Submitted:
16 April 2025
Posted:
17 April 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
- Ethics Statement
- Sertoli cell culture
- Animals and Genotyping
- RT-PCR
- Histological Examination
- Male Fertility Test
- Immunofluorescence Staining
- Cell counts
- Sperm counting
- Isolation of Sertoli cells from testis and labeling of F-actin with phalloidin
- Statistical analysis
Results
- Spag6l mRNA is expressed in Sertoli cells
- Generation of Sertoli cell-specific Spag6l knockout mice
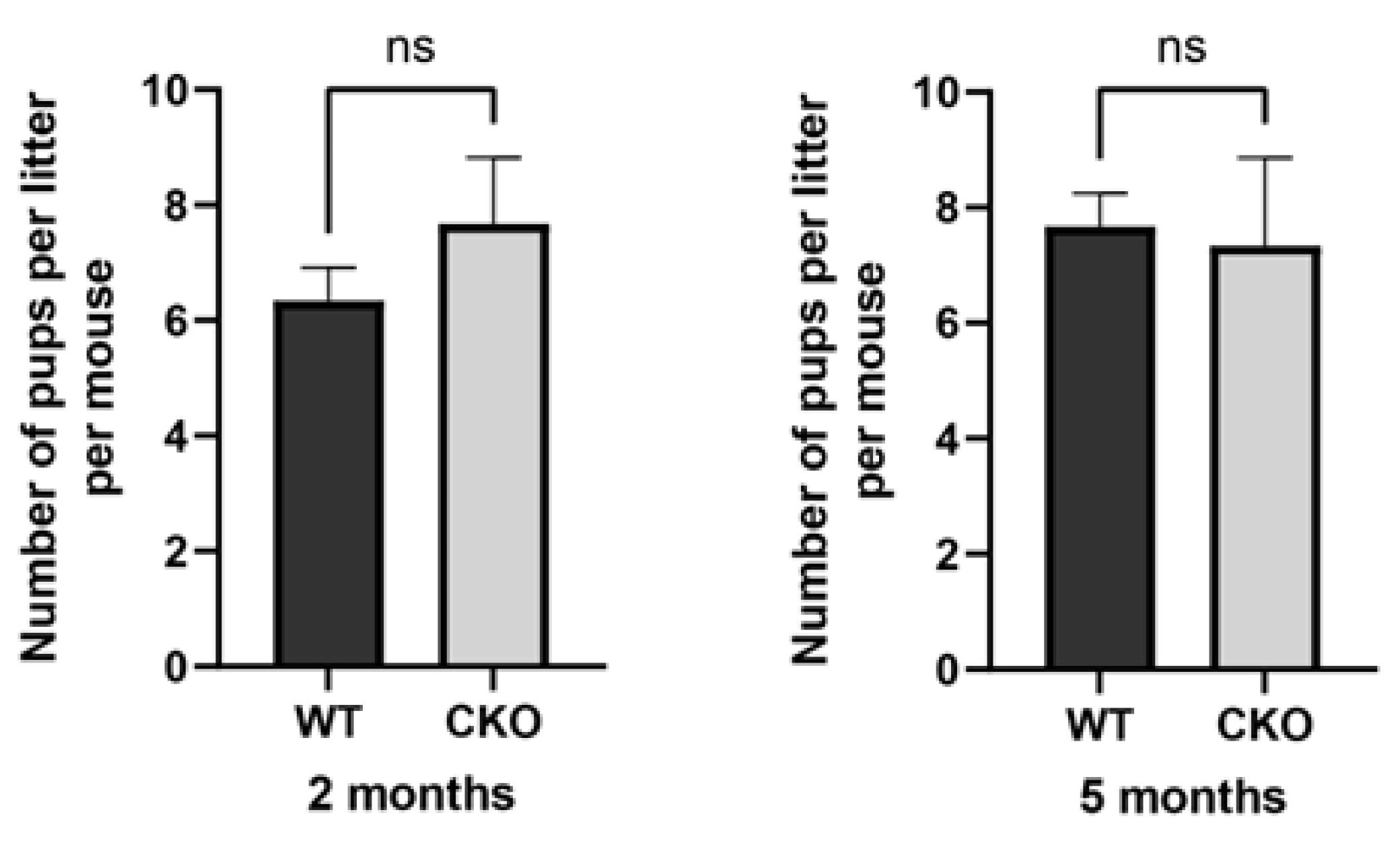
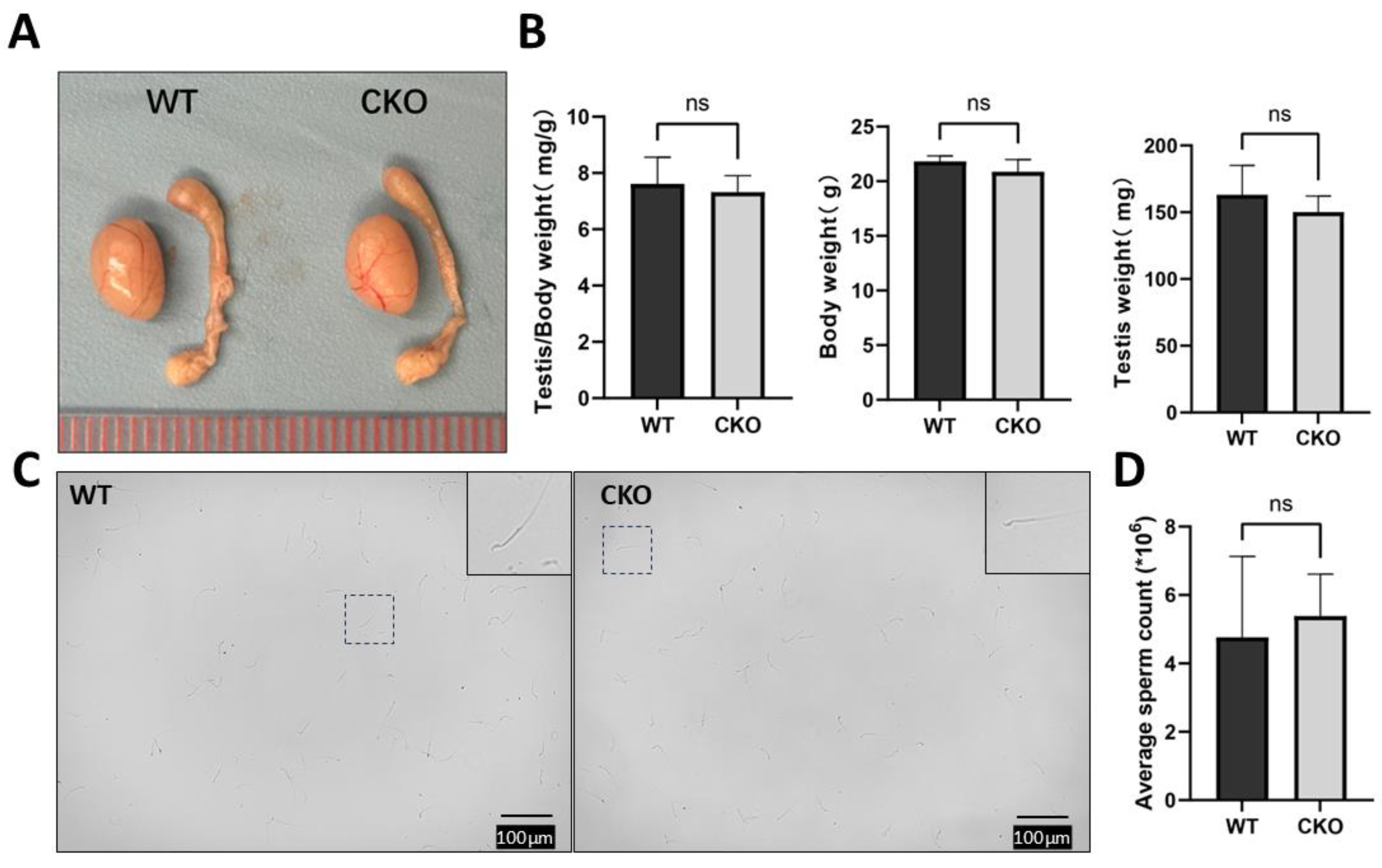
- The cKO male mice had normal fertility with normal testis size, sperm number and motility
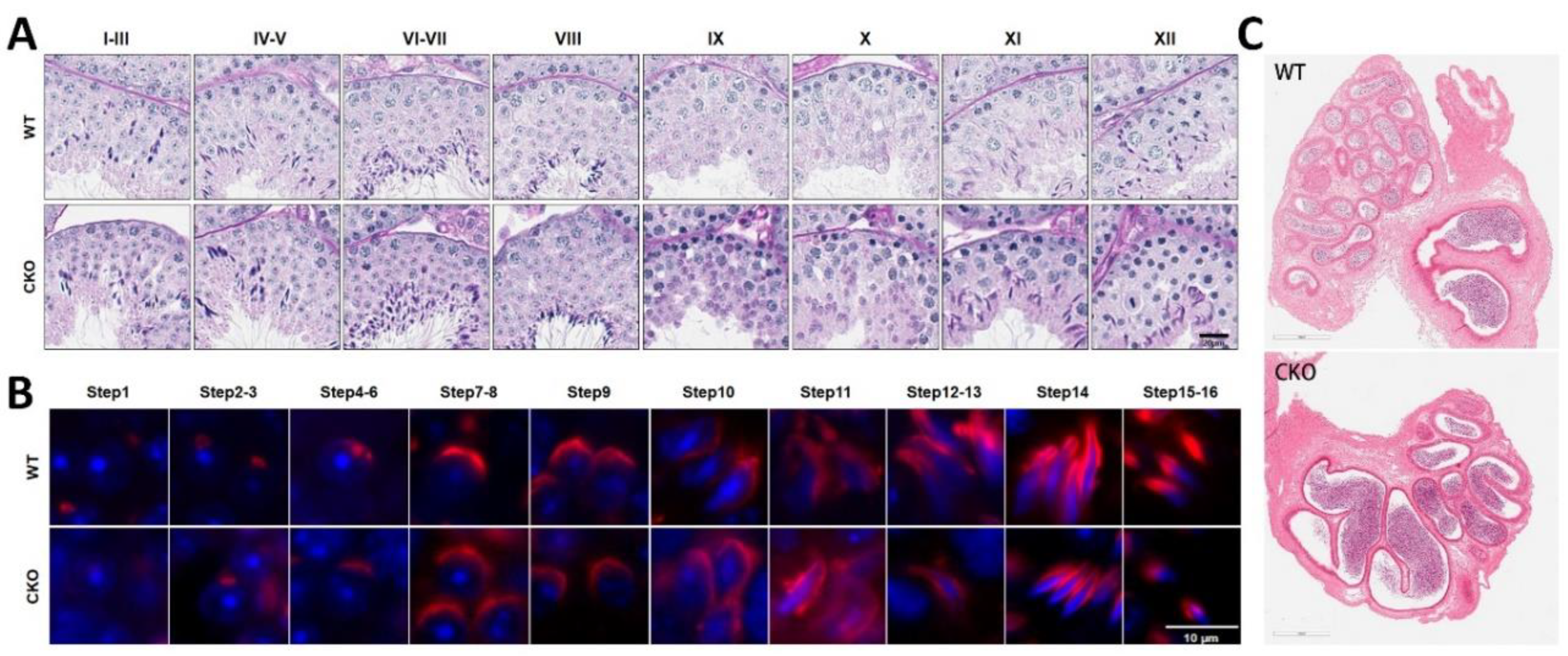
- Spermatogenesis is normal in the Spag6l cKO mice
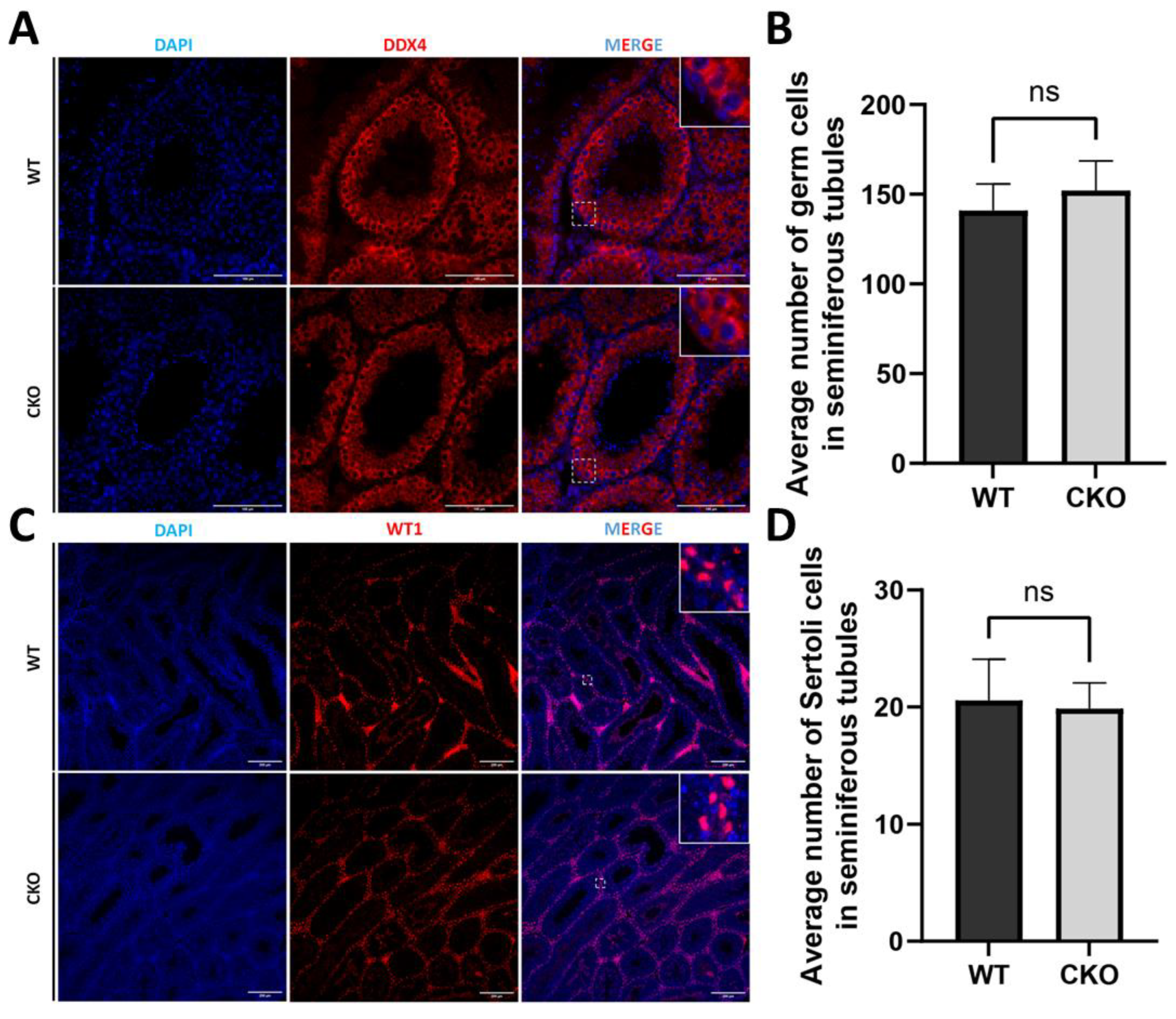
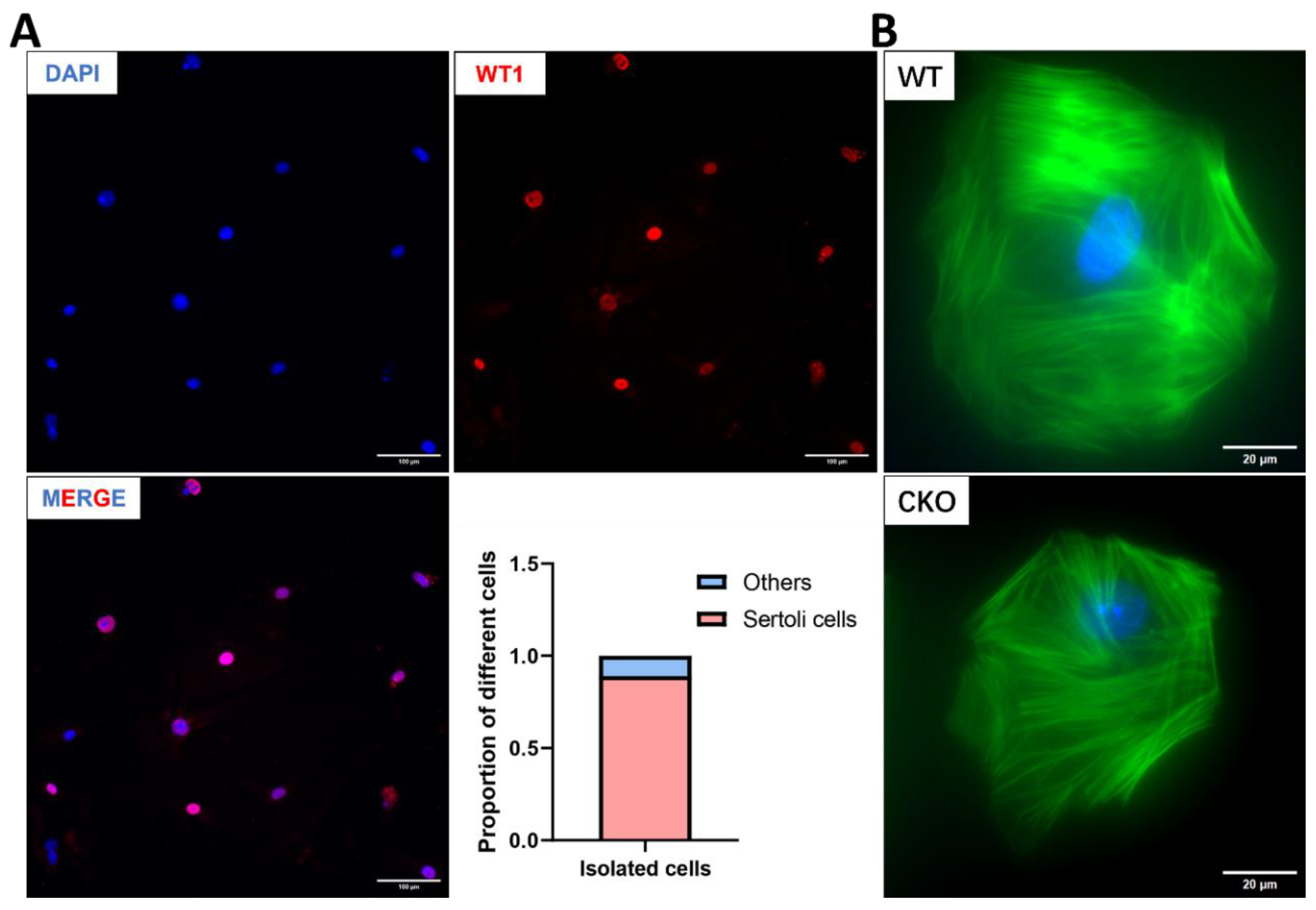
- Disruption of Spag6l in Sertoli cells did not change the population of germ cells and Sertoli cells in the seminiferous tubules.
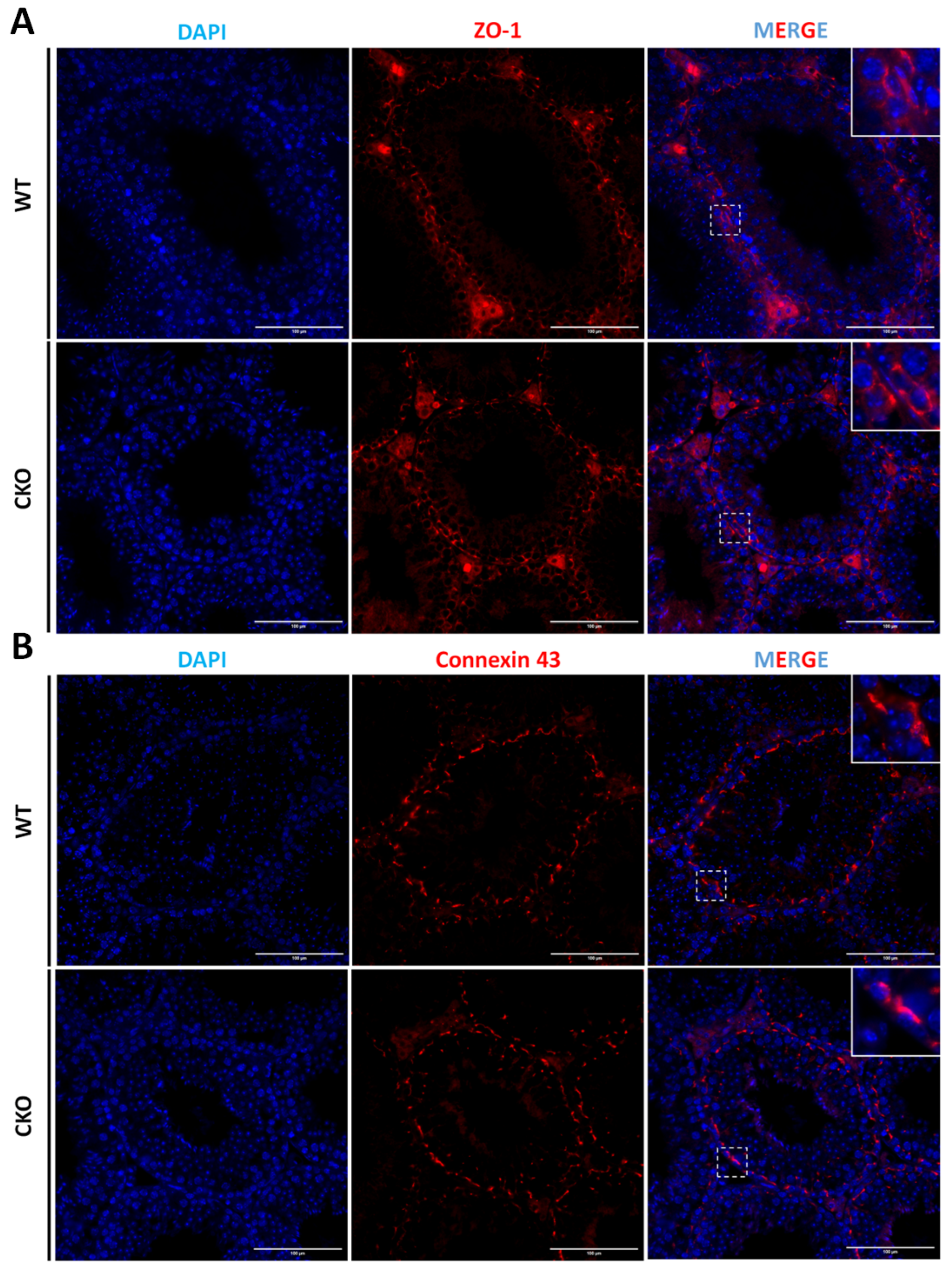
- The Sertoli cells maintain a normal cellular junction in the absence of SPAG6L
- The cytoskeleton of Sertoli cells show normal structure in the absence of SPAG6L
Discussion
Supplementary Materials
Acknowledgments
References
- Smith, E.F.; Lefebvre, P.A. PF16 encodes a protein with armadillo repeats and localizes to a single microtubule of the central apparatus in Chlamydomonas flagella. Journal of Cell Biology 1996, 132, 359–370. [Google Scholar] [CrossRef]
- Neilson, L.I.; et al. cDNA cloning and characterization of a human sperm antigen (SPAG6) with homology to the product of the Chlamydomonas PF16 locus. Genomics 1999, 60, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; et al. SPAG6 regulates cell apoptosis through the TRAIL signal pathway in myelodysplastic syndromes. Oncology Reports 2017, 37, 2839–2846. [Google Scholar] [CrossRef]
- Yin, J.; et al. SPAG6 silencing induces apoptosis in the myelodysplastic syndrome cell line SKM-1 via the PTEN/PI3K/AKT signaling pathway in vitro and in vivo. Int J Oncol 2018, 53, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; et al. SPAG6 silencing induces autophagic cell death in SKM-1 cells via the AMPK/mTOR/ULK1 signaling pathway. Oncology Letters 2020, 20, 551–560. [Google Scholar] [CrossRef]
- Jiang, M.; et al. Upregulation of SPAG6 in Myelodysplastic Syndrome: Knockdown Inhibits Cell Proliferation via AKT/FOXO Signaling Pathway. DNA Cell Biol 2019, 38, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.R.; et al. SPAG6 promotes cell proliferation and inhibits apoptosis through the PTEN/PI3K/AKT pathway in Burkitt lymphoma. Oncology Reports 2020, 44, 2021–2030. [Google Scholar] [CrossRef]
- Altenberger, C.; et al. SPAG6 and L1TD1 are transcriptionally regulated by DNA methylation in non-small cell lung cancers. Mol Cancer 2017, 16, 1. [Google Scholar] [CrossRef]
- Mulaw, M.A.; et al. CALM/AF10-positive leukemias show upregulation of genes involved in chromatin assembly and DNA repair processes and of genes adjacent to the breakpoint at 10p12. Leukemia 2012, 26, 1012–1019. [Google Scholar] [CrossRef]
- Lonergan, K.M.; et al. Identification of novel lung genes in bronchial epithelium by serial analysis of gene expression. American Journal of Respiratory Cell and Molecular Biology 2006, 35, 651–661. [Google Scholar] [CrossRef]
- Silina, K.; et al. Sperm-associated Antigens as Targets for Cancer Immunotherapy: Expression Pattern and Humoral Immune Response in Cancer Patients. Journal of Immunotherapy 2011, 34, 28–44. [Google Scholar] [CrossRef] [PubMed]
- Kitchen, M.O.; et al. Methylation of HOXA9 and ISL1 Predicts Patient Outcome in High-Grade Non-Invasive Bladder Cancer. PLoS One 2015, 10, e0137003. [Google Scholar] [CrossRef]
- Abe, M.; et al. Identification of genes targeted by CpG island methylator phenotype in neuroblastomas, and their possible integrative involvement in poor prognosis. Oncology 2008, 74, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; et al. Patients with severe asthenoteratospermia carrying SPAG6 or RSPH3 mutations have a positive pregnancy outcome following intracytoplasmic sperm injection. J Assist Reprod Genet 2020, 37, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; et al. Lineage-specific duplications of Muroidea Faim and Spag6 genes and atypical accelerated evolution of the parental Spag6 gene. J Mol Evol 2013, 77, 119–129. [Google Scholar] [CrossRef]
- Yap, Y.T.; et al. The Ancient and Evolved Mouse Sperm-Associated Antigen 6 Genes Have Different Biologic Functions In Vivo. Cells 2022, 11. [Google Scholar] [CrossRef]
- Sapiro, R.; et al. Male infertility, impaired sperm motility, and hydrocephalus in mice deficient in sperm-associated antigen 6. Molecular and Cellular Biology 2002, 22, 6298–6305. [Google Scholar] [CrossRef]
- Teves, M.E.; et al. Sperm-associated antigen 6 (SPAG6) deficiency and defects in ciliogenesis and cilia function: polarity, density, and beat. PLoS One 2014, 9, e107271. [Google Scholar] [CrossRef]
- Li, X.; et al. Otitis media in sperm-associated antigen 6 (Spag6)-deficient mice. PLoS One 2014, 9, e112879. [Google Scholar] [CrossRef]
- Li, W.; et al. Sperm Associated Antigen 6 (SPAG6) Regulates Fibroblast Cell Growth, Morphology, Migration and Ciliogenesis. Scientific Reports 2015, 5. [Google Scholar] [CrossRef]
- Cooley, L.F.; et al. Impaired immunological synapse in sperm associated antigen 6 (SPAG6) deficient mice. Scientific Reports 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.H.; et al. The sperm-associated antigen 6 interactome and its role in spermatogenesis. Reproduction 2019, 158, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Man, Y.; et al. Generation of floxed Spag6l mice and disruption of the gene by crossing to a Hprt-Cre line. Genesis 2023, 61, e23512. [Google Scholar] [CrossRef] [PubMed]
- McKenna, E.D.; et al. The Tubulin Code, from Molecules to Health and Disease. Annual Review of Cell and Developmental Biology 2023, 39, 331–361. [Google Scholar] [CrossRef]
- O'Donnell, L.; Smith, L.B.; Rebourcet, D. Sertoli cells as key drivers of testis function. Seminars in Cell & Developmental Biology 2022, 121, 2–9. [Google Scholar]
- Zirkin, B.R.; Papadopoulos, V. Leydig cells: formation, function, and regulation. Biology of Reproduction 2018, 99, 101–111. [Google Scholar] [CrossRef]
- Ni, F.D.; Hao, S.L.; Yang, W.X. Multiple signaling pathways in Sertoli cells: recent findings in spermatogenesis. Cell Death & Disease 2019, 10. [Google Scholar]
- Mruk, D.D.; Cheng, C.Y. Sertoli-Sertoli and Sertoli-germ cell interactions and their significance in germ cell movement in the seminiferous epithelium during spermatogenesis. Endocrine Reviews 2004, 25, 747–806. [Google Scholar]
- Wu, S.W.; et al. Crosstalk between Sertoli and Germ Cells in Male Fertility. Trends in Molecular Medicine 2020, 26, 215–231. [Google Scholar] [CrossRef]
- Li, L.X.; et al. Regulation of spermatid polarity by the actin-and microtubule (MT)-based cytoskeletons. Seminars in Cell & Developmental Biology 2018, 81, 88–96. [Google Scholar]
- Mcguinness, M.P.; et al. Relationship of a Mouse Sertoli-Cell Line (Msc-1) to Normal Sertoli Cells. Biology of Reproduction 1994, 51, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Lécureuil, C.; et al. Sertoli and granulosa cell-specific Cre recombinase activity in transgenic mice. Genesis 2002, 33, 114–118. [Google Scholar] [CrossRef]
- Eskola, V.; et al. Stable transfection of the rat follicle-stimulating hormone receptor complementary DNA into an immortalized murine Sertoli cell line. Molecular and Cellular Endocrinology 1998, 139, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; et al. Assessment of estradiol-induced gene regulation and proliferation in an immortalized mouse immature Sertoli cell line. Life Sci 2016, 148, 268–278. [Google Scholar] [CrossRef]
- Wang, Y.; Lui, W.Y. Opposite effects of interleukin-1alpha and transforming growth factor-beta2 induce stage-specific regulation of junctional adhesion molecule-B gene in Sertoli cells. Endocrinology 2009, 150, 2404–2412. [Google Scholar] [CrossRef]
- Takai, Y.; et al. The Immunoglobulin-Like Cell Adhesion Molecule Nectin and Its Associated Protein Afadin. Annual Review of Cell and Developmental Biology 2008, 24, 309–342. [Google Scholar] [CrossRef] [PubMed]
- Carmona, B.; et al. Tubulin Post-Translational Modifications: The Elusive Roles of Acetylation. Biology-Basel 2023, 12. [Google Scholar] [CrossRef]
- Tang, E.I.; Mruk, D.D.; Cheng, C.Y. Regulation of microtubule (MT)-based cytoskeleton in the seminiferous epithelium during spermatogenesis. Seminars in Cell & Developmental Biology 2016, 59, 35–45. [Google Scholar]
- França, L.R.; et al. BLOOD-TISSUE BARRIERS Morphofunctional and Immunological Aspects of the Blood-Testis and Blood-Epididymal Barriers. Biology and Regulation of Blood-Tissue Barriers 2013, 763, 237–259. [Google Scholar]
- Pelletier, R.M. The blood-testis barrier: the junctional permeability, the proteins and the lipids. Progress in Histochemistry and Cytochemistry 2011, 46, 49–127. [Google Scholar] [CrossRef]
- Qian, X.J.; et al. Actin binding proteins, spermatid transport and spermiation. Seminars in Cell & Developmental Biology 2014, 30, 75–85. [Google Scholar]
- Ou, Y.; et al. Primary cilia in the developing pig testis. Cell and Tissue Research 2014, 358, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.F.; et al. The Emerging Role of Sperm-Associated Antigen 6 Gene in the Microtubule Function of Cells and Cancer. Mol Ther Oncolytics 2019, 15, 101–107. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



