerick S, Frentsch M, Romagnani C, et al. The early cellular signatures of protective immunity induced by live viral vaccination. European journal of immunology 2012;42:2363-73. [CrossRef]
- Luiza-Silva M, Campi-Azevedo AC, Batista MA, Martins MA, Avelar RS, da Silveira Lemos D, et al. Cytokine signatures of innate and adaptive immunity in 17DD yellow fever vaccinated children and its association with the level of neutralizing antibody. Journal of Infectious Diseases 2011;204:873-83. [CrossRef]
- Reis LR, da Costa-Rocha IA, Campi-Azevedo AC, Peruhype-Magalhães V, Coelho-dos-Reis JG, Costa-Pereira C, et al. Exploratory study of humoral and cellular immunity to 17DD yellow fever vaccination in children and adults residents of areas without circulation of yellow fever virus. Vaccine 2022;40:798-810. [CrossRef]
- Bovay A, Nassiri S, Maby-El Hajjami H, Marcos Mondejar P, Akondy RS, Ahmed R, et al. Minimal immune response to booster vaccination against Yellow Fever associated with pre-existing antibodies. Vaccine 2020;38:2172-82. [CrossRef]
- Hepburn MJ, Kortepeter MG, Pittman PR, Boudreau EF, Mangiafico JA, Buck PA, et al. Neutralizing antibody response to booster vaccination with the 17d yellow fever vaccine. Vaccine 2006;24:2843-9. [CrossRef]
- Wieten RW, Goorhuis A, Jonker EFF, de Bree GJ, de Visser AW, van Genderen PJJ, et al. 17D yellow fever vaccine elicits comparable long-term immune responses in healthy individuals and immune-compromised patients. J Infect 2016;72:713-22. [CrossRef]
- Reinhardt B, Jaspert R, Niedrig M, Kostner C, L'age-Stehr J. Development of viremia and humoral and cellular parameters of immune activation after vaccination with yellow fever virus strain 17D: a model of human flavivirus infection. Journal of medical virology 1998;56:159-67.
- Stryhn A, Kongsgaard M, Rasmussen M, Harndahl MN, Osterbye T, Bassi MR, et al. A Systematic, Unbiased Mapping of CD8(+) and CD4(+) T Cell Epitopes in Yellow Fever Vaccinees. Front Immunol 2020;11:1836.
- James EA, LaFond RE, Gates TJ, Mai DT, Malhotra U, Kwok WW. Yellow fever vaccination elicits broad functional CD4+ T cell responses that recognize structural and nonstructural proteins. J Virol 2013;87:12794-804. [CrossRef]
- Wec AZ, Haslwanter D, Abdiche YN, Shehata L, Pedreno-Lopez N, Moyer CL, et al. Longitudinal dynamics of the human B cell response to the yellow fever 17D vaccine. Proc Natl Acad Sci U S A 2020;117:6675-85. [CrossRef]
- Maciel Jr M, Cruz FdSP, Cordeiro MT, da Motta MA, Cassemiro KMSdM, Maia RdCC, et al. A DNA vaccine against yellow fever virus: development and evaluation. PLoS neglected tropical diseases 2015;9:e0003693.
- Querec TD, Akondy RS, Lee EK, Cao W, Nakaya HI, Teuwen D, et al. Systems biology approach predicts immunogenicity of the yellow fever vaccine in humans. Nat Immunol 2009;10:116-25. [CrossRef]
- Miller JD, van der Most RG, Akondy RS, Glidewell JT, Albott S, Masopust D, et al. Human effector and memory CD8+ T cell responses to smallpox and yellow fever vaccines. Immunity 2008;28:710-22. [CrossRef]
- Wrammert J, Miller J, Akondy R, Ahmed R. Human immune memory to yellow fever and smallpox vaccination. J Clin Immunol 2009;29:151-7. [CrossRef]
- Ahmed R, Akondy RS. Insights into human CD8(+) T-cell memory using the yellow fever and smallpox vaccines. Immunol Cell Biol 2011;89:340-5.
- Piras-Douce F, Broudic K, Chautard E, Raynal F, Courtois V, Gautheron S, et al. Evaluation of safety and immuno-efficacy of a next generation live-attenuated yellow fever vaccine in cynomolgus macaques. Vaccine 2023;41:1457-70. [CrossRef]
- Fuertes Marraco SA, Soneson C, Cagnon L, Gannon PO, Allard M, Maillard SA, et al. Long-lasting stem cell–like memory CD8+ T cells with a naïve-like profile upon yellow fever vaccination. Science translational medicine 2015;7:282ra48-ra48.
- Kling K, Domingo C, Bogdan C, Duffy S, Harder T, Howick J, et al. Duration of Protection After Vaccination Against Yellow Fever: A Systematic Review and Meta-Analysis. Clin Infect Dis 2022;75:2266-74. [CrossRef]
- Vaccines CGfSoYF. Duration of immunity in recipients of two doses of 17DD yellow fever vaccine. Vaccine 2019;37:5129-35.
- Collaborative group for studies on yellow fever v. Duration of post-vaccination immunity against yellow fever in adults. Vaccine 2014;32:4977-84.
- Wigg de Araujo Lagos L, de Jesus Lopes de Abreu A, Caetano R, Braga JU. Yellow fever vaccine safety in immunocompromised individuals: a systematic review and meta-analysis. J Travel Med 2023;30. [CrossRef]
- Lindsey NP, Rabe IB, Miller ER, Fischer M, Staples JE. Adverse event reports following yellow fever vaccination, 2007-13. J Travel Med 2016;23. [CrossRef]
- de Menezes Martins R, da Luz Fernandes Leal M, Homma A. Serious adverse events associated with yellow fever vaccine. Human Vaccines & Immunotherapeutics 2015;11:2183-7. [CrossRef]
- E. Thomas R, L. Lorenzetti D, Spragins W, Jackson D, Williamson T. Reporting rates of yellow fever vaccine 17D or 17DD-associated serious adverse events in pharmacovigilance data bases: systematic review. Current Drug Safety 2011;6:145-54. [CrossRef]
- Kelso JM, Mootrey GT, Tsai TF. Anaphylaxis from yellow fever vaccine. Journal of allergy and clinical immunology 1999;103:698-701.
- Bae H-G, Domingo C, Tenorio A, de Ory F, Muñoz J, Weber P, et al. Immune response during adverse events after 17D-derived yellow fever vaccination in Europe. The Journal of infectious diseases 2008;197:1577-84. [CrossRef]
- Chan CY, Chan KR, Chua CJ, Nur Hazirah S, Ghosh S, Ooi EE, et al. Early molecular correlates of adverse events following yellow fever vaccination. JCI Insight 2017;2. [CrossRef]
- Thomas RE, Lorenzetti DL, Spragins W, Jackson D, Williamson T. The safety of yellow fever vaccine 17D or 17DD in children, pregnant women, HIV+ individuals, and older persons: systematic review. Am J Trop Med Hyg 2012;86:359-72. [CrossRef]
- Vasconcelos PFC, Luna EJ, Galler R, Silva LJ, Coimbra TL, Barros VLRS, et al. Serious adverse events associated with yellow fever 17DD vaccine in Brazil: a report of two cases. The Lancet 2001;358:91-7. [CrossRef]
- Nordin JD, Parker ED, Vazquez-Benitez G, Kharbanda EO, Naleway A, Marcy SM, et al. Safety of the yellow fever vaccine: a retrospective study. Journal Of Travel Medicine 2013;20:368-73. [CrossRef]
- Martins RdM, Maia MdLdS, Santos EMd, Cruz RLdS, dos Santos PRG, Carvalho SMD, et al. Yellow Fever Vaccine Post-marketing Surveillance in Brazil. Procedia in Vaccinology 2010;2:178-83.
- Thomas RE, Lorenzetti DL, Spragins W, Jackson D, Williamson T. Active and passive surveillance of yellow fever vaccine 17D or 17DD-associated serious adverse events: systematic review. Vaccine 2011;29:4544-55. [CrossRef]
- Belmusto-Worn VE, Sanchez JL, McCARTHY K, Nichols R, Bautista CT, Magill AJ, et al. Randomized, double-blind, phase III, pivotal field trial of the comparative immunogenicity, safety, and tolerability of two yellow fever 17D vaccines (ARILVAXTM and YF-VAX (R)) in healthy infants and. American Journal of Tropical Medicine and Hygiene 2005;72:189-97.
- Lecomte E, Laureys G, Verbeke F, Domingo Carrasco C, Van Esbroeck M, Huits R. A clinician’s perspective on yellow fever vaccine-associated neurotropic disease. Journal of Travel Medicine 2020;27. [CrossRef]
- McMahon AW, Eidex RB, Marfin AA, Russell M, Sejvar JJ, Markoff L, et al. Neurologic disease associated with 17D-204 yellow fever vaccination: a report of 15 cases. Vaccine 2007;25:1727-34. [CrossRef]
- de Andrade Gandolfi F, Estofolete CF, Wakai MC, Negri AF, Barcelos MD, Vasilakis N, et al. Yellow Fever Vaccine-Related Neurotropic Disease in Brazil Following Immunization with 17DD. Vaccines (Basel) 2023;11. [CrossRef]
- Monath TP. Review of the risks and benefits of yellow fever vaccination including some new analyses. Expert review of vaccines 2012;11:427-48. [CrossRef]
- Struchiner CJ, Luz PM, Dourado I, Sato HK, Aguiar SG, Ribeiro JG, et al. Risk of fatal adverse events associated with 17DD yellow fever vaccine. Epidemiol Infect 2004;132:939-46. [CrossRef]
- Chippaux JP, Chippaux A. Yellow fever in Africa and the Americas: a historical and epidemiological perspective. J Venom Anim Toxins Incl Trop Dis 2018;24:20. [CrossRef]
- Chen LH, Wilson ME. Yellow fever control: current epidemiology and vaccination strategies. Trop Dis Travel Med Vaccines 2020;6:1. [CrossRef]
- de Oliveira Figueiredo P, Stoffella-Dutra AG, Barbosa Costa G, Silva de Oliveira J, Dourado Amaral C, Duarte Santos J, et al. Re-Emergence of Yellow Fever in Brazil during 2016-2019: Challenges, Lessons Learned, and Perspectives. Viruses 2020;12.
- Nomhwange T, Jean Baptiste AE, Ezebilo O, Oteri J, Olajide L, Emelife K, et al. The resurgence of yellow fever outbreaks in Nigeria: a 2-year review 2017-2019. BMC Infect Dis 2021;21:1054. [CrossRef]
- Diagne MM, Ndione MHD, Gaye A, Barry MA, Diallo D, Diallo A, et al. Yellow Fever Outbreak in Eastern Senegal, 2020-2021. Viruses 2021;13. [CrossRef]
- Salomon OD, Arias AR. The second coming of urban yellow fever in the Americas: looking the past to see the future. An Acad Bras Cienc 2022;94:e20201252.
- Zhao S, Stone L, Gao D, He D. Modelling the large-scale yellow fever outbreak in Luanda, Angola, and the impact of vaccination. PLoS Negl Trop Dis 2018;12:e0006158. [CrossRef]
- Mensah EA, Gyasi SO, Nsubuga F, Alali WQ. A proposed One Health approach to control yellow fever outbreaks in Uganda. One Health Outlook 2024;6:9. [CrossRef]
- Ortiz-Martínez Y, Patiño-Barbosa AM, Rodriguez-Morales AJ. Yellow fever in the Americas: the growing concern about new epidemics. F1000Research 2017;6. [CrossRef]
- Cunha MdP, Duarte-Neto AN, Pour SZ, Ortiz-Baez AS, Černý J, Pereira BBdS, et al. Origin of the São Paulo Yellow Fever epidemic of 2017–2018 revealed through molecular epidemiological analysis of fatal cases. Scientific Reports 2019;9.
- Tomori O. Yellow fever: the recurring plague. Crit Rev Clin Lab Sci 2004;41:391-427.
- Faria NR, Kraemer MU, Hill SC, Góes de Jesus J, Aguiar Rd, Iani FC, et al. Genomic and epidemiological monitoring of yellow fever virus transmission potential. Science 2018;361:894-9. [CrossRef]
- do Carmo Cupertino M, Garcia R, Gomes AP, de Paula SO, Mayers N, Siqueira-Batista R. Epidemiological, prevention and control updates of yellow fever outbreak in Brazil. Asian Pacific Journal of Tropical Medicine 2019;12:49-59. [CrossRef]
- Collins ND, Barrett AD. Live Attenuated Yellow Fever 17D Vaccine: A Legacy Vaccine Still Controlling Outbreaks In Modern Day. Curr Infect Dis Rep 2017;19:14. [CrossRef]
- Kraemer MU, Faria NR, Reiner RC, Golding N, Nikolay B, Stasse S, et al. Spread of yellow fever virus outbreak in Angola and the Democratic Republic of the Congo 2015–16: a modelling study. The Lancet infectious diseases 2017;17:330-8. [CrossRef]
- Thomas C, Michaud C, Gaillet M, Carrión-Nessi FS, Forero-Peña DA, Lacerda MVG, et al. Yellow Fever Reemergence Risk in the Guiana Shield: a Comprehensive Review of Cases Between 1990 and 2022. Current Tropical Medicine Reports 2023;10:138-45. [CrossRef]
- Wasserman S, Tambyah PA, Lim PL. Yellow fever cases in Asia: primed for an epidemic. International Journal of Infectious Diseases 2016;48:98-103.
- Ndeffo-Mbah ML, Pandey A. Global Risk and Elimination of Yellow Fever Epidemics. J Infect Dis 2020;221:2026-34. [CrossRef]
- Jean K, Hamlet A, Benzler J, Cibrelus L, Gaythorpe KAM, Sall A, et al. Eliminating yellow fever epidemics in Africa: Vaccine demand forecast and impact modelling. PLoS Negl Trop Dis 2020;14:e0008304. [CrossRef]
- Silva T, Nogueira de Sa A, Prates EJS, Rodrigues DE, Silva T, Matozinhos FP, et al. Yellow fever vaccination before and during the covid-19 pandemic in Brazil. Rev Saude Publica 2022;56:45.
- Casey RM, Harris JB, Ahuka-Mundeke S, Dixon MG, Kizito GM, Nsele PM, et al. Immunogenicity of Fractional-Dose Vaccine during a Yellow Fever Outbreak - Final Report. N Engl J Med 2019;381:444-54. [CrossRef]
- Doshi RH, Mukadi PK, Casey RM, Kizito GM, Gao H, Nguete UB, et al. Immunological response to fractional-dose yellow fever vaccine administered during an outbreak in Kinshasa, Democratic Republic of the Congo: results 5 years after vaccination from a prospective cohort study. Lancet Infect Dis 2024;24:611-8. [CrossRef]
- Nnaji CA, Shey MS, Adetokunboh OO, Wiysonge CS. Immunogenicity and safety of fractional dose yellow fever vaccination: A systematic review and meta-analysis. Vaccine 2020;38:1291-301. [CrossRef]
- Vannice K, Wilder-Smith A, Hombach J. Fractional-Dose Yellow Fever Vaccination - Advancing the Evidence Base. N Engl J Med 2018;379:603-5. [CrossRef]
- Roukens AHE, Visser LG. Fractional-dose yellow fever vaccination: an expert review. J Travel Med 2019;26. [CrossRef]
- Manikandan S, Mathivanan A, Bora B, Hemaladkshmi P, Abhisubesh V, Poopathi S. A review on vector borne disease transmission: Current strategies of mosquito vector control. Indian Journal of Entomology 2023;503-13. [CrossRef]
- Kleinert RDV, Montoya-Diaz E, Khera T, Welsch K, Tegtmeyer B, Hoehl S, et al. Yellow Fever: Integrating Current Knowledge with Technological Innovations to Identify Strategies for Controlling a Re-Emerging Virus. Viruses 2019;11. [CrossRef]
- Garske T, Van Kerkhove MD, Yactayo S, Ronveaux O, Lewis RF, Staples JE, et al. Yellow Fever in Africa: estimating the burden of disease and impact of mass vaccination from outbreak and serological data. PLoS Med 2014;11:e1001638. [CrossRef]
- Raimundo SM, Yang HM, Massad E. Modeling Vaccine Preventable Vector-Borne Infections: Yellow Fever as a Case Study. Journal of Biological Systems 2016;24:193-216. [CrossRef]
- Hansen CA, Barrett ADT. The Present and Future of Yellow Fever Vaccines. Pharmaceuticals 2021;14. [CrossRef]
- Medina-Magues LG, Muhe J, Jasny E, Medina-Magues ES, Roth N, Lopera-Madrid J, et al. Immunogenicity and protective activity of mRNA vaccine candidates against yellow fever virus in animal models. NPJ Vaccines 2023;8:31. [CrossRef]
- Monath TP, Seligman SJ, Robertson JS, Guy B, Hayes EB, Condit RC, et al. Live virus vaccines based on a yellow fever vaccine backbone: standardized template with key considerations for a risk/benefit assessment. Vaccine 2015;33:62-72. [CrossRef]
- Al-Halifa S, Gauthier L, Arpin D, Bourgault S, Archambault D. Nanoparticle-Based Vaccines Against Respiratory Viruses. Front Immunol 2019;10:22. [CrossRef]
- Servadio JL, Munoz-Zanzi C, Convertino M. Environmental determinants predicting population vulnerability to high yellow fever incidence. R Soc Open Sci 2022;9:220086. [CrossRef]
- Adrien N, Hyde TB, Gacic-Dobo M, Hombach J, Krishnaswamy A, Lambach P. Differences between coverage of yellow fever vaccine and the first dose of measles-containing vaccine: A desk review of global data sources. Vaccine 2019;37:4511-7. [CrossRef]
- Wu JT, Peak CM, Leung GM, Lipsitch M. Fractional dosing of yellow fever vaccine to extend supply: a modelling study. The Lancet 2016;388:2904-11. [CrossRef]
- Gubler DJ, Almeida MAB, Cardoso JdC, dos Santos E, da Fonseca DF, Cruz LL, et al. Surveillance for Yellow Fever Virus in Non-Human Primates in Southern Brazil, 2001–2011: A Tool for Prioritizing Human Populations for Vaccination. PLoS Neglected Tropical Diseases 2014;8.
- Selemane I. Epidemiological monitoring of the last outbreak of yellow fever in Brazil - An outlook from Portugal. Travel Med Infect Dis 2019;28:46-51. [CrossRef]
- Andrade MS, Campos FS, Oliveira CH, Oliveira RS, Campos AAS, Almeida MAB, et al. Fast surveillance response reveals the introduction of a new yellow fever virus sub-lineage in 2021, in Minas Gerais, Brazil. Mem Inst Oswaldo Cruz 2022;117:e220127. [CrossRef]
- Hyde TB, Andrus JK, Dietz VJ, Integrated All VPDSWG, Andrus JK, Hyde TB, et al. Critical issues in implementing a national integrated all-vaccine preventable disease surveillance system. Vaccine 2013;31 Suppl 3:C94-8. [CrossRef]
- Zhao Y, Zhang X, Shu S, Sun Y, Feng X, Zhang S. Yellow Fever: A Re-Emerging Threat. Health 2018;10:1431-48. [CrossRef]
- Cruz ACR, Hernandez LHA, Aragao CF, da Paz TYB, da Silva SP, da Silva FS, et al. The Importance of Entomo-Virological Investigation of Yellow Fever Virus to Strengthen Surveillance in Brazil. Trop Med Infect Dis 2023;8.
- Aliaga-Samanez A, Real R, Segura M, Marfil-Daza C, Olivero J. Yellow fever surveillance suggests zoonotic and anthroponotic emergent potential. Commun Biol 2022;5:530. [CrossRef]
- Mantilla-Granados JS, Sarmiento-Senior D, Manzano J, Calderon-Pelaez MA, Velandia-Romero ML, Buitrago LS, et al. Multidisciplinary approach for surveillance and risk identification of yellow fever and other arboviruses in Colombia. One Health 2022;15:100438. [CrossRef]
- Williams DT, Mackenzie JS, Bingham J. Flaviviruses. Diseases of swine 2019;530-43.
- Subramaniam KS, Lant S, Goodwin L, Grifoni A, Weiskopf D, Turtle L. Two Is Better Than One: Evidence for T-Cell Cross-Protection Between Dengue and Zika and Implications on Vaccine Design. Front Immunol 2020;11:517. [CrossRef]
- Zuckerman JN, Hatz C, Kantele A. Review of current typhoid fever vaccines, cross-protection against paratyphoid fever, and the European guidelines. Expert Rev Vaccines 2017;16:1029-43. [CrossRef]
- Cadenas-Fernández E, Barroso-Arévalo S, Kosowska A, Díaz-Frutos M, Gallardo C, Rodríguez-Bertos A, et al. Challenging boundaries: is cross-protection evaluation necessary for African swine fever vaccine development? A case of oral vaccination in wild boar. Frontiers in Immunology 2024;15. [CrossRef]
- Possas C, Lourenço-de-Oliveira R, Tauil PL, Pinheiro FdP, Pissinatti A, Cunha RVd, et al. Yellow fever outbreak in Brazil: the puzzle of rapid viral spread and challenges for immunisation. Memórias do Instituto Oswaldo Cruz 2018;113:e180278. [CrossRef]
- de Oliveira Figueiredo P, Stoffella-Dutra AG, Barbosa Costa G, Silva de Oliveira J, Dourado Amaral C, Duarte Santos J, et al. Re-emergence of yellow fever in Brazil during 2016–2019: challenges, lessons learned, and perspectives. Viruses 2020;12:1233.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




