Submitted:
26 May 2025
Posted:
27 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CSPH | Clinically Significant Portal Hypertension |
| EGD | Esophagogastroduodenoscopy |
| GOV2 | Type 2 Gastroesophageal Varices |
| JRSH | Japanese Research Society for Portal Hypertension |
| OR | Odds Ratios |
| PHG | Portal Hypertension Gastrophaty |
| PHPs | Portal Hypertension Polyps |
References
- Asrani, S.K.; Devarbhavi, H.; Eaton, J.; Kamath, P.S. The burden of liver diseases in the world. J Hepatol 2019, 70:151-171. [CrossRef]
- Turco, L.; Villanueva, C.; Albillos, A.; Genescà, J.; Garcia-Pagan, J.C.; Calleja, J.L.; et al. β blockers to prevent decompensation of cirrhosis in patients with clinically significant portal hypertension (PREDESCI): a randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2019, 393:1597-1608.
- de Franchis, R.; Bosch, J.; Garcia-Tsao, G.; Reiberger, T.; Ripoll, C. Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol 2022, 76:959-974.
- Scarlata, G.G.M.; Ismaiel, A.; Gambardella, M.L.; Leucuta, D.C.; Luzza, F.; Dumitrascu D.L.; Abenavoli, L. Use of Non-Invasive Biomarkers and Clinical Scores to Predict the Complications of Liver Cirrhosis: A Bicentric Experience. Medicina (Kaunas) 2024, 12;60:1854. [CrossRef]
- Urrunaga, N.H.; Rockey, D.C. Portal hypertensive gastropathy and colopathy. Clin Liver Dis 2014, 18:389-406. [CrossRef]
- Topal, F.; Akbulut, S.; Karahan, C.; Günav, S.; Saritas Yüksel, E.; Topal, FE. Portal Hypertensive Polyps as a Gastroscopic Finding in Liver Cirrhosis. Gastroenterol Res Pract 2020, 2020:9058909.
- Seleem, W.M.; Hanafy, A.S. Management of a Portal Hypertensive Polyp: Case Report of a Rare Entity. Gastrointest Tumors 2019, 6:137-141. [CrossRef]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J Hepatol 2018, 69:406-460.
- Gambardella, M.L.; Luigiano, C.; La Torre, G.; Scarlata, G.G.M.; Luzza, F.; Abenavoli, L. Portal hypertension-associated gastric pathology: role of endoscopic banding ligation. Minerva Gastroenterol 2025. [CrossRef]
- Sersté, T.; Melot, C.; Francoz, C.; Durand, F.; Rautou, P.E.; Valla, D.; Moreau, R.; Lebrec, D. Deleterious effects of beta-blockers on survival in patients with cirrhosis and refractory ascites. Hepatology 2010, 52:1017-22. [CrossRef]
- Bossen, L.; Krag, A.; Vilstrup, H.; Watson, H.; Jepsen, P. Non-selective betablockers do not affect mortality in cirrhosis patients with ascites: Post hoc analysis of three RCTs with 1198 patients. Hepatology 2016, 63:1968–1976.
- Bang, U.C.; Benfield, T.; Hyldstrup, L.; Jensen, J.E.; Bendtsen, F. Effect of propranolol on survival in patients with decompensated cirrhosis: a nationwide study based Danish patient registers. Liver Int 2016, 36:1304–1312. [CrossRef]
- Elwakil, R.; Al Breedy, A.M.; Gabal, H.H. Effect of endoscopic variceal abliteration by band ligation on portal hypertensive gastro-duadenopathy: endoscopic and pathological study. Hepatol Int 2016, 10:965-973. [CrossRef]
- Tripathi, D.; Ferguson, J.W.; Kochar, N.; Leithead, J.A.; Therapondos, G.; McAvoy, N.C.; et al. Randomized controlled trial of carvedilol versus variceal band ligation for the prevention of the first variceal bleed. Hepatology 2009, 50:825-833. [CrossRef]
- Lo, G.H.; Lai, K.H.; Cheng, J.S.; Hsu, P.I.; Chen, T.A.; Wang, E.M.; et al. The effects of endoscopic variceal ligation and propranolol on portal hypertensive gastropathy: a prospective, controlled trial. Gastrointest Endosc 2001, 53:579-84. [CrossRef]
- Elnaser, S.S.; El-Ebiary, S.; Bastawi, M.B.; Shafei, A.L.; Abd-Elhafee, A. Effect of endoscopic injection sclerotherapy and variceal ligation on development of portal hypertensive gastropathy and duodenopathy. J Egypt Soc Parasitol 2005, 35:253-64.
- McCormack, T.T.; Rose, J.D.; Smith, P.M.; Johnson, A.G. Perforating veins and blood flow in oesophageal varices. Lancet 1983, 2:1442-4. [CrossRef]
- Yüksel, O.; Köklü, S.; Arhan, M.; Yolcu, O.F.; Ertuğrul, I.; Odemiş, B.; et al. Effects of esophageal varice eradication on portal hypertensive gastropathy and fundal varices: a retrospective and comparative study. Dig Dis Sci 2006, 51:27-30. [CrossRef]
- Lahbabi, M.; Mellouki, I.; Aqodad, N.; Elabkari, M.; Elyousfi, M.; Ibrahimi, S.A.; et al. Esophageal variceal ligation in the secondary prevention of variceal bleeding: Result of long term follow-up. Pan Afr Med J 2013, 15:3. [CrossRef]
- Hou, M.C.; Lin, H.C.; Chen, C.H.; Kuo, B.I.; Perng, C.L.; Lee, F.Y.; et al. Changes in portal hypertensive gastropathy after endoscopic variceal sclerotherapy or ligation: an endoscopic observation. Gastrointest Endosc 1995, 42:139-44. [CrossRef]
- Kanke, K.; Ishida, M.; Yajima, N.; Saito, M.; Suzuki, Y.; Masuyama, H.; et al. Gastric mucosal congestion following endoscopic variceal ligation--analysis using reflectance spectrophotometry. Nihon Shokakibyo Gakkai Zasshi 1996, 93:701-6.
- Vianna, A.; Hayes, P.C.; Moscoso, G.; Driver, M.; Portmann, B.; Westaby, D.; et al. Normal venous circulation of the gastroesophageal junction: A route to understanding varices. Gastroenterology 1987, 93:876-89.
- Yamamoto, Y.; Sezai, S.; Sakurabayashi, S.; Hirano, M.; Oka, H. Effect of hepatic collateral hemodynamics on gastric mucosal blood flow in patients with liver cirrhosis. Dig Dis Sci 1992, 37:1319-23. [CrossRef]
- Lemmers, A.; Evrard, S.; Demetter, P.; Verset, G.; Gossum, A.V.; Adler, M.; et al. Gastrointestinal polypoid lesions: a poorly known endoscopic feature of portal hypertension. United European Gastroenterol J 2014, 2:189-96. [CrossRef]
- Panackel, C.; Joshy, H.; Sebastian, B.; Thomas, R.; Mathai, S.K. Gastric antral polyps: a manifestation of portal hypertensive gastropathy. Indian J Gastroenterol 2013, 32:206-7. [CrossRef]
- Livovsky, D.M.; Pappo, O.; Skarzhinsky, G.; Peretz, A.; Turvall, E.; Ackerman, Z. Gastric Polyp Growth during Endoscopic Surveillance for Esophageal Varices or Barrett's Esophagus. Isr Med Assoc J 2016, 18:267-71.
- Kara, D.; Hüsing-Kabar, A.; Schmidt, H.; Grünewald, I.; Chandhok, G.; Maschmeier, M.; et al. Portal Hypertensive Polyposis in Advanced Liver Cirrhosis: The Unknown Entity? Can J. Gastroenterol Hepatol 2018, 2018:2182784.
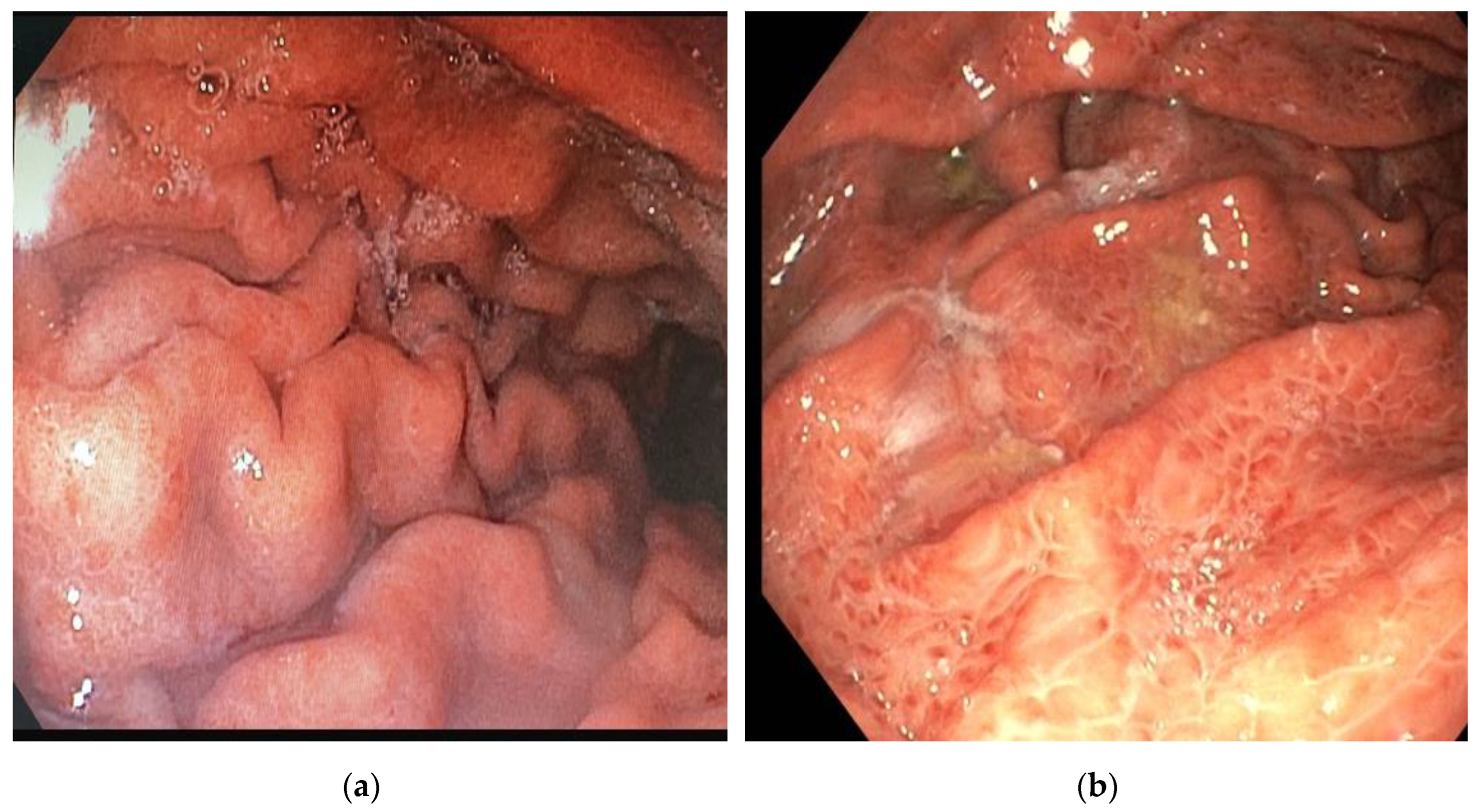
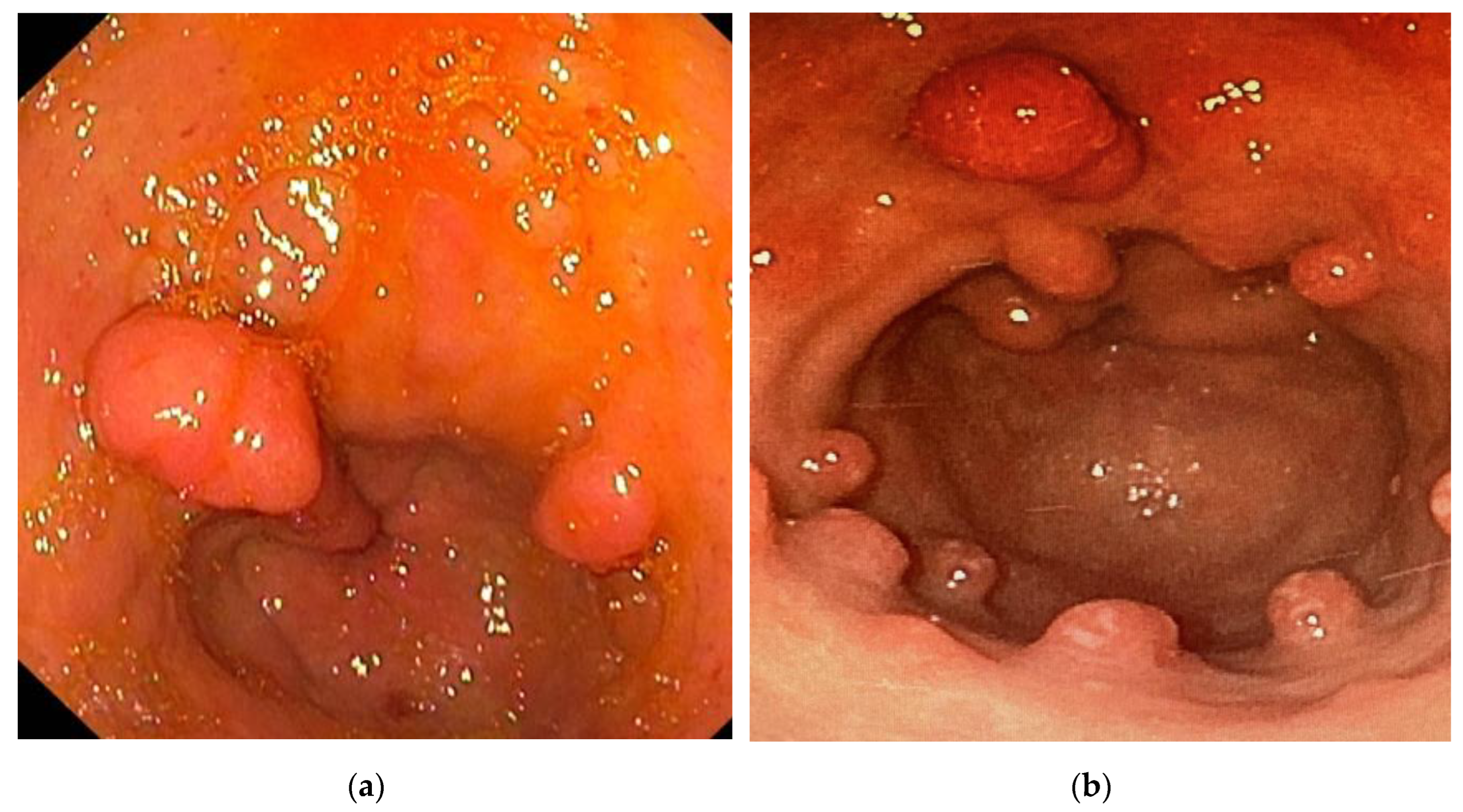
| Patients (N=44) | |
|---|---|
| Demographic and Anthropometric data | |
| Age (years) | 64±12 |
| Male gender, n (%) | 36 (82) |
| BMI (Kg/m2) | 24±5 |
| Disease characteristics | |
| Disease duration (years) | 4±5 |
| CHILD PUGH | |
| A, n (%) | 18 (41) |
| B, n (%) | 20 (45) |
| C, n (%) | 6 (14) |
| CHILD PUGH numeric | 7±2 |
| Disease etiology | |
| Viral , n (%) | 9 (21) |
| Alcoholic, n (%) | 19 (43) |
| Metabolic, n (%) | 16 (36) |
| Hepatic Encephalopathy, n (%) | 44 (100%) |
| Ascites, n (%) | 18 (41) |
| Hepatocarcinoma, n (%) | 8 (18) |
| Score | |
| APRI | 1±2 |
| FIB-4 | 10±8 |
| Dysmetabolic comorbidities, n (%) | |
| T2DM | 15 (34) |
| Hypertension | 18 (41) |
| Treatments | |
| Emergency legature, n (%) | 16 (36) |
| Election ligature, n(%) | 28 (64) |
| Beta-blockers, n (%) | 22 (50) |
| with PHG (n=26) | without PHG (n=18) | p-value | |
|---|---|---|---|
| Demographic and Anthropometric | |||
| Age (years) | 64±11 | 62±14 | 0.517 |
| Male gender, n (%) | 23 (89) | 13 (72) | 0.170 |
| BMI (Kg/m2) | 24±5 | 24±5 | 0.893 |
| Disease characteristics | |||
| Disease duration (years) | 4±4 | 4±7 | 0.674 |
| CHILD PUGH numeric | 8±2 | 7±2 | 0.065 |
| Disease etiology | 0.124 | ||
| Viral , n (%) | 8 (31) | 1 (5) | 0.124 |
| Alcoholic, n (%) | 10 (38) | 9 (50) | 0.124 |
| Metabolic, n (%) | 8 (31) | 8(44) | 0.124 |
| Hepatic Encephalopathy, n (%) | 26 (100) | 18 (100) | |
| Ascites,y n (%) | 14 (54) | 4 (22) | 0.036 |
| Hepatocarcinoma, n (%) | 5 (19) | 3 (17) | 0.828 |
| Dysmetabolic comorbidities | |||
| T2DM | 8 (31) | 7 (39) | 0.576 |
| Hypertension | 9 (35) | 9 (50) | 0.307 |
| Biohumoral parameters | |||
| Hb (g/dL) | 9±2 | 8±2 | 0.139 |
| PLT (10^9/µL) | 71±46 | 88±66 | 0.268 |
| INR | 1±0 | 1±0 | 0.726 |
| Albumin (g/dL) | 3±0 | 3±0 | 0.317 |
| Creatinine (mg/dL) | 1±0 | 1±0 | 0.277 |
| GGT (U/L) | 206±396 | 125±107 | 0.407 |
| ALT (UI/L) | 68±148 | 37±26 | 0.381 |
| AST (UI/L) | 100±163 | 53±52 | 0.252 |
| Bilirubin (g/dL) | 3±2 | 2±1 | 0.072 |
| Score | |||
| FIB-4 | 11±8 | 8±9 | 0.170 |
| APRI SCORE | 1±2 | 1±1 | 1.412 |
| Treatments | |||
| Emergency legature, n (%) | 15 (58) | 13 (72) | 0.325 |
| Election ligature, n(%) | 11 (42) | 5 (28) | 0.325 |
| Beta-blockers, n (%) | 16 (61) | 6 (33) | 0.066 |
| PHG not worsened (n=16) |
PHG worsened (n=28) | OR (95%CI) | p-value | |
|---|---|---|---|---|
| Demographic and Anthropometric data | ||||
| Age (years) | 66±9 | 62±13 | 1 (0.9-1.0) | 0.294 |
| Male gender, n (%) | 13(81) | 23 (82) | 0.9 (0.2-4.6) | 0.941 |
| BMI (Kg/m2) | 24±5 | 24±5 | 1 (0.9-1.1) | 0.707 |
| Disease characteristics | ||||
| Disease duration (years) | 4±3 | 4±6 | 1.0 (0.9-1.1) | 0.637 |
| CHILD PUGH numeric | 8±2 | 7±2 | 0.8 (0.5-1.1) | 0.171 |
| Disease etiology | ||||
| Viral , n (%) | 5(31) | 4(14) | 1.6 (0.7-3.7) | 0.283 |
| Alcoholic, n (%) | 6(37) | 13(46) | 1.6 (0.7-3.7) | 0.283 |
| Metabolic, n (%) | 5(31) | 11(39) | 1.6 (0.7-3.7) | 0.283 |
| Hepatic Encephalopathy, n (%) | 16(100) | 28 (100) | ||
| Ascites, n (%) | 10(62) | 8(28) | 4.2 (1.1-15.3) | 0.032 |
| Hepatocarcinoma, n (%) | 3(19) | 5(18) | 1.0 (0.2-5.1) | 0.941 |
| Gastric varices T0, n (%) | 0(0) | 3 | 0 | 0.999 |
| PHPs T0, n (%) | 1(6) | 1 | 1.8 (0.1-31) | 0.695 |
| Dysmetabolic comorbidities | ||||
| T2DM | 4 (25) | 11(39) | 0.5 (0.1-2.0) | 0.340 |
| Hypertension | 6 (37) | 12(43) | 1.2 (0.3-4.4) | 0.728 |
| Biohumoral parameters | ||||
| Hb (g/dL) | 9±3 | 8±2 | 0.835 (0.6-1.1) | 0.167 |
| PLT (10^9/µL) | 79±44 | 77±50 | 1.0 (0.98-1.02) | 0.876 |
| INR | 1±0 | 1±0 | 2.2 (0.2-25.9) | 0.524 |
| Albumin (g/dL) | 3±0 | 3±0 | 1.2 (0.4-3.7) | 0.741 |
| Creatinine (mg/dL) | 1.2±1 | 0.8±0 | 0.4 (0.1-1.2) | 0.107 |
| GGT (U/L) | 221±493 | 145±134 | 0.99 (0.99-1.0) | 0.467 |
| ALT (UI/L) | 88±185 | 36±32 | 0.99 (0.98-1.0) | 0.280 |
| AST (UI/L) | 120±1202 | 58±55 | 0.97 (0.9-1.0) | 0.191 |
| Bilirubin (g/dL) | 2±2 | 2±2 | 0.95 (0.7-1.3) | 0.761 |
| Score | ||||
| FIB-4 | 11±8 | 9±8 | 0.97 (0.9-1) | 0.432 |
| APRI SCORE | 2±3 | 1±1 | 0.9 (0.6-1.2) | 0.362 |
| Treatment | ||||
| Emergency legature, n (%) | 8(50) | 20(72) | 0.4 (0.1-1.4) | 0.160 |
| Election ligature, n (%) | 8(50) | 8(28) | 0.4 (0.1-1.4) | 0.160 |
| Beta blockers, n (%) | 11 (69) | 11(39) | 3.4 (0.9-12.5) | 0.050 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




